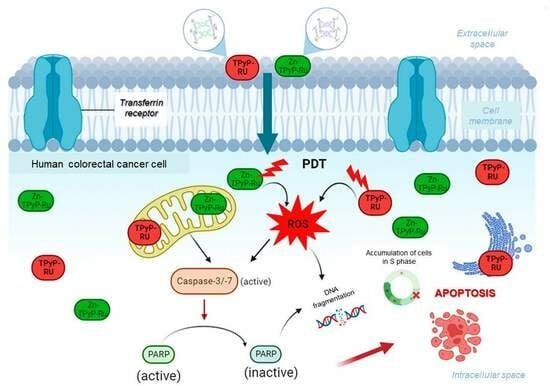
A Combination of Ruthenium Complexes and Photosensitizers to Treat Colorectal Cancer
Abstract
:1. Introduction
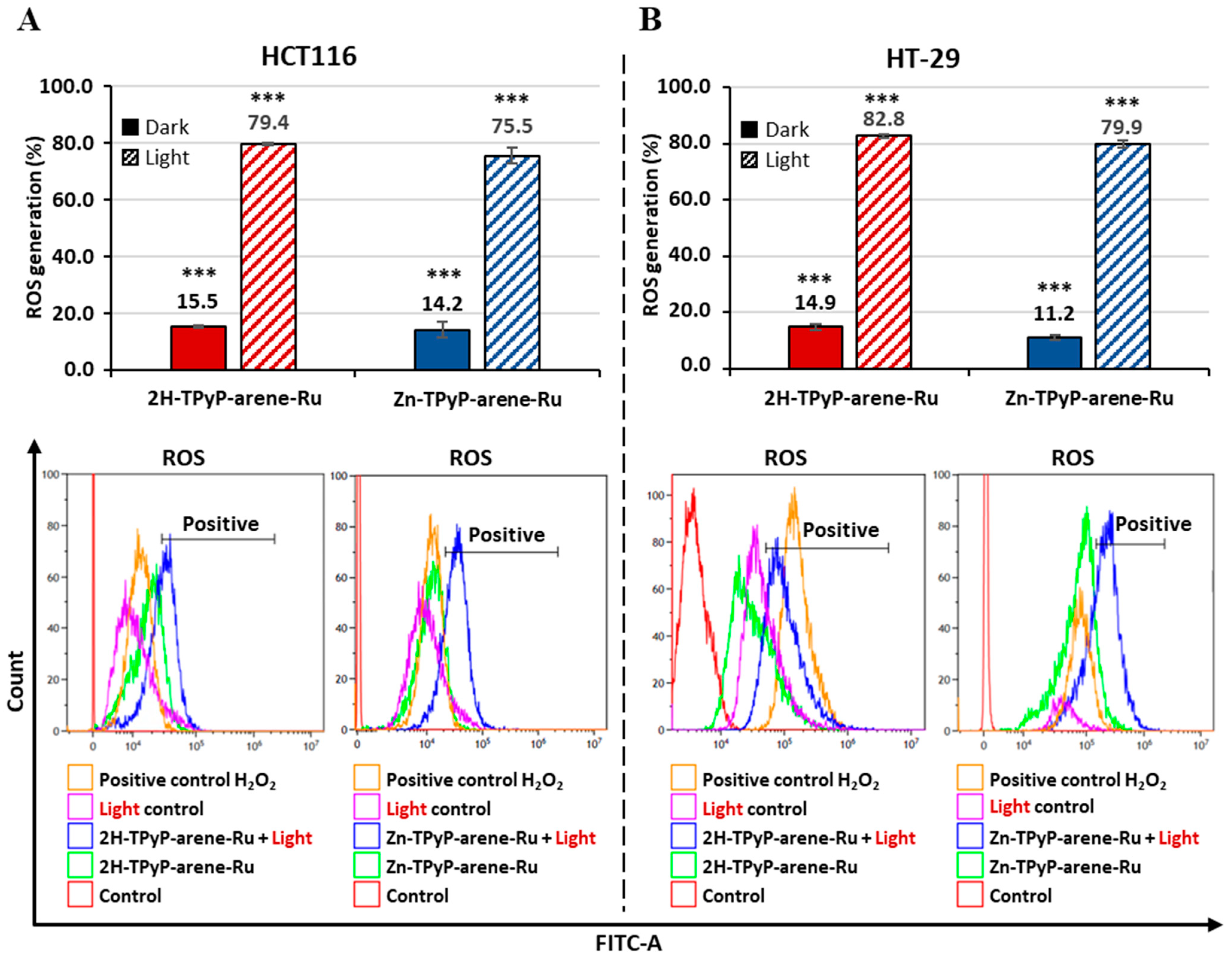
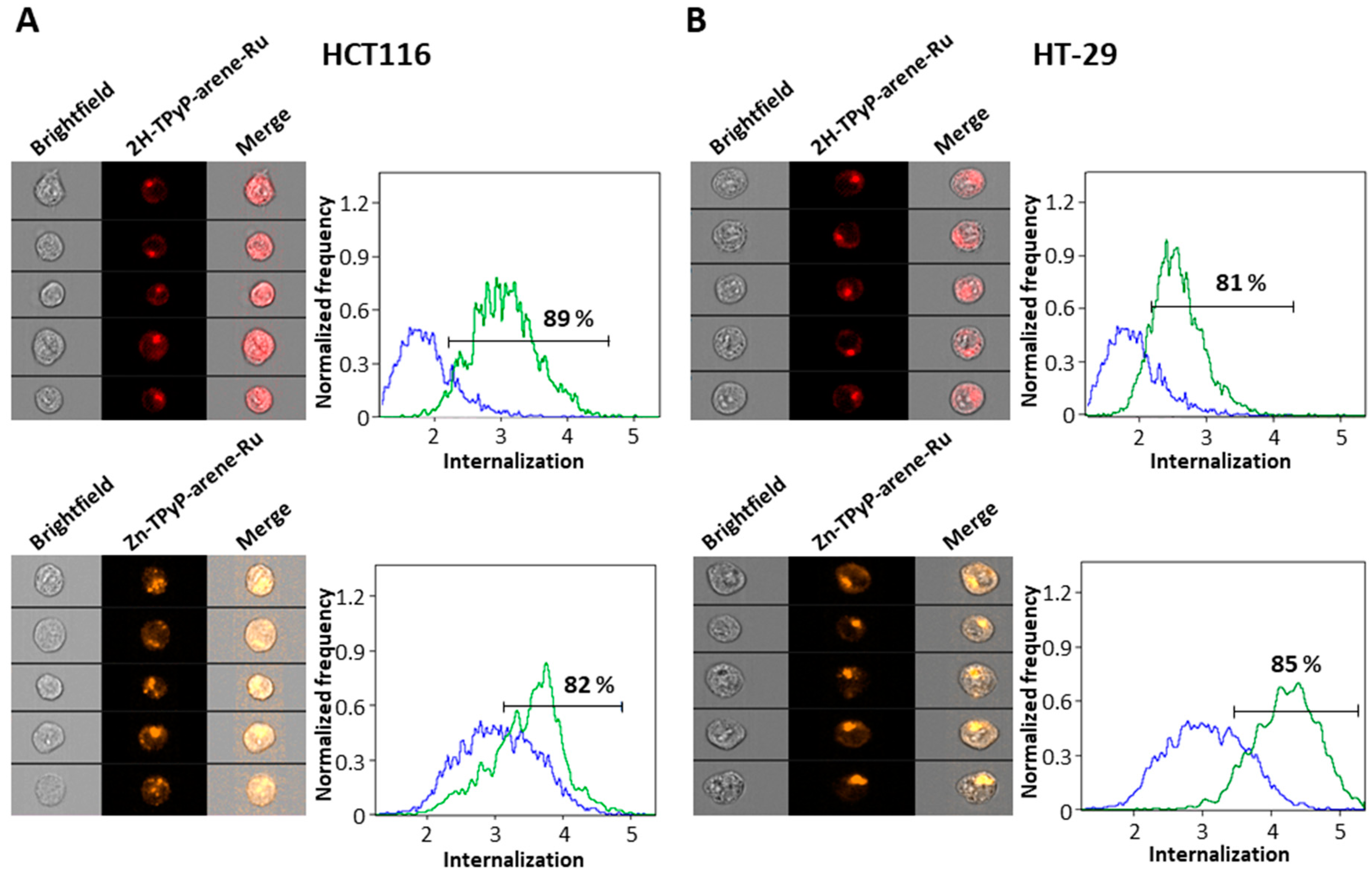
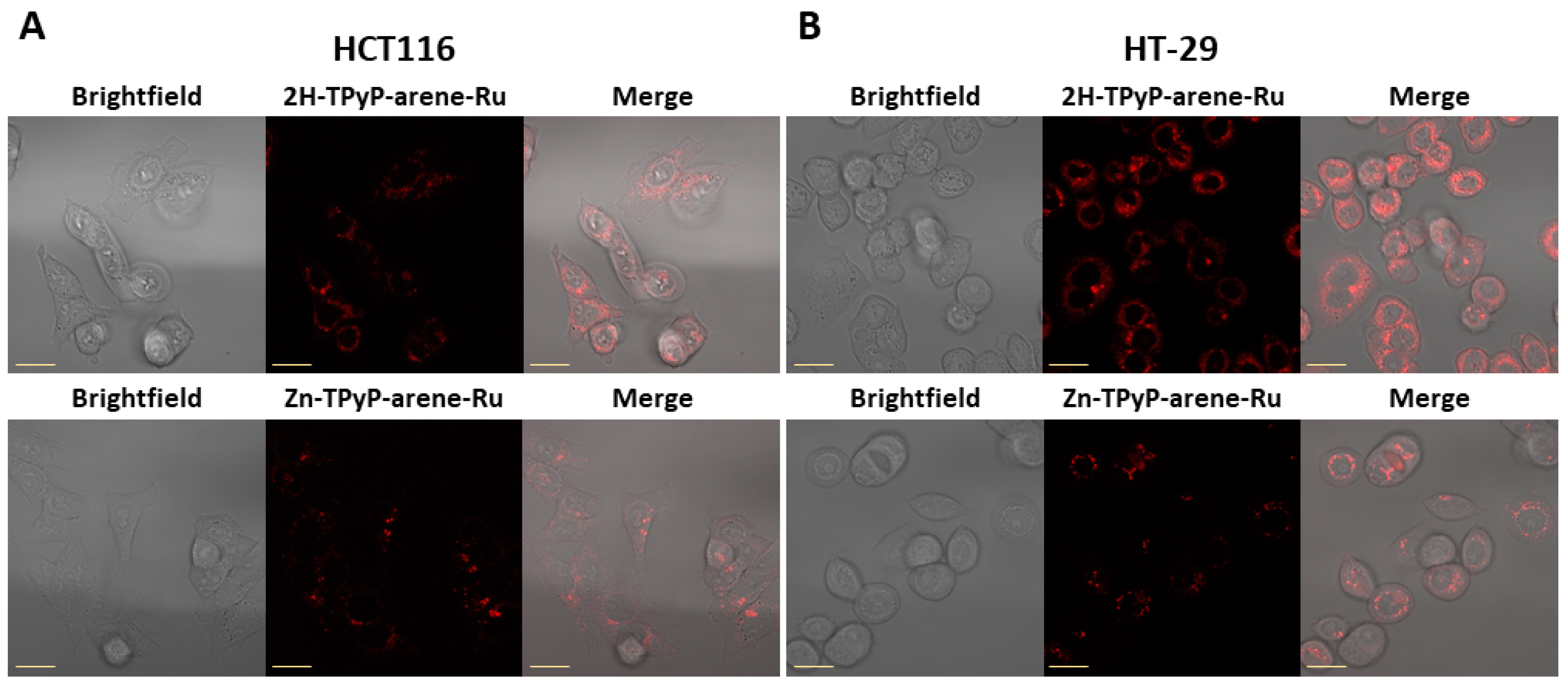
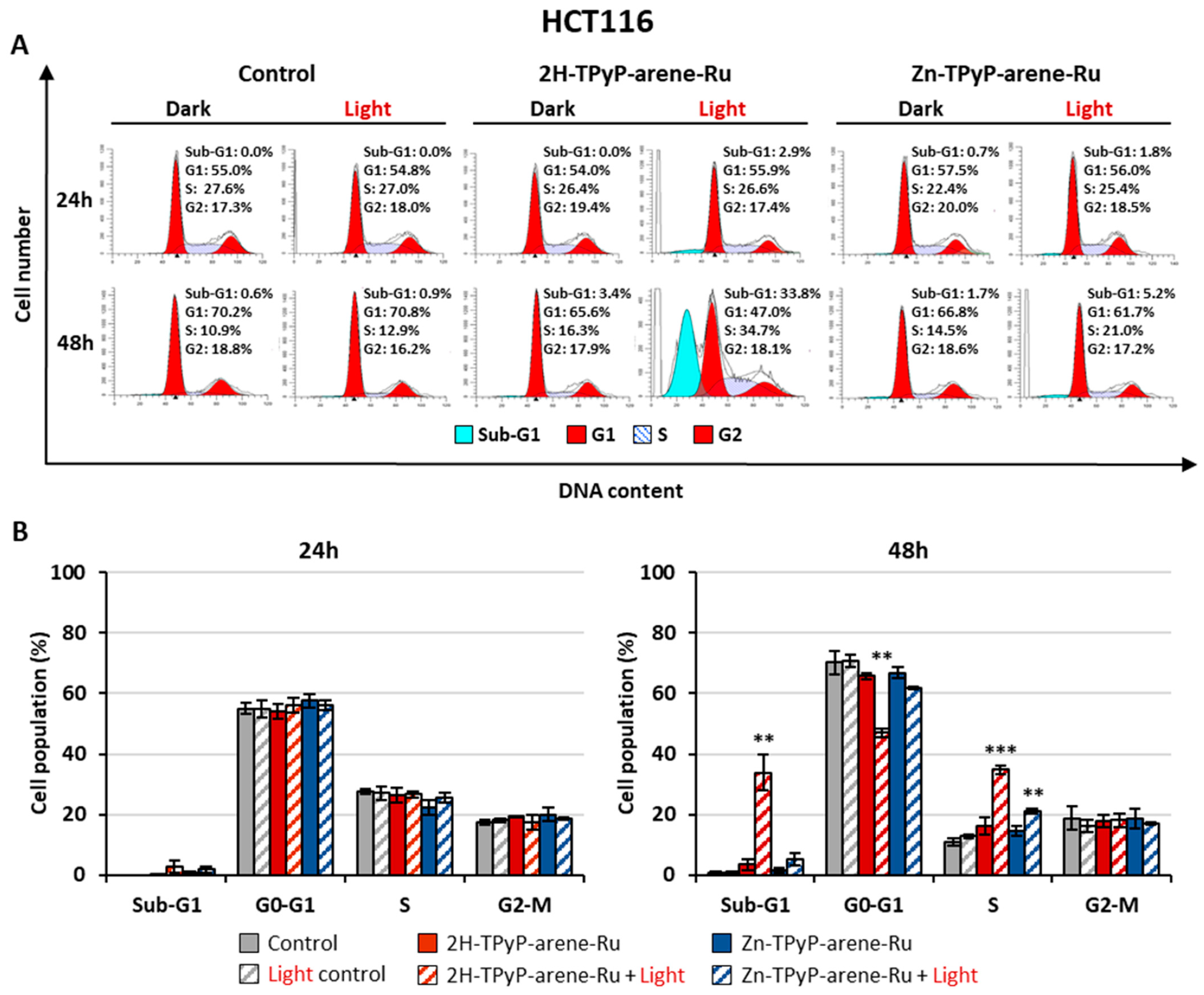
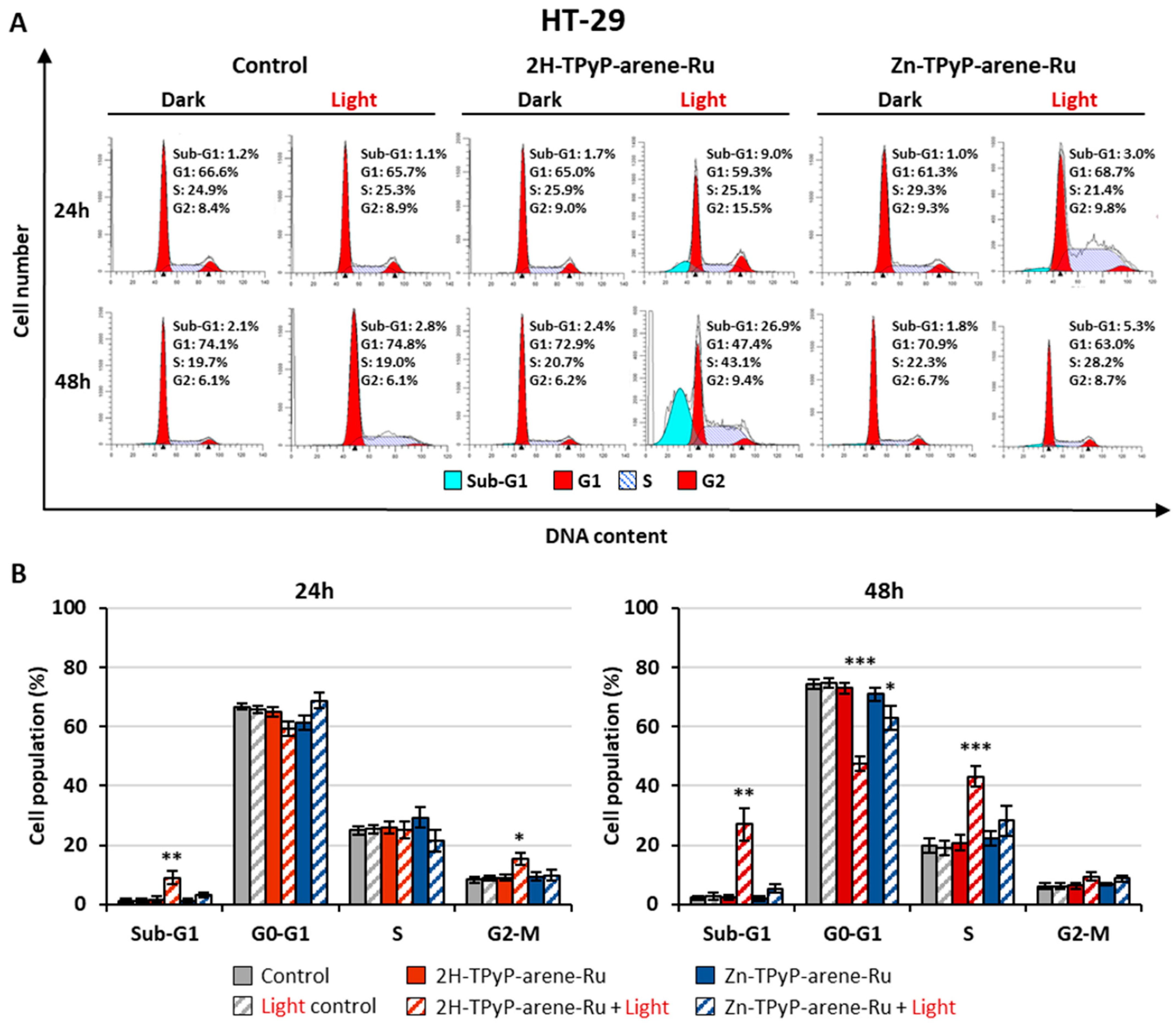
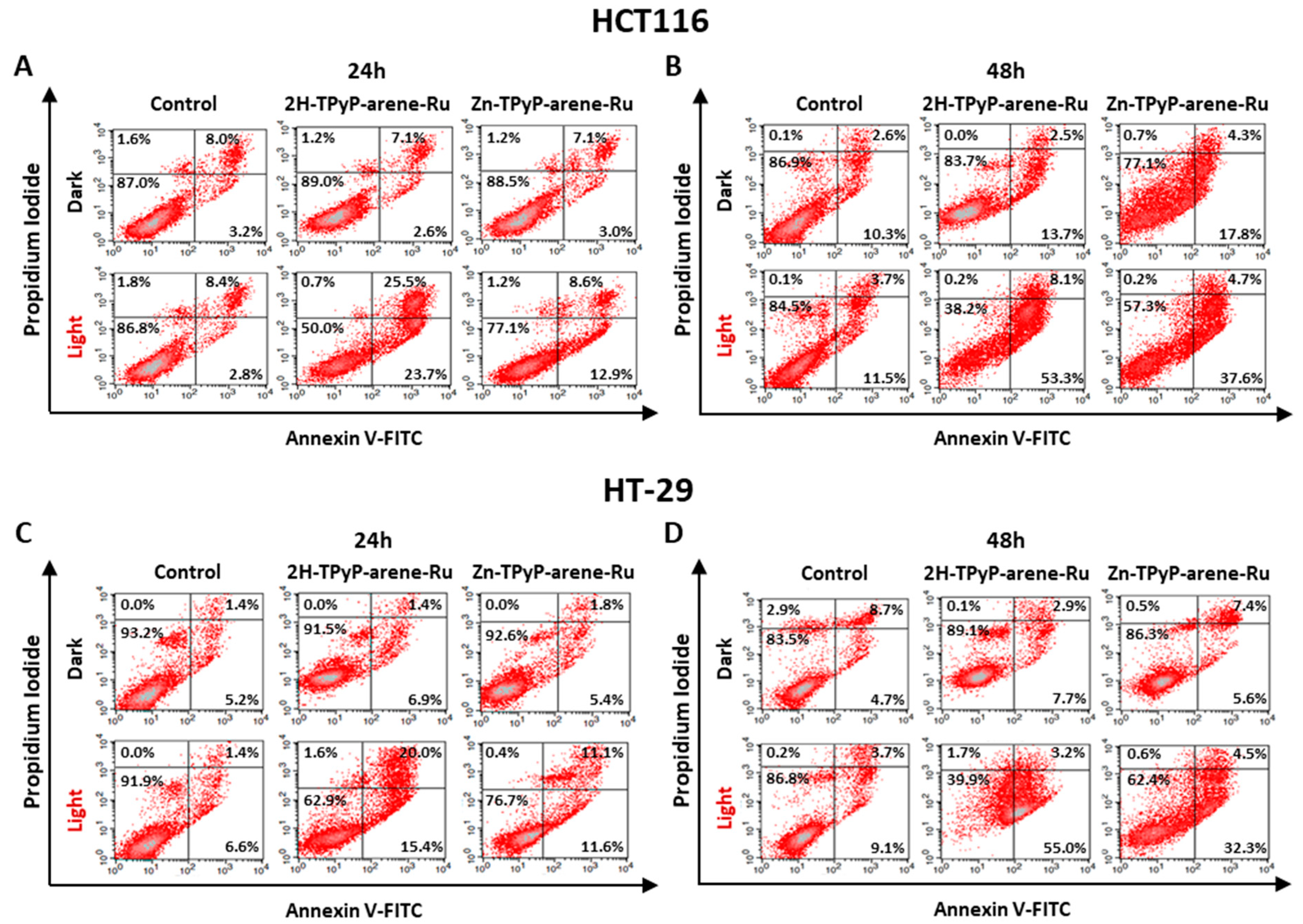
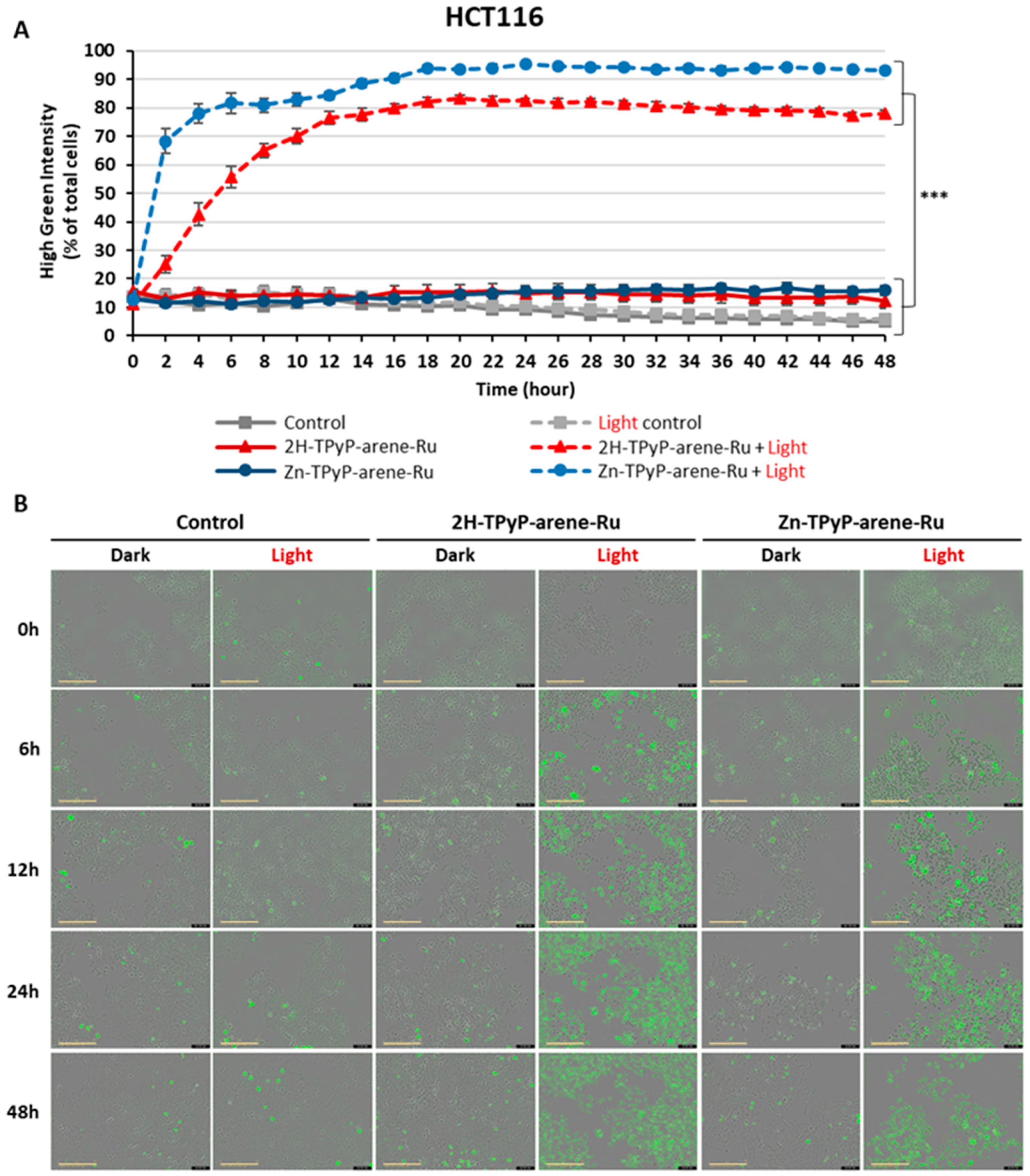
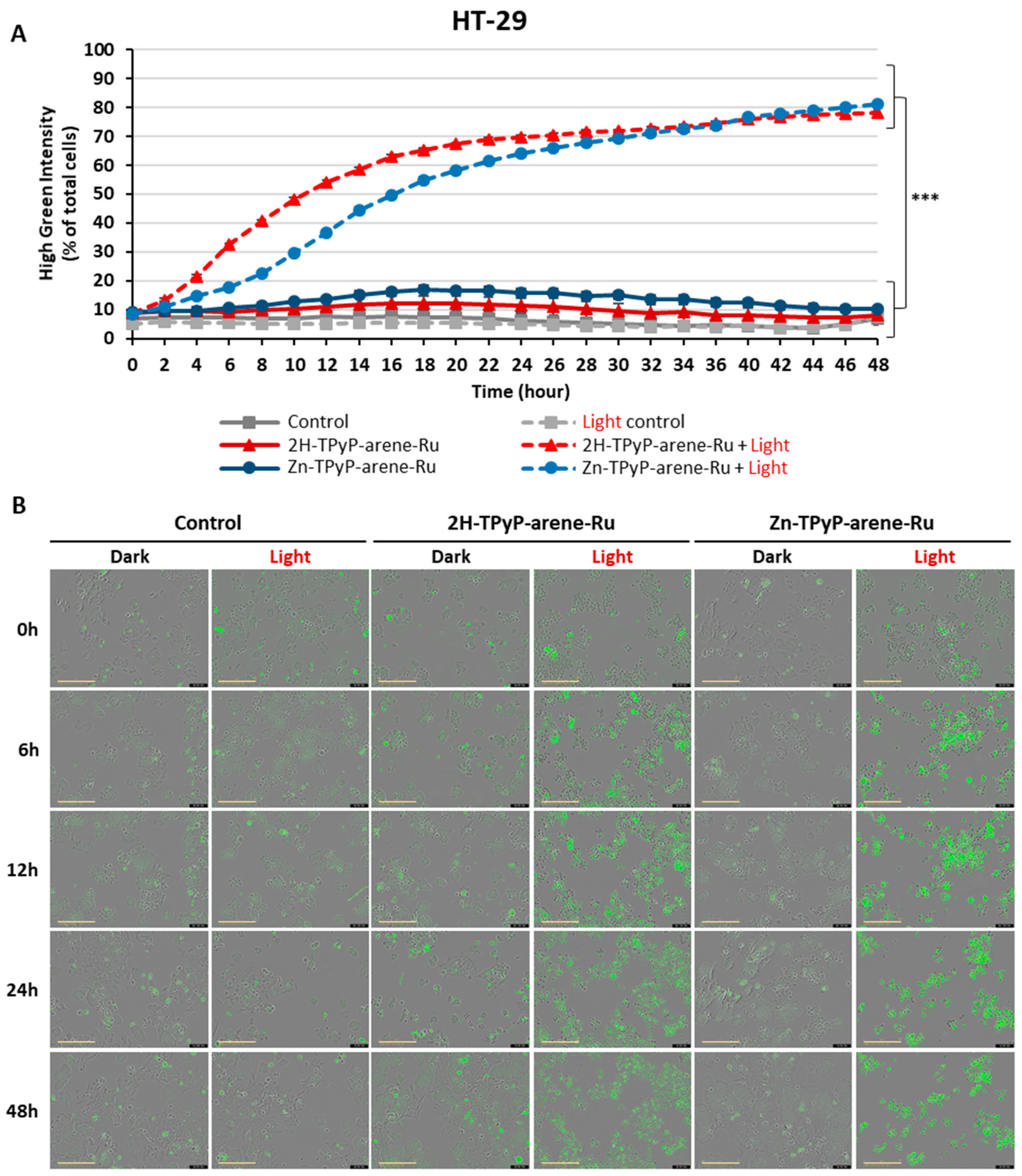
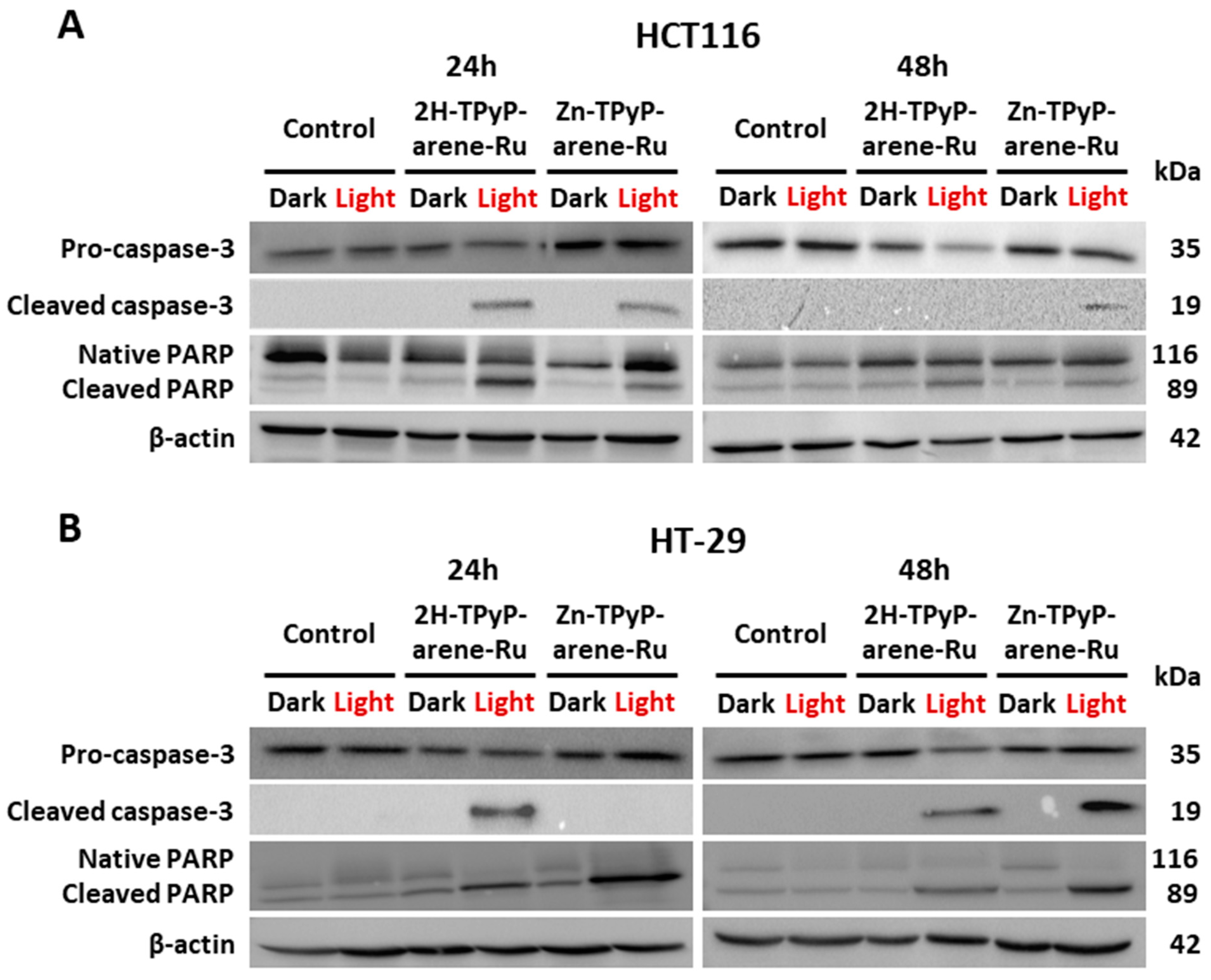
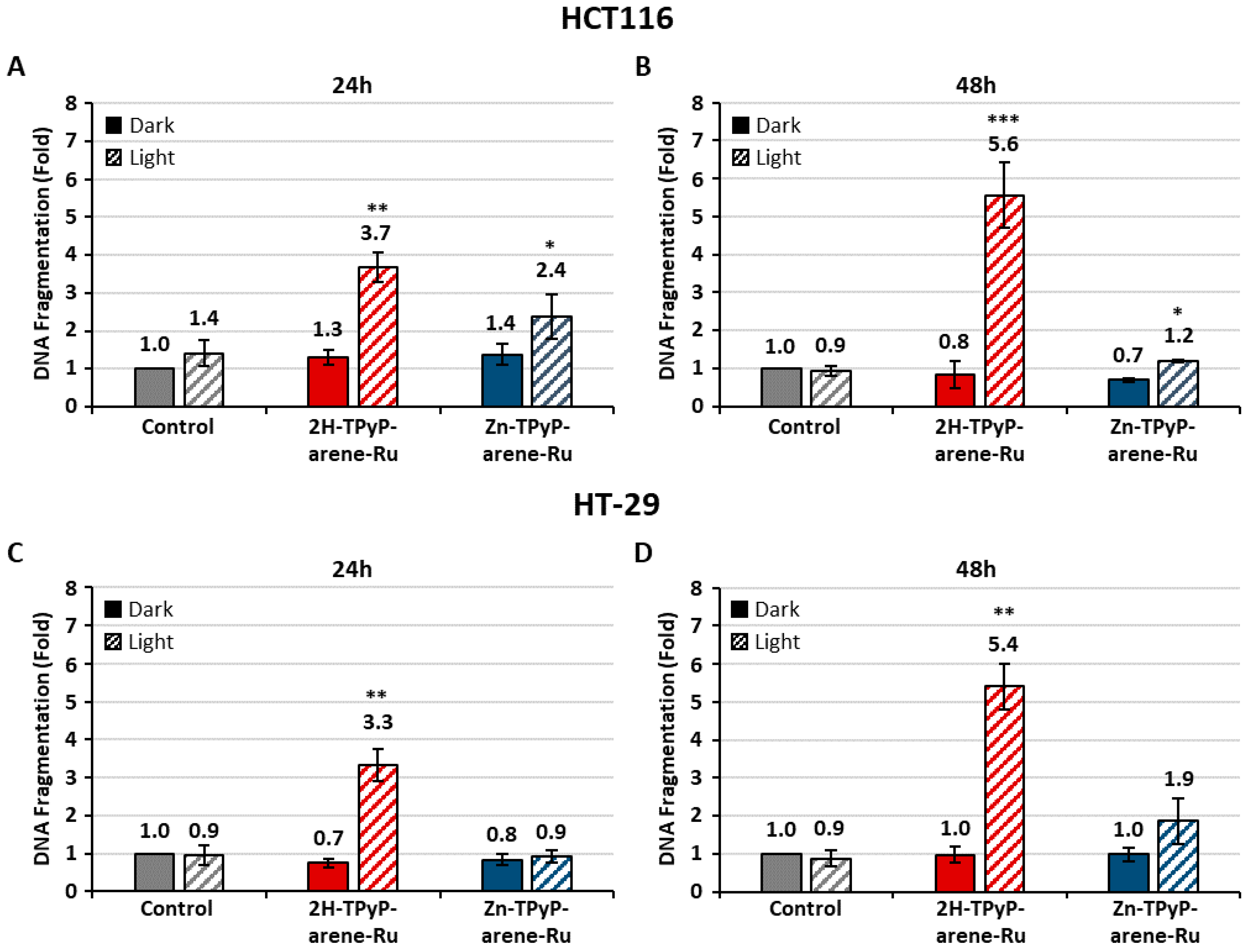
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Cell Culture and Treatment
3.3. Cytotoxicity and Phototoxicity
3.4. Intracellular ROS Production
3.5. Cellular Internalization
3.5.1. Flow Cytometry with AMNIS Imaging
3.5.2. Confocal Microscopy
3.6. Cell Cycle Analysis
3.7. Mechanisms of Apoptosis
3.7.1. Annexin V-FITC/PI Dual Staining Assay
3.7.2. Quantitative Analysis of Activated Caspases-3/-7
3.7.3. Protein Extraction and Western Blot Analysis
3.7.4. DNA Fragmentation
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Labianca, R.; Beretta, G.D.; Kildani, B.; Milesi, L.; Merlin, F.; Mosconi, S.; Pessi, M.A.; Prochilo, T.; Quadri, A.; Gatta, G.; et al. Colon cancer. Crit. Rev. Oncol. Hematol. 2010, 74, 106–133. [Google Scholar] [CrossRef] [PubMed]
- Colonna, M. Chapter 1—Épidémiologie. In Cancérologie Colorectale; Faucheron, J.-L., Ed.; Elsevier Masson: Paris, France, 2020; pp. 1–10. [Google Scholar]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Hobeika, C.; Lefevre, J. Chapter 4—Bilan du cancer colorectal. In Cancérologie Colorectale; Faucheron, J.-L., Ed.; Elsevier Masson: Paris, France, 2020; pp. 31–50. [Google Scholar]
- Nagaraju, G.P.; Shukla, D.; Vishvakarma, N.K. (Eds.) Colon Cancer Diagnosis and Therapy: Volume 1; Springer: Cham, Switzerland, 2021. [Google Scholar] [CrossRef]
- Li-Chang, H.H.; Kirsch, R.; Conner, J.; Sari, A.; Pollett, A.; El-Zimaity, H.; Jain, D.; Celli, R.; Reid, S.L.; Riddell, R.H. Atlas of Intestinal Pathology: Volume 1: Neoplastic Diseases of the Intestines; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Ishida, H.; Koda, K. Recent Advances in the Treatment of Colorectal Cancer; Springer: Singapore, 2019. [Google Scholar] [CrossRef]
- Baatrup, G. Multidisciplinary Treatment of Colorectal Cancer: Staging—Treatment—Pathology—Palliation; Springer: Cham, Switzerland, 2021. [Google Scholar] [CrossRef]
- Messersmith, W.A.; Ahnen, D.J. Targeting EGFR in colorectal cancer. N. Engl. J. Med. 2008, 359, 1834–1836. [Google Scholar] [CrossRef] [PubMed]
- Kozovska, Z.; Gabrisova, V.; Kucerova, L. Colon cancer: Cancer stem cells markers, drug resistance and treatment. Biomed. Pharmacother. 2014, 68, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Kawczyk-Krupka, A.; Bugaj, A.M.; Latos, W.; Zaremba, K.; Wawrzyniec, K.; Sieroń, A. Photodynamic therapy in colorectal cancer treatment: The state of the art in clinical trials. Photodiagn. Photodyn. Ther. 2015, 12, 545–553. [Google Scholar] [CrossRef]
- Gu, B.; Wang, B.; Li, X.; Feng, Z.; Ma, C.; Gao, L.; Yu, Y.; Zhang, J.; Zheng, P.; Wang, Y.; et al. Photodynamic therapy improves the clinical efficacy of advanced colorectal cancer and recruits immune cells into the tumor immune microenvironment. Front. Immunol. 2022, 13, 1050421. [Google Scholar] [CrossRef]
- Sharifkazemi, H.; Amini, S.M.; Ortakand, R.K.; Narouie, B. A Review of Photodynamic Therapy in Different Types of Tumors. J. Transl. Res. Urol. 2022, 4, 61–70. [Google Scholar] [CrossRef]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D.; et al. Photodynamic therapy of cancer: An update. CA Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef]
- Chilakamarthi, U.; Giribabu, L. Photodynamic Therapy: Past, Present and Future. Chem. Rec. 2017, 17, 775–802. [Google Scholar] [CrossRef]
- Bretin, L.; Pinon, A.; Bouramtane, S.; Ouk, C.; Richard, L.; Perrin, M.L.; Chaunavel, A.; Carrion, C.; Bregier, F.; Sol, V.; et al. Photodynamic Therapy Activity of New Porphyrin-Xylan-Coated Silica Nanoparticles in Human Colorectal Cancer. Cancers 2019, 11, 1474. [Google Scholar] [CrossRef]
- Hodgkinson, N.; Kruger, C.A.; Abrahamse, H. Targeted photodynamic therapy as potential treatment modality for the eradication of colon cancer and colon cancer stem cells. Tumour Biol. 2017, 39, 1010428317734691. [Google Scholar] [CrossRef] [PubMed]
- Castano, A.P.; Mroz, P.; Hamblin, M.R. Photodynamic therapy and anti-tumour immunity. Nat. Rev. Cancer. 2006, 6, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, A.F.; De Almeida, D.R.Q.; Terra, L.F.; Baptista, M.S.; Labriola, L. Photodynamic therapy in cancer treatment—An update review. J. Cancer Metastasis Treat. 2019, 5, 25. [Google Scholar] [CrossRef]
- Sellera, F.P.; Nascimento, C.L.; Ribeiro, M.S. Photodynamic Therapy in Veterinary Medicine: From Basics to Clinical Practice; Springer: Cham, Switzerland, 2016. [Google Scholar] [CrossRef]
- Baptista, M.S.; Cadet, J.; Di Mascio, P.; Ghogare, A.A.; Greer, A.; Hamblin, M.R.; Lorente, C.; Nunez, S.C.; Ribeiro, M.S.; Thomas, A.H.; et al. Type I and Type II Photosensitized Oxidation Reactions: Guidelines and Mechanistic Pathways. Photochem. Photobiol. 2017, 93, 912–919. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, S.; Knap, B.; Przystupski, D.; Saczko, J.; Kędzierska, E.; Knap-Czop, K.; Kotlińska, J.; Michel, O.; Kotowski, K.; Kulbacka, J. Photodynamic therapy—Mechanisms, photosensitizers and combinations. J. Biomed. Pharmacother. 2018, 106, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Delaire, J.; Piard, J.; Méallet-Renault, R.; Clavier, G. Photophysique et photochimie: Des fondements aux applications. In QuinteSciences; EDP Sciences: Les Ulis, France, 2016. [Google Scholar]
- Nowak-Stepniowska, A.; Pergoł, P.; Padzik-Graczyk, A. Photodynamic method of cancer diagnosis and therapy-mechanisms and applications. Postep. Biochem. 2013, 59, 53–63. [Google Scholar]
- Luksiene, Z. Photodynamic therapy: Mechanism of action and ways to improve the efficiency of treatment. Medicina 2003, 39, 1137–1150. [Google Scholar]
- Juzeniene, A.; Moan, J. The history of PDT in Norway Part one: Identification of basic mechanisms of general PDT. Photodiagn. Photodyn. Ther. 2007, 4, 3–11. [Google Scholar] [CrossRef]
- Fonseca, S.M.; Pina, J.; Arnaut, L.G.; Seixas de Melo, J.; Burrows, H.D.; Chattopadhyay, N.; Alcacer, L.; Charas, A.; Morgado, J.; Monkman, A.P.; et al. Triplet-state and singlet oxygenformation in fluorene-based alternating copolymers. J. Phys. Chem. B 2006, 110, 8278–8283. [Google Scholar] [CrossRef]
- Kessel, D. Death pathways associated with photodynamic therapy. Med. Laser Appl. 2006, 21, 219–224. [Google Scholar] [CrossRef]
- Zhu, W.; Gao, Y.H.; Liao, P.Y.; Chen, D.Y.; Sun, N.N.; Nguyen Thi, P.A.; Yan, Y.J.; Wu, X.F.; Chen, Z.L. Comparison between porphin, chlorin and bacteriochlorin derivatives for photodynamic therapy: Synthesis, photophysical properties, and biological activity. Eur. J. Med. Chem. 2018, 160, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Köberle, B.; Schoch, S. Platinum Complexes in Colorectal Cancer and Other Solid Tumors. Cancers 2021, 13, 2073. [Google Scholar] [CrossRef] [PubMed]
- Lazarević, T.; Rilak, A.; Bugarčić, Ž.D. Platinum, palladium, gold and ruthenium complexes as anticancer agents: Current clinical uses, cytotoxicity studies and future perspectives. Eur. J. Med. Chem. 2017, 142, 8–31. [Google Scholar] [CrossRef]
- Alessio, E.; Mestroni, G.; Bergamo, A.; Sava, G. Ruthenium antimetastatic agents. Curr. Top. Med. Chem. 2004, 4, 1525–1535. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Kim, C.Y.; Nam, T.G. Ruthenium Complexes as Anticancer Agents: A Brief History and Perspectives. Drug Des. Dev. Ther. 2020, 14, 5375–5392. [Google Scholar] [CrossRef]
- Schmitt, F.; Govindaswamy, P.; Süss-Fink, G.; Ang, W.H.; Dyson, P.J.; Juillerat-Jeanneret, L.; Therrien, B. Ruthenium porphyrin compounds for photodynamic therapy of cancer. J. Med. Chem. 2008, 51, 1811–1816. [Google Scholar] [CrossRef]
- Rani-Beeram, S.; Meyer, K.; McCrate, A.; Hong, Y.; Nielsen, M.; Swavey, S. A Fluorinated Ruthenium Porphyrin as a Potential Photodynamic Therapy Agent: Synthesis, Characterization, DNA Binding, and Melanoma Cell Studies. Inorg. Chem. 2008, 47, 11278–11283. [Google Scholar] [CrossRef]
- Monro, S.; Colón, K.L.; Yin, H.; Roque, J.; Konda, P.; Gujar, S.; Thummel, R.P.; Lilge, L.; Cameron, C.G.; McFarland, S.A. Transition Metal Complexes and Photodynamic Therapy from a Tumor-Centered Approach: Challenges, Opportunities, and Highlights from the Development of TLD1433. Chem. Rev. 2019, 119, 797–828. [Google Scholar] [CrossRef]
- Gallardo-Villagrán, M.; Paulus, L.; Charissoux, J.L.; Leger, D.Y.; Vergne-Salle, P.; Therrien, B.; Liagre, B. Ruthenium-based assemblies incorporating tetrapyridylporphyrin panels: A photosensitizer delivery strategy for the treatment of rheumatoid arthritis by photodynamic therapy. Dalton Trans. 2022, 51, 9673–9680. [Google Scholar] [CrossRef]
- Barry, N.P.E.; Zava, O.; Dyson, P.J.; Therrien, B. Synthesis, Characterization and Anticancer Activity of Porphyrin-Containing Organometallic Cubes. Aust. J. Chem. 2010, 63, 1529–1537. [Google Scholar] [CrossRef]
- Baskaran, R.; Lee, J.; Yang, S.G. Clinical development of photodynamic agents and therapeutic applications. Biomater. Res. 2018, 22, 25. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.W.-Y.; Zhang, M.; Li, D.; Li, M.; Wong, A.S.-T. Enhanced anti-cancer activities of a gold(III) pyrrolidinedithiocarbamato complex incorporated in a biodegradable metal-organic framework. J. Inorg. Biochem. 2016, 163, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, F.; Govindaswamy, P.; Zava, O.; Süss-Fink, G.; Juillerat-Jeanneret, L.; Therrien, B. Combined arene ruthenium porphyrins as chemotherapeutics and photosensitizers for cancer therapy. J. Biol. Inorg. Chem. 2009, 14, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Bogoeva, V.; Siksjø, M.; Sæterbø, K.G.; Melø, T.B.; Bjørkøy, A.; Lindgren, M.; Gederaas, O.A. Ruthenium porphyrin-induced photodamage in bladder cancer cells. Photodiagn. Photodyn. Ther. 2016, 14, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Gallardo-Villagrán, M.; Paulus, L.; Charissoux, J.L.; Sutour, S.; Vergne-Salle, P.; Leger, D.Y.; Liagre, B.; Therrien, B. Evaluation of Ruthenium-Based Assemblies as Carriers of Photosensitizers to Treat Rheumatoid Arthritis by Photodynamic Therapy. Pharmaceutics 2021, 13, 2104. [Google Scholar] [CrossRef]
- Silva, S.L.R.; Baliza, I.R.S.; Dias, R.B.; Sales, C.B.S.; Gurgel Rocha, C.A.; Soares, M.B.P.; Correa, R.S.; Batista, A.A.; Bezerra, D.P. Ru(II)-thymine complex causes DNA damage and apoptotic cell death in human colon carcinoma HCT116 cells mediated by JNK/p38/ERK1/2 via a p53-independent signaling. Sci. Rep. 2019, 9, 11094. [Google Scholar] [CrossRef]
- Mishra, A.K.; Mishra, L. Ruthenium Chemistry; Pan Stanford Publishing Pte. Ltd.: Singapore, 2018. [Google Scholar] [CrossRef]
- Lu, Y.; Shen, T.; Yang, H.; Gu, W. Ruthenium Complexes Induce HepG2 Human Hepatocellular Carcinoma Cell Apoptosis and Inhibit Cell Migration and Invasion through Regulation of the Nrf2 Pathway. Int. J. Mol. Sci. 2016, 17, 775. [Google Scholar] [CrossRef]
- Barry, N.P.E.; Austeri, M.; Lacour, J.; Therrien, B. Highly Efficient NMR Enantiodiscrimination of Chiral Octanuclear Metalla-Boxes in Polar Solvent. Organometallics 2009, 28, 4894–4897. [Google Scholar] [CrossRef]
- Rioux, B.; Pinon, A.; Gamond, A.; Martin, F.; Laurent, A.; Champavier, Y.; Barette, C.; Liagre, B.; Fagnère, C.; Sol, V.; et al. Synthesis and biological evaluation of chalcone-polyamine conjugates as novel vectorized agents in colorectal and prostate cancer chemotherapy. Eur. J. Med. Chem. 2021, 222, 113586. [Google Scholar] [CrossRef]
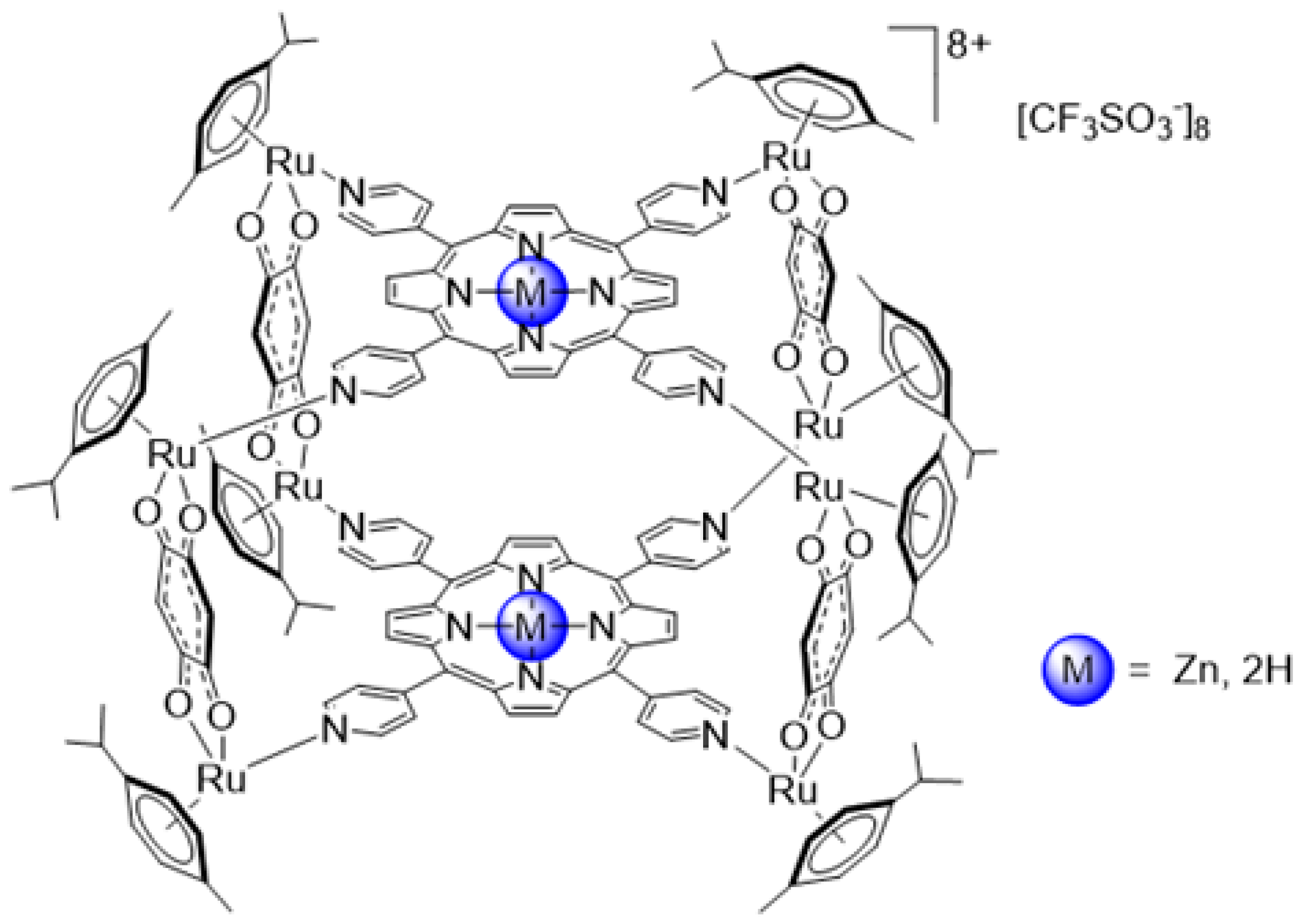
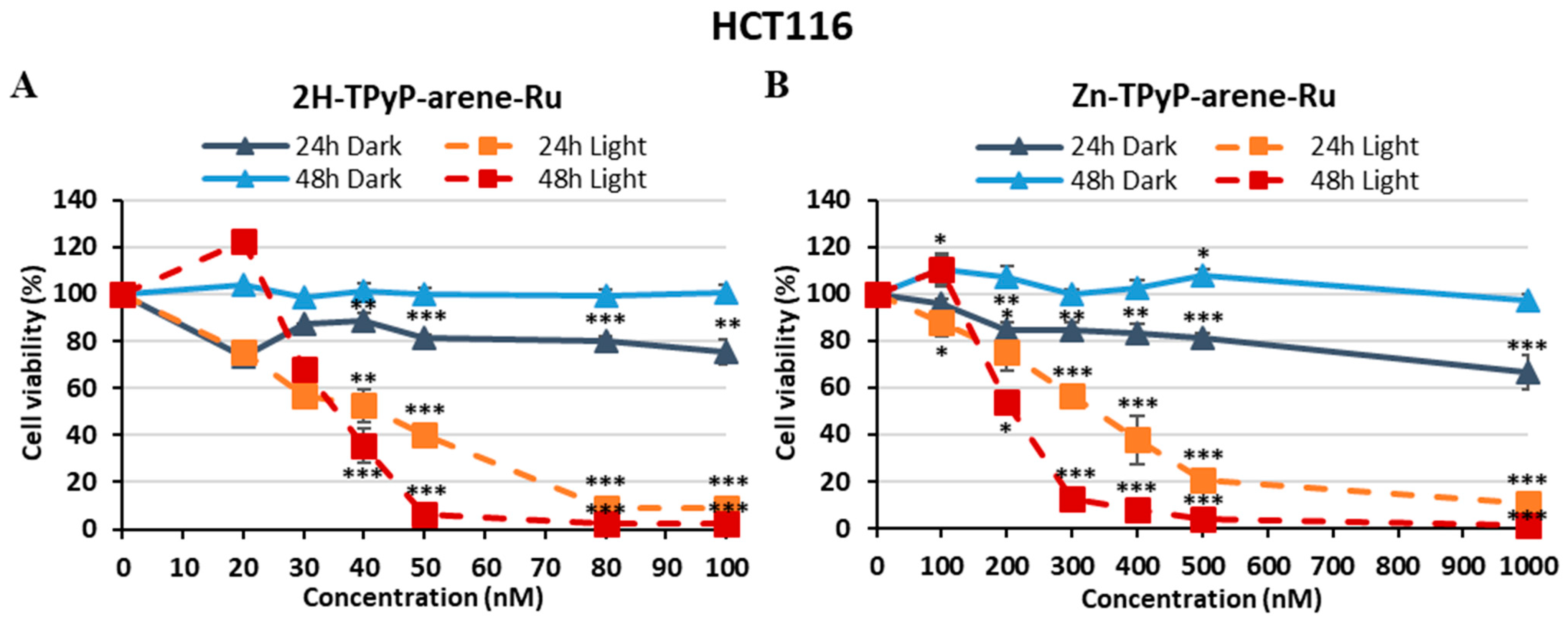
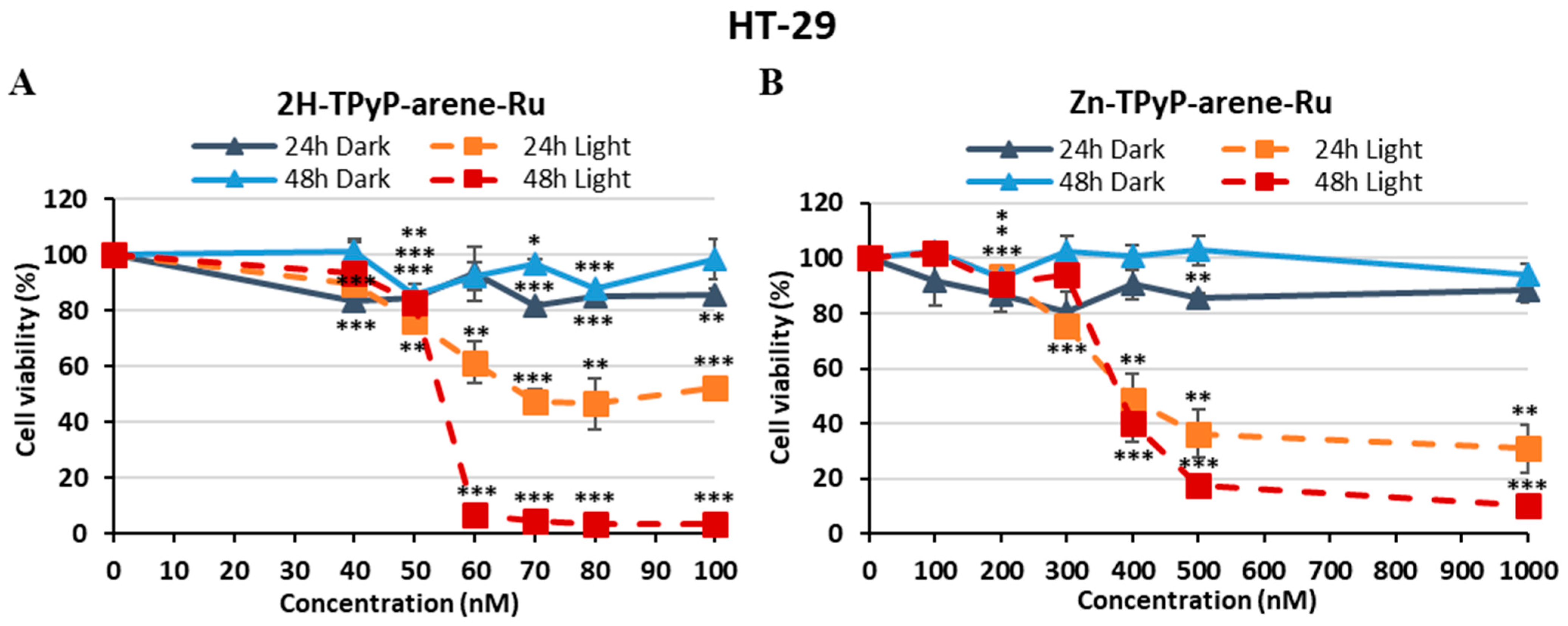
| PS | IC50 (nM) | PI (IC50 Dark/IC50 Light) | ||||
|---|---|---|---|---|---|---|
| Light 24 h | Light 48 h | Dark | 24 h | 48 h | ||
| HCT116 | 2H-TPyP-arene-Ru | 41.9 | 35.2 | > 100 | > 2.38 | > 2.84 |
| Zn-TPyP-arene-Ru | 331 | 207 | > 1000 | > 3.02 | > 4.83 | |
| HT-29 | 2H-TPyP-arene-Ru | 67.8 | 54.1 | > 100 | > 1.47 | > 1.84 |
| Zn-TPyP-arene-Ru | 394 | 379 | > 1000 | > 2.53 | > 2.63 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Massoud, J.; Pinon, A.; Gallardo-Villagrán, M.; Paulus, L.; Ouk, C.; Carrion, C.; Antoun, S.; Diab-Assaf, M.; Therrien, B.; Liagre, B. A Combination of Ruthenium Complexes and Photosensitizers to Treat Colorectal Cancer. Inorganics 2023, 11, 451. https://doi.org/10.3390/inorganics11120451
Massoud J, Pinon A, Gallardo-Villagrán M, Paulus L, Ouk C, Carrion C, Antoun S, Diab-Assaf M, Therrien B, Liagre B. A Combination of Ruthenium Complexes and Photosensitizers to Treat Colorectal Cancer. Inorganics. 2023; 11(12):451. https://doi.org/10.3390/inorganics11120451
Chicago/Turabian StyleMassoud, Jacquie, Aline Pinon, Manuel Gallardo-Villagrán, Lucie Paulus, Catherine Ouk, Claire Carrion, Sayed Antoun, Mona Diab-Assaf, Bruno Therrien, and Bertrand Liagre. 2023. "A Combination of Ruthenium Complexes and Photosensitizers to Treat Colorectal Cancer" Inorganics 11, no. 12: 451. https://doi.org/10.3390/inorganics11120451
APA StyleMassoud, J., Pinon, A., Gallardo-Villagrán, M., Paulus, L., Ouk, C., Carrion, C., Antoun, S., Diab-Assaf, M., Therrien, B., & Liagre, B. (2023). A Combination of Ruthenium Complexes and Photosensitizers to Treat Colorectal Cancer. Inorganics, 11(12), 451. https://doi.org/10.3390/inorganics11120451