1. Introduction
The rising demand for energy around the globe is leading to economic and environmental problems, as the current supply of energy is highly reliant on fossil fuels. New sources and systems of energy should be explored to reduce the dependence on scarce, expensive, and polluting fossil fuels. Biomass is accounted as one of the most abundant and available wastes, and it is a carbon carrier that can be used to produce bio-energy [
1]. However, raw biomass is less attractive due to the existence of several drawbacks, such as high moisture content, low energy density, hydrophilicity, and high alkali content which makes transportation, storage, and usage of the biomass difficult [
2]. Thermochemical and biochemical conversion technologies, such as torrefaction, pyrolysis, gasification, hydrothermal carbonization (HTC), and anaerobic digestion (AD) are used to tackle these barriers. Among these technologies, anaerobic digestion and hydrothermal carbonization are highly taken into account, due to their relatively lower temperatures and their ability to deal with high moisture content biomass [
3].
HTC is a wet process that involves the reaction of biomass with hot (180–260 °C), pressurized (2–6 MPa) liquid water. During this process, numerous chemical reactions take place, including hydrolysis, condensation, and decarboxylation [
4]. The produced char is called hydrochar to distinguish it from biochar, which is obtained from non-hydrothermal processes, such as torrefaction and pyrolysis [
3]. The inputs to this system usually include raw biomass, water, and energy (heat) and the outputs are hydrochar, a gas stream and a liquid stream (process water) [
5]. AD is a collection of processes, by which microorganisms break down biodegradable material in the absence of oxygen. The main product of AD is biogas, which consists of methane, carbon dioxide, and traces of other gases [
6]. In addition to biogas, digestate is a by-product of AD that consists of left-over indigestible material and dead micro-organisms. Biomass can also directly combust to produce energy. Although thermochemical processes (like HTC), and biochemical processes (like AD), are highly noticed in the recent years, direct combustion (where the raw biomass is burnt in excess air to produce heat) is perhaps the simplest way to convert biomass to bioenergy [
7,
8,
9]. Hence, the proposed systems to convert biomass to bioenergy should be compared with the direct combustion.
Research on biomass conversion has been extensively conducted in the lab scale; however, there is a sensible gap regarding the practical and industrial design and environmental assessments. The integration of the biomass treating processes can not only make the whole process more energy-efficient, but it can also further reduce the waste considerably based on the circular economy [
4]. Reza et al. [
10] used the digestate from an AD process operating at thermophilic conditions (50–60 °C) as a feed for HTC. The energy recovery of this combination was 20% higher than energy recovery from HTC and 60% higher than the one for AD. The AD-HTC process that was proposed by Reza et al. [
10] can be reversed to HTC-AD [
11], such that the biomass is firstly fed to the HTC process, where its alkali content dissolves in the process water (PW) and it can produce hydrochar with lower ash content that is more suitable for boilers [
12]. It is worthy to mention that proper process condition should be considered to ensure that the ash content will reduce in HTC; otherwise, there is a possibility of increase in ash content in some cases [
13]. In addition to the use of hydrochar for power generation, the process water can enter an existing AD system. The pH of the PW is in the range of 2.7–3.5 [
14,
15] and, hence, it can accelerate the hydrolysis step in AD. Ease of conversion in the AD system is expected, as hydrolysis is considered to be the limiting factor of AD systems [
16].
This study is aimed at introducing two different scenarios for bioenergy production while using direct combustion for one scenario and a combination of hydrothermal carbonization and anaerobic digestion for the other one. Both scenarios integrate the power cycles and the biomass conversion processes with heat recovery and internal energy supply. The required data for HTC were obtained from lab-scale experiments. These data include hydrochar’s mass yield, heating value, and elemental composition. Engineering Equation Solver (EES) developed the governing thermodynamic equations of the systems. The main goals of this study are to develop and perform numerical analysis on an integrated HTC-AD scenario for converting biomass to energy, and compare the performance of electricity production by the proposed method with direct combustion of the raw biomass.
2. Materials and Methods
Both systems that were analyzed in this study can work with a variety of biomass. For this study, sawdust has been considered as the feedstock, which is tiny pieces of wood that fall as powder from wood as it is cut by a saw [
17]. Sawdust is one of the most abundant biomass sources, with more than five-million tonnes of sawdust surplus were available in Canada alone in 2004 [
18].
Some of the main primary datasets were obtained by performing lab-scale experiments. The HTC experiments were conducted while using a Parr 600 mL series 4560 mini reactor (Parr Instrument Company, Moline, IL, USA) and approximately 20 g of sawdust was used for each experiment. For HTC, dry biomass to water mass ratio of 1:5 was used. After stirring the sawdust and water mixture in the reactor, it was sealed and pressurized by nitrogen gas to ensure that the pressure inside the reactor is always above saturation pressure of water. The reactor was then heated up to 220 °C and kept at this temperature for 40 min. The reactor was then immediately submerged into ice water until the temperature decreased to below 30 °C. The liquid–hydrochar mixture was removed from the reactor and then separated while using filter paper. The solid sample was placed inside a tin crucible and then left in a furnace overnight at 105 °C to dry.
A higher heating value (HHV) test was performed on the raw feed and solid products of HTC while using the IKA bomb calorimeter (IKAWorks, Wilmington, NC, USA). Moreover, the proximate analysis and ultimate analysis were performed on the obtained hydrochar and the raw sample. More details on the procedure of these characterization tests and the composition of the products can be found in Ref. [
15].
In the HTC-AD scenario, HTC was scaled up and combined with Rankine, and Brayton cycles and anaerobic digestion process.
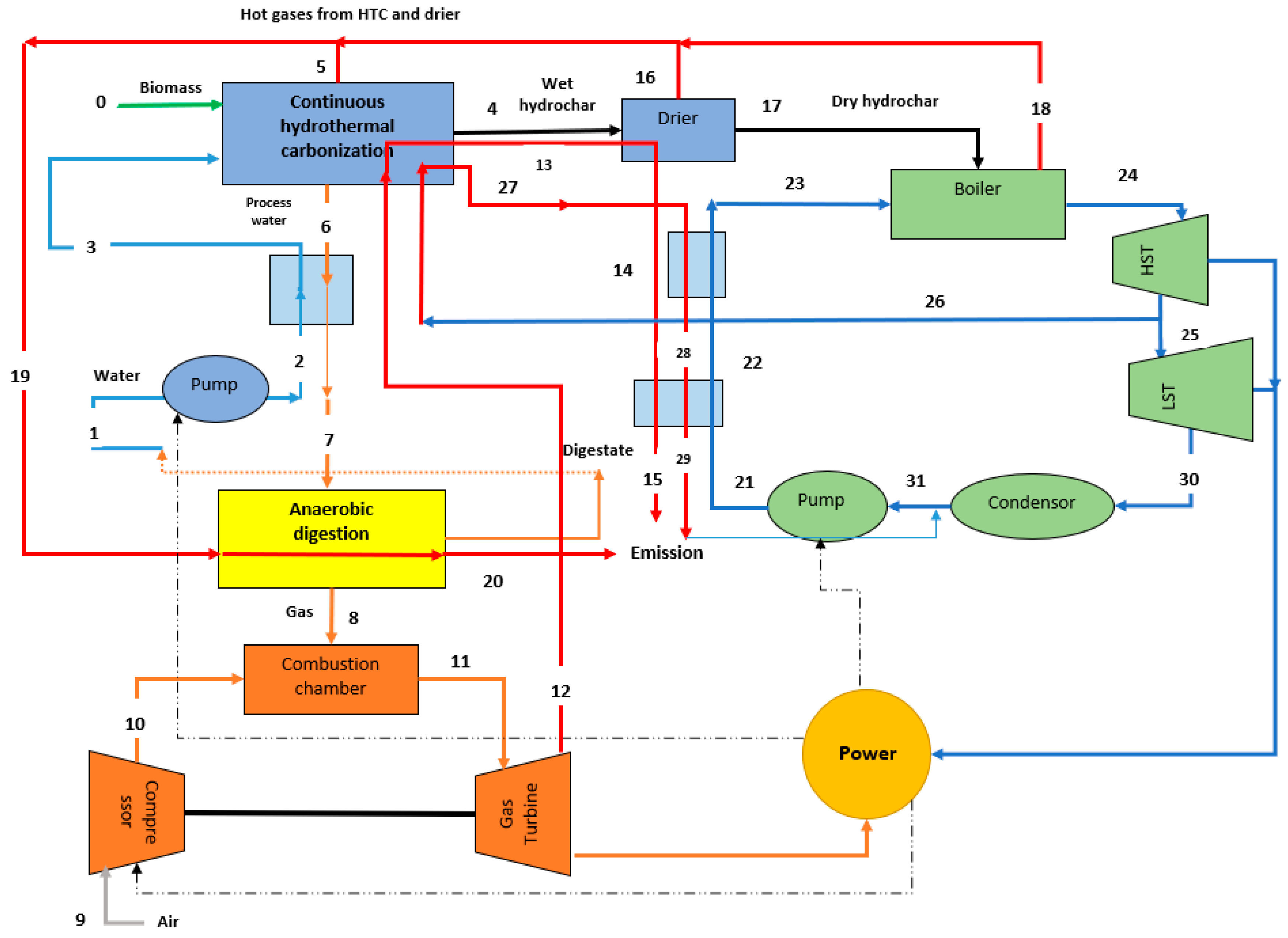
Figure 1 shows the schematic of scenario 1. As is shown in
Figure 1, biomass is mixed with hot pressurized water. It is worth mentioning that, unlike the lab-scale experiments, the HTC reactor is not pressurized with nitrogen gas, but is instead pressurized with the water that enters the reactor while using a high-pressure pump. Moreover, the required heat for HTC is provided by a heat exchanger that recovers the heat from the Rankine and Brayton cycles. The main product of HTC is wet hydrochar that is utilized in a Rankine cycle after getting dried in the drier.
Table 1 is a stream table to help understand the conditions of each stream in the HTC-AD scenario. The liquid by-product of the HTC exchanges its heat with the water stream entering the HTC reactor and then enters into the AD system. A gas stream enriched in methane and hydrogen that enters the Brayton cycle to produce more power is the main output of this AD system. The gas turbine’s exhaust is at high temperature [
19,
20]; hence, before releasing this hot stream to the atmosphere, its heat is transferred to the HTC reactor. If the gas turbine’s exhaust heat is insufficient for the HTC process, excess heat is supplied from the lower stages of the steam turbine of the Rankine cycle. The power that is generated by the steam turbine is used wherever electricity was required (for the pumps and the compressor).
As shown in
Figure 2, the biomass is introduced to the drier as the first processing step in this scenario (direct combustion (DC)). Afterward, the dried biomass is utilized in a boiler to convert a water stream to steam, which can then produce the power after expansion in the turbine.
Table 2 reports the stream table of the DC scenario.
Several assumptions, such as the operational conditions of each equipment section, should be considered, as both systems incorporate several types of equipment. Moreover, some of the data was not obtained via experiments. Hence, this section clarifies the assumptions and data obtained from the literature.
It was assumed that HTC is scaled up to a continuous reactor and the flow rate of the biomass to this reactor is 0.016 kg/s. The same mass flow rate was considered for the DC scenario. The initial moisture content of the biomass was 10% and a 1:5 dry biomass to water weight ratio was considered for HTC.
The process water from HTC is acidic and it contains water, acetic acid, and a number of other organic acids [
15]. According to Kambo et al. [
14], these organic acids mainly contain acetic acid, 5-Hydroxyl Methyl Furfural (HMF), and levulinic acid.
Table 3 reports the composition of the process water that was considered in this study. It is worthwhile to note that a more accurate and detailed research study on HTC should consider the presence of other organic compounds, such as glycolic acid or formic acid. In this research due to the high number of equations only three of the components were considered.
The chemical reactions of these acids in AD were considered based on Buswell and Muller [
21] and they are as follows:
It should be noted that the presence of other elements, such as nitrogen (in other feedstocks such as algae), would result in nitrogen compunds in the bio-oil and bio-gas. Hence, the Equations (1)–(3) should be updated for other feedstocks. Moreover, the addition of materials that help to buffer pH changes and regulate the pH might be required, as the pH of the organic materials entering the AD is around 3, and most of the commercial AD systems work in a pH range of 6.7–7.8 [
22].
The composition of the gas stream exiting the HTC reactor was considered to be all carbon dioxide due to its high abundance relative to other process gases, as discussed by Yan et al. [
23]. The combustion of the biomass and hydrochar in the boilers, as well as the combustion of the obtained gases from AD in the combustion chamber, was considered with 150% of excess air.
It was assumed that the system operates at steady-state and steady flow conditions. Moreover, the HTC temperature was 220 °C, the AD system operates at 35 °C, the PW leaves the HTC reactor at 180 °C, the air consists of 79% nitrogen, and 21% oxygen and the air, biogas, and combustion products are considered as ideal gases. Regarding the temperature of the PW, it should be noted that this temperature is highly dependent on the design of a continuous HTC system that can discharge the process water at that temperature. Given that, to date, there is no clear report regarding such a reactor, a sensitivity analysis will be performed to investigate the effects of lower temperatures of PW. The efficiencies for all exchangers, turbines, and compressors were considered as 80%, whereas the efficiency was assumed as 70% for the pumps [
24]. A temperature approach of 10 °C was considered for all heat exchangers. Moreover, the design is such that the boiler and the gas turbine exhaust supply the required heats for the drier and the HTC reactor, respectively. The compressor and pumps use a portion of the produced power.
Table 4 reports other assumptions considered in the EES codes of the scenarios.