The Effects of Conjugated Linoleic Acids on Cancer
Abstract
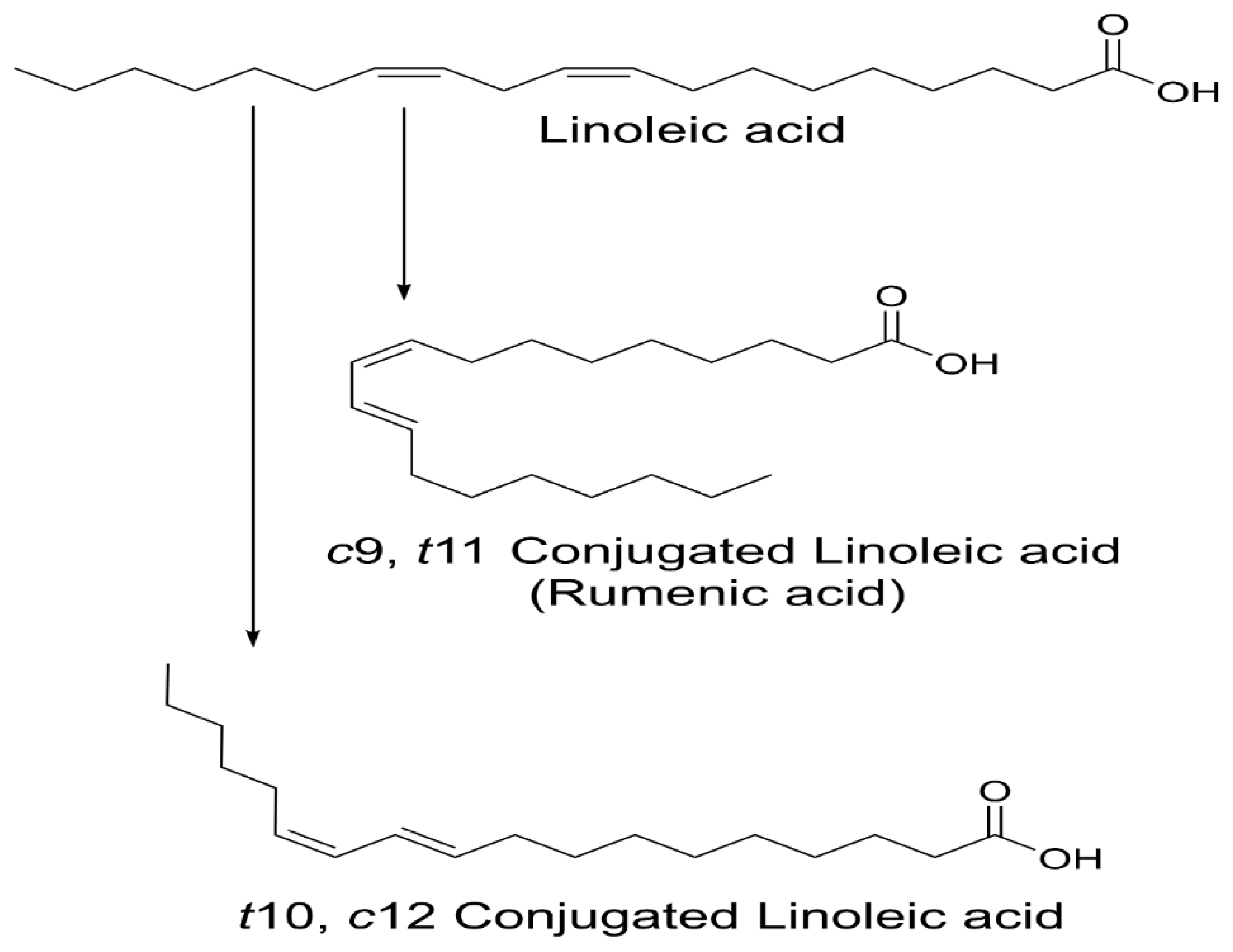
1. Introduction
2. Animal Models
2.1. Colon Cancer
2.2. Breast and Mammary Cancer
2.3. Prostate Cancer
2.4. Pancreatic and Liver Cancer
3. Cellular Models
3.1. Colon Cancer
3.2. Breast and Mammary Cancer
3.3. Prostate Cancer
3.4. Liver Cancer
3.5. Skin Cancer
4. Clinical Studies
4.1. Colon Cancer
4.2. Breast Cancer
5. Cellular Mechanisms by Which CLA May Inhibit Cancer
5.1. Anti-Cancer Initiation
5.2. Anti-Cancer Promotion
5.3. Anti-Proliferative
6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ander, B.P.; Dupasquier, C.M.; Prociuk, M.A.; Pierce, G.N. Polyunsaturated fatty acids and their effects on cardiovascular disease. Exp. Clin. Cardiol. 2003, 8, 164–172. [Google Scholar]
- Koba, K.; Yanagita, T. Health benefits of conjugated linoleic acid (CLA). Obes. Res. Clin. Pract. 2014, 8, e525–e532. [Google Scholar] [CrossRef] [PubMed]
- Albers, R.; van der Wielen, R.P.; Brink, E.J.; Hendriks, H.F.; Dorovska-Taran, V.N.; Mohede, I.C. Effects of cis-9, trans-11 and trans-10, cis-12 conjugated linoleic acid (CLA) isomers on immune function in healthy men. Eur. J. Clin. Nutr. 2003, 57, 595–603. [Google Scholar] [CrossRef]
- Salsinha, A.S.; Pimentel, L.L.; Fontes, A.L.; Gomes, A.M.; Rodríguez-Alcalá, L.M. Microbial Production of Conjugated Linoleic Acid and Conjugated Linolenic Acid Relies on a Multienzymatic System. Microbiol. Mol. Biol. Rev. 2018, 82, e00019-18. [Google Scholar] [CrossRef]
- Kepler, C.R.; Tucker, W.P.; Tove, S.B. Biohydrogenation of Unsaturated Fatty Acids: V. Stereospecificity of proton addition and mechanism of action of linoleic acid Δ12-cis, Δ11-trans-isomerase from Butyrivibrio fibrisolvens. J. Biol. Chem. 1971, 246, 2765–2771. [Google Scholar] [CrossRef]
- Gorissen, L.; Leroy, F.; De Vuyst, L.; De Smet, S.; Raes, K. Bacterial production of conjugated linoleic and linolenic Acid in foods: A technological challenge. Crit. Rev. Food Sci. Nutr. 2015, 55, 1561–1574. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.O.; Kim, C.S.; Cho, S.K.; Choi, H.J.; Ji, G.E.; Oh, D.-K. Bioconversion of linoleic acid into conjugated linoleic acid during fermentation and by washed cells of Lactobacillus reuteri. Biotechnol. Lett. 2003, 25, 935–938. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.-Y.; Yen, C.-L. Inhibition of Lipid Peroxidation by Lactobacillus acidophilus and Bifidobacterium longum. J. Agric. Food Chem. 1999, 47, 3661–3664. [Google Scholar] [CrossRef]
- Dahiya, D.K.; Puniya, A.K. Isolation, molecular characterization and screening of indigenous lactobacilli for their abilities to produce bioactive conjugated linoleic acid (CLA). J. Food Sci. Technol. 2017, 54, 792–801. [Google Scholar] [CrossRef]
- Chung, S.H.; Kim, I.H.; Park, H.G.; Kang, H.S.; Yoon, C.S.; Jeong, H.Y.; Choi, N.J.; Kwon, E.G.; Kim, Y.J. Synthesis of Conjugated Linoleic Acid by Human-Derived Bifidobacterium breve LMC 017: Utilization as a Functional Starter Culture for Milk Fermentation. J. Agric. Food Chem. 2008, 56, 3311–3316. [Google Scholar] [CrossRef]
- Dhiman, T.R.; Nam, S.H.; Ure, A.L. Factors affecting conjugated linoleic acid content in milk and meat. Crit. Rev. Food Sci. Nutr. 2005, 45, 463–482. [Google Scholar] [CrossRef]
- Yang, B.; Chen, H.; Stanton, C.; Ross, R.P.; Zhang, H.; Chen, Y.Q.; Chen, W. Review of the roles of conjugated linoleic acid in health and disease. J. Funct. Foods 2015, 15, 314–325. [Google Scholar] [CrossRef]
- Booth, R.G.; Kon, S.A. A study of seasonal variation in butter fat: A seasonal spectroscopic variation in the fatty acid fraction. Biochem. J. 1935, 29, 133–137. [Google Scholar] [CrossRef]
- Pariza, M.W.; Ashoor, S.H.; Chu, F.S. Mutagens in heat-processed meat, bakery and cereal products. Food Cosmet Toxicol. 1979, 17, 429–430. [Google Scholar] [CrossRef]
- Ha, Y.L.; Grimm, N.K.; Pariza, M.W. Anticarcinogens from fried ground beef: Heat-altered derivatives of linoleic acid. Carcinogenesis 1987, 8, 1881–1887. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K. Occurrence of conjugated linoleic acid in ruminant products and its physiological functions. Anim. Sci. J. 2005, 76, 291–303. [Google Scholar] [CrossRef]
- Kelley, N.S.; Hubbard, N.E.; Erickson, K.L. Conjugated linoleic acid isomers and cancer. J. Nutr. 2007, 137, 2599–2607. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Lee, H.J.; Cho, H.Y.; Kim, Y.J. Role of the conjugated linoleic acid in the prevention of cancer. Crit. Rev. Food Sci. Nutr. 2005, 45, 135–144. [Google Scholar] [CrossRef]
- Khanal, R. Potential health benefits of conjugated linoleic acid (CLA): A Review. Anim. Biosci. 2004, 17, 1315–1328. [Google Scholar] [CrossRef]
- Den Hartigh, L.J. Conjugated linoleic acid effects on cancer, obesity, and atherosclerosis: A review of pre-clinical and human trials with current perspectives. Nutrients. 2019, 11, 370. [Google Scholar] [CrossRef]
- Pariza, M.W.; Hargraves, W.A. A beef-derived mutagenesis modulator inhibits initiation of mouse epidermal tumors by 7,12-dimethylbenz[a]anthracene. Carcinogenesis 1985, 6, 591–593. [Google Scholar] [CrossRef] [PubMed]
- Azain, M.J. Conjugated linoleic acid and its effects on animal products and health in single-stomached animals. Proc. Nutr Soc. 2003, 62, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; Ryu, J.H.; Ha, Y.L.; Park, J.H.Y. Dietary conjugated linoleic acid (CLA) induces apoptosis of colonic mucosa in 1,2-dimethylhydrazine-treated rats: A possible mechanism of the anticarcinogenic effect by CLA. Br. J. Nutr. 2001, 86, 549–555. [Google Scholar] [CrossRef]
- Shiraishi, R.; Iwakiri, R.; Fujise, T.; Kuroki, T.; Kakimoto, T.; Takashima, T.; Sakata, Y.; Tsunada, S.; Nakashima, Y.; Yanagita, T.; et al. Conjugated linoleic acid suppresses colon carcinogenesis in azoxymethane-pretreated rats with long-term feeding of diet containing beef tallow. J. Gastroenterol. 2010, 45, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Mandir, N.; Goodlad, R.A. Conjugated linoleic acids differentially alter polyp number and diameter in the Apcmin/+ mouse model of intestinal cancer. Cell Prolif. 2008, 41, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Petrik, M.B.; McEntee, M.F.; Johnson, B.T.; Obukowicz, M.G.; Whelan, J. Highly unsaturated (n-3) fatty acids, but not alpha-linolenic, conjugated linoleic or gamma-linolenic acids, reduce tumorigenesis in Apc(Min/+) mice. J. Nutr. 2000, 130, 2434–2443. [Google Scholar] [CrossRef]
- Soel, S.M.; Choi, O.S.; Bang, M.H.; Yoon Park, J.H.; Kim, W.K. Influence of conjugated linoleic acid isomers on the metastasis of colon cancer cells in vitro and in vivo. J. Nutr. Biochem. 2007, 18, 650–657. [Google Scholar] [CrossRef]
- Nichenametla, S.; South, E.; Exon, J. Interaction of conjugated linoleic acid, sphingomyelin, and butyrate on formation of colonic aberrant crypt foci and immune functions in rats. J. Toxicol. Env. Health A 2004, 67, 469–481. [Google Scholar] [CrossRef]
- Kim, K.-H.; Park, H.-S. Dietary supplementation of conjugated linoleic acid reduces colon tumor incidence in DMH-treated rats by increasing apoptosis with modulation of biomarkers. Nutrition 2003, 19, 772–777. [Google Scholar] [CrossRef]
- Sasaki, T.; Fujii, K.; Yoshida, K.; Shimura, H.; Sasahira, T.; Ohmori, H.; Kuniyasu, H. Peritoneal metastasis inhibition by linoleic acid with activation of PPARgamma in human gastrointestinal cancer cells. Virchows Arch. 2006, 448, 422–427. [Google Scholar] [CrossRef]
- Ip, C.; Scimeca, J.A. Conjugated linoleic acid and linoleic acid are distinctive modulators of mammary carcinogenesis. Nutr Cancer 1997, 27, 131–135. [Google Scholar] [CrossRef]
- Białek, M.; Białek, A.; Czauderna, M. Conjugated Linoleic Acid Isomers Affect Profile of Lipid Compounds and Intensity of Their Oxidation in Heart of Rats with Chemically-Induced Mammary Tumors-Preliminary Study. Nutrients 2019, 11, 2032. [Google Scholar] [CrossRef]
- Visonneau, S.; Cesano, A.; Tepper, S.A.; Scimeca, J.A.; Santoli, D.; Kritchevsky, D. Conjugated linoleic acid suppresses the growth of human breast adenocarcinoma cells in SCID mice. Anticancer Res. 1997, 17, 969–973. [Google Scholar]
- Hubbard, N.E.; Lim, D.; Summers, L.; Erickson, K.L. Reduction of murine mammary tumor metastasis by conjugated linoleic acid. Cancer Lett. 2000, 150, 93–100. [Google Scholar] [CrossRef]
- Tao, X.M.; Wang, J.C.; Wang, J.B.; Feng, Q.; Gao, S.Y.; Zhang, L.R.; Zhang, Q. Enhanced anticancer activity of gemcitabine coupling with conjugated linoleic acid against human breast cancer in vitro and in vivo. Eur. J. Pharm Biopharm. 2012, 82, 401–409. [Google Scholar] [CrossRef]
- Lavillonnière, F.; Chajès, V.; Martin, J.C.; Sébédio, J.L.; Lhuillery, C.; Bougnoux, P. Dietary purified cis-9,trans-11 conjugated linoleic acid isomer has anticarcinogenic properties in chemically induced mammary tumors in rats. Nutr. Cancer 2003, 45, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Liu, P.; Yang, X.; Li, H.; Li, H.; Guo, Y.; Meng, X.; Liu, X. The dietary c9, t11-conjugated linoleic acid enriched from butter reduces breast cancer progression in vivo. J. Food Biochem. 2020, 44, e13163. [Google Scholar] [CrossRef]
- Ip, C.; Banni, S.; Angioni, E.; Carta, G.; McGinley, J.; Thompson, H.J.; Barbano, D.; Bauman, D. Conjugated Linoleic Acid-Enriched Butter Fat Alters Mammary Gland Morphogenesis and Reduces Cancer Risk in Rats. J. Nutr. 1999, 129, 2135–2142. [Google Scholar] [CrossRef]
- Cesano, A.; Visonneau, S.; Scimeca, J.A.; Kritchevsky, D.; Santoli, D. Opposite effects of linoleic acid and conjugated linoleic acid on human prostatic cancer in SCID mice. Anticancer Res. 1998, 18, 1429–1434. [Google Scholar] [PubMed]
- Yang, H.; Holcroft, J.; Glickman, B.W.; de Boer, J.G. Conjugated linoleic acid inhibits mutagenesis by 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine in the prostate of Big Blue rats. Mutagenesis 2003, 18, 195–200. [Google Scholar] [CrossRef][Green Version]
- Jung, M.Y.; Choi, N.J.; Oh, C.H.; Shin, H.K.; Yoon, S.H. Selectively Hydrogenated Soybean Oil Exerts Strong Anti-Prostate Cancer Activities. Lipids 2011, 46, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Cohen, L.A.; Zhao, Z.; Pittman, B.; Scimeca, J. Effect of soy protein isolate and conjugated linoleic acid on the growth of Dunning R-3327-AT-1 rat prostate tumors. Prostate 2003, 54, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Kilian, M.; Mautsch, I.; Gregor, J.I.; Stahlknecht, P.; Jacobi, C.A.; Schimke, I.; Guski, H.; Wenger, F.A. Influence of conjugated vs. conventional linoleic acid on liver metastasis and hepatic lipidperoxidation in BOP-induced pancreatic cancer in Syrian hamster. Prostaglandins Leukot Essent Fat. Acids 2002, 67, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Kilian, M.; Mautsch, I.; Gregor, J.I.; Heinichen, D.; Jacobi, C.A.; Schimke, I.; Guski, H.; Müller, J.M.; Wenger, F.A. Influence of conjugated and conventional linoleic acid on tumor growth and lipid peroxidation in pancreatic adenocarcinoma in hamster. Prostaglandins Leukot Essent Fat. Acids 2003, 69, 67–72. [Google Scholar] [CrossRef]
- Kohno, H.; Suzuki, R.; Yasui, Y.; Hosokawa, M.; Miyashita, K.; Tanaka, T. Pomegranate seed oil rich in conjugated linolenic acid suppresses chemically induced colon carcinogenesis in rats. Cancer Sci. 2004, 95, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, M.; Ikeda, A.; Hirao, A.; Tanaka, Y.; Miyazaki, Y.; Rikimaru, T.; Shimada, M.; Sugimachi, K.; Tachibana, H.; Yamada, K. Effect of dietary conjugated linoleic acid on the in vivo growth of rat hepatoma dRLh-84. Nutr. Cancer 2001, 40, 140–148. [Google Scholar] [CrossRef]
- Cho, H.J.; Kim, W.K.; Kim, E.J.; Jung, K.C.; Park, S.; Lee, H.S.; Tyner, A.L.; Park, J.H.Y. Conjugated linoleic acid inhibits cell proliferation and ErbB3 signaling in HT-29 human colon cell line. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 284, G996–G1005. [Google Scholar] [CrossRef]
- Cho, H.J.; Kim, W.K.; Jung, J.I.; Kim, E.J.; Lim, S.S.; Kwon, D.Y.; Park, J.H. Trans-10,cis-12, not cis-9,trans-11, conjugated linoleic acid decreases ErbB3 expression in HT-29 human colon cancer cells. World J. Gastroenterol. 2005, 11, 5142–5150. [Google Scholar]
- Palombo, J.D.; Ganguly, A.; Bistrian, B.R.; Menard, M.P. The antiproliferative effects of biologically active isomers of conjugated linoleic acid on human colorectal and prostatic cancer cells. Cancer Lett. 2002, 177, 163–172. [Google Scholar] [CrossRef]
- Kim, E.J.; Holthuizen, P.E.; Park, H.S.; Ha, Y.L.; Jung, K.C.; Park, J.H.Y. Trans-10,cis-12-conjugated linoleic acid inhibits Caco-2 colon cancer cell growth. Am. J. Physiol. Gastrointest. Liver Physiol. 2002, 283, G357–G367. [Google Scholar] [CrossRef][Green Version]
- Lampen, A.; Leifheit, M.; Voss, J.; Nau, H. Molecular and cellular effects of cis-9, trans-11-conjugated linoleic acid in enterocytes: Effects on proliferation, differentiation, and gene expression. Biochim. Biophys. Acta (Bba) Mol. Cell Biol. Lipids 2005, 1735, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.Y.; Tyner, A.L.; Park, J.-B.; Lee, J.-Y.; Choi, Y.H.; Park, J.H.Y. Inhibition of colon cancer cell proliferation by the dietary compound conjugated linoleic acid is mediated by the CDK inhibitor p21CIP1/WAF1. J. Cell. Physiol. 2005, 205, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.Q.; Yang, Y.M.; Wang, Q.; Gao, Y.H.; Liu, J.R.; Zhang, J.S.; Wang, X.L.; Liu, R.H. Effects of c9, t11-conjugated linoleic acid on adhesion of human gastric carcinoma cell line SGC-7901. World J. Gastroenterol. 2004, 10, 1392–1396. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.S.; Huang, Y.W.; Liu, S.; Chang, H.L.; Ye, W.; Shu, S.; Sugimoto, Y.; Funk, J.A.; Smeaks, D.D.; Hill, L.N.; et al. Conjugated linoleic acid (CLA) modulates prostaglandin E2 (PGE2) signaling in canine mammary cells. Anticancer Res. 2006, 26, 889–898. [Google Scholar]
- Sorenmo, K. Canine mammary gland tumors. Vet. Clin. N. Am. Small Anim Pr. 2003, 33, 573–596. [Google Scholar] [CrossRef]
- O’Shea, M.; Devery, R.; Lawless, F.; Murphy, J.; Stanton, C. Milk fat conjugated linoleic acid (CLA) inhibits growth of human mammary MCF-7 cancer cells. Anticancer Res. 2000, 20, 3591–3601. [Google Scholar] [PubMed]
- El Roz, A.; Bard, J.M.; Huvelin, J.M.; Nazih, H. The anti-proliferative and pro-apoptotic effects of the trans9,trans11 conjugated linoleic acid isomer on MCF-7 breast cancer cells are associated with LXR activation. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 265–272. [Google Scholar] [CrossRef]
- Miglietta, A.; Bozzo, F.; Bocca, C.; Gabriel, L.; Trombetta, A.; Belotti, S.; Canuto, R.A. Conjugated linoleic acid induces apoptosis in MDA-MB-231 breast cancer cells through ERK/MAPK signalling and mitochondrial pathway. Cancer Lett. 2006, 234, 149–157. [Google Scholar] [CrossRef]
- Durgam, V.R.; Fernandes, G. The growth inhibitory effect of conjugated linoleic acid on MCF-7 cells is related to estrogen response system. Cancer Lett. 1997, 116, 121–130. [Google Scholar] [CrossRef]
- Park, Y.; Allen, K.G.; Shultz, T.D. Modulation of MCF-7 breast cancer cell signal transduction by linoleic acid and conjugated linoleic acid in culture. Anticancer Res. 2000, 20, 669–676. [Google Scholar]
- Rakib, M.A.; Kim, Y.S.; Jang, W.J.; Choi, B.D.; Kim, J.O.; Kong, I.K.; Ha, Y.L. Attenuation of 12-O-Tetradecanoylphorbol-13-acetate (TPA)-Induced Gap Junctional Intercellular Communication (GJIC) Inhibition in MCF-10A Cells by c9, t11-Conjugated Linoleic Acid. J. Agric. Food Chem. 2010, 58, 12022–12030. [Google Scholar] [CrossRef]
- Rakib, M.A.; Kim, Y.S.; Jang, W.J.; Jang, J.S.; Kang, S.J.; Ha, Y.L. Preventive Effect of t,t-Conjugated Linoleic Acid on 12-O-Tetradecanoylphorbol-13-acetate-Induced Inhibition of Gap Junctional Intercellular Communication in Human Mammary Epithelial MCF-10A Cells. J. Agric. Food Chem. 2011, 59, 4164–4170. [Google Scholar] [CrossRef]
- Kim, E.J.; Shin, H.-K.; Cho, J.S.; Lee, S.K.; Won, M.H.; Kim, J.W.; Park, J.H.Y. trans-10,cis-12 Conjugated Linoleic Acid Inhibits the G1-S Cell Cycle Progression in DU145 Human Prostate Carcinoma Cells. J. Med. Food 2006, 9, 293–299. [Google Scholar] [CrossRef]
- Ochoa, J.J.; Farquharson, A.J.; Grant, I.; Moffat, L.E.; Heys, S.D.; Wahle, K.W.J. Conjugated linoleic acids (CLAs) decrease prostate cancer cell proliferation: Different molecular mechanisms for cis -9, trans -11 and trans -10, cis -12 isomers. Carcinogenesis 2004, 25, 1185–1191. [Google Scholar] [CrossRef]
- Yamasaki, M.; Chujo, H.; Koga, Y.; Oishi, A.; Rikimaru, T.; Shimada, M.; Sugimachi, K.; Tachibana, H.; Yamada, K. Potent cytotoxic effect of the trans10, cis12 isomer of conjugated linoleic acid on rat hepatoma dRLh-84 cells. Cancer Lett. 2002, 188, 171–180. [Google Scholar] [CrossRef]
- Yamasaki, M.; Miyamoto, Y.; Chujo, H.; Nishiyama, K.; Tachibana, H.; Yamada, K. Trans10, cis12-conjugated linoleic acid induces mitochondria-related apoptosis and lysosomal destabilization in rat hepatoma cells. Biochim. Biophys. Acta (Bba) Mol. Cell Biol. Lipids 2005, 1735, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Muzio, G.; Maggiora, M.; Oraldi, M.; Trombetta, A.; Canuto, R.A. PPARα and PP2A are involved in the proapoptotic effect of conjugated linoleic acid on human hepatoma cell line SK-HEP-1. Int. J. Cancer 2007, 121, 2395–2401. [Google Scholar] [CrossRef]
- Yamasaki, M.; Nagatomo, T.; Matsuyama, T.; Ikeho, Y.; Kato, E.; Nishiyama, K.; Sakakibara, Y.; Suiko, M.; Nishiyama, K. Conjugated linoleic acids inhibit hypoxia inducible factor-1α stabilization under hypoxic condition in human hepatocellular carcinoma cells. J. Oleo Sci. 2012, 61, 491–496. [Google Scholar] [CrossRef]
- Liu, K.L.; Belury, M.A. Conjugated linoleic acid modulation of phorbol ester-induced events in murine keratinocytes. Lipids 1997, 32, 725–730. [Google Scholar] [CrossRef] [PubMed]
- McCrorie, T.A.; Keaveney, E.M.; Wallace, J.M.W.; Binns, N.; Livingstone, M.B.E. Human health effects of conjugated linoleic acid from milk and supplements. Nutr. Res. Rev. 2011, 24, 206–227. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, A.; Banu, J.; Rahman, M.; Causey, J.; Fernandes, G. Biological effects of conjugated linoleic acids in health and disease. J. Nutr. Biochem. 2006, 17, 789–810. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.-S. Biological effects of conjugated linoleic acid on obesity-related cancers. Chem. Biol. Interact. 2014, 224, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Mohammadzadeh, M.; Faramarzi, E.; Mahdavi, R.; Nasirimotlagh, B.; Asghari, J.M. Effect of conjugated linoleic acid supplementation on inflammatory factors and matrix metalloproteinase enzymes in rectal cancer patients undergoing chemoradiotherapy. Integr Cancer Ther. 2013, 12, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Bergkvist, L.; Wolk, A. High-fat dairy food and conjugated linoleic acid intakes in relation to colorectal cancer incidence in the Swedish Mammography Cohort. Am J Clin Nutr. 2005, 82, 894–900. [Google Scholar] [CrossRef]
- Arab, A.; Akbarian, S.G.; Ghiyasvand, R.; Miraghajani, M. The effects of conjugated linoleic acids on breast cancer: A systematic review. Adv. Biomed. Res. 2016, 5, 115. [Google Scholar] [PubMed]
- Aro, A.; Männistö, S.; Salminen, I.; Ovaskainen, M.L.; Kataja, V.; Uusitupa, M. Inverse association between dietary and serum conjugated linoleic acid and risk of breast cancer in postmenopausal women. Nutr. Cancer 2000, 38, 151–157. [Google Scholar] [CrossRef]
- Knekt, P.; Järvinen, R.; Seppänen, R.; Pukkala, E.; Aromaa, A. Intake of dairy products and the risk of breast cancer. Br. J. Cancer 1996, 73, 687–691. [Google Scholar] [CrossRef]
- Chajes, V.; Lavillonniere, F.; Maillard, V.; Giraudeau, B.; Jourdan, M.L.; Sebedio, J.L.; Bougnoux, P. Conjugated linoleic acid content in breast adipose tissue of breast cancer patients and the risk of metastasis. Nutr. Cancer 2003, 45, 17–23. [Google Scholar] [CrossRef]
- Voorrips, L.E.; Brants, H.A.M.; Kardinaal, A.F.M.; Hiddink, G.J.; van den Brandt, P.A.; Goldbohm, R.A. Intake of conjugated linoleic acid, fat, and other fatty acids in relation to postmenopausal breast cancer: The Netherlands Cohort Study on Diet and Cancer. Am. J. Clin. Nutr. 2002, 76, 873–882. [Google Scholar] [CrossRef]
- McCann, S.E.; Ip, C.; Ip, M.M.; McGuire, M.K.; Muti, P.; Edge, S.B.; Trevisan, M.; Freudenheim, J.L. Dietary intake of conjugated linoleic acids and risk of premenopausal and postmenopausal breast cancer, Western New York Exposures and Breast Cancer Study (WEB Study). Cancer Epidemiol. Biomark. Prev. 2004, 13, 1480–1484. [Google Scholar]
- Oh, Y.S.; Lee, H.S.; Cho, H.J.; Lee, S.G.; Jung, K.C.; Park, J.H. Conjugated linoleic acid inhibits DNA synthesis and induces apoptosis in TSU-Pr1 human bladder cancer cells. Anticancer Res. 2003, 23, 4765–4772. [Google Scholar]
- Ha, Y.L.; Storkson, J.; Pariza, M.W. Inhibition of benzo(a)pyrene-induced mouse forestomach neoplasia by conjugated dienoic derivatives of linoleic acid. Cancer Res. 1990, 50, 1097–1101. [Google Scholar]
- Yu, L. Free Radical Scavenging Properties of Conjugated Linoleic Acids. J. Agric. Food Chem. 2001, 49, 3452–3456. [Google Scholar] [CrossRef] [PubMed]
- Shultz, T.D.; Chew, B.P.; Seaman, W.R.; Luedecke, L.O. Inhibitory effect of conjugated dienoic derivatives of linoleic acid and beta-carotene on the in vitro growth of human cancer cells. Cancer Lett. 1992, 63, 125–133. [Google Scholar] [CrossRef]
- Lalithadevi, B.; Muthiah, N.S.; Murty, K.S.N. Antioxidant activity of conjugated linoleic acid. Asian J. Pharm. Clin. Res. 2018, 11, 169. [Google Scholar]
- Belury, M.A.; Kempa-Steczko, A. Conjugated linoleic acid modulates hepatic lipid composition in mice. Lipids 1997, 32, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Urquhart, P.; Parkin, S.M.; Rogers, J.S.; Bosley, J.A.; Nicolaou, A. The effect of conjugated linoleic acid on arachidonic acid metabolism and eicosanoid production in human saphenous vein endothelial cells. Biochim. Biophys Acta 2002, 1580, 150–160. [Google Scholar] [CrossRef]
- Kemp, M.Q.; Jeffy, B.D.; Romagnolo, D.F. Conjugated linoleic acid inhibits cell proliferation through a p53-dependent mechanism: Effects on the expression of G1-restriction points in breast and colon cancer cells. J. Nutr. 2003, 133, 3670–3677. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.J.; Kim, E.J.; Lim, S.S.; Kim, M.K.; Sung, M.K.; Kim, J.S.; Park, J.H. Trans-10,cis-12, not cis-9,trans-11, conjugated linoleic acid inhibits G1-S progression in HT-29 human colon cancer cells. J. Nutr. 2006, 136, 893–898. [Google Scholar] [CrossRef]
- Ip, C.; Ip, M.M.; Loftus, T.; Shoemaker, S.; Shea-Eaton, W. Induction of apoptosis by conjugated linoleic acid in cultured mammary tumor cells and premalignant lesions of the rat mammary gland. Cancer Epidemiol. Biomark. Prev. 2000, 9, 689–696. [Google Scholar]
- Wang, L.S.; Huang, Y.W.; Liu, S.; Yan, P.; Lin, Y.C. Conjugated linoleic acid induces apoptosis through estrogen receptor alpha in human breast tissue. BMC Cancer 2008, 8, 208. [Google Scholar] [CrossRef] [PubMed]
- Bostwick, D.G.; Meiers, I. Prostate. In Modern Surgical Pathology, 2nd ed.; Weidner, N., Cote, R., Suster, S., Weiss, L., Eds.; Saunders: Collingwood, ON, Canada, 2009; Volume 2, pp. 1121–1180. [Google Scholar]
- Borner, C. The Bcl-2 protein family: Sensors and checkpoints for life-or-death decisions. Mol. Immunol. 2003, 39, 615–647. [Google Scholar] [CrossRef]
- Ou, L.; Ip, C.; Lisafeld, B.; Ip, M.M. Conjugated linoleic acid induces apoptosis of murine mammary tumor cells via Bcl-2 loss. Biochem. Biophys. Res. Commun. 2007, 356, 1044–1049. [Google Scholar] [CrossRef] [PubMed]
- Cichosz, G.; Czeczot, H.; Bielecka, M. The anticarcinogenic potential of milk fat. Ann. Agric. Env. Med. 2020, 27, 512–518. [Google Scholar] [CrossRef]
- Miller, A.; McGrath, E.; Stanton, C.; Devery, R. Vaccenic acid (t11-18:1) is converted to c9, t11-CLA in MCF-7 and SW480 cancer cells. Lipids 2003, 38, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Ip, C.; Dong, Y.; Thompson, H.J.; Bauman, D.E.; Ip, M.M. Control of rat mammary epithelium proliferation by conjugated linoleic acid. Nutr. Cancer 2001, 39, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.W.; Chew, B.P.; Wong, T.S.; Hosick, H.L.; Boylston, T.D.; Shultz, T.D. Effects of dietary conjugated linoleic acid on lymphocyte function and growth of mammary tumors in mice. Anticancer Res. 1997, 17, 987–993. [Google Scholar] [PubMed]

| CancerType | Inducer | Animal | CLA Source | Effects | Reference |
|---|---|---|---|---|---|
| Colon | DMD 1 | SD rats | CLA mixture | Incresed apoptotic index, lower number of tumors. | Park et al. [23] |
| Colon | AOM 2 | SD rats | CLA mixture | Incresed apoptosis, and activity of Caspase-3. | Shiraishi et al. [24] |
| Colon | Crypt fission | Apc(min/+) mice | c9, t11-CLA t10, c12-CLA | Decrease polyp size. Increase polyp size. | Mandir et al. [25] |
| Colon | Unhealthy diet | Apc(min/+) mice | CLA mixture | No effect. | Petrik et al. [26] |
| Colon and pulmonary | NMU 3 | BALB/c mice | c9, t11-CLA t10, c12-CLA | Inhibit cancer cell migration, reduced pulmonary nodules. | Soel et al. [27] |
| Colon | AOM | SD rats | c9, t11-CLA | Inhibit aberrant crypts, enhanced killer cell activiy. | Nichenametla et al. [28] |
| Colon | DMH 4 | SD rats | CLA mixture | Reduced tumor incidence, increase apoptosis. | Kim et al. [29] |
| Colon | AOM | BALB/c mice | CLA mixture | Decreased metastatic foci, activation PPRn. | Sasaki et al. [30] |
| Mammary | DMBA 5 | SD rats | CLA mixture | No effect. | Ip et al. [31] |
| Mammary and breast | DMBA | SD rats | CLA mixture | Inhibit PUFA oxidation. | Bialek et al. [32] |
| Breast | DMBA | SCID rats | CLA mixture | Reduced tumor growth. | Visonneau et al. [33] |
| Mammary | DMBA | BALB/c mice | CLA mixture | Prolong tumor latency, decresed tumor burden. | Hubbard et al. [34] |
| Breast | DMBA | BALB/c mice | c9, t11-CLA | Reduced tumor growth in conjugate with gemcitabine. | Tao et al. [35] |
| Mammary | MNU 6 | SD rats | c9, t11-CLA | Decressed tumor mass per animal. | Lavillonneiere et al. [36] |
| Mammry and breast | DMBA | SD rats | c9, t11-CLA | Decreased tumor weight and volume. | Zeng et al. [37] |
| Breast | MNU | SD rats | CLA mixture | Decresed terminal and bud cell density. | Ip et al. [38] |
| Prostate | DMBA | SCID mice | CLA mixture | Inhibit cancer cell grovth. | Cesano et al. [39] |
| Prostate | PhIP 7 | Big blue rats | CLA mixture | Inhibit the formation of PhIP→DNA. | Yang et al. [40] |
| Prostate | AIN-76A diet | Copenhagen rats | CLA mixture | Apoptotic activity and citotoxycity. | Jung et al. [41] |
| Prostate | PhIP | Copenhagen rats | CLA mixture | Not beneficiary alone, effect with isoflavone. | Cohen et al. [42] |
| Pancreas | BOP 8 | Syrian hamster | CLA mixture | No effect. | Kilian et al. [43,44] |
| Liver | AOM | Male F344 rats | c9, t11-CLA | Tumor inhibition, increse expression of PPRn. | Kohno et al. [45] |
| Liver | AOM | Donryu rats | CLA mixture | Induce hepatic lipid peroxidation and accumulation. | Yamasaki et al. [46] |
| CancerType | Cell Lines | CLA Source | Effects | Reference |
|---|---|---|---|---|
| Colon | HT-29 | CLA mixture | Inhibition proliferation by ErbB3 signaling. | Cho et al. [47] |
| Colon | HT-29 | c9, t11-CLA, t10, c12-CLA | Inhibition proliferation by ErbB3 signaling. | Cho et al. [48] |
| Prostate and colon | HT-29, PC-3, MIP-101 | c9, t11-CLA, t10, c12-CLA | Inhibition effect, caspase dependent. | Palombo et al. [49] |
| Colon | Caco-2 | t10, c12-CLA c9, t11-CLA | t10, c12-CLA inhibition proliferation. Decresed insulin-like growth factor II (IGF-II) expression. c9, t11-CLA no effect. | Kim et al. [50] |
| Colon | Caco-2 | c9, t11-CLA | Antiproliferative effect, repressed expresiion c-myc, cyclin D1 in a PPARn | Lampen et al. [51] |
| Colon | HT-29 | CLA mixture | Induced cell cycle arrest at G0/G1 phase | Lim et al. [52] |
| Gastric | SGS-7901 | c9, t11-CLA | Reduced adhesion. Increased the level of ECD and α-catenin. Decrease ICAM-1 and VCAM-1 | Chen et al. [53] |
| Breast | MCF-7, MDA-MB-231- ERα | CLA mixture | Induced cell apoptosis by caspase activity. | Wang et al. [54], Sorenmo et al. [55] |
| Breast | MCF-7 | c9, t11-CLA t10, c12-CLA | c9, t11-CLA more effective suppress growth. t10, c12-CLA less effective. | O’Shea et al. [56] |
| Breast | MCF-7 | c9, t11-CLA, t9, t11-CLA t10, c12-CLA | t9, t11-CLA induced apoptosis, incresed mMRA levels of LXR genes. c9, t11-CLA and t10, c12-CLA less potent. | El Roz et al. [57] |
| Breast | MDA-MB-231 | CLA mixture | ERK reduction and upregulation of pro-apoptotic protein Bak | Miglietta et al. [58] |
| Breast | MCF-7, MDA-MB-231 | CLA mixture | Inhibition cell cycle, c-myc expression. | Durgam et al. [59] |
| Breast | MCF-7 | CLA mixture LA | Inhibition on growth. Stimulation of growth. | Park et al. [60] |
| Breast | MCF-10A | t9, t11-CLA, t10, t12-CLA c9, t11-CLA | The anti-promotional activity of t9, t11 and t10, t12-CLA were less potent than c9, t11-CLA. | Rakib et al. [61,62] |
| Prostate | DU145 | t10, c12-CLA c9, t11-CLA | t10, c12-CLA inhibited the G1-S cell cycle progression. c9, t11-CLA no effect. | Kim et al. [63] |
| Prostate | PC-3 | t10, c12-CLA c9, t11-CLA | t10, c12-CLA apoptosis and cell cycle control. c9, t11-CLA-Regulation of genes for arachidonic acid metabolism. | Ochoa et al. [64] |
| Liver | dRLh-84 | c9, t11-CLA, t10, c12-CLA | Different cytotoxic effects. | Yamasaki et al. [65,66] |
| Liver | SK-HEP-1 | CLA mixture | Increased PPARα and proapoptotic proteins. Activating apoptotic pathways. | Muzio et al. [67] |
| Liver | HepG2 | c9, t11-CLA, t10, c12-CLA | Inhibited cell proliferation and induced apoptotic cell death under hypoxia. | Yamasaki et al. [68] |
| Skin | HEL-30 | Radioactive (1-14C) CLA | Partial inhibition of tumor promotion. | Liu et al. [69] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dachev, M.; Bryndová, J.; Jakubek, M.; Moučka, Z.; Urban, M. The Effects of Conjugated Linoleic Acids on Cancer. Processes 2021, 9, 454. https://doi.org/10.3390/pr9030454
Dachev M, Bryndová J, Jakubek M, Moučka Z, Urban M. The Effects of Conjugated Linoleic Acids on Cancer. Processes. 2021; 9(3):454. https://doi.org/10.3390/pr9030454
Chicago/Turabian StyleDachev, Marko, Jana Bryndová, Milan Jakubek, Zdeněk Moučka, and Marian Urban. 2021. "The Effects of Conjugated Linoleic Acids on Cancer" Processes 9, no. 3: 454. https://doi.org/10.3390/pr9030454
APA StyleDachev, M., Bryndová, J., Jakubek, M., Moučka, Z., & Urban, M. (2021). The Effects of Conjugated Linoleic Acids on Cancer. Processes, 9(3), 454. https://doi.org/10.3390/pr9030454







