High-Pressure Processing for the Production of Added-Value Claw Meat from Edible Crab (Cancer pagurus)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Processing
2.2.1. High-Pressure Treatments
2.2.2. Water Immersion Cooking
2.2.3. Thermal Pasteurization
2.3. Analytical Determinations
2.3.1. Thermal Transition Properties
2.3.2. Processing Yield
2.3.3. Moisture, Protein, and Ash Content
2.3.4. Transverse Relaxation Time (T2)
2.3.5. pH
2.3.6. Total Volatile Basic Nitrogen
2.3.7. Visual Appearance and Color
2.3.8. Fatty Acid Profile
2.3.9. Microbial Inactivation and Microbial Counts
2.3.10. Salt Content
2.4. Statistical Analysis
3. Results and Discussion
3.1. Thermal Transition Properties of Claw Muscle Proteins
3.2. Processing Yield and Moisture, Protein, and Ash Content
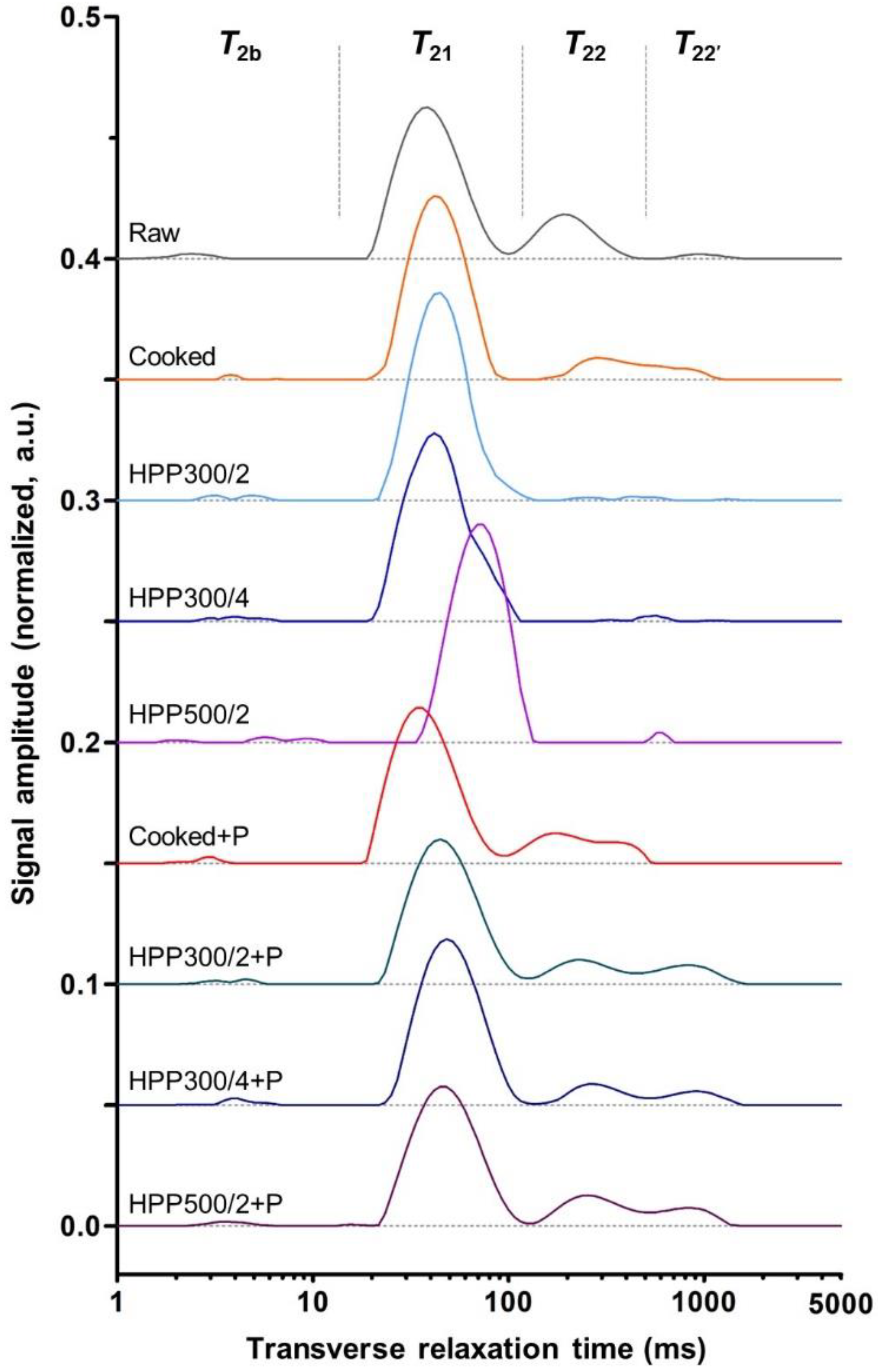
3.3. Muscle Water Distribution and Mobility (Transverse Relaxation Time, T2)
3.4. pH and Total Volatile Basic Nitrogen
3.5. Visual Appearance and Color
3.6. Fatty Acid Profile
3.7. Microbial Inactivation and Microbial Counts
3.8. Salt Diffusion into the Claw Meat
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Klaoudatos, D.; Conides, A.; Anastasopoulou, A.; Dulcic, J. Age, growth, mortality and sex ratio of the inshore population of the edible crab, Cancer pagurus (Linnaeus 1758) in South Wales (UK). J. Appl. Ichthyol. 2013, 29, 579–586. [Google Scholar] [CrossRef]
- Eurostat. Landings of Fishery Products. Edible Crab (Cancer pagurus). Available online: http://ec.europa.eu/eurostat/web/fisheries/data/database (accessed on 15 March 2021).
- Wiech, M.; Vik, E.; Duinker, A.; Frantzen, S.; Bakke, S.; Maage, A. Effects of cooking and freezing practices on the distribution of cadmium in different tissues of the brown crab (Cancer pagurus). Food Control. 2017, 75, 14–20. [Google Scholar] [CrossRef]
- Acrunet (Atlantic Crab Resource Users Network). Activity 5: Characterisation, Analysis and Modelling of the European Brown Crab Industry. Available online: http://www.acrunet.eu/acrunet-deliverables (accessed on 15 March 2021).
- Dima, J.B.; Barón, P.J.; Zaritzky, N.E. Pasteurization conditions and evaluation of quality parameters of frozen packaged crab meat. J. Aquat. Food Prod. Technol. 2015, 25, 745–759. [Google Scholar] [CrossRef]
- Ward, D.R.; Nickelson, R.; Finne, G.; Hopson, D.J. Processing technologies and their effects on microbiological properties, thermal processing efficiency, and yield of blue crab. Mar. Fish. Rev. 1983, 45, 38–43. [Google Scholar]
- Niamnuy, C.; Devahastin, S.; Soponronnarit, S. Quality Changes of Shrimp during Boiling in Salt Solution. J. Food Sci. 2007, 72, S289–S297. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Andrés, J.M.; Charoux, C.M.G.; Cullen, P.J.; Tiwari, B.K.; Pérez, J. Chemical Modifications of Lipids and Proteins by Nonthermal Food Processing Technologies. J. Agric. Food Chem. 2018, 66, 5041–5054. [Google Scholar] [CrossRef]
- Campus, M. High Pressure Processing of Meat, Meat Products and Seafood. Food Eng. Rev. 2010, 2, 256–273. [Google Scholar] [CrossRef]
- Aubourg, S.P. Impact of high-pressure processing on chemical constituents and nutritional properties in aquatic foods: A review. Int. J. Food Sci. Technol. 2017, 53, 873–891. [Google Scholar] [CrossRef]
- Nanda, P.K.; Das, A.K.; Dandapat, P.; Dhar, P.; Bandyopadhyay, S.; Dib, A.L.; Lorenzo, J.M.; Gagaoua, M. Nutritional aspects, flavour profile and health benefits of crab meat based novel food products and valorisation of processing waste to wealth: A review. Trends Food Sci. Technol. 2021, 112, 252–267. [Google Scholar] [CrossRef]
- Martínez, M.; Velazquez, G.; Cando, D.; Núñez-Flores, R.; Borderías, A.; Moreno, H. Effects of high pressure processing on protein fractions of blue crab (Callinectes sapidus) meat. Innov. Food Sci. Emerg. Technol. 2017, 41, 323–329. [Google Scholar] [CrossRef]
- Ye, T.; Chen, X.; Chen, Z.; Yao, H.; Wang, Y.; Lin, L.; Lu, J. Quality and microbial community of high pressure shucked crab (Eriocheir sinensis) meat stored at 4 °C. J. Food Process. Preserv. 2021, 45, e15330. [Google Scholar] [CrossRef]
- Ye, T.; Chen, X.; Chen, Z.; Liu, R.; Wang, Y.; Lin, L.; Lu, J. Quality characteristics of shucked crab meat (Eriocheir sinensis) processed by high pressure during superchilled storage. J. Food Biochem. 2021, e13708. [Google Scholar] [CrossRef]
- Leadley, C.; Shaw, H.; Green, A.; Burling, S.; Bhandari, D. Pilot Trials to Determine the Benefits of High Pressure Processing (HPP) for Seafood in the UK; Report on Phase 2 Studies. Report No. FMT/REP/95900/2; CCFRA: Chipping Campden, UK, 2008. [Google Scholar]
- Elamin, W.M.; Endan, J.; Yusof, Y.-A.; Shamsudin, R.; Ahmedov, A. Effect of high pressure processing on sensory and physical attributes of Malaysian shrimps and mud crabs. Am. Sci. Res. J. Eng. Technol. Sci. (ASRJETS) 2015, 14, 91–96. [Google Scholar]
- Kristoffersen, S.; Siikavuopio, S.; Dahl, R.; Jakobsen, R.; Tidemann, E. Kongekrabbe—Evaluering av Metoder for Foredling [Red King Crab—Evaluation of Methods for Valorization]; Report No. 32/2009; Nofima Marin: Tromsø, Norway, 2009; Available online: https://www.nofima.no/filearchive/Rapport%2032-2009.pdf (accessed on 15 March 2021).
- ECFF (European Chilled Food Federation). Recommendations for the Production of Prepackaged Chilled Food; The European Chilled Food Federation: Helsinki, Finland, 2006; Available online: http://www.ecff.net/images/ECFF_Recommendations_2nd_ed_18_12_06.pdf (accessed on 15 March 2021).
- FDA (Food and Drug Administration). Fish and Fishery Products Hazards and Controls Guidance, 4th ed.; Department of Health and Human Services, Food and Drug Administration, Center for Food Safety and Applied Nutrition: Washington, DC, USA, 2011. Available online: https://www.fda.gov/downloads/Food/GuidanceRegulation/UCM253143.pdf (accessed on 15 March 2021).
- AOAC. Official Methods of Analysis of AOAC International, 17th ed.; AOAC International: Gaithersburg, MD, USA, 2002. [Google Scholar]
- Li, X.; Liu, S.; Su, W.; Cai, L. Physical quality changes of precooked Chinese shrimp Fenneropenaeus chinensis and correlation to water distribution and mobility by low-field NMR during frozen storage. J. Food Process. Preserv. 2017, 41, 13220. [Google Scholar] [CrossRef]
- Lorentzen, G.; Rotabakk, B.T.; Olsen, S.H.; Skuland, A.V.; Siikavuopio, S.I. Shelf life of snow crab clusters (Chionoecetes opilio) stored at 0 and 4 °C. Food Control. 2016, 59, 454–460. [Google Scholar] [CrossRef]
- Malle, P.; Poumeyrol, M. A New Chemical Criterion for the Quality Control of Fish: Trimethylamine/Total Volatile Basic Nitrogen (%). J. Food Prot. 1989, 52, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Brunton, N.P.; Mason, C.; Collins, M.J. Rapid microwave assisted preparation of fatty acid methyl esters for the analysis of fatty acid profiles in foods. J. Anal. Chem. 2015, 70, 1218–1224. [Google Scholar] [CrossRef]
- Gangopadhyay, N.; Rai, D.K.; Brunton, N.P.; Gallagher, E.; Harrison, S.M. Fatty acids, sterols and tocols in Irish barley varieties: Profiling and correlation analysis. Eur. J. Lipid Sci. Technol. 2016, 119, 1600213. [Google Scholar] [CrossRef]
- Lascorz, D.; Torella, E.; Lyng, J.G.; Arroyo, C. The potential of ohmic heating as an alternative to steam for heat processing shrimps. Innov. Food Sci. Emerg. Technol. 2016, 37, 329–335. [Google Scholar] [CrossRef]
- Benjakul, S.; Sutthipan, N. Comparative study on chemical composition, thermal properties and microstructure between the muscle of hard shell and soft shell mud crabs. Food Chem. 2009, 112, 627–633. [Google Scholar] [CrossRef]
- Dima, J.B.; Barón, P.J.; Zaritzky, N.E. Mathematical modeling of the heat transfer process and protein denaturation during the thermal treatment of Patagonian marine crabs. J. Food Eng. 2012, 113, 623–634. [Google Scholar] [CrossRef]
- Balny, C.; Masson, P. Effects of high pressure on proteins. Food Rev. Int. 1993, 9, 611–628. [Google Scholar] [CrossRef]
- Rossner, K.L.; Sherman, R.G. Organization of a Skeletal Muscle Insertion in the Crab Carcinus maenas. Trans. Am. Microsc. Soc. 1976, 95, 46. [Google Scholar] [CrossRef] [PubMed]
- Dang, T.T.; Gringer, N.; Jessen, F.; Olsen, K.; Bøknæs, N.; Nielsen, P.L.; Orlien, V. Emerging and potential technologies for facilitating shrimp peeling: A review. Innov. Food Sci. Emerg. Technol. 2018, 45, 228–240. [Google Scholar] [CrossRef]
- McDermott, A.; Whyte, P.; Brunton, N.; Lyng, J.; Bolton, D.J. Increasing the Yield of Irish Brown Crab (Cancer pagurus) during Processing without Adversely Affecting Shelf-Life. Foods 2018, 7, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maulvault, A.L.; Anacleto, P.; Lourenço, H.M.; Carvalho, M.L.; Nunes, M.L.; Marques, A. Nutritional quality and safety of cooked edible crab (Cancer pagurus). Food Chem. 2012, 133, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Roer, R.; Dillaman, R. The structure and calcification of the crustacean cuticle. Am. Zool. 1984, 24, 893–909. [Google Scholar] [CrossRef]
- Barrento, S.; Marques, A.; Teixeira, B.; Mendes, R.; Bandarra, N.; Vaz-Pires, P.; Nunes, M.L. Chemical composition, cholesterol, fatty acid and amino acid in two populations of brown crab Cancer pagurus: Ecological and human health implications. J. Food Compos. Anal. 2010, 23, 716–725. [Google Scholar] [CrossRef]
- Martínez-Maldonado, M.A.; León, J.A.R.-D.; Méndez-Montealvo, M.G.; Morales-Sánchez, E.; Velazquez, G. Effect of the Cooking Process on the Gelling Properties of Whole and Minced Jumbo Lump of Blue Crab (Callinectes sapidus). J. Aquat. Food Prod. Technol. 2018, 27, 418–429. [Google Scholar] [CrossRef]
- Niamnuy, C.; Devahastin, S.; Soponronnarit, S. Changes in protein compositions and their effects on physical changes of shrimp during boiling in salt solution. Food Chem. 2008, 108, 165–175. [Google Scholar] [CrossRef]
- Jantakoson, T.; Kijroongrojana, K.; Benjakul, S. Effect of high pressure and heat treatments on black tiger shrimp (Penaeus monodon Fabricius) muscle protein. Int. Aquat. Res. 2012, 4, 19. [Google Scholar] [CrossRef] [Green Version]
- Silva, J.L.; Foguel, D.; Royer, C.A. Pressure provides new insights into protein folding, dynamics and structure. Trends Biochem. Sci. 2001, 26, 612–618. [Google Scholar] [CrossRef]
- de Oliveira, F.A.; Neto, O.C.; dos Santos, L.M.R.; Ferreira, E.H.R.; Rosenthal, A. Effect of high pressure on fish meat quality-A review. Trends Food Sci. Technol. 2017, 66, 1–19. [Google Scholar] [CrossRef]
- Hedges, N.D.; Goodband, R.M. The Influence of High Hydrostatic Pressure on the Water Holding Capacity of Fish Muscle. Presentation at the 1st Trans-Atlantic Fisheries Technology Conference (TAFT 2003), Reykjavik, Iceland. Available online: http://digital.csic.es/bitstream/10261/90401/1/TAFT_2003_Proceedings.pdf (accessed on 15 March 2021).
- Erikson, U.; Standal, I.B.; Aursand, I.G.; Veliyulin, E.; Aursand, M. Use of NMR in fish processing optimization: A review of recent progress. Magn. Reson. Chem. 2012, 50, 471–480. [Google Scholar] [CrossRef]
- Cheng, S.; Tang, Y.; Zhang, T.; Song, Y.; Wang, X.; Wang, H.; Wang, H.; Tan, M. An approach for monitoring the dynamic states of water in shrimp during drying process with LF-NMR and MRI. Dry. Technol. 2017, 36, 841–848. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, W.; Zhou, F.; Zheng, Y.; Wang, X. Tenderness and histochemistry of muscle tissues from Eriocheir sinensis. Food Biosci. 2020, 34, 100479. [Google Scholar] [CrossRef]
- Zhang, L.; Yin, M.; Wang, X. Meat texture, muscle histochemistry and protein composition of Eriocheir sinensis with different size traits. Food Chem. 2021, 338, 127632. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.; Liu, A.; Zheng, J.; Wang, P.; Yin, S. High pressure processing and water holding capacity of sea bass skeletal muscle. J. Aquat. Food Prod. Technol. 2015, 24, 740–751. [Google Scholar] [CrossRef]
- Kaur, B.P.; Kaushik, N.; Rao, P.S.; Mishra, H.N. Chilled storage of high pressure processed black tiger shrimp (Penaeus monodon). J. Aquat. Food Prod. Technol. 2015, 24, 283–299. [Google Scholar] [CrossRef]
- Yi, J.; Xu, Q.; Hu, X.; Dong, P.; Liao, X.; Zhang, Y. Shucking of bay scallop (Argopecten irradians) using high hydrostatic pressure and its effect on microbiological and physical quality of adductor muscle. Innov. Food Sci. Emerg. Technol. 2013, 18, 57–64. [Google Scholar] [CrossRef]
- Knorr, D.; Heinz, V.; Buckow, R. High pressure application for food biopolymers. Biochim. Biophys. Acta Proteins Proteom. 2006, 1764, 619–631. [Google Scholar] [CrossRef]
- Grossi, A.; Olsen, K.; Bolumar, T.; Rinnan, Å.; Øgendal, L.H.; Orlien, V. The effect of high pressure on the functional properties of pork myofibrillar proteins. Food Chem. 2016, 196, 1005–1015. [Google Scholar] [CrossRef]
- Yang, J.; Powers, J.R. Effects of high pressure on food proteins. In High Pressure Processing of Food: Principles, Technology and Applications; Balasubramaniam, V.M., Barbosa-Cánovas, G.V., Lelieveld, H.L.M., Eds.; Springer: New York, NY, USA, 2016; pp. 353–389. [Google Scholar] [CrossRef]
- Bertram, H.C.; Kristensen, M.; Andersen, H.J. Functionality of myofibrillar proteins as affected by pH, ionic strength and heat treatment–A low-field NMR study. Meat Sci. 2004, 68, 249–256. [Google Scholar] [CrossRef]
- McDonnell, C.K.; Allen, P.; Morin, C.; Lyng, J.G. The effect of ultrasonic salting on protein and water–protein interactions in meat. Food Chem. 2014, 147, 245–251. [Google Scholar] [CrossRef]
- Xuan, X.-T.; Cui, Y.; Lin, X.-D.; Yu, J.-F.; Liao, X.-J.; Ling, J.-G.; Shang, H.-T. Impact of high hydrostatic pressure on the shelling efficacy, physicochemical properties, and microstructure of fresh razor clam (Sinonovacula constricta). J. Food Sci. 2018, 83, 284–293. [Google Scholar] [CrossRef]
- McDonnell, C.K.; Allen, P.; Duggan, E.; Arimi, J.M.; Casey, E.; Duane, G.; Lyng, J.G. The effect of salt and fibre direction on water dynamics, distribution and mobility in pork muscle: A low field NMR study. Meat Sci. 2013, 95, 51–58. [Google Scholar] [CrossRef]
- Li, W.; Wang, P.; Xu, X.; Xing, T.; Zhou, G. Use of low-field nuclear magnetic resonance to characterize water properties in frozen chicken breasts thawed under high pressure. Eur. Food Res. Technol. 2014, 239, 183–188. [Google Scholar] [CrossRef]
- Bertram, H.C.; Dønstrup, S.; Karlsson, A.H.; Andersen, H.J. Continuous distribution analysis of T2 relaxation in meat—An approach in the determination of water-holding capacity. Meat Sci. 2002, 60, 279–285. [Google Scholar] [CrossRef]
- Lian, F.; Lyng, J.G.; Brunton, N.P. Application of the Check-All-that-Apply (CATA) Sensory Method to Identify Drivers of Liking in ready-to-eat Meat of edible crab (Cancer pagurus). Presented at the 47th Conference of the West European Fish Technologists’ Association (WEFTA 2017), Dublin, Ireland, 9–12 October 2017. [Google Scholar]
- Pearce, K.L.; Rosenvold, K.; Andersen, H.J.; Hopkins, D.L. Water distribution and mobility in meat during the conversion of muscle to meat and ageing and the impacts on fresh meat quality attributes-A review. Meat Sci. 2011, 89, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Anupama, T.K.; Laly, S.J.; Kumar, K.N.A.; Sankar, T.V.; Ninan, G. Biochemical and microbiological assessment of crucifix crab (Charybdis feriatus) stored at 4 °C. J. Aquat. Food Prod. Technol. 2018, 27, 531–541. [Google Scholar] [CrossRef]
- Shahidi, F. Proteins from seafood processing discards. In Seafood Proteins; Sikorski, Z.E., Bonnie, S.P., Shahidi, F., Eds.; Chapman & Hall: New York, NY, USA, 1994; pp. 171–193. [Google Scholar] [CrossRef]
- Anacleto, P.; Teixeira, B.; Marques, P.; Pedro, S.; Nunes, M.L.; Marques, A. Shelf-life of cooked edible crab (Cancer pagurus) stored under refrigerated conditions. LWT Food Sci. Technol. 1994, 44, 1376–1382. [Google Scholar] [CrossRef]
- Bindu, J.; Ginson, J.; Kamalakanth, C.K.; Asha, K.K.; Gopal, T.K.S. Physico-chemical changes in high pressure treated Indian white prawn (Fenneropenaeus indicus) during chill storage. Innov. Food Sci. Emerg. Technol. 2013, 17, 37–42. [Google Scholar] [CrossRef]
- Jun, J.-Y.; Jung, M.-J.; Kim, D.-S.; Jeong, I.-H.; Kim, B.-M. Postmortem changes in physiochemical and sensory properties of red snow crab (Chionoecetes japonicus) leg muscle during freeze storage. Fish. Aquat. Sci. 2017, 20, 13. [Google Scholar] [CrossRef] [Green Version]
- Biji, K.B.; Shamseer, R.M.; Mohan, C.O.; Ravishankar, C.N.; Mathew, S.; Gopal, T.K.S. Effect of thermal processing on the biochemical constituents of green mussel (Perna viridis) in Tin-free-steel cans. J. Food Sci. Technol. 2015, 52, 6804–6809. [Google Scholar] [CrossRef] [Green Version]
- Commission Regulation (EC). No. 1022/2008 of 17 October 2008 amending Regulation (EC) No. 2074/2005 as regards the total volatile basic nitrogen (TVB-N) limits. Off. J. Eur. Union 2008, 052, 18–20. [Google Scholar]
- Truong, B.Q.; Buckow, R.; Stathopoulos, C.E.; Nguyen, M.H. Advances in high-pressure processing of fish muscles. Food Eng. Rev. 2014, 7, 109–129. [Google Scholar] [CrossRef]
- Medler, S.; Mykles, D.L. Muscle structure, fiber types, and physiology. In The Natural History of the Crustacea Physiology; Cheng, E.S., Thiel, M., Eds.; Oxford University Press: Oxford, UK, 2015; pp. 103–133. [Google Scholar]
- Wade, N.M.; Gabaudan, J.; Glencross, B.D. A review of carotenoid utilisation and function in crustacean aquaculture. Rev. Aquac. 2015, 9, 141–156. [Google Scholar] [CrossRef]
- Kruk, Z.A.; Yun, H.; Rutley, D.L.; Lee, E.J.; Kim, Y.J.; Jo, C. The effect of high pressure on microbial population, meat quality and sensory characteristics of chicken breast fillet. Food Control. 2011, 22, 6–12. [Google Scholar] [CrossRef]
- Sazonova, S.; Grube, M.; Shvirksts, K.; Galoburda, R.; Gramatina, I. FTIR spectroscopy studies of high pressure-induced changes in pork macromolecular structure. J. Mol. Struct. 2019, 1186, 377–383. [Google Scholar] [CrossRef]
- Humaid, S.; Nayyar, D.; Bolton, J.; Skonberg, D.I. Physicochemical Properties and Consumer Acceptance of High-Pressure Processed, Sous Vide-Cooked Lobster Tails. J. Food Sci. 2019, 84, 3454–3462. [Google Scholar] [CrossRef] [PubMed]
- Lorente-Cebrián, S.; Costa, A.G.V.; Navas-Carretero, S.; Zabala, M.; Laiglesia, L.M.; Martínez, J.A.; Moreno-Aliaga, M.J. An update on the role of omega-3 fatty acids on inflammatory and degenerative diseases. J. Physiol. Biochem. 2015, 71, 341–349. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The Importance of the Omega-6/Omega-3 Fatty Acid Ratio in Cardiovascular Disease and Other Chronic Diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Keum, Y.-S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolism, and significance—A review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef]
- Commission Regulation (EU). No. 116/2010 of 9 February 2010 amending Regulation (EC) No. 1924/2006 of the European Parliament and of the Council with regard to the list of nutrition claims. Off. J. Eur. Union 2010, 001, 16–18. [Google Scholar]
- Cruz-Romero, M.C.; Kerry, J.P.; Kelly, A.L. Fatty acids, volatile compounds and colour changes in high-pressure-treated oysters (Crassostrea gigas). Innov. Food Sci. Emerg. Technol. 2008, 9, 54–61. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; Montero, P.; Fernández-Martín, F.; Calvo, M.; Gómez-Guillén, M. The effect of high-pressure treatment on functional components of shrimp (Litopenaeus vannamei) cephalothorax. Innov. Food Sci. Emerg. Technol. 2016, 34, 154–160. [Google Scholar] [CrossRef] [Green Version]
- Medina-Meza, I.G.; Barnaba, C.; Barbosa-Cánovas, G.V. Effects of high pressure processing on lipid oxidation: A review. Innov. Food Sci. Emerg. Technol. 2014, 22, 1–10. [Google Scholar] [CrossRef]
- Guyon, C.; Meynier, A.; de Lamballerie, M. Protein and lipid oxidation in meat: A review with emphasis on high-pressure treatments. Trends Food Sci. Technol. 2016, 50, 131–143. [Google Scholar] [CrossRef]
- Risso, S.J.; Carelli, A.A. Effects of conservation method and time on fatty acid composition, taste and microstructure of southern king crab (Lithodes santolla Molina, 1782) meat. J. Aquat. Food Prod. Technol. 2017, 26, 731–743. [Google Scholar] [CrossRef]
- Yagiz, Y.; Kristinsson, H.G.; Balaban, M.O.; Welt, B.A.; Ralat, M.; Marshall, M.R. Effect of high pressure processing and cooking treatment on the quality of Atlantic salmon. Food Chem. 2009, 116, 828–835. [Google Scholar] [CrossRef]
- Aubourg, S.P.; Tabilo-Munizaga, G.; Reyes, J.E.; Rodríguez, A.; Pérez-Won, M. Effect of high-pressure treatment on microbial activity and lipid oxidation in chilled coho salmon. Eur. J. Lipid Sci. Technol. 2010, 112, 362–372. [Google Scholar] [CrossRef]
- Suklim, K.; Flick, G. The effect of high hydrostatic pressure on the microbiological, physical, and sensory quality of fresh blue crab (Callinectes sapidus) meat. Presented at the 2nd Joint Trans-Atlantic Fisheries Technology Conference (TAFT 2006), Québec City, QC, Canada, 29 October–1 November 2006. [Google Scholar]
- Linton, M.; Mc Clements, J.M.J.; Patterson, M.F. Changes in the microbiological quality of shellfish, brought about by treatment with high hydrostatic pressure. Int. J. Food Sci. Technol. 2003, 38, 713–727. [Google Scholar] [CrossRef]
- Yi, J.; Zhang, L.; Ding, G.; Hu, X.; Liao, X.; Zhang, Y. High hydrostatic pressure and thermal treatments for ready-to-eat wine-marinated shrimp: An evaluation of microbiological and physicochemical qualities. Innov. Food Sci. Emerg. Technol. 2013, 20, 16–23. [Google Scholar] [CrossRef]
- Robson, A.A.; Kelly, M.S.; Latchford, J.W. Effect of temperature on the spoilage rate of whole, unprocessed crabs: Carcinus maenas, Necora puber and Cancer pagurus. Food Microbiol. 2007, 24, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Humaid, S.; Nayyar, D.; Bolton, J.; Perkins, B.; Skonberg, D.I. Refrigerated shelf-life evaluation of high pressure processed, raw and sous vide cooked lobster. High Press. Res. 2020, 40, 444–463. [Google Scholar] [CrossRef]
- Georget, E.; Sevenich, R.; Reineke, K.; Mathys, A.; Heinz, V.; Callanan, M.; Rauh, C.; Knorr, D. Inactivation of microorganisms by high isostatic pressure processing in complex matrices: A review. Innov. Food Sci. Emerg. Technol. 2015, 27, 1–14. [Google Scholar] [CrossRef]
- Suklim, K.; Flick, G.J.; Bourne, D.W.; Granata, L.A.; Eifert, J.; Williams, R.; Popham, D.; Wittman, R. Pressure-Induced Germination and Inactivation of Bacillus cereus Spores and Their Survival in Fresh Blue Crab Meat (Callinectes sapidus) During Storage. J. Aquat. Food Prod. Technol. 2008, 17, 322–337. [Google Scholar] [CrossRef]
- Condón-Abanto, S.; Arroyo, C.; Álvarez, I.; Condón, S.; Lyng, J. Application of ultrasound in combination with heat and pressure for the inactivation of spore forming bacteria isolated from edible crab (Cancer pagurus). Int. J. Food Microbiol. 2016, 223, 9–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ward, B.Q.; Carroll, B.J.; Garrett, E.S.; Reese, G.B. Survey of the US Gulf Coast for the presence of Clostridium botulinum. Appl. Microbiol. 1967, 15, 629–636. [Google Scholar] [CrossRef]
- Skinner, G.E.; Morrissey, T.R.; Patazca, E.; Loeza, V.; Halik, L.A.; Schill, K.M.; Reddy, N.R. Effect of High Pressures in Combination with Temperature on the Inactivation of Spores of Nonproteolytic Clostridium botulinum Types B and F. J. Food Prot. 2018, 81, 261–271. [Google Scholar] [CrossRef]
- Leng, X.; Zhang, L.; Huang, M.; Xu, X.; Zhou, G. Mass transfer dynamics during high pressure brining of chicken breast. J. Food Eng. 2013, 118, 296–301. [Google Scholar] [CrossRef]
- Ros-Polski, V.; Koutchma, T.; Xue, J.; Defelice, C.; Balamurugan, S. Effects of high hydrostatic pressure processing parameters and NaCl concentration on the physical properties, texture and quality of white chicken meat. Innov. Food Sci. Emerg. Technol. 2015, 30, 31–42. [Google Scholar] [CrossRef]
- Villacís, M.; Rastogi, N.; Balasubramaniam, V. Effect of high pressure on moisture and NaCl diffusion into turkey breast. LWT 2008, 41, 836–844. [Google Scholar] [CrossRef] [Green Version]
- Condón-Abanto, S.; Arroyo, C.; Álvarez, I.; Brunton, N.; Whyte, P.; Lyng, J. An assessment of the application of ultrasound in the processing of ready-to-eat whole brown crab (Cancer pagurus). Ultrason. Sonochemistry 2018, 40, 497–504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lian, F.; Måge, I.; Lorentzen, G.; Siikavuopio, S.I.; Øverbø, K.; Vang, B.; Lindberg, D. Exploring the effect of inhibitors, cooking and freezing on melanosis in snow crab (Chionoecetes opilio) clusters. Food Control. 2018, 92, 255–266. [Google Scholar] [CrossRef]




| Raw | Cooked | HPP300/2 | HPP300/4 | HPP500/2 | Cooked+P | HPP300/2+P | HPP300/4+P | HPP500/2+P | |
|---|---|---|---|---|---|---|---|---|---|
| Processing yield (%) | 0.0 | −5.2 ± 1.2 c | 0.9 ± 0.6 a | 1.3 ± 0.9 a | 1.2 ± 1.0 a | −7.7 ± 1.0 d | −1.2 ± 0.4 b | −0.6 ± 0.4 b | −1.4 ± 0.9 b |
| Moisture (%) | 76.9 ± 1.6 bcd | 74.7 ± 1.2 de | 78.5 ± 2.1 bc | 79.3 ± 2.0 ab | 81.8 ± 1.8 a | 72.4 ± 1.1 e | 76.2 ± 0.9 cd | 78.6 ± 1.6 bc | 76.5 ± 1.9 cd |
| Protein (%) | 18.0 ± 0.7 bc | 18.6 ± 0.6 ab | 16.7 ± 0.6 cd | 16.5 ± 0.7 cd | 15.2 ± 0.7 d | 19.9 ± 1.0 a | 17.9 ± 0.8 bc | 18.3 ± 0.8 b | 16.2 ± 1.2 d |
| Ash (%) | 2.26 ± 0.08 cd | 2.64 ± 0.10 ab | 2.05 ± 0.09 de | 2.06 ± 0.06 d | 1.83 ± 0.18 e | 2.74 ± 0.17 a | 2.55 ± 0.10 ab | 2.51 ± 0.09 b | 2.48 ± 0.17 bc |
| pH | 6.92 ± 0.04 f | 7.36 ± 0.04 b | 7.01 ± 0.03 e | 7.04 ± 0.02 de | 7.09 ± 0.02 d | 7.46 ± 0.03 a | 7.27 ± 0.03 c | 7.30 ± 0.02 c | 7.41 ± 0.03 ab |
| TVB-N (mg N/100 g) | 19.3 ± 1.7 e | 32.1 ± 1.9 d | 18.6 ± 0.5 e | 17.7 ± 1.1 e | 16.3 ± 0.7 e | 52.9 ± 2.9 a | 35.1 ± 2.2 cd | 36.8 ± 2.8 c | 40.7 ± 2.1 b |
| Color | |||||||||
| L* | n.d. | 76.93 ± 0.48 d | 66.89 ± 2.03 f | 69.62 ± 0.62 e | 77.78 ± 0.59 bcd | 77.36 ± 0.29 cd | 77.97 ± 0.42 bc | 78.42 ± 0.91 b | 82.50 ± 0.91 a |
| a* | n.d. | −3.55 ± 0.08 c | −1.30 ± 0.19 a | −1.03 ± 0.18 a | −2.82 ± 0.56 b | −2.78 ± 0.07 b | −2.78 ± 0.22 b | −2.74 ± 0.41 b | −2.71 ± 0.19 b |
| b* | n.d. | 6.07 ± 0.37 b | 0.40 ± 1.07 c | −0.48 ± 0.64 d | −2.01 ± 0.91 e | 6.52 ± 0.20 b | 7.48 ± 0.95 a | 6.86 ± 1.11 ab | 6.77 ± 1.09 ab |
| ΔE*cooked | n.d. | 0.0 | 11.8 ± 1.8 a | 10.1 ± 0.4 ab | 8.2 ± 0.8 bc | 1.0 ± 0.2 d | 2.0 ± 0.5 d | 1.9 ± 0.6 d | 5.7 ± 0.6 c |
| ΔE*cooked+P | n.d. | 1.0 ± 0.1 d | 12.2 ± 1.8 a | 10.6 ± 0.4 ab | 8.6 ± 0.8 b | 0.0 | 1.2 ± 0.6 d | 1.2 ± 0.6 d | 5.2 ± 0.6 c |
| T2b | T21 | T22 | T22′ | |||||
|---|---|---|---|---|---|---|---|---|
| Center (ms) | Area (%) | Center (ms) | Area (%) | Center (ms) | Area (%) | Center (ms) | Area (%) | |
| Raw | 2.5 ± 0.4 c | 0.1 ± 0.0 | 37.8 ± 1.9 cd | 37.4 ± 8.0 bc | 195.8 ± 20.2 de | 45.9 ± 10.2 a | 1004.3 ± 132.2 a | 16.7 ± 3.1 b |
| Cooked | 5.8 ± 2.2 ab | 0.1 ± 0.0 | 42.5 ± 1.4 bc | 37.1 ± 6.2 bc | 290.8 ± 17.7 c | 21.4 ± 4.8 bcd | 548.4 ± 97.1 c | 42.0 ± 10.2 a |
| HPP300/2 | 4.0 ± 0.9 bc | 0.1 ± 0.0 | 46.1 ± 6.1 bc | 79.8 ± 4.8 a | 483.0 ± 61.3 b | 11.7 ± 1.3 de | 863.7 ± 96.8 ab | 14.2 ± 2.2 b |
| HPP300/4 | 4.1 ± 0.9 bc | 0.1 ± 0.1 | 45.3 ± 11.3 bc | 81.1 ± 4.8 a | 596.6 ± 102.2 a | 18.8 ± 4.8 cde | ||
| HPP500/2 | 7.5 ± 1.6 a | 0.2 ± 0.0 | 72.3 ± 7.8 a | 90.4 ± 1.9 a | 589.0 ± 21.3 a | 9.4 ± 0.9 e | ||
| Cooked+P | 2.8 ± 0.3 c | 0.1 ± 0.0 | 35.2 ± 1.2 d | 39.4 ± 6.4 b | 176.7 ± 18.7 e | 29.9 ± 3.7 b | 326.1 ± 54.2 d | 30.6 ± 4.4 ab |
| HPP300/2+P | 3.8 ± 0.7 bc | 0.1 ± 0.0 | 45.4 ± 1.3 bc | 29.9 ± 11.1 bc | 232.2 ± 7.8 cde | 23.1 ± 7.3 bc | 800.2 ± 78.9 b | 47.0 ± 17.0 a |
| HPP300/4+P | 4.5 ± 0.7 bc | 0.1 ± 0.0 | 48.3 ± 2.0 b | 37.0 ± 8.1 bc | 265.5 ± 14.2 cd | 22.4 ± 4.9 bc | 816.3 ± 146.0 ab | 40.6 ± 11.7 a |
| HPP500/2+P | 3.9 ± 0.6 bc | 0.1 ± 0.0 | 47.2 ± 4.0 bc | 26.1 ± 4.6 c | 249.9 ± 13.7 cde | 31.4 ± 4.6 b | 826.7 ± 41.0 b | 42.4 ± 7.2 a |
| Fatty Acid | Raw | Cooked | HPP300/2 | HPP300/4 | HPP500/2 | Cooked+P | HPP300/2+P | HPP300/4+P | HPP500/2+P |
|---|---|---|---|---|---|---|---|---|---|
| 16:0 | 12.0 ± 0.9 ab | 12.2 ± 0.8 ab | 11.3 ± 0.7 b | 12.4 ± 0.3 ab | 12.1 ± 0.8 ab | 12.7 ± 0.5 a | 11.6 ± 0.6 ab | 12.4 ± 0.2 ab | 12.2 ± 0.9 ab |
| 18:0 | 4.3 ± 0.5 ab | 5.4 ± 1.0 a | 4.6 ± 0.2 ab | 4.1 ± 0.4 b | 4.3 ± 0.5 ab | 4.9 ± 1.2 ab | 4.7 ± 0.2 ab | 4.2 ± 0.4 b | 4.4 ± 0.4 ab |
| ∑ SFA 1 | 18.3 ± 0.7 | 19.2 ± 1.4 | 18.0 ± 0.8 | 18.2 ± 0.8 | 19.0 ± 2.6 | 19.2 ± 0.7 | 18.3 ± 1.1 | 18.3 ± 0.7 | 18.2 ± 1.0 |
| 16:1 | 9.1 ± 0.9 | 7.2 ± 2.3 | 6.7 ± 2.3 | 7.3 ± 1.1 | 6.8 ± 1.9 | 8.2 ± 2.6 | 6.0 ± 1.5 | 7.2 ± 1.2 | 6.8 ± 2.1 |
| 18:1n−9c | 18.5 ± 2.7 | 21.2 ± 1.1 | 18.6 ± 3.7 | 18.9 ± 0.6 | 19.7 ± 2.0 | 20.5 ± 0.9 | 17.9 ± 3.8 | 18.8 ± 0.8 | 19.6 ± 1.2 |
| 18:1n−7 | 7.1 ± 1.1 | 5.5 ± 0.2 | 6.7 ± 1.0 | 6.6 ± 0.2 | 6.6 ± 0.9 | 6.4 ± 1.3 | 6.6 ± 1.0 | 6.5 ± 0.2 | 6.6 ± 1.1 |
| ∑ MUFA 2 | 36.7 ± 1.7 a | 35.5 ± 2.0 ab | 34.6 ± 0.1 ab | 34.7 ± 1.6 ab | 34.8 ± 1.9 ab | 36.6 ± 3.0 a | 33.0 ± 1.1 b | 34.5 ± 1.9 ab | 34.7 ± 2.4 ab |
| 20:4n−6 | 4.5 ± 0.7 | 5.3 ± 1.0 | 6.2 ± 1.8 | 6.1 ± 0.6 | 5.8 ± 1.9 | 4.4 ± 0.4 | 5.9 ± 1.7 | 5.9 ± 0.5 | 5.8 ± 1.6 |
| ∑ (n−6) | 6.0 ± 0.8 | 8.5 ± 0.6 | 8.7 ± 2.3 | 8.2 ± 1.0 | 7.9 ± 3.0 | 6.5 ± 1.8 | 8.4 ± 2.0 | 8.0 ± 0.9 | 7.9 ± 2.6 |
| 20:5n−3 (EPA) | 27.4 ± 2.1 | 24.3 ± 2.2 | 25.7 ± 1.8 | 25.2 ± 2.3 | 26.9 ± 1.0 | 26.3 ± 2.9 | 26.9 ± 3.0 | 25.7 ± 1.7 | 27.5 ± 1.6 |
| 22:5n−3 | 1.6 ± 0.9 | 0.7 ± 0.4 | 2.1 ± 1.4 | 1.7 ± 0.5 | 0.9 ± 0.2 | 0.8 ± 0.2 | 2.0 ± 1.1 | 1.5 ± 0.4 | 1.0 ± 0.3 |
| 22:6n−3 (DHA) | 10.0 ± 1.4 | 11.8 ± 0.6 | 10.8 ± 1.2 | 12.0 ± 2.0 | 10.5 ± 2.5 | 10.6 ± 1.8 | 11.4 ± 1.9 | 11.9 ± 1.8 | 10.7 ± 2.1 |
| ∑ (n−3) | 39.0 ± 1.6 | 36.9 ± 1.5 | 38.6 ± 1.9 | 38.9 ± 0.8 | 38.3 ± 2.0 | 37.7 ± 1.4 | 40.3 ± 2.3 | 39.1 ± 0.6 | 39.2 ± 0.8 |
| ∑ PUFA 3 | 45.0 ± 1.7 ab | 45.3 ± 1.6 ab | 47.4 ± 0.7 ab | 47.1 ± 1.6 ab | 46.2 ± 4.1 ab | 44.2 ± 2.4 b | 48.7 ± 0.6 a | 47.1 ± 1.5 ab | 47.1 ± 3.4 ab |
| EPA + DHA (mg/g) | 0.92 ± 0.07 d | 1.07 ± 0.10 cd | 1.08 ± 0.17 bcd | 1.31 ± 0.22 abc | 0.94 ± 0.12 d | 1.07 ± 0.07 cd | 1.36 ± 0.21 ab | 1.41 ± 0.20 a | 1.13 ± 0.11 abcd |
| ∑ (n−3)/∑ (n−6) | 6.6 ± 1.0 | 4.4 ± 0.4 | 4.7 ± 1.6 | 4.8 ± 0.6 | 5.5 ± 2.3 | 6.2 ± 1.7 | 5.0 ± 1.6 | 4.9 ± 0.5 | 5.4 ± 2.0 |
| ∑ PUFA 3/∑ SFA1 | 2.5 ± 0.1 | 2.4 ± 0.2 | 2.6 ± 0.2 | 2.6 ± 0.2 | 2.5 ± 0.5 | 2.3 ± 0.1 | 2.7 ± 0.2 | 2.6 ± 0.1 | 2.6 ± 0.3 |
| PI | 3.1 ± 0.3 | 3.0 ± 0.4 | 3.2 ± 0.2 | 3.0 ± 0.2 | 3.1 ± 0.2 | 2.9 ± 0.2 | 3.3 ± 0.2 | 3.0 ± 0.1 | 3.2 ± 0.3 |
| Sea Salt Concentration (w/v) in the Processing Medium | ||
|---|---|---|
| 1% | 5% | |
| Raw | 1.10 ± 0.09 * | 1.10 ± 0.09 * |
| Cooked | 1.08 ± 0.07 ef* | 1.22 ± 0.06 cd |
| HPP300/2 | 1.02 ± 0.07 f* | 1.31 ± 0.08 c |
| HPP300/4 | 1.08 ± 0.07 ef* | 1.49 ± 0.07 b |
| HPP500/2 | 1.15 ± 0.06 de* | 1.71 ± 0.10 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lian, F.; De Conto, E.; Del Grippo, V.; Harrison, S.M.; Fagan, J.; Lyng, J.G.; Brunton, N.P. High-Pressure Processing for the Production of Added-Value Claw Meat from Edible Crab (Cancer pagurus). Foods 2021, 10, 955. https://doi.org/10.3390/foods10050955
Lian F, De Conto E, Del Grippo V, Harrison SM, Fagan J, Lyng JG, Brunton NP. High-Pressure Processing for the Production of Added-Value Claw Meat from Edible Crab (Cancer pagurus). Foods. 2021; 10(5):955. https://doi.org/10.3390/foods10050955
Chicago/Turabian StyleLian, Federico, Enrico De Conto, Vincenzo Del Grippo, Sabine M. Harrison, John Fagan, James G. Lyng, and Nigel P. Brunton. 2021. "High-Pressure Processing for the Production of Added-Value Claw Meat from Edible Crab (Cancer pagurus)" Foods 10, no. 5: 955. https://doi.org/10.3390/foods10050955







