Ex-Vivo Exposure on Biological Tissues in the 2-μm Spectral Range with an All-Fiber Continuous-Wave Holmium Laser
Abstract
:1. Introduction
2. Materials and Methods
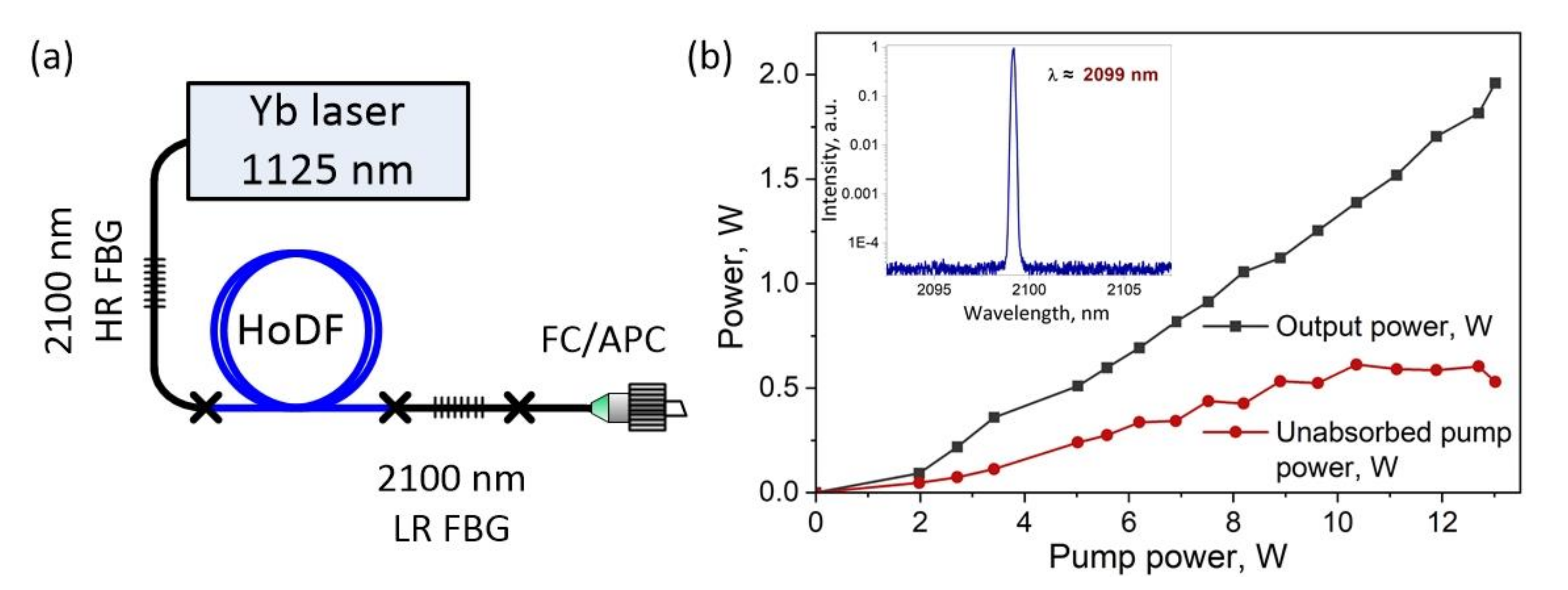
2.1. Ho-Doped Fiber Laser Operating in Continuous-Wave Mode
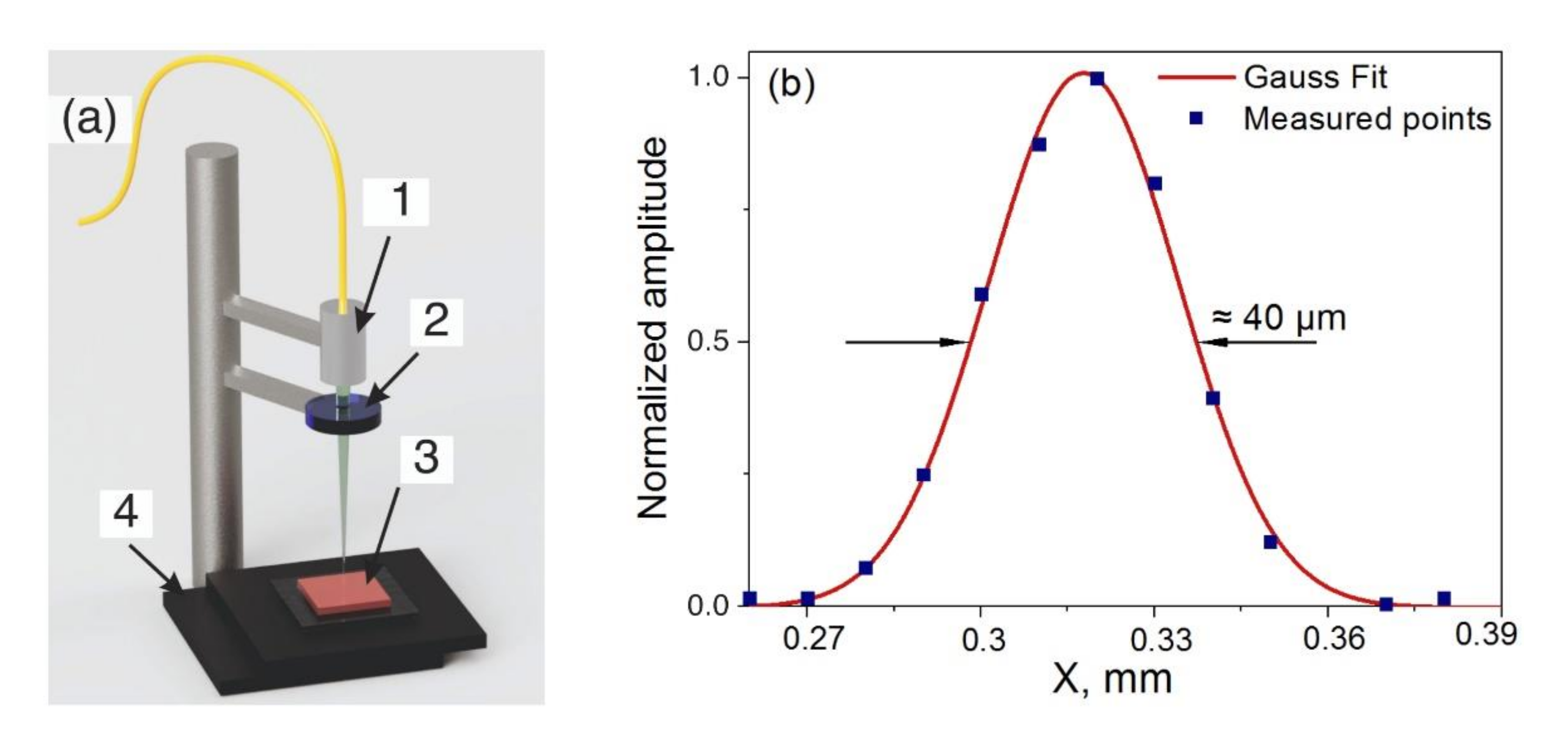
2.2. Ex-Vivo Tissue Preparation and Experimental Setup
2.3. Laser Parameters of Exposure
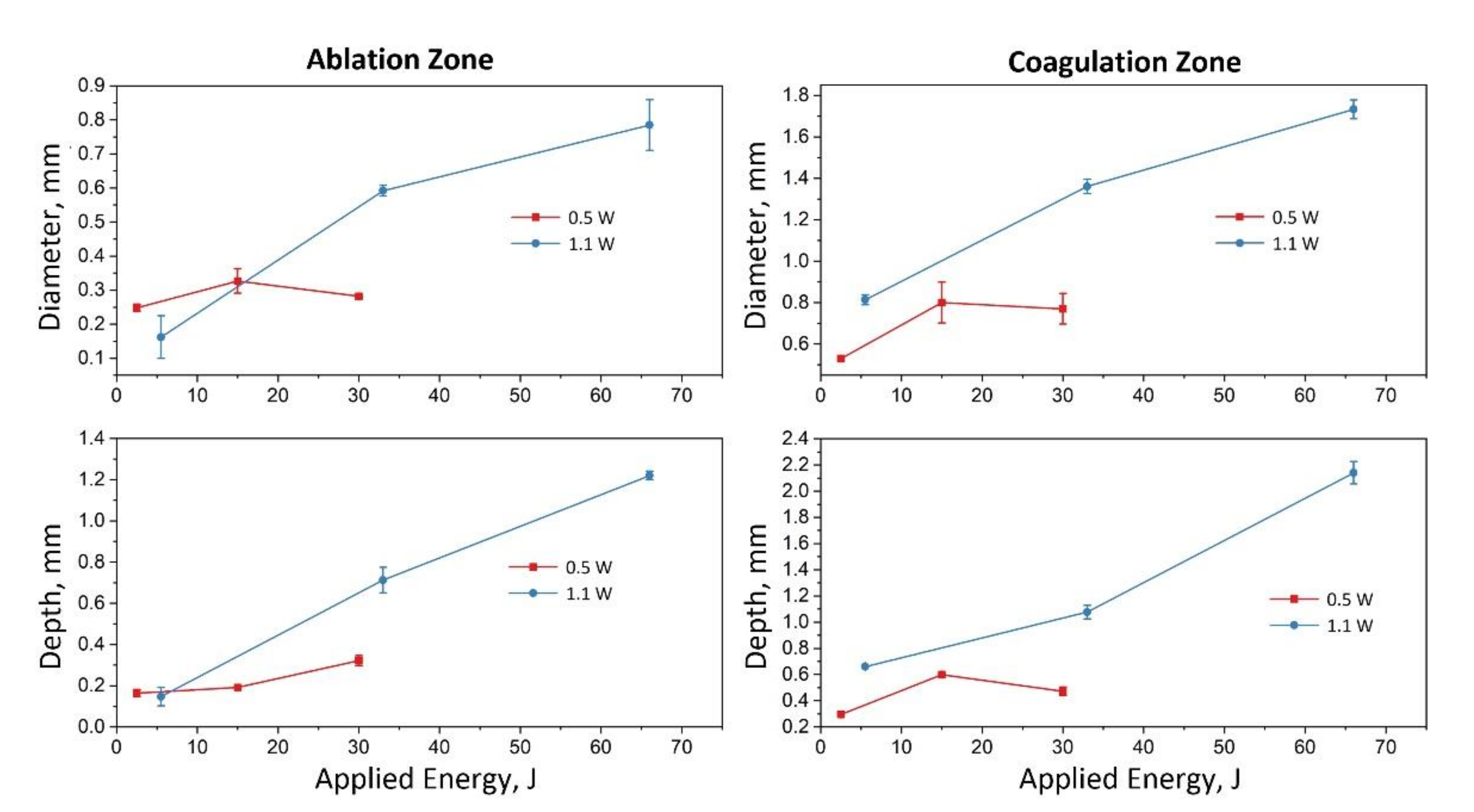
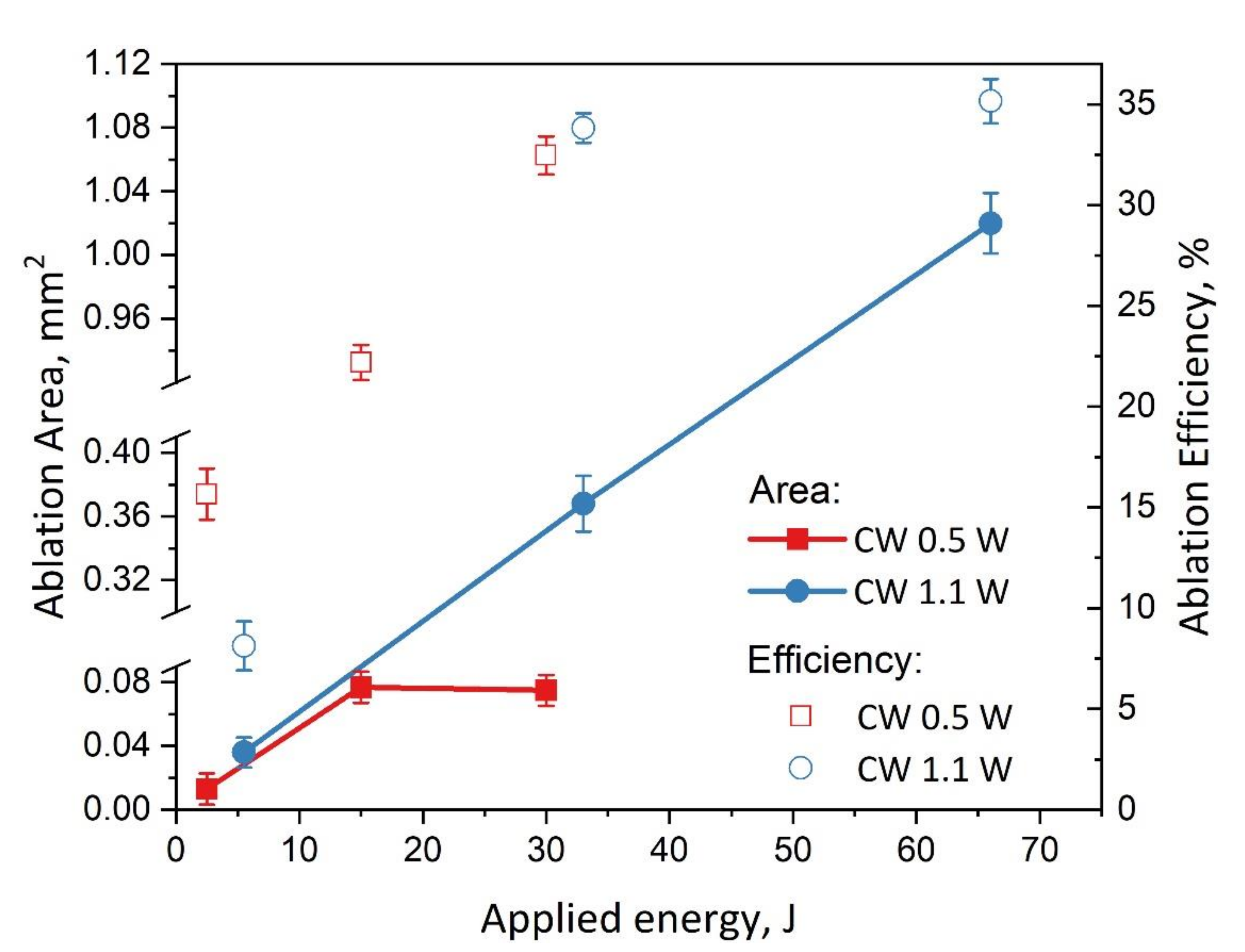
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shi, W.; Fang, Q.; Zhu, X.; Norwood, R.A.; Peyghambarian, N. Fiber lasers and their applications. Appl. Opt. 2014, 53, 6554–6568. [Google Scholar] [CrossRef]
- Gursel, A.T. Fiber lasers and their medical applications. In Optical Amplifiers—A Few Different Dimensions; Intech Open: London, UK, 2018. [Google Scholar] [CrossRef] [Green Version]
- Holmen, L.G.; Shardlow, P.C.; Barua, P.; Sahu, J.K.; Simakov, N.; Hemming, A.; Clarkson, W.A. Tunable holmium-doped fiber laser with multiwatt operation from 2025 nm to 2200 nm. Opt. Lett. 2019, 44, 4131–4134. [Google Scholar] [CrossRef] [PubMed]
- Antipov, S.O.; Kamynin, V.A.; Medvedkov, O.I.; Marakulin, A.V.; Minashina, L.A.; Kurkov, A.S.; Baranikov, A.V. Holmium fibre laser emitting at 2.21 μm. Quantum Electron. 2013, 43, 603. [Google Scholar] [CrossRef]
- Alekseev, N.E.; Izyneev, A.A.; Kravchenko, V.B.; Rudnitskii, Y.P. Influence of the concentration quenching and of water on the energy characteristics of neodymium-activated glasses. Sov. J. Quantum Electron. 1975, 4, 1111. [Google Scholar] [CrossRef]
- Falconi, M.C.; Laneve, D.; Prudenzano, F. Advances in mid-IR fiber lasers: Tellurite, fluoride and chalcogenide. Fibers 2017, 5, 23. [Google Scholar] [CrossRef] [Green Version]
- Ma, J.; Qin, Z.; Xie, G.; Qian, L.; Tang, D. Review of mid-infrared mode-locked laser sources in the 2.0 μm–3.5 μm spectral region. Appl. Phys. Rev. 2019, 6, 021317. [Google Scholar] [CrossRef] [Green Version]
- Filatova, S.A.; Shcherbakov, I.A.; Tsvetkov, V.B. Optical properties of animal tissues in the wavelength range from 350 to 2600 nm. J. Biomed. Opt. 2017, 22, 035009. [Google Scholar] [CrossRef]
- Serebryakov, V.A.; Boĭko, É.V.; Petrishchev, N.N.; Yan, A.V. Medical applications of mid-IR lasers. Problems and prospects. J. Opt. Technol. 2010, 77, 6–17. [Google Scholar] [CrossRef]
- Huusmann, S.; Wolters, M.; Kramer, M.W.; Bach, T.; Teichmann, H.O.; Eing, A.; Herrmann, T.R. Tissue damage by laser radiation: An in vitro comparison between Tm: YAG and Ho: YAG laser on a porcine kidney model. Springerplus 2016, 5, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antipov, O.L.; Zakharov, N.G.; Fedorov, M.; Shakhova, N.M.; Prodanets, N.N.; Snopova, L.B.; Sharkov, V.V.; Sroka, R. Cutting effects induced by 2 μm laser radiation of cw Tm: YLF and cw and Q-switched Ho: YAG lasers on ex-vivo tissue. Med. Laser Appl. 2011, 26, 67–75. [Google Scholar] [CrossRef]
- Serebryakov, V.S.; Boĭko, É.V.; Kalintsev, A.G.; Kornev, A.F.; Narivonchik, A.S.; Pavlova, A.L. Mid-IR laser for high-precision surgery. J. Opt. Technol. 2015, 82, 781–788. [Google Scholar] [CrossRef]
- Jones, P.S. Development and characterisation of holmium and erbium lasers for the ablation of biological tissue. Ph.D. Thesis, University of London, University College London, London, UK, 1993. [Google Scholar]
- Blackmon, R.L.; Fried, N.M.; Irby, P.B. Comparison of holmium: YAG and thulium fiber laser lithotripsy: Ablation thresholds, ablation rates, and retropulsion effects. J. Biomed. Opt. 2011, 16, 071403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alagha, H.Z.; Gülsoy, M. Photothermal ablation of liver tissue with 1940-nm thulium fiber laser: An ex vivo study on lamb liver. J. Biomed. Opt. 2016, 21, 015007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tunc, B.; Gulsoy, M. Tm: Fiber laser ablation with real-time temperature monitoring for minimizing collateral thermal damage: Ex vivo dosimetry for ovine brain. Lasers Surg. Med. 2013, 45, 48–56. [Google Scholar] [CrossRef]
- Sliney, D.H.; Trokel, S.L. Medical Lasers and Their Safe Use, 1st ed.; Springer Science & Business Media: New York, NY, USA, 1993; p. 230. [Google Scholar] [CrossRef]
- Vogel, A.; Venugopalan, V. Mechanisms of pulsed laser ablation of biological tissues. Chem. Rev. 2003, 103, 577–644. [Google Scholar] [CrossRef] [Green Version]
- Hoy, C.L.; Ferhanoglu, O.; Yildirim, M.; Kim, K.H.; Karajanagi, S.S.; Chan, K.M.C.; Kobler, J.B.; Zeitels, S.M.; Ben-Yakar, A. Clinical ultrafast laser surgery: Recent advances and future directions. IEEE J. Sel. Top. Quantum Electron. 2013, 20, 242–255. [Google Scholar] [CrossRef]
- Pierce, M.C.; Jackson, S.D.; Dickinson, M.R.; King, T.A. Laser-tissue interaction with a high-power 2-µm fiber laser: Preliminary studies with soft tissue. Lasers Surg. Med. 1999, 25, 407–413. [Google Scholar] [CrossRef]
- Verdaasdonk, R.M.; Borst, C.; Van Gemert, M.J.C. Explosive onset of continuous wave laser tissue ablation. Phys. Med. Biol. 1990, 35, 1129–1144. [Google Scholar] [CrossRef] [PubMed]
- Domankevitz, Y.; McMillan, K.; Nishioka, N.S. Characterization of tissue ablation with a continuous wave holmium laser. Lasers Surg. Med. 1996, 19, 97–102. [Google Scholar] [CrossRef]
- LeCarpentier, G.L.; Motamedi, M.; McMath, L.P.; Rastegar, S.; Welch, A.J. Continuous wave laser ablation of tissue: Analysis of thermal and mechanical events. IEEE Trans. Biomed. Eng. 1993, 40, 188–200. [Google Scholar] [CrossRef]
- Herrmann, T.R.; Liatsikos, E.N.; Nagele, U.; Traxer, O.; Merseburger, A.S. EAU Guidelines Panel on Lasers. Eur. Urol. 2012, 61, 783–795. [Google Scholar] [CrossRef]
- Wollin, T.A.; Denstedt, J.D. The Holmium Laser in Urology. J. Clin. Laser Med. Surg. 1998, 16, 13–20. [Google Scholar] [CrossRef]
- Wieneke, S.; Gerhard, C. Lasers in Medical Diagnosis and Therapy; IOP Publishing: Bristol, UK, 2018. [Google Scholar] [CrossRef]
- Skandalakis, G.P.; Rivera, D.R.; Rizea, C.D.; Bouras, A.; Jesu Raj, J.G.; Bozec, D.; Hadjipanayis, C.G. Hyperthermia treatment advances for brain tumors. Int. J. Hyperth. 2020, 37, 3–19. [Google Scholar] [CrossRef]
- Kashiwagi, S. Laser adjuvant for vaccination. FASEB J. 2020, 34, 3485–3500. [Google Scholar] [CrossRef] [Green Version]
- Belykh, E.; Yagmurlu, K.; Martirosyan, N.L.; Lei, T.; Izadyyazdanabadi, M.; Malik, K.M.; Byvaltsev, V.A.; Nakaji, P.; Preul, M.C. Laser application in neurosurgery. Surg. Neurol. Int. 2017, 8, 274. [Google Scholar] [CrossRef]
- Pyo, H.; Kim, H.; Kang, H.W. Evaluations on laser ablation of ex vivo porcine stomach tissue for development of Ho: YAG-assisted endoscopic submucosal dissection (ESD). Lasers Med. Sci. 2021, 36, 1437–1444. [Google Scholar] [CrossRef]
- Huang, Y.; Jivraj, J.; Zhou, J.; Ramjist, J.; Wong, R.; Gu, X.; Yang, V.X. Pulsed and CW adjustable 1942 nm single-mode all-fiber Tm-doped fiber laser system for surgical laser soft tissue ablation applications. Opt. Express 2016, 24, 16674–16686. [Google Scholar] [CrossRef]
- Tunc, B.; Gulsoy, M. Stereotaxic laser brain surgery with 1940-nm Tm: Fiber laser: An in vivo study. Lasers Surg. Med. 2019, 51, 643–652. [Google Scholar] [CrossRef]
- Scholle, K.; Lamrini, S.; Koopmann, P.; Fuhrberg, P. 2 µm laser sources and their possible applications. In Frontiers in Guidedwave Optics and Optoelectronics; Bishnu, P., Ed.; IntechOpen: Rijeka, Croatia, 2010; p. 674. [Google Scholar] [CrossRef] [Green Version]
- Fried, N.M.; Murray, K.E. High-power thulium fiber laser ablation of urinary tissues at 1.94 µm. J. Endourol. 2005, 19, 25–31. [Google Scholar] [CrossRef] [Green Version]
- Khoder, W.; Zilinberg, K.; Waidelich, R.M.; Stief, C.G.; Becker, A.J.; Pangratz, T.; Henning, G.; Sroka, R. Ex vivo comparison of the tissue effects of six laser wavelengths for potential use in laser supported partial nephrectomy. J. Biomed. Opt. 2012, 17, 068005. [Google Scholar] [CrossRef]
- Bashkatov, A.N.; Genina, E.A.; Tuchin, V.V. Optical properties of skin, subcutaneous, and muscle tissues: A review. J. Innov. Opt. Health Sci. 2011, 4, 9–38. [Google Scholar] [CrossRef]
- Kopyeva, M.S.; Filatova, S.A.; Kamynin, V.A.; Chekhlova, T.K.; Tsvetkov, V.B. Comparison of Continuous Wave and Ultrashort Pulsed Holmium-doped Fiber Lasers Exposure on Ex-vivo Tissue. In Proceedings of the 2021 Conference on Lasers and Electro-Optics Europe & European Quantum Electronics Conference (CLEO/Europe-EQEC), Munich, Germany, 20–24 June 2021; IEEE: New York, NY, USA, 2021; p. 1. [Google Scholar] [CrossRef]
- El-Sherif, A.F.; King, T.A. Soft and hard tissue ablation with short-pulse high peak power and continuous thulium-silica fibre lasers. Lasers Med. Sci. 2003, 18, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Tunc, B.; Gulsoy, M. The Comparison of Thermal Effects of a 1940-nm Tm: Fiber Laser and 980-nm Diode Laser on Cortical Tissue: Stereotaxic Laser Brain Surgery. Lasers Surg. Med. 2020, 52, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Image Processing and Analysis in Java. Available online: https://imagej.nih.gov/ij/index.html (accessed on 29 October 2021).





| Output Power, W | Power Density, W/cm2 | Exposure Time, s | Applied Energy, J |
|---|---|---|---|
| 0.3 | 24 × 103 | 5 | 1.5 |
| 30 | 9 | ||
| 60 | 18 | ||
| 0.5 | 40 × 103 | 5 | 2.5 |
| 30 | 15 | ||
| 60 | 30 | ||
| 1.1 | 88 × 103 | 5 | 5.5 |
| 30 | 33 | ||
| 60 | 66 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopyeva, M.S.; Filatova, S.A.; Kamynin, V.A.; Trikshev, A.I.; Kozlikina, E.I.; Astashov, V.V.; Loschenov, V.B.; Tsvetkov, V.B. Ex-Vivo Exposure on Biological Tissues in the 2-μm Spectral Range with an All-Fiber Continuous-Wave Holmium Laser. Photonics 2022, 9, 20. https://doi.org/10.3390/photonics9010020
Kopyeva MS, Filatova SA, Kamynin VA, Trikshev AI, Kozlikina EI, Astashov VV, Loschenov VB, Tsvetkov VB. Ex-Vivo Exposure on Biological Tissues in the 2-μm Spectral Range with an All-Fiber Continuous-Wave Holmium Laser. Photonics. 2022; 9(1):20. https://doi.org/10.3390/photonics9010020
Chicago/Turabian StyleKopyeva, Mariya S., Serafima A. Filatova, Vladimir A. Kamynin, Anton I. Trikshev, Elizaveta I. Kozlikina, Vadim V. Astashov, Victor B. Loschenov, and Vladimir B. Tsvetkov. 2022. "Ex-Vivo Exposure on Biological Tissues in the 2-μm Spectral Range with an All-Fiber Continuous-Wave Holmium Laser" Photonics 9, no. 1: 20. https://doi.org/10.3390/photonics9010020
APA StyleKopyeva, M. S., Filatova, S. A., Kamynin, V. A., Trikshev, A. I., Kozlikina, E. I., Astashov, V. V., Loschenov, V. B., & Tsvetkov, V. B. (2022). Ex-Vivo Exposure on Biological Tissues in the 2-μm Spectral Range with an All-Fiber Continuous-Wave Holmium Laser. Photonics, 9(1), 20. https://doi.org/10.3390/photonics9010020







