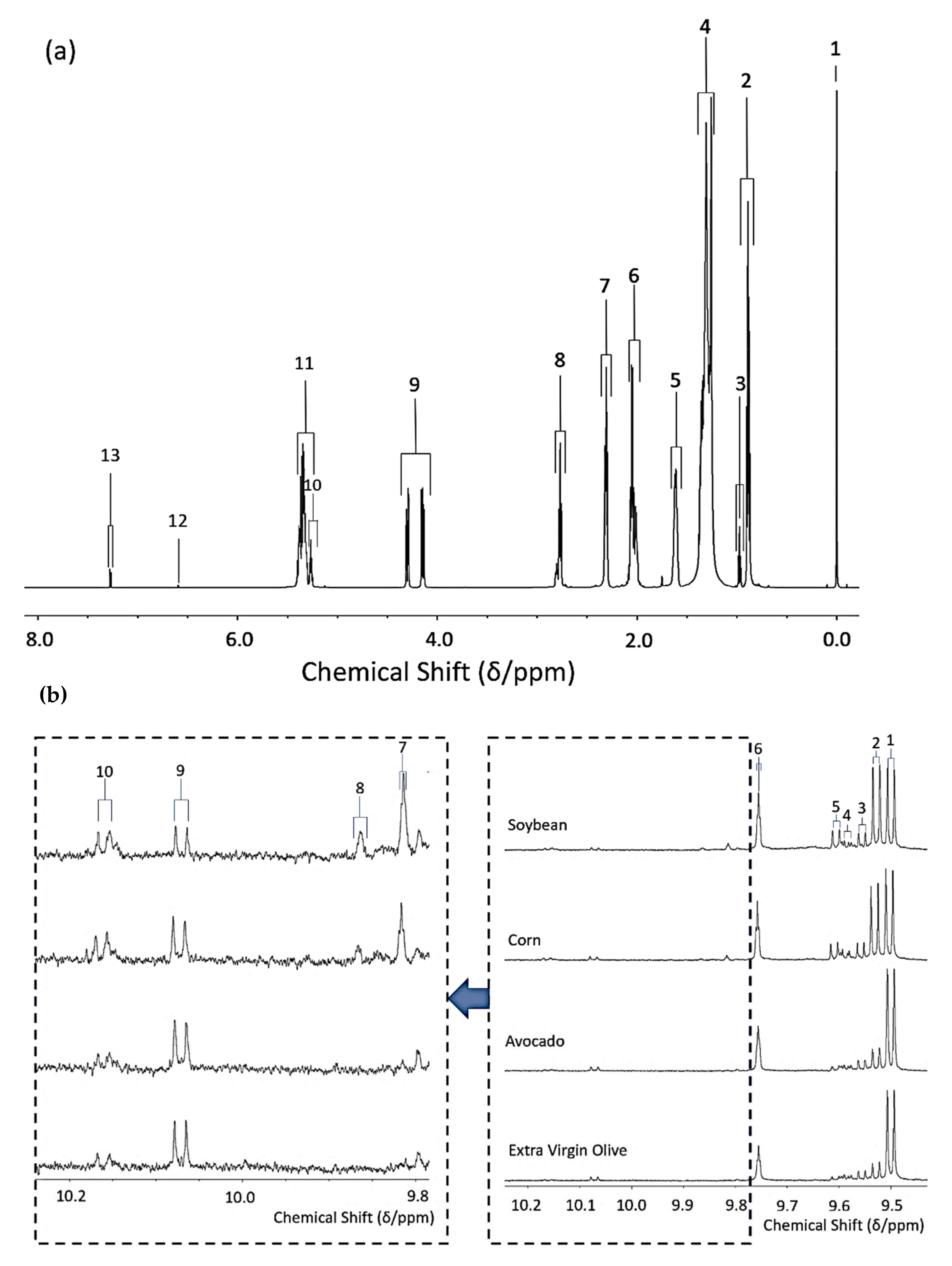
3.1. 1H NMR Analysis of the FA Contents of Culinary Oils before and after Exposure to LSSFEs
Figure 1a shows the major triacylglycerol (TAG) resonances detectable in the
1H NMR profile of a typical soybean oil sample as an example. Full assignments for each of these signals are provided in
Table 1, and these were based on their chemical shift values, coupling patterns and coupling constants as previously reported [
1,
3,
15,
16,
17].
In order to determine the lipid compositional profiles of the cooking oils evaluated, their MUFA, PUFA and SFA (molar %) contents were computed and monitored at increasing time-points throughout the complete 90 min thermal stressing cycle. Major TAG lipid signals were normalized to that of the total FA terminal-CH
3 groups (
t, δ = 0.84–0.99 ppm). Signals employed for these calculations were the unsaturated ω-3 FA acyl group-terminal-C
H3 (
t. δ = 0.95–0.99 ppm); FA acyl group -CH=CH-CH
2- (
m, δ = 1.96–2.10 ppm);
bis-allylic-CH=CH-C
H2-CH=CH- protons of linoleoylglycerols (
m, δ = 2.75–2.79 ppm); ω-3 FA acyl group
bis-allylic-CH=CH-C
H2-CH=CH- protons of linolenoylglycerols (
m, δ = 2.79–2.82 ppm), and the -C
H2OH functions of TAG backbones (
dd,dd, δ = 4.12–4.32 ppm) as documented in [
17] (
Table 1). Moreover,
Table S1 in the Supplementary Materials section provides a full listing of the
1H NMR-determined FA compositions at the 0, 5, 10, 20, 30, 60 and 90 min LSSFE time-points for all six oils monitored. In view of the very low contents of ω-3 FAs present in extra-virgin olive, avocado and corn oils (i.e., <1.2 weight % for each of them), it was not possible to monitor LSSFE-induced changes in its contents in these products in view of the superimposition of its characteristic terminal-CH
3 group resonance at δ = 0.95–0.99 ppm on that of the similarly intense
13C satellite of the ω-6 FA signal (δ = 0.84–0.91 ppm). Therefore, only the weight % literature values for the unheated oils are provided in
Table S1 for comparative purposes.
An evaluation of the oil FA compositions prior to and following thermal stressing episodes (
Table S1, Supplementary Materials) demonstrated that the greatest thermally induced modifications to their lipid composition profiles were observed for both soybean oils tested, and corn oil, in view of their relatively high PUFA contents. Notably, the relative SFA content levels of the oils increased significantly in these products in view of a corresponding decrease in overall PUFA and, to a much lesser extent, MUFA contents. However, as expected, time-dependent decreases in overall UFA contents were evident across all six oils tested, although markedly greater decreases in total PUFA contents were observed in corn and soybean oils, as expected. Indeed, their total PUFA levels were reduced by 5–10 molar % at the 90 min LSSFE time-point. Culinary oils with higher MUFA contents, however, exhibited the smallest FA compositional changes post-thermal stressing, i.e., those observed for the two extra-virgin olive oils investigated. Avocado oil, however, underwent some moderate changes in its composition, specifically increases in its SFA (>7%) and decreases in its PUFA (>3%) molar contents, although such changes were not as prominent as those observed for soybean and corn oils. These modifications in FA concentrations demonstrate the high susceptibilities of culinary oil PUFAs to thermo-oxidation and degradation during LSSFEs, and also highlight the fact that MUFA-rich oils display a much greater resistance against oxidative deterioration. Hence, from a toxicological standpoint, these high MUFA content oils are generally more suitable for commercial or domestic high-temperature frying practices. For the total UFA and SFA contents, the orders of decrease and increase in their magnitudes were SBO2 ≈ SBO1 ≈ CO1 > IEVOO1 ≈ AVO1 > SEVOO1 (starting from the 10 min time-point), and CO1 ≈ SBO2 > SBO1 > IEVOO1 ≈AVO1 > SEVOO1 (from the 20 min time-point), respectively.
ANOVA of the FA composition datasets revealed extremely highly significant differences between both oil type and sampling time-point (0–90 min) mean values (
Table 2). Indeed, such differences had
p values ranging from <10
−177 to <10
−23 (oil type), and <10
−51 to <10
−25 (LSSFE time-point). Bar diagrams of these mean values, with their associated 95% confidence intervals (CIs), are shown in
Figure S1 of the Supplementary Materials section. Interestingly, throughout the course of these LSSFEs, the MUFA contents of these oils increased for PUFA-rich soybean and corn oils from the 30 min time-point (a consequence of primary PUFA consumption), whereas those of the extra-virgin olive and avocado oils decreased from this time-point, which indicates the delayed onset of MUFA peroxidation, a process following primary PUFA oxidation. Moreover, the PUFA contents of the two soybean and single corn oil products decreased from 20 min, whereas those of both extra-virgin olive and the avocado oil products were lowered from the 30 min time-point.
As expected, the culinary oil x time-point first-order interaction effect was also extremely significant for all oils tested (
p < 10
−50 to <10
−18), and this ratifies the contrasting responses of each oil type to increasing LSSFE lengths; for example, differential rates and extents PUFA losses from soybean/corn oils and extra-virgin olive/avocado oils (
Figure S1b), and likewise the contrasting elevations and reductions in oil MUFA contents for these two groups respectively (
Figure S1a). Our results also demonstrated that the ‘health-friendly’ ω-3 PUFAs, which are readily
1H NMR-detectable in soybean oil products, were particularly susceptible to thermal degradation during the LSSFE heating process, and therefore such vegetable and other oils marketed with the nutritional benefits offered by such FAs therein contain significantly diminished levels of these nutrients after being exposed to commercial or domestic frying or cooking processes. For these data, the specific ω-3 FA function resonances present in both soybean oil products tested showed reductions in the contents of linolenoylglycerols of 1.0 mol. % for SBO1, and 1.5 mol. % for SBO2 at the 90 min LSSFE sampling time-point (
Supplementary Materials, Table S1 and Figure S1e). Furthermore, despite the presence of tocopherol antioxidants in such products [
18], the realm of aldehydic LOPs generated remained in abundance (
Figure 2). Indeed, these results corroborate with those of previous studies, which have indicated that antioxidants such as naturally present α-TOH, and added tocopherol esters and BHA, exhibit only very limited antioxidant properties at high frying temperatures [
3,
8,
17]. Moreover, α-TOH has been shown to be thermally unstable at temperatures as low as 140 °C [
19].
3.2. 1H NMR Monitoring of the Evolution of Secondary Aldehydic LOPs in Culinary Oils during Their Exposure to LSSFEs
All aldehyde species investigated are
1H NMR-detectable in the 9.00–10.20 ppm region of the spectral profiles obtained as their -C
HO functional group protons, and individual aldehyde assignments and the multiplicities of resonances are highlighted in
Table 3 as resonance assignment codes 1–10 (
Figure 1b). These aldehyde signals were integrated relative to the total terminal-CH
3 group resonances of total oil FAs, and their concentrations are expressed in mmol./mol. total FA units. From recent studies featuring determinations of the molecular structures of aldehydic LOPs derived from the peroxidation of plentiful ω-3 FAs in marine oils [
15], the saturated nature of those giving rise to -C
HO resonances located within the 9.70–9.90 ppm regions of spectra acquired on its thermo-oxidised samples (i.e., both high- and low-molecular-mass
n-alkanals, and 4-oxo-
n-alkanals), was confirmed by the acquisition of 2D
1H-
1H COSY spectra. Indeed, all adjacent
1H nucleus correlations observed for resonances within this region were found to be localised in the δ = 2.37–2.80 ppm spectral sector, an observation confirming the presence of saturated methylene (-CH
2-) groups at the α-position for all species involved.
As expected, aldehyde formation increases with increasing thermal stressing period (
Figure 2), and these values were also clearly critically dependent on oil nature and composition, with the highest levels of all aldehydes monitored being generated in PUFA-rich soybean and corn oils. Indeed, this time-dependent trend was observed across all aldehyde species, with the exception of (
E)-2-alkenals, for which one of the soybean oil products tested (SBO1) generated slightly lower quantities at the 90 min time-point. However, (
E)-2-Alkenals have also been shown to be produced as prominent secondary LOPs arising from the oxidation of oleoylglycerols in oil products rich in these FAs [
1,
16]. As noted from
Figure 2, the relationship observed between aldehyde concentrations produced and LSSFE heating time is sigmoidal (i.e., S-shaped), since the lipid peroxidation process is an autocatalytic, self-propagating process, and is dependent on the primary formation and subsequent fragmentation of lipid hydroperoxides such as PUFA-derived CHPDs; a full list of mean concentration values for 9 of the aldehydes generated in each oil product at each LSSFE sampling time-point is provided in
Table S2 of the Supplementary Materials. Prior to the 30 min time-point of LSSFE exposure, secondary aldehydic LOPs detectable in extra-virgin olive oil samples were present at only low quantities. However, this was not in the case for soybean and corn oils (
Figure 2), with significant amounts being generated at this time-point. Indeed, ordering of the magnitudes of the levels of aldehydic LOPs generated in these oil products at this time-point only gave a ranking of SBO2 > SBO1 > CO1 > AVO1 > IEVOO1 > SEVOO1 for total α,β-unsaturated aldehydes, and SBO2 ≈ CO ≈ AVO1 > SBO1 > IEVOO1 > SEVOO1 for arguably less toxic
n-alkanals (
Table S3 of Supplementary Materials). These values are again reflective of the relative PUFA and MUFA contents of these oils, with the latter being much more resistant to peroxidation than the former.
Notwithstanding, it appears that the greater susceptibilities of linolenoylglycerol species to peroxidation is responsible for the higher α,β-unsaturated aldehyde levels found in both soybean oil products at this quite low LSSFE time-point, i.e., a higher rate of CHPD generation and fragmentation to such aldehydes than that observed with linoleoylglycerols. In general, corn oil and one of the soybean oil products (SBO2) produced the highest levels of aldehydes throughout the thermal stressing time course at 180 °C. Although at the 90 min time-point, these levels were very similar for (
E,E)-alka-2,4-dienals, 4,5-epoxy-(
E)-2-alkenals, 4-hydroxy-/4-hydroperoxy-(
E)-2-alkenals,
n-alkanals. and 4-oxo-2-alkanals produced in these oils, concentrations of (
E)-2-alkenals were somewhat greater in corn oil than in SBO2. However, the SBO2 product generated higher levels of LMM
n-alkanals than both corn oil and the other soybean oil product (SBO1); these SBO2 product concentrations were also greater for (
E,Z)-alka-2,4-dienals (
Figure 2 and
Table S3). The higher LMM
n-alkanal concentrations, including propanal, generated in both SBO products, are consistent with their specific generation from their higher contents of ω-3 FA TAG species [
1,
6].
Although the source of 4-oxo-
n-alkanals is not simply explicable from a full survey conducted on literature available, Dick et al. [
20] found that 4-hydroxynonanal (HNA) arose from the actions of alkenal oxidoreductases on the linoleoylglycerol peroxidation product 4-hydroxy-(
E)-2-nonenal (4-HNE) in vivo, and Kubatova et al. [
21] noted that 4-oxo-
n-nonanal, a highly potent lysyl alkylating agent, was a product generated in their experiments focused on the metabolism of HNE in central nervous system (CNS) models. Hence, this process appears to involve a sequentially mediated reduction of the (
E)-2-nonenal chain to its saturated nonanal derivative via alkenal oxidoreductase activities, and the possible oxidation of its 4-position primary hydroxyl function through the actions of any available alcohol dehydrogenases [
22]. However, since 4-oxo-(
E)-nonenal (4-ONE) is a known lipid peroxidation product [
22], it is feasible that a direct uncatalyzed chemical pathway involving the generation of 4-oxo-
n-alkanals directly from reduction of their corresponding 4-oxo-(
E)-2-alkenal species, i.e., 4-oxo-
n-nonanal from linoleoylglycerol-derived 4-ONE, and 4-oxo-
n-hexanal from linolenoylglycerol-derived 4-oxo-(
E)-2-hexenal, occurs in culinary oils following prolonged LSSFE thermal stressing periods, i.e., those exposed for ≥60 min Interestingly, a correlation analysis of the complete dataset featuring all time-points revealed that there was a strong positive correlation between 4-oxo-
n-alkanal and the combined 4-hydroxy/4-hydroperoxy-(
E)-2-alkenal levels in mmol./mol total FA units (r = 0.89), as may be expected if this hypothesis was correct (4-hydroxy-(
E)-2-alkenals arise from reduction of their corresponding hydroperoxy- derivatives [
17]).
Since 4-oxo-
n-alkanals produced at relatively high levels in soybean oil products at LSSFE time-points of ≥60 min were also found to be strongly correlated with LMM
n-alkanals (r = 0.93), and that both these aldehydic LOPs strongly loaded on the same PC in PCA analysis (
Section 3.3), it appears that the 4-oxo-
n-alkanal species monitored has a precursor that arises from the peroxidation of linolenoylglycerols, i.e., 4-oxo-(
E)-hexenal, which potentially generates 4-oxo-
n-hexanal at high temperatures.
Univariate ANOVA of the time-dependent aldehyde level datasets obtained from all culinary oils investigated demonstrated that differences between oil product and sampling time-point (0–90 min) mean values were both very highly significantly different for all 9 aldehydes individually evaluated (
p < 10
−6 in each case). Individual differences between these values for the different oils at each time-point, and between the time-points themselves, are also clearly visible in the mean ± SEM plots shown in
Figure 2;
post-hoc Bonferroni-corrected two sample comparisons made between their least square mean (LSM) values are reported in
Table S4 (Supplementary Materials). Notably, the culinary oil x time-point first-order interaction effect was also very highly significant for all aldehydes evaluated (
p < 10
−6 again), and this confirms the differential responses of aldehydic LOP concentration variables with respect to LSSFE heating time-point for each oil examined, as might be expected in view of their differing FA contents and hence differing patterns of LOPs generated (both primary and secondary), along with differing aldehydic LOP b.pts. This is also clearly apparent in
Figure 2. With the exception of avocado oil, all of the oils tested did not evolve any
1H NMR-detectable 4-oxo-
n-alkanals, LMM
n-alkanals, nor (
Z)-2-alkenals until the 60 min LSSFE time-point.
From these
post-hoc test results, it should be noted that soybean oil product SBO2 had least-squares mean (LSM) values that were not univariately significantly different from those of PUFA-rich corn oil for (
E)-2-alkenals, 4,5-epoxy-(
E)-2-alkenals, combined 4-hydroxy-/4-hydroperoxy-(
E)-2-alkenals,
n-alkanals, 4-oxo-
n-alkanals and (
Z)-2-alkenals. However, for both alka-2,4-dienal isomers (i.e., (
E,E)- and (
E,Z)-forms), which are PUFA- but not MUFA-derived secondary LOPs, significantly higher concentrations were found in the SBO2 product over that of corn oil. Moreover, as expected, in view of their relatively high ω-3 FA contents, both soybean oil products evaluated yielded significantly greater LMM
n-alkanal levels than that of corn oil, notably at the 60 and 90 min LSSFE time-points (
Table S4). For this LSM analysis, there was no significant difference observed between the two different soybean oils evaluated. Additionally, a UV statistical comparison of the ratios of LMM to high-molecular-mass
n-alkanal LOP concentrations produced at the 90 min LSSFE time-point demonstrated that the values for soybean oils SBO1 and SBO2 (0.040 and 0.052, respectively) were indeed both significantly higher than that found for corn oil (0.027),
p < 0.01 (ANOVA Welch test), and this again provided evidence that linolenoylglycerol species serve as substrates for the LMM homologues.
As expected from previous investigations [
1], (
E)-2-alkenals were only marginally generated up until the 90 min heating period in extra-virgin olive oils (
Figure 2), most notably in the Spanish product. Conversely, corn oil produced the highest level of this class of α,β-unsaturated aldehydes, with soybean and avocado oils also generating relatively high concentrations.
3.3. Multivariate Chemometric Analyses of Product-Dependent 1H NMR-Detectable Aldehydic LOP Concentration Signatures Found in Thermally Stressed Culinary Oil Products
Figure 3a shows ANOVA-based heatmaps of all aldehyde levels determined at the 90 min time-point only, and in addition to clearly demonstrating the much higher levels of these secondary LOPs generated in the two soybean and the single corn oil products examined, this also provides some evidence for product-distinctive patterns of these toxins, for example higher levels of LMM- and 4-oxo-
n-alkanals, the former known to arise from fragmentation of CHPDs generated from the primary peroxidation of ω-3 FAs in soybean oils [
1,
6]. The associated AHC analysis performed revealed two major analyte clusterings, with four sub-clusterings for one of these. The top left-hand side major cluster comprised (
E)-2- and (
Z)-2-alkenals only, which are expected to be associated since they are both PUFA-derived secondary LOPs [
23,
24], and the latter species has been postulated to arise from thermal isomerism of the former [
1,
25]; (
E)-2-alkenals may, however, also be generated from the peroxidation of MUFA substrates. Notwithstanding, the second major cluster contains all other aldehyde analyte variables. The downwards first (1), second (2), third (3) and fourth (4) sub-clusters within the bottom left-hand side major cluster feature (1) both (
E,E)- and (
E.Z)-alka-2,4-dienal isomers, which again could be related in view of a possible thermal isomeric interconversion process [
1]; (2)
n-alkanals alone, which are derived the peroxidation of both PUFA and MUFA sources; (3) 4,5-epoxy- and 4-hydroxy-/4-hydroperoxy-(
E)-2-alkenals, which both arise from PUFA peroxidation, the former from their corresponding (
E.E)-alka-2,4-dienal precursors; and (4) LMM- and 4-oxo-
n-alkanals, which presumably both arise from the peroxidation of ω-3 PUFAs in view of their strong positive correlation, as noted above.
However, it should also be noted that 2-heptenal isomers may be generated from alka-2,4-decadienal decomposition, together with acrolein, hexanal, acetaldehyde, butenal, 2-heptenal, 2-octenal, glyoxal, (
E)-2-buten-1,4-dial and benzaldehyde [
26]. Furthermore, alka-2,4-dienals may degrade to 2,3- or 4,5-epoxyaldehydes, which are then further degenerated to composites of either isomeric 2-octenals and acetaldehyde, or glyoxal and 2-octene [
27,
28].
Figure 3b, however, shows a corresponding heatmap of mean replicate aldehyde levels, in this case performed following the application of constant sum normalisation (CSN) in order to permit comparisons of proportionate contributions of aldehyde levels towards the total observed within each oil product studied, i.e., for the purpose of recognising any oil-dependent characteristic patterns of these LOPs. Indeed, this heatmap confirmed that at the 90 min LSSFE time-point, the soybean oil products contained proportionately higher levels of both (
E,E)- and (
E,Z)-alka-2,4-dienals, and 4,5-epoxy-(
E)-2-alkenals, the latter being derived from (
E,E)-alka-2,4-dienals [
17]. Moreover, LMM
n-alkanals were proportionately present at greater concentrations in both soybean oil products over those of all other oils investigated, and this is attributable to these oils’ considerably higher ω-3 PUFA contents. Additionally, one of the soybean oil products (SBO1) had very high proportionate levels of the 4-hydroxy-/4-hydroperoxy-(
E)-2-alkenal classification, although both soybean oil products contained similar proportionate concentrations of this class of aldehydes to those of the corn oil samples analysed. Similarly, the AHC analysis arising therefrom was found to be similar to that obtained without the use of CSN. Indeed, these clustering features again corresponded to 4,5-epoxy-(
E)-2-alkenals being derived from (
E,E)-alka-2,4-dienals [
17], ω-3 FA-sourced LMM
n-alkanals being associated with 4-oxo-
n-alkanals, high-molecular-mass
n-alkanals and (
E)-2-alkenals originating from both MUFA and PUFA sources, and (
Z)-2-alkenals arising from the thermally induced isomerisation of their corresponding (
E)-forms [
1].
Subsequently, PCA was performed on the
1H NMR-determined aldehydic LOP dataset, i.e., all 9 aldehydes monitored, and this included all 0–90 min sampling time-points involved for each of the 6 culinary oil products exposed to LSSFEs at 180 °C. The optimal model for this MV analysis, which was developed with Varimax rotation and Kaiser normalisation, contained 3 orthogonal factors (principal components, PCs,
Table 4). Of the 3 PCs isolable, the first, second and third accounted for 40.3, 32.2 and 25.6% of the total variance respectively (the fourth PC accounted for only 1.2%); these approximately corresponded to PC-loadings of 4, 3 and 2 aldehydic LOPs, respectively. Although, as expected, some aldehyde classifications loaded significantly on 2 or even all 3 PCs (detailed below), the strongest loadings for PC1 were found for (
E,E)- and (
E.Z)-2,4-alkadienals, combined 4-hydroxy-/4-hydroperoxy-(
E)-2-alkenals, and 4,5-epoxy-(
E)-2-alkenals; (
E)- and (
Z)-2-alkenals, and higher-molecular-mass
n-alkanals on PC2; and 4-oxo-
n-alkanals and LMM
n-alkanals on PC3. For PC1, these observations are again explicable by the reported route of generation of 4,5-epoxy-(
E)-2-alkenals from (
E.E)-2,4-alkadienals [
17], the possible thermal isomerism of this di-unsaturated aldehyde to its corresponding (
E,Z)- form [
1,
25], and presumably also a common linoleoylglycerol source for all PC1-loading aldehydes featured, including the 4-hydroxy-/4-hydroperoxy-(
E)-2-alkenal classification, in which case they would be present as nonenal homologues. For PC2, both (
E)-2-alkenals and
n-alkanals arise from the peroxidation of MUFAs as well as PUFAs, and (
Z)-2-alkenals are generated from the thermal isomerism of their (
E)-derivatives. Moreover, for PC3, LMM
n-alkanals arise from peroxidised linolenoylglycerol sources, and arguably also 4-oxo-
n-alkanal (as 4-oxo-
n-hexanal), as considered above.
Hence, despite at least some aldehydes exerting a significant loading vector impact on PCs other than that upon which they loaded the most strongly, it appears that those loading the strongest on PCs 1, 2 and 3 may originally arise from common sources, and these results are consistent with linoleoylglycerol, oleoylglycerol and linolenoylglycerol origins respectively, although the thermally-induced isomerism of selected aldehyde classes, and the further degradation of more structurally complex ones to simpler species [
1], clearly complicates this form of analysis and its interpretation. Such co-loadings of aldehyde predictor variables on up to two other PCs are explicable by (1) the differential UFA sources of differing homologues of the same classes of aldehydes, (e.g., (
E)-2-alkenals arising from the fragmentation of either CHPDs or HPMs, which are, in turn, derived from the peroxidation of PUFAs and MUFAs, respectively, and 4-hydroxy-(
E)-2-alkenals derived from either linoleoyl- or linolenoylglycerol sources, i.e., 4-hydroxy-(
E)-2-nonenal and -hexenal form the former, and 4-hydroxy-(
E)-2-heptenal and -pentenal from the latter); and (2) differential mechanisms available for their post-generation isomerism and/or decomposition.
For the 90 min LSSFE time-point dataset, both PLS-DA and OPLS-DA scores plots demonstrated a high level of discriminatory power between each oil type investigated (as shown for the latter in
Figure 4a). Indeed, although there was a small level of overlap between the CO1 and SBO1 sample clusters, aldehyde patterns arising from both soybean oil products remained distinct from all the other culinary oils evaluated. For this discrimination, cross-validation Q
2 and R
2Y values obtained for the OPLS-DA model were 0.73 and 0.85, respectively (permutation testing
p value <5.0 × 10
−4), and those for the PLS-DA model were 0.67 and 0.86 respectively (
p = 0.007). Considering that there were six different culinary oils for comparison, these values are indeed excellent, and demonstrate a high level of discriminatory potential for both MV analysis techniques employed. Variable importance parameters (VIPs) for the aldehyde predictor variables (
Figure 4b) were in the order (
E)-2-alkenals > (
Z)-2-alkenals >
n-alkanals > 4-hydroxy-/4-hydroperoxy-(
E)-2-alkenals > 4,5-epoxy-(
E)-2-alkenals (VIP values all >1), followed by 4-oxo-
n-alkanals > LMM
n-alkanals > (
E,E)-alka-2,4-dienals (VIP values 0.70–0.92). Since (
E,Z)-alka-2,4-dienals had a VIP parameter of only <0.40, its overall discriminatory power was of little or no value.
However, one cautionary note and limitation of the work performed in the current study is that the ω-3 PUFA peroxidation product propanal has a very low b.pt (only 49 °C) [
2], and therefore levels monitored in thermally stressed culinary oil products, particularly soybean oils, are likely to represent only a small or very small proportion of the total generated from their exposure to the thermo-oxidative LSSFEs featured. In point of fact, our solution-state
1H NMR studies only detect and determine the amounts of aldehydic LOPs which are remaining in these oil products following their exposure to high-temperature LSSFEs, a process which clearly gives rise to the vaporisation of many of them, most especially those with b.pts which lie some way below that of the thermal stressing temperature employed (180 °C), as reviewed in [
2]. Nevertheless, these ‘oil solution’ forms of aldehydes largely represent the fractions of these toxins which are transferable to fried foods during such thermo-oxidative episodes, and arguably also those which are ingestible by humans when consuming fried foods such as potato chips [
1,
2]. As expected, potato chip levels of nascent aldehydic LOP toxins appear to be linearly proportional to the mass of fryer-used culinary oils taken up by these commonly consumed foods throughout the frying process [
1].
3.4. Overview of Experimental Results Obtained
One major objective of the current study was to conduct both UV and MV 1H NMR-based chemometrics analysis in order to explore the acylglycerol FA sources of aldehydic LOPs, and employ ‘state-of-the-art’ chemometrics strategies to distinguish between the signatures of aldehydic LOPs generated in thermally stressed ω-3 FA-containing soybean oils from those yielded from the thermo-oxidation of other culinary frying oils, including an ω-3 FA-deplete culinary oil with a similar total PUFA content (corn oil).
Whereas ω-6 and ω-3 FAs are both present in in natural and refined soybean oil products, the major ω-3 FA therein, i.e., linolenic acid, thermo-oxidatively deteriorates more rapidly, and to a greater extent than ω-6 FAs. Moreover, the homologue status and patterns of different classes of LOPs generated therefrom, notably aldehydes, are clearly distinct from those arising from the peroxidation of ω-6 FAs [8.9]. Hence, the breakdown of these highly peroxidatively-susceptible FAs in soybean oils during their exposure to standard frying practices was expected to give rise to a differential pattern and higher levels of aldehydic LOPs when compared to those derived from ω-6 FAs (predominantly linoleic acid) on a molar equivalent basis [
1,
8].
Herein, we found that the relatively higher ω-3 FA contents of the soybean oil products tested generated higher levels of LMM
n-alkanal LOPs over those of all other oils tested, although this observation was more pronounced for one of the two products investigated here (SBO2,
Figure 2). Since both these oil products have very similar FA contents, this observation may be ascribable to higher levels of chain-breaking antioxidants present in the more peroxidation-resistant brand (SBO1), which was manufactured by an expeller-pressing process, and therefore may be expected to contain larger amounts of these protective agents. However, levels of both α- and ω-tocopherols in refined soybean oils are sub-millimolar (
Section 3.6), and the markedly lower concentration of the former has indicated that such oil products may not represent an acceptable source of dietary vitamin E [
29]. Nevertheless, such antioxidant levels may indeed offer at least some health and storage (shelf-life longevity) benefits to consumers.
Overall, when the PUFA and MUFA contents of cooking oils are higher and lower respectively in culinary oil products, greater levels of aldehydes are produced. As anticipated, all aldehydes derived from PUFA peroxidation alone, along with those arising from both MUFA and PUFA degeneration such as (E)-2-alkenals, were increased in concentration with increasing oil PUFA content in view of their more rapid rate of peroxidation over that of MUFAs, and also an increased rate of decomposition of primary CHPDs than that of HPM peroxidation products.
However, 2-alkenals and alka-2,4-dienals have also been shown to thermally degrade at a temperature of 200 °C. Indeed, Zamora et al. [
30] explored the thermally-induced decomposition of 2-pentenal and -octenal, and 2,4-heptadienal and 2,4-decadienal, in order to determine their stability at this high temperature, and also to identify and monitor the levels of products arising therefrom. Results acquired demonstrated that both these classes of aldehydes rapidly degraded in the presence of air and an aqueous buffering system, generating formaldehyde and acetaldehyde, along with products arising from >CH=CH< bond cleavage: specifically, propanal and hexanal from 2-pentenal and 2-octenal, respectively, and propanal/2-pentenal and hexanal/2-octenal admixtures from 2,4-heptadienal and 2,4-decadienal, respectively (glyoxal and fumaraldehyde were also significant degradation products). Therefore, propanal, which arises as a secondary LOP from the peroxidation of linolenoylglycerol substrates, may also be yielded from the thermally induced decomposition of either 2-pentenal or 2,4-heptadienal. Fortunately, both these aldehydes are also derived from ω-3 FA peroxidation [
15,
31,
32]. For the studies reported in Ref. [
30], the activation energy of >CH=CH< double bond cleavage was found to be quite low (∼25 kJ/mol), and overall, the levels of alkanals generated (10–18%) were greater than that of 2-alkenals (∼1%). These results are comparable to those reported in references [
27] and [
28].
Although the above thermally-induced decompositions of higher homologue α,β-unsaturated aldehydes to lower ones is promoted by exposure to atmospheric O
2, and the presence of an aqueous buffering system, it should be noted that refined, unused culinary vegetable oils usually contains a water content of approximately 0.1% (
w/
w), and more importantly used frying oil contains 0.3–1.5% (
w/
w) of it at standard frying temperatures [
33]. Hence, it is anticipated that culinary oil water may strongly facilitate these reactions during standard frying practices, most especially in water-rich foods fried in such oil media.
1H NMR analysis performed at an operating frequency of 600 MHz can simultaneously identify and quantify a range of aldehydic LOPs (up to 10 noted in this study), and therefore provides a greater overall evaluation of the oxidative deterioration status of oils exposed to standard or unusual frying practices than that conducted at lower field strengths, e.g., at only 400 MHz. Moreover, high-resolution NMR spectroscopy can successfully resolve resonances arising from different geometric (configurational) isomers of different classes of α,β-unsaturated aldehydes, and has the ability to simultaneously determine the nature and level of UFA acyl chain degradations (
Table S1 and
Figure S1).
3.6. Deleterious Health Effects Potentially Arising from the Ingestion or Inhalation of Toxic Aldehydic LOPs, and Strategies for Their Circumvention in Soybean Frying Oils
In this investigation, high-resolution
1H NMR analysis has established that higher levels of aldehydes are observed in PUFA-rich rather than MUFA-rich oils, as expected [
36,
37,
38]. Moreover, the pattern of aldehydic LOPs generated in soybean oils was found to differ significantly from that of corn oil, which has a similar total PUFA, but much lower ω-3 FA content. The deleterious health risks presented by the ingestion of high concentrations of aldehydes, e.g., those present in fried foods such as French fries (which contain up to 30% (
w/
w) or even higher quantities of LOP-containing pre-heated oils [
39]), or alternatively directly inhaled in their vapourised forms, afford significant concerns to both consumers and restaurant employees respectively.
Studies dating back over the past 25 years or so [
1,
2,
3,
39,
40,
41] have shown that the ingestion of dietary aldehydic LOPs is associated with the induction, development and/or perpetuation of deleterious health effects in humans dietarily ingesting them in foods, particularly fried foods, and these include neurodegenerative disorders [
42], atherosclerosis and its cardiovascular sequelae [
43,
44], cancer [
45] and liver damage [
46], for example, together with a range of further non-communicable diseases [
34]. Globally, cardiovascular diseases (CVDs) have remained the leading cause of death in humans for the past 15 years [
47]. Moreover, fried food intakes, which are known to contain aldehydic LOPs [
1,
39], have been correlated with all-cause, cancer and CVD mortalities in a recent prospective cohort study [
48]. Cytotoxic and genotoxic aldehydes generated from culinary frying or cooking practices have previously been shown to be absorbed from the gut into the systemic circulation in vivo [
40], where they may gain access to critical organs and tissues which potentially serve as toxicological targets. Observations within eukaryotic models have also observed that aldehydes cause oxidative stress damage to cells, and chemical modification of DNA [
49]. Indeed, the biomolecularly-destructive effects of aldehyde species have been demonstrated in animal models as histopathological modifications of the liver, colon and jejunum [
46], and that report confirmed that these essential organs were damaged following the oral administration of pre-heated, PUFA-rich cooking oils, but not of corresponding unheated products. Moreover, once absorbed into the systemic circulation, these aldehydes have the ability to propagate foam cell formation from macrophages, a critical stage in the pathogeneses of atherosclerosis [
40,
44]. Furthermore, it has been demonstrated in a rat model that the ingestion of LOPs administered as thermally-stressed safflower oil markedly enhanced the occurrence of malformations in embryos [
50], i.e., they exert teratogenic properties.
Of much interest, 4-ONE, the unsaturated derivative of 4-oxo-(
E)-nonanal, and generated in thermally stressed soybean oils (putatively as the 4-oxo-(
E)-hexanal homologue from peroxidised linolenoylglycerols), has been found to exert a higher level of toxicity than the much more highly studied 4-HNE in a number of experimental models. For example, data available in reference [
51] revealed that 4-ONE potently alters mitochondrial function by acting as an effector of lipid peroxidation within this environment.
Intriguingly, the inhalation of vapourised aldehydes generated during high-temperature frying or wok-cooking practices has been correlated with a higher prevalence of bronchitis in epidemiological studies [
52]. Indeed, it was found that women aged 20–40 who had been exposed to cooking oil fumes >21 times per week had a >4-fold higher risk of developing chronic bronchitis over those who were exposed <14 times a week. Epidemiological studies performed on non-smoking Taiwanese women have demonstrated an increased incidence of lung cancer amongst those who cooked more meals per day [
53]. Intriguingly, it was also noted that the risk of lung cancer was higher if women waited until visible fumes were produced, prior to cooking episodes [
53]. Deaths attributable to lung cancer have also been ranked as a leading cause of mortality worldwide (World Health Organisation 2016) [
47], and this study therefore provided a further understanding of alternative, non-tobacco smoking, causes of this condition. Notably, in 2016, soybean oil was rated the most commonly consumed vegetable oil in China (44%), with rapeseed, palm and peanut oils rating second, third and fourth (24, 18 and 9%), respectively [
54]. Elevated levels of urinary oxidative stress biomarkers, specifically 1-hydroxypyrene, malondialdehyde (MDA) and 8-hydroxy-2′-deoxyguanosine, were observed in restaurant workers routinely exposed to cooking oil fumes [
55,
56]; MDA presumably at least partially arises from cooking oil fume ingestion, since this aldehyde is a well-known cytotoxic and genotoxic LOP arising from the oxidation of ω-3 FAs [
2]. Indeed, a significant proportion of aldehydic LOPs have boiling-points (b.pts) close to, below or much below those of standard frying temperatures (
ca. 180 °C) [
2]. In 2013, Lee and Gany [
57] conducted a literature review of the risks of lung cancer associated with cooking oil fume inhalation, and they found that all investigations which explored the mechanisms associated with these increased risks uncovered evidence for mutagens and/or carcinogens in cooking oil fume extracts, and/or relevant molecular routes for DNA damage or carcinogenesis. These researchers therefore strongly concluded that further studies should be performed in order to further explore at-risk groups based in the USA, along with the development and validation of powerful interventions focused on the curtailment of these risks.
From the results acquired in the current study, PUFA-rich oils, notably soybean and corn oils, should not be recommended for high-temperature frying practices in view of their high susceptibilities to peroxidative degradation. Indeed, we recommend that MUFA-rich oils, including extra-virgin olive and avocado oils, should be utilised for industrial, restaurant-based and domestic frying practices instead because of their relatively high resistance against thermally-induced peroxidation.
Evidence presented in the current study suggests that the employment of genetically engineered, high oleoylglycerol content soybean oils would be more suitable for high-temperature culinary frying or cooking purposes, in view of substantial reductions in PUFA content and consequently also those of toxic aldehydes formed during such frying episodes. By silencing the GmFAD3 gene, both linolenic acid and SFA contents can be reduced in soybean oil, a process which increases its stability and resistance to thermally- induced oxidation [
58]. Alternatively, in order to prevent aldehyde formation, changes in cooking practices, including the restriction of O
2 from the system, for example, via the instigation of vacuum frying techniques, would also serve to markedly reduce the formation of aldehydes and other LOPs during the thermal stressing of such frying oils. This methodology has been shown to reduce the contents of acrylamide in potato crisps by 98%, and also lowered polymeric TAG species in culinary oils evaluated [
59]. Results obtained by Albertos et al. [
60] corroborate the benefits of vacuum frying practices, including the diminished degradation of ω-3 FAs and tocopherols, as well as reduced concentrations of lipid hydroperoxides and their carbonyl fragmentation products. In view of the lower temperatures used during these cooking practices, many ’health-friendly’ agents, including chain-breaking antioxidants and ω-3 FAs themselves remain relatively thermally stable. A further approach is pressure frying, a strategy which has been adopted for the preparation of fried chicken products in numerous commercial restaurants focused on such fast-food commodities [
61]. However, the cost and required user-knowledge and familiarity with these frying techniques represent two factors which may render their instigation and common use unattractive to fast-food vendors, especially those managing small, non-global restaurants.
Another alternative involves the blending of two oils, for example one high in PUFAs, such as soybean oil, and others high in MUFAs such as camellia oil, in order to reduce lipid peroxidation and hence enhance frying stability. This has been explored when these two oils underwent deep-frying events, and results acquired showed that the peroxide and
p-anisidine values of such blends were diminished significantly with higher camellia:soybean oil ratios [
62].
An additional strategy features the supplementation of soybean oil products with dietary lipid-soluble, peroxyl radical-scavenging chain-breaking antioxidants. However, this vegetable oil already contains naturally occurring tocopherols (tocopherols and tocotrienols are present in different classes and sources of vegetable oils at a range of concentrations [
29]). The antioxidant effectiveness of these agents depends on their derivative (aromatic methyl substituent positional) forms, and their relative activities in this context are, of course, concentration-dependent [
63]. Typically, soybean oil contains higher and lower levels of the γ- and α-tocopherol forms, respectively, than those found in other PUFA-rich oils such as sunflower and rapeseed oils, although these levels are comparable to those found in corn oil [
63]. However, previous investigations have demonstrated that tocopherols and other phenolic antioxidants are only poorly protective against the peroxidation of PUFAs taking place during high-temperature frying episodes, a consequence of (1) their inabilities to combat the vast thermally induced peroxidative cascade of autocatalytic free radical reactions occurring therein, especially when present at relatively low concentrations, which are insufficient to counter these effects, and (2) their thermal instabilities at these temperatures [
3,
19,
40]. Moreover, tocopherols have b.pts which are not much greater than that of standard frying temperature [
40], and therefore significant evaporative losses of these antioxidants can result from their exposure to standard frying temperatures. Alpha- and γ-tocopherol levels present in soybean oil have been reported as 71 ± 6 and 273 ± 11 mg/kg, i.e., 0.13 and 0.51 mmol./kg, respectively [
29], values which are notably lower than those of other cooking oils, and which from the results acquired in the current study, are clearly insufficient to retard the peroxidation process during high-temperature frying practices.
Nevertheless, polyphenols, which are powerful chain-breaking antioxidants, have been shown to offer strong protective activities against the thermo-oxidation of culinary oil PUFAs [
64], and also suppress the thermal degradation of α-tocopherol at temperatures of 100–120 °C [
65], and therefore supplementation of natural soybean oil samples with such agents may indeed serve to enhance the their safety margins when employed for domestic or commercial frying operations, which are usually conducted at 160–190 °C, although for standard frying practices it is 180 °C [
30]. Indeed, using the Rancimat technique for evaluating the oxidative stability of culinary oils, it has been revealed that supplementation of edible vegetable oils with a ‘spectrum’ of polyphenols contained within an olive leaf extract increased the oil deterioration induction time from 19–54% [
64], the highest increase being observed with sunflower oil, for which a ‘protection factor’ of 1.54 was recorded.
Although quite rich in tocopherols, vegetable seed oils frequently employed for high-temperature frying purposes are, unfortunately, almost completely devoid of other phenolic antioxidant compounds, and therefore their supplementation with such polyphenolic antioxidants, for example those present in olive leaf extracts [
64], may indeed be valuable for the protection of UFAs therein against adventitious peroxidation during periods of storage; such an approach may even offer some level of protection of UFA-rich frying oils when exposed to high-temperature frying episodes. However, olive oil embodies both tocopherols and phenolic compounds as antioxidants, especially the extra-virgin forms which are mainly used as salad oils. Hence, in addition to their high MUFA contents, phenolic and polyphenolic antioxidants contained within the two extra-virgin olive oil products investigated in the current study may also serve to enhance their resistivities against peroxidative degradation. Following ingestion, polyphenols also putatively act as preventative agents against a range of degenerative diseases such as atherosclerosis and selected cancers, together with systemic inflammatory conditions, all of which involve excessive ‘oxidative stress’ inputs in vivo [
66].