Rendering Fat and Heavy Fischer-Tropsch Waxes Mixtures (0–100%) Fast Pyrolysis Tests for the Production of Ethylene and Propylene
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Comission. Energy Roadmap—Impact Assessment and Scenario Analysis; European Comission: Brussels, Belgium, 2011; pp. 1–192. [Google Scholar]
- European Environment Agency. Share of Renewable Energy in Gross Final Energy Consumption in Europe; European Environment Agency: Copenhagen, Denmark, 2015. [Google Scholar]
- U.S. Energy Information Administration. International Energy Outlook 2019 with Projections to 2050; U.S. Energy Information Administration, Office of Energy Analysis, U.S. Department of Energy: Washington, DC, USA, 2019.
- Karaba, A.; Dvořakova, V.; Jan Patera, J.; Zamostny, P. Improving the steam-cracking efficiency of naphtha feedstocks by mixed/separate processing. J. Anal. Appl. Pyrol. 2020, 146, 104768. [Google Scholar] [CrossRef]
- Hsu, C.S.; Robinson, P.R. Petroleum System and Occurrence. In Petroleum Science and Technology; Springer: Cham, Switzerland, 2019; pp. 67–82. [Google Scholar] [CrossRef]
- Moreira, J.V. Steam Cracking: Kinetics and Feed Characterisation; Instituto Superior Tecnico: Lisbon, Portugal, November 2015. Available online: https://fenix.tecnico.ulisboa.pt/downloadFile/1126295043834327/JVM_ExtendedAbstract.pdf (accessed on 25 January 2021).
- Dupain, X.; Krul, R.A.; Makkee, M.; Moulijn, J.A. Are Fischer–Tropsch waxes good feedstocks for fluid catalytic cracking units? Catal. Today 2005, 106, 288–292. [Google Scholar] [CrossRef]
- Fogassy, G.; Thegarid, N.; Schuurman, Y.; Mirodatos, C. The fate of bio-carbon in FCC co-processing products. Green Chem. 2012, 14, 1367–1371. [Google Scholar] [CrossRef]
- Herink, T.; Belohlav, Z.; Zamostny, P.; Doskocil, J. Application of Hydrocarbon Cracking Experiments to Ethylene Unit Control and Optimization. Petrol. Chem. 2006, 46, 237–245. [Google Scholar] [CrossRef]
- Hidalgo, J.M.; Tišler, Z.; Vráblík, A.; Velvarská, R.; Lederer, J. Acid-modified phonolite and foamed zeolite as supports for NiW catalysts for deoxygenation of waste rendering fat. React. Kinet. Mech. Cat. 2019, 126, 773–793. [Google Scholar] [CrossRef]
- Karaba, A.; Rozhon, J.; Patera, J.; Hajek, J.; Zamostny, P. Fischer-Tropsch Wax from Renewable Resources as an Excellent Feedstock for the Steam-Cracking Process. Chem. Eng. Technol. 2020, 44, 329–338. [Google Scholar] [CrossRef]
- Simacek, P.; Kubicka, D.; Pospisil, M.; Rubas, M.; Hora, L.; Sebor, G. Fischer–Tropsch product as a co-feed for refinery hydrocracking unit. Fuel 2013, 105, 432–439. [Google Scholar] [CrossRef]
- Kang, J.; Ma, W.; Keogh, R.A.; Shafer, W.D.; Jacobs, G.; Davis, B.H. Hydrocracking and Hydroisomerization of n-Hexadecane, n-Octacosane and Fischer–Tropsch Wax Over a Pt/SiO2–Al2O3 Catalyst. Catal. Lett. 2012, 142, 1295–1305. [Google Scholar] [CrossRef]
- Wang, F.; Xu, J.; Ren, J.; Li, Y. Experimental investigation and modeling of steam cracking of Fischer–Tropsch naphtha for light olefins. Chem. Eng. Process. 2010, 49, 51–58. [Google Scholar] [CrossRef]
- Dennis, J.O.; Lei, G.D. High purity naphthas for the production of ethylene and propylene. World Intellectual Property. Organization Patent International Publication Number WO2004069961A2, 19 August 2004. [Google Scholar]
- Zamostny, P.; Belohalv, Z.; Starkbaumova, L. A Multipurpose Micro-pulse Reactor for Studying Gas-phase Reactions. Chem. Biochem. Eng. Q. 2007, 21, 105–113. [Google Scholar]
- Zamostny, P.; Belohlav, Z.; Starkbaumova, L.; Patera, J. Experimental study of hydrocarbon structure effects on the composition of its pyrolysis products. J. Anal. Appl. Pyrolysis 2010, 87, 207–216. [Google Scholar] [CrossRef]
- Belohlav, Z.; Pavlik, D.; Herink, T.; Svoboda, P.; Zamostny, P.; Marek, J. Use of Pyrolysis Gas Chromatography for Evaluation of Thermal Cracking of Naphtha. Chem. Listy 2002, 96, 325–329. [Google Scholar]
- Belohlav, Z.; Herink, T.; Lederer, J.; Marek, J.; Rachova, N.; Svoboda, P.; Zamostny, P.; Vojtova, D. Evaluation of pyrolysis feedstock by pyrolysis gas chromatography. Pet. Chem. 2005, 45, 118–125. [Google Scholar]
- Smidrkal, J.; Belohlav, Z.; Zamostny, P.; Filip, V. Olefin production through pyrolysis of triacylglycerols. Lipid Technol. 2009, 21, 220–223. [Google Scholar] [CrossRef]
- Zamostny, P.; Belohlav, Z.; Smidrkal, J. Production of olefins via steam cracking of vegetable oils. J. Resour. Conserv. Recycl. 2012, 59, 47–51. [Google Scholar] [CrossRef]
- Murat, M.; Lederer, J.; Rodová, A.; Hidalgo Herrador, J.M. Hydrodeoxygenation and pyrolysis of free fatty acids obtained from waste rendering fat. Eclet. Quim. 2020, 45, 28–36. [Google Scholar] [CrossRef]
- Pinto, F.; Paradela, F.; Gulyurtlu, I.; Ramos, A.M. Prediction of liquid yields from the pyrolysis of waste mixtures using response surface methodology. Fuel Process. Technol. 2013, 116, 271–283. [Google Scholar] [CrossRef]
- Ganesamoorthi, B.; Kalaivanan, S.; Dinesh, R.; Naveenkumar, T.; Anand, K. Optimization Technique using Response Surface Method for USMW process. Procedia Soc. Behav. Sci. 2015, 189, 169–174. [Google Scholar] [CrossRef]
- Amghizar, I.; Vandewalle, L.A.; Van Geem, K.M.; Marin, G.B. New Trends in Olefin Production. Engineering 2017, 3, 171–178. [Google Scholar] [CrossRef]
- Maher, K.D.; Bressler, D.C. Pyrolysis of triglyceride materials for the production of renewable fuels and chemicals. Bioresour. Technol. 2007, 98, 2351–2368. [Google Scholar] [CrossRef]
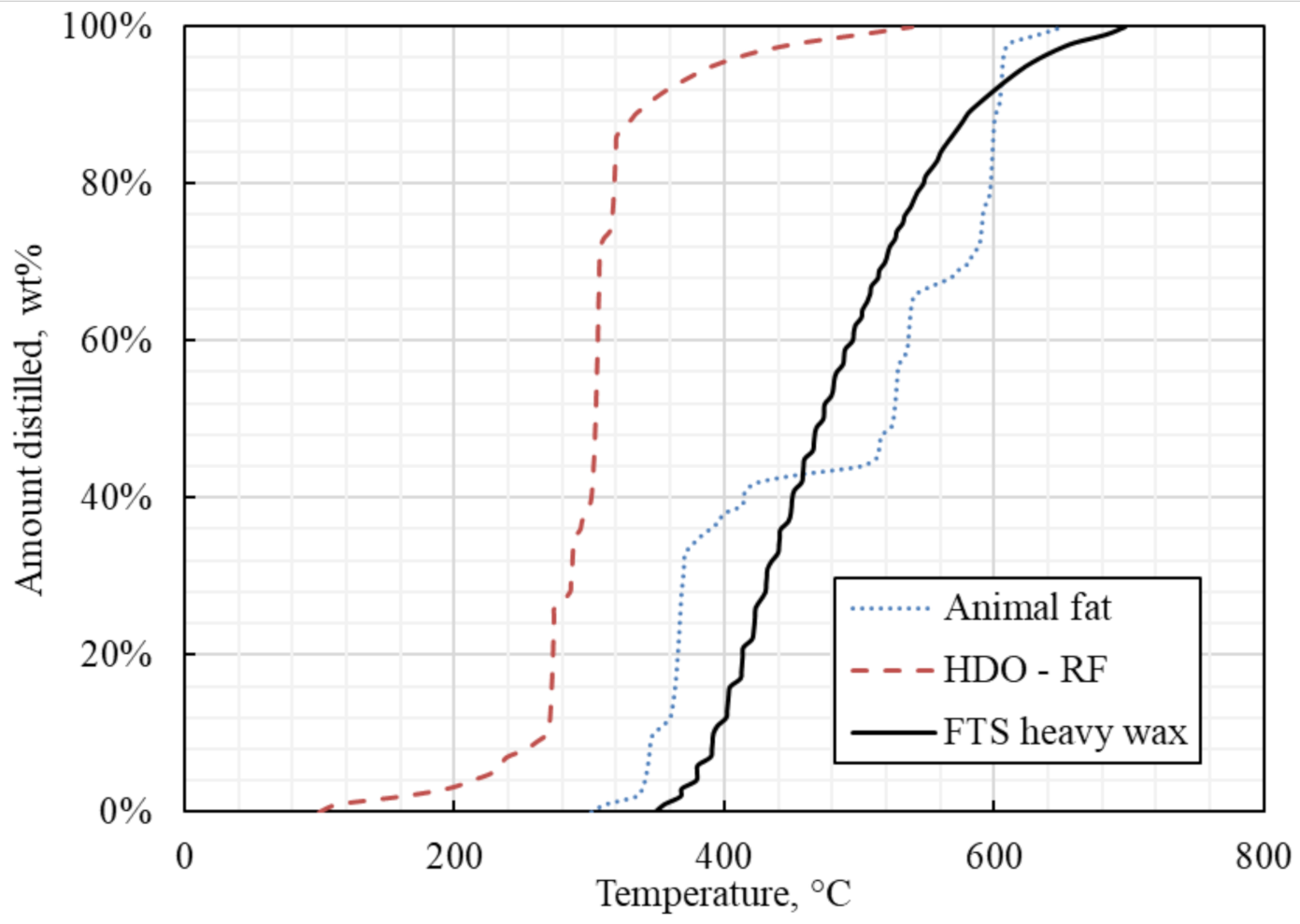

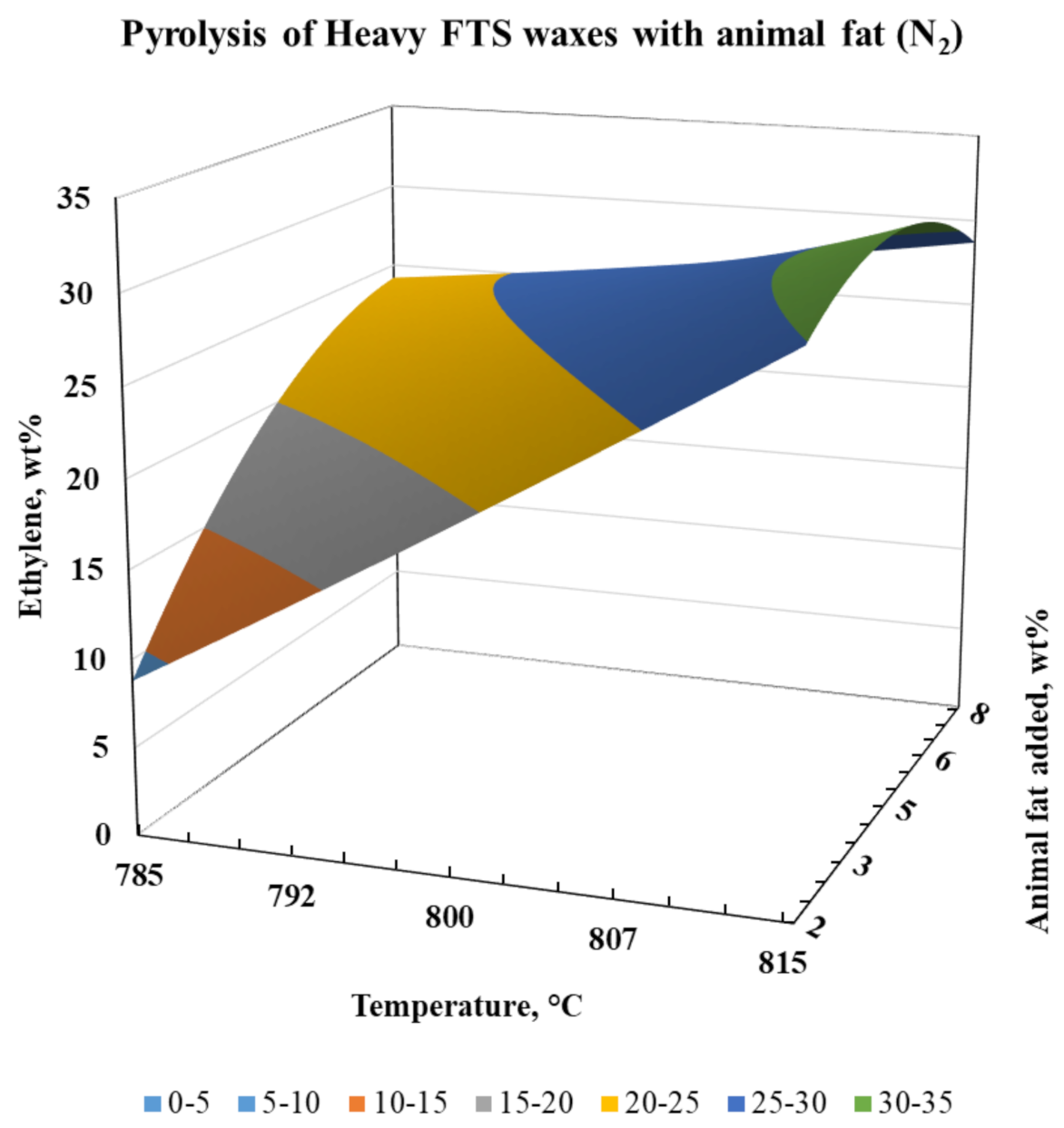

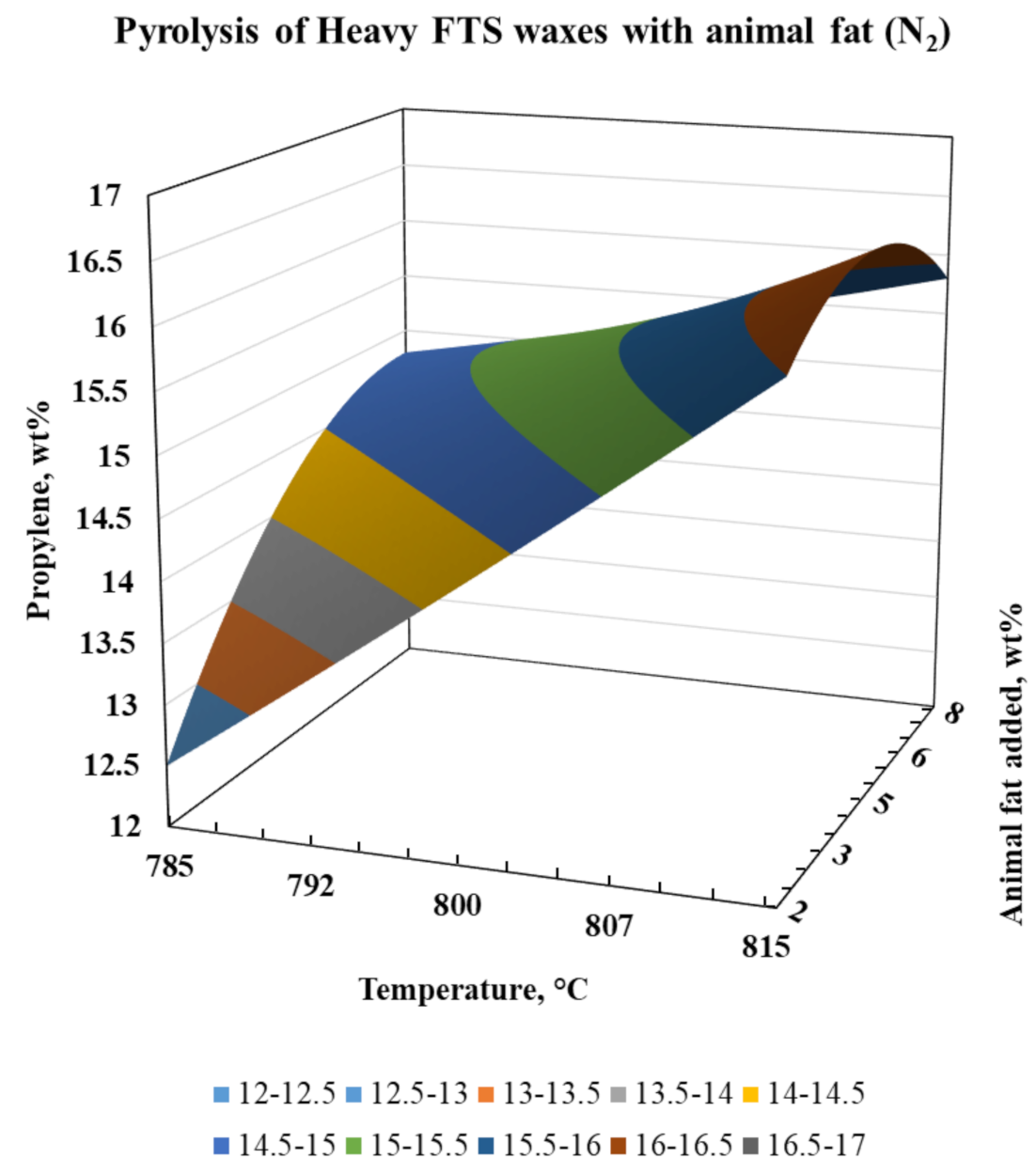

| Metal Composition | Amount | Units |
|---|---|---|
| Al | <0.4 | mg kg−1 |
| Ca | 110 | mg kg−1 |
| Cr | 6.38 | mg kg−1 |
| Fe | 13.3 | mg kg−1 |
| K | 218 | mg kg−1 |
| Mg | 16.6 | mg kg−1 |
| Mn | < 0.2 | mg kg−1 |
| Na | 216 | mg kg−1 |
| Ni | <0.2 | mg kg−1 |
| P | 50.1 | mg kg−1 |
| Ti | <0.2 | mg kg−1 |
| C | 76.6 | wt% |
| H | 12.3 | wt% |
| S | -- | mg kg−1 |
| N | 800 | mg kg−1 |
| Oxygen by difference | 11.0 | wt% |
| Ash content1 | 0.3 | wt% |
| Acid value2 | 65.5 | mg KOH g−1 |
| Water content | 2650.3 | mg kg−1 |
| Amount Distilled, wt% | Temperature, °C | nCx, (n-alkanes) |
|---|---|---|
| Initial Boiling Point | 348.34 | nC20 |
| 1% | 355.16 | nC21 |
| 5% | 379.39 | nC22-nC23 |
| 10% | 391.36 | nC24 |
| 20% | 413.18 | nC25-nC30 |
| 30% | 431.04 | nC30-nC31 |
| 40% | 449.88 | nC31-nC32 |
| 50% | 472.67 | nC32-nC34 |
| 60% | 494.51 | nC34-nC35 |
| 70% | 518.57 | nC36-nC37 |
| 80% | 547.05 | nC38-nC39 |
| 90% | 586.37 | nC39-nC42 |
| Final Boiling Point | 696.48 | nC43+ |
| Material | FT Residue |
|---|---|
| C, % | 84.5 |
| H, % | 14.7 |
| N, ppm | 83.2 |
| S, ppm | 29.6 |
| H/C ratio | 2.07 |
| Test Name | Natural Variables | Coded Factors | ||
|---|---|---|---|---|
| Temperature, °C | RF, wt% | Z1 | Z2 | |
| 1 | 785 | 2 | - | - |
| 2 | 815 | 2 | + | - |
| 3 | 785 | 8 | - | + |
| 4 | 815 | 8 | + | + |
| 5 | 800 | 4 | = | = |
| 6 | 800 | 4 | = | = |
| 7 | 800 | 4 | = | = |
| 8 | 800 | 4 | = | = |
| Sample | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Temperature, °C- RF content, wt% | 785-2 | 815-2 | 785-8 | 815-8 | 800-4 | 800-4 | 800-4 | 800-4 |
| Methane | 4.90 | 3.94 | 5.35 | 5.98 | 5.49 | 5.12 | 5.50 | 5.82 |
| Ethane | 1.45 | 2.77 | 3.72 | 3.47 | 4.23 | 4.02 | 3.88 | 4.30 |
| Ethylene | 8.85 | 29.94 | 24.53 | 28.74 | 23.82 | 23.04 | 25.24 | 25.21 |
| Propane | 0.80 | 0.75 | 0.72 | 0.61 | 0.95 | 0.98 | 0.74 | 0.92 |
| Propylene | 12.49 | 16.03 | 14.82 | 15.81 | 14.95 | 14.29 | 15.32 | 15.76 |
| Acethylene | 0.01 | 0.01 | 0.09 | 0.21 | 0.10 | 0.08 | 0.12 | 0.11 |
| iso-butane | 0.06 | 0.10 | 0.07 | 0.12 | 0.07 | 0.07 | 0.09 | 0.08 |
| Propadiene | 0.23 | 0.18 | 0.17 | 0.12 | 0.21 | 0.23 | 0.16 | 0.20 |
| n-butane | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| t-2-butene | 0.00 | 0.00 | 0.18 | 0.25 | 0.17 | 0.00 | 0.22 | 0.18 |
| 1-butene | 6.6 | 7.76 | 6.10 | 6.27 | 6.15 | 6.33 | 6.50 | 6.38 |
| i-butene | 0.18 | 0.10 | 0.30 | 0.30 | 0.33 | 0.30 | 0.31 | 0.33 |
| c-2-butene | 0.22 | 0.36 | 0.36 | 0.34 | 0.39 | 0.36 | 0.35 | 0.39 |
| butadiene | 5.30 | 7.08 | 6.04 | 6.95 | 6.19 | 5.99 | 6.45 | 4.00 |
| C6-C7 | 26.30 | 25.35 | 29.74 | 26.38 | 28.67 | 28.81 | 28.55 | 29.71 |
| Toluene | 0.0 | 0.15 | 1.07 | 0.59 | 1.07 | 1.25 | 0.98 | 0.98 |
| C7+ and others | 32.6 | 5.45 | 6.74 | 3.85 | 7.19 | 9.13 | 5.60 | 5.63 |
| EPropylene | 0.00 | 0.00 | 0.00 | 0.00 | 2.10 | 5.65 | −3.57 | −3.57 |
| RPropylene | 0.99 | |||||||
| SSEPropylene | 3.56 | |||||||
| EEthylene | 0.03 | −0.03 | −0.01 | 0.01 | 0.84 | 5.53 | −1.55 | −4.30 |
| REthylene | 0.99 | |||||||
| SSEEthylene | 1.16 | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murat, M.; Gholami, Z.; Šimek, J.; Rodríguez-Padrón, D.; Hidalgo-Herrador, J.M. Rendering Fat and Heavy Fischer-Tropsch Waxes Mixtures (0–100%) Fast Pyrolysis Tests for the Production of Ethylene and Propylene. Processes 2021, 9, 367. https://doi.org/10.3390/pr9020367
Murat M, Gholami Z, Šimek J, Rodríguez-Padrón D, Hidalgo-Herrador JM. Rendering Fat and Heavy Fischer-Tropsch Waxes Mixtures (0–100%) Fast Pyrolysis Tests for the Production of Ethylene and Propylene. Processes. 2021; 9(2):367. https://doi.org/10.3390/pr9020367
Chicago/Turabian StyleMurat, Martyna, Zahra Gholami, Josef Šimek, Daily Rodríguez-Padrón, and José Miguel Hidalgo-Herrador. 2021. "Rendering Fat and Heavy Fischer-Tropsch Waxes Mixtures (0–100%) Fast Pyrolysis Tests for the Production of Ethylene and Propylene" Processes 9, no. 2: 367. https://doi.org/10.3390/pr9020367
APA StyleMurat, M., Gholami, Z., Šimek, J., Rodríguez-Padrón, D., & Hidalgo-Herrador, J. M. (2021). Rendering Fat and Heavy Fischer-Tropsch Waxes Mixtures (0–100%) Fast Pyrolysis Tests for the Production of Ethylene and Propylene. Processes, 9(2), 367. https://doi.org/10.3390/pr9020367






