Innovative Ultrasound-Assisted Approaches towards Reduction of Heavy Metals and Iodine in Macroalgal Biomass
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Treatment Conditions
2.3. Cadmium, Mercury and Total Arsenic Analysis
2.4. Iodine Analysis
2.5. Determination of Removal Efficacy
2.6. Statistical Analysis
3. Results
3.1. As, Cd, Hg, and I Content in Untreated, Wild-Harvested L. hyperborea
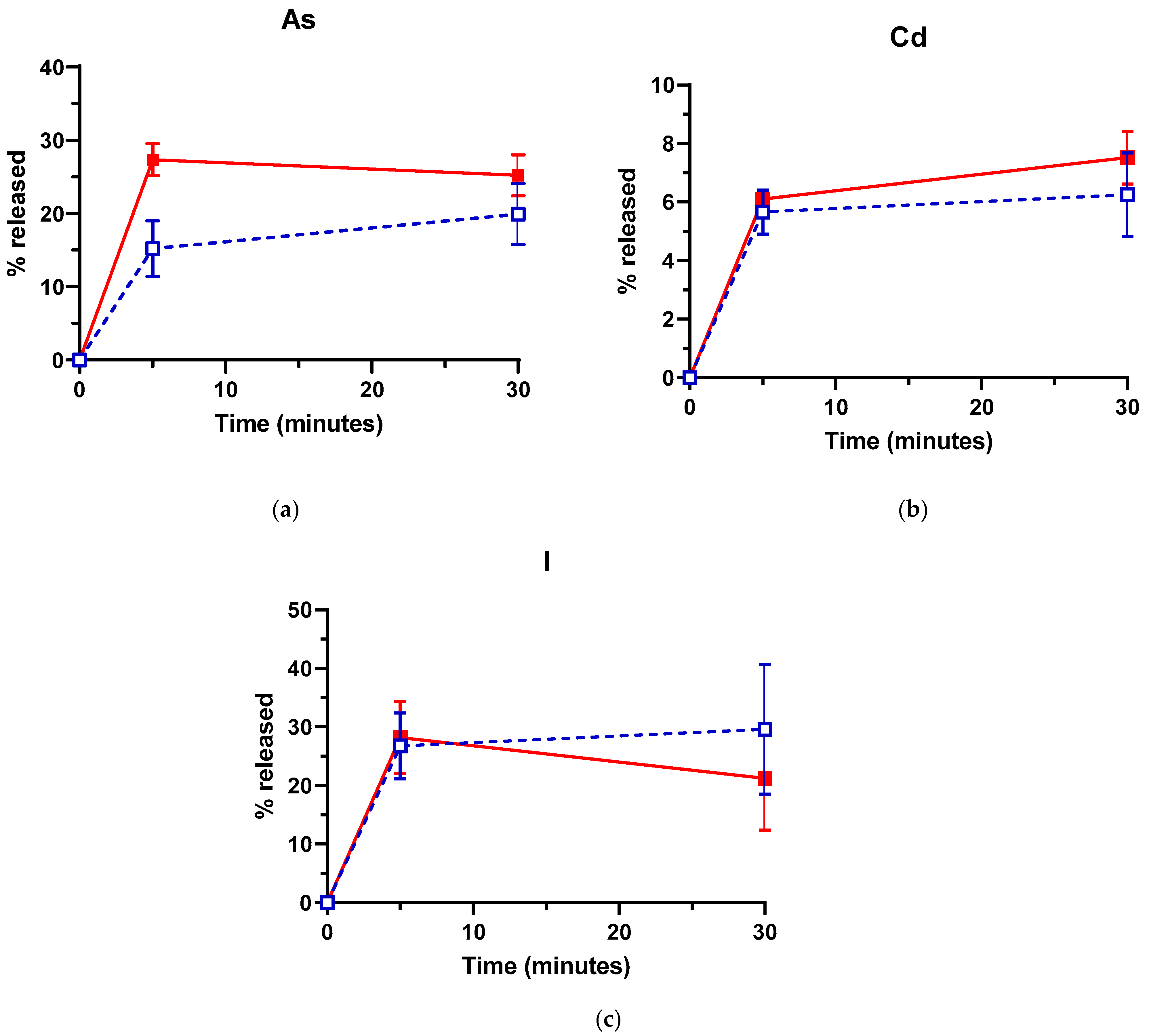
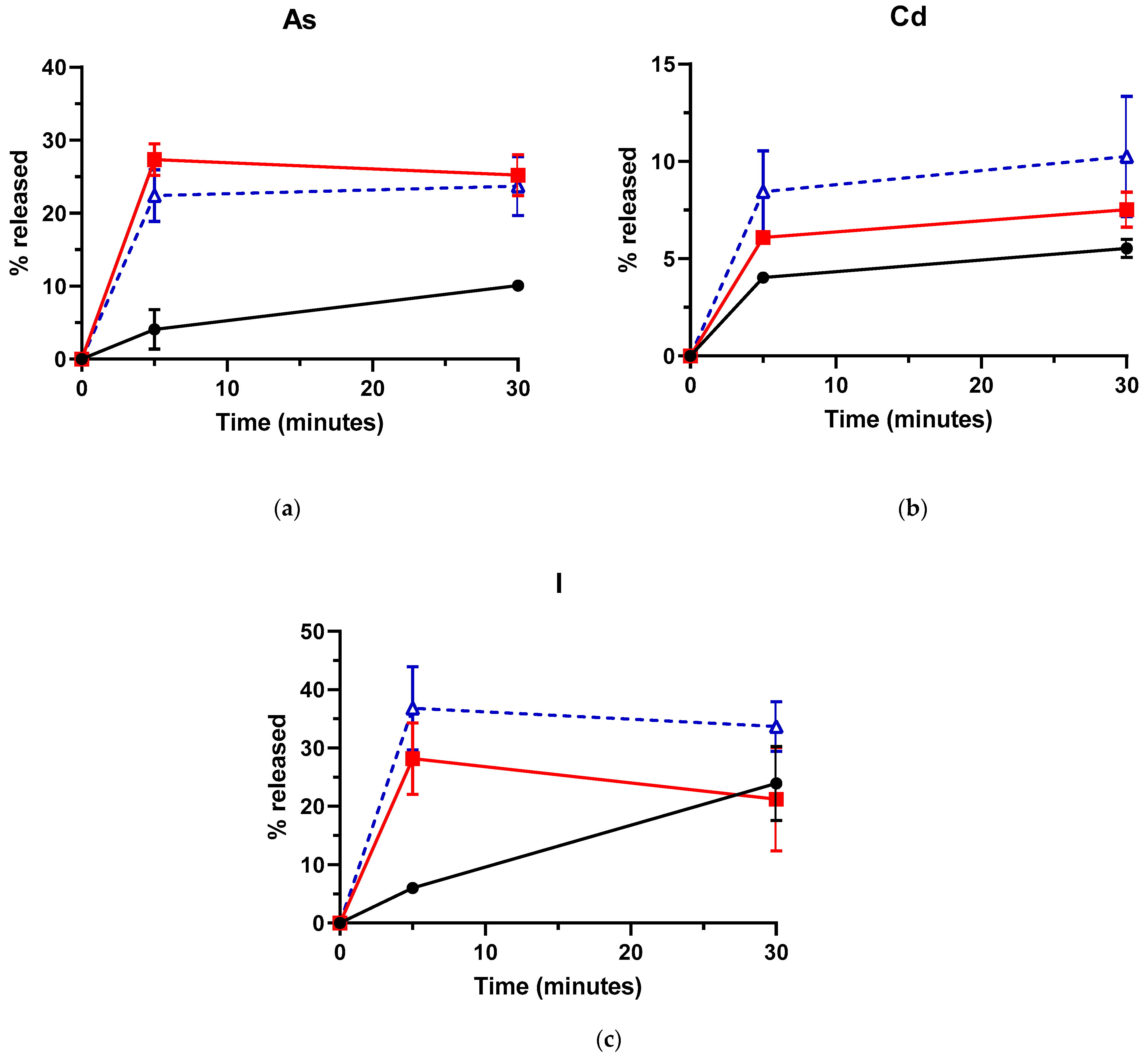
3.2. Effect of US on As, Cd and I Removal from L. hyperborea Blades
3.3. Influence of Treatment Temperature on As, Cd and I Removal by US
3.4. Influence of the Addition of EDTA on As, Cd and I Removal by US
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Bocanegra, A.; Bastida, S.; Benedí, J.; Ródenas, S.; Sánchez-Muniz, F.J. Characteristics and Nutritional and Cardiovascular-Health Properties of Seaweeds. J. Med. Food. 2009, 12, 236–258. [Google Scholar] [CrossRef]
- Holland, J. Seaweed on Track to Become Europe’s Next Big Superfood Trend. Available online: https://www.seafoodsource.com/news/food-safety-health/seaweed-on-track-to-become-europe-s-next-big-superfood-trend (accessed on 6 July 2019).
- Banach, J.L.; Hoek-van den Hil, E.F.; van der Fels-Klerx, H.J. Food Safety Hazards in the European Seaweed Chain. Compr. Rev. Food. Sci. Food. Saf. 2020, 19, 332–364. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. The Global Status of Seaweed Production, Trade and Utilization. Available online: http://www.fao.org/in-action/globefish/publications/details-publication/en/c/1154074/ (accessed on 5 February 2021).
- Fleurence, J. Seaweed as Food. In Seaweed in Health and Disease Prevention; Fleurence, J., Levine, I., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 149–167. ISBN 978-0-12-802772-1. [Google Scholar]
- Lorenzo, J.M.; Agregán, R.; Munekata, P.E.S.; Franco, D.; Carballo, J.; Şahin, S.; Lacomba, R.; Barba, F.J. Proximate Composition and Nutritional Value of Three Macroalgae: Ascophyllum Nodosum, Fucus Vesiculosus and Bifurcaria Bifurcata. Mar. Drugs. 2017, 15, 360. [Google Scholar] [CrossRef]
- Ganesan, A.R.; Tiwari, U.; Rajauria, G. Seaweed Nutraceuticals and Their Therapeutic Role in Disease Prevention. Food. Sci. Hum. Well. 2019, 8, 252–263. [Google Scholar] [CrossRef]
- Roohinejad, S.; Koubaa, M.; Barba, F.J.; Saljoughian, S.; Amid, M.; Greiner, R. Application of Seaweeds to Develop New Food Products with Enhanced Shelf-Life, Quality and Health-Related Beneficial Properties. Int. Food Res. J. 2017, 99, 1066–1083. [Google Scholar] [CrossRef]
- Cardoso, S.; Carvalho, L.; Silva, P.M.; Rodrigues, M.I.; Pereira, O.; Pereira, L. Bioproducts from Seaweeds: A Review with Special Focus on the Iberian Peninsula. Curr. Org. Chem. 2014, 18, 896–917. [Google Scholar] [CrossRef]
- Circuncisão, A.R.; Catarino, M.D.; Cardoso, S.M.; Silva, A.M.S. Minerals from Macroalgae Origin: Health Benefits and Risks for Consumers. Mar. Drugs. 2018, 16, 400. [Google Scholar] [CrossRef]
- Cardoso, S.M.; Pereira, O.R.; Seca, A.M.L.; Pinto, D.C.G.A.; Silva, A.M.S. Seaweeds as Preventive Agents for Cardiovascular Diseases: From Nutrients to Functional Foods. Mar. Drugs. 2015, 13, 6838–6865. [Google Scholar] [CrossRef]
- Davis, T.A.; Volesky, B.; Mucci, A. A Review of the Biochemistry of Heavy Metal Biosorption by Brown Algae. Water. Res. 2003, 37, 4311–4330. [Google Scholar] [CrossRef]
- European Union (EU) Recommendation (EU). 2018/464 of 19 March 2018 on the Monitoring of Metals and Iodine in Seaweed, Halophytes and Products Based on Seaweed. Off. J. Eur. Union 2018, 61, 16–18. [Google Scholar]
- Scientific Committee on Food; Scientific Panel on Dietetic Products, Nutrition and Allergies; European Food Safety Authority (EFSA). Tolerable Upper Intake Levels for Vitamins and Minerals; Scientific Committee on Food; Scientific Panel on Dietetic Products, Nutrition and Allergies; European Food Safety Authority (EFSA): Parma, Italy, 2006. [Google Scholar]
- Stévant, P.; Marfaing, H.; Duinker, A.; Fleurence, J.; Rustad, T.; Sandbakken, I.; Chapman, A. Biomass Soaking Treatments to Reduce Potentially Undesirable Compounds in the Edible Seaweeds Sugar Kelp (Saccharina Latissima) and Winged Kelp (Alaria Esculenta) and Health Risk Estimation for Human Consumption. J. Appl. Phycol. 2018, 30, 2047–2060. [Google Scholar] [CrossRef]
- Biancarosa, I.; Belghit, I.; Bruckner, C.G.; Liland, N.S.; Waagbø, R.; Amlund, H.; Heesch, S.; Lock, E.-J. Chemical Characterization of 21 Species of Marine Macroalgae Common in Norwegian Waters: Benefits of and Limitations to Their Potential Use in Food and Feed. J. Sci. Food. Agric. 2018, 98, 2035–2042. [Google Scholar] [CrossRef]
- Lüning, K.; Mortensen, L. European Aquaculture of Sugar Kelp (Saccharina Latissima) for Food Industries: Iodine Content and Epiphytic Animals as Major Problems. Bot. Mar. 2015, 58, 449–455. [Google Scholar] [CrossRef]
- Besada, V.; Andrade, J.M.; Schultze, F.; González, J.J. Heavy Metals in Edible Seaweeds Commercialised for Human Consumption. J. Mar. Syst. 2009, 75, 305–313. [Google Scholar] [CrossRef]
- Khaled, A.; Hessein, A.; Abdel-Halim, A.M.; Morsy, F.M. Distribution of Heavy Metals in Seaweeds Collected along Marsa-Matrouh Beaches, Egyptian Mediterranean Sea. Egypt. J. Aquat. Res. 2014, 40, 363–371. [Google Scholar] [CrossRef]
- Commission Regulation (EC). No 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs (Text with EEA Relevance). Off. J. Eur. Union 2006, 364, 5–24. [Google Scholar]
- Żbikowski, R.; Szefer, P.; Latała, A. Distribution and Relationships between Selected Chemical Elements in Green Alga Enteromorpha Sp. from the Southern Baltic. Environ. Pollut. 2006, 143, 435–448. [Google Scholar] [CrossRef]
- Brown, M.T.; Hodgkinson, W.M.; Hurd, C.L. Spatial and Temporal Variations in the Copper and Zinc Concentrations of Two Green Seaweeds from Otago Harbour, New Zealand. Mar. Environ. Res. 1999, 47, 175–184. [Google Scholar] [CrossRef]
- Roleda, M.Y.; Marfaing, H.; Desnica, N.; Jónsdóttir, R.; Skjermo, J.; Rebours, C.; Nitschke, U. Variations in Polyphenol and Heavy Metal Contents of Wild-Harvested and Cultivated Seaweed Bulk Biomass: Health Risk Assessment and Implication for Food Applications. Food Control 2019, 95, 121–134. [Google Scholar] [CrossRef]
- Brito, G.B.; de Souza, T.L.; Bressy, F.C.; Moura, C.W.N.; Korn, M.G.A. Levels and Spatial Distribution of Trace Elements in Macroalgae Species from the Todos Os Santos Bay, Bahia, Brazil. Mar. Pollut. Bull. 2012, 64, 2238–2244. [Google Scholar] [CrossRef]
- Nielsen, C.W.; Holdt, S.L.; Sloth, J.J.; Marinho, G.S.; Sæther, M.; Funderud, J.; Rustad, T. Reducing the High Iodine Content of Saccharina Latissima and Improving the Profile of Other Valuable Compounds by Water Blanching. Foods 2020, 9, 569. [Google Scholar] [CrossRef]
- Almela, C.; Clemente, M.J.; Vélez, D.; Montoro, R. Total Arsenic, Inorganic Arsenic, Lead and Cadmium Contents in Edible Seaweed Sold in Spain. Food. Chem. Toxicol. 2006, 44, 1901–1908. [Google Scholar] [CrossRef]
- Brandon, E.F.A.; Janssen, P.J.C.M.; de Wit-Bos, L. Arsenic: Bioaccessibility from Seaweed and Rice, Dietary Exposure Calculations and Risk Assessment. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess 2014, 31, 1993–2003. [Google Scholar] [CrossRef]
- Nitschke, U.; Stengel, D.B. Quantification of Iodine Loss in Edible Irish Seaweeds during Processing. J. Appl. Phycol. 2016, 28, 3527–3533. [Google Scholar] [CrossRef]
- Park, G.; Kang, D.; Davaatseren, M.; Shin, C.; Kang, G.-J.; Chung, M.-S. Reduction of Total, Organic, and Inorganic Arsenic Content in Hizikia Fusiforme (Hijiki). Food. Sci. Biotechnol. 2019, 28, 615–622. [Google Scholar] [CrossRef]
- Superior Health Council Arsenic and Other Elements in Algae and Dietary Supplements Based on Algae, Superior Health Council. Available online: https://www.absym-bvas.be/images/spf_sante/Conseil_superieur_de_la_sante/CSS_9149.pdf (accessed on 8 January 2021).
- Bruhn, A.; Brynning, G.; Johansen, A.; Lindegaard, M.S.; Sveigaard, H.H.; Aarup, B.; Fonager, L.; Andersen, L.L.; Rasmussen, M.B.; Larsen, M.M.; et al. Fermentation of Sugar Kelp (Saccharina Latissima)—Effects on Sensory Properties, and Content of Minerals and Metal. J. Appl. Phycol. 2019, 31, 3175–3187. [Google Scholar] [CrossRef]
- Blikra, M.J.; Løvdal, T.; Vaka, M.R.; Roiha, I.S.; Lunestad, B.T.; Lindseth, C.; Skipnes, D. Assessment of Food Quality and Microbial Safety of Brown Macroalgae (Alaria Esculenta and Saccharina Latissima). J. Sci. Food Agric. 2019, 99, 1198–1206. [Google Scholar] [CrossRef]
- Ho, K.K.H.Y.; Redan, B.W. Impact of Thermal Processing on the Nutrients, Phytochemicals, and Metal Contaminants in Edible Algae. Crit. Rev. Food Sci. Nutr. 2020, 1–19. [Google Scholar] [CrossRef]
- Rajauria, G.; Jaiswal, A.K.; Abu-Ghannam, N.; Gupta, S. Effect of Hydrothermal Processing on Colour, Antioxidant and Free Radical Scavenging Capacities of Edible Irish Brown Seaweeds. IFST 2010, 45, 2485–2493. [Google Scholar] [CrossRef]
- Mateluna, C.; Figueroa, V.; Ortiz, J.; Aguilera, J.M. Effect of Processing on Texture and Microstructure of the Seaweed Durvillaea Antarctica. J. Appl. Phycol. 2020, 32, 4211–4219. [Google Scholar] [CrossRef]
- Cox, S.; Gupta, S.; Abu-Ghannam, N. Application of Response Surface Methodology to Study the Influence of Hydrothermal Processing on Phytochemical Constituents of the Irish Edible Brown Seaweed Himanthalia Elongata. Bot. Mar. 2011, 54, 471–480. [Google Scholar] [CrossRef]
- Poojary, M.M.; Barba, F.J.; Aliakbarian, B.; Donsì, F.; Pataro, G.; Dias, D.A.; Juliano, P. Innovative Alternative Technologies to Extract Carotenoids from Microalgae and Seaweeds. Mar. Drugs. 2016, 14, 214. [Google Scholar] [CrossRef]
- Domínguez-González, R.; Moreda-Piñeiro, A.; Bermejo-Barrera, A.; Bermejo-Barrera, P. Application of Ultrasound-Assisted Acid Leaching Procedures for Major and Trace Elements Determination in Edible Seaweed by Inductively Coupled Plasma-Optical Emission Spectrometry. Talanta 2005, 66, 937–942. [Google Scholar] [CrossRef]
- Muniz-Naviero, O.; Moreda-Pineiro, A.; Bermejo-Barrera, A.; Bermejo-Barrera, P. AAS Determination of Total Arsenic and Toal Inorganic Arsenic in Seaweed After High-Temperature Ultrasound-Assisted Extraction. At. Spectrosc. 2004, 25, 79–87. [Google Scholar]
- Romarís-Hortas, V.; Moreda-Piñeiro, A.; Bermejo-Barrera, P. Microwave Assisted Extraction of Iodine and Bromine from Edible Seaweed for Inductively Coupled Plasma-Mass Spectrometry Determination. Talanta 2009, 79, 947–952. [Google Scholar] [CrossRef]
- Romarís-Hortas, V.; Bermejo-Barrera, P.; Moreda-Piñeiro, A. Ultrasound-Assisted Enzymatic Hydrolysis for Iodinated Amino Acid Extraction from Edible Seaweed before Reversed-Phase High Performance Liquid Chromatography-Inductively Coupled Plasma-Mass Spectrometry. J. Chromatogr. A. 2013, 1309, 33–40. [Google Scholar] [CrossRef]
- Wang, X.; Xu, J.; Wang, L.; Gao, X.; Fu, X.; Zhao, Y. Optimization of Microwave-Ultrasound-Assisted Enzymatic Hydrolysis Extraction of Iodine Amino Acids in Laminaria by High Performance Liquid Chromatography with a Photodiode Array Detector. Algal. Res. 2019, 39, 101452. [Google Scholar] [CrossRef]
- Charoux, C.M.G.; O’Donnell, C.P.; Tiwari, B.K. Chapter 9—Ultrasound Processing and Food Quality. In Ultrasound: Advances for Food Processing and Preservation; Bermudez-Aguirre, D., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 215–235. ISBN 978-0-12-804581-7. [Google Scholar]
- Zhang, Z.-H.; Wang, L.-H.; Zeng, X.-A.; Han, Z.; Brennan, C.S. Non-Thermal Technologies and Its Current and Future Application in the Food Industry: A Review. IJFST 2019, 54, 1–13. [Google Scholar] [CrossRef]
- Astráin-Redín, L.; Ciudad-Hidalgo, S.; Raso, J.; Condón, S.; Cebrián, G.; Álvarez, I. Application of High-Power Ultrasound in the Food Industry | IntechOpen. In Sonochemical Reactions; Karakuş, S., Ed.; IntechOpen: London, UK, 2019; pp. 103–126. [Google Scholar]
- Condón-Abanto, S.; Raso, J.; Arroyo, C.; Lyng, J.G.; Condón, S.; Álvarez, I. Evaluation of the Potential of Ultrasound Technology Combined with Mild Temperatures to Reduce Cadmium Content of Edible Crab (Cancer Pagurus). Ultrason. Sonochem. 2018, 48, 550–554. [Google Scholar] [CrossRef]
- Zhou, J.L.; Huang, P.L.; Lin, R.G. Sorption and Desorption of Cu and Cd by Macroalgae and Microalgae. Environ. Pollut. 1998, 101, 67–75. [Google Scholar] [CrossRef]
- Gall, E.A.; Küpper, F.C.; Kloareg, B. A Survey of Iodine Content in Laminaria Digitata. Bot. Mar. 2004, 47, 30–37. [Google Scholar] [CrossRef]
- MacArtain, P.; Gill, C.I.R.; Brooks, M.; Campbell, R.; Rowland, I.R. Nutritional Value of Edible Seaweeds. Nutr. Rev. 2007, 65, 535–543. [Google Scholar] [CrossRef]
- Nitschke, U.; Ruth, A.A.; Dixneuf, S.; Stengel, D.B. Molecular Iodine Emission Rates and Photosynthetic Performance of Different Thallus Parts of Laminaria Digitata (Phaeophyceae) during Emersion. Planta 2011, 233, 737–748. [Google Scholar] [CrossRef]
- Schiener, P.; Black, K.D.; Stanley, M.S.; Green, D.H. The Seasonal Variation in the Chemical Composition of the Kelp Species Laminaria Digitata, Laminaria Hyperborea, Saccharina Latissima and Alaria Esculenta. J. Appl. Phycol. 2015, 27, 363–373. [Google Scholar] [CrossRef]
- Villare, R.; Puente, X.; Carballeira, A. Seasonal Variation and Background Levels of Heavy Metals in Two Green Seaweeds. Environ. Pollut. 2002, 119, 79–90. [Google Scholar] [CrossRef]
- Hwang, Y.O.; Park, S.G.; Park, G.Y.; Choi, S.M.; Kim, M.Y. Total Arsenic, Mercury, Lead, and Cadmium Contents in Edible Dried Seaweed in Korea. Food. Addit. Contam. Part B Surveill 2010, 3, 7–13. [Google Scholar] [CrossRef]
- Maehre, H.K.; Malde, M.K.; Eilertsen, K.-E.; Elvevoll, E.O. Characterization of Protein, Lipid and Mineral Contents in Common Norwegian Seaweeds and Evaluation of Their Potential as Food and Feed. J. Sci. Food. Agric. 2014, 94, 3281–3290. [Google Scholar] [CrossRef]
- Yamada, M.; Yamamoto, K.; Ushihara, Y.; Kawai, H. Variation in Metal Concentrations in the Brown Alga Undaria Pinnatifida in Osaka Bay, Japan. Phycol. Res. 2007, 55, 222–230. [Google Scholar] [CrossRef]
- Desideri, D.; Cantaluppi, C.; Ceccotto, F.; Meli, M.A.; Roselli, C.; Feduzi, L. Essential and Toxic Elements in Seaweeds for Human Consumption. J. Toxicol. Environ. Health. A 2016, 79, 112–122. [Google Scholar] [CrossRef]
- Ronan, J.M.; Stengel, D.B.; Raab, A.; Feldmann, J.; O’Hea, L.; Bralatei, E.; McGovern, E. High Proportions of Inorganic Arsenic in Laminaria Digitata but Not in Ascophyllum Nodosum Samples from Ireland. Chemosphere 2017, 186, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Taylor, V.F.; Jackson, B.P. Concentrations and Speciation of Arsenic in New England Seaweed Species Harvested for Food and Agriculture. Chemosphere 2016, 163, 6–13. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Technical Report Series 95—Evaluation of Certain Contaminants in Food: Seventy-Second Report of the Joint FAO/WHO Expert Committee in Food Additives; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Rose, M.; Lewis, J.; Langford, N.; Baxter, M.; Origgi, S.; Barber, M.; MacBain, H.; Thomas, K. Arsenic in Seaweed--Forms, Concentration and Dietary Exposure. Food. Chem. Toxicol. 2007, 45, 1263–1267. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.F. Arsenic Toxicity and Potential Mechanisms of Action. Toxicol. Lett. 2002, 133, 1–16. [Google Scholar] [CrossRef]
- Almela, C.; Algora, S.; Benito, V.; Clemente, M.J.; Devesa, V.; Súñer, M.A.; Vélez, D.; Montoro, R. Heavy Metal, Total Arsenic, and Inorganic Arsenic Contents of Algae Food Products. J. Agric. Food Chem. 2002, 50, 918–923. [Google Scholar] [CrossRef] [PubMed]
- CEVA. Edible Seaweed and Microalgae—Regulatory Status in France and Europe; CEVA: Pen Lan Peninsula, France, 2014. [Google Scholar]
- AFSSA. Opinion of the French Food Safety Agency on the Recommended Maximum Inorganic Arsenic Content of Laminaria and Consumption of These Seaweeds in Light of Their High Iodine Content; AFSSA: Austin, TX, USA, 2009. [Google Scholar]
- Cherry, P.; O’Hara, C.; Magee, P.J.; McSorley, E.M.; Allsopp, P.J. Risks and Benefits of Consuming Edible Seaweeds. Nutr. Rev. 2019, 77, 307–329. [Google Scholar] [CrossRef]
- ANSES. OPINION of the French Agency for Food, Environmental and Occupational Health & Safety on the Risk of Excess Iodine Intake from the Consumption of Seaweed in Foodstuffs; ANSES Opinion Request No 2017-SA-0086; ANSES: Buenos Aires, Argentina, 2018. [Google Scholar]
- Monteiro, M.S.; Sloth, J.; Holdt, S.; Hansen, M. Analysis and Risk Assessment of Seaweed. EFSA J. 2019, 17, e170915. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA) Scientific Opinion on Dietary Reference Values for Iodine. EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). EFSA J. 2014, 12, 3660. [Google Scholar]
- Zava, T.T.; Zava, D.T. Assessment of Japanese Iodine Intake Based on Seaweed Consumption in Japan: A Literature-Based Analysis. Thyroid Res. 2011, 4, 14. [Google Scholar] [CrossRef] [PubMed]
- Raize, O.; Argaman, Y.; Yannai, S. Mechanisms of Biosorption of Different Heavy Metals by Brown Marine Macroalgae. Biotechnol. Bioeng. 2004, 87, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Lomax, C.; Liu, W.-J.; Wu, L.; Xue, K.; Xiong, J.; Zhou, J.; McGrath, S.P.; Meharg, A.A.; Miller, A.J.; Zhao, F.-J. Methylated Arsenic Species in Plants Originate from Soil Microorganisms. New Phytol. 2012, 193, 665–672. [Google Scholar] [CrossRef]
- Güven, K.C.; Akyüz, K.; Yurdun, T. Selectivity of Heavy Metal Binding by Algal Polysaccharides. Environ. Toxicol. Chem. 1995, 47, 65–70. [Google Scholar] [CrossRef]
- Henriques, B.; Teixeira, A.; Figueira, P.; Reis, A.T.; Almeida, J.; Vale, C.; Pereira, E. Simultaneous Removal of Trace Elements from Contaminated Waters by Living Ulva Lactuca. Sci. Total Environ. 2019, 652, 880–888. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Chai, C.; Qian, Q.; Yan, X.; Fan, X. Determination of Chemical Species of Iodine in Some Seaweeds (I). Sci. Total Environ. 1997, 204, 215–221. [Google Scholar] [CrossRef]
- Küpper, F.C.; Carpenter, L.J.; McFiggans, G.B.; Palmer, C.J.; Waite, T.J.; Boneberg, E.-M.; Woitsch, S.; Weiller, M.; Abela, R.; Grolimund, D.; et al. Iodide Accumulation Provides Kelp with an Inorganic Antioxidant Impacting Atmospheric Chemistry. PNAS 2008, 105, 6954–6958. [Google Scholar] [CrossRef] [PubMed]
- Seymour, I.J.; Burfoot, D.; Smith, R.L.; Cox, L.A.; Lockwood, A. Ultrasound Decontamination of Minimally Processed Fruits and Vegetables. IFST 2002, 37, 547–557. [Google Scholar] [CrossRef]
- Knorr, D.; Zenker, M.; Heinz, V.; Lee, D.-U. Applications and Potential of Ultrasonics in Food Processing. Trends Food Sci. Technol. 2004, 15, 261–266. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Sicaire, A.-G.; Meullemiestre, A.; Fabiano-Tixier, A.-S.; Abert-Vian, M. Ultrasound Assisted Extraction of Food and Natural Products. Mechanisms, Techniques, Combinations, Protocols and Applications. A Review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Di Palma, L.; Mecozzi, R. Heavy Metals Mobilization from Harbour Sediments Using EDTA and Citric Acid as Chelating Agents. J. Hazard. Mater. 2007, 147, 768–775. [Google Scholar] [CrossRef]
- Rahman, M.A.; Hasegawa, H.; Ueda, K.; Maki, T.; Rahman, M.M. Influence of EDTA and Chemical Species on Arsenic Accumulation in Spirodela Polyrhiza L. (Duckweed). Ecotoxicol. Environ. Saf. 2008, 70, 311–318. [Google Scholar] [CrossRef][Green Version]
- Hasselström, L.; Visch, W.; Gröndahl, F.; Nylund, G.M.; Pavia, H. The Impact of Seaweed Cultivation on Ecosystem Services—A Case Study from the West Coast of Sweden. Mar. Pollut. Bull. 2018, 133, 53–64. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noriega-Fernández, E.; Sone, I.; Astráin-Redín, L.; Prabhu, L.; Sivertsvik, M.; Álvarez, I.; Cebrián, G. Innovative Ultrasound-Assisted Approaches towards Reduction of Heavy Metals and Iodine in Macroalgal Biomass. Foods 2021, 10, 649. https://doi.org/10.3390/foods10030649
Noriega-Fernández E, Sone I, Astráin-Redín L, Prabhu L, Sivertsvik M, Álvarez I, Cebrián G. Innovative Ultrasound-Assisted Approaches towards Reduction of Heavy Metals and Iodine in Macroalgal Biomass. Foods. 2021; 10(3):649. https://doi.org/10.3390/foods10030649
Chicago/Turabian StyleNoriega-Fernández, Estefanía, Izumi Sone, Leire Astráin-Redín, Leena Prabhu, Morten Sivertsvik, Ignacio Álvarez, and Guillermo Cebrián. 2021. "Innovative Ultrasound-Assisted Approaches towards Reduction of Heavy Metals and Iodine in Macroalgal Biomass" Foods 10, no. 3: 649. https://doi.org/10.3390/foods10030649
APA StyleNoriega-Fernández, E., Sone, I., Astráin-Redín, L., Prabhu, L., Sivertsvik, M., Álvarez, I., & Cebrián, G. (2021). Innovative Ultrasound-Assisted Approaches towards Reduction of Heavy Metals and Iodine in Macroalgal Biomass. Foods, 10(3), 649. https://doi.org/10.3390/foods10030649





