Recent Advances in the Mechanisms and Regulation of QS in Dairy Spoilage by Pseudomonas spp.
Abstract
:1. Introduction
2. Pseudomonas spp. as Major Cause of Spoilage in Dairy Chain
2.1. Spoilage Traits Caused by Proteolytic and Lipolytic Activities
2.2. Discoloration as a Spoilage Trait
3. Biofilm by Food-Related Psychrotrophic Pseudomonads and Its Impact on Dairy Industry
| Pseudomonas Species | Sources of Isolation | Experimental Conditions Assayed for Biofilm Formation | References |
|---|---|---|---|
| P. fluorescens | Burrata, Butter Curd Cheese, Mozzarella cheese, Milk Scamorza, Brine Ice machine, Mozzarella manufacturing plant. | TSB supplemented with 0.2% of glucose, 10 °C at least 48 h of incubation or 30 °C at least 24 h of incubation | [8,78] |
| Spoiled milk | TSB, 25 °C, 16 h and exposure for 5 min to 100 ppm chlorine dioxide | [88] | |
| Floor, drains, pipes, and valves of different processing equipment (raw milk cooling tank, milk separator, skim tank, cream tank, homogenizer, pasteurization vat, milk storage vats, cheese vat, cheese ripening room, and packaging area) | TSB and citrate minimal medium at 4 °C; citrate minimal medium at 30 °C for 48 h | [82] | |
| Overhead pipe filler in a dairy processing plant | Diluted nutrient medium consisting of 0.05 g/L glucose, 0.025 g/L peptone and 0.0125 g/L yeast extract in 0.02 M phosphate buffer (pH 7), in a flow cell reactor system for 7 days | [89] | |
| Fresh cheeses | M63 15 °C and 30 °C for 72 h | [9] | |
| Raw milk | Stainless steel coupons immersed in TSB or skimmed milk at 7 °C for 7 days. | [43] | |
| Raw milk, pasteurised milk, curd, whey, cheeses environmental surfaces (collected after routine cleaning process) and environmental air from 8 different areas of a dairy industry | On stainless steel coupons immersed in Ultra-high temperature whole milk at exposure temperatures of 7, 13, 27, 41, and 47 °C and contact times of 0, 1.2, 4, 6.8, and 8 days. | [90] | |
| Mozzarella cheese | Ricotta-based medium 12 °C for 168 h; | [91] | |
| Dairy product | Pipes filled with skimmed milk diluted 1/10 at 20 °C at 8 rpm | [92] | |
| Dairy wastewater | TSB (3 g/L) + Ca2+ (0.1–1 M) at 30 °C, 150 rpm, overnight 18 h | [93] | |
| P. putida | Dairy products | M63 at 15 and 30 °C for 72 h | [94] |
| Mozzarella cheese, curd | TSB supplemented with 0.2% of glucose, 10 °C at least 48 h of incubation and 30 °C at least 24 h of incubation; | [8] | |
| Milk processing line (e.g., balance tank) | BHI, 22 °C and 30 °C, 70 rpm. | [95] | |
| Dairy plants | Native or modified-surface plate heat exchanger during the pasteurization of raw milk for 17 h. | [83] | |
| P. granadensis | Mozzarella cheese | TSB supplemented with 0.2% of glucose, 10 °C at least 48 h of incubation and 30 °C at least 24 h of incubation. | [8] |
| P. brenneri | |||
| P. brenneri | Raw milk | TSB, 7 °C for 7 days and stainless-steel coupons immersed in TSB or skimmed milk at 7 °C for 7 days. | [30] |
| P. koreensis | Mozzarella cheese, brine | TSB, 7 °C for 7 days and stainless-steel coupons immersed in TSB or skimmed milk at 7 °C for 7 days. | [8] |
| Raw milk | TSB, 7 °C for 7 days | [30] | |
| P. veronii | Raw milk | Stainless steel surface immersed in TSB or skimmed milk at 7 °C for 96 h. | [96] |
| TSB, 7 °C for 7 days | [30] | ||
| P. fragi | Stainless-steel surfaces of raw milk tankers floor drains (cooling chamber, and cutting, washing and processing areas of the plant) | Stainless steel surface of an inadequately cleaned tanker immersed in a sterile reconstituted skim milk at fluctuating temperature at a wide range of temperature (16–37 °C) for a maximum of 24 h | [97] |
| Raw milk | TSB, 7 °C for 7 days and stainless-steel coupons immersed in TSB or skimmed milk at 7 °C for 7 days | [30] | |
| P. lactis | Mozzarella cheese | M63 at 15 and 30 °C for 72 h LB at 15 and 30 °C for 48 h | [35] |
| P. gessardii | Mozzarella cheese | M63 at 15 and 30 °C for 72 h | [94] |
| P. cedrina | Raw milk | TSB, 7 °C for 7 days and stainless-steel coupons immersed in TSB or skimmed milk at 7 °C for 7 days | [30] |
| P. azotoformans | |||
| P. extremorientalis | TSB, 7 °C for 7 days | [30] | |
| P. libanensis | TSB, 7 °C for 7 days and stainless-steel coupons immersed in TSB or skimmed milk at 7 °C for 7 days | [30] | |
| P. lundensis | |||
| P. lurida | |||
| P. simiae | TSB, 7 °C for 7 days and stainless-steel coupons immersed in TSB or skimmed milk at 7 °C from 96 to 7 days | [30] | |
| P. yamanorum | TSB, 7 °C for 7 days and stainless-steel coupons immersed in TSB or skimmed milk at 7 °C for 7 days | [30] | |
| P. rhodesia | |||
| P. rhodesia | Milk processing line (e.g., balance tank) | Native or modified-surface plate heat exchanger during the pasteurization of raw milk for 17 h | [83] |
| P. chlororaphis | |||
| P. mucidolens | Floor drains (cooling chamber, and cutting, washing and processing areas of the plant) | nr | [98] |
| P. vancouverensis |
4. Quorum Sensing in Pseudomonas spp.
4.1. P. fluorescens
4.2. P. lactis
4.3. P. putida
4.4. P. fragi and P. gessardii
5. QS-Regulated Spoilage Traits in Dairy-Borne Pseudomonas spp.
5.1. Proteases, Lipases and Phospholipases
5.2. Pigments
5.3. Off Flavours
6. Methodological Advances, Perspectives and Concluding Remarks
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO; IFAD; UNICEF; WFP; WHO. The State of Food Security and Nutrition in the World 2019. Safeguarding against Economic Slowdowns and Downturns; FAO: Rome, Italy, 2019; p. 239. [Google Scholar] [CrossRef]
- Grimaccia, E.; Naccarato, A. Food insecurity in Europe: A gender perspective. Soc. Ind. Res. 2020, 1–19. [Google Scholar] [CrossRef]
- Laborde, D.; Martin, W.; Swinnen, J.; Vos, R. COVID-19 risks to global food security. Science 2020, 369, 500–502. [Google Scholar] [CrossRef]
- UN General Assembly, Transforming Our World: The 2030 Agenda for Sustainable Development, 21 October 2015, A/RES/70/1. Available online: https://www.refworld.org/docid/57b6e3e44 (accessed on 3 November 2021).
- Santeramo, F.G.; Lamonaca, E. Food Loss–Food Waste–Food Security: A New Research Agenda. Sustainability 2021, 13, 4642. [Google Scholar] [CrossRef]
- Martin, N.H.; Boor, K.J.; Wiedmann, M. Effect of post-pasteurization contamination on fluid milk quality. J. Dairy Sci. 2018, 101, 861–870. [Google Scholar] [CrossRef] [Green Version]
- Martin, N.H.; Torres-Frenzel, P.; Wiedmann, M. Controlling dairy product spoilage to reduce food loss and waste. J. Dairy Sci. 2020, 104, 1251–1261. [Google Scholar] [CrossRef] [PubMed]
- Rossi, C.; Serio, A.; Chaves-López, C.; Anniballi, F.; Auricchio, B.; Goffredo, E.; Cenci-Goga, B.T.; Lista, F.; Fillo, S.; Paparella, A. Biofilm formation, pigment production and motility in Pseudomonas spp. isolated from the dairy industry. Food Cont. 2018, 86, 241–248. [Google Scholar]
- Quintieri, L.; Zühlke, D.; Fanelli, F.; Caputo, L.; Liuzzi, V.C.; Logrieco, A.F.; Hirschfeld, C.; Becher, D.; Riedel, K. Proteomic analysis of the food spoiler Pseudomonas fluorescens ITEM 17298 reveals the antibiofilm activity of the pepsin-digested bovine lactoferrin. Food Microbiol. 2019, 82, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.G.; Luke, R.K.J. Siderophore production and utilization by milk spoilage Pseudomonas species. J. Dairy Sci. 2010, 93, 1355–1363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jamuna, B.A.; Ravishankar, R.V. Quorum sensing regulation and inhibition of exoenzyme production and biofilm formation in the food spoilage bacteria Pseudomonas psychrophila PSPF19. Food Biotechnol. 2014, 28, 293–308. [Google Scholar] [CrossRef]
- Machado, I.; Silva, L.R.; Giaouris, E.D.; Melo, L.F.; Simões, M. Quorum sensing in food spoilage and natural-based strategies for its inhibition. Food Res. Int. 2020, 127, 108754. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Wang, N.; Sadiq, F.A.; He, G. RNA sequencing reveals the involvement of quorum sensing in dairy spoilage caused by psychrotrophic bacteria. LWT 2020, 127, 109384. [Google Scholar] [CrossRef]
- Li, T.; Wang, D.; Ren, L.; Mei, Y.; Ding, T.; Li, Q.; Chen, H.; Li, J. Involvement of exogenous N-acyl-homoserine lactones in spoilage potential of Pseudomonas fluorescens isolated from refrigerated turbot. Front. Microbiol. 2019, 10, 2716. [Google Scholar] [CrossRef]
- Hassan, S.; Ahmad, T.; Bashir, M.; Kiran, G.S.; Selvin, J. Novel Perspectives on the Quorum Sensing Inhibitors (QSIs)/Quorum Quenchers (QQs) in Food Preservation and Spoilage. In Implication of Quorum Sensing and Biofilm Formation in Medicine, Agriculture and Food Industry; Bramhachari, P.V., Ed.; Springer: Singapore, 2019; pp. 269–298. [Google Scholar]
- Bai, A.J.; Vittal, R.R. Quorum sensing inhibitory and anti-biofilm activity of essential oils and their in vivo efficacy in food systems. Food Biotechnol. 2014, 28, 269–292. [Google Scholar] [CrossRef]
- Shobharani, P.; Agrawal, R. Interception of quorum sensing signal molecule by furanone to enhance shelf life of fermented milk. Food Cont. 2010, 21, 61–69. [Google Scholar] [CrossRef]
- Myszka, K.; Tomaś, N.; Wolko, Ł.; Szwengiel, A.; Grygier, A.; Nuc, K.; Majcher, M. In situ approaches show the limitation of the spoilage potential of Juniperus phoenicea L. essential oil against cold-tolerant Pseudomonas fluorescens KM24. Appl. Microbiol. Biotechnol. 2021, 105, 4255–4268. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Feng, L.; Lu, H.; Zhu, J.; Kumar, V.; Liu, X. Transcriptomic analysis of the food spoilers Pseudomonas fluorescens reveals the antibiofilm of carvacrol by interference with intracellular signaling processes. Food Cont. 2021, 127, 108115. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Chen, J.; Koseki, S.; Yang, Q.; Yu, H.; Fu, L. Screening and preservation application of quorum sensing inhibitors of Pseudomonas fluorescens and Shewanella baltica in seafood products. LWT 2021, 149, 111749. [Google Scholar] [CrossRef]
- Gill, C.O.; Suisted, J.R. The effects of temperature and growth rate on the proportion of unsaturated fatty acids in bacterial lipids. J. Gen. Microbiol. 1978, 104, 31–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neumeyer, K.; Ross, T.; McMeekin, T.A. Development of a predictive model to describe the effects of temperature and water activity on the growth of spoilage pseudomonads. Int. J. Food Microbiol. 1997, 38, 45–54. [Google Scholar] [CrossRef]
- Stoops, J.; Maes, P.; Claes, J.; Van Campenhout, L. Growth of Pseudomonas fluorescens in modified atmosphere packaged tofu. Lett. Appl. Microbiol. 2012, 54, 195–202. [Google Scholar] [CrossRef]
- Nicodeme, M.; Grill, J.P.; Humbert, G.; Gaillard, J.L. Extracellular protease activity of different Pseudomonas strains: Dependence of proteolytic activity on culture conditions. J. Appl. Microbiol. 2005, 99, 641–648. [Google Scholar] [CrossRef]
- Law, B.A.; Andrews, A.T.; Sharpe, M.E. Gelation of ultra-high-temperature-sterilized milk by proteases from a strain of Pseudomonas fluorescens isolated from raw milk. J. Dairy Res. 1977, 44, 145–148. [Google Scholar]
- De Jonghe, V.; Coorevits, A.; Van Hoorde, K.; Messens, W.; Van Landschoot, A.; De Vos, P.; Heyndrickx, M. Influence of storage conditions on the growth of Pseudomonas species in refrigerated raw milk. Appl. Environ. Microbiol. 2011, 77, 460–470. [Google Scholar] [CrossRef] [Green Version]
- Stoeckel, M.; Lidolt, M.; Achberger, V.; Glück, C.; Krewinkel, M.; Stressler, T.; von Neubeck, M.; Wenning, M.; Scherer, S.; Fischer, L.; et al. Growth of Pseudomonas weihenstephanensis, Pseudomonas proteolytica and Pseudomonas sp. in raw milk: Impact of residual heat-stable enzyme activity on stability of UHT milk during shelf-life. Int. Dairy J. 2016, 59, 20–28. [Google Scholar] [CrossRef]
- Volk, V.; Glück, C.; Leptihn, S.; Ewert, J.; Stressler, T.; Fischer, L. Two heat resistant endopeptidases from Pseudomonas species with destabilizing potential during milk storage. J. Agric. Food Chem. 2018, 67, 905–915. [Google Scholar] [CrossRef]
- Machado, S.G.; da Silva, F.L.; Bazzolli, D.M.; Heyndrickx, M.; Costa, P.M.D.A.; Vanetti, M.C.D. Pseudomonas spp. and Serratia liquefaciens as predominant spoilers in cold raw milk. J. Food Sci. 2015, 80, M1842–M1849. [Google Scholar]
- Yuan, L.; Sadiq, F.A.; Liu, T.J.; Li, Y.; Gu, J.S.; Yang, H.Y.; He, G.Q. Spoilage potential of psychrotrophic bacteria isolated from raw milk and the thermo-stability of their enzymes. J. Zhejiang Univ. Sci. B 2018, 19, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Baruzzi, F.; Lagonigro, R.; Quintieri, L.; Morea, M.; Caputo, L. Occurrence of non-lactic acid bacteria populations involved in protein hydrolysis of cold-stored high moisture Mozzarella cheese. Food Microbiol. 2012, 30, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Scatamburlo, T.M.; Yamazi, A.K.; Cavicchioli, V.Q.; Pieri, F.A.; Nero, L.A. Spoilage potential of Pseudomonas species isolated from goat milk. J. Dairy Sci. 2015, 98, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Liu, H.; Dong, L.; Zheng, N.; Xing, M.; Zhang, Y.; Zhao, S.; Wang, J. Identification and proteolytic activity quantification of Pseudomonas spp. isolated from different raw milks at storage temperatures. J. Dairy Sci. 2018, 101, 2897–2905. [Google Scholar] [PubMed] [Green Version]
- Ottaviani, F.; Disegna, L. Muffe e lieviti nei prodotti e negli ambienti caseari. Latte 1987, 12, 779–811. [Google Scholar]
- Quintieri, L.; Caputo, L.; De Angelis, M.; Fanelli, F. Genomic Analysis of Three Cheese-Borne Pseudomonas lactis with Biofilm and Spoilage-Associated Behavior. Microorganisms 2020, 8, 1208. [Google Scholar] [CrossRef] [PubMed]
- Martin, N.H.; Murphy, S.C.; Ralyea, R.D.; Wiedmann, M.; Boor, K.J. When cheese gets the blues: Pseudomonas fluorescens as the causative agent of cheese spoilage. J. Dairy Sci. 2011, 94, 3176–3183. [Google Scholar] [CrossRef]
- Carrascosa, C.; Millán, R.; Jaber, J.R.; Lupiola, P.; del Rosario-Quintana, C.; Mauricio, C.; Sanjuán, E. Blue pigment in fresh cheese produced by Pseudomonas fluorescens. Food Cont. 2015, 54, 95–102. [Google Scholar] [CrossRef]
- da Silva Rodrigues, R.; Machado, S.G.; de Carvalho, A.F.; Nero, L.A. Pseudomonas sp. as the causative agent of anomalous blue discoloration in Brazilian fresh soft cheese (Minas Frescal). Int. Dairy J. 2021, 117, 105020. [Google Scholar] [CrossRef]
- Carminati, D.; Bonvini, B.; Rossetti, L.; Zago, M.; Tidona, F.; Giraffa, G. Investigation on the presence of blue pigment-producing Pseudomonas strains along a production line of fresh mozzarella cheese. Food Cont. 2019, 100, 321–328. [Google Scholar] [CrossRef]
- Cantoni, C.; Soncini, G.; Milesi, S.; Cocolin, L.; Iacumin, L.; Comi, G. Colorazioni anomale e rigonfiamento di formaggi fusi e mozzarelle. Ind. Aliment. 2006, 45, 276–281. [Google Scholar]
- Evanowski, R.L.; Reichler, S.J.; Kent, D.J.; Martin, N.H.; Boor, K.J.; Wiedmann, M. Pseudomonas azotoformans causes gray discoloration in HTST fluid milk. J. Dairy Sci. 2017, 100, 7906–7909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kornacki, J.L.; Flowers, R.S.; Bradley, J.R.L. Microbiology of Butter. In Applied Dairy Microbiology, 2nd ed.; Marcel Dekker, Inc.: New York, NY, USA, 2001; pp. 127–150. [Google Scholar]
- Deeth, H.C.; Fitz-Gerald, C.H. Lipolytic enzymes and hydrolytic rancidity. In Advanced Dairy Chemistry Volume 2 Lipids; Springer: Boston, MA, USA, 2006; pp. 481–556. [Google Scholar]
- Samaržija, D.; Zamberlin, Š.; Pogačić, T. Psychrotrophic bacteria and their negative effects on milk and dairy products quality. Mljekarstvo: J. Dairy Prod. Proc. Imp. 2012, 62, 77–95. [Google Scholar]
- Kumar, H.; Franzetti, L.; Kaushal, A.; Kumar, D. Pseudomonas fluorescens: A potential food spoiler and challenges and advances in its detection. Ann. Microbiol. 2019, 69, 873–883. [Google Scholar] [CrossRef]
- Corsetti, A.; Rossi, J.; Gobbetti, M. Interactions between yeasts and bacteria in the smear surface-ripened cheeses. Int. J. Food Microbiol. 2001, 69, 1–10. [Google Scholar] [CrossRef]
- Fox, P.F.; Stepaniak, L. Isolation and some properties of extracellular heat-stable lipases from Pseudomonas fluorescens strain AFT 36. J. Dairy Res. 1983, 50, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Jay, J.M.; Loessner, M.J.; Golden, D.A. Modern Food Microbiology; Springer: New York, NY, USA, 2005. [Google Scholar]
- McPhee, J.D.; Griffiths, M.W. Pseudomonas spp. In Encyclopaedia of Dairy Sciences; Roginski, H., Fuquay, W.J., Fox, F.P., Eds.; Academic Press: New York, NY, USA, 2002; Volume 4, pp. 2340–2350. [Google Scholar]
- Hammad, A.M. Spoilage potential of Pseudomonas spp. isolated from domiati cheese. Assiut Vet. Med. J. 2015, 61, 18–23. [Google Scholar]
- Farkye, N.Y.; Vedamuthu, E.R. Microbiology of soft cheeses. In Dairy Microbiology Handbook. The Microbiology of Milk and Milk Products; Robinson, R.K., Ed.; John Wiley and Sons: Hoboken, NJ, USA, 2002; pp. 479–513. [Google Scholar]
- Paludetti, L.F.; Kelly, A.L.; Gleeson, D. Effect of thermoresistant protease of Pseudomonas fluorescens on rennet coagulation properties and proteolysis of milk. J. Dairy Sci. 2020, 103, 4043–4055. [Google Scholar] [CrossRef]
- Stuknytė, M.; Decimo, M.; Colzani, M.; Silvetti, T.; Brasca, M.; Cattaneo, S.; Aldini, G.; De Noni, I. Extracellular thermostable proteolytic activity of the milk spoilage bacterium Pseudomonas fluorescens PS19 on bovine caseins. J. Dairy Sci. 2016, 99, 4188–4195. [Google Scholar] [CrossRef]
- D’Incecco, P.; Brasca, M.; Rosi, V.; Morandi, S.; Ferranti, P.; Picariello, G.; Pellegrino, L. Bacterial proteolysis of casein leading to UHT milk gelation: An applicative study. Food Chem. 2019, 292, 217–226. [Google Scholar] [CrossRef]
- Crudden, A.; Fox, P.F.; Kelly, A.L. Factors affecting the hydrolytic action of plasmin in milk. Int. Dairy J. 2005, 15, 305–313. [Google Scholar] [CrossRef]
- Mara, O.; Roupie, C.; Duffy, Y.A.; Kelly, A.L. The Curd-forming Properties of Milk as affected by the action of plasmin. Int. Dairy J. 1998, 8, 807–812. [Google Scholar] [CrossRef]
- Liu, M.; Wang, H.; Griffiths, M.W. Regulation of alkaline metalloprotease promoter by N-acyl homoserine lactone quorum sensing in Pseudomonas fluorescens. J. Appl. Microbiol. 2007, 103, 2174–2184. [Google Scholar] [CrossRef]
- Liao, C.H.; McCallus, D.E. Biochemical and genetic characterization of an extracellular protease from Pseudomonas fluorescens CY091. Appl. Environ. Microbiol. 1998, 64, 914–921. [Google Scholar] [CrossRef] [Green Version]
- Quintieri, L.; Pistillo, B.R.; Caputo, L.; Favia, P.; Baruzzi, F. Bovine lactoferrin and lactoferricin on plasma-deposited coating against spoilage Pseudomonas spp. Inn. Food Sci. Emerg. Technol. 2013, 20, 215–222. [Google Scholar] [CrossRef]
- Caputo, L.; Quintieri, L.; Bianchi, D.M.; Decastelli, L.; Monaci, L.; Visconti, A.; Baruzzi, F. Pepsin-digested bovine lactoferrin prevents Mozzarella cheese blue discoloration caused by Pseudomonas fluorescens. Food Microbiol. 2015, 46, 15–24. [Google Scholar] [CrossRef]
- Ledenbach, L.H.; Marshall, R.T. Microbiological spoilage of dairy products. In Compendium of the Microbiological Spoilage of Food and Beverages Industries; Doyle, M.P., Sperber, W.H., Eds.; Springer: New York, NY, USA, 2009; pp. 41–68. [Google Scholar]
- Morales, P.; Fernandez-Garcia, E.; Nunez, M. Production of volatile compounds in cheese by Pseudomonas fragi strains of dairy origin. J. Food Prot. 2005, 68, 1399–1407. [Google Scholar] [CrossRef]
- Deeth, H.C. Lipases from Milk and Other Sources. In Agents of Change. Food Engineering Series; Kelly, A.L., Larsen, L.B., Eds.; Springer: Cham, Switzerland, 2021. [Google Scholar] [CrossRef]
- Decimo, M.; Brasca, M.; Ordóñez, J.A.; Cabeza, M.C. Fatty acids released from cream by psychrotrophs isolated from bovine raw milk. Int. J. Dairy Technol. 2017, 70, 339–344. [Google Scholar] [CrossRef]
- Deeth, H.C.; Fitzgerald, C.H. Lipolytic enzymes and hydrolytic rancidity in milk and milk products. In Developments in Dairy Chemistry; Fox, P.F., Ed.; Elsevier: Amsterdam, The Netherlands; Applied Science Publishers: London, UK, 1983; Volume 2, pp. 195–239. [Google Scholar]
- Ghoddusi, H.; Özer, B. Microbiology of cream, butter, ice cream and related products. In Dairy Microbiology and Biochemistry–Recent Developments; Ozer, B., Akdemir-Evrendilek, G., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 245–270. [Google Scholar]
- Soncini, G.; Marchisio, E.; Cantoni, C. Causes of chromatic alterations in Mozzarella cheese. Ind. Alim. 1998, 37, 850–855. [Google Scholar]
- Franzetti, L.; Scarpellini, M. Characterisation of Pseudomonas spp. isolated from foods. Annals Microbiol. 2007, 57, 39–47. [Google Scholar]
- Palleroni, N.J.; Genus, I. Pseudomonas Migula 1894. In Bergey’s Manual of Systematic Bacteriology, 2nd ed.; Brenner, D.J., Krieg, N.R., Staley, J.T., Eds.; The Proteobacteria, Part B, The Gammaproteobacteria; Springer: New York, NY, USA, 2005; Volume 2, pp. 323–379. [Google Scholar]
- RASFF. Rapid Alert System for Food and Feed. Annual Report. 2010. Available online: https://op.europa.eu/en/publication-detail/-/publication/7de58882-f5c5-4e28-b8b5-0ebf9836dbdf/language-en/format-PDF/source-174744260 (accessed on 12 December 2020).
- Nogarol, C.; Acutis, P.L.; Bianchi, D.M.; Maurella, C.; Peletto, S.; Gallina, S.; Adriano, D.; Zuccon, F.; Borrello, S.; Caramelli, M.; et al. Molecular characterization of Pseudomonas fluorescens isolates involved in the Italian “blue mozzarella” event. J. Food Prot. 2013, 76, 500–504. [Google Scholar] [CrossRef]
- Andreani, N.A.; Martino, M.E.; Fasolato, L.; Carraro, L.; Montemurro, F.; Mioni, R.; Bordin, P.; Cardazzo, B. Tracking the blue: A MLST approach to characterise the Pseudomonas fluorescens group. Food Microbiol. 2014, 39, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Quintieri, L.; Fanelli, F.; Zühlke, D.; Caputo, L.; Logrieco, A.F.; Albrecht, D.; Riedel, K. Biofilm and pathogenesis-related proteins in the foodborne P. fluorescens ITEM 17298 with distinctive phenotypes during cold storage. Front. Microbiol. 2020, 11, 991. [Google Scholar] [PubMed]
- Fanelli, F.; Liuzzi, V.C.; Quintieri, L.; Mulè, G.; Baruzzi, F.; Logrieco, A.F.; Caputo, L. Draft genome sequence of Pseudomonas fluorescens strain ITEM 17298, associated with cheese spoilage. Genome Announc. 2017, 5, e01141–e01217. [Google Scholar] [CrossRef] [Green Version]
- European Commission. Regulation (EC) No. 178/2002 of the European Parliament and of the Council of 28 January 2002 laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety. Off. J. Eur. Comm. 2002, L31, 1–24. [Google Scholar]
- European Commission. Regulation (EU) 2019/1381 of the European Parliament and of the Council of 20 June 2019 on the transparency and sustainability of the EU risk assessment in the food chain. Off. J. Eur. Comm. 2019, L231, 1–28. [Google Scholar]
- Flemming, H.-C.; Wingender, J.; Szewzyk, U.; Steinberg, P.; Rice, S.A.; Kjelleberg, S. Biofilms: An emergent form of bacterial life. Nat. Rev. Microbiol. 2016, 14, 563–575. [Google Scholar] [CrossRef]
- Rossi, C.; Chaves-López, C.; Serio, A.; Goffredo, E.; Goga, B.T.C.; Paparella, A. Influence of incubation conditions on biofilm formation by Pseudomonas fluorescens isolated from dairy products and dairy manufacturing plants. Italian J. Food Saf. 2016, 5, 5793. [Google Scholar] [CrossRef] [PubMed]
- Fysun, O.; Kern, H.; Wilke, B.; Langowski, H.C. Evaluation of factors influencing dairy biofilm formation in filling hoses of food-processing equipment. Food Bioprod. Process. 2019, 113, 39–48. [Google Scholar] [CrossRef]
- Marchand, S.; De Block, J.; De Jonghe, V.; Coorevits, A.; Heyndrickx, M.; Herman, L. Biofilm formation in milk production and processing environments; influence on milk quality and safety. Comp. Rev. Food Sci. Food Saf. 2012, 11, 133–147. [Google Scholar] [CrossRef]
- Srey, S.; Jahid, I.K.; Ha, S.D. Ha Biofilm formation in food industries. A food safety concern Food Cont. 2013, 31, 572–585. [Google Scholar] [CrossRef]
- Aswathanarayan, J.B.; Vittal, R.R. Attachment and biofilm formation of Pseudomonas fluorescens PSD4 isolated from a dairy processing line. Food Sci. Biotechnol. 2014, 23, 1903–1910. [Google Scholar] [CrossRef]
- Jindal, S.; Anand, S.; Metzger, L.; Amamcharla, J. A comparison of biofilm development on stainless steel and modified-surface plate heat exchangers during a 17-h milk pasteurization run. J. Dairy Sci. 2018, 101, 2921–2926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aviat, F.; Gerhards, C.; Rodriguez-Jerez, J.J.; Michel, V.; Bayon, I.L.; Ismail, R.; Federighi, M. Microbial safety of wood in contact with food: A review. Comp. Rev. Food Sci. Food Saf. 2016, 15, 491–505. [Google Scholar] [CrossRef]
- Steinka, I. Chemical and microbiological aspects of the interaction between food and food packages. In Food Packaging Hygiene; Barone, C., Bolzoni, L., Caruso, G., Montanari, A., Parisi, S., Steinka, I., Eds.; Springer: Cham, Switzerland, 2015; pp. 79–104. [Google Scholar]
- Fanelli, F.; Caputo, L.; Quintieri, L. Phenotypic and genomic characterization of Pseudomonas putida ITEM 17297 spoiler of fresh vegetables: Focus on biofilm and antibiotic resistance interaction. Curr. Res. Food Sci. 2021, 4, 74–82. [Google Scholar] [CrossRef]
- Carrascosa, C.; Raheem, D.; Ramos, F.; Saraiva, A.; Raposo, A. Microbial biofilms in the food industry—A comprehensive review. Int. J. Environ. Res. Pub. Health 2021, 18, 2014. [Google Scholar] [CrossRef]
- Lindsay, D.; Brözel, V.S.; Mostert, J.F.; Von Holy, A. Differential efficacy of a chlorine dioxide-containing sanitizer against single species and binary biofilms of a dairy-associated Bacillus cereus and a Pseudomonas fluorescens isolate. J. Appl. Microbiol. 2002, 92, 352–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simões, M.; Simões, L.C.; Vieira, M.J. Physiology and behavior of Pseudomonas fluorescens single and dual strain biofilms under diverse hydrodynamics stresses. Int. J. Food Microbiol. 2008, 128, 309–316. [Google Scholar] [CrossRef] [Green Version]
- Castro, M.S.R.; da Silva Fernandes, M.; Kabuki, D.Y.; Kuaye, A.Y. Modelling Pseudomonas fluorescens and Pseudomonas aeruginosa biofilm formation on stainless steel surfaces and controlling through sanitisers. Int. Dairy J. 2021, 114, 104945. [Google Scholar] [CrossRef]
- Maggio, F.; Rossi, C.; Chaves-López, C.; Serio, A.; Valbonetti, L.; Pomilio, F.; Chiavaroli, A.P.; Paparella, A. Interactions between L. monocytogenes and P. fluorescens in Dual-Species Biofilms under Simulated Dairy Processing Conditions. Foods 2021, 10, 176. [Google Scholar] [CrossRef] [PubMed]
- Bénézech, T.; Faille, C. Two-phase kinetics of biofilm removal during CIP. Respective roles of mechanical and chemical effects on the detachment of single cells vs. cell clusters from a Pseudomonas fluorescens biofilm. J. Food Eng. 2018, 219, 121–128. [Google Scholar]
- Dixon, M.J.L.; Flint, S.H.; Palmer, J.S.; Love, R.; Chabas, C.; Beuger, A.L. The effect of calcium on biofilm formation in dairy wastewater. Water Pract. Technol. 2018, 13, 400–409. [Google Scholar] [CrossRef]
- Quintieri, L.; Fanelli, F.; Caputo, L. Antibiotic resistant Pseudomonas spp. spoilers in fresh dairy products: An underestimated risk and the control strategies. Foods 2019, 8, 372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chumkhunthod, P.; Schraft, H.; Griffiths, M.W. Rapid monitoring method to assess efficacy of sanitizers against Pseudomonas putida biofilms. J. Food Prot. 1998, 61, 1043–1046. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Mohammadpour, H.; Gharibi, D.; Borujeni, M.P. Identification of Pseudomonas jessenii and Pseudomonas gessardii as the most proteolytic Pseudomonas isolates in Iranian raw milk and their impact on stability of sterilized milk during storage. J. Dairy Res. 2020, 87, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Teh, K.H.; Flint, S.; Palmer, J.; Andrewes, P.; Bremer, P.; Lindsay, D. Biofilm—An unrecognised source of spoilage enzymes in dairy products? Int. Dairy J. 2014, 34, 32–40. [Google Scholar] [CrossRef]
- Dzieciol, M.; Schornsteiner, E.; Muhterem-Uyar, M.; Stessl, B.; Wagner, M.; Schmitz-Esser, S. Bacterial diversity of floor drain biofilms and drain waters in a Listeria monocytogenes contaminated food processing environment. Int. J. Food Microbiol. 2016, 223, 33–40. [Google Scholar] [CrossRef]
- D’Souza, G.; Pandian, E.; Hosea, S. A Nearly Fatal Case of Pseudomonas fluorescens Bacteremia Secondary to a Naturopathic Intravenous Vitamin Infusion. J. Investig. Med. High Impact Case Rep. 2021, 9. [Google Scholar] [CrossRef]
- Sasahara, K.C.; Zottola, E.A. Biofilm Formation by Listeria monocytogenes Utilizes a Primary Colonizing Microorganism in Flowing Systems. J. Food Prot. 1993, 56, 1022–1028. [Google Scholar] [CrossRef]
- Simões, M.; Simões, L.C.; Pereira, M.O.; Vieira, M.J. Antagonism between Bacillus cereus and Pseudomonas fluorescens in planktonic systems and in biofilms. Biofouling 2008, 24, 339–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kives, J.; Guadarrama, D.; Orgaz, B.; Rivera-Sen, A.; Vazquez, J.; SanJose, C. Interactions in biofilms of Lactococcus lactis ssp. cremoris and Pseudomonas fluorescens cultured in cold UHT milk. J. Dairy Sci. 2005, 88, 4165–4171. [Google Scholar] [PubMed]
- Alvarez-Ordóñez, A.; Coughlan, L.M.; Briandet, R.; Cotter, P.D. Biofilms in food processing environments: Challenges and opportunities. Ann. Rev. Food Sci. Technol. 2019, 10, 173–195. [Google Scholar] [CrossRef]
- Giaouris, E.; Chorianopoulos, N.; Doulgeraki, A.; Nychas, G.J. Co-culture with Listeria monocytogenes within a dual-species biofilm community strongly increases resistance of Pseudomonas putida to benzalkonium chloride. PLoS ONE 2013, 8, e77276. [Google Scholar] [CrossRef] [Green Version]
- Nadell, C.D.; Xavier, J.B.; Levin, S.A.; Foster, K.R. The evolution of quorum sensing in bacterial biofilms. PLoS Biol. 2008, 6, e14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galloway, W.R.; Hodgkinson, J.T.; Bowden, S.D.; Welch, M.; Spring, D.R. Quorum sensing in Gram-negative bacteria: Small-molecule modulation of AHL and AI-2 quorum sensing pathways. Chem. Rev. 2011, 111, 28–67. [Google Scholar] [CrossRef]
- Ding, T.; Li, T.; Li, J. Virtual screening for quorum-sensing inhibitors of Pseudomonas fluorescens P07 from a food-derived compound database. J. Appl. Microbiol. 2019, 127, 763–777. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ji, L.; Wang, X.; Li, J.; Zhu, J.; Sun, A. Role of RpoS in stress resistance, quorum sensing and spoilage potential of Pseudomonas fluorescens. Int. J. Food Microbiol. 2018, 270, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.L.; Uelinton, M.P.; Riedel, K.; Vanetti, M.C.; Mantovani, H.C.; De Araújo, E.F. Lack of AHL-based quorum sensing in Pseudomonas fluorescens isolated from milk. Brazilian J. Microbiol. 2014, 45, 1039–1046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, R.; Zhu, J.; Feng, L.; Li, J.; Liu, X. Characterization of LuxI/LuxR and their regulation involved in biofilm formation and stress resistance in fish spoilers Pseudomonas fluorescens. Int. J. Food Microbiol. 2019, 297, 60–71. [Google Scholar] [CrossRef]
- Gray, K.M.; Garey, J.R. The evolution of bacterial LuxI and LuxR quorum sensing regulators. Microbiology 2001, 147, 2379–2387. [Google Scholar] [CrossRef] [Green Version]
- El-Sayed, A.K.; Hothersall, J.; Thomas, C.M. Quorum-sensing-dependent regulation of biosynthesis of the polyketide antibiotic mupirocin in Pseudomonas fluorescens NCIMB 10586. Microbiology 2001, 147, 2127–2139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laue, B.E.; Jiang, Y.; Chhabra, S.R.; Jacob, S.; Stewart, G.S.A.B.; Hardman, A.; Downie, J.A.; O’Gara, F.; Williams, P. The biocontrol strain Pseudomonas fluorescens F113 produces the Rhizobium small bacteriocin, N-(3-hydroxy-7-cis-tetradecanoyl)homoserine lactone, via HdtS, a putative novel Nacylhomoserine lactone synthase. Microbiology 2000, 146, 2469–2480. [Google Scholar] [CrossRef] [Green Version]
- Wei, H.L.; Zhang, L.Q. Quorum-sensing system influences root colonization and biological control ability in Pseudomonas fluorescens 2P24. Antonie Van Leeuwenhoek 2006, 89, 267–280. [Google Scholar] [CrossRef]
- Scales, B.S.; Dickson, R.P.; LiPuma, J.J.; Huffnagle, G.B. Microbiology, genomics, and clinical significance of the Pseudomonas fluorescens species complex, an unappreciated colonizer of humans. Clin. Microbiol. Rev. 2014, 27, 927–948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, H.K.; Suárez-Moreno, Z.R.; Degrassi, G.; Subramoni, S.; González, J.F.; Venturi, V. Bacterial LuxR solos have evolved to respond to different molecules including signals from plants. Front Plant Sci. 2013, 4, 447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Workentine, M.L.; Chang, L.; Ceri, H.; Turner, R.J. The GacS–GacA two-component regulatory system of Pseudomonas fluorescens: A bacterial two-hybrid analysis. FEMS Microbiol. Lett. 2009, 292, 50–56. [Google Scholar] [CrossRef]
- Venturi, V. Regulation of quorum sensing in Pseudomonas. FEMS Microbiol. Rev. 2005, 30, 274–291. [Google Scholar] [CrossRef] [Green Version]
- Yan, Q.; Wu, X.G.; Wei, H.L.; Wang, H.M.; Zhang, L.Q. Differential control of the PcoI/PcoR quorum-sensing system in Pseudomonas fluorescens 2P24 by sigma factor RpoS and the GacS/GacA two-component regulatory system. Microbiol. Res. 2009, 164, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, J.; Zhu, J.; Du, P.; Sun, A. Combined transcriptome and proteome analysis of rpoS regulon reveals its role in spoilage potential of Pseudomonas fluorescens. Front. Microbiol. 2019, 10, 94. [Google Scholar] [CrossRef] [Green Version]
- Liang, F.; Zhang, B.; Yang, Q.; Zhang, Y.; Zheng, D.; Zhang, L.Q.; Yan, Q.; Wu, X. Cyclic-di-GMP Regulates the Quorum-Sensing System and Biocontrol Activity of Pseudomonas fluorescens 2P24 through the RsmA and RsmE Proteins. Appl. Environ. Microbiol. 2020, 86, e02016–e02020. [Google Scholar] [CrossRef]
- Von Neubeck, M.; Huptas, C.; Glück, C.; Krewinkel, M.; Stoeckel, M.; Stressler, T.; Fischer, L.; Hinrichs, J.; Scherer, S.; Wenning, M. Pseudomonas lactis sp. nov. and Pseudomonas paralactis sp. nov. isolated from bovine raw milk. Int. J. Syst. Evol. Microbiol. 2017, 67, 1656–1664. [Google Scholar]
- Huang, J.J.; Petersen, A.; Whiteley, M.; Leadbetter, J.R. Identification of QuiP, the product of gene PA1032, as the second acyl-homoserine lactone acylase of Pseudomonas aeruginosa PAO1. Appl. Environ. Microbiol. 2006, 72, 1190–1197. [Google Scholar] [CrossRef] [Green Version]
- Sappington, K.J.; Dandekar, A.A.; Oinuma, K.I.; Greenberg, E.P. Reversible signal binding bythe Pseudomonas aeruginosa quorum-sensing signal receptor LasR. MBio 2011, 2, e00011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirakawa, H.; Oda, Y.; Phattarasukol, S.; Armour, C.D.; Castle, J.C.; Raymond, C.K.; Lappala, C.R.; Schaefer, A.L.; Harwood, C.S.; Greenberg, E.P. Activity of the Rhodopseudomonas palustris p-coumaroyl-homoserine lactone-responsive transcription factor RpaR. J. Bacteriol. 2011, 193, 2598–2607. [Google Scholar] [CrossRef] [Green Version]
- Kong, P. Evaluation of a novel endophytic Pseudomonas lactis strain for control of boxwood blight. J. Environ. Hortic. 2019, 37, 39–43. [Google Scholar] [CrossRef]
- Steidle, A.; Allesen-Holm, M.; Riedel, K.; Berg, G.; Givskov, M.; Molin, S.; Eberl, L. Identification and characterization of an N-acylhomoserine lactone-dependent quorum-sensing system in Pseudomonas putida strain IsoF. Appl. Environ. Microbiol. 2002, 68, 6371–6382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bertani, I.; Rampioni, G.; Leoni, L.; Venturi, V. The Pseudomonas putida Lon protease is involved in N-acyl homoserine lactone quorum sensing regulation. BMC Microbiol. 2007, 7, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sauer, K.; Camper, A.K. Characterization of phenotypic changes in Pseudomonas putida in response to surface-associated growth. J. Bacteriol. 2001, 183, 6579–6589. [Google Scholar] [CrossRef] [Green Version]
- Bertani, I.; Venturi, V. Regulation of the N-acyl homoserine lactone-dependent quorum-sensing system in rhizosphere Pseudomonas putida WCS358 and cross-talk with the stationary-phase RpoS sigma factor and the global regulator GacA. Appl. Environ. Microbiol. 2004, 70, 5493–5502. [Google Scholar] [CrossRef] [Green Version]
- Dubern, J.F.; Lugtenberg, B.J.; Bloemberg, G.V. The ppuI-rsaL-ppuR quorum-sensing system regulates biofilm formation of Pseudomonas putida PCL1445 by controlling biosynthesis of the cyclic lipopeptides putisolvins I and II. J. Bacteriol. 2006, 188, 2898–2906. [Google Scholar] [CrossRef] [Green Version]
- Caldera, L.; Franzetti, L.V.; Van Coillie, E.; De Vos, P.; Stragier, P.; De Block, J.; Heyndrickx, M. Identification, enzymatic spoilage characterization and proteolytic activity quantification of Pseudomonas spp. isolated from different foods. Food Microbiol. 2016, 54, 142–153. [Google Scholar]
- Ferrocino, I.; Ercolini, D.; Villani, F.; Moorhead, S.M.; Griffiths, M.W. Pseudomonas fragi strains isolated from meat do not produce N-acyl homoserine lactones as signal molecules. J. Food Prot. 2009, 72, 2597–2601. [Google Scholar] [CrossRef]
- Singha, L.P.; Kotoky, R.; Pandey, P. Draft genome sequence of Pseudomonas fragi strain DBC, which has the ability to degrade high-molecular-weight polyaromatic hydrocarbons. Genome Announc. 2017, 5, e01347–e01417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, J.; Cheng, J.; Wang, Y.; Shen, X. The Pseudomonas Quinolone Signal (PQS): Not Just for Quorum Sensing Anymore. Front. Cell. Infect. Microbiol. 2018, 8, 230. [Google Scholar] [CrossRef] [PubMed]
- Wettstadt, S.; Llamas, M.A. Role of regulated proteolysis in the communication of bacteria with the environment. Front. Mol. Biosci. 2020, 7, 586497. [Google Scholar] [CrossRef]
- Bai, A.J.; Vittal, R.R. Bacterial quorum sensing and food industry. Compr. Rev. Food Sci. Food Saf. 2011, 10, 183–193. [Google Scholar] [CrossRef]
- McCarthy, C.N.; Woods, R.G.; Beacham, I.R. Regulation of the aprX-lipA operon of Pseudomonas fluorescens B52: Differential regulation of the proximal and distal genes, encoding protease and lipase, by ompR-envZ. FEMS Microbiol. Lett. 2004, 241, 243–248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Machado, S.G.; Baglinière, F.; Marchand, S.; Van Coillie, E.; Vanetti, M.C.; De Block, J.; Heyndrickx, M. The biodiversity of the microbiota producing heat-resistant enzymes responsible for spoilage in processed bovine milk and dairy products. Front. Microbiol. 2017, 8, 302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ammor, M.S.; Flórez, A.B.; van Hoek, A.H.; de Los Reyes-Gavilán, C.G.; Aarts, H.J.; Margolles, A.; Mayo, B. Molecular characterization of intrinsic and acquired antibiotic resistance in lactic acid bacteria and bifidobacteria. J. Mol. Microbiol. Biotechnol. 2008, 14, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Rosenau, F.; Jaeger, K.E. Bacterial lipases from Pseudomonas: Regulation of gene expression and mechanisms of secretion. Biochimie 2000, 82, 1023–1032. [Google Scholar] [CrossRef]
- Prigent-Combaret, C.; Brombacher, E.; Vidal, O.; Ambert, A.; Lejeune, P.; Landini, P.; Dorel, C. Complex regulatory network controls initial adhesion and biofilm formation in Escherichia coli via regulation of the csgD gene. J. Bacteriol. 2001, 183, 7213–7223. [Google Scholar] [CrossRef] [Green Version]
- Sacherer, P.; Défago, G.; Haas, D. Extracellular protease and phospholipase C are controlled by the global regulatory gene gacA in the biocontrol strain Pseudomonas fluorescens CHA0. FEMS Microbiol. Lett. 1994, 116, 155–160. [Google Scholar] [CrossRef]
- Armes, A.C.; Buchan, A. Cyclic di-GMP is integrated into a hierarchical quorum sensing network regulating antimicrobial production and biofilm formation in Roseobacter clade member Rhodobacterales Strain Y4I. Front. Mar. Sci. 2021, 8, 681551. [Google Scholar] [CrossRef]
- Cude, W.N.; Buchan, A. Acyl-homoserine lactone-based quorum sensing in the Roseobacter clade: Complex cell-to-cell communication controls multiple physiologies. Front. Microbiol. 2013, 4, 336. [Google Scholar] [CrossRef] [Green Version]
- Lan, L.; Murray, T.S.; Kazmierczak, B.I.; He, C. Pseudomonas aeruginosa OspR is an oxidative stress sensing regulator that affects pigment production, antibiotic resistance and dissemination during infection. Mol. Microbiol. 2010, 75, 76–91. [Google Scholar] [CrossRef] [Green Version]
- Andreani, N.A.; Carraro, L.; Zhang, L.; Vos, M.; Cardazzo, B. Transposon mutagenesis in Pseudomonas fluorescens reveals genes involved in blue pigment production and antioxidant protection. Food Microbiol. 2019, 82, 497–503. [Google Scholar] [CrossRef]
- Reichler, S.J.; Martin, N.H.; Evanowski, R.L.; Kovac, J.; Wiedmann, M.; Orsi, R.H. A century of gray: A genomic locus found in 2 distinct Pseudomonas spp. is associated with historical and contemporary color defects in dairy products worldwide. J. Dairy Sci. 2019, 102, 5979–6000. [Google Scholar] [PubMed] [Green Version]
- Bishop, T.F.; Martin, L.W.; Lamont, I.L. Activation of a cell surface signaling pathway in Pseudomonas aeruginosa requires ClpP protease and new sigma factor synthesis. Front. Microbiol. 2017, 8, 2442. [Google Scholar] [CrossRef] [PubMed]
- Wilderman, P.J.; Vasil, A.I.; Johnson, Z.; Wilson, M.J.; Cunliffe, H.E.; Lamont, I.L.; Vasil, M.L. Characterization of an endoprotease (PrpL) encoded by a PvdS-regulated gene in Pseudomonas aeruginosa. Infect. Immun. 2001, 69, 5385–5394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azzara, C.D.; Campbell, L.B. Off-flavors of dairy products. In Developments in Food Science; Off-Flavors in Foods and Beverages; Charalambous, G., Ed.; Elsevier: Amsterdam, The Netherlands, 1992; pp. 329–374. [Google Scholar]
- Andreani, N.A.; Carraro, L.; Fasolato, L.; Balzan, S.; Lucchini, R.; Novelli, E.; Cardazzo, B. Characterisation of the thermostable protease AprX in strains of Pseudomonas fluorescens and impact on the shelf-life of dairy products: Preliminary results. Ital. J. Food Saf. 2016, 5, 6175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gloria, M.B.A.; Saraiva, P.R.; Rigueira, J.C.; Brandao, S.C. Bioactive amines changes in raw and sterilised milk inoculated with Pseudomonas fluorescens stored at different temperatures. Int. J. Dairy Technol. 2011, 64, 45–51. [Google Scholar] [CrossRef] [Green Version]
- Luengo, J.M.; Olivera, E.R. Catabolism of biogenic amines in Pseudomonas species. Environ. Microbiol. 2020, 22, 1174–1192. [Google Scholar] [CrossRef] [Green Version]
- Bernier, S.P.; Nguyen, D.T.; Sokol, P.A. A LysR-type transcriptional regulator in Burkholderia cenocepacia influences colony morphology and virulence. Infect. Immun. 2008, 76, 38–47. [Google Scholar] [CrossRef] [Green Version]
- Chan, K.G.; Liu, Y.C.; Chang, C.Y. Inhibiting N-acyl-homoserine lactone synthesis and quenching Pseudomonas quinolone quorum sensing to attenuate virulence. Front. Microbiol. 2015, 6, 1173. [Google Scholar] [CrossRef]
- O’Toole, G.A.; Kolter, R. Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: A genetic analysis. Mol. Microbiol. 1998, 28, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Pratt, L.A.; Kolter, R. Genetic analyses of bacterial biofilm formation. Curr. Opin. Microbiol. 1999, 2, 598–603. [Google Scholar] [CrossRef]
- Williamson, L.L.; Borlee, B.R.; Schloss, P.D.; Guan, C.; Allen, H.K.; Handelsman, J. Intracellular screen to identify metagenomic clones that induce or inhibit a quorum-sensing biosensor. Appl. Environ. Microbiol. 2005, 71, 6335–6344. [Google Scholar] [CrossRef] [Green Version]
- Federle, M.J. Autoinducer-2-based chemical communication in bacteria: Complexities of interspecies signaling. Contrib. Microbiol. 2009, 16, 18–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pereira, C.S.; Thompson, J.A.; Xavier, K.B. AI-2-mediated signalling in bacteria. FEMS Microbiol Rev. 2013, 37, 156–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kimura, N. Metagenomic approaches to understanding phylogenetic diversity in quorum sensing. Virulence 2014, 5, 433–442. [Google Scholar] [CrossRef]
- García-Contreras, R.; Nuñez-López, L.; Jasso-Chávez, R.; Kwan, B.W.; Belmont, J.A.; Rangel-Vega, A.; Maeda, T.; Wood, T. Quorum sensing enhancement of the stress response promotes resistance to quorum quenching and prevents social cheating. ISME J. 2015, 9, 115–125. [Google Scholar] [CrossRef]
- Wang, Y.; Rattray, J.B.; Thomas, S.A.; Gurney, J.; Brown, S.P. In Silico bacteria evolve robust cooperation via complex quorum-sensing strategies. Sci. Rep. 2020, 10, 8628. [Google Scholar] [CrossRef] [PubMed]
- Ofria, C.; Wilke, C.O. Avida: A software platform for research in computational evolutionary biology. Art. Life 2004, 10, 191–229. [Google Scholar] [CrossRef]
- Knibbe, C.; Beslon, G.; Lefort, V.; Chaudier, F.; Fayard, J.M. Self-adaptation of genome size in artificial organisms. In Advances in Artificial Life; Capcarrère, M.S., Freitas, A.A., Bentley, P.J., Johnson, C.G., Timmis, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 423–432. [Google Scholar]
- Miller, C.; Gilmore, J. Detection of quorum-sensing molecules for pathogenic molecules using cell-based and cell-free biosensors. Antibiotics 2020, 9, 259. [Google Scholar] [CrossRef]
- Zhang, B.; Powers, R. Analysis of bacterial biofilms using NMR-based metabolomics. Fut. Med. Chem. 2012, 4, 1273–1306. [Google Scholar] [CrossRef] [Green Version]
- Davenport, P.W.; Griffin, J.L.; Welch, M. Quorum sensing is accompanied by global metabolic changes in the opportunistic human pathogen Pseudomonas aeruginosa. J. Bacteriol. 2015, 197, 2072–2082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinto, U.M.; de Souza Viana, E.; Martins, M.L.; Vanetti, M.C.D. Detection of acylated homoserine lactones in gram-negative proteolytic psychrotrophic bacteria isolated from cooled raw milk. Food Cont. 2007, 18, 1322–1327. [Google Scholar] [CrossRef]
- Hentzer, M.; Givskov, M. Pharmacological inhibition of quorum sensing for the treatment of chronic bacterial infections. J. Clin. Investig. 2003, 112, 1300–1307. [Google Scholar] [CrossRef]
- Jakobsen, T.H.; Bragason, S.K.; Phipps, R.K.; Christensen, L.D.; Van Gennip, M.; Alhede, M.; Skindersoe, M.; Larsen, T.O.; Høiby, N.; Bjarnsholt, T.; et al. Food as a source for quorum sensing inhibitors: Iberin from horseradish revealed as a quorum sensing inhibitor of Pseudomonas aeruginosa. Appl. Environ. Microbiol. 2012, 78, 2410–2421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skandamis, P.N.; Nychas, G.J.E. Quorum sensing in the context of food microbiology. Appl. Environ. Microbiol. 2012, 78, 5473–5482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Della Pepa, T.; Elshafie, H.S.; Capasso, R.; De Feo, V.; Camele, I.; Nazzaro, F.; Scognamiglio, M.R.; Caputo, L. Antimicrobial and phytotoxic activity of Origanum heracleoticum and O. majorana essential oils growing in cilento (Southern Italy). Molecules 2019, 24, 2576. [Google Scholar] [CrossRef] [Green Version]
- Gopu, V.; Meena, C.K.; Shetty, P.H. Quercetin influences quorum sensing in food borne bacteria: In-Vitro and In-Silico evidence. PLoS ONE 2015, 10, e0134684. [Google Scholar] [CrossRef] [Green Version]

| Spoilage Traits | Pseudomonas Species | Dairy Product | References |
|---|---|---|---|
| Proteolysis | |||
| Milk creaming, sediment formation, gelation, bitterness | P. fluorescens, P. weihenstephanensis, P. proteolytica, Pseudomonas spp. | UHT milk | [25,26,27] |
| P. panacis | Skimmed milk | [28] | |
| P. azotoformans | Raw milk | [29,30] | |
| P. gessardii | Milk | [26,31] | |
| P. proteolytica | |||
| P. fluorescens | Unpasteurized goat milk | [32] | |
| Pseudomonas spp. | Non-bovine raw milk | [33] | |
| Bitterness, Mozzarella skin wrinkling/peeling, cheese softness, sediment formation | P. lundensis, P gessardii | Mozzarella cheese, skimmed milk | [31] |
| P. fluorescens | Crescenza cheese | [34] | |
| P. fluorescens, P. fragi | Mozzarella cheese | [31] | |
| P. fluorescens, P. putida | Cheddar cheese | [25] | |
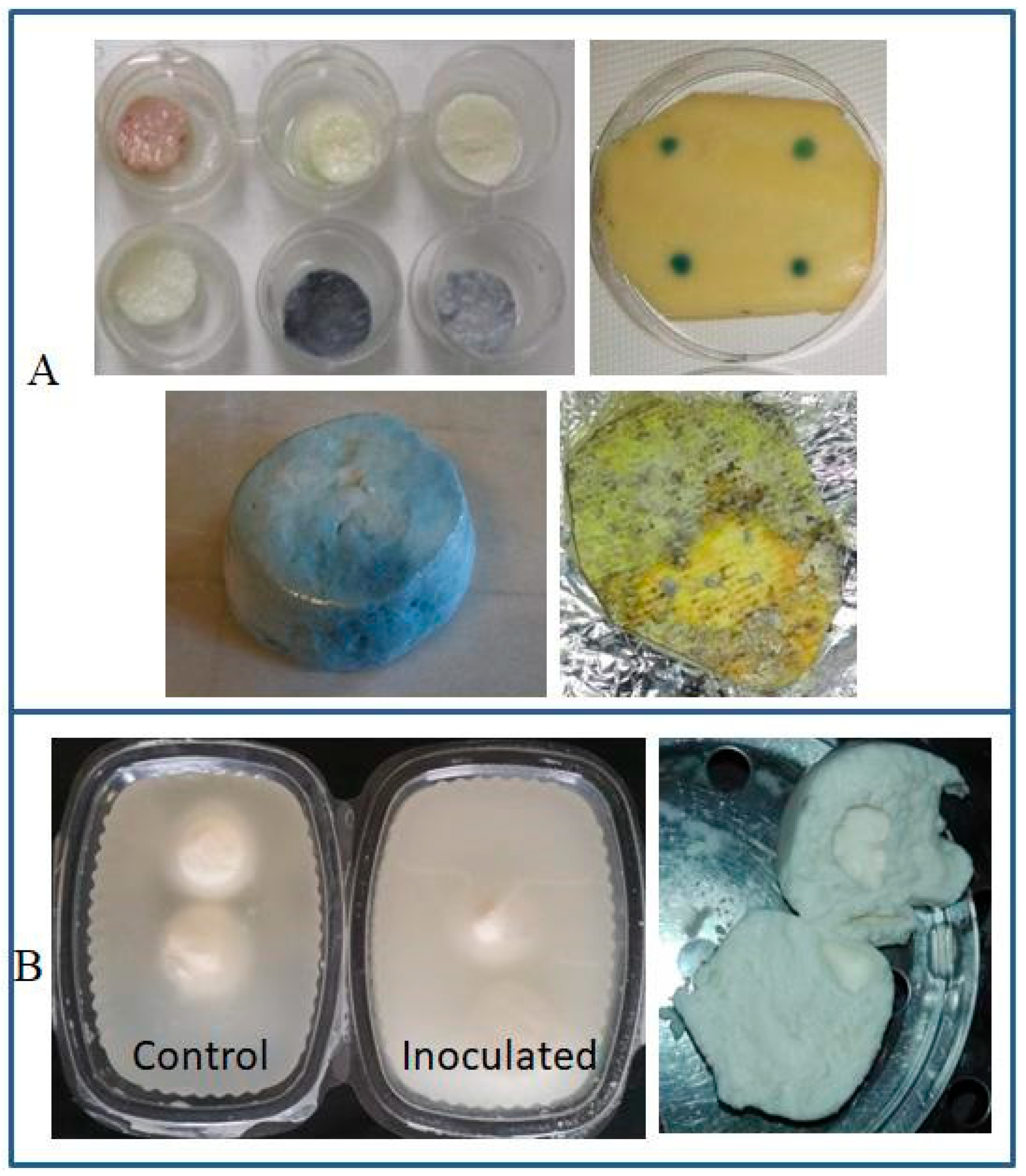
| Discoloration | |||
| Blue | P. fluorescens, P. lactis | Mozzarella cheese | [8,35] |
| P. fluorescens | Latin-style fresh cheeses | [36,37] | |
| P. carnis, Pseudomonas spp. | Brazilian fresh soft cheese | [38] | |
| P. fluorescens | Mozzarella processing fluids | [39] | |
| Orange or Orange-red-brown | P. aureofaciens, P. gessardii, P. putida biovar II | Mozzarella cheese | [40] |
| Greenish | P. fluorescens | ||
| Fluorescent (yellow-green) | P. fluorescens, P. putida, P. palleronii | ||
| Grayish | P. azotoformans | HTST milk | [41] |
| P. fragi | Milk | [40] | |
| Black | P. mephitica, P. nigrifaciens | Butter | [42] |
| Lipolysis | |||
| Rancidity off-flavors straw-berry flavor bitterness soapy | Pseudomonas spp., P. fluorescens | Sterilized milk | [43,44,45] |
| P. fluorescens | Ripened semi-hard cheese | [46,47] | |
| P. fragi, P. putrefaciens, Pseudomonas spp. | Cream, butter | [44,48,49] | |
| Pseudomonas spp. | Domiati cheese | [50] | |
| Pseudomonas spp. | Soft cheese | [51] | |
| Modification of rennet coagulation time and curd firmness | Pseudomonas spp., P. fluorescens | Cheese Cheddar cheese | [44,52] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quintieri, L.; Caputo, L.; Brasca, M.; Fanelli, F. Recent Advances in the Mechanisms and Regulation of QS in Dairy Spoilage by Pseudomonas spp. Foods 2021, 10, 3088. https://doi.org/10.3390/foods10123088
Quintieri L, Caputo L, Brasca M, Fanelli F. Recent Advances in the Mechanisms and Regulation of QS in Dairy Spoilage by Pseudomonas spp. Foods. 2021; 10(12):3088. https://doi.org/10.3390/foods10123088
Chicago/Turabian StyleQuintieri, Laura, Leonardo Caputo, Milena Brasca, and Francesca Fanelli. 2021. "Recent Advances in the Mechanisms and Regulation of QS in Dairy Spoilage by Pseudomonas spp." Foods 10, no. 12: 3088. https://doi.org/10.3390/foods10123088
APA StyleQuintieri, L., Caputo, L., Brasca, M., & Fanelli, F. (2021). Recent Advances in the Mechanisms and Regulation of QS in Dairy Spoilage by Pseudomonas spp. Foods, 10(12), 3088. https://doi.org/10.3390/foods10123088









