Rosemary Essential Oils as a Promising Source of Bioactive Compounds: Chemical Composition, Thermal Properties, Biological Activity, and Gastronomical Perspectives
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. GC/MS Analysis
2.3. Contents of Major and Trace Elements
2.4. Thermal Analysis
2.4.1. Thermal Characterization of EO
2.4.2. Oxidative Stability of Sunflower Oil with Different Share of EO
2.5. Antimicrobial Activity of Samples
2.6. Statistical Analysis
3. Results
3.1. Chemical Profiles of Essential Oils
3.2. Antimicrobial Activity of Essential Oils
3.3. Thermal Analysis of Essential Oils
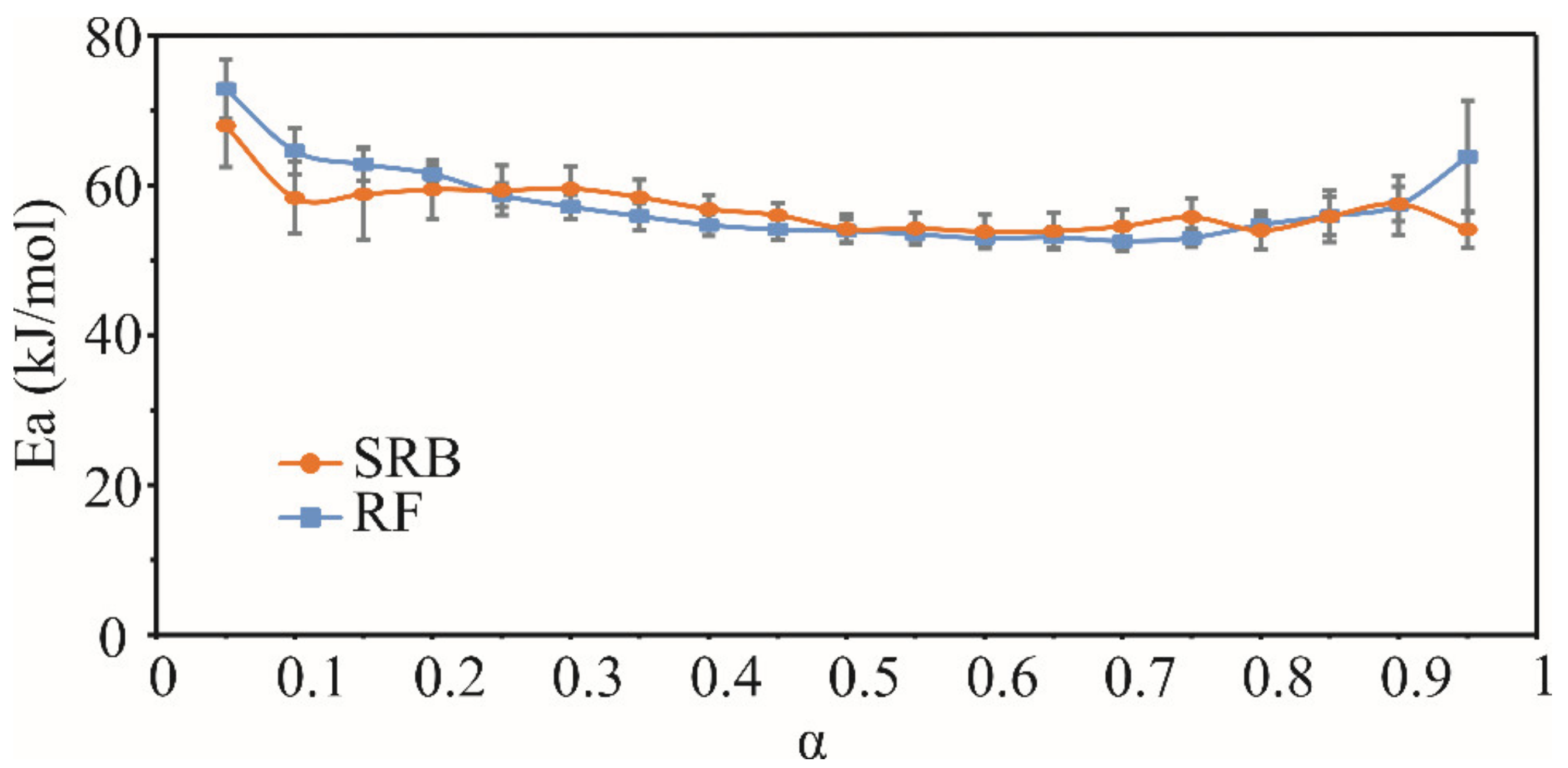
3.3.1. Thermal Properties
3.3.2. The Effect of Added RF and SRB EO on the Oxidative Stability of Sunflower Oil
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zaouali, Y.; Bouzaine, T.; Boussaid, M. Essential oils composition in two Rosmarinus officinalis L. varieties and incidence for antimicrobial and antioxidant activities. Food Chem. Toxicol. 2010, 48, 3144–3152. [Google Scholar] [CrossRef] [PubMed]
- Lemos, M.F.; Lemos, M.F.; Pacheco, H.P.; Endringer, D.C.; Scherer, R. Seasonality modifies rosemary’s composition and biological activity. Ind. Crops Prod. 2015, 70, 41–47. [Google Scholar] [CrossRef]
- Jordán, M.J.; Lax, V.; Rota, M.C.; Lorán, S.; Sotomayor, J.A. Effect of the phenological stage on the chemical composition, and antimicrobial and antioxidant properties of Rosmarinus officinalis L essential oil and its polyphenolic extract. Ind. Crops Prod. 2013, 48, 144–152. [Google Scholar] [CrossRef]
- Bajalan, I.; Rouzbahani, R.; Pirbalouti, A.G.; Maggi, F. Antioxidant and antibacterial activities of the essential oils obtained from seven Iranian populations of Rosmarinus officinalis. Ind. Crops Prod. 2017, 107, 305–311. [Google Scholar] [CrossRef]
- Pellegrini, M.; Ricci, A.; Serio, A.; Chaves-López, C.; Mazzarrino, G.; D’Amato, S.; Lo Sterzo, C.; Paparella, A. Characterization of Essential Oils Obtained from Abruzzo Autochthonous Plants: Antioxidant and Antimicrobial Activities Assessment for Food Application. Foods 2018, 7, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teixeira, B.; Marques, A.; Ramos, C.; Neng, N.R.; Nogueira, J.M.F.; Saraiva, J.A.; Nunes, M.L. Chemical composition and antibacterial and antioxidant properties of commercial essential oils. Ind. Crops Prod. 2013, 43, 587–595. [Google Scholar] [CrossRef]
- Karakaya, S.; El, S.N.; Karagozlu, N.; Sahin, S.; Sumnu, G.; Bayramoglu, B. Microwave-assisted hydrodistillation of essential oil from rosemary. J. Food Sci. Technol. 2014, 51, 1056–1065. [Google Scholar] [CrossRef] [Green Version]
- Bousbia, N.; Abert Vian, M.; Ferhat, M.A.; Petitcolas, E.; Meklati, B.Y.; Chemat, F. Comparison of two isolation methods for essential oil from rosemary leaves: Hydrodistillation and microwave hydrodiffusion and gravity. Food Chem. 2009, 114, 355–362. [Google Scholar] [CrossRef]
- Wang, D.; Fan, W.; Guan, Y.; Huang, H.; Yi, T.; Ji, J. Oxidative stability of sunflower oil flavored by essential oil from Coriandrum sativum L. during accelerated storage. LWT 2018, 98, 268–275. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Zhang, Y.; Zu, Y.; Yang, L.; Lu, Q.; Wang, W. Antioxidant effects of rosemary extracts on sunflower oil compared with synthetic antioxidants. Int. J. Food Sci. Technol. 2014, 49, 385–391. [Google Scholar] [CrossRef]
- Mezza, G.N.; Borgarello, A.V.; Grosso, N.R.; Fernandez, H.; Pramparo, M.C.; Gayol, M.F. Antioxidant activity of rosemary essential oil fractions obtained by molecular distillation and their effect on oxidative stability of sunflower oil. Food Chem. 2018, 242, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Kamkar, A.; Javan, A.J.; Asadi, F.; Kamalinejad, M. The antioxidative effect of Iranian Mentha pulegium extracts and essential oil in sunflower oil. Food Chem. Toxicol. 2010, 48, 1796–1800. [Google Scholar] [CrossRef]
- Lercker, G.; Rodriguez-Estrada, M.T. Cholesterol oxidation mechanisms. In Cholesterol and Phytosterol Oxidation Products: Analysis, Occurrence, and Biological Effects; Guardiola, F., Dutta, P.C., Codony, R., Savage, G.P., Eds.; AOCS Press: Champaign, IL, USA, 2002; pp. 1–25. [Google Scholar]
- Sikwese, F.E.; Duodu, K.G. Antioxidant effect of a crude phenolic extract from sorghum bran in sunflower oil in the presence of ferric ions. Food Chem. 2007, 104, 324–331. [Google Scholar] [CrossRef] [Green Version]
- Padmashree, A.; Roopa, N.; Semwal, A.D.; Sharma, G.K.; Agathian, G.; Bawa, A.S. Star-anise (Illicium verum) and black caraway (Carum nigrum) as natural antioxidants. Food Chem. 2007, 104, 59–66. [Google Scholar] [CrossRef]
- Wang, D.; Meng, Y.; Zhao, X.; Fan, W.; Yi, T.; Wang, X. Sunflower oil flavored by essential oil from Punica granatum cv. Heyinshiliu peels improved its oxidative stability and sensory properties. LWT 2019, 111, 55–61. [Google Scholar] [CrossRef]
- Micić, D.; Ostojić, S.; Pezo, L.; Blagojević, S.; Pavlić, B.; Zeković, Z.; Đurović, S. Essential oils of coriander and sage: Investigation of chemical profile, thermal properties and QSRR analysis. Ind. Crops Prod. 2019, 138, 111438. [Google Scholar] [CrossRef]
- Šojić, B.; Pavlić, B.; Tomović, V.; Ikonić, P.; Zeković, Z.; Kocić-Tanackov, S.; Đurović, S.; Škaljac, S.; Jokanović, M.; Ivić, M. Essential oil versus supercritical fluid extracts of winter savory (Satureja montana L.)—Assessment of the oxidative, microbiological and sensory quality of fresh pork sausages. Food Chem. 2019, 287, 280–286. [Google Scholar] [CrossRef]
- Friedman, H.L. Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Application to a phenolic plastic. J. Polym. Sci. Part C Polym. Symp. 1964, 6, 183–195. [Google Scholar] [CrossRef]
- Vyazovkin, S.; Chrissafis, K.; Di Lorenzo, M.L.; Koga, N.; Pijolat, M.; Roduit, B.; Sbirrazzuoli, N.; Suñol, J.J. ICTAC Kinetics Committee recommendations for collecting experimental thermal analysis data for kinetic computations. Thermochim. Acta 2014, 590, 1–23. [Google Scholar] [CrossRef]
- Micić, D.M.; Ostojić, S.B.; Simonović, M.B.; Krstić, G.; Pezo, L.L.; Simonović, B.R. Kinetics of blackberry and raspberry seed oils oxidation by DSC. Thermochim. Acta 2015, 601, 39–44. [Google Scholar] [CrossRef]
- Riabov, P.A.; Micić, D.; Božović, R.B.; Jovanović, D.V.; Tomić, A.; Šovljanski, O.; Filip, S.; Tosti, T.; Ostojić, S.; Blagojević, S.; et al. The chemical, biological and thermal characteristics and gastronomical perspectives of Laurus nobilis essential oil from different geographical origin. Ind. Crops Prod. 2020, 151, 112498. [Google Scholar] [CrossRef]
- Pavlić, B.; Teslić, N.; Vidaković, A.; Vidović, S.; Velićanski, A.; Versari, A.; Radosavljević, R.; Zeković, Z. Sage processing from by-product to high quality powder: I. Bioactive potential. Ind. Crops Prod. 2017, 107, 81–89. [Google Scholar] [CrossRef]
- Bozin, B.; Mimica-Dukic, N.; Samojlik, I.; Jovin, E. Antimicrobial and Antioxidant Properties of Rosemary and Sage (Rosmarinus officinalis L. and Salvia officinalis L., Lamiaceae) Essential Oils. J. Agric. Food Chem. 2007, 55, 7879–7885. [Google Scholar] [CrossRef]
- Gachkar, L.; Yadegari, D.; Rezaei, M.; Taghizadeh, M.; Astaneh, S.; Rasooli, I. Chemical and biological characteristics of Cuminum cyminum and Rosmarinus officinalis essential oils. Food Chem. 2007, 102, 898–904. [Google Scholar] [CrossRef]
- Stephanos, J.J.; Addison, A.W. Chemistry of Metalloproteins: Problems and Solutions in Bioinorganic Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2014. [Google Scholar]
- Đurović, S.; Pavlić, B.; Šorgić, S.; Popov, S.; Savić, S.; Petronijević, M.; Radojković, M.; Cvetanović, A.; Zeković, Z. Chemical composition of stinging nettle leaves obtained by different analytical approaches. J. Funct. Foods 2017, 32, 18–26. [Google Scholar] [CrossRef]
- Roat-Malone, R.M. Bioinorganic Chemistry: A Short Course, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2007. [Google Scholar]
- USDA–NAL. Dietary Reference Intakes (DRIs): Estimated Average Requirements. Available online: https://www.nal.usda.gov/sites/default/files/fnic_uploads/recommended_intakes_individuals.pdf (accessed on 8 February 2021).
- Abdollahi, M.; Rezaei, M.; Farzi, G. A novel active bionanocomposite film incorporating rosemary essential oil and nanoclay into chitosan. J. Food Eng. 2012, 111, 343–350. [Google Scholar] [CrossRef]
- Deba, F.; Xuan, T.D.; Yasuda, M.; Tawata, S. Chemical composition and antioxidant, antibacterial and antifungal activities of the essential oils from Bidens pilosa Linn. var. Radiata. Food Control 2008, 19, 346–352. [Google Scholar] [CrossRef]
- Griffin, S.G.; Wyllie, S.G.; Markham, J.L.; Leach, D.N. The role of structure and molecular properties of terpenoids in determining their antimicrobial activity. Flavour Fragr. J. 1999, 14, 322–332. [Google Scholar] [CrossRef]
- Zengin, H.; Baysal, A. Antibacterial and antioxidant activity of essential oil terpenes against pathogenic and spoilage-forming bacteria and cell structure-activity relationships evaluated by SEM microscopy. Molecules 2014, 19, 17773–17798. [Google Scholar] [CrossRef] [Green Version]
- Trombetta, D.; Castelli, F.; Sarpietro, M.G.; Venuti, V.; Cristani, M.; Daniele, C.; Saija, A.; Mazzanti, G.; Bisignano, G. Mechanisms of Antibacterial Action of Three Monoterpenes. Antimicrob. Agents Chemother. 2005, 49, 2474–2478. [Google Scholar] [CrossRef] [Green Version]
- Uribe, S.; Ramirez, J.; Peña, A. Effects of beta-pinene on yeast membrane functions. J. Bacteriol. 1985, 161, 1195–1200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uribe, S.; Pena, A. Toxicity of allelopathic monoterpene suspensions on yeast dependence on droplet size. J. Chem. Ecol. 1990, 16, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Sikkema, J.; de Bont, J.A.; Poolman, B. Interactions of cyclic hydrocarbons with biological membranes. J. Biol. Chem. 1994, 269, 8022–8028. [Google Scholar] [CrossRef]
- Andrade-Ochoa, S.; Nevárez-Moorillón, G.V.; Sánchez-Torres, L.E.; Villanueva-García, M.; Sánchez-Ramírez, B.E.; Rodríguez-Valdez, L.M.; Rivera-Chavira, B.E. Quantitative structure-activity relationship of molecules constituent of different essential oils with antimycobacterial activity against Mycobacterium tuberculosis and Mycobacterium bovis. BMC Complement. Altern. Med. 2015, 15, 332. [Google Scholar] [CrossRef] [Green Version]
- Bhavaniramya, S.; Vishnupriya, S.; Al-Aboody, M.S.; Vijayakumar, R.; Baskaran, D. Role of essential oils in food safety: Antimicrobial and antioxidant applications. Grain Oil Sci. Technol. 2019, 2, 49–55. [Google Scholar] [CrossRef]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial Activity of Some Essential Oils-Present Status and Future Perspectives. Medicines 2017, 4, 58. [Google Scholar] [CrossRef] [Green Version]
- ChemSpider|Search and Share Chemistry. Available online: http://www.chemspider.com (accessed on 8 February 2021).
- Vyazovkin, S.; Burnham, A.K.; Criado, J.M.; Pérez-Maqueda, L.A.; Popescu, C.; Sbirrazzuoli, N. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim. Acta 2011, 520, 1–19. [Google Scholar] [CrossRef]
- Cheng, Y.; Huang, Y.; Alexander, K.; Dollimore, D. A thermal analysis study of methyl salicylate. Thermochim. Acta 2001, 367–368, 23–28. [Google Scholar] [CrossRef]
- Prado, J.R.; Vyazovkin, S. Activation energies of water vaporization from the bulk and from laponite, montmorillonite, and chitosan powders. Thermochim. Acta 2011, 524, 197–201. [Google Scholar] [CrossRef]
- Özcan, M.M.; Arslan, D. Antioxidant effect of essential oils of rosemary, clove and cinnamon on hazelnut and poppy oils. Food Chem. 2011, 129, 171–174. [Google Scholar] [CrossRef]
- Özcan, M.; Akgül, A. Antioxidant activity of extracts and essential oils from Turkish spices on sunflower oil. Acta Aliment. 1995, 24, 81–90. [Google Scholar]
- Wojtunik, K.A.; Ciesla, L.M.; Waksmundzka-Hajnos, M. Model Studies on the Antioxidant Activity of Common Terpenoid Constituents of Essential Oils by Means of the 2,2-Diphenyl-1-picrylhydrazyl Method. J. Agric. Food Chem. 2014, 62, 9088–9094. [Google Scholar] [CrossRef] [PubMed]


| Compound | SRB | RF | ||
|---|---|---|---|---|
| Content (%) | Content (mg/g EO) | Content (%) | Content (mg/g EO) | |
| Cyclic monoterpenes | ||||
| Myrcene | 2.43 | 3.9 ± 0.1 a | 0.05 | 0.15 ± 0.02 b |
| Cyclic monoterpenes | ||||
| α-Thujene | 0.39 | - | 0.55 | - |
| α-Pinene | 23.00 | 282 ± 5 a | 17.76 | 122 ± 1 b |
| α-Fenchene | 0.09 | - | 0.22 | - |
| Camphene | 9.99 | - | 8.36 | - |
| β-Pinene | 3.49 | 49.9 ± 0.9 a | 5.19 | 37.7 ± 0.2 b |
| α-Phellandrene | 0.59 | - | N.D. * | - |
| β-Phellandrene | 0.13 | - | N.D. | - |
| α-Terpinene | 0.25 | 26.4 ± 0.3 | ND. | - |
| Limonene | 4.18 | 63.4 ± 0.5 a | 3.11 | 27.4 ± 0.3 b |
| γ-Terpinene | 0.43 | 5.1 ± 0.1 | N.D. | - |
| Terpinolene | 0.48 | - | N.D. | - |
| Acyclic oxygenated mnoterpenes | ||||
| Linalool | 0.85 | 15.6 ± 0.2 a | 0.84 | 10.0 ± 0.1 b |
| Cyclic oxygenated monoterpenes | ||||
| Eucalyptol | 17.79 | 177 ± 3 a | 23.40 | 169 ± 2 b |
| α-Pinene oxide | N.D. | - | 0.47 | - |
| Fenchone | 0.01 | 0.02 ± 0.00 b | 0.02 | 0.86 ± 0.05 a |
| α-Campholenal | N.D. | - | 0.58 | - |
| Isothujol | 0.02 | - | N.D. | - |
| Camphor | 14.39 | 149 ± 3 a | 17.17 | 104 ± 2 b |
| Bornyl acetate | 2.39 | 22.5 ± 0.3 a | 3.31 | 19.3 ± 0.1 b |
| Pinocarvone | N.D. | - | 0.20 | - |
| Terpinen-4-ol | 1.19 | 14.8 ± 0.1 | N.D. | - |
| Myrtenal | 0.04 | - | 0.33 | - |
| cis-Sabinol | 0.08 | - | 0.14 | - |
| Isoborneol | N.D. | - | 0.11 | 0.61 ± 0.02 |
| α-Terpineol | 2.30 | 51.0 ± 0.5 | N.D. | - |
| Borneol | 2.39 | 20.2 ± 0.2 b | 4.56 | 27.7 ± 0.3 a |
| Carvone | N.D. | - | 0.07 | 3.86 ± 0.06 |
| Myrtenol | 0.21 | - | 0.25 | - |
| trans-Carveol | 0.17 | - | 0.05 | - |
| p-Cymene-8-ol | N.D. | - | 0.08 | - |
| Verbenone | 2.33 | - | 0.30 | - |
| cis-Verbenol | 0.15 | - | 0.25 | - |
| Cyclic aromatic monoterpenes | ||||
| p-Cymene | 4.51 | 33.6 ± 0.4 b | 10.91 | 41.3 ± 0.4 a |
| m-Cymene | N.D. | - | 0.02 | 0.07 ± 0.01 |
| Sesquiterpenes | ||||
| α-Copaene | 0.18 | - | 0.03 | - |
| α-Cubebene | 0.12 | - | 0.07 | - |
| trans-β-Caryophyllene | 3.22 | 29.7 ± 0.2 a | 0.63 | 4.06 ± 0.09 b |
| Humulene | 1.22 | - | N.D. | - |
| Caryophyllene oxide | 0.47 | - | 0.97 | - |
| Other | ||||
| 2-Methyl-3-octanone | 0.44 | - | N.D. | - |
| 3-Octanol | 0.03 | - | N.D. | - |
| 1-Octen-3-ol | 0.08 | - | N.D. | - |
| Element (mg/kg) | SRB | RF |
|---|---|---|
| Bulk elements | ||
| K | 1.14 ± 0.08 a | 1.26 ± 0.05 a |
| Na | 8.46 ± 0.15 a | 8.17 ± 0.36 a |
| Mg | 1.18 ± 0.09 a | 0.89 ± 0.01 b |
| Ca | 7.21 ± 0.23 b | 9.90 ± 0.20 a |
| Trace elements | ||
| Co | 0.032 ± 0.001 | N.D. * |
| Cr | 9.50 ± 0.02 a | 0.025 ± 0.002 b |
| Cu | 0.132 ± 0.009 a | 0.041 ± 0.005 b |
| Fe | 5.66 ± 0.04 a | 1.58 ± 0.17 b |
| Li | 0.022 ± 0.002 a | 0.012 ± 0.001 b |
| Mn | 0.261 ± 0.009 a | 0.022 ± 0.002 b |
| Al | 0.83 ± 0.03 a | 0.81 ± 0.09 a |
| Sr | 0.020 ± 0.004 a | 0.012 ± 0.002 a |
| Ba | 0.05 ± 0.02 a | 0.004 ± 0.001 a |
| Ni | 2.06 ± 0.01 a | 0.23 ± 0.02 b |
| Zn | 0.048 ± 0.001 b | 0.107 ± 0.012 a |
| Se | 0.44 ± 0.07 a | 0.47 ± 0.10 a |
| P | 1067 ± 2 a | 1053 ± 10 a |
| S | 53.61 ± 0.08 a | 6.06 ± 0.04 b |
| Polluting elements | ||
| Pb | 0.064 ± 0.003 b | 0.115 ± 0.005 a |
| As | N.D. * | N.D. |
| Cd | 0.024 ± 0.003 a | 0.011 ± 0.001 b |
| Group | Tested Strains | Disk Diffusion Method (15 µL of EO Concentration 100%) | Positive Control | Negative Control | |||
|---|---|---|---|---|---|---|---|
| Tested Samples of EO | Antibiotic/Antimicotic | DMSO 5% | |||||
| SRB | RF | CHL | TET | Actidion | |||
| G(-) bacteria | E.coli ATCC 25922 | 32 ± 3 b | 40.0 ± 0.0 a | 29 ± 2 b | 21.0 ± 0.0 c | - | nd * |
| P. aeruginosa ATCC 27853 | 10.0 ± 0.0 c | 21 ± 3 a | 12.3 ± 0.6 bc | 14.7 ± 0.6 b | - | nd | |
| G(+) bacteria | B. cereus ATCC 11778 | 12 ± 1 d | 40.0 ± 0.0 a | 30.3 ± 0.6 b | 28 ± 1 c | - | nd |
| S. aureus ATCC 25923 | 32 ± 2 b | 40.0 ± 0.0 a | 29.7 ± 0.6 b | 26.0 ± 0.0 c | - | nd | |
| Yeast | S. cerevisiae ATCC 9763 | 33 ± 2 b | 40.0 ± 0.0 a | - | - | 40.0 ± 0.0 a | nd |
| Fungi | A. brasiliensis ATCC 16404 | 11 ± 2 c | 33.0 ± 0.0 a | - | - | 26.3 ± 0.6 b | nd |
| Group | Tested Strains | MIC [%] | |
|---|---|---|---|
| Tested Samples of EO | |||
| SRB | RF | ||
| G(−) bacteria | E. coli ATCC 25922 | 12.5 | 0.8 |
| P. aeruginosa ATCC 27853 | 50.0 | 6.3 | |
| G(+) bacteria | B. cereus ATCC 11778 | 12.5 | 0.8 |
| S.aureus ATCC 25923 | 6.3 | 0.8 | |
| Yeast | S. cerevisiae ATCC 9763 | >50.0 | 1.6 |
| Fungi | A. brasiliensis ATCC 16404 | >50.0 | 1.6 |
| Parameters | SRB | RF |
|---|---|---|
| DSC | ||
| Ton (°C) | 179 ± 1 b | 192 ± 2 a |
| Tp (°C) | 204 ± 2 b | 207 ± 1 a |
| Toff (°C) | 223 ± 2 a | 221 ± 1 a |
| ΔH (J/g) | 320 ± 10 a | 256 ± 9 b |
| TGA | ||
| Tp (°C) | 71 ± 2 b | 74 ± 1 a |
| Ts (°C) | 29 ± 2 a | 30 ± 3 a |
| Te (°C) | 121 ± 2 a | 122 ± 2 a |
| Residue at Te (%) | 1.9 ± 0.5 a | 1.5 ± 0.3 a |
| Percentage of Added EO (% w/w) | OIT (min) | |
|---|---|---|
| SRB | RF | |
| 0 | 18 ± 1 | |
| 0.1 | 19.4 ± 0.8 AB,a | 20 ± 1 A,a |
| 0.5 | 20.7 ± 0.8 AB,a,* | 20.2 ± 0.2 A,a |
| 1 | 21 ± 1 A,a,* | 18.5 ± 0.4 A,b |
| 5 | 18.3 ± 0.6 BC,a | 10.8 ± 0.8 B,b,* |
| 10 | 16.3 ± 0.8 C,a | 5 ± 1 C,b,* |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Micić, D.; Đurović, S.; Riabov, P.; Tomić, A.; Šovljanski, O.; Filip, S.; Tosti, T.; Dojčinović, B.; Božović, R.; Jovanović, D.; et al. Rosemary Essential Oils as a Promising Source of Bioactive Compounds: Chemical Composition, Thermal Properties, Biological Activity, and Gastronomical Perspectives. Foods 2021, 10, 2734. https://doi.org/10.3390/foods10112734
Micić D, Đurović S, Riabov P, Tomić A, Šovljanski O, Filip S, Tosti T, Dojčinović B, Božović R, Jovanović D, et al. Rosemary Essential Oils as a Promising Source of Bioactive Compounds: Chemical Composition, Thermal Properties, Biological Activity, and Gastronomical Perspectives. Foods. 2021; 10(11):2734. https://doi.org/10.3390/foods10112734
Chicago/Turabian StyleMicić, Darko, Saša Đurović, Pavel Riabov, Ana Tomić, Olja Šovljanski, Snežana Filip, Tomislav Tosti, Biljana Dojčinović, Rade Božović, Dušan Jovanović, and et al. 2021. "Rosemary Essential Oils as a Promising Source of Bioactive Compounds: Chemical Composition, Thermal Properties, Biological Activity, and Gastronomical Perspectives" Foods 10, no. 11: 2734. https://doi.org/10.3390/foods10112734









