New Perspectives on Iron-Based Nanostructures
Abstract
:1. Introduction
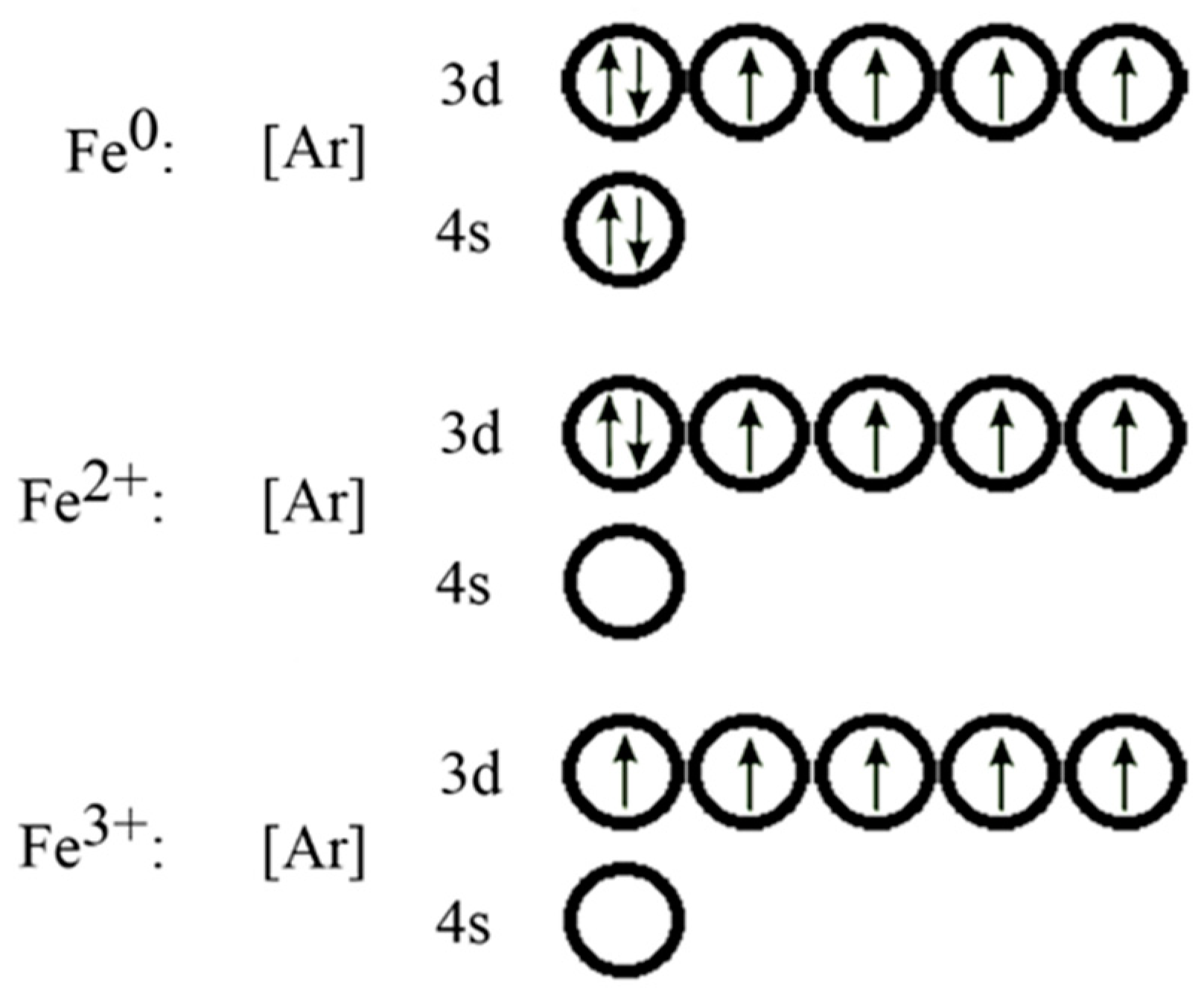
2. Zero-Valent Iron
3. Iron Oxides
3.1. Ferrous Oxide (FeO)
3.2. Ferric Oxide (Fe2O3)
3.2.1. Anhydrous Ferric Oxides
Hematite (α-Fe2O3)
Beta-Fe2O3 (β-Fe2O3)
Maghemite (γ-Fe2O3)
ε-Fe2O3
Zeta-Fe2O3 (ζ-Fe2O3)
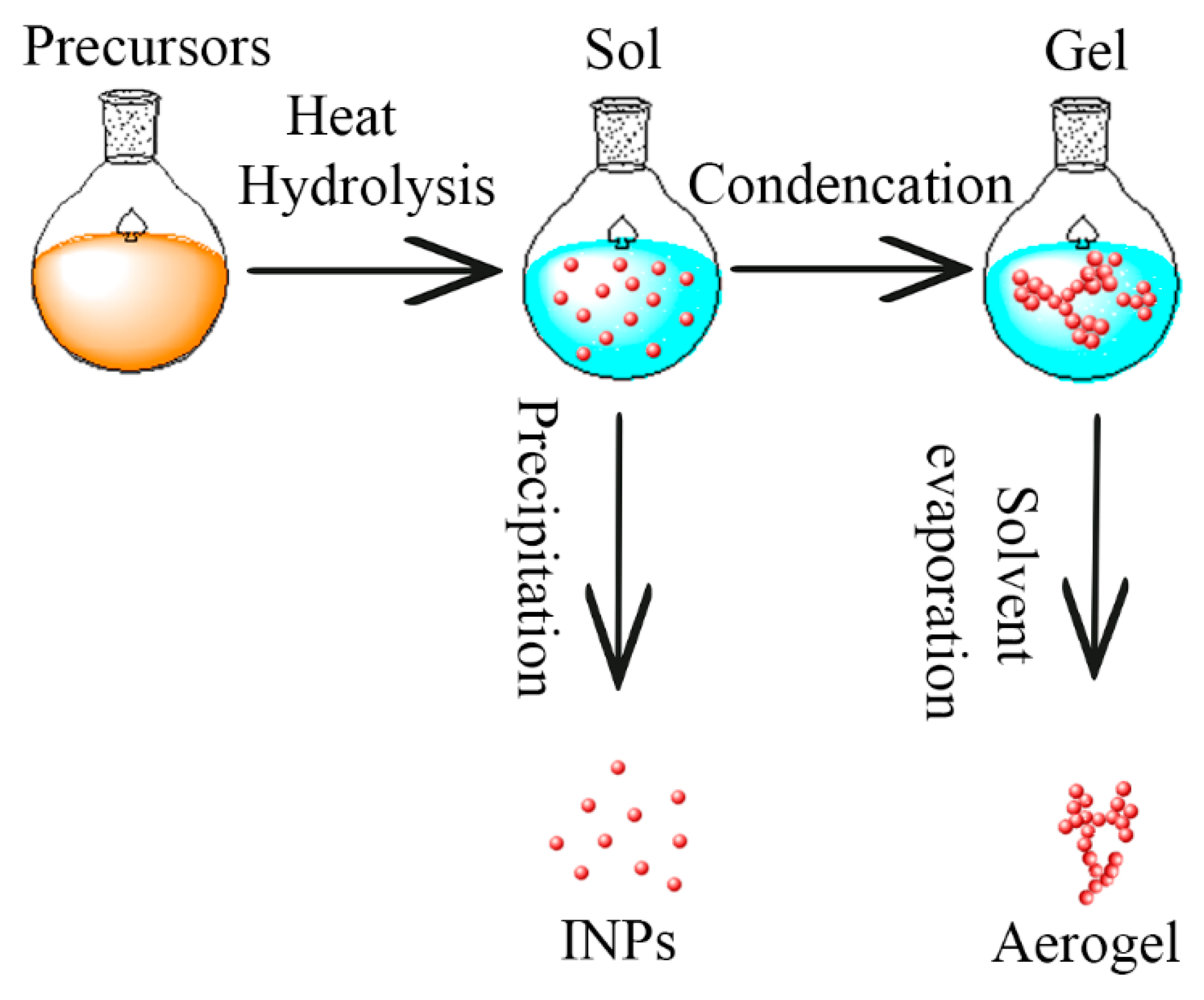
3.2.2. Hydrous Ferric Oxide (Ferrihydrite)
3.3. Ferrous Ferric Oxide (Fe3O4)
4. Ferric Oxide-Hydroxide (Ferric Oxyhydroxide)
4.1. Anhydrous Ferric Oxyhydroxides
4.1.1. Goethite (α-FeOOH)
4.1.2. Akaganeite (β-FeOOH)
4.1.3. Lepidocrocite (γ-FeOOH)
4.1.4. Feroxyhyte (δ-FeOOH)
4.1.5. Schwertmannite
4.2. Hydrated Ferric Oxyhydroxides
5. Iron Hydroxides
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Iron Ore Mine Production by Country 2014–2018. Available online: https://www.statista.com/statistics/267380/iron-ore-mine-production-by-country/ (accessed on 30 March 2019).
- Fan, M.; Yuan, P.; Chen, T.; He, H.; Yuan, A.; Chen, K.; Zhu, J.; Liu, D. Synthesis, characterization and size control of zerovalent iron nanoparticles anchored on montmorillonite. Chin. Sci. Bull. 2010, 55, 1092–1099. [Google Scholar] [CrossRef]
- Xiaomin, N.; Xiaobo, S.; Huagui, Z.; Dongen, Z.; Dandan, Y.; Qingbiao, Z. Studies on the one-step preparation of iron nanoparticles in solution. J. Cryst. Growth 2005, 275, 548–553. [Google Scholar] [CrossRef]
- dos Santos, F.S.; Lago, F.R.; Yokoyama, L.; Fonseca, F.V. Synthesis and characterization of zero-valent iron nanoparticles supported on SBA-15. J. Mater. Res. Technol. 2017, 6, 178–183. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Zare-Hoseinabadi, A.; Berenjian, A.; Ghasemi, Y. Green synthesis and characterization of zero-valent iron nanoparticles using stinging nettle (Urtica dioica) leaf extract. Green Proc. Synth. 2017, 6, 469–475. [Google Scholar] [CrossRef]
- Taghizadeh, S.-M.; Berenjian, A.; Taghizadeh, S.; Ghasemi, Y.; Taherpour, A.; Sarmah, A.K.; Ebrahiminezhad, A. One-put green synthesis of multifunctional silver iron core-shell nanostructure with antimicrobial and catalytic properties. Ind. Crop. Prod. 2019, 130, 230–236. [Google Scholar] [CrossRef]
- Akbari, A.; Mohamadzadeh, F. New method of synthesis of stable zero valent iron nanoparticles (nZVI) by chelating agent diethylene triamine penta acetic acid (DTPA) and removal of radioactive uranium from ground water by using Iron nanoparticle. J. Nanostruct. 2012, 2, 175–181. [Google Scholar] [CrossRef]
- Sun, Y.-P.; Li, X.-q.; Cao, J.; Zhang, W.-x.; Wang, H.P. Characterization of zero-valent iron nanoparticles. Adv. Colloid Interface Sci. 2006, 120, 47–56. [Google Scholar] [CrossRef]
- Üzüm, Ç.; Shahwan, T.; Eroğlu, A.E.; Hallam, K.R.; Scott, T.B.; Lieberwirth, I. Synthesis and characterization of kaolinite-supported zero-valent iron nanoparticles and their application for the removal of aqueous Cu2+ and Co2+ ions. Appl. Clay Sci. 2009, 43, 172–181. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.-N.; Yang, L.-X.; Shi, X.-Y.; Li, I.-C.; Biazik, J.M.; Ratinac, K.R.; Chen, D.-H.; Thordarson, P.; Shieh, D.-B.; Braet, F. The selective growth inhibition of oral cancer by iron core-gold shell nanoparticles through mitochondria-mediated autophagy. Biomaterials 2011, 32, 4565–4573. [Google Scholar] [CrossRef]
- Yang, L.-X.; Wu, Y.-N.; Wang, P.-W.; Huang, K.-J.; Su, W.-C.; Shieh, D.-B. Silver-coated zero-valent iron nanoparticles enhance cancer therapy in mice through lysosome-dependent dual programed cell death pathways: Triggering simultaneous apoptosis and autophagy only in cancerous cells. J. Mater. Chem. B 2020, 8, 4122–4131. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Zare-Hoseinabadi, A.; Sarmah, A.K.; Taghizadeh, S.; Ghasemi, Y.; Berenjian, A. Plant-Mediated synthesis and applications of iron nanoparticles. Mol. Biotechnol. 2018, 60, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Wang, X.; Xing, B. Removal of labile arsenic from flooded paddy soils with a novel extractive column loaded with quartz-supported nanoscale zero-valent iron. Environ. Pollut. 2019, 255, 113249. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Li, P.; Yu, H.; Zhang, T.; Qiu, F. Controlled fabrication of functionalized nanoscale zero-valent iron/celluloses composite with silicon as protective layer for arsenic removal. Chem. Eng. Res. Des. 2019, 151, 242–251. [Google Scholar] [CrossRef]
- Tso, C.-p.; Shih, Y.-h. Effect of carboxylic acids on the properties of zerovalent iron toward adsorption and degradation of trichloroethylene. J. Environ. Manag. 2018, 206, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Ertosun, F.M.; Cellat, K.; Eren, O.; Gül, Ş.; Kuşvuran, E.; Şen, F. Comparison of nanoscale zero-valent iron, fenton, and photo-fenton processes for degradation of pesticide 2, 4-dichlorophenoxyacetic acid in aqueous solution. SN Appl. Sci. 2019, 1, 1491. [Google Scholar] [CrossRef] [Green Version]
- Shojaei, S.; Shojaei, S. Optimization of process variables by the application of response surface methodology for dye removal using nanoscale zero-valent iron. Int. J. Environ. Sci. Technol. 2019, 16, 4601–4610. [Google Scholar] [CrossRef]
- Ji, H.; Zhu, Y.; Duan, J.; Liu, W.; Zhao, D. Reductive immobilization and long-term remobilization of radioactive pertechnetate using bio-macromolecules stabilized zero valent iron nanoparticles. Chin. Chem. Lett. 2019, 30, 2163–2168. [Google Scholar] [CrossRef]
- Lee, C.; Kim, J.Y.; Lee, W.I.; Nelson, K.L.; Yoon, J.; Sedlak, D.L. Bactericidal effect of zero-valent iron nanoparticles on Escherichia coli. Environ. Sci. Technol. 2008, 42, 4927–4933. [Google Scholar] [CrossRef] [Green Version]
- Cornell, R.M.; Schwertmann, U. The Iron OXIDES: Structure, Properties, Reactions, Occurrences and Uses; John Wiley & Sons: Hoboken, NJ, USA, 2003. [Google Scholar]
- Redl, F.X.; Black, C.T.; Papaefthymiou, G.C.; Sandstrom, R.L.; Yin, M.; Zeng, H.; Murray, C.B.; O’Brien, S.P. Magnetic, electronic, and structural characterization of nonstoichiometric iron oxides at the nanoscale. J. Am. Chem. Soc. 2004, 126, 14583–14599. [Google Scholar] [CrossRef]
- Guntlin, C.P.; Ochsenbein, S.T.; WÖrle, M.; Erni, R.; Kravchyk, K.V.; Kovalenko, M.V. Popcorn-shaped FexO (Wüstite) nanoparticles from a single-source precursor: Colloidal synthesis and magnetic properties. Chem. Mater. 2018, 30, 1249–1256. [Google Scholar] [CrossRef] [Green Version]
- Martinez, A.I.; Garcia-Lobato, M.; Perry, D.L. Study of the properties of iron oxide nanostructures. In Research in Nanotechnology Developments; Barrañón, A., Ed.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2009; Volume 19, pp. 184–193. [Google Scholar]
- Strobel, R.; Pratsinis, S.E. Direct synthesis of maghemite, magnetite and wustite nanoparticles by flame spray pyrolysis. Adv. Powder Technol. 2009, 20, 190–194. [Google Scholar] [CrossRef]
- Chen, C.-J.; Chiang, R.-K.; Lai, H.-Y.; Lin, C.-R. Characterization of monodisperse wüstite nanoparticles following partial oxidation. J. Phys. Chem. C 2010, 114, 4258–4263. [Google Scholar] [CrossRef]
- Gheisari, M.; Mozaffari, M.; Acet, M.; Amighian, J. Preparation and investigation of magnetic properties of wüstite nanoparticles. J. Magn. Magn. Mater. 2008, 320, 2618–2621. [Google Scholar] [CrossRef]
- Papaefthymiou, G.; Redl, F.; Black, C.; Sandstrom, R.; Yin, M.; Murray, C.; O’Brien, S. Hybrid magnetic nanoparticles derived from wüstite disproportionation reactions at the nanoscale. In ICAME 2005; Springer: Berlin/Heidelberg, Germany, 2006; pp. 239–245. [Google Scholar] [CrossRef]
- Ahmadipour, M.; Rao, K.V.; Rajendar, V. Formation of nanoscale Mg(x)Fe(1-X)O (x = 0.1, 0.2, 0.4) structure by solution combustion: Effect of fuel to oxidizer ratio. J. Nanomater. 2012, 2012, 163909. [Google Scholar] [CrossRef]
- Chen, C.-J.; Chiang, R.-K.; Kamali, S.; Wang, S.-L. Synthesis and controllable oxidation of monodisperse cobalt-doped wüstite nanoparticles and their core–shell stability and exchange-bias stabilization. Nanoscale 2015, 7, 14332–14343. [Google Scholar] [CrossRef]
- Situ, S.F.; Samia, A.C.S. Highly efficient antibacterial iron oxide@carbon nanochains from wustite precursor nanoparticles. ACS Appl. Mater. Interfaces 2014, 6, 20154–20163. [Google Scholar] [CrossRef]
- Chavez-Galan, J.; Almanza, R. Solar filters based on iron oxides used as efficient windows for energy savings. Sol. Energy 2007, 81, 13–19. [Google Scholar] [CrossRef]
- Campos, E.A.; Pinto, D.V.B.S.; Oliveira, J.I.S.d.; Mattos, E.d.C.; Dutra, R.d.C.L. Synthesis, characterization and applications of iron oxide nanoparticles-a short review. J. Aerosp. Technol. Manag. 2015, 7, 267–276. [Google Scholar] [CrossRef] [Green Version]
- Lassoued, A.; Dkhil, B.; Gadri, A.; Ammar, S. Control of the shape and size of iron oxide (α-Fe2O3) nanoparticles synthesized through the chemical precipitation method. Results Phys. 2017, 7, 3007–3015. [Google Scholar] [CrossRef]
- Tadic, M.; Panjan, M.; Damnjanovic, V.; Milosevic, I. Magnetic properties of hematite (α-Fe2O3) nanoparticles prepared by hydrothermal synthesis method. Appl. Surf. Sci. 2014, 320, 183–187. [Google Scholar] [CrossRef]
- Kefeni, K.K.; Msagati, T.A.; Nkambule, T.T.; Mamba, B.B. Synthesis and application of hematite nanoparticles for acid mine drainage treatment. J. Environ. Chem. Eng. 2018, 6, 1865–1874. [Google Scholar] [CrossRef]
- Asoufi, H.M.; Al-Antary, T.M.; Awwad, A.M. Green route for synthesis hematite (α-Fe2O3) nanoparticles: Toxicity effect on the green peach aphid, Myzus persicae (Sulzer). Environ. Nanotechnol. Monit. Manag. 2018, 9, 107–111. [Google Scholar] [CrossRef]
- Narayanan, K.B.; Han, S.S. One-pot green synthesis of hematite (α-Fe2O3) nanoparticles by ultrasonic irradiation and their in vitro cytotoxicity on human keratinocytes CRL-2310. J. Cluster Sci. 2016, 27, 1763–1775. [Google Scholar] [CrossRef]
- Taghavi, F.S.; Ramazani, A.; Golfar, Z.; Woo, J.S. Green synthesis of α-Fe2O3 (hematite) nanoparticles using tragacanth Gel. J. Appl. Chem. Res. 2017, 11, 19–27. [Google Scholar]
- Ali, H.R.; Nassar, H.N.; El-Gendy, N.S. Green synthesis of α-Fe2O3 using Citrus reticulum peels extract and water decontamination from different organic pollutants. Energ. Source Part A 2017, 39, 1425–1434. [Google Scholar] [CrossRef]
- Ahmmad, B.; Leonard, K.; Islam, M.S.; Kurawaki, J.; Muruganandham, M.; Ohkubo, T.; Kuroda, Y. Green synthesis of mesoporous hematite (α-Fe2O3) nanoparticles and their photocatalytic activity. Adv. Powder Technol. 2013, 24, 160–167. [Google Scholar] [CrossRef]
- Rufus, A.; Sreeju, N.; Philip, D. Synthesis of biogenic hematite (α-Fe2O3) nanoparticles for antibacterial and nanofluid applications. RSC Adv. 2016, 6, 94206–94217. [Google Scholar] [CrossRef]
- Supattarasakda, K.; Petcharoen, K.; Permpool, T.; Sirivat, A.; Lerdwijitjarud, W.J.P.t. Control of hematite nanoparticle size and shape by the chemical precipitation method. Powder Technol. 2013, 249, 353–359. [Google Scholar] [CrossRef]
- Anthony, J.W.; Bideaux, R.A.; Bladh, K.W.; Nichols, M.C. Handbook of Mineralogy; Mineralogical Society of America: Chantilly, VA, USA, 2004. [Google Scholar]
- Basavegowda, N.; Mishra, K.; Lee, Y.R. Synthesis, characterization, and catalytic applications of hematite (α-Fe2O3) nanoparticles as reusable nanocatalyst. Adv. Nat. Sci. 2017, 8, 025017. [Google Scholar] [CrossRef]
- Nazeer, A.A.; Udhayakumar, S.; Mani, S.; Dhanapal, M.; Vijaykumar, S.D. Surface modification of Fe2O3 and MgO nanoparticles with agrowastes for the treatment of chlorosis in Glycine max. Nano Converg. 2018, 5, 23. [Google Scholar] [CrossRef]
- Danno, T.; Nakatsuka, D.; Kusano, Y.; Asaoka, H.; Nakanishi, M.; Fujii, T.; Ikeda, Y.; Takada, J. Crystal structure of β-Fe2O3 and topotactic phase transformation to α-Fe2O3. Cryst. Growth Des. 2013, 13, 770–774. [Google Scholar] [CrossRef]
- Zhang, N.; Guo, Y.; Wang, X.; Zhang, S.; Li, Z.; Zou, Z. Beta-Fe2O3 nanoparticle-assembled film for photoelectrochemical water splitting. Dalton Trans. 2017, 46, 10673–10677. [Google Scholar] [CrossRef]
- Kumar, A.; Singhal, A. Synthesis of colloidal β-Fe2O3 nanostructures—Influence of addition of Co2+ on their morphology and magnetic behavior. Nanotechnology 2007, 18, 475703. [Google Scholar] [CrossRef]
- Nurdin, I.; Johan, M.; Yaacob, I.; Ang, B.; Andriyana, A. Synthesis, characterisation and stability of superparamagnetic maghemite nanoparticle suspension. Mater. Res. Innov. 2014, 18, 200–203. [Google Scholar] [CrossRef]
- Aliahmad, M.; Moghaddam, N.N. Synthesis of maghemite (γ-Fe2O3) nanoparticles by thermal-decomposition of magnetite (Fe3O4) nanoparticles. Mater. Sci.-Poland 2013, 31, 264–268. [Google Scholar] [CrossRef]
- Múzquiz-Ramos, E.; Guerrero-Chávez, V.; Macías-Martínez, B.; López-Badillo, C.; García-Cerda, L. Synthesis and characterization of maghemite nanoparticles for hyperthermia applications. Ceram. Int. 2015, 41, 397–402. [Google Scholar] [CrossRef]
- Guivar, J.A.R.; Sadrollahi, E.; Menzel, D.; Fernandes, E.G.R.; López, E.O.; Torres, M.M.; Arsuaga, J.M.; Arencibia, A.; Litterst, F.J. Magnetic, structural and surface properties of functionalized maghemite nanoparticles for copper and lead adsorption. RSC Adv. 2017, 7, 28763–28779. [Google Scholar] [CrossRef] [Green Version]
- Frison, R.; Cernuto, G.; Cervellino, A.; Zaharko, O.; Colonna, G.M.; Guagliardi, A.; Masciocchi, N. Magnetite–maghemite nanoparticles in the 5–15 nm range: Correlating the core–shell composition and the surface structure to the magnetic properties. a total scattering study. Chem. Mater. 2013, 25, 4820–4827. [Google Scholar] [CrossRef]
- Kera, N.H.; Bhaumik, M.; Pillay, K.; Ray, S.S.; Maity, A. Selective removal of toxic Cr(VI) from aqueous solution by adsorption combined with reduction at a magnetic nanocomposite surface. J. Colloid Interface Sci. 2017, 503, 214–228. [Google Scholar] [CrossRef]
- Prado, Y.; Daffé, N.; Michel, A.; Georgelin, T.; Yaacoub, N.; Greneche, J.-M.; Choueikani, F.; Otero, E.; Ohresser, P.; Arrio, M.-A. Enhancing the magnetic anisotropy of maghemite nanoparticles via the surface coordination of molecular complexes. Nat. Commun. 2015, 6, 10139. [Google Scholar] [CrossRef]
- Mazarío, E.; Helal, A.S.; Stemper, J.; Mayoral, A.; Decorse, P.; Chevillot-Biraud, A.; Novak, S.; Perruchot, C.; Lion, C.; Losno, R. Maghemite nanoparticles bearing di(amidoxime) groups for the extraction of uranium from wastewaters. AIP Adv. 2017, 7, 056702. [Google Scholar] [CrossRef]
- Palmqvist, N.M.; Seisenbaeva, G.A.; Svedlindh, P.; Kessler, V.G. Maghemite nanoparticles acts as nanozymes, improving growth and abiotic stress tolerance in Brassica napus. Nanoscale Res. Lett. 2017, 12, 631. [Google Scholar] [CrossRef] [PubMed]
- Tavakkoli, M.; Kallio, T.; Reynaud, O.; Nasibulin, A.G.; Sainio, J.; Jiang, H.; Kauppinen, E.I.; Laasonen, K. Maghemite nanoparticles decorated on carbon nanotubes as efficient electrocatalysts for the oxygen evolution reaction. J. Mater. Chem. A 2016, 4, 5216–5222. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.; Chen, G.; Lo, I.M. Removal and recovery of Cr(VI) from wastewater by maghemite nanoparticles. Water Res. 2005, 39, 4528–4536. [Google Scholar] [CrossRef]
- Jiang, W.; Pelaez, M.; Dionysiou, D.D.; Entezari, M.H.; Tsoutsou, D.; O’Shea, K. Chromium(VI) removal by maghemite nanoparticles. Chem. Eng. J. 2013, 222, 527–533. [Google Scholar] [CrossRef]
- Avram, A.; Radoi, A.; Schiopu, V.; Avram, M.; Gavrila, H. Synthesis and characterization of γ-Fe2O3 nanoparticles for applications in magnetic hyperthermia. Synthesis 2011, 10, 1. [Google Scholar]
- Mercante, L.; Melo, W.; Granada, M.; Troiani, H.; Macedo, W.; Ardison, J.; Vaz, M.; Novak, M. Magnetic properties of nanoscale crystalline maghemite obtained by a new synthetic route. J. Magn. Magn. Mater. 2012, 324, 3029–3033. [Google Scholar] [CrossRef] [Green Version]
- Kuchma, E.A.; Zolotukhin, P.V.; Belanova, A.A.; Soldatov, M.A.; Lastovina, T.A.; Kubrin, S.P.; Nikolsky, A.V.; Mirmikova, L.I.; Soldatov, A.V. Low toxic maghemite nanoparticles for theranostic applications. Int. J. Nanomed. 2017, 12, 6365–6371. [Google Scholar] [CrossRef] [Green Version]
- Kumar, N.; Kulkarni, K.; Behera, L.; Verma, V. Preparation and characterization of maghemite nanoparticles from mild steel for magnetically guided drug therapy. J. Mater. Sci. Mater. Med. 2017, 28, 116. [Google Scholar] [CrossRef]
- Blanc-Beguin, F.; Nabily, S.; Gieraltowski, J.; Turzo, A.; Querellou, S.; Salaun, P. Cytotoxicity and GMI bio-sensor detection of maghemite nanoparticles internalized into cells. J. Magn. Magn. Mater. 2009, 321, 192–197. [Google Scholar] [CrossRef]
- Perlstein, B.; Lublin-Tennenbaum, T.; Marom, I.; Margel, S. Synthesis and characterization of functionalized magnetic maghemite nanoparticles with fluorescent probe capabilities for biological applications. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 92, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Hai, J.; Piraux, H.; Mazario, E.; Volatron, J.; Ha-Duong, N.; Decorse, P.; Lomas, J.; Verbeke, P.; Ammar, S.; Wilhelm, C. Maghemite nanoparticles coated with human serum albumin: Combining targeting by the iron-acquisition pathway and potential in photothermal therapies. J. Mater. Chem. B 2017, 5, 3154–3162. [Google Scholar] [CrossRef] [PubMed]
- González, B.; Ruiz-Hernández, E.; Feito, M.J.; de Laorden, C.L.; Arcos, D.; Ramírez-Santillán, C.; Matesanz, C.; Portolés, M.T.; Vallet-Regí, M. Covalently bonded dendrimer-maghemite nanosystems: Nonviral vectors for in vitro gene magnetofection. J. Mater. Chem. 2011, 21, 4598–4604. [Google Scholar] [CrossRef]
- Hai, J.; Piraux, H.; Mazario, E.; Volatron, J.; Ha-Duong, N.; Decorse, P.; Espinosa, A.; Whilem, C.; Verbeke, P.; Gazeau, F. Multifunctionality of maghemite nanoparticles functionalized by HSA for drug delivery. In Proceedings of the 2017 IEEE International Magnetics Conference (INTERMAG), Dublin, Ireland, 24–28 April 2017; pp. 1–2. [Google Scholar]
- Štarha, P.; Stavárek, M.; Tuček, J.; Trávníček, Z. 4-Aminobenzoic acid-coated maghemite nanoparticles as potential anticancer drug magnetic carriers: A case study on highly cytotoxic cisplatin-like complexes involving 7-azaindoles. Molecules 2014, 19, 1622–1634. [Google Scholar] [CrossRef] [PubMed]
- Thünemann, A.F.; Schütt, D.; Kaufner, L.; Pison, U.; Möhwald, H. Maghemite nanoparticles protectively coated with poly (ethylene imine) and poly (ethylene oxide)-b lock-poly (glutamic acid). Langmuir 2006, 22, 2351–2357. [Google Scholar] [CrossRef]
- Liu, J.; Detrembleur, C.; Debuigne, A.; De Pauw-Gillet, M.-C.; Mornet, S.; Vander Elst, L.; Laurent, S.; Duguet, E.; Jérôme, C. Glucose-, pH-and thermo-responsive nanogels crosslinked by functional superparamagnetic maghemite nanoparticles as innovative drug delivery systems. J. Mater. Chem. B 2014, 2, 1009–1023. [Google Scholar] [CrossRef]
- Blyakhman, F.A.; Safronov, A.P.; Zubarev, A.Y.; Shklyar, T.F.; Makeyev, O.G.; Makarova, E.B.; Melekhin, V.V.; Larrañaga, A.; Kurlyandskaya, G.V. Polyacrylamide ferrogels with embedded maghemite nanoparticles for biomedical engineering. Results Phys. 2017, 7, 3624–3633. [Google Scholar] [CrossRef]
- Kim, J.-J.; Singh, R.K.; Seo, S.-J.; Kim, T.-H.; Kim, J.-H.; Lee, E.-J.; Kim, H.-W. Magnetic scaffolds of polycaprolactone with functionalized magnetite nanoparticles: Physicochemical, mechanical, and biological properties effective for bone regeneration. RSC Adv. 2014, 4, 17325–17336. [Google Scholar] [CrossRef]
- Ngadiman, N.H.A.; Idris, A.; Irfan, M.; Kurniawan, D.; Yusof, N.M.; Nasiri, R. γ-Fe2O3 nanoparticles filled polyvinyl alcohol as potential biomaterial for tissue engineering scaffold. J. Mech. Behav. Biomed. Mater. 2015, 49, 90–104. [Google Scholar] [CrossRef]
- Fallahiarezoudar, E.; Ahmadipourroudposht, M.; Idris, A.; Yusof, N.M. Optimization and development of Maghemite (γ-Fe2O3) filled poly-L-lactic acid (PLLA)/thermoplastic polyurethane (TPU) electrospun nanofibers using Taguchi orthogonal array for tissue engineering heart valve. Mater. Sci. Eng. C 2017, 76, 616–627. [Google Scholar] [CrossRef]
- Chirita, M.; Grozescu, I.; Taubert, L.; Radulescu, H.; Princz, E. Fe2O3-nanoparticles, physical properties and their photochemical and photoelectrochemical applications. Chem. Bull. 2009, 54, 1–8. [Google Scholar]
- Tucek, J.; Zboril, R.; Namai, A.; Ohkoshi, S.-i. ε-Fe2O3: An advanced nanomaterial exhibiting giant coercive field, millimeter-wave ferromagnetic resonance, and magnetoelectric coupling. Chem. Mater. 2010, 22, 6483–6505. [Google Scholar] [CrossRef]
- López-Sánchez, J.; Muñoz-Noval, A.; Serrano, A.; Abuín, M.; de la Figuera, J.; Marco, J.; Pérez, L.; Carmona, N.; de la Fuente, O.R. Growth, structure and magnetism of ε-Fe2O3 in nanoparticle form. RSC Adv. 2016, 6, 46380–46387. [Google Scholar] [CrossRef]
- Ohkoshi, S.-i.; Namai, A.; Yamaoka, T.; Yoshikiyo, M.; Imoto, K.; Nasu, T.; Anan, S.; Umeta, Y.; Nakagawa, K.; Tokoro, H. Mesoscopic bar magnet based on ε-Fe2O3 hard ferrite. Sci. Rep. 2016, 6, 27212. [Google Scholar] [CrossRef]
- Tuček, J.; Machala, L.; Ono, S.; Namai, A.; Yoshikiyo, M.; Imoto, K.; Tokoro, H.; Ohkoshi, S.-i.; Zbořil, R. Zeta-Fe2O3–A new stable polymorph in iron(III) oxide family. Sci. Rep. 2015, 5, 15091. [Google Scholar] [CrossRef] [Green Version]
- Lynch, S.; Batty, L.; Byrne, P. Environmental risk of metal mining contaminated river bank sediment at redox-transitional zones. Minerals 2014, 4, 52–73. [Google Scholar] [CrossRef] [Green Version]
- Mohapatra, M.; Anand, S. Synthesis and applications of nano-structured iron oxides/hydroxides—A review. Int. J. Eng. Sci. Technol. 2010, 2, 127–146. [Google Scholar] [CrossRef] [Green Version]
- Eusterhues, K.; Wagner, F.E.; Häusler, W.; Hanzlik, M.; Knicker, H.; Totsche, K.U.; KÖgel-Knabner, I.; Schwertmann, U. Characterization of ferrihydrite-soil organic matter coprecipitates by X-ray diffraction and Mossbauer spectroscopy. Environ. Sci. Technol. 2008, 42, 7891–7897. [Google Scholar] [CrossRef]
- Poulson, R.L.; Johnson, C.M.; Beard, B.L. Iron isotope exchange kinetics at the nanoparticulate ferrihydrite surface. Am. Mineral. 2005, 90, 758–763. [Google Scholar] [CrossRef]
- Chasteen, N.D.; Harrison, P.M. Mineralization in ferritin: An efficient means of iron storage. J. Struct. Biol. 1999, 126, 182–194. [Google Scholar] [CrossRef] [Green Version]
- Liu, G.; Debnath, S.; Paul, K.W.; Han, W.; Hausner, D.B.; Hosein, H.-A.; Michel, F.M.; Parise, J.B.; Sparks, D.L.; Strongin, D.R. Characterization and surface reactivity of ferrihydrite nanoparticles assembled in ferritin. Langmuir 2006, 22, 9313–9321. [Google Scholar] [CrossRef] [PubMed]
- Baldi, F.; Marchetto, D.; Battistel, D.; Daniele, S.; Faleri, C.; De Castro, C.; Lanzetta, R. Iron-binding characterization and polysaccharide production by Klebsiella oxytoca strain isolated from mine acid drainage. J. Appl. Microbiol. 2009, 107, 1241–1250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baldi, F.; Marchetto, D.; Paganelli, S.; Piccolo, O. Bio-generated metal binding polysaccharides as catalysts for synthetic applications and organic pollutant transformations. N. Biotechnol. 2011, 29, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Baldi, F.; Minacci, A.; Pepi, M.; Scozzafava, A. Gel sequestration of heavy metals by Klebsiella oxytoca isolated from iron mat. FEMS Microbiol. Ecol. 2001, 36, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Kianpour, S.; Ebrahiminezhad, A.; Mohkam, M.; Tamaddon, A.M.; Dehshahri, A.; Heidari, R.; Ghasemi, Y. Physicochemical and biological characteristics of the nanostructured polysaccharide-iron hydrogel produced by microorganism Klebsiella oxytoca. J. Basic Microbiol. 2016, 2016, 132–140. [Google Scholar] [CrossRef]
- Kianpour, S.; Ebrahiminezhad, A.; Deyhimi, M.; Negahdaripour, M.; Raee, M.j.; Mohkam, M.; Rezaee, H.; Irajie, C.; Berenjian, A.; Ghasemi, Y. Structural characterization of polysaccharide-coated iron oxide nanoparticles produced by Staphylococcus warneri, isolated from a thermal spring. J. Basic Microbiol. 2019, 2019, 1–10. [Google Scholar] [CrossRef]
- Kianpour, S.; Ebrahiminezhad, A.; Negahdaripour, M.; Mohkam, M.; Mohammadi, F.; Niknezhad, S.; Ghasemi, Y. Characterization of biogenic Fe (III)-binding exopolysaccharide nanoparticles produced by Ralstonia sp. SK03. Biotechnol. Prog. 2018, 34, 1167–1176. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Zare, M.; Kiyanpour, S.; Berenjian, A.; Niknezhad, S.V.; Ghasemi, Y. Biosynthesis of xanthan gum coated iron nanoparticles by using Xanthomonas campestris. IET Nanobiotechnol. 2017, 151, 684–691. [Google Scholar] [CrossRef]
- Stolyar, S.; Yaroslavtsev, R.; Bayukov, O.; Balaev, D.; Krasikov, A.; Iskhakov, R.; Vorotynov, A.; Ladygina, V.; Purtov, K.; Volochaev, M. Preparation, structure and magnetic properties of synthetic ferrihydrite nanoparticles. J. Phys. Conf. Ser. 2018, 994, 012003. [Google Scholar] [CrossRef]
- Villacís-García, M.; Ugalde-Arzate, M.; Vaca-Escobar, K.; Villalobos, M.; Martínez-Villegas, N. Laboratory synthesis of goethite and ferrihydrite of controlled particle sizes. Bol. Soc. Geol. Mex. 2015, 67, 433–446. [Google Scholar] [CrossRef]
- Sokolov, I.; Cherkasov, V.; Vasilyeva, A.; Bragina, V.; Nikitin, M. Paramagnetic colloidal ferrihydrite nanoparticles for MRI contrasting. Colloids Surf. Physicochem. Eng. Aspects 2018, 539, 46–52. [Google Scholar] [CrossRef]
- Balaev, D.; Krasikov, A.; Dubrovskiy, A.; Popkov, S.; Stolyar, S.; Bayukov, O.; Iskhakov, R.; Ladygina, V.; Yaroslavtsev, R. Magnetic properties of heat treated bacterial ferrihydrite nanoparticles. J. Magn. Magn. Mater. 2016, 410, 171–180. [Google Scholar] [CrossRef]
- Rout, K.; Mohapatra, M.; Anand, S. 2-Line ferrihydrite: Synthesis, characterization and its adsorption behaviour for removal of Pb(II), Cd(II), Cu(II) and Zn(II) from aqueous solutions. Dalton Trans. 2012, 41, 3302–3312. [Google Scholar] [CrossRef] [PubMed]
- Antelo, J.; Arce, F.; Fiol, S. Arsenate and phosphate adsorption on ferrihydrite nanoparticles. synergetic interaction with calcium ions. Chem. Geol. 2015, 410, 53–62. [Google Scholar] [CrossRef]
- Tosco, T.; Bosch, J.; Meckenstock, R.U.; Sethi, R. Transport of ferrihydrite nanoparticles in saturated porous media: Role of ionic strength and flow rate. Environ. Sci. Technol. 2012, 46, 4008–4015. [Google Scholar] [CrossRef] [Green Version]
- Xiang, A.; Yan, W.; Koel, B.E.; Jaffé, P.R. Poly(acrylic acid) coating induced 2-line ferrihydrite nanoparticle transport in saturated porous media. J. Nanopart. Res. 2013, 15, 1705. [Google Scholar] [CrossRef]
- Pariona, N.; Martinez, A.I.; Hdz-García, H.; Cruz, L.A.; Hernandez-Valdes, A. Effects of hematite and ferrihydrite nanoparticles on germination and growth of maize seedlings. Saudi J. Biol. Sci. 2017, 24, 1547–1554. [Google Scholar] [CrossRef] [Green Version]
- Majidi, S.; Zeinali Sehrig, F.; Farkhani, S.M.; Soleymani Goloujeh, M.; Akbarzadeh, A. Current methods for synthesis of magnetic nanoparticles. Artif. Cells Nanomed. Biotechnol. 2016, 44, 722–734. [Google Scholar] [CrossRef]
- Döpke, C.; Grothe, T.; Steblinski, P.; Klöcker, M.; Sabantina, L.; Kosmalska, D.; Blachowicz, T.; Ehrmann, A. Magnetic nanofiber mats for data storage and transfer. Nanomaterials 2019, 9, 92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Domingos, D.G.; Henriques, R.O.; Xavier, J.A.; Junior, N.L.; da Costa, R.H.R. Increasing activated sludge aggregation by magnetite nanoparticles addition. Water Sci. Technol. 2019, 79, 993–999. [Google Scholar] [CrossRef]
- Alfaro, I.; Molina, L.; González, P.; Gaete, J.; Valenzuela, F.; Marco, J.F.; Sáez, C.; Basualto, C. Silica-coated magnetite nanoparticles functionalized with betaine and their use as an adsorbent for Mo(VI) and Re(VII) species from acidic aqueous solutions. J. Ind. Eng. Chem. 2019, 78, 271–283. [Google Scholar] [CrossRef]
- Molina, L.; Gaete, J.; Alfaro, I.; Ide, V.; Valenzuela, F.; Parada, J.; Basualto, C. Synthesis and characterization of magnetite nanoparticles functionalized with organophosphorus compounds and its application as an adsorbent for La(III), Nd(III) and Pr(III) ions from aqueous solutions. J. Mol. Liq. 2019, 275, 178–191. [Google Scholar] [CrossRef]
- Veisi, H.; Razeghi, S.; Mohammadi, P.; Hemmati, S. Silver nanoparticles decorated on thiol-modified magnetite nanoparticles (Fe3O4/SiO2-Pr-S-Ag) as a recyclable nanocatalyst for degradation of organic dyes. Mater. Sci. Eng. C 2019, 97, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.; Lin, M.; Huang, J.; Zhou, C.; Tian, W.; Yu, H.; Jiang, X.; Ye, J.; Shi, Y.; Xiao, Y. The recent advances of magnetic nanoparticles in Medicine. J. Nanomater. 2018, 2018, 7805147. [Google Scholar] [CrossRef] [Green Version]
- Thanh, B.T.; Hai, T.H.; Van, P.H.; Minh Tung, L.; Lee, J. Detection of hepatitis B surface antigen by immunoassay using magnetite nanoparticles binding hepatitis B surface antibody. Geosystem Eng. 2019, 22, 206–213. [Google Scholar] [CrossRef]
- Gupta, A.K.; Wells, S. Surface-modified superparamagnetic nanoparticles for drug delivery: Preparation, characterization, and cytotoxicity studies. IEEE Trans. NanoBiosci. 2004, 3, 66–73. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Davaran, S.; Rasoul-Amini, S.; Barar, J.; Moghadam, M.; Ghasemi, Y. Synthesis, characterization and anti-Listeria monocytogenes effect of amino acid coated magnetite nanoparticles. Curr. Nanosci. 2012, 8, 868–874. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Ghasemi, Y.; Rasoul-Amini, S.; Barar, J.; Davaran, S. Impact of amino-acid coating on the synthesis and characteristics of iron-oxide nanoparticles (IONs). Bull. Korean Chem. Soc. 2012, 33, 3957–3962. [Google Scholar] [CrossRef] [Green Version]
- Ebrahiminezhad, A.; Ghasemi, Y.; Rasoul-Amini, S.; Barar, J.; Davaran, S. Preparation of novel magnetic fluorescent nanoparticles using amino acids. Colloids Surf. B. 2013, 102, 534–539. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Rasoul-Amini, S.; Davaran, S.; Barar, J.; Ghasemi, Y. Impacts of iron oxide nanoparticles on the invasion power of Listeria monocytogenes. Curr. Nanosci. 2014, 10, 382–388. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Rasoul-Amini, S.; Kouhpayeh, A.; Davaran, S.; Barar, J.; Ghasemi, Y. Impacts of amine functionalized iron oxide nanoparticles on HepG2 cell line. Curr. Nanosci. 2015, 11, 113–119. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Varma, V.; Yang, S.; Berenjian, A. Magnetic immobilization of Bacillus subtilis natto cells for menaquinone-7 fermentation. Appl. Microbiol. Biotechnol. 2016, 100, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Ebrahiminezhad, A.; Varma, V.; Yang, S.; Ghasemi, Y.; Berenjian, A. Synthesis and application of amine functionalized iron oxide nanoparticles on menaquinone-7 fermentation: A step towards process intensification. Nanomaterials 2015, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Gholami, A.; Rasoul-Amini, S.; Ebrahiminezhad, A.; Abootalebi, N.; Niroumand, U.; Ebrahimi, N.; Ghasemi, Y. Magnetic properties and antimicrobial effect of amino and lipoamino acid coated iron oxide nanoparticles. Minerva Biotecnol. 2016, 28, 177–186. [Google Scholar]
- Gholami, A.; Rasoul-amini, S.; Ebrahiminezhad, A.; Seradj, S.H.; Ghasemi, Y. Lipoamino acid coated superparamagnetic iron oxide nanoparticles concentration and time dependently enhanced growth of human hepatocarcinoma cell line (Hep-G2). J. Nanomater. 2015, 2015, 451405. [Google Scholar] [CrossRef] [Green Version]
- Berry, C.C.; Wells, S.; Charles, S.; Curtis, A.S.G. Dextran and albumin derivatised iron oxide nanoparticles: Influence on fibroblasts in vitro. Biomaterials 2003, 24, 4551–4557. [Google Scholar] [CrossRef]
- Raee, M.J.; Ebrahiminezhad, A.; Gholami, A.; Ghoshoon, M.B.; Ghasemi, Y. Magnetic immobilization of recombinant E. coli producing extracellular asparaginase: An effective way to intensify downstream process. Sep. Sci. Technol. 2018, 53, 1397–1404. [Google Scholar] [CrossRef]
- Ranmadugala, D.; Ebrahiminezhad, A.; Manley-Harris, M.; Ghasemi, Y.; Berenjian, A. Iron oxide nanoparticles in modern microbiology and biotechnology. Crit. Rev. Microbiol. 2017, 43, 493–507. [Google Scholar] [CrossRef]
- Ranmadugala, D.; Ebrahiminezhad, A.; Manley-Harris, M.; Ghasemi, Y.; Berenjian, A. Magnetic immobilization of bacteria using iron oxide nanoparticles. Biotechnol. Lett. 2017, 40, 237–248. [Google Scholar] [CrossRef]
- Seifan, M.; Sarmah, A.K.; Samani, A.K.; Ebrahiminezhad, A.; Ghasemi, Y.; Berenjian, A. Mechanical properties of bio self-healing concrete containing immobilized bacteria with iron oxide nanoparticles. Appl. Microbiol. Biotechnol. 2018, 102, 4489–4498. [Google Scholar] [CrossRef]
- Seifan, M.; Ebrahiminezhad, A.; Ghasemi, Y.; Berenjian, A. Microbial calcium carbonate precipitation with high affinity to fill the concrete pore space: Nanobiotechnological approach. Bioprocess Biosyst. Eng. 2019, 42, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Seifan, M.; Ebrahiminezhad, A.; Ghasemi, Y.; Samani, A.K.; Berenjian, A. Amine-modified magnetic iron oxide nanoparticle as a promising carrier for application in bio self-healing concrete. Appl. Microbiol. Biotechnol. 2017, 102, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Seifan, M.; Ebrahiminezhad, A.; Ghasemi, Y.; Samani, A.K.; Berenjian, A. The role of magnetic iron oxide nanoparticles in the bacterially induced calcium carbonate precipitation. Appl. Microbiol. Biotechnol. 2018, 102, 3595–3606. [Google Scholar] [CrossRef] [PubMed]
- Seifan, M.; Sarmah, A.K.; Ebrahiminezhad, A.; Ghasemi, Y.; Samani, A.K.; Berenjian, A. Bio-reinforced self-healing concrete using magnetic iron oxide nanoparticles. Appl. Microbiol. Biotechnol. 2018, 102, 2167–2178. [Google Scholar] [CrossRef] [PubMed]
- Ernst, C.; Bartel, A.; Elferink, J.W.; Huhn, J.; Eschbach, E.; Schönfeld, K.; Feßler, A.T.; Oberheitmann, B.; Schwarz, S. Improved DNA extraction and purification with magnetic nanoparticles for the detection of methicillin-resistant Staphylococcus aureus. Vet. Microbiol. 2019, 230, 45–48. [Google Scholar] [CrossRef]
- Haghighi, A.H.; Faghih, Z.; Khorasani, M.T.; Farjadian, F. Antibody conjugated onto surface modified magnetic nanoparticles for separation of HER2+ breast cancer cells. J. Magn. Magn. Mater. 2019, 490, 165479. [Google Scholar] [CrossRef]
- Orhan, H.; Evli, S.; Dabanca, M.B.; Başbülbül, G.; Uygun, M.; Uygun, D.A. Bacteria killer enzyme attached magnetic nanoparticles. Mater. Sci. Eng. C 2019, 94, 558–564. [Google Scholar] [CrossRef]
- Berovic, M.; Berlot, M.; Kralj, S.; Makovec, D. A new method for the rapid separation of magnetized yeast in sparkling wine. Biochem. Eng. J. 2014, 88, 77–84. [Google Scholar] [CrossRef]
- Taghizadeh, S.-M.; Ebrahiminezhad, A.; Ghoshoon, M.B.; Dehshahri, A.; Berenjian, A.; Ghasemi, Y. Magnetic immobilization of Pichia pastoris cells for the production of recombinant human serum albumin. Nanomaterials 2020, 10, 111. [Google Scholar] [CrossRef] [Green Version]
- Taghizadeh, S.M.; Berenjian, A.; Chew, K.W.; Show, P.L.; Mohd Zaid, H.F.; Ramezani, H.; Ghasemi, Y.; Raee, M.J.; Ebrahiminezhad, A. Impact of magnetic immobilization on the cell physiology of green unicellular algae Chlorella vulgaris. Bioengineered 2020, 11, 141–153. [Google Scholar] [CrossRef] [Green Version]
- Ansari, F.; Grigoriev, P.; Libor, S.; Tothill, I.E.; Ramsden, J.J. DBT degradation enhancement by decorating Rhodococcus erythropolis IGST8 with magnetic Fe3O4 nanoparticles. Biotechnol. Bioeng. 2009, 102, 1505–1512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ranmadugala, D.; Ebrahiminezhad, A.; Manley-Harris, M.; Ghasemi, Y.; Berenjian, A. Impact of 3–aminopropyltriethoxysilane-coated iron oxide nanoparticles on menaquinone-7 production using B. subtilis. Nanomaterials 2017, 7, 350. [Google Scholar] [CrossRef] [PubMed]
- Ranmadugala, D.; Ebrahiminezhad, A.; Manley-Harris, M.; Ghasemi, Y.; Berenjian, A. The effect of iron oxide nanoparticles on Bacillus subtilis biofilm, growth and viability. Process Biochem. 2017, 62, 231–240. [Google Scholar] [CrossRef]
- Ranmadugala, D.; Ebrahiminezhad, A.; Manley-Harris, M.; Ghasemi, Y.; Berenjian, A. Reduced biofilm formation in Menaquinone-7 production process by optimizing the composition of the cultivation medium. Trends Pharma. Sci. 2017, 3, 245–254. [Google Scholar]
- Taghizadeh, S.-M.; Jafari, S.; Ahmad-Kiadaliri, T.; Mobasher, M.A.; Lal, N.; Raee, M.J.; Berenjian, A.; Ghasemi, Y.; Ebrahiminezhad, A. Magnetic immobilisation as a promising approach against bacteriophage infection. Mater. Res. Express. 2019, 6. [Google Scholar] [CrossRef]
- Dieudonné, A.; Pignol, D.; Prévéral, S. Magnetosomes: Biogenic iron nanoparticles produced by environmental bacteria. Appl. Microbiol. Biotechnol. 2019, 103, 3637–3649. [Google Scholar] [CrossRef]
- Yamazaki, T.; Suzuki, Y.; Kouduka, M.; Kawamura, N. Dependence of bacterial magnetosome morphology on chemical conditions in deep-sea sediments. Earth. Planet. Sci. Lett. 2019, 513, 135–143. [Google Scholar] [CrossRef]
- Gorobets, S.; Gorobets, O.; Bulaievska, M.; Sharay, I. Detection of biogenic magnetic nanoparticles in ethmoid bones of migratory and non-migratory fishes. SN Appl. Sci. 2019, 1, 63. [Google Scholar] [CrossRef] [Green Version]
- Gorobets, S.; Gorobets, O.; Magerman, A.; Gorobets, Y.; Sharay, I. Biogenic magnetic nanoparticles in plants. arXiv 2019, arXiv:1901.07212. [Google Scholar]
- Murros, K.E.; Salminen, J.; Wasiljeff, J.; Macías-Sánchez, E.; Soinne, L.S.; Faivre, D.; Valtonen, J.; Pohja, M.; Saari, P.; Pesonen, L. Magnetic nanoparticles in human cervical skin. Front. Med. 2019, 6, 123. [Google Scholar] [CrossRef]
- Van de Walle, A.; Sangnier, A.P.; Abou-Hassan, A.; Curcio, A.; Hémadi, M.; Menguy, N.; Lalatonne, Y.; Luciani, N.; Wilhelm, C. Biosynthesis of magnetic nanoparticles from nano-degradation products revealed in human stem cells. Proc. Natl. Acad. Sci. USA 2019, 116, 4044–4053. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brok, E.; Frandsen, C.; Madsen, D.E.; Jacobsen, H.; Birk, J.; Lefmann, K.; Bendix, J.; Pedersen, K.; Boothroyd, C.; Berhe, A. Magnetic properties of ultra-small goethite nanoparticles. J. Phys. D Appl. Phys. 2014, 47, 365003. [Google Scholar] [CrossRef]
- van der Zee, C.; Roberts, D.R.; Rancourt, D.G.; Slomp, C.P. Nanogoethite is the dominant reactive oxyhydroxide phase in lake and marine sediments. Geology 2003, 31, 993–996. [Google Scholar] [CrossRef]
- Ghanbariasad, A.; Taghizadeh, S.-M.; Show, P.L.; Nomanbhay, S.; Berenjian, A.; Ghasemi, Y.; Ebrahiminezhad, A. Controlled synthesis of iron oxyhydroxide (FeOOH) nanoparticles using secretory compounds from Chlorella vulgaris microalgae. Bioengineered 2019, 10, 390–396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, V.; Kynicky, J.; Ambrozova, P.; Adam, V. Microwave-Assisted Synthesis of Goethite Nanoparticles Used for Removal of Cr (VI) from Aqueous Solution. Materials 2017, 10, 783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, M.K.; Poinern, G.E.J.; Issa, T.B.; Singh, P. Arsenic adsorption on goethite nanoparticles produced through hydrazine sulfate assisted synthesis method. Korean J. Chem. Eng. 2012, 29, 95–102. [Google Scholar] [CrossRef]
- Kar, S.; Equeenuddin, S.M. Adsorption of hexavalent chromium using natural goethite: Isotherm, thermodynamic and kinetic study. J. Geol. Soc. India 2019, 93, 285–292. [Google Scholar] [CrossRef]
- Adegoke, H.; Adekola, F.; Ashola, M. Adsorptive removal of hexavalent chromium using synthetic goethite nanoparticles. Niger. J. Chem. Res. 2018, 23, 20–38. [Google Scholar]
- Tiraferri, A.; Hernandez, L.A.S.; Bianco, C.; Tosco, T.; Sethi, R. Colloidal behavior of goethite nanoparticles modified with humic acid and implications for aquifer reclamation. J. Nanopart. Res. 2017, 19, 107. [Google Scholar] [CrossRef]
- Kalishyn, Y.; Bychko, I.; Polunkin, E.; Kameneva, T.; Strizhak, P. Enhancement in the oxidative stability of commercial gasoline fuel by the goethite nanoparticles. ChemRxiv. Preprint 2018. [Google Scholar] [CrossRef]
- Zhao, J.; Lin, W.; Chang, Q.; Li, W.; Lai, Y. Adsorptive characteristics of akaganeite and its environmental applications: A review. Environ. Technol. Rev. 2012, 1, 114–126. [Google Scholar] [CrossRef]
- Kim, J.; Grey, C.P. 2H and 7Li solid-state MAS NMR study of local environments and lithium adsorption on the iron(III) oxyhydroxide, akaganeite (β-FeOOH). Chem. Mater. 2010, 22, 5453–5462. [Google Scholar] [CrossRef]
- Mohapatra, J.; Mitra, A.; Tyagi, H.; Bahadur, D.; Aslam, M. Iron oxide nanorods as high-performance magnetic resonance imaging contrast agents. Nanoscale 2015, 7, 9174–9184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasparis, G.; Erdocio, A.S.; Tuffnell, J.M.; Thanh, N.T.K. Synthesis of size-tuneable β-FeOOH nanoellipsoids and a study of their morphological and compositional changes by reduction. CrystEngComm 2019, 21, 1293–1301. [Google Scholar] [CrossRef] [Green Version]
- Yuan, Z.-Y.; Su, B.-L. Surfactant-assisted nanoparticle assembly of mesoporous β-FeOOH (akaganeite). Chem. Phys. Lett. 2003, 381, 710–714. [Google Scholar] [CrossRef]
- Faria, M.C.; Rosemberg, R.S.; Bomfeti, C.A.; Monteiro, D.S.; Barbosa, F.; Oliveira, L.C.; Rodriguez, M.; Pereira, M.C.; Rodrigues, J.L. Arsenic removal from contaminated water by ultrafine δ-FeOOH adsorbents. Chem. Eng. J. 2014, 237, 47–54. [Google Scholar] [CrossRef]
- Zhang, Y.-X.; Jia, Y. A facile solution approach for the synthesis of akaganéite (β-FeOOH) nanorods and their ion-exchange mechanism toward As(V) ions. Appl. Surf. Sci. 2014, 290, 102–106. [Google Scholar] [CrossRef]
- Legg, B.A. The Formation and Aggregation of Iron Oxyhydroxide Nanoparticles in the Aqueous Environment. Ph.D. Thesis, UC Berkeley, Berkeley, CA, USA, 2013. [Google Scholar]
- Majzlan, J.; Mazeina, L.; Navrotsky, A. Enthalpy of water adsorption and surface enthalpy of lepidocrocite (γ-FeOOH). Geochim. Cosmochim. Acta 2007, 71, 615–623. [Google Scholar] [CrossRef]
- Vindedahl, A.M.; Strehlau, J.H.; Arnold, W.A.; Penn, R.L. Organic matter and iron oxide nanoparticles: Aggregation, interactions, and reactivity. Environ. Sci. Nano 2016, 3, 494–505. [Google Scholar] [CrossRef]
- Navarro, G.; Acevedo, R.; Soto, A.; Herane, M. Synthesis and characterization of lepidocrocite and its potential applications in the adsorption of pollutant species. J. Phys. Conf. Ser. 2008, 134, 012023. [Google Scholar] [CrossRef]
- Lee, G.; Kim, S.; Choi, B.; Huh, S.; Chang, Y.; Kim, B.; Park, J.; Oh, S. Magnetic properties of needle-like α-FeOOH and γ-FeOOH nanoparticles. J. Korean Phys. Soc. 2004, 45, 1019–1024. [Google Scholar]
- Ewing, F. The crystal structure of lepidocrocite. J. Chem. Phys. 1935, 3, 420–424. [Google Scholar] [CrossRef]
- Liu, A.; Liu, J.; Pan, B.; Zhang, W.-x. Formation of lepidocrocite (γ-FeOOH) from oxidation of nanoscale zero-valent iron (nZVI) in oxygenated water. RSC Adv. 2014, 4, 57377–57382. [Google Scholar] [CrossRef]
- Agarwal, A.; Joshi, H.; Kumar, A. Synthesis, characterization and application of nano lepidocrocite and magnetite in the degradation of carbon tetrachloride. S. Afr. J. Chem. 2011, 64, 218–224. [Google Scholar]
- Sheydaei, M.; Aber, S. Preparation of nano-lepidocrocite and an investigation of its ability to remove a metal complex dye. Clean 2013, 41, 890–898. [Google Scholar] [CrossRef]
- Alexandratos, V.G.; Behrends, T.; Van Cappellen, P. Fate of adsorbed U(VI) during sulfidization of lepidocrocite and hematite. Environ. Sci. Technol. 2017, 51, 2140–2150. [Google Scholar] [CrossRef]
- O’Loughlin, E.J.; Gorski, C.A.; Flynn, T.M.; Scherer, M.M. Electron donor utilization and secondary mineral formation during the bioreduction of lepidocrocite by Shewanella putrefaciens CN32. Minerals 2019, 9, 434. [Google Scholar] [CrossRef] [Green Version]
- Koch, C.B.; Oxborrow, C.; Mørup, S.; Madsen, M.; Quinn, A.; Coey, J. Magnetic properties of feroxyhyte (δ-FeOOH). Phys. Chem. Miner. 1995, 22, 333–341. [Google Scholar] [CrossRef]
- Chen, P.; Xu, K.; Li, X.; Guo, Y.; Zhou, D.; Zhao, J.; Wu, X.; Wu, C.; Xie, Y. Ultrathin nanosheets of feroxyhyte: A new two-dimensional material with robust ferromagnetic behavior. Chem. Sci. 2014, 5, 2251–2255. [Google Scholar] [CrossRef]
- Chukhrov, F.; Zvyagin, B.; Gorshkov, A.; Yermilova, L.; Korovushkin, V.; Rudnitskaya, Y.S.; Yakubovskaya, N.Y. Feroxyhyte, a new modification of FeOOH. Int. Geol. Rev. 1977, 19, 873–890. [Google Scholar] [CrossRef]
- Nishida, N.; Amagasa, S.; Kobayashi, Y.; Yamada, Y. Synthesis of Cu-doped δ-FeOOH nanoparticles by a wet chemical method. J. Nanopart. Res. 2018, 20, 181. [Google Scholar] [CrossRef]
- Nishida, N.; Amagasa, S.; Ito, H.; Kobayashi, Y.; Yamada, Y. Manganese-doped feroxyhyte nano-urchins produced by chemical methods. Hyperfine Interact. 2018, 239, 33. [Google Scholar] [CrossRef]
- Jurkin, T.; Štefanić, G.; Dražić, G.; Gotić, M. Synthesis route to δ-FeOOH nanodiscs. Mater. Lett. 2016, 173, 55–59. [Google Scholar] [CrossRef]
- Pinto, I.S.; Pacheco, P.H.; Coelho, J.V.; Lorençon, E.; Ardisson, J.D.; Fabris, J.D.; de Souza, P.P.; Krambrock, K.W.; Oliveira, L.C.; Pereira, M.C. Nanostructured δ-FeOOH: An efficient Fenton-like catalyst for the oxidation of organics in water. Appl. Catal. B 2012, 119, 175–182. [Google Scholar] [CrossRef]
- Chagas, P.; Da Silva, A.C.; Passamani, E.C.; Ardisson, J.D.; de Oliveira, L.C.A.; Fabris, J.D.; Paniago, R.M.; Monteiro, D.S.; Pereira, M.C. δ-FeOOH: A superparamagnetic material for controlled heat release under AC magnetic field. J. Nanopart. Res. 2013, 15, 1544. [Google Scholar] [CrossRef]
- Li, Y.; Fu, F.; Cai, W.; Tang, B. Synergistic effect of mesoporous feroxyhyte nanoparticles and Fe(II) on phosphate immobilization: Adsorption and chemical precipitation. Powder Technol. 2019, 345, 786–795. [Google Scholar] [CrossRef]
- Pinakidou, F.; Katsikini, M.; Paloura, E.; Simeonidis, K.; Mitraka, E.; Mitrakas, M. Monitoring the role of Mn and Fe in the As-removal efficiency of tetravalent manganese feroxyhyte nanoparticles from drinking water: An X-ray absorption spectroscopy study. J. Colloid Interface Sci. 2016, 477, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Kokkinos, E.; Soukakos, K.; Kostoglou, M.; Mitrakas, M. Cadmium, mercury, and nickel adsorption by tetravalent manganese feroxyhyte: Selectivity, kinetic modeling, and thermodynamic study. Environ. Sci. Pollut. Res. 2018, 25, 12263–12273. [Google Scholar] [CrossRef] [PubMed]
- Tresintsi, S.; Simeonidis, K.; Estradé, S.; Martinez-Boubeta, C.; Vourlias, G.; Pinakidou, F.; Katsikini, M.; Paloura, E.C.; Stavropoulos, G.; Mitrakas, M. Tetravalent manganese feroxyhyte: A novel nanoadsorbent equally selective for As(III) and As(V) removal from drinking water. Environ. Sci. Technol. 2013, 47, 9699–9705. [Google Scholar] [CrossRef]
- Hu, J.; Lo, I.M.; Chen, G. Performance and mechanism of chromate (VI) adsorption by δ-FeOOH-coated maghemite (γ-Fe2O3) nanoparticles. Sep. Purif. Technol. 2007, 58, 76–82. [Google Scholar] [CrossRef]
- Pereira, M.C.; Garcia, E.M.; da Silva, A.C.; Lorençon, E.; Ardisson, J.D.; Murad, E.; Fabris, J.D.; Matencio, T.; de Castro Ramalho, T.; Rocha, M.V.J. Nanostructured δ-FeOOH: A novel photocatalyst for water splitting. J. Mater. Chem. 2011, 21, 10280–10282. [Google Scholar] [CrossRef]
- da Silva Rocha, T.; Nascimento, E.S.; da Silva, A.C.; dos Santos Oliveira, H.; Garcia, E.M.; de Oliveira, L.C.A.; Monteiro, D.S.; Rodriguez, M.; Pereira, M.C. Enhanced photocatalytic hydrogen generation from water by Ni(OH)2 loaded on Ni-doped δ-FeOOH nanoparticles obtained by one-step synthesis. RSC Adv. 2013, 3, 20308–20314. [Google Scholar] [CrossRef]
- Bigham, J.; Carlson, L.; Murad, E. Schwertmannite, a new iron oxyhydroxysulphate from Pyhäsalmi, Finland, and other localities. Mineral. Mag. 1994, 58, 641–648. [Google Scholar] [CrossRef] [Green Version]
- Sestu, M.; Navarra, G.; Carrero, S.; Valvidares, S.M.; Aquilanti, G.; Pérez-Lopez, R.; Fernandez-Martinez, A. Whole-nanoparticle atomistic modeling of the schwertmannite structure from total scattering data. J. Appl. Crystallogr. 2017, 50, 1617–1626. [Google Scholar] [CrossRef]
- Zhang, Z.; Bi, X.; Li, X.; Zhao, Q.; Chen, H. Schwertmannite: Occurrence, properties, synthesis and application in environmental remediation. RSC Adv. 2018, 8, 33583–33599. [Google Scholar] [CrossRef] [Green Version]
- Paikaray, S.; Schröder, C.; Peiffer, S. Schwertmannite stability in anoxic Fe(II)-rich aqueous solution. Geochim. Cosmochim. Acta 2017, 217, 292–305. [Google Scholar] [CrossRef]
- Miyata, N.; Takahashi, A.; Fujii, T.; Hashimoto, H.; Takada, J. Biosynthesis of Schwertmannite and Goethite in a bioreactor with acidophilic Fe(II)-oxidizing betaproteobacterium strain GJ-E10. Minerals 2018, 8, 98. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Yang, M.; Yao, T.; Xiong, H. Isolation, identification and arsenic-resistance of Acidithiobacillus ferrooxidans HX3 producing schwertmannite. J. Environ. Sci. 2014, 26, 1463–1470. [Google Scholar] [CrossRef]
- Song, Y.; Liu, Y.; Wang, H. Comparison of the biological and chemical synthesis of schwertmannite at a consistent Fe2+ oxidation efficiency and the effect of extracellular polymeric substances of Acidithiobacillus ferrooxidans on biomineralization. Materials 2018, 11, 1739. [Google Scholar] [CrossRef] [Green Version]
- Kumpulainen, S.; Räisänen, M.-L.; Von der Kammer, F.; Hofmann, T. Ageing of synthetic and natural schwertmannites at pH 2–8. Clay Miner. 2008, 43, 437–448. [Google Scholar] [CrossRef]
- Qiao, X.; Liu, L.; Shi, J.; Zhou, L.; Guo, Y.; Ge, Y.; Fan, W.; Liu, F. Heating changes bio-schwertmannite microstructure and arsenic(III) removal efficiency. Minerals 2017, 7, 9. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, J.; Zhang, S.; Zhou, L.; Xu, J.; Ge, Y.; Fan, W.; Liu, F. Schwertmannite adherence to the reactor wall during the bio-synthesis process and deterioration of its structural characteristics and arsenic(III) removal efficiency. Minerals 2017, 7, 64. [Google Scholar] [CrossRef]
- Birch, W.D.; Pring, A.; Reller, A.; Schmalle, H.W. Bernalite, Fe(OH)3, a new mineral from Broken Hill, New South Wales: Description and structure. Am. Mineral. 1993, 78, 827–834. [Google Scholar]
- Welch, M.; Crichton, W.; Ross, N. Compression of the perovskite-related mineral bernalite Fe(OH)3 to 9 GPa and a reappraisal of its structure. Mineral. Mag. 2005, 69, 309–315. [Google Scholar] [CrossRef]
- Han, J.; Ro, H.-M. Identification of bernalite transformation and tridentate arsenate complex at nano-goethite under effects of drying, pH and surface loading. Sci. Rep. 2018, 8, 8369. [Google Scholar] [CrossRef]
- Grundl, T.; Delwiche, J. Kinetics of ferric oxyhydroxide precipitation. J. Contam. Hydrol. 1993, 14, 71–87. [Google Scholar] [CrossRef]
- Chen, Z. Synthesis of monodisperse organic-and water-soluble iron oxide nanoparticles using Fe(OH)3 as precursor. J. Exp. Nanosci. 2014, 9, 406–414. [Google Scholar] [CrossRef]
- Pinto, P.S.; Lanza, G.D.; Ardisson, J.D.; Lago, R.M. Controlled dehydration of Fe(OH)3 to Fe2O3: Developing mesopores with complexing iron species for the adsorption of β-Lactam antibiotics. J. Braz. Chem. Soc. 2019, 30, 310–317. [Google Scholar] [CrossRef]
- Thuy, L.T.X.; Cuong, L.P.; Yabutani, T. Removal of trace metals from aqueous solution by Fe(OH)3 coprecipitation and flotation using poly-glutamic acid. Int. Res. J. Pure Appl. Chem. 2014, 4, 797–804. [Google Scholar] [CrossRef]
- Arefi, M.; Saberi, D.; Karimi, M.; Heydari, A. Superparamagnetic Fe(OH)3@Fe3O4 nanoparticles: An efficient and recoverable catalyst for tandem oxidative amidation of alcohols with amine hydrochloride salts. ACS Comb. Sci. 2015, 17, 341–347. [Google Scholar] [CrossRef]
- Yan, J.; Tang, H.; Lin, Z.; Anjum, M.N.; Zhu, L. Efficient degradation of organic pollutants with ferrous hydroxide colloids as heterogeneous Fenton-like activator of hydrogen peroxide. Chemosphere 2012, 87, 111–117. [Google Scholar] [CrossRef]





© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taghizadeh, S.-M.; Berenjian, A.; Zare, M.; Ebrahiminezhad, A. New Perspectives on Iron-Based Nanostructures. Processes 2020, 8, 1128. https://doi.org/10.3390/pr8091128
Taghizadeh S-M, Berenjian A, Zare M, Ebrahiminezhad A. New Perspectives on Iron-Based Nanostructures. Processes. 2020; 8(9):1128. https://doi.org/10.3390/pr8091128
Chicago/Turabian StyleTaghizadeh, Seyedeh-Masoumeh, Aydin Berenjian, Marziyeh Zare, and Alireza Ebrahiminezhad. 2020. "New Perspectives on Iron-Based Nanostructures" Processes 8, no. 9: 1128. https://doi.org/10.3390/pr8091128




