Co-Occurrence of Cyanotoxins and Phycotoxins in One of the Largest Southeast Asian Brackish Waterbodies: A Preliminary Study at the Tam Giang—Cau Hai Lagoon (Vietnam)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Handling Methods and Sample Collection
2.2. Laboratory Analytical Methods
2.2.1. Nutrients and Chlorophyll-a and Phycocyanin Analysis
- Ve = volume of buffer extract in mL.
- Vs = volume of water sample in Liters.
- I = path length of cuvette in cm.
- F = Dilution factor (i.e., if the 663 Abs is >0.99 with the 1 cm cell, diluting, re-analyzing, and inserting the dilution factor in the equation).
- E = The volume of acetone used for the extraction (mL).
- V = The volume of water filtered (L).
- L = The cell path length (cm).
2.2.2. Phytoplankton Diversity
2.2.3. Toxins Analysis in Water and Shellfish Samples
3. Results
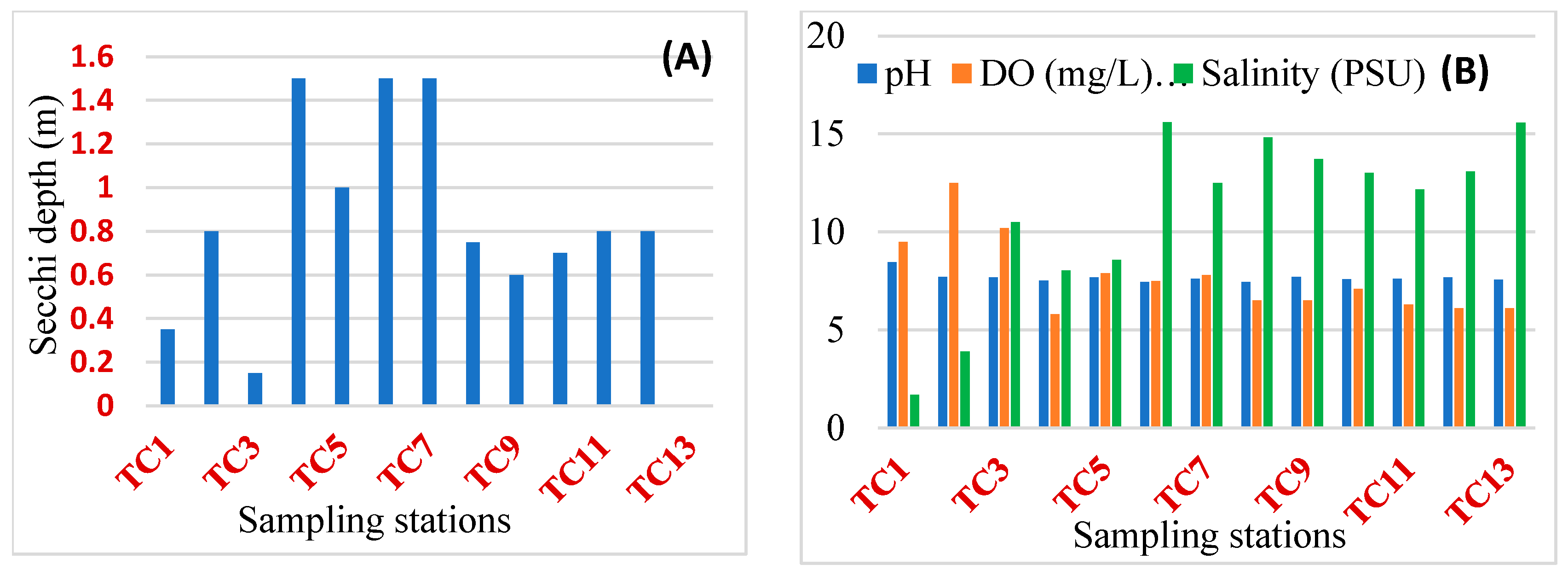
3.1. Physical Chemical Parameters
3.2. Abundance of Phytoplankton Species
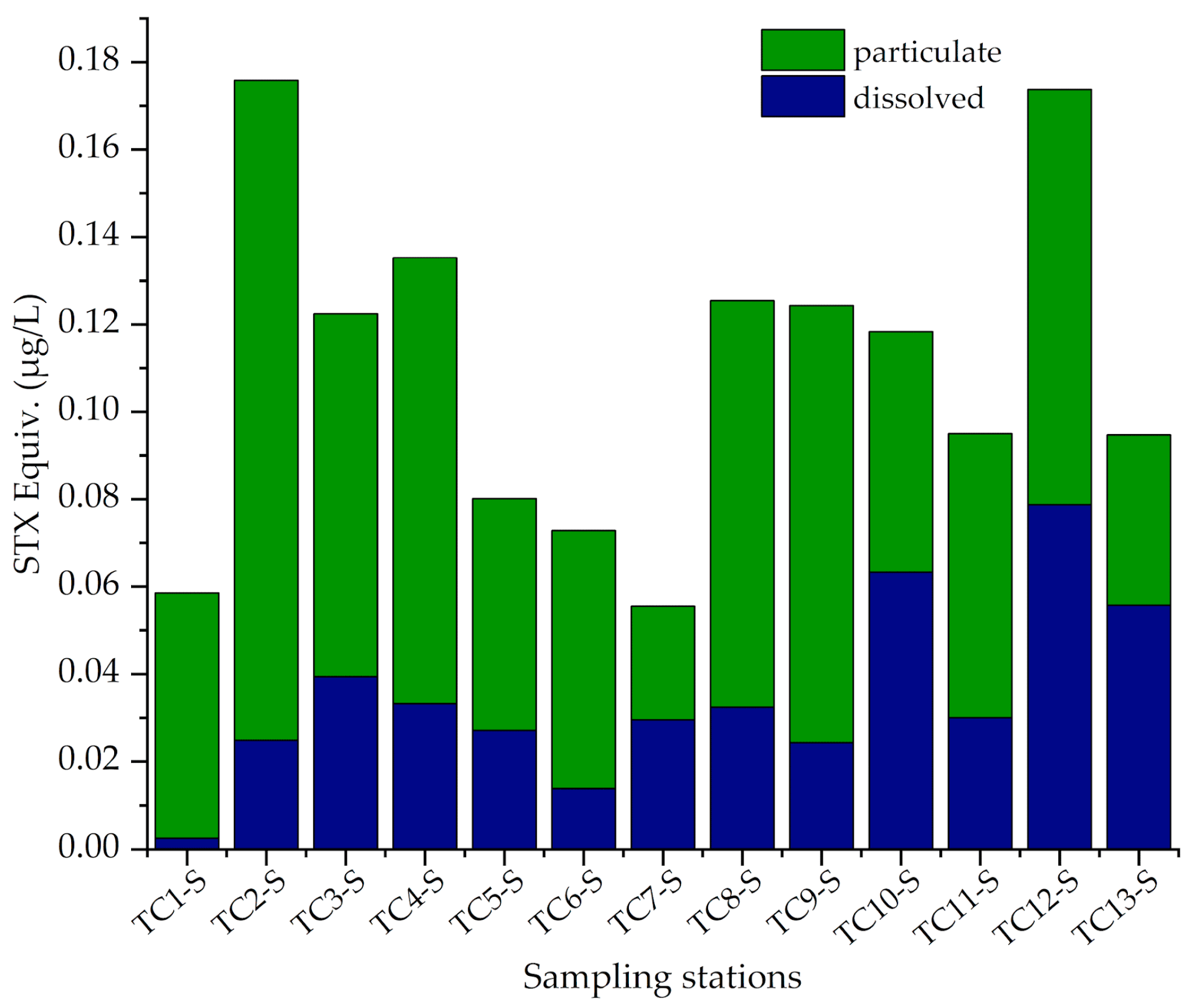
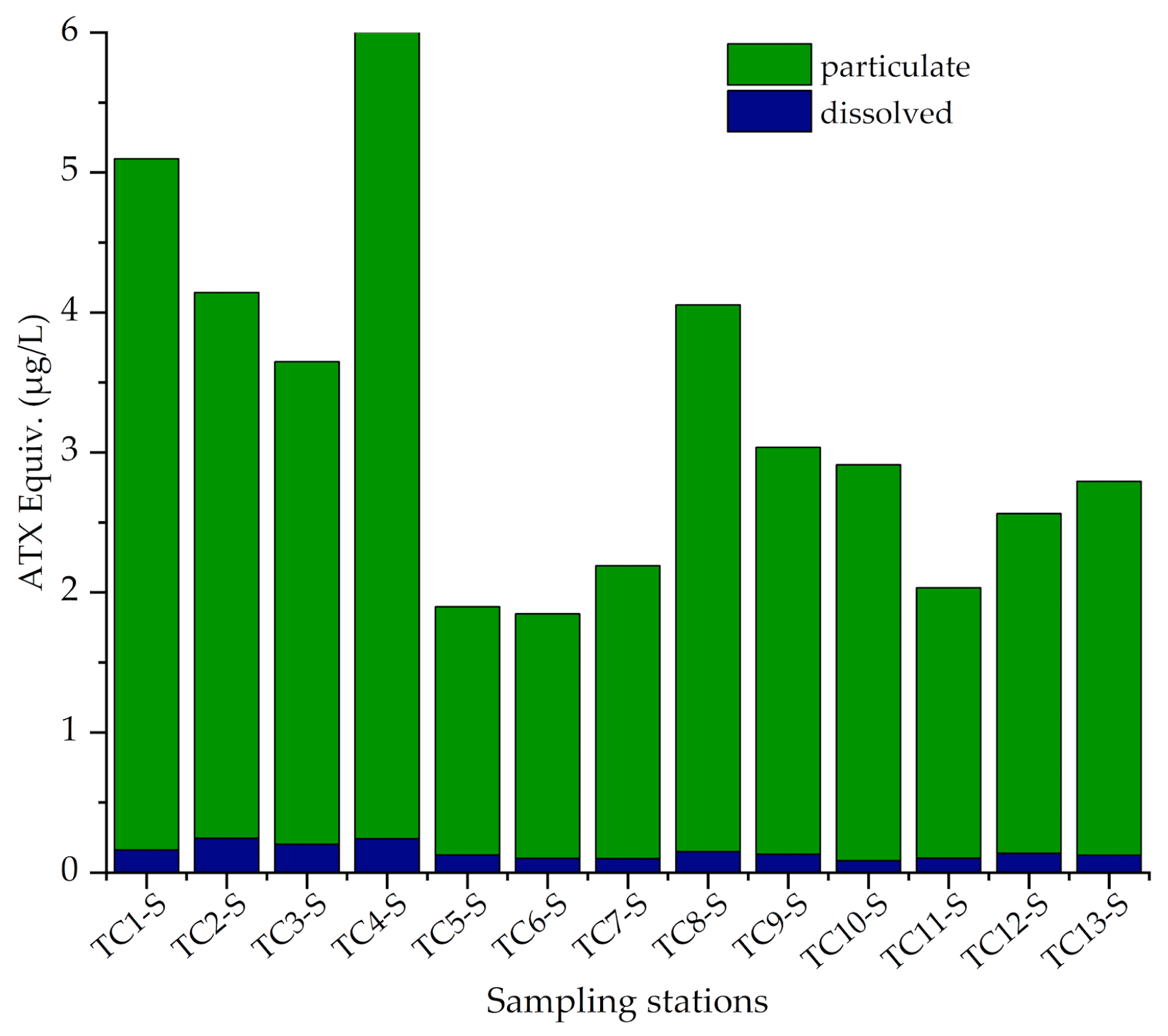
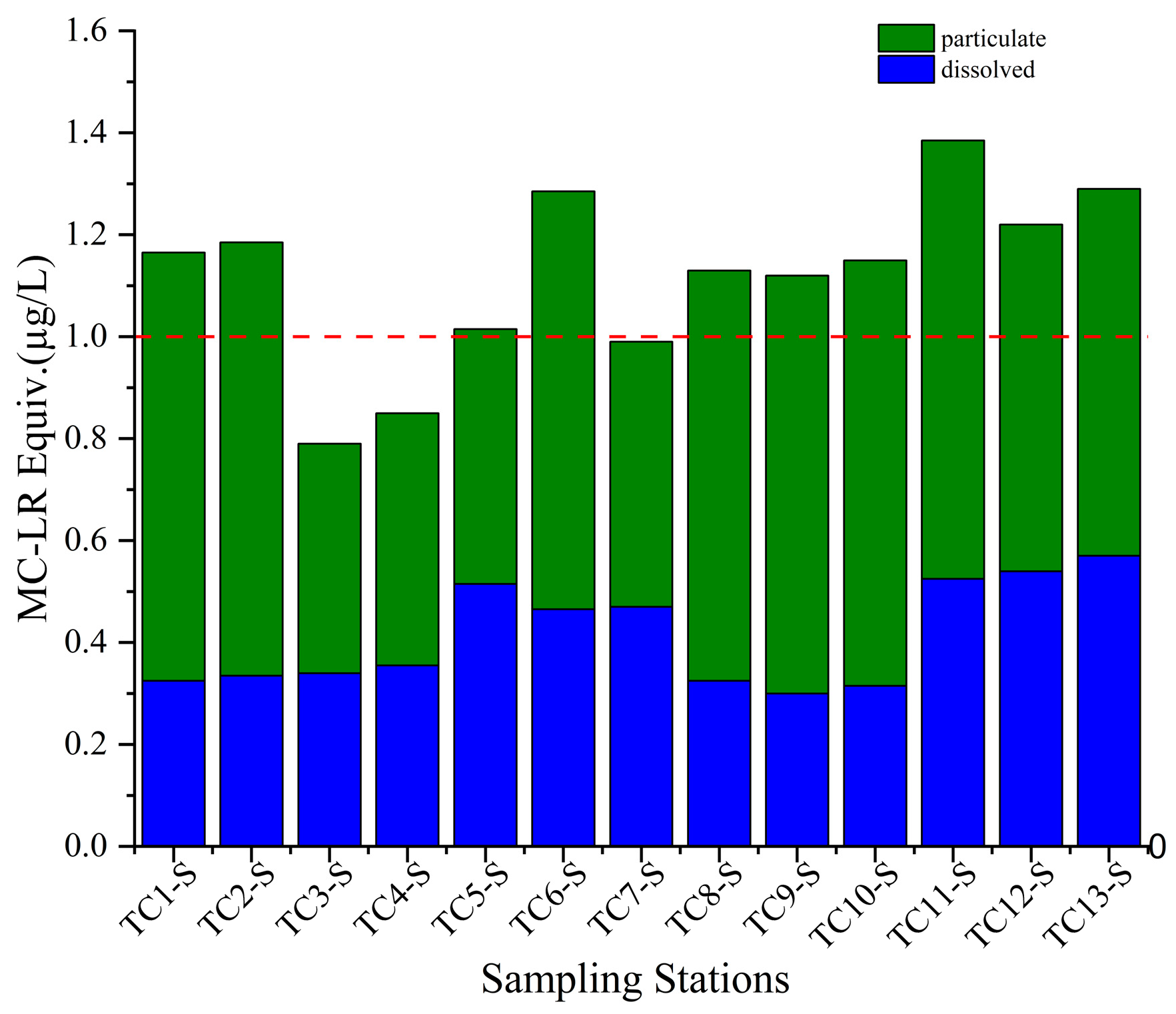
3.3. Toxin Concentrations in Water Samples
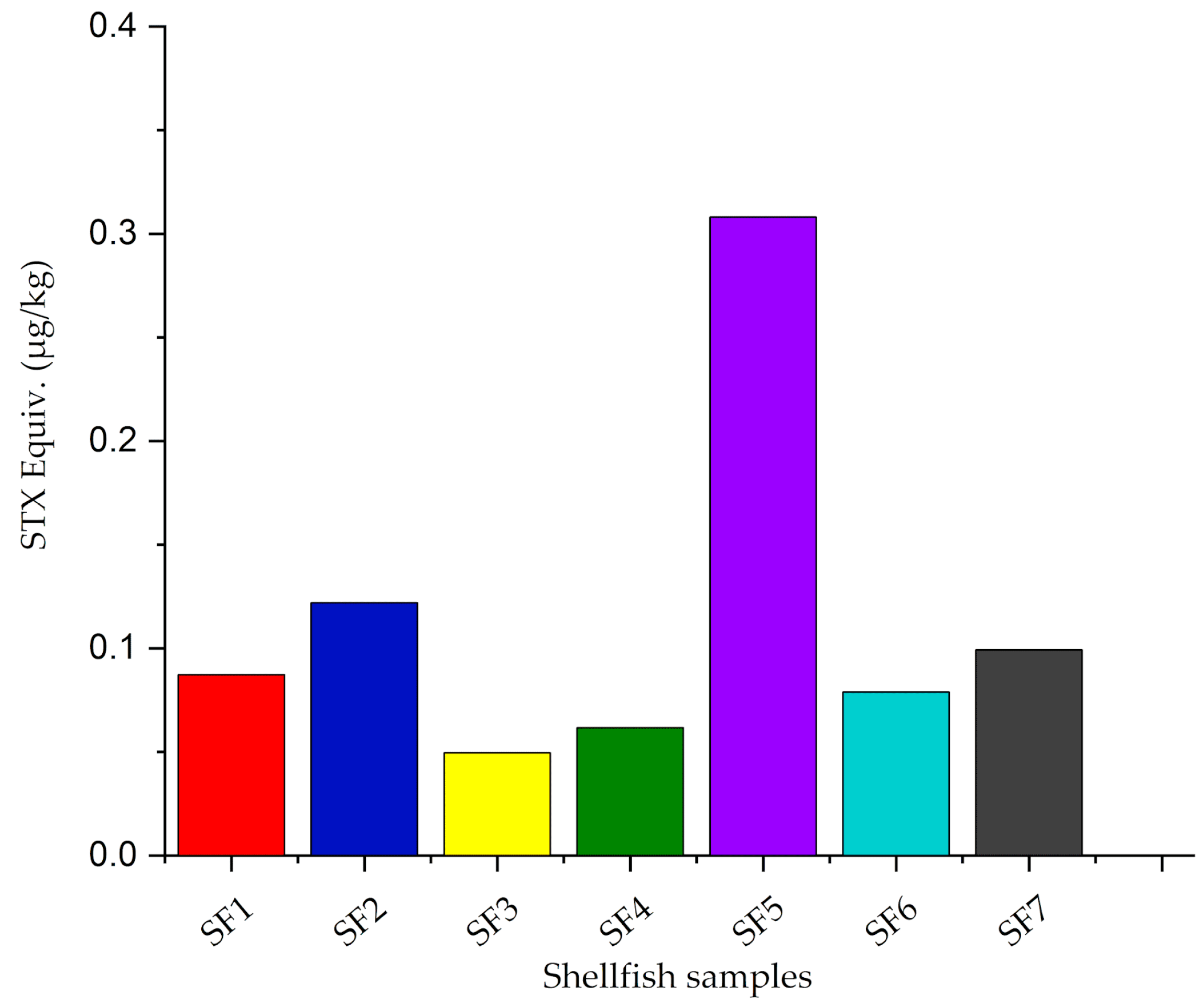
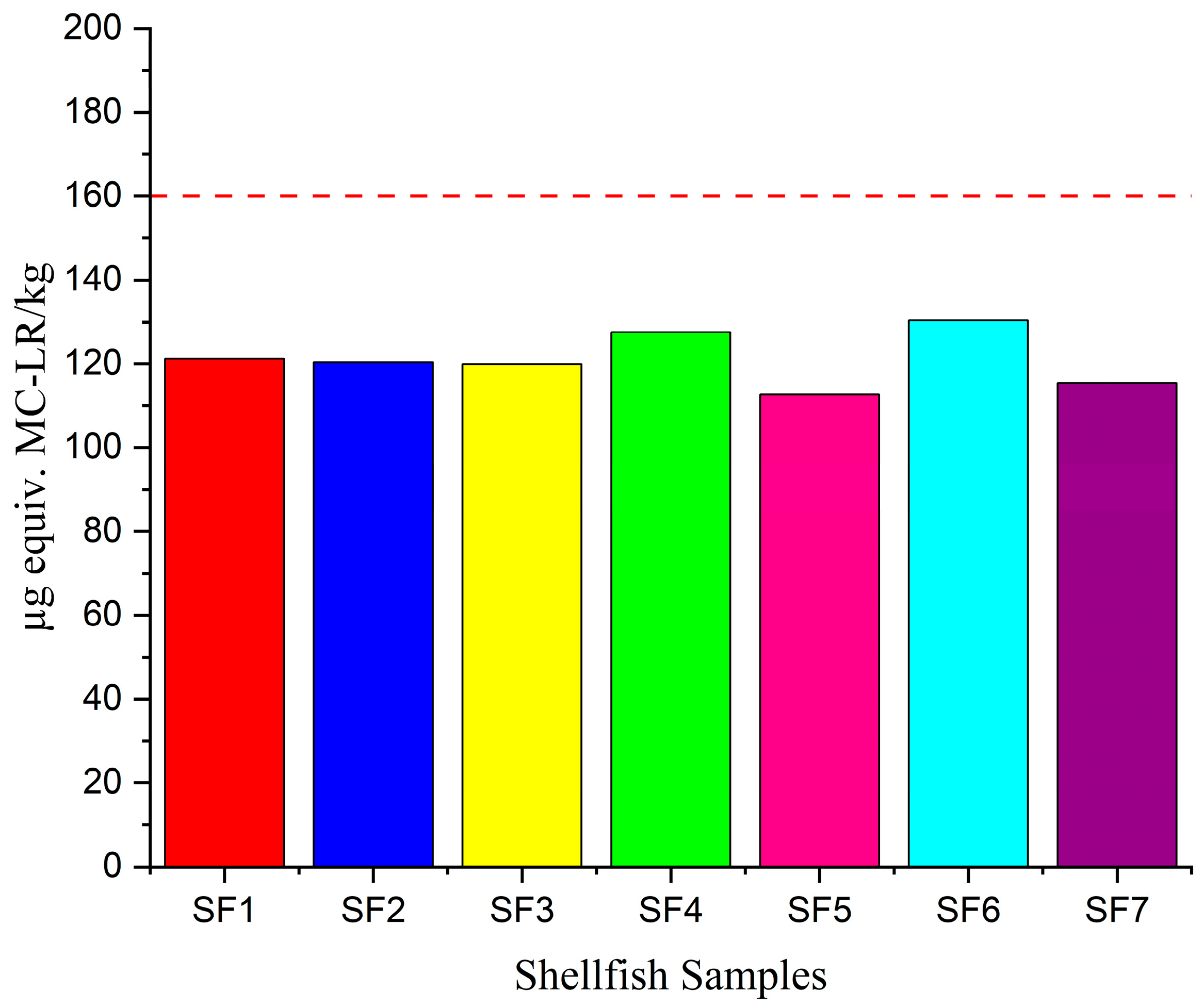
3.4. Toxin Levels in Shellfish Samples
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. Physical Chemical Parameters from Each Sampling Site on Tam Giang lagoon (Vietnam)
| Site | Coordinates | Depth (m) | Secchi Disk (m) | D.O (mg/L) | Temp (°C) | pH | Turbidity (NTU) | Salinity (PSU) | NH4 (mg/L) | NO2 (mg/L) | NO3 (mg/L) | TN (mg/L) | PO4 (mg/L) | TP (mg/L) | Chl-a (mg/L) | PC (mg/L) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TC1 | N 16°33′52.7″ E 107°37′26.6″ | 0.5 | 0.35 | 9.5 | 34.75 | 8.45 | 45 | 0.17 | 0.12 | 0.001 | 0.09 | 0.582 | LOD * | 0.062 | 30.71 | 0.08 |
| TC2 | N 16°35′10.4″ E 107°34′40.0″ | 2 | 0.8 | 12.5 | 33.6 | 7.7 | 34.7 | 3.9 | 0.07 | 0.0040 | 0.14 | 0.819 | LOD | 0.075 | 20.03 | 0.06 |
| TC3 | N 16°36’49.1″ E 107°31’41.9″ | ~2.3 | 0.15 | 10.16 | 33.2 | 7.69 | 2 | 10.5 | 0.08 | 0.003 | 0.80 | 1.220 | LOD | 0.076 | 0.00 | 0.01 |
| TC4 | N 16°37’34.4″ E 107°30’18.6″ | ~1.5 | 1.5 | 5.8 | 34.3 | 7.51 | 50 | 8.03 | 0.07 | 0.0097 | 0.57 | 1.487 | LOD | 0.106 | 42.72 | 0.01 |
| TC5 | N 16°37’13.8″ E 107°30’7.4″ | 4.1 | 1 | 7.9 | 32.63 | 7.68 | 12.2 | 8.58 | 0.10 | 0.0067 | 0.79 | 1.012 | LOD | 0.060 | 0.00 | 0.02 |
| TC6 | N 16°38’36.5″ E 107°27’55.5″ | 3.40 | 1.5 | 7.5 | 33.34 | 7.45 | 13.5 | 15.6 | 0.08 | 0.006 | 0.43 | 1.060 | LOD | 0.060 | 9.35 | 0.01 |
| TC7 | N 16°39’12.3″ E 107°26’35″ | 12.5 | 1.5 | 7.8 | 31.6 | 7.62 | 8.2 | 12.5 | 0.06 | 0.002 | 1.11 | 1.100 | LOD | 0.042 | 9.35 | 0.02 |
| TC8 | N 16°29’26.8″ E 107°43’59.0″ | 14.8 | 0.75 | 6.5 | 33.5 | 6.5 | 23.6 | 14.82 | 0.11 | 0.002 | 1.14 | 0.774 | LOD | 0.071 | 6.23 | 0.05 |
| TC9 | N 16°27’01.0″E 107°45’30.4″ | 13.7 | 0.6 | 6.5 | 34.2 | 6.5 | 16.8 | 13.71 | 0.10 | 0.002 | 0.95 | 0.622 | LOD | 0.067 | 4.45 | 0.02 |
| TC10 | N 16°25’26.9″ E 107°46′46.4″ | 13 | 0.7 | 7.1 | 34.5 | 7.1 | 16.2 | 13.01 | 0.10 | 0.001 | 0.94 | 0.523 | LOD | 0.067 | 11.57 | 0.02 |
| TC11 | N 16°23′35.7″ E 107°48′23.1″ | 12.2 | 0.8 | 6.3 | 34.3 | 6.3 | 15 | 12.17 | 0.10 | 0.002 | 0.99 | 0.678 | LOD | 0.069 | 4.45 | 0.03 |
| TC12 | N 16°22′23.6″ E 107°49′54.2″ | 13.1 | 0.8 | 6.1 | 34.3 | 6.1 | 17.9 | 13.07 | 0.11 | 0.001 | 0.99 | 0.795 | LOD | 0.071 | 8.01 | 0.01 |
| TC13 | N 16°39′41″ E 107°26′43″ | 15.6 | - | 6.1 | 34.7 | 6.1 | 11 | 15.56 | 0.14 | 0.002 | 1.07 | 0.974 | LOD | 0.064 | 0.00 | 0.02 |
Appendix A.2. Formula Used to Calculate Carlson’s Trophic State Index
| POINTS | CTSI (SECHHI) | CSTI (TP) | CSTI (CHL-A) | CSTI | Attributes |
|---|---|---|---|---|---|
| TC 1 | 75.12793681 | 63.59996901 | 64.19361423 | 67.64051 | Eutrophic dominance of blue-green algae, algal scum |
| TC 2 | 63.21549857 | 66.35498552 | 60.00038845 | 63.19029 | Eutrophic dominance of blue-green algae, algal scum |
| TC 3 | 87.33749898 | 66.665105 | 0 | 51.3342 | Eutrophic, decreased-transparency, warm-water fisheries only |
| TC 4 | 54.15724779 | 71.34064102 | 67.43328518 | 64.31039 | Eutrophic dominance of blue-green algae, algal scum |
| TC 5 | 60 | 63.21541289 | 0 | 41.0718 | Mesotrophic, moderately clear, but increasing probability of anoxia during the summer |
| TC 6 | 54.15724779 | 63.21541289 | 52.52379454 | 56.63215 | Eutrophic, decreased transparency, warm-water fisheries only |
| TC 7 | 54.15724779 | 58.12772866 | 53.8337375 | 55.3729 | Eutrophic, decreased transparency, warm-water fisheries only |
| TC 8 | 64.14549866 | 65.56663295 | 0 | 43.23738 | Mesotrophic water moderately clear, probability of anoxia during the summer |
| TC 9 | 67.36099724 | 64.85614442 | 48.54618182 | 60.25444 | Eutrophic, decreased transparency, warm-water fisheries only |
| TC 10 | 65.13968594 | 64.85614442 | 45.24538918 | 58.41374 | Eutrophic, decreased transparency, warm-water fisheries only |
| TC 11 | 63.21549857 | 65.21576405 | 54.61895646 | 61.01674 | Eutrophic, algal scum probable, extensive macrophyte problems |
| TC 12 | 63.21549857 | 65.56663295 | 47.03396366 | 58.60537 | Eutrophic, decreased transparency, warm-water fisheries only |
| TC 13 | 83.19200032 | 64.10882694 | 51.01157637 | 66.10413 | Eutrophi, algal scum probable, extensive macrophyte problems |
References
- Beaulieu, J.; DelSontro, T.; Downing, J. Eutrophication will increase methane emissions from lakes and impoundments during the 21st century. Nat. Commun. 2019, 10, 1375. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Yang, S.; Zhao, D.; Hu, C.; Xu, W.; Anderson, D.M.; Li, Y.; Song, X.P.; Boyce, D.G.; Gibson, L.; et al. Coastal phytoplankton blooms expand and intensify in the 21st century. Nature 2023, 615, 280–284. [Google Scholar] [CrossRef]
- Anderson, D.M.; Fensin, E.; Gobler, C.J.; Hoeglund, A.E.; Hubbard, K.A.; Kulis, D.M.; Landsberg, J.H.; Lefebvre, K.A.; Provoost, P.; Richlen, M.L. Marine harmful algal blooms (HABs) in the United States: History, current status and future trends. Harmful Algae 2021, 102, 101975. [Google Scholar] [CrossRef] [PubMed]
- De Souza, M.C.; Crossetti, L.O.; Becker, V. Effects of temperature increase and nutrient enrichment on phytoplankton functional groups in a Brazilian semi-arid reservoir. Acta Limnol. Bras. 2018, 30, e215. [Google Scholar] [CrossRef]
- Gobler, C.J. Climate change and harmful algal blooms: Insights and perspective. Harmful Algae 2020, 91, 101731. [Google Scholar] [CrossRef]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Rigosi, A.; Carey, C.C.; Ibelings, B.W.; Brookes, J. The interaction between climate warming and eutrophication to promote cyanobacteria is dependent on trophic state and varies among taxa. Limnol. Oceanogr. 2014, 59, 99–114. [Google Scholar] [CrossRef]
- Trainer, V.L.; Moore, S.K.; Hallegraeff, G.; Kudela, R.M.; Clement, A.; Mardones, J.I.; Cochlan, W.P. Pelagic harmful algal blooms and climate change: Lessons from nature’s experiments with extremes. Harmful Algae 2020, 91, 101591. [Google Scholar] [CrossRef] [PubMed]
- Bashir, F.; Bashir, A.; Bouaïcha, N.; Chen, L.; Codd, G.A.; Neilan, B.A.; Xu, W.; Ziko, L.; Rajput, V.D.; Minkina, T.; et al. Cyanotoxins, biosynthetic gene clusters, and factors modulating cyanotoxin biosynthesis. World J. Microbiol. Biotechnol. 2023, 39, 241. [Google Scholar] [CrossRef]
- Otero, P.; Silva, M. Emerging marine biotoxins in European waters: Potential risks and analytical challenges. Mar. Drugs 2022, 20, 199. [Google Scholar] [CrossRef]
- Ettoumi, A.; El Khalloufi, F.; El Ghazali, I.; Oudra, B.; Amrani, A.; Nasri, H.; Bouaïcha, N. Bioaccumulation of cyanobacterial toxins in aquatic organisms and its consequences for public health. In Zooplankton and Phytoplankton: Types, Characteristics and Ecology; Nova Science: Hauppauge, NY, USA, 2011; Volume 201, pp. 1–34. [Google Scholar] [CrossRef]
- Karlson, B.; Andersen, P.; Arneborg, L.; Cembella, A.; Eikrem, W.; John, U.; Joy, J.; Klemm, K.; Kobos, J.; Lehtinen, S. Harmful algal blooms and their effects in coastal seas of Northern Europe. Harmful Algae 2021, 102, 101989. [Google Scholar] [CrossRef]
- Lad, A.; Breidenbach, J.D.; Su, R.C.; Murray, J.; Kuang, R.; Mascarenhas, A.; Najjar, J.; Patel, S.; Hegde, P.; Youssef, M. As We Drink and Breathe: Adverse Health Effects of Microcystins and Other Harmful Algal Bloom Toxins in the Liver, Gut, Lungs and Beyond. Life 2022, 12, 418. [Google Scholar] [CrossRef]
- Menezes, C.; Nova, R.; Vale, M.; Azevedo, J.; Vasconcelos, V.; Pinto, C. First description of an outbreak of cattle intoxication by cyanobacteria (blue-green algae) in the South of Portugal. Bov. Pract. 2019, 53, 66–70. [Google Scholar] [CrossRef]
- Starr, M.; Lair, S.; Michaud, S.; Scarratt, M.; Quilliam, M.; Lefaivre, D.; Robert, M.; Wotherspoon, A.; Michaud, R.; Ménard, N.; et al. Multispecies mass mortality of marine fauna linked to a toxic dinoflagellate bloom. PLoS ONE 2017, 12, e0176299. [Google Scholar] [CrossRef]
- Turner, A.D.; Turner, F.R.I.; White, M.; Hartnell, D.; Crompton, C.G.; Bates, N.; Egginton, J.; Branscombe, L.; Lewis, A.M.; Maskrey, B.H. Confirmation Using Triple Quadrupole and High-Resolution Mass Spectrometry of a Fatal Canine Neurotoxicosis following Exposure to Anatoxins at an Inland Reservoir. Toxins 2022, 14, 804. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.Z. Neurotoxins from marine dinoflagellates: A brief review. Mar. Drugs 2008, 6, 349–371. [Google Scholar] [CrossRef] [PubMed]
- Wood, R. Acute animal and human poisonings from cyanotoxin exposure—A review of the literature. Environ. Int. 2016, 91, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Hallegraeff, G.M.; Anderson, D.M.; Belin, C.; Bottein, M.-Y.D.; Bresnan, E.; Chinain, M.; Enevoldsen, H.; Iwataki, M.; Karlson, B.; McKenzie, C.H.; et al. Perceived global increase in algal blooms is attributable to intensified monitoring and emerging bloom impacts. Commun Earth Env. 2021, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Dirks, C.; Cappelli, P.; Blomqvist, M.; Ekroth, S.; Johansson, M.B.; Persson, M.; Drakare, S.; Pekar, H.; Muratovic, A.Z. Cyanotoxin occurrence and diversity in 98 cyanobacterial blooms from Swedish lakes and the Baltic Sea. Mar. Drugs 2024, 22, 199. [Google Scholar] [CrossRef]
- Fristachi, A.; Sinclair, J.L.; Hall, S.; Berkman, J.A.H.; Boyer, G.; Burkholder, J.M.; Burns, J.W.; Carmichael, W.W.; DuFour, A.; Frazier, W.; et al. Occurrence of cyanobacterial harmful algal blooms workgroup report. In Springer Ebooks; Springer: New York, NY, USA, 2008; pp. 45–103. [Google Scholar] [CrossRef]
- Halinen, K.; Jokela, J.; Fewer, D.P.; Wahlsten, M.; Sivonen, K. Direct evidence for production of microcystins by Anabaena strains from the Baltic Sea. Appl. Environ. Microbiol. 2007, 73, 6543–6550. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Fulton, R.S. Ecology of harmful cyanobacteria. In Ecology of Harmful Algae; Graneli, E., Turner, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 95–107. [Google Scholar]
- Sivonen, K.; Jones, G. Cynobacterial toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; WHO, E & FN Spon: London, UK; New York, NY, USA, 1999; pp. 41–95. [Google Scholar]
- Karjalainen, M.; Engström-Öst, J.; Korpinen, S.; Peltonen, H.; Pääkkönen, J.; Rönkkönen, S.; Suikkanen, S.; Viitasalo, M. Ecosystem consequences of cyanobacteria in the Northern Baltic Sea. Ambio 2007, 36, 195–202. [Google Scholar] [CrossRef]
- Stal, L.J.; Albertano, P.; Bergman, B.; von Brockel, K.; Gallon, J.R.; Hayes, P.K.; Sivonen, K.; Walsby, A.E. BASIC: Baltic Sea cyanobacteria. An investigation of the structure and dynamics of water blooms of cyanobacteria in the Baltic Sea—Responses to a changing environment. Cont. Shelf Res. 2003, 23, 1695–1714. [Google Scholar] [CrossRef]
- Capone, D.G.; Zehr, J.P.; Paerl, H.W.; Bergman, B.; Carpenter, E. Trichodesmium, a globally significant marine cyanobacterium. Science 1997, 276, 1221–1229. [Google Scholar] [CrossRef]
- LaRoche, J.; Breitbarth, E. Importance of the diazotrophs as a source of new nitrogen in the ocean. J. Sea Res. 2005, 53, 67–91. [Google Scholar] [CrossRef]
- Kononen, K.; Niemi, Å. Long-term variation in phytoplankton composition at the entrance to the Gulf of Finland. Ophelia 1984, (Suppl. S3), 101–110. [Google Scholar]
- Niemistö, L.; Rinne, I.; Melvasalo, T.; Niemi, Å. Blue-green algae and their nitrogen fixation in the Baltic Sea in 1980, 1982 and 1984. Merentutkimuslaitos 1989, 17, 3–20. [Google Scholar]
- Rinne, I.; Melvasalo, T.; Niemi, Å.; Niemistö, L. Studies on nitrogen in the Gulf of Bothnia. Finn. Mar. Res. 1981, 248, 117–127. [Google Scholar]
- Dao, T.S.; Cronberg, G.; Nimptsch, J.; Do-Hong, L.-C.; Wiegand, C. Toxic cyanobacteria from Tri An Reservoir, Vietnam. Nova Hedwig. 2010, 90, 433–448. [Google Scholar] [CrossRef]
- Duong, D.T. Taxonomy of Cyanobacteria of Vietnam; Agricultural Publishing House: Ha Noi, Vietnam, 1996; p. 220. [Google Scholar]
- Duong, T.; Le, T.; Dao, T.S.; Pflugmacher, S.; Rochelle-Newall, E.; Hoang, T.; VU, T.; Ho, C.; Dang, D. Seasonal variation of cyanobacteria and microcystins in the Nui Coc Reservoir, Northern Vietnam. J. Appl. Phycol. 2013, 25, 1065–1075. [Google Scholar] [CrossRef]
- Hummert, C.; Dahlmann, J.; Reinhardt, K.; Dang, H.P.H.; Dang, D.K.; Luckas, B. Liquid chromatography-mass spectrometry identification of microcystins in Microcystis aeruginosa strain from lake Thanh Cong, Hanoi, Vietnam. Chromatographia 2001, 54, 569–575. [Google Scholar] [CrossRef]
- Nguyen, T.T. Algae from the Central-Highland of Vietnam; Annals of Ho Chi Minh City University: Ho Chi Minh City, Vietnam, 1983; pp. 113–118. [Google Scholar]
- Pham, T.L.; Shimizu, K.; Dao, T.-S.; Motoo, U. First report on free and covalently bound microcystins in fish and bivalves from Vietnam: Assessment of risks to humans. Environ. Toxicol. Chem. 2017, 36, 2953–2957. [Google Scholar] [CrossRef] [PubMed]
- Shirota, A. The Plankton of South Vietnam. Freshwater and Marine Plankton; Overseas Technical Cooperation Agency Japan: Tokyo, Japan, 1966. [Google Scholar]
- Louzao, M.C.; Vilariño, N.; Vale, C.; Costas, C.; Cao, A.; Raposo-Garcia, S.; Vieytes, M.R.; Botana, L.M. Current Trends and New Challenges in Marine Phycotoxins. Mar. Drugs 2022, 20, 198. [Google Scholar] [CrossRef]
- Mafra, L.L.; De Souza, D.A.; Menezes, M.; Schramm, M.A.; Hoff, R.B. Marine biotoxins: Latest advances and challenges toward seafood safety, using Brazil as a case study. Curr. Opin. Food Sci. 2023, 53, 101078. [Google Scholar] [CrossRef]
- Bouaïcha, N.; Miles, C.O.; Beach, D.G.; Labidi, Z.; Djabri, A.; Benayache, N.Y.; Nguyen-Quang, T. Structural diversity, characterization and toxicology of microcystins. Toxins 2019, 11, 714. [Google Scholar] [CrossRef]
- Peacock, M.B.; Gibble, C.M.; Senn, D.B.; Cloern, J.E.; Kudela, R.M. Blurred lines: Multiple freshwater and marine algal toxins at the land-sea interface of San Francisco Bay, California. Harmful Algae 2018, 73, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Thung, D. Environment and Resources in the Tam Giang–Cau Hai Lagoon. In Integrated Management of Lagoon Activities—IMOLA Project Report; People’s Committee of Thua Thien Hue Province, FAO and Cooperazione Italian: Hue, Vietnam, 2007. [Google Scholar]
- Huong, T.T.T.; Berkes, F. Diversity of resource use and property rights in Tam Giang Lagoon, Vietnam. Int. J. Commons 2011, 5, 130–149. [Google Scholar] [CrossRef]
- Tuyen, T.V. Dynamics of property rights in the Tam Giang Lagoon. In Lessons in Resource Management from the Tam Giang Lagoon; Brzeski, V.J., Newkirk, G.F., Eds.; Coastal Resources Research Network (CoRR), Dalhousie University: Halifax, NS, Canada, 2002; pp. 39–52. [Google Scholar]
- Tuyen, T.V.; Armitage, D.; Marschke, M. Livelihoods and ecomanagement in the Tam Giang lagoon, Vietnam. Ocean Coast. Manag. 2010, 53, 327–335. [Google Scholar] [CrossRef]
- Phap, T.T.; Mien, L.V.; Thuan, L.T.N. Sustainable Development of Aquaculture in Tam Giang Lagoon. In International Development Research Centre (IDRC) Report; Hue University of Agriculture and Forestry: Hue, Vietnam, 2002; Available online: https://idl-bnc-idrc.dspacedirect.org/server/api/core/bitstreams/aba923ba-7837-434d-a9f1-4f939a8d4b25/content (accessed on 19 December 2023).
- Kanda, J. Determination of ammonium in seawater based on the indophenol reaction with o-phenylphenol (OPP). Water Res. 1995, 29, 2746–2750. [Google Scholar] [CrossRef]
- Fox, J.B. Kinetics and mechanisms of the Griess reaction. Anal. Chem. 1979, 51, 1493–1502. [Google Scholar] [CrossRef]
- Rump, H.H.; Krist, H. Laboratory Manual for the Examination of Water, Waste Water and Soil; VCH: New York, NY, USA, 1992. [Google Scholar]
- Standard Methods Committee of the American Public Health Association, American Water Works Association, and Water Environment Federation. 4500-NO3− nitrogen (nitrate). In Standard Methods for the Examination of Water and Wastewater; Lipps, W.C., Baxter, T.E., Braun-Howland, E., Eds.; APHA Press: Washington, DC, USA, 1998. [Google Scholar]
- Yepremian, C.; Catherine, A.; Bernard, C.; Congestri, R.; Elersek, T.; Pilkaityte, R. Phycocyanin Extraction and Determination. In Phytoplankton Pigments: Characterization, Chemotaxonomy and Applications in Oceanography, 1st ed.; Jeffrey, S.W., Wright, M., Jensen, L., Eds.; Cambridge University Press: Cambridge, UK, 2017; pp. 35–50. [Google Scholar] [CrossRef]
- Environmental Sciences Section, Inorganic Chemistry Unit, & Wisconsin State Lab of Hygiene. In ESS Method 150.1: Chlorophyll—Spectrophotometric; 465 Henry Mall: Madison WI, USA, 1991; pp. 3-359–3-362. Available online: https://polk.wateratlas.usf.edu/upload/documents/methd150.pdf (accessed on 1 June 2023).
- Sano, M.; Makabe, R.; Kurosawa, N.; Moteki, M.; Odate, T. Effects of Lugol’s iodine on long-term preservation of marine plankton samples for molecular and stable carbon and nitrogen isotope analyses. Limnol. Oceanogr. Methods 2020, 18, 635–643. [Google Scholar] [CrossRef]
- OECD (The Organization for Economic Co-operation and Development). Paris, International, “Eutrophisation des eaux. Méthodes de surveillance, d’évaluation et de lutte.” GéoProdig, Portail, Ortail D’information Géographique. Available online: https://geoprodig.cnrs.fr/items/show/156307 (accessed on 22 July 2024).
- Carlson, R.E. More complication in the chlorophyll.a-Secchi’s disc relationship. Limnol. Oceanogr. 1980, 25, 378–382. [Google Scholar] [CrossRef]
- Nguyen, T.T.L.; Pham, N.T.T.; Tran, T.M.H. Occurrences of Microcystis spp. and microcystins in some cyanobacterial blooms in freshwater bodies in Vietnam. VNU J. Sci. Nat. Sci. Technol. 2010, 26, 172–177. [Google Scholar]
- Panou, M.; Zervou, S.-K.; Kaloudis, T.; Hiskia, A.; Gkelis, S. A Greek Cylindrospermopsis Raciborskii Strain: Missing Link in Tropic Invader’s Phylogeography Tale. Harmful Algae 2018, 80, 96–106. [Google Scholar] [CrossRef]
- Wasmund, N. Occurrence of cyanobacterial blooms in the Baltic Sea in relation to environmental conditions. Int. Rev. Hydrobiol. 1997, 82, 169–184. [Google Scholar] [CrossRef]
- Lehman, P.W.; Boyer, G.; Hall, C.; Waller, S.; Gehrts, K. Distribution and toxicity of a new colonial microcystis aeruginosa bloom in the San Francisco Bay estuary, California. Hydrobiologia 2005, 541, 87–99. [Google Scholar] [CrossRef]
- Tanabe, Y.; Hodoki, Y.; Sano, T.; Tada, K.; Watanabe, M.M. Adaptation of the freshwater bloom-forming cyanobacterium Microcystis aeruginosa to brackish water is driven by recent horizontal transfer of sucrose genes. Front. Microbiol. 2018, 9, 1150. [Google Scholar] [CrossRef]
- Tarafdar, L.; Mohapatra, M.; Muduli, P.R.; Kumar, A.; Mishra, D.R.; Rastogi, G. Co-occurrence patterns and environmental factors associated with rapid onset of Microcystis aeruginosa bloom in a tropical coastal lagoon. J. Environ. Manag. 2023, 325, 116580. [Google Scholar] [CrossRef]
- Miller, M.A.; Kudela, R.M.; Mekebri, A.; Crane, D.; Oates, S.C.; Tinker, M.T.; Staedler, M.; Miller, W.A.; Toy-Choutka, S.; Dominik, C.; et al. Evidence for a novel marine harmful algal bloom: Cyanotoxin (microcystin) transfer from land to sea otters. PLoS ONE 2010, 5, e12576. [Google Scholar] [CrossRef]
- Tonk, L.; Bosch, K.; Visser, P.; Huisman, J. Salt tolerance of the harmful cyanobacterium Microcystis aeruginosa. Aquat. Microb. Ecol. 2007, 46, 117–123. [Google Scholar] [CrossRef]
- World Health Organization. Manganese in Drinking-Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; No. WHO/SDE/WSH/03.04/104; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Benayache, N.Y.; Afri-Mehennaoui, F.Z.; Kherief-Nacereddine, S.; Vo-Quoc, B.; Hushchyna, K.; Nguyen-Quang, T.; Bouaïcha, N. Massive fish death associated with the toxic cyanobacterial Planktothrix sp. bloom in the Béni-Haroun Reservoir (Algeria). Environ. Sci. Pollut. Res. 2022, 29, 80849–80859. [Google Scholar] [CrossRef] [PubMed]
- Vuong, T.T.T.; Hao, L.T.H.; Minh, T.B.; Chi, L.D. Spatial an1 qd seasonal variation of diarrheic shellfish poisoning (DSP) toxins in bivalve mollusks from some coastal regions of Vietnam and assessment of potential health risks. Mar. Pollut. Bull. 2018, 133, 911–919. [Google Scholar]
- Le, T.T.; Nguyen, V.N.; Fukuyo, Y. Dinophysis (Dinophyceae) in Vietnamese waters. Coast. Mar. Sci. 2012, 35, 73–77. [Google Scholar]
- Ngo, H.P. Survey of Biotoxin in ASEAN Region; Article “Vietnam”. In Technical Compilation of Biotoxins Monitoring in ASEAN Region 2009–2012; Southeast Asian Fisheries Development Center: Singapore, 2012; pp. 65–68. [Google Scholar]
- Gibble, C.M.; Peacock, M.B.; Kudela, R.M. Evidence of freshwater algal toxins in marine shellfish: Implications for human and aquatic health. Harmful Algae 2016, 59, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Onofrio, M.D.; Egerton, T.A.; Reece, K.S.; Pease, S.K.D.; Sanderson, M.P.; Jones, W., III; Yeargan, E.; Roach, A.; DeMent, C.; Wood, A.; et al. Spatiotemporal distribution of phycotoxins within nearshore waters of the Chesapeake Bay and Virginia coastal bays. Harmful Algae 2021, 103, 101993. [Google Scholar] [CrossRef]
- Straquadine, N.R.W.; Kudela, R.M.; Gobler, C.J. Hepatotoxic shellfish poisoning: Accumulation of microcystins in Eastern oysters (Crassostrea virginica) and Asian clams (Corbicula fluminea) exposed to wild and cultured populations of the harmful cyanobacteria, Microcystis. Harmful Algae 2022, 115, 102236. [Google Scholar] [CrossRef]
- Pease, S.K.D.; Egerton, T.A.; Reece, K.S.; Sanderson, M.P.; Onofrio, M.D.; Evan, Y.; Wood, A.; Roach, A.; Huang, I.-S.W.; Scott, G.P.; et al. Co-occurrence of marine and freshwater phycotoxins in oysters, and analysis of possible predictors for management. Toxicon 2023, 19, 100166. [Google Scholar] [CrossRef]
- Chorus, I.; Welker, M. (Eds.) Toxic Cyanobacteria in Water, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2021; on behalf of the World Health Organization, Geneva, CH. [Google Scholar]
- Butler, N.; Carlisle, J.; Linville, R. Toxicological Summary and Suggested Action Levels to Reduce Potential Adverse Health Effects of Six Cyanotoxins. In Toxicological Summary and Suggested Action Levels to Reduce Potential Adverse Health Effects of Six Cyanotoxins; Office of Environmental Health Hazard Assessment California Environmental Protection Agency, 2012; Final Report. Available online: https://www.waterboards.ca.gov/water_issues/programs/tmdl/records/state_board/2016/ref4294.pdf (accessed on 1 June 2024).
- Mulvenna, V.; Dale, K.; Priestly, B.; Mueller, U.; Humpage, A.; Shaw, G.; Allinson, G.; Falconer, I. Health risk assessment for cyanobacterial toxins in seafood. Int. J. Environ. Res. Public Health 2012, 9, 807–820. [Google Scholar] [CrossRef]
- Nguyen, T.A.; Picot, C.; Tran, T.L.; Carpentier, F.G.; Roudot, A.C.; Parent-Massin, D.M. Evaluation of shellfish consumption in Nha Trang City, Southern coastal Vietnam. Malays J Nutr. 2012, 18, 37–45. [Google Scholar]
- Nishiwaki-Matsushima, R.; Ohta, T.; Nishiwaki, S.; Suganuma, M.; Kohyama, T.; Ishikawa, T.; Carmichael, W.W.; Fujiki, H. Liver tumor promotion by the cyanobacterial cyclic peptide toxin microcystin-LR. J. Cancer Res. Clin. Oncol. 1992, 118, 420–424. [Google Scholar] [CrossRef]
- Grosse, Y.; Baan, R.; Straif, K.; Ghissassi, E.F.; Cogliani, V. on behalf of the WHO International Agency for Research on Cancer Monograph Working Group. Carcinogenicity of nitrate, nitrite, and cyanobacterial peptide toxins. Lancet Oncol. 2006, 7, 628–629. [Google Scholar] [CrossRef] [PubMed]
- Foss, A.J.; Miles, C.O.; Samdal, I.A.; Løvberg, K.E.; Wilkins, A.L.; Rise, F.; Jaabæk, J.A.H.; McGowan, P.C.; Aubel, M.T. Analysis of free and metabolized microcystins in samples following a bird mortality event. Harmful Algae 2018, 80, 117–129. [Google Scholar] [CrossRef] [PubMed]


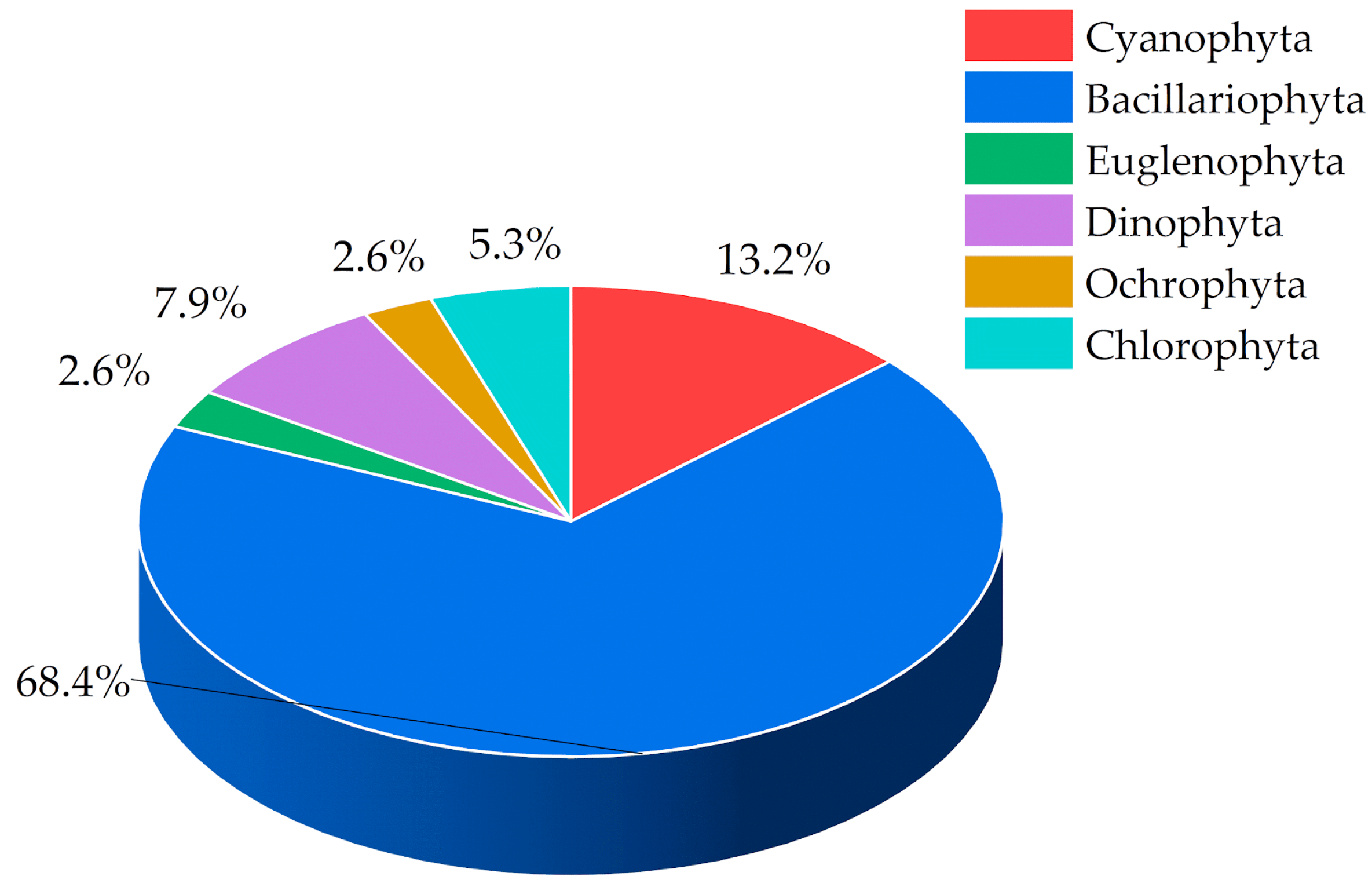

| Phylum | Genera |
|---|---|
| Bacillariophyta (diatoms) | Amphiphrora sp., Aulacoseira granulata, Bacillaria paxillifera, Chaetoceros sp., Coscinodiscus radiates, Cylindrotheca closterium, Cymbella sp., Guinardia flaccida, Leptocylindrus sp., Melosira sp., Navicula sp., Nitzschia sp., Pleurosigma angulatum, Rhizosolenia sp., Tabellaria sp., Thalassionema frauenfeldii |
| Cyanophyta | Anabaena (actually Dolichospermum) sp., Raphidiopsis raciborskii, Pseudanabaena sp., Oscillatoria sp. |
| Dinophyta | Protoperidinium sp., Dinophysis sp. |
| Chlorophyta | Chlamydomonas sp., Closterium sp., Desmidium baileyi |
| Ochrophyta | Dinobryon sertularia |
| Euglenophyta | Euglena viridis |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sahoo, D.; Tran, N.K.N.; Nguyen, T.G.-H.; Ho, T.T.H.; Phan, T.T.H.; Hoang, D.T.H.; Binh, N.H.; Nguyen, T.T.L.; Doc, L.Q.; Bouaïcha, N.; et al. Co-Occurrence of Cyanotoxins and Phycotoxins in One of the Largest Southeast Asian Brackish Waterbodies: A Preliminary Study at the Tam Giang—Cau Hai Lagoon (Vietnam). Limnol. Rev. 2024, 24, 335-353. https://doi.org/10.3390/limnolrev24030020
Sahoo D, Tran NKN, Nguyen TG-H, Ho TTH, Phan TTH, Hoang DTH, Binh NH, Nguyen TTL, Doc LQ, Bouaïcha N, et al. Co-Occurrence of Cyanotoxins and Phycotoxins in One of the Largest Southeast Asian Brackish Waterbodies: A Preliminary Study at the Tam Giang—Cau Hai Lagoon (Vietnam). Limnological Review. 2024; 24(3):335-353. https://doi.org/10.3390/limnolrev24030020
Chicago/Turabian StyleSahoo, Devleena, Ngoc Khanh Ni Tran, Thi Gia-Hang Nguyen, Thi Thu Hoai Ho, Thi Thuy Hang Phan, Duong Thu Huong Hoang, Ngo Huu Binh, Thi Thu Lien Nguyen, Luong Quang Doc, Noureddine Bouaïcha, and et al. 2024. "Co-Occurrence of Cyanotoxins and Phycotoxins in One of the Largest Southeast Asian Brackish Waterbodies: A Preliminary Study at the Tam Giang—Cau Hai Lagoon (Vietnam)" Limnological Review 24, no. 3: 335-353. https://doi.org/10.3390/limnolrev24030020
APA StyleSahoo, D., Tran, N. K. N., Nguyen, T. G.-H., Ho, T. T. H., Phan, T. T. H., Hoang, D. T. H., Binh, N. H., Nguyen, T. T. L., Doc, L. Q., Bouaïcha, N., & Nguyen-Quang, T. (2024). Co-Occurrence of Cyanotoxins and Phycotoxins in One of the Largest Southeast Asian Brackish Waterbodies: A Preliminary Study at the Tam Giang—Cau Hai Lagoon (Vietnam). Limnological Review, 24(3), 335-353. https://doi.org/10.3390/limnolrev24030020







