Synthesis and Applications of Halide Perovskite Nanocrystals in Optoelectronics
Abstract
1. Introduction
2. Synthesis of Perovskite Nanocrystals
2.1. Synthetic Methods
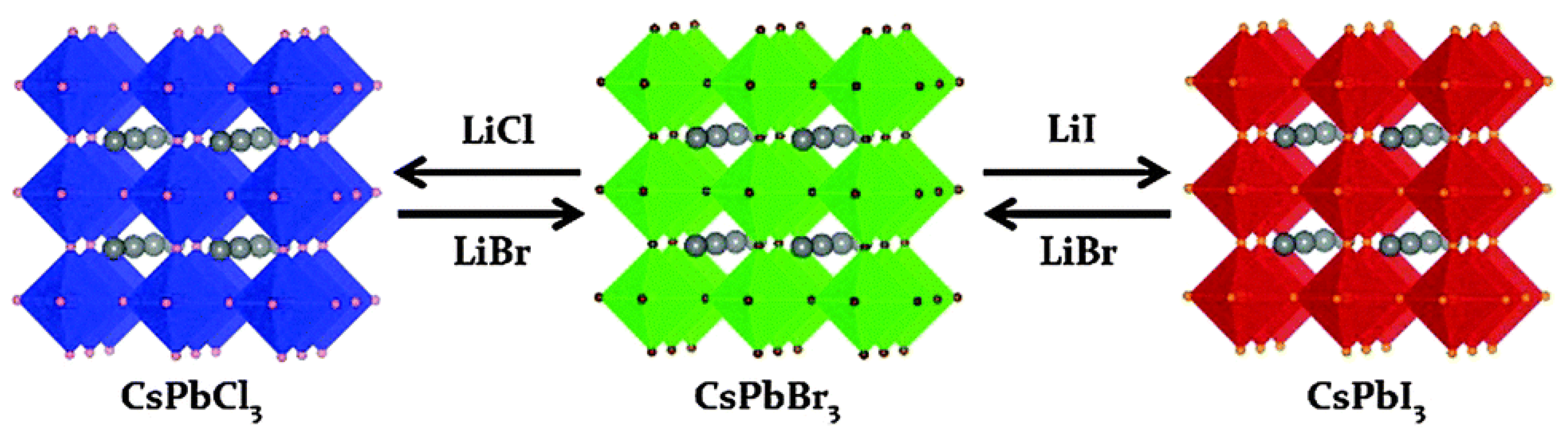
2.2. Improving Methods of Synthesizing Halide Perovskite Nanocrystals
3. Light-Emitting Diodes
3.1. Introduction of the Perovskite Nanocrystal LEDs
3.2. Optimizing Nanocrystal Perovskite Device for Wavelengths of Blue and Cyan Light (450 nm–500 nm)
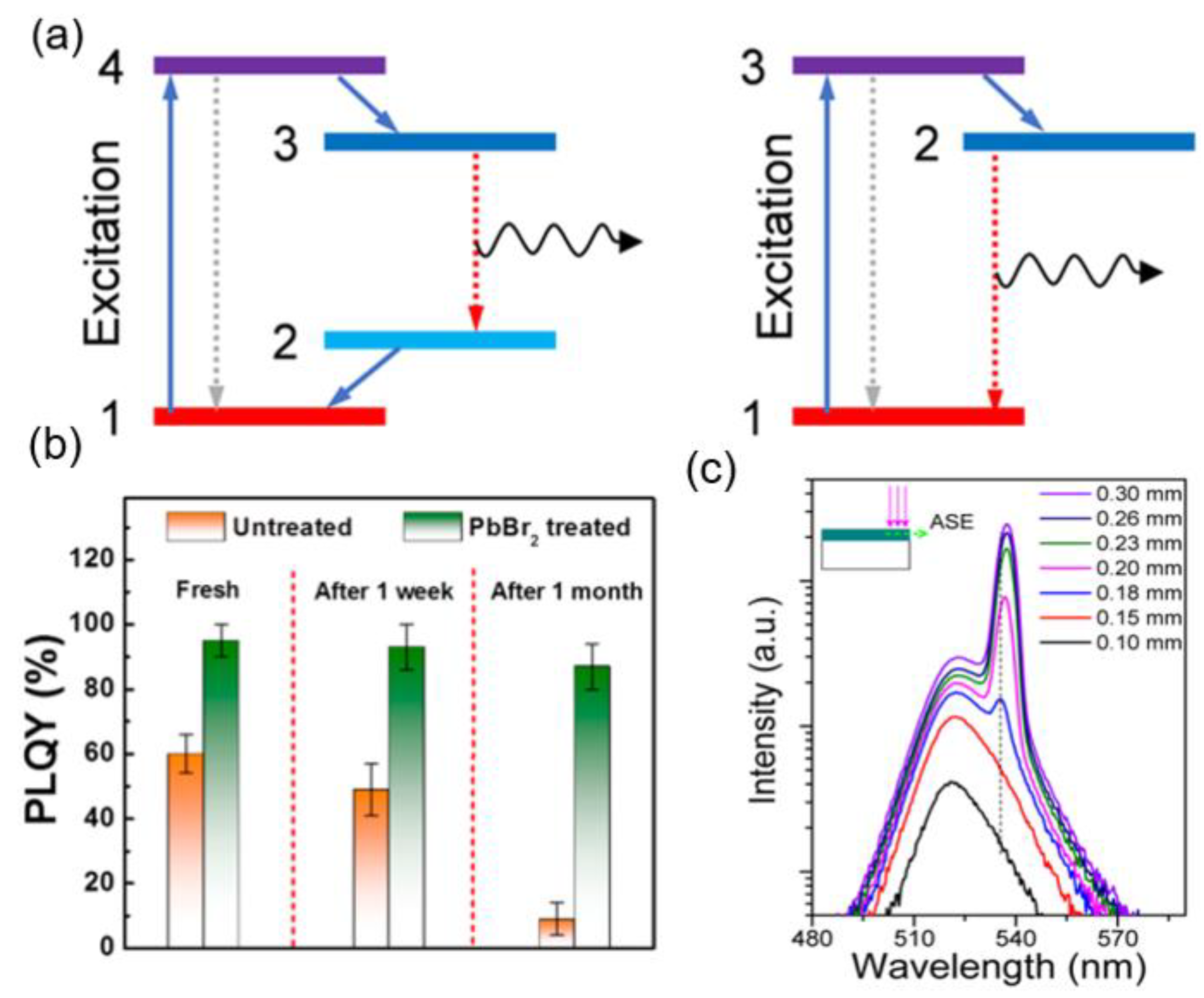
3.3. Optimizing Nanocrystal Perovskite Device for Wavelengths of Green Light (495 nm–570 nm)
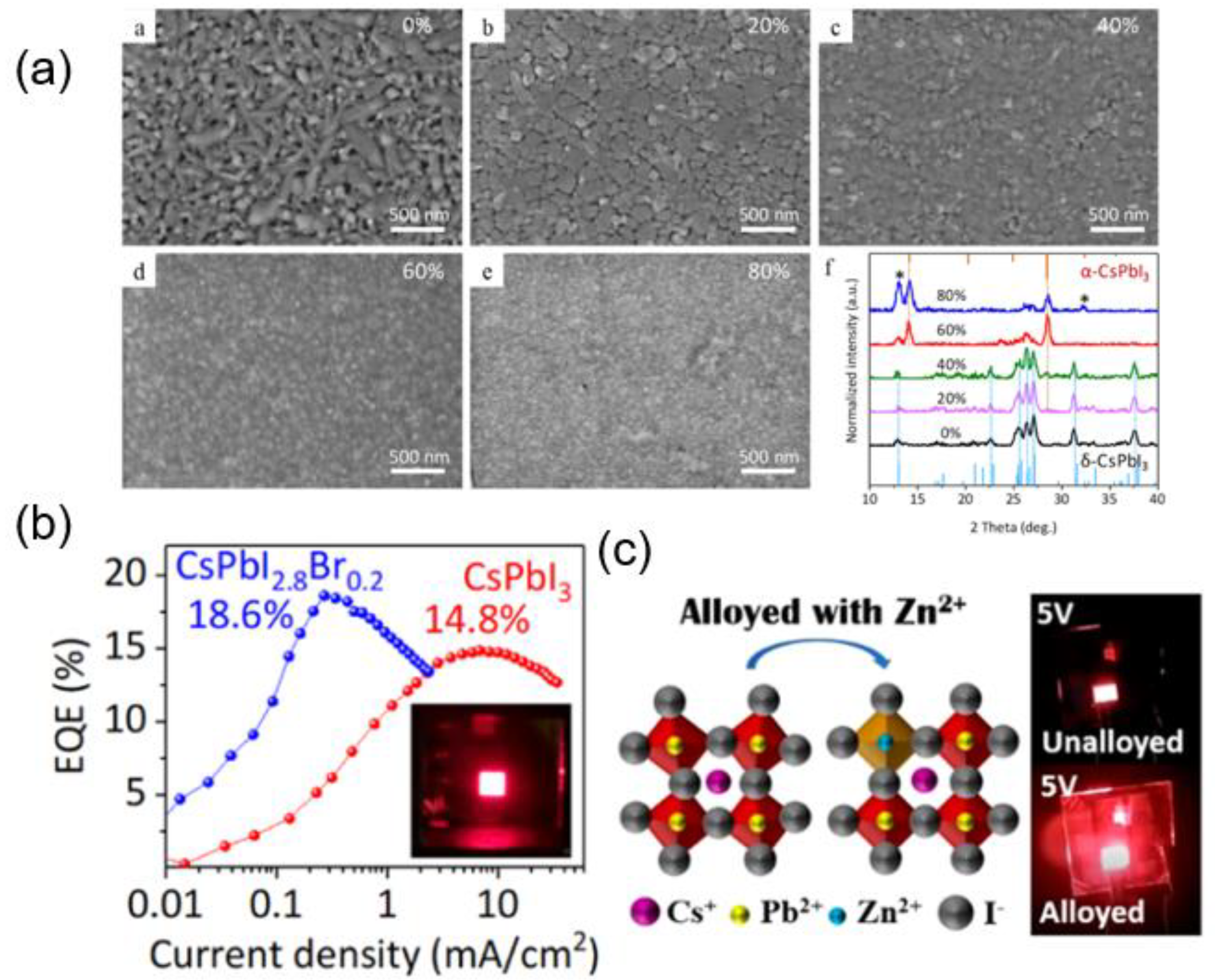
3.4. Optimizing Nanocrystal Perovskite Device for Wavelengths of Orange and Red Light (590 nm–750 nm)
4. Solar Cells
4.1. The Development of Halide Perovskite Nanocrystals Solar Cells
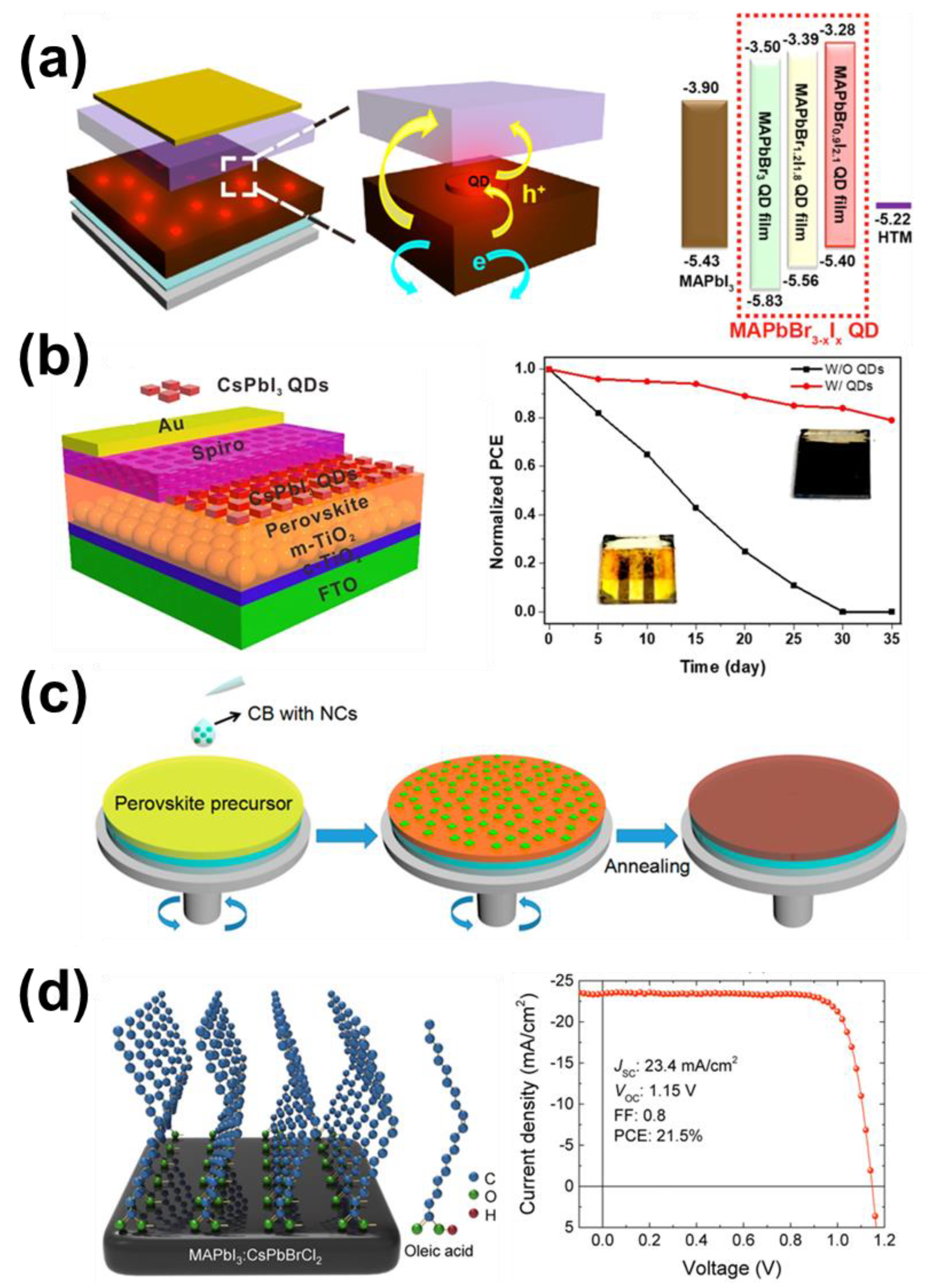
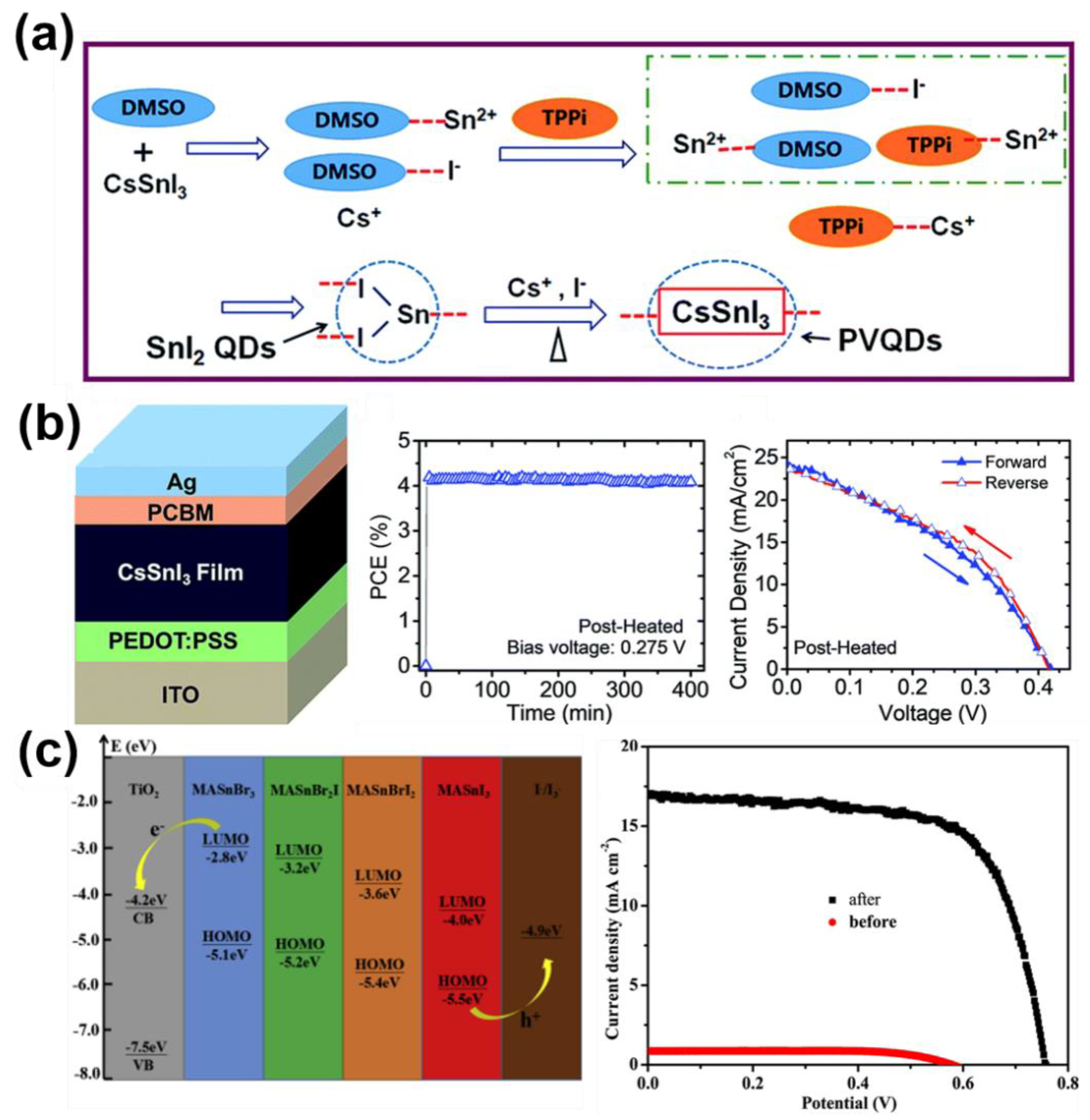
4.2. The Solar Cells of Halide Perovskites Nanocrystals as Active Layers
4.3. Halide Perovskite NCs in Traditional Perovskite Solar Cells
5. Photodetectors
5.1. Introduction of Perovskite Nanocrystal Photodetectors
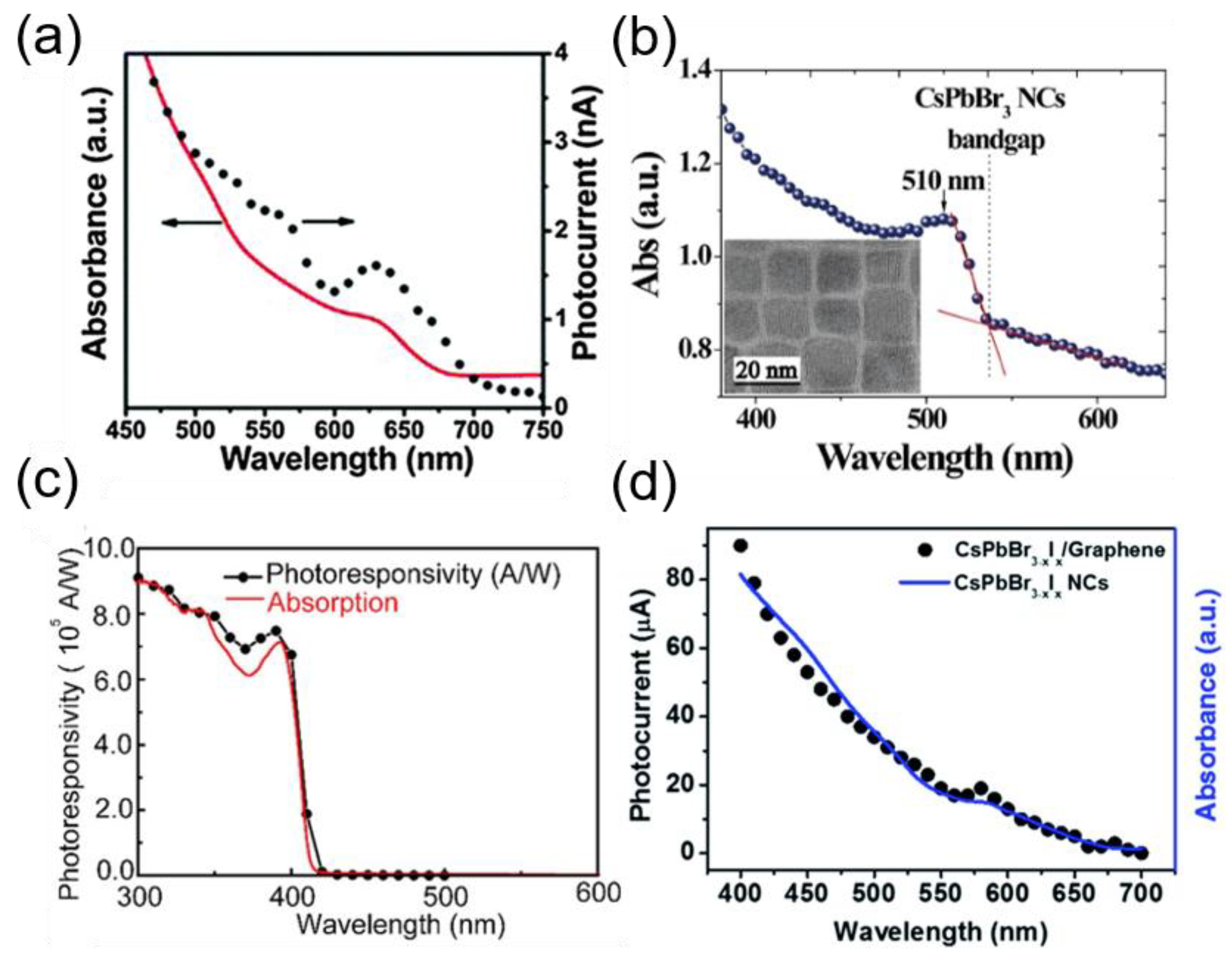
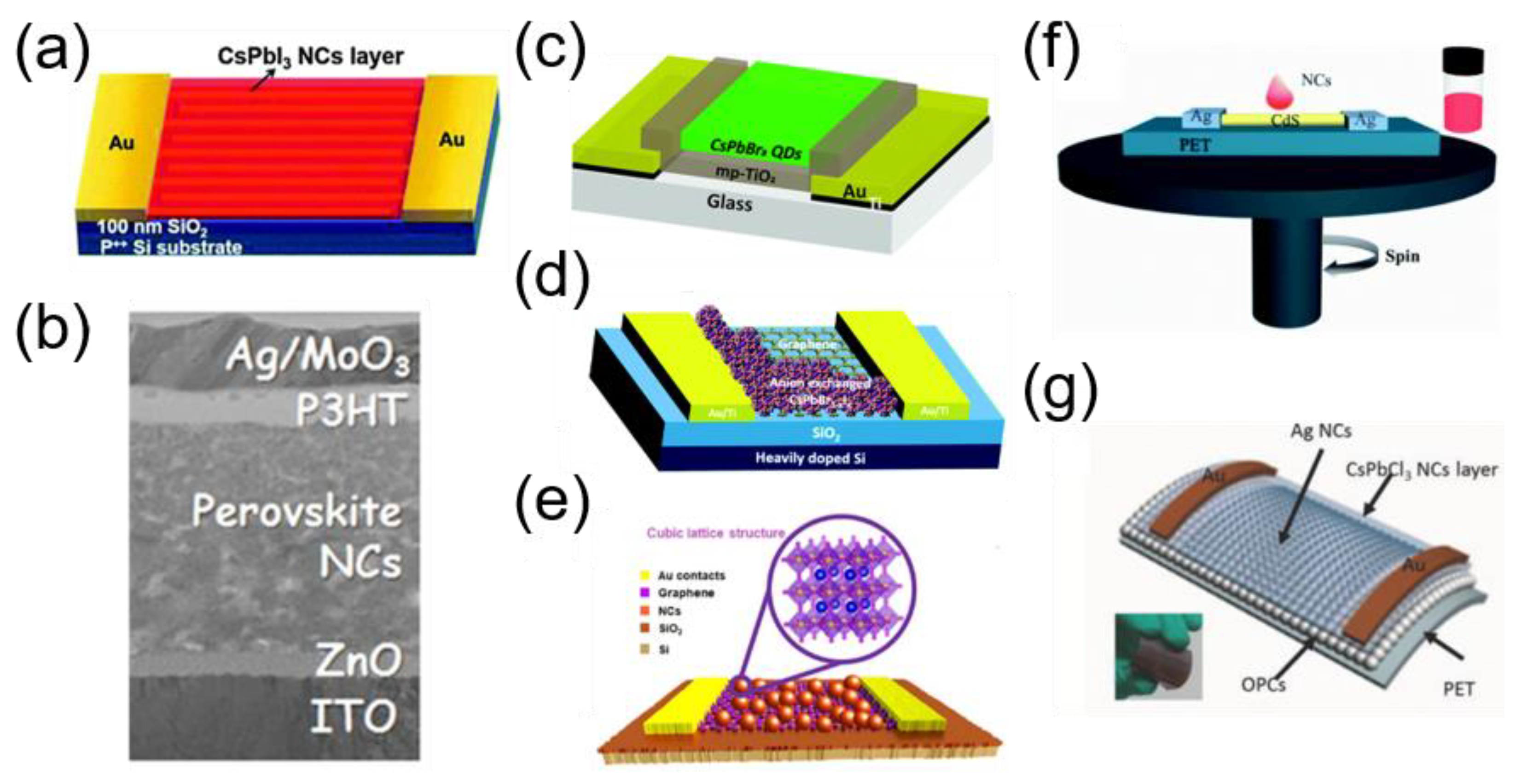
5.2. Structure of Perovskite NC-Based Photodetectors
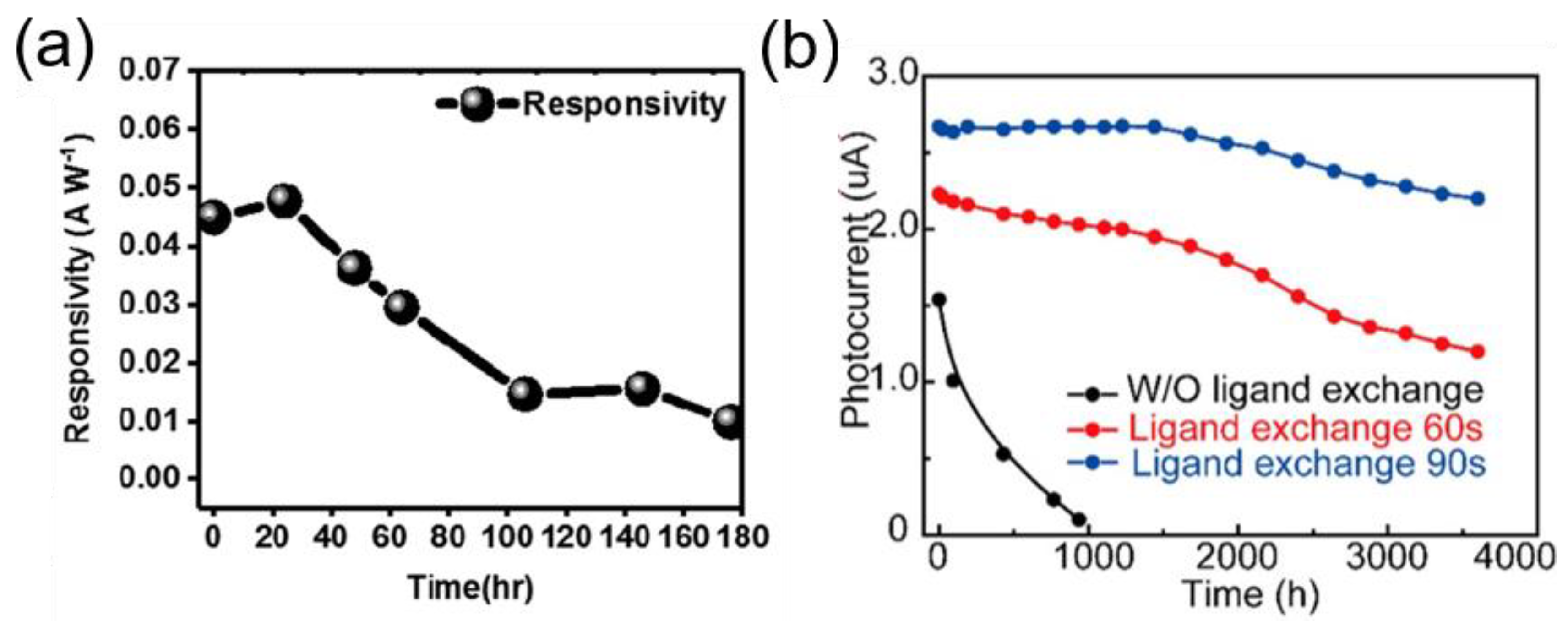
5.3. Strategies to Improve Instability in Ambient
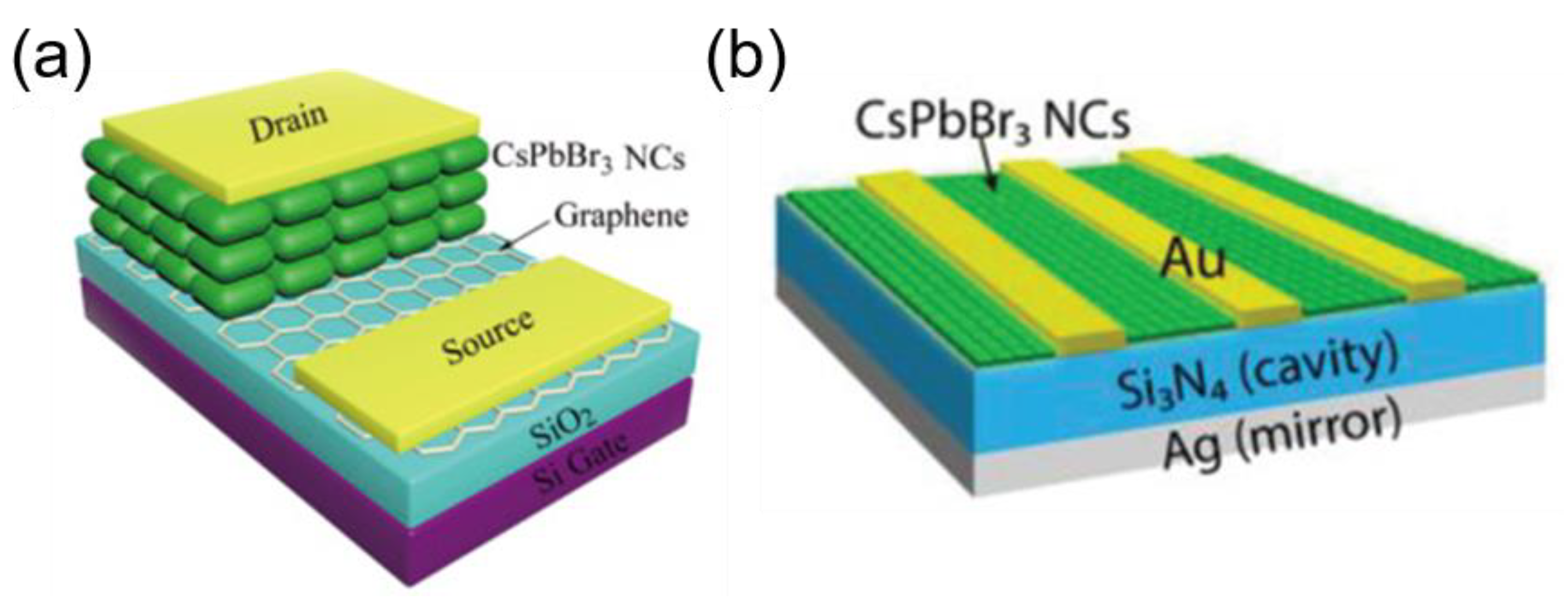
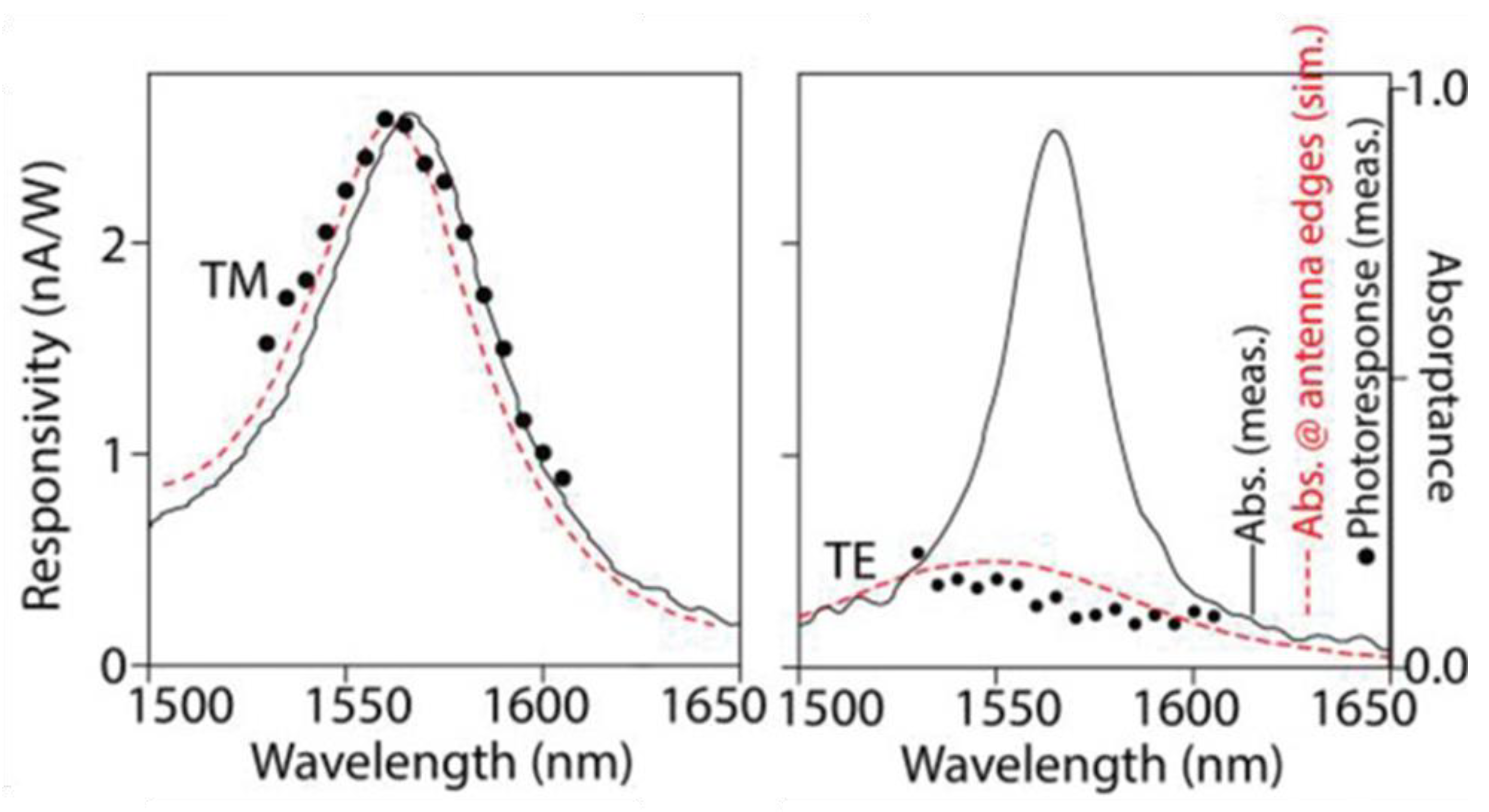
5.4. New Architecture of Perovskite NC-Based Photodetectors
6. Laser
6.1. Introduction of Laser Device
6.2. Application of Perovskite NCs in Laser
6.3. Application of Perovskite NCs in Nonlinear Optics
7. Lead-Free Halide Perovskite Nanocrystal Application
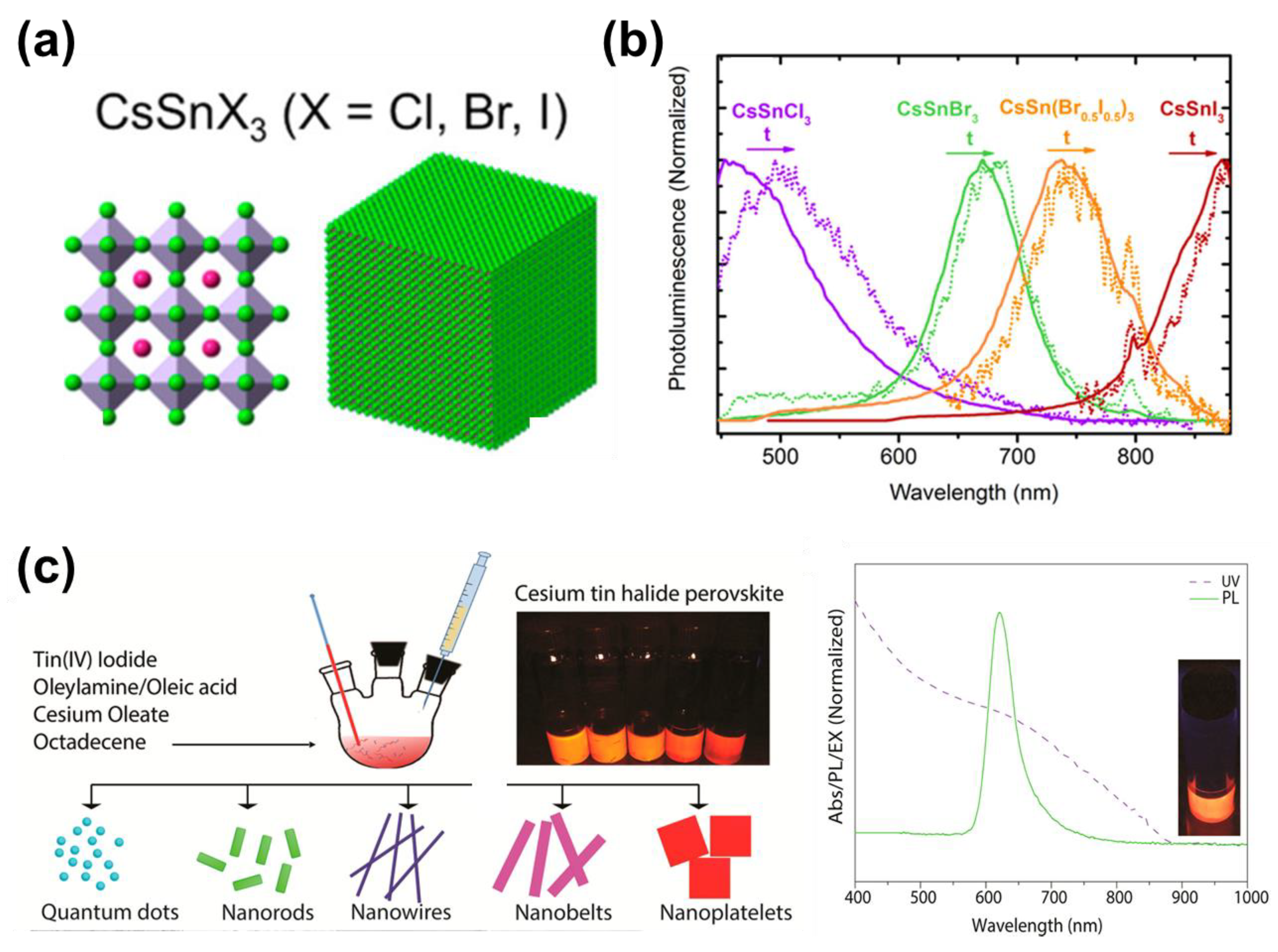
7.1. Lead-Free Halide Perovskite Nanocrystals
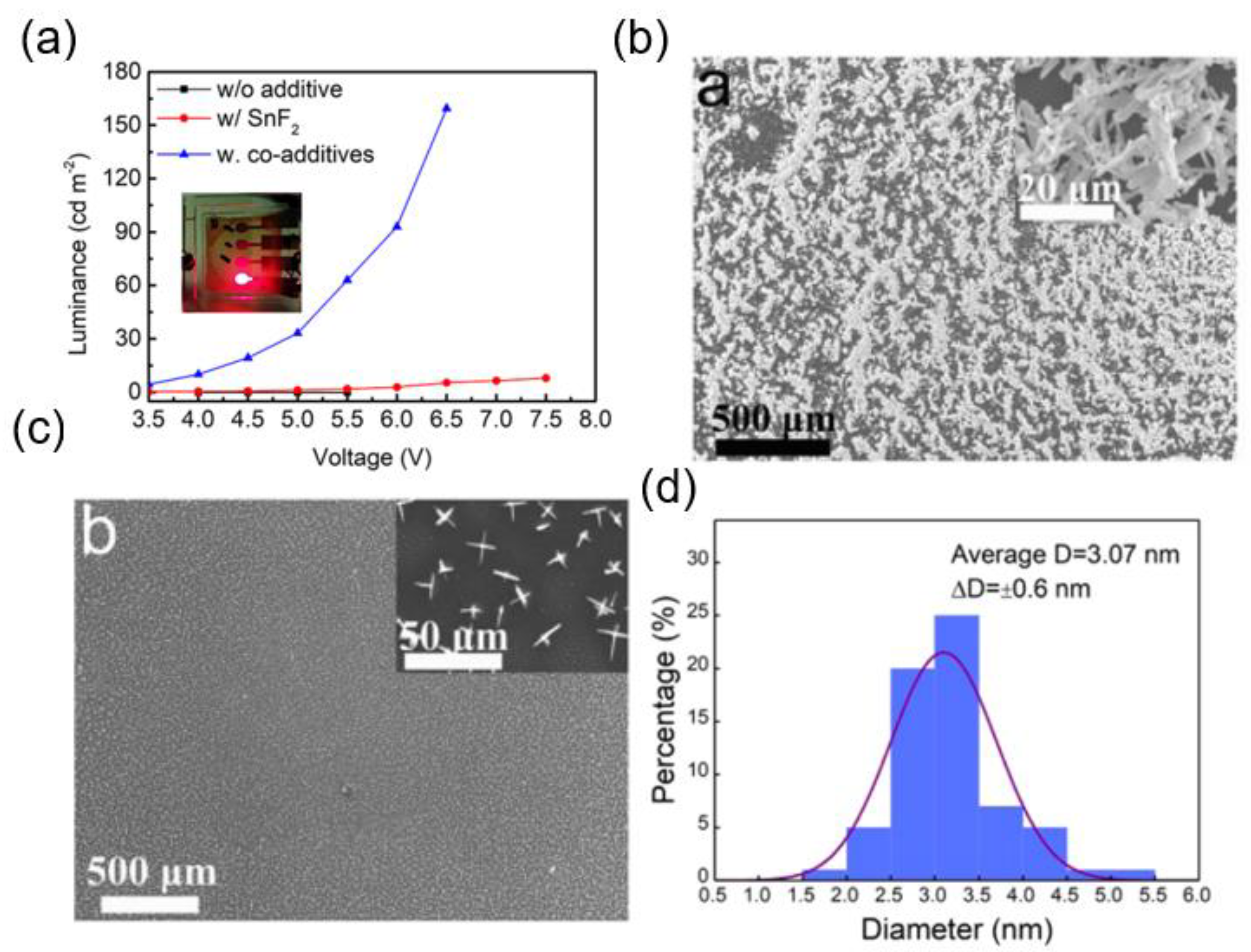
7.2. Lead-Free Perovskite Nanocrystal Light-Emitting Diodes
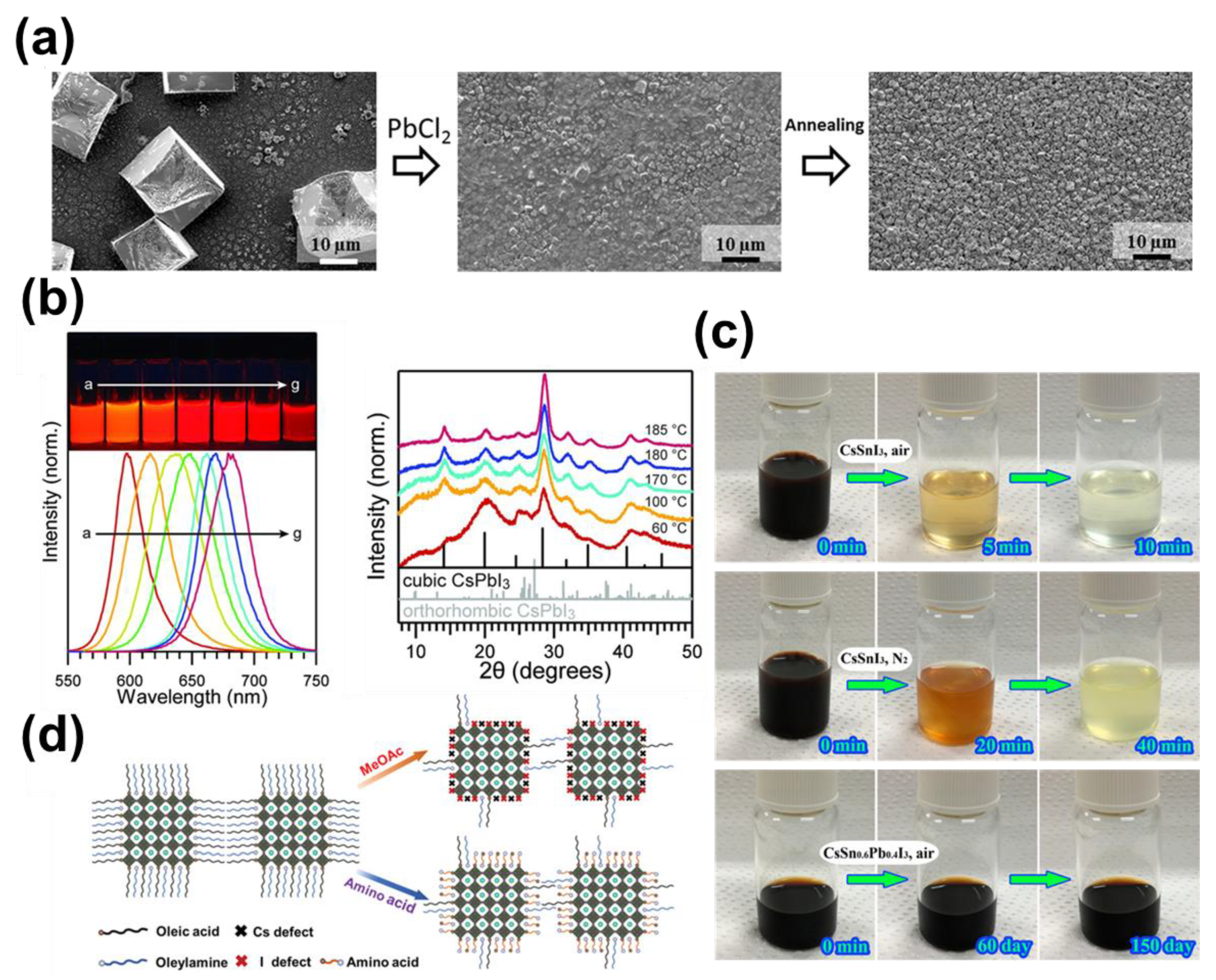
7.3. Lead-Free Perovskite Nanocrystal Solar Cells
8. Conclusions and Outlook
Funding
Conflicts of Interest
References
- Kojima, A.; Teshima, K.; Shirai, Y.; Miyasaka, T. Organometal Halide Perovskites as Visible-Light Sensitizers for Photovoltaic Cells. J. Am. Chem. Soc. 2009, 131, 6050–6051. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Troughton, J.; Gasparini, N.; Lin, Y.; Wei, M.; Hou, Y.; Liu, J.; Song, K.; Chen, Z.; Yang, C.; et al. Quantum Dots Supply Bulk- and Surface-Passivation Agents for Efficient and Stable Perovskite Solar Cells. Joule 2019, 3, 1963–1976. [Google Scholar] [CrossRef]
- Han, D.; Imran, M.; Zhang, M.; Chang, S.; Wu, X.-G.; Zhang, X.; Tang, J.; Wang, M.; Ali, S.; Li, X.; et al. Efficient Light-Emitting Diodes Based on in Situ Fabricated FAPbBr3 Nanocrystals: The Enhancing Role of the Ligand-Assisted Reprecipitation Process. ACS Nano 2018, 12, 8808–8816. [Google Scholar] [CrossRef] [PubMed]
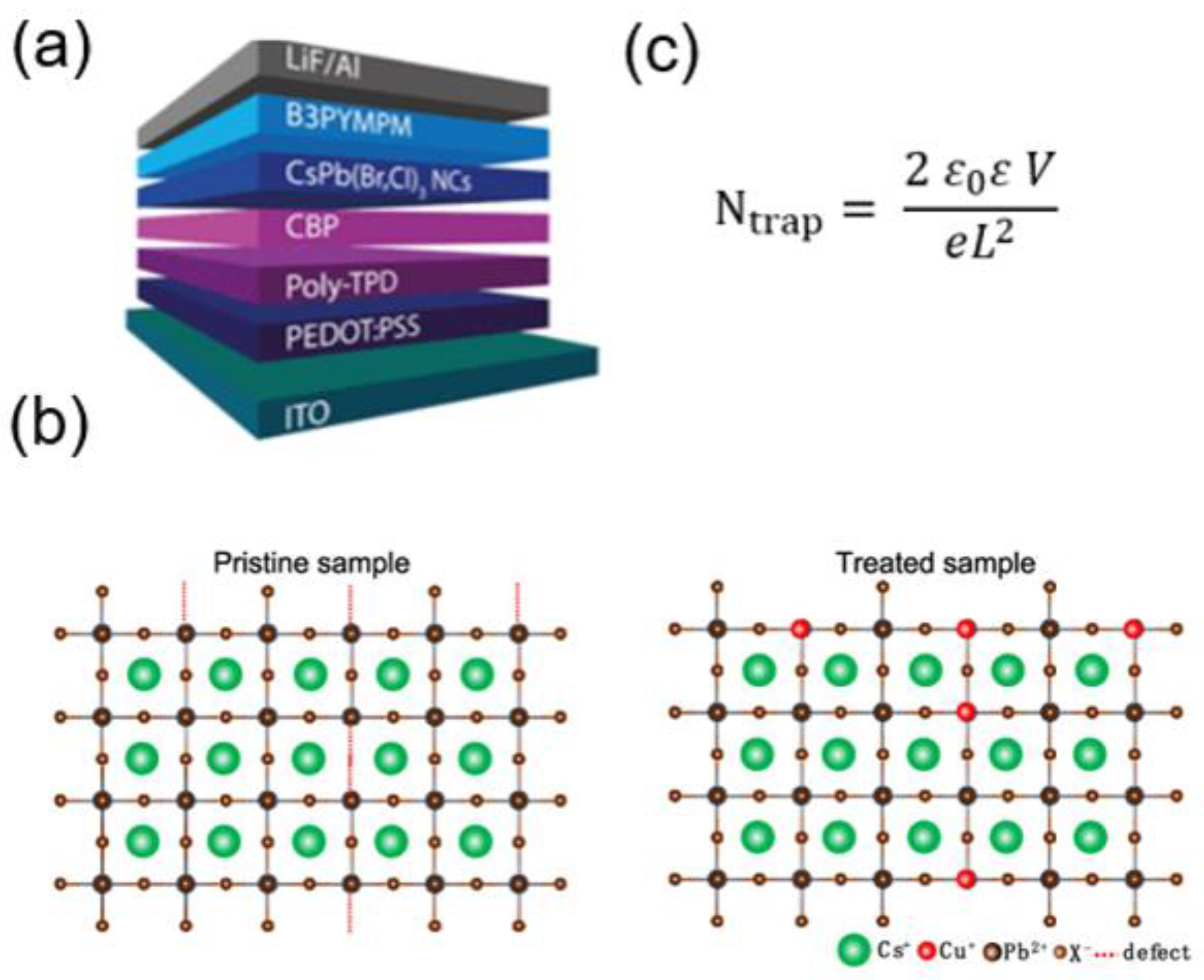
- Gao, L.; Cheng, T.; Wang, Q.; Gou, L.; Zhang, Y.; Zhang, X.; Tan, C.-H.; Zheng, W.; McLachlan, M.A.; Zhang, J. Color-Stable and High-Efficiency Blue Perovskite Nanocrystal Light-Emitting Diodes via Monovalent Copper Ion Lowering Lead Defects. ACS Appl. Mater. Interfaces 2021, 13, 55380–55390. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Zhang, Y.; Kershaw, S.V.; Li, T.; Wang, C.; Zhang, X.; Wang, W.; Li, D.; Wang, Y.; Lu, M.; et al. Zn-Alloyed CsPbI3 Nanocrystals for Highly Efficient Perovskite Light-Emitting Devices. Nano Lett. 2019, 19, 1552–1559. [Google Scholar] [CrossRef]
- Ramasamy, P.; Lim, D.-H.; Kim, B.; Lee, S.-H.; Lee, M.-S.; Lee, J.-S. All-Inorganic Cesium Lead Halide Perovskite Nanocrystals for Photodetector Applications. Chem. Commun. 2016, 52, 2067–2070. [Google Scholar] [CrossRef]
- Gong, M.; Sakidja, R.; Goul, R.; Ewing, D.; Casper, M.; Stramel, A.; Elliot, A.; Wu, J.Z. High-Performance All-Inorganic CsPbCl3 Perovskite Nanocrystal Photodetectors with Superior Stability. ACS Nano 2019, 13, 1772–1783. [Google Scholar] [CrossRef]
- Xing, J.; Liu, X.F.; Zhang, Q.; Ha, S.T.; Yuan, Y.W.; Shen, C.; Sum, T.C.; Xiong, Q. Vapor Phase Synthesis of Organometal Halide Perovskite Nanowires for Tunable Room-Temperature Nanolasers. Nano Lett. 2015, 15, 4571–4577. [Google Scholar] [CrossRef]
- Zhu, H.; Fu, Y.; Meng, F.; Wu, X.; Gong, Z.; Ding, Q.; Gustafsson, M.V.; Trinh, M.T.; Jin, S.; Zhu, X.-Y. Lead Halide Perovskite Nanowire Lasers with Low Lasing Thresholds and High Quality Factors. Nat. Mater. 2015, 14, 636–642. [Google Scholar] [CrossRef]
- Koscher, B.A.; Swabeck, J.K.; Bronstein, N.D.; Alivisatos, A.P. Essentially Trap-Free CsPbBr3 Colloidal Nanocrystals by Postsynthetic Thiocyanate Surface Treatment. J. Am. Chem. Soc. 2017, 139, 6566–6569. [Google Scholar] [CrossRef]
- Manoli, A. Surface Functionalization of CsPbBr3 Νanocrystals for Photonic Applications. ACS Appl. Nano Mater 2021, 4, 5084–5097. [Google Scholar] [CrossRef]
- Bodnarchuk, M.I.; Boehme, S.C.; Ten Brinck, S.; Bernasconi, C.; Shynkarenko, Y.; Krieg, F.; Widmer, R.; Aeschlimann, B.; Günther, D.; Kovalenko, M.V.; et al. Rationalizing and Controlling the Surface Structure and Electronic Passivation of Cesium Lead Halide Nanocrystals. ACS Energy Lett. 2019, 4, 63–74. [Google Scholar] [CrossRef]
- Li, H.; Lin, H.; Ouyang, D.; Yao, C.; Li, C.; Sun, J.; Song, Y.; Wang, Y.; Yan, Y.; Wang, Y.; et al. Efficient and Stable Red Perovskite Light-Emitting Diodes with Operational Stability > 300 h. Adv. Mater. 2021, 33, e2008820. [Google Scholar] [CrossRef]
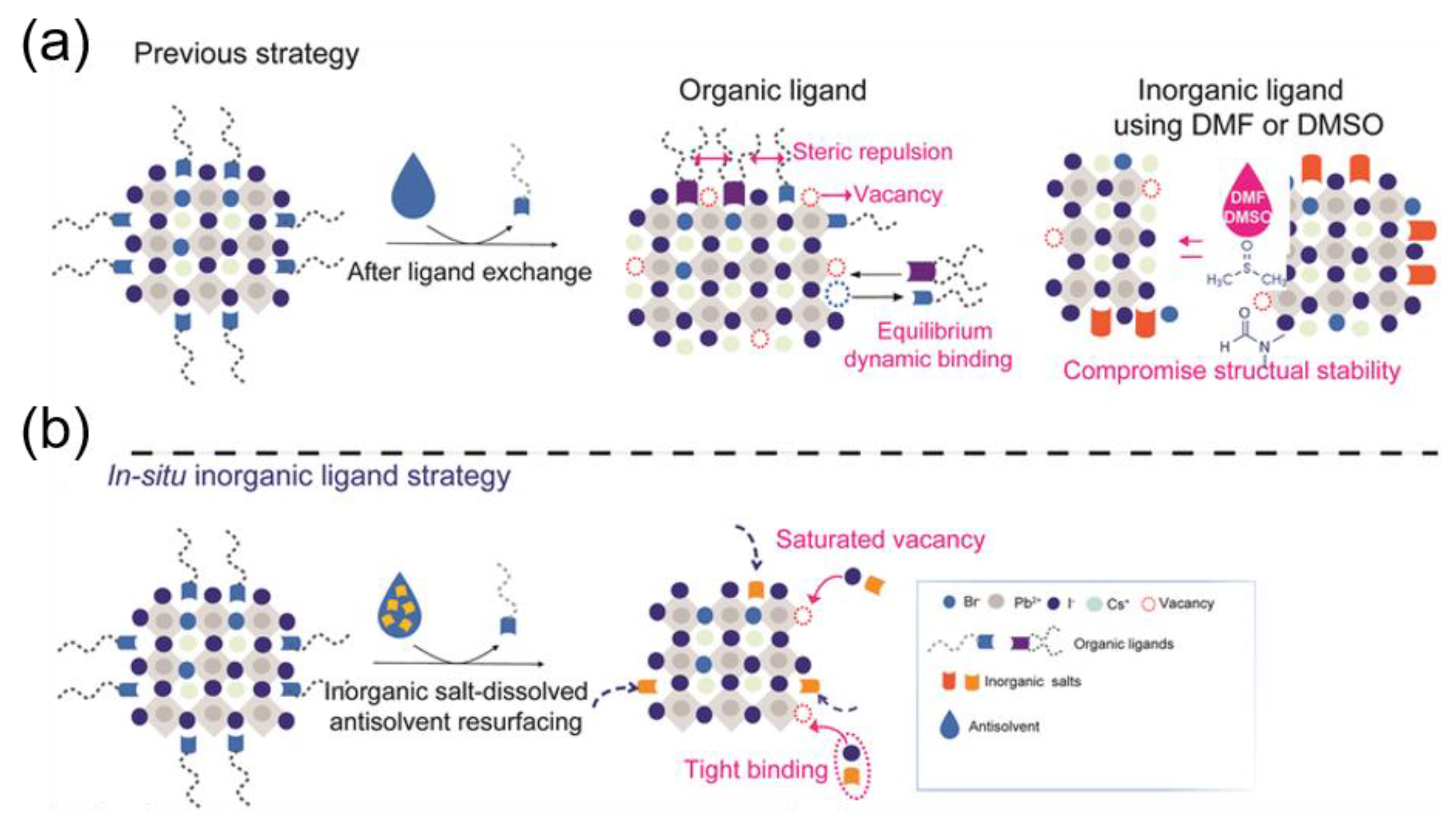
- Wang, Y.-K.; Singh, K.; Li, J.-Y.; Dong, Y.; Wang, X.-Q.; Pina, J.M.; Yu, Y.-J.; Sabatini, R.; Liu, Y.; Ma, D.; et al. In Situ Inorganic Ligand Replenishment Enables Bandgap Stability in Mixed-Halide Perovskite Quantum Dot Solids. Adv. Mater. 2022, 34, e2200854. [Google Scholar] [CrossRef]
- Woo Choi, J.; Woo, H.C.; Huang, X.; Jung, W.-G.; Kim, B.-J.; Jeon, S.-W.; Yim, S.-Y.; Lee, J.-S.; Lee, C.-L. Organic–Inorganic Hybrid Perovskite Quantum Dots with High PLQY and Enhanced Carrier Mobility through Crystallinity Control by Solvent Engineering and Solid-State Ligand Exchange. Nanoscale 2018, 10, 13356–13367. [Google Scholar] [CrossRef]
- Bi, C.; Kershaw, S.V.; Rogach, A.L.; Tian, J. Improved Stability and Photodetector Performance of CsPbI 3 Perovskite Quantum Dots by Ligand Exchange with Aminoethanethiol. Adv. Funct. Mater. 2019, 29, 1902446. [Google Scholar] [CrossRef]
- Suh, Y.-H.; Kim, T.; Choi, J.W.; Lee, C.-L.; Park, J. High-Performance CsPbX3 Perovskite Quantum-Dot Light-Emitting Devices via Solid-State Ligand Exchange. ACS Appl. Nano Mater. 2018, 1, 488–496. [Google Scholar] [CrossRef]
- Vickers, E.T.; Enlow, E.E.; Delmas, W.G.; DiBenedetto, A.C.; Chowdhury, A.H.; Bahrami, B.; Dreskin, B.W.; Graham, T.A.; Hernandez, I.N.; Carter, S.A.; et al. Enhancing Charge Carrier Delocalization in Perovskite Quantum Dot Solids with Energetically Aligned Conjugated Capping Ligands. ACS Energy Lett. 2020, 5, 817–825. [Google Scholar] [CrossRef]
- Kwak, D.-H.; Lim, D.-H.; Ra, H.-S.; Ramasamy, P.; Lee, J.-S. High Performance Hybrid Graphene–CsPbBr3−xIx Perovskite Nanocrystal Photodetector. RSC Adv. 2016, 6, 65252–65256. [Google Scholar] [CrossRef]
- Li, D.; Zhou, D.; Xu, W.; Chen, X.; Pan, G.; Zhou, X.; Ding, N.; Song, H. Plasmonic Photonic Crystals Induced Two-Order Fluorescence Enhancement of Blue Perovskite Nanocrystals and Its Application for High-Performance Flexible Ultraviolet Photodetectors. Adv. Funct. Mater. 2018, 28, 1804429. [Google Scholar] [CrossRef]
- Li, H.; Lin, W.; Ma, L.; Liu, Y.; Wang, Y.; Li, A.; Jin, X.; Xiong, L. High-Performance Broadband Photodetectors Based on All-Inorganic Perovskite CsPb(Br/I)3 Nanocrystal/CdS-Microwire Heterostructures. RSC Adv. 2021, 11, 11663–11671. [Google Scholar] [CrossRef] [PubMed]
- Che, Y.; Cao, X.; Zhang, Y.; Yao, J. High Performance CsPbBr3 Perovskite Nanocrystal Vertical Phototransistor with Graphene Electrode. Opt. Mater. 2020, 100, 109664. [Google Scholar] [CrossRef]
- Hu, Q.; Guo, J.; Lu, M.; Lu, P.; Zhang, Y.; Yu, W.W.; Bai, X. Efficient and Stable Mg2+ -Doped CsPbCl3 Nanocrystals for Violet LEDs. J. Phys. Chem. Lett. 2021, 12, 8203–8211. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, X.; Tan, Y.; Liang, D.; Zou, Y.; Cai, L.; Wu, T.; Wen, K.; Wang, Y.; Li, Y.; et al. Strontium Ion B-site Substitution for Spectral-stable Blue Emitting Perovskite Light-emitting Diodes. Adv. Opt. Mater. 2020, 8, 2001073. [Google Scholar] [CrossRef]
- Tan, C.S. Lead-Free Ultra-Wide Direct Bandgap Perovskite EACaI3. IEEE Trans. Nanotechnol. 2022, 21, 66–70. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Z.; Xu, J.; Dong, Y.; Chen, B.; Park, S.M.; Ma, D.; Lee, S.; Huang, J.E.; Teale, S.; et al. Wide-Bandgap Perovskite Quantum Dots in Perovskite Matrix for Sky-Blue Light-Emitting Diodes. J. Am. Chem. Soc. 2022, 144, 4009–4016. [Google Scholar] [CrossRef]
- Wang, L.; Guo, Q.; Duan, J.; Xie, W.; Ji, G.; Li, S.; Chen, C.; Li, J.; Yang, L.; Tan, Z.; et al. Exploration of Nontoxic Cs3CeBr6 for Violet Light-Emitting Diodes. ACS Energy Lett. 2021, 6, 4245–4254. [Google Scholar] [CrossRef]
- Chiba, T.; Sato, J.; Ishikawa, S.; Takahashi, Y.; Ebe, H.; Sumikoshi, S.; Ohisa, S.; Kido, J. Neodymium Chloride-Doped Perovskite Nanocrystals for Efficient Blue Light-Emitting Devices. ACS Appl. Mater. Interfaces 2020, 12, 53891–53898. [Google Scholar] [CrossRef]
- Luo, J.; Yang, L.; Tan, Z.; Xie, W.; Sun, Q.; Li, J.; Du, P.; Xiao, Q.; Wang, L.; Zhao, X.; et al. Efficient Blue Light Emitting Diodes Based on Europium Halide Perovskites. Adv. Mater. 2021, 33, e2101903. [Google Scholar] [CrossRef]
- Cai, T.; Wang, J.; Li, W.; Hills-Kimball, K.; Yang, H.; Nagaoka, Y.; Yuan, Y.; Zia, R.; Chen, O. Mn2+/Yb3+ Codoped CsPbCl3 Perovskite Nanocrystals with Triple-Wavelength Emission for Luminescent Solar Concentrators. Adv. Sci. 2020, 7, 2001317. [Google Scholar] [CrossRef]
- Cao, F.; Tian, W.; Wang, M.; Wang, M.; Li, L. Stability Enhancement of Lead-free CsSnI 3 Perovskite Photodetector with Reductive Ascorbic Acid Additive. InfoMat 2020, 2, 577–584. [Google Scholar] [CrossRef]
- Tan, C.S. Transition Metal Ions in Methylammonium Chloride Perovskites. ACS Omega 2022, 7, 1412–1419. [Google Scholar] [CrossRef]
- Pan, G.; Bai, X.; Xu, W.; Chen, X.; Zhai, Y.; Zhu, J.; Shao, H.; Ding, N.; Xu, L.; Dong, B.; et al. Bright Blue Light Emission of Ni2+ Ion-Doped CsPbClxBr3-x Perovskite Quantum Dots Enabling Efficient Light-Emitting Devices. ACS Appl. Mater. Interfaces 2020, 12, 14195–14202. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Q.; Liu, X.; Feng, J.; He, L.; Li, H.; Li, C.; Zhang, H. Simultaneous Enhancement of Photoluminescence and Stability of CsPbCl3 Perovskite Enabled by Titanium Ion Dopant. J. Phys. Chem. Lett. 2021, 12, 10746–10752. [Google Scholar] [CrossRef]
- De, A.; Das, S.; Mondal, N.; Samanta, A. Highly Luminescent Violet- and Blue-Emitting Stable Perovskite Nanocrystals. ACS Materials Lett. 2019, 1, 116–122. [Google Scholar] [CrossRef]
- Liu, M.; Zhong, G.; Yin, Y.; Miao, J.; Li, K.; Wang, C.; Xu, X.; Shen, C.; Meng, H. Aluminum-Doped Cesium Lead Bromide Perovskite Nanocrystals with Stable Blue Photoluminescence Used for Display Backlight. Adv. Sci. 2017, 4, 1700335. [Google Scholar] [CrossRef]
- Thapa, S.; Adhikari, G.C.; Zhu, H.; Grigoriev, A.; Zhu, P. Zn-Alloyed All-Inorganic Halide Perovskite-Based White Light-Emitting Diodes with Superior Color Quality. Sci. Rep. 2019, 9, 18636. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, A.-Y.; Yu, J.C.; Nam, Y.S.; Choi, Y.; Park, J.; Song, M.H. Surface Ligand Engineering for Efficient Perovskite Nanocrystal-Based Light-Emitting Diodes. ACS Appl. Mater. Interfaces 2019, 11, 8428–8435. [Google Scholar] [CrossRef]
- Zheng, W.; Wan, Q.; Zhang, Q.; Liu, M.; Zhang, C.; Wang, B.; Kong, L.; Li, L. High-Efficiency Perovskite Nanocrystal Light-Emitting Diodes via Decorating NiOx on the Nanocrystal Surface. Nanoscale 2020, 12, 8711–8719. [Google Scholar] [CrossRef]
- Zhang, F.; Zhong, H.; Chen, C.; Wu, X.-G.; Hu, X.; Huang, H.; Han, J.; Zou, B.; Dong, Y. Brightly Luminescent and Color-Tunable Colloidal CH3NH3PbX3 (X = Br, I, Cl) Quantum Dots: Potential Alternatives for Display Technology. ACS Nano 2015, 9, 4533–4542. [Google Scholar] [CrossRef]
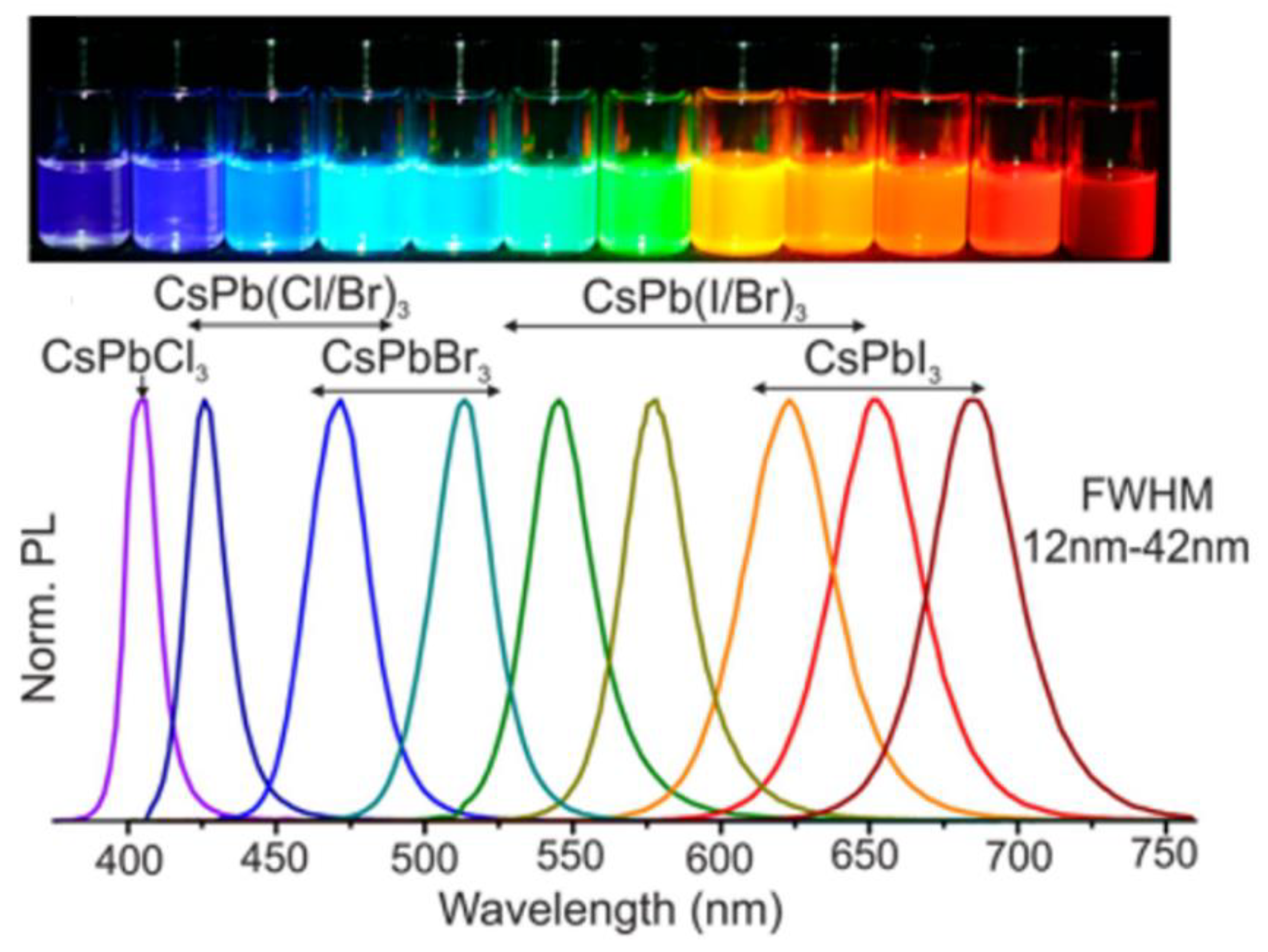
- Nedelcu, G.; Protesescu, L.; Yakunin, S.; Bodnarchuk, M.I.; Grotevent, M.J.; Kovalenko, M.V. Fast Anion-Exchange in Highly Luminescent Nanocrystals of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, I). Nano Lett. 2015, 15, 5635–5640. [Google Scholar] [CrossRef] [PubMed]
- Akkerman, Q.A.; D’Innocenzo, V.; Accornero, S.; Scarpellini, A.; Petrozza, A.; Prato, M.; Manna, L. Tuning the Optical Properties of Cesium Lead Halide Perovskite Nanocrystals by Anion Exchange Reactions. J. Am. Chem. Soc. 2015, 137, 10276–10281. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zou, Y.; Wu, L.; Pan, Q.; Yang, D.; Hu, H.; Tan, Y.; Zhong, Q.; Xu, Y.; Liu, H.; et al. Solvothermal Synthesis of High-Quality All-Inorganic Cesium Lead Halide Perovskite Nanocrystals: From Nanocube to Ultrathin Nanowire. Adv. Funct. Mater. 2017, 27, 1701121. [Google Scholar] [CrossRef]
- Tang, B.; Hu, Y.; Lu, J.; Dong, H.; Mou, N.; Gao, X.; Wang, H.; Jiang, X.; Zhang, L. Energy Transfer and Wavelength Tunable Lasing of Single Perovskite Alloy Nanowire. Nano Energy 2020, 71, 104641. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, Q.; Zhang, C.; Wang, R.; Wu, H.; Zhang, X.; Xing, G.; Yu, W.W.; Wang, X.; Zhang, Y.; et al. Two-Photon-Pumped Perovskite Semiconductor Nanocrystal Lasers. J. Am. Chem. Soc. 2016, 138, 3761–3768. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.-S.; Guo, S.; Makarov, N.S.; Klimov, V.I. Room Temperature Single-Photon Emission from Individual Perovskite Quantum Dots. ACS Nano 2015, 9, 10386–10393. [Google Scholar] [CrossRef]
- Roo, J.D. Highly Dynamic Ligand Binding and Light Absorption Coefficient of Cesium Lead Bromide Perovskite Nanocrystals. ACS Nano 2016, 10, 2071–2081. [Google Scholar] [CrossRef]
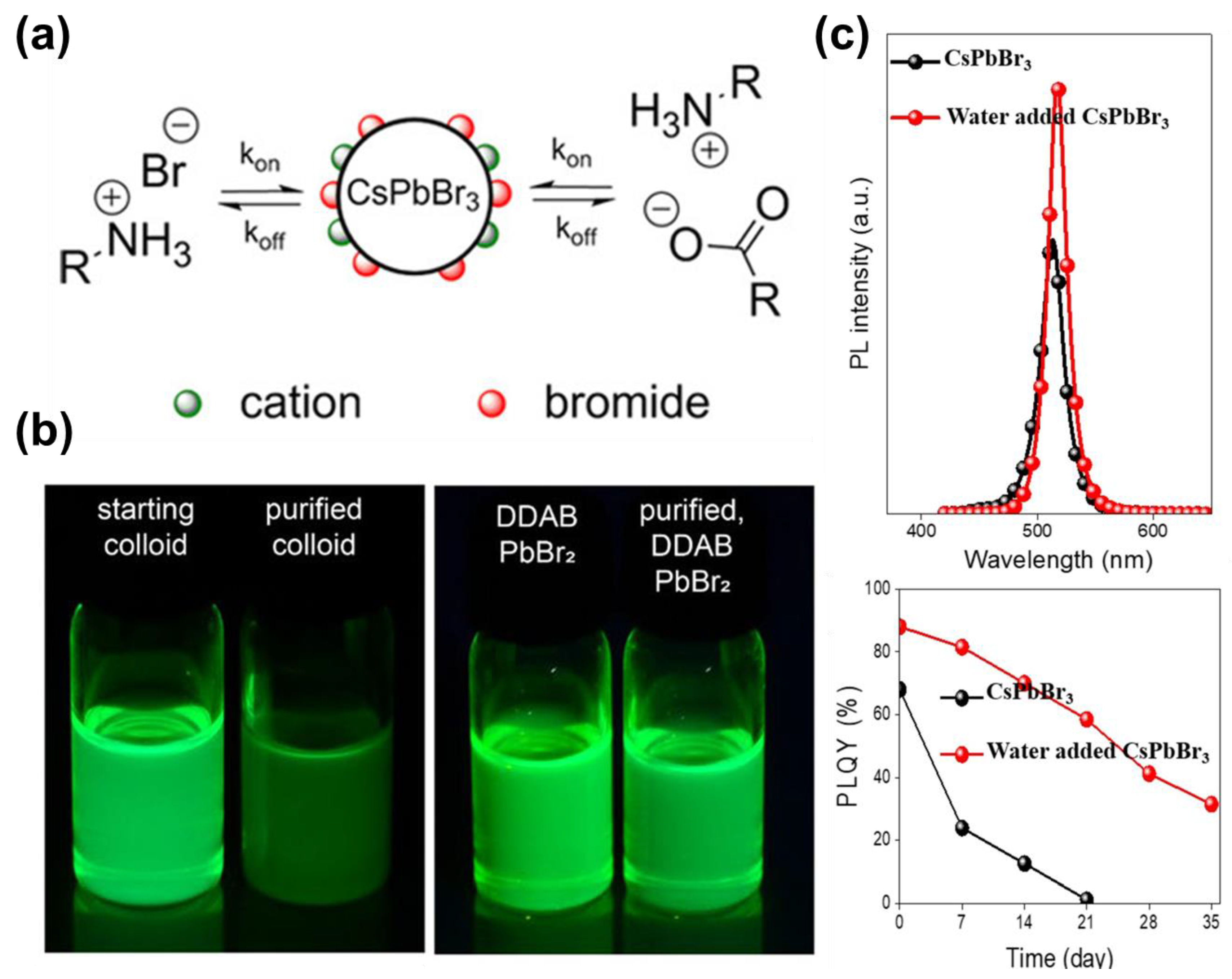
- Kim, M.; Kim, J.H.; Kim, M.; Kim, C.S.; Choi, J.W.; Choi, K.; Lee, J.H.; Park, J.; Kang, Y.-C.; Jin, S.-H.; et al. Enhanced Photoluminescence Quantum Efficiency and Stability of Water Assisted CsPbBr3 Perovskite Nanocrystals. J. Ind. Eng. Chem. 2020, 88, 84–89. [Google Scholar] [CrossRef]
- Protesescu, L.; Yakunin, S.; Bodnarchuk, M.I.; Krieg, F.; Caputo, R.; Hendon, C.H.; Yang, R.X.; Walsh, A.; Kovalenko, M.V. Nanocrystals of Cesium Lead Halide Perovskites (CsPbX₃, X = Cl, Br, and I): Novel Optoelectronic Materials Showing Bright Emission with Wide Color Gamut. Nano Lett. 2015, 15, 3692–3696. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Wolf, C.; Kim, Y.-T.; Cho, H.; Kwon, W.; Do, S.; Sadhanala, A.; Park, C.G.; Rhee, S.-W.; Im, S.H.; et al. Highly Efficient Light-Emitting Diodes of Colloidal Metal-Halide Perovskite Nanocrystals beyond Quantum Size. ACS Nano 2017, 11, 6586–6593. [Google Scholar] [CrossRef]
- Ochsenbein, S.T.; Krieg, F.; Shynkarenko, Y.; Rainò, G.; Kovalenko, M.V. Engineering Color-Stable Blue Light-Emitting Diodes with Lead Halide Perovskite Nanocrystals. ACS Appl. Mater. Interfaces 2019, 11, 21655–21660. [Google Scholar] [CrossRef]
- Lee, J.-W.; Choi, Y.J.; Yang, J.-M.; Ham, S.; Jeon, S.K.; Lee, J.Y.; Song, Y.-H.; Ji, E.K.; Yoon, D.-H.; Seo, S.; et al. In-Situ Formed Type I Nanocrystalline Perovskite Film for Highly Efficient Light-Emitting Diode. ACS Nano 2017, 11, 3311–3319. [Google Scholar] [CrossRef]
- Yan, F.; Xing, J.; Xing, G.; Quan, L.; Tan, S.T.; Zhao, J.; Su, R.; Zhang, L.; Chen, S.; Zhao, Y.; et al. Highly Efficient Visible Colloidal Lead-Halide Perovskite Nanocrystal Light-Emitting Diodes. Nano Lett. 2018, 18, 3157–3164. [Google Scholar] [CrossRef]
- Cho, H.; Jeong, S.-H.; Park, M.-H.; Kim, Y.-H.; Wolf, C.; Lee, C.-L.; Heo, J.H.; Sadhanala, A.; Myoung, N.; Yoo, S.; et al. Overcoming the Electroluminescence Efficiency Limitations of Perovskite Light-Emitting Diodes. Science 2015, 350, 1222–1225. [Google Scholar] [CrossRef]
- Xiao, Z.; Kerner, R.A.; Tran, N.; Zhao, L.; Scholes, G.D.; Rand, B.P. Engineering Perovskite Nanocrystal Surface Termination for Light-emitting Diodes with External Quantum Efficiency Exceeding 15%. Adv. Funct. Mater. 2019, 29, 1807284. [Google Scholar] [CrossRef]
- Cheng, G.; Liu, Y.; Chen, T.; Chen, W.; Fang, Z.; Zhang, J.; Ding, L.; Li, X.; Shi, T.; Xiao, Z. Efficient All-Inorganic Perovskite Light-Emitting Diodes with Improved Operation Stability. ACS Appl. Mater. Interfaces 2020, 12, 18084–18090. [Google Scholar] [CrossRef]
- Zhao, L.; Yeh, Y.-W.; Tran, N.L.; Wu, F.; Xiao, Z.; Kerner, R.A.; Lin, Y.L.; Scholes, G.D.; Yao, N.; Rand, B.P. In Situ Preparation of Metal Halide Perovskite Nanocrystal Thin Films for Improved Light-Emitting Devices. ACS Nano 2017, 11, 3957–3964. [Google Scholar] [CrossRef]
- Im, J.-H.; Lee, C.-R.; Lee, J.-W.; Park, S.-W.; Park, N.-G. 6.5% Efficient Perovskite Quantum-Dot-Sensitized Solar Cell. Nanoscale 2011, 3, 4088–4093. [Google Scholar] [CrossRef]
- Kim, H.-S.; Lee, C.-R.; Im, J.-H.; Lee, K.-B.; Moehl, T.; Marchioro, A.; Moon, S.-J.; Humphry-Baker, R.; Yum, J.-H.; Moser, J.E.; et al. Lead Iodide Perovskite Sensitized All-Solid-State Submicron Thin Film Mesoscopic Solar Cell with Efficiency Exceeding 9%. Sci. Rep. 2012, 2, 591. [Google Scholar] [CrossRef]
- Burschka, J.; Pellet, N.; Moon, S.-J.; Humphry-Baker, R.; Gao, P.; Nazeeruddin, M.K.; Grätzel, M. Sequential Deposition as a Route to High-Performance Perovskite-Sensitized Solar Cells. Nature 2013, 499, 316–319. [Google Scholar] [CrossRef]
- Chen, K.; Jin, W.; Zhang, Y.; Yang, T.; Reiss, P.; Zhong, Q.; Bach, U.; Li, Q.; Wang, Y.; Zhang, H.; et al. High Efficiency Mesoscopic Solar Cells Using CsPbI3 Perovskite Quantum Dots Enabled by Chemical Interface Engineering. J. Am. Chem. Soc. 2020, 142, 3775–3783. [Google Scholar] [CrossRef] [PubMed]
- Akkerman, Q.A.; Gandini, M.; Di Stasio, F.; Rastogi, P.; Palazon, F.; Bertoni, G.; Ball, J.M.; Prato, M.; Petrozza, A.; Manna, L. Strongly Emissive Perovskite Nanocrystal Inks for High-Voltage Solar Cells. Nat. Energy 2017, 2, 16194. [Google Scholar] [CrossRef]
- Yuan, J.; Bi, C.; Wang, S.; Guo, R.; Shen, T.; Zhang, L.; Tian, J. Spray-coated Colloidal Perovskite Quantum Dot Films for Highly Efficient Solar Cells. Adv. Funct. Mater. 2019, 29, 1906615. [Google Scholar] [CrossRef]
- Tidhar, Y.; Edri, E.; Weissman, H.; Zohar, D.; Hodes, G.; Cahen, D.; Rybtchinski, B.; Kirmayer, S. Crystallization of Methyl Ammonium Lead Halide Perovskites: Implications for Photovoltaic Applications. J. Am. Chem. Soc. 2014, 136, 13249–13256. [Google Scholar] [CrossRef] [PubMed]
- Swarnkar, A.; Marshall, A.R.; Sanehira, E.M.; Chernomordik, B.D.; Moore, D.T.; Christians, J.A.; Chakrabarti, T.; Luther, J.M. Quantum Dot-Induced Phase Stabilization of α-CsPbI3 Perovskite for High-Efficiency Photovoltaics. Science 2016, 354, 92–95. [Google Scholar] [CrossRef]
- Liu, F.; Ding, C.; Zhang, Y.; Ripolles, T.S.; Kamisaka, T.; Toyoda, T.; Hayase, S.; Minemoto, T.; Yoshino, K.; Dai, S.; et al. Colloidal Synthesis of Air-Stable Alloyed CsSn1-XPbxI3 Perovskite Nanocrystals for Use in Solar Cells. J. Am. Chem. Soc. 2017, 139, 16708–16719. [Google Scholar] [CrossRef]
- Jia, D.; Chen, J.; Yu, M.; Liu, J.; Johansson, E.M.J.; Hagfeldt, A.; Zhang, X. Dual Passivation of CsPbI3 Perovskite Nanocrystals with Amino Acid Ligands for Efficient Quantum Dot Solar Cells. Small 2020, 16, e2001772. [Google Scholar] [CrossRef]
- Sanehira, E.M.; Marshall, A.R.; Christians, J.A.; Harvey, S.P.; Ciesielski, P.N.; Wheeler, L.M.; Schulz, P.; Lin, L.Y.; Beard, M.C.; Luther, J.M. Enhanced Mobility CsPbI3 Quantum Dot Arrays for Record-Efficiency, High-Voltage Photovoltaic Cells. Sci. Adv. 2017, 3, eaao4204. [Google Scholar] [CrossRef]
- Wheeler, L.M.; Sanehira, E.M.; Marshall, A.R.; Schulz, P.; Suri, M.; Anderson, N.C.; Christians, J.A.; Nordlund, D.; Sokaras, D.; Kroll, T.; et al. Targeted Ligand-Exchange Chemistry on Cesium Lead Halide Perovskite Quantum Dots for High-Efficiency Photovoltaics. J. Am. Chem. Soc. 2018, 140, 10504–10513. [Google Scholar] [CrossRef]
- Wang, Q.; Jin, Z.; Chen, D.; Bai, D.; Bian, H.; Sun, J.; Zhu, G.; Wang, G.; Liu, S.F. M-Graphene Crosslinked CsPbI3 Quantum Dots for High Efficiency Solar Cells with Much Improved Stability. Adv. Energy Mater. 2018, 8, 1800007. [Google Scholar] [CrossRef]
- Chen, K.; Zhong, Q.; Chen, W.; Sang, B.; Wang, Y.; Yang, T.; Liu, Y.; Zhang, Y.; Zhang, H. Short-chain Ligand-passivated Stable α-CsPbI3 Quantum Dot for All-inorganic Perovskite Solar Cells. Adv. Funct. Mater. 2019, 29, 1900991. [Google Scholar] [CrossRef]
- Cha, M.; Da, P.; Wang, J.; Wang, W.; Chen, Z.; Xiu, F.; Zheng, G.; Wang, Z.-S. Enhancing Perovskite Solar Cell Performance by Interface Engineering Using CH3NH3PbBr0.9I2.1 Quantum Dots. J. Am. Chem. Soc. 2016, 138, 8581–8587. [Google Scholar] [CrossRef]
- Liu, C.; Hu, M.; Zhou, X.; Wu, J.; Zhang, L.; Kong, W.; Li, X.; Zhao, X.; Dai, S.; Xu, B.; et al. Efficiency and Stability Enhancement of Perovskite Solar Cells by Introducing CsPbI3 Quantum Dots as an Interface Engineering Layer. NPG Asia Mater. 2018, 10, 552–561. [Google Scholar] [CrossRef]
- Zai, H.; Zhu, C.; Xie, H.; Zhao, Y.; Shi, C.; Chen, Z.; Ke, X.; Sui, M.; Chen, C.; Hu, J.; et al. Congeneric Incorporation of CsPbBr3 Nanocrystals in a Hybrid Perovskite Heterojunction for Photovoltaic Efficiency Enhancement. ACS Energy Lett. 2018, 3, 30–38. [Google Scholar] [CrossRef]
- Yang, W.; Su, R.; Luo, D.; Hu, Q.; Zhang, F.; Xu, Z.; Wang, Z.; Tang, J.; Lv, Z.; Yang, X.; et al. Surface Modification Induced by Perovskite Quantum Dots for Triple-Cation Perovskite Solar Cells. Nano Energy 2020, 67, 104189. [Google Scholar] [CrossRef]
- Sim, K.M.; Swarnkar, A.; Nag, A.; Chung, D.S. Phase Stabilized α-CsPbI3 Perovskite Nanocrystals for Photodiode Applications. Laser Photon. Rev. 2018, 12, 1700209. [Google Scholar] [CrossRef]
- Jansen-van Vuuren, R.D.; Armin, A.; Pandey, A.K.; Burn, P.L.; Meredith, P. Organic Photodiodes: The Future of Full Color Detection and Image Sensing. Adv. Mater. 2016, 28, 4766–4802. [Google Scholar] [CrossRef]
- Pierre, A.; Deckman, I.; Lechêne, P.B.; Arias, A.C. High Detectivity All-Printed Organic Photodiodes. Adv. Mater. 2015, 27, 6411–6417. [Google Scholar] [CrossRef]
- Konstantatos, G.; Clifford, J.; Levina, L.; Sargent, E.H. Sensitive Solution-Processed Visible-Wavelength Photodetectors. Nat. Photonics 2007, 1, 531–534. [Google Scholar] [CrossRef]
- Zhou, L.; Yu, K.; Yang, F.; Zheng, J.; Zuo, Y.; Li, C.; Cheng, B.; Wang, Q. All-Inorganic Perovskite Quantum Dot/Mesoporous TiO2 Composite-Based Photodetectors with Enhanced Performance. Dalton Trans. 2017, 46, 1766–1769. [Google Scholar] [CrossRef]
- Surendran, A.; Yu, X.; Begum, R.; Tao, Y.; Wang, Q.J.; Leong, W.L. All Inorganic Mixed Halide Perovskite Nanocrystal-Graphene Hybrid Photodetector: From Ultrahigh Gain to Photostability. ACS Appl. Mater. Interfaces 2019, 11, 27064–27072. [Google Scholar] [CrossRef] [PubMed]
- Talapin, D.V.; Lee, J.-S.; Kovalenko, M.V.; Shevchenko, E.V. Prospects of Colloidal Nanocrystals for Electronic and Optoelectronic Applications. Chem. Rev. 2010, 110, 389–458. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Tan, Z.; Wu, C.; Wang, X.; Mohammad, M.A.; Xie, D.; Yang, Y.; Wang, J.; Li, L.-J.; Xu, J.; et al. Novel Field-Effect Schottky Barrier Transistors Based on Graphene-MoS2 Heterojunctions. Sci. Rep. 2014, 4, 5951. [Google Scholar] [CrossRef]
- Wang, Z.; Lin, C.-C.; Ho, Y.-L.; Xiang, R.; Maruyama, S.; Chen, C.-W.; Delaunay, J.-J. Self-patterned CsPbBr 3 Nanocrystal Based Plasmonic Hot-carrier Photodetector at Telecommunications Wavelengths. Adv. Opt. Mater. 2021, 9, 2101474. [Google Scholar] [CrossRef]
- Gao, W.; Yu, S.F. Reality or Fantasy—Perovskite Semiconductor Laser Diodes. EcoMat 2021, 3, e12077. [Google Scholar] [CrossRef]
- Chen, J.; Du, W.; Shi, J.; Li, M.; Wang, Y.; Zhang, Q.; Liu, X. Perovskite Quantum Dot Lasers. InfoMat 2020, 2, 170–183. [Google Scholar] [CrossRef]
- Wang, Y.; Zhi, M.; Chang, Y.-Q.; Zhang, J.-P.; Chan, Y. Stable, Ultralow Threshold Amplified Spontaneous Emission from CsPbBr3 Nanoparticles Exhibiting Trion Gain. Nano Lett. 2018, 18, 4976–4984. [Google Scholar] [CrossRef]
- Protesescu, L.; Yakunin, S.; Bodnarchuk, M.I.; Bertolotti, F.; Masciocchi, N.; Guagliardi, A.; Kovalenko, M.V. Monodisperse Formamidinium Lead Bromide Nanocrystals with Bright and Stable Green Photoluminescence. J. Am. Chem. Soc. 2016, 138, 14202–14205. [Google Scholar] [CrossRef]
- Hu, Z.; Liu, Z.; Zhan, Z.; Shi, T.; Du, J.; Tang, X.; Leng, Y. Advances in Metal Halide Perovskite Lasers: Synthetic Strategies, Morphology Control, and Lasing Emission. Adv. Photon. 2021, 3, 034002. [Google Scholar] [CrossRef]
- Huang, C.; Wang, K.; Yang, Z.; Jiang, L.; Liu, R.; Su, R.; Zhou, Z.-K.; Wang, X. Up-Conversion Perovskite Nanolaser with Single Mode and Low Threshold. J. Phys. Chem. C Nanomater. Interfaces 2017, 121, 10071–10077. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Nalla, V.; Zeng, H.; Sun, H. Solution-Processed Low Threshold Vertical Cavity Surface Emitting Lasers from All-Inorganic Perovskite Nanocrystals. Adv. Funct. Mater. 2017, 27, 1605088. [Google Scholar] [CrossRef]
- Yuan, S.; Chen, D.; Li, X.; Zhong, J.; Xu, X. In Situ Crystallization Synthesis of CsPbBr3 Perovskite Quantum Dot-Embedded Glasses with Improved Stability for Solid-State Lighting and Random Upconverted Lasing. ACS Appl. Mater. Interfaces 2018, 10, 18918–18926. [Google Scholar] [CrossRef]
- Eaton, S.W.; Lai, M.; Gibson, N.A.; Wong, A.B.; Dou, L.; Ma, J.; Wang, L.-W.; Leone, S.R.; Yang, P. Lasing in Robust Cesium Lead Halide Perovskite Nanowires. Proc. Natl. Acad. Sci. USA 2016, 113, 1993–1998. [Google Scholar] [CrossRef]
- Fu, Y.; Zhu, H.; Stoumpos, C.C.; Ding, Q.; Wang, J.; Kanatzidis, M.G.; Zhu, X.; Jin, S. Broad Wavelength Tunable Robust Lasing from Single-Crystal Nanowires of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, I). ACS Nano 2016, 10, 7963–7972. [Google Scholar] [CrossRef]
- Xu, J.; Semin, S.; Rasing, T.; Rowan, A.E. Organized Chromophoric Assemblies for Nonlinear Optical Materials: Towards (Sub)Wavelength Scale Architectures. Small 2015, 11, 1113–1129. [Google Scholar] [CrossRef]
- Walters, G.; Sutherland, B.R.; Hoogland, S.; Shi, D.; Comin, R.; Sellan, D.P.; Bakr, O.M.; Sargent, E.H. Two-Photon Absorption in Organometallic Bromide Perovskites. ACS Nano 2015, 9, 9340–9346. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Zhao, X.; Xiao, L.; Zeng, H.; Sun, H. Nonlinear Absorption and Low-Threshold Multiphoton Pumped Stimulated Emission from All-Inorganic Perovskite Nanocrystals. Nano Lett. 2016, 16, 448–453. [Google Scholar] [CrossRef]
- Amendola, V.; Fortunati, I.; Marega, C.; Abdelhady, A.L.; Saidaminov, M.I.; Bakr, O.M. High-Purity Hybrid Organolead Halide Perovskite Nanoparticles Obtained by Pulsed-Laser Irradiation in Liquid. Chemphyschem 2017, 18, 1047–1054. [Google Scholar] [CrossRef]
- Jellicoe, T.C.; Richter, J.M.; Glass, H.F.J.; Tabachnyk, M.; Brady, R.; Dutton, S.E.; Rao, A.; Friend, R.H.; Credgington, D.; Greenham, N.C.; et al. Synthesis and Optical Properties of Lead-Free Cesium Tin Halide Perovskite Nanocrystals. J. Am. Chem. Soc. 2016, 138, 2941–2944. [Google Scholar] [CrossRef]
- Wang, A.; Yan, X.; Zhang, M.; Sun, S.; Yang, M.; Shen, W.; Pan, X.; Wang, P.; Deng, Z. Controlled Synthesis of Lead-Free and Stable Perovskite Derivative Cs2SnI6 Nanocrystals via a Facile Hot-Injection Process. Chem. Mater. 2016, 28, 8132–8140. [Google Scholar] [CrossRef]
- Dey, A.; Ye, J.; De, A.; Debroye, E.; Ha, S.K.; Bladt, E.; Kshirsagar, A.S.; Wang, Z.; Yin, J.; Wang, Y.; et al. State of the Art and Prospects for Halide Perovskite Nanocrystals. ACS Nano 2021, 15, 10775–10981. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, W.; Zhou, D.; Ma, X.; Chen, X.; Pan, G.; Zhu, J.; Ji, Y.; Ding, N.; Song, H. Cesium Tin Halide Perovskite Quantum Dots as an Organic Photoluminescence Probe for Lead Ion. J. Lumin. 2019, 216, 116711. [Google Scholar] [CrossRef]
- Mu, H.; Hu, F.; Wang, R.; Jia, J.; Xiao, S. Effects of In-Situ Annealing on the Electroluminescence Performance of the Sn-Based Perovskite Light-Emitting Diodes Prepared by Thermal Evaporation. J. Lumin. 2020, 226, 117493. [Google Scholar] [CrossRef]
- Heo, J.-M.; Cho, H.; Lee, S.-C.; Park, M.-H.; Kim, J.S.; Kim, H.; Park, J.; Kim, Y.-H.; Yun, H.J.; Yoon, E.; et al. Bright Lead-Free Inorganic CsSnBr3 Perovskite Light-Emitting Diodes. ACS Energy Lett. 2022, 7, 2807–2815. [Google Scholar] [CrossRef]
- Li, T.; Mo, X.; Peng, C.; Lu, Q.; Qi, C.; Tao, X.; Ouyang, Y.; Zhou, Y. Distinct Green Electroluminescence from Lead-Free CsCuBr2 Halide Micro-Crosses. Chem. Commun. 2019, 55, 4554–4557. [Google Scholar] [CrossRef]
- Jun, T.; Sim, K.; Iimura, S.; Sasase, M.; Kamioka, H.; Kim, J.; Hosono, H. Lead-Free Highly Efficient Blue-Emitting Cs3Cu2I5 with 0D Electronic Structure. Adv. Mater. 2018, 30, e1804547. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, Y.; Deng, H.; Farooq, U.; Yang, X.; Khan, J.; Tang, J.; Song, H. High Quantum Yield Blue Emission from Lead-Free Inorganic Antimony Halide Perovskite Colloidal Quantum Dots. ACS Nano 2017, 11, 9294–9302. [Google Scholar] [CrossRef]
- Wang, M.; Wang, W.; Ma, B.; Shen, W.; Liu, L.; Cao, K.; Chen, S.; Huang, W. Lead-Free Perovskite Materials for Solar Cells. Nanomicro Lett. 2021, 13, 62. [Google Scholar] [CrossRef]
- Wang, Y.; Tu, J.; Li, T.; Tao, C.; Deng, X.; Li, Z. Convenient Preparation of CsSnI3 Quantum Dots, Excellent Stability, and the Highest Performance of Lead-Free Inorganic Perovskite Solar Cells so Far. J. Mater. Chem. A Mater. Energy Sustain. 2019, 7, 7683–7690. [Google Scholar] [CrossRef]
- Xu, H.; Yuan, H.; Duan, J.; Zhao, Y.; Jiao, Z.; Tang, Q. Lead-Free CH3NH3SnBr3-XIx Perovskite Quantum Dots for Mesoscopic Solar Cell Applications. Electrochim. Acta 2018, 282, 807–812. [Google Scholar] [CrossRef]
- Schileo, G.; Grancini, G. Lead or No Lead? Availability, Toxicity, Sustainability and Environmental Impact of Lead-Free Perovskite Solar Cells. J. Mater. Chem. C Mater. Opt. Electron. Devices 2021, 9, 67–76. [Google Scholar] [CrossRef]
- Yuan, S.; Zheng, X.; Shen, W.-S.; Liu, J.; Cui, L.-S.; Zhang, C.; Tian, Q.-S.; Wu, J.-J.; Zhou, Y.-H.; Wang, X.-D.; et al. Overcoming Degradation Pathways to Achieve Stable Blue Perovskite Light-Emitting Diodes. ACS Energy Lett. 2022, 7, 1348–1354. [Google Scholar] [CrossRef]




| Ref | Ligand | Result | Perovskite | PLQY (%) |
|---|---|---|---|---|
| [14] | KI inorganic ligand | The mobility increased | CsPb(IxBr1−x)3 NC | 95% |
| [15] | Hexyl amine ligand | Hole mobility increased | CH3NH3PbBr3 QDs | 82% |
| [15] | Octyl amine ligand | Hole mobility increased | CH3NH3PbBr3 QDs | 78% |
| [16] | 2-aminoethanethiol (AET) | Higher electron mobility | CsPbI3 QDs | 51% |
| [17] | aromatic acid/amine | Dense and uniform thin film | CsPbBr3 QDs | N/A |
| [18] | DPPA + TCA | faster charge transport time | MAPbBr3 QDs | 90% |
| [18] | DPPA + TFCA | faster charge transport time | MAPbBr3 QDs | 46% |
| Ref | Perovskite | Modified | EL Peak | EQE (%) |
|---|---|---|---|---|
| [4] | CsPbClBr2 | Add Cu+ | 479 nm | 3.78% |
| [51] | CsPb(Br/Cl)3 | Adding Br and Cl in different ratios | 463 nm | 1.2% |
| Ref | Perovskite | Modified | EL Peak | EQE (%) |
|---|---|---|---|---|
| [3] | FAPbBr3 | Add DPPA-Br as a ligand | 528 nm | 16.3% |
| [38] | CsPbBr3 | Change ligand (QABs) | 513 nm | 9.71% |
| [52] | MAPbBr3 | Change ratio MABr and PbBr2 | 540 nm | 8.21% |
| [53] | MAPbBr3 | Two layers ETL | 525 nm | 12.9% |
| Ref | Perovskite | Modified | EL Peak | EQE (%) |
|---|---|---|---|---|
| [5] | CsPb0.64Zn0.36I3 | Add Zn2+ | 668 nm | 15.1% |
| [56] | CsPbI3 | Add 4-F-PMAI | 692 nm | 14.8% |
| [57] | MAPbI3 | Add 4-F-PMAI | 749 nm | 7.9% |
| Ref | Rise Time (s) | Decay Time (s) |
|---|---|---|
| [7] | 0.3 | 0.35 |
| [19] | 0.81 | 3.65 |
| [22] | 0.02 | 0.036 |
| [81] | 0.2736 | 2.26 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.P.; Li, H.C.; Huang, Y.C.; Tan, C.S. Synthesis and Applications of Halide Perovskite Nanocrystals in Optoelectronics. Inorganics 2023, 11, 39. https://doi.org/10.3390/inorganics11010039
Wang YP, Li HC, Huang YC, Tan CS. Synthesis and Applications of Halide Perovskite Nanocrystals in Optoelectronics. Inorganics. 2023; 11(1):39. https://doi.org/10.3390/inorganics11010039
Chicago/Turabian StyleWang, Yen Po, Hsin Chieh Li, Yan Chi Huang, and Chih Shan Tan. 2023. "Synthesis and Applications of Halide Perovskite Nanocrystals in Optoelectronics" Inorganics 11, no. 1: 39. https://doi.org/10.3390/inorganics11010039
APA StyleWang, Y. P., Li, H. C., Huang, Y. C., & Tan, C. S. (2023). Synthesis and Applications of Halide Perovskite Nanocrystals in Optoelectronics. Inorganics, 11(1), 39. https://doi.org/10.3390/inorganics11010039







