The Feasibility of Using Pulsed-Vacuum in Stimulating Calcium-Alginate Hydrogel Balls
Abstract
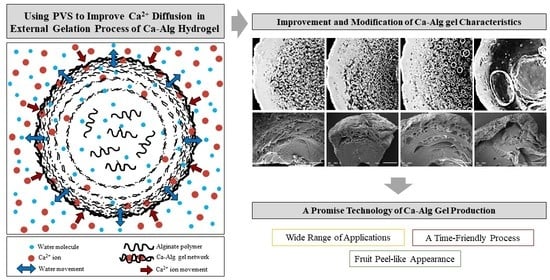
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Formation of Alginate Balls
2.3. Preparation of Calcium-Alginate Hydrogel Balls
2.4. Pulsed-Vacuum Stimulation Process
2.5. Determination of Weight Loss
2.6. Determination of Diffusion Coefficient of Calcium Ion
2.7. Volume Shrinkage Measurement
2.8. Textural Analysis
2.9. Image Acquisition
2.10. Characterization of Hydrogel Structure
2.11. Statistical Analysis
3. Results and Discussion
3.1. Diffusion Coefficients of Calcium Ion
3.2. Weight Reduction and Shrinkage
3.3. Textural Characteristics
3.4. Microstructural Characteristics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ramdhan, T.; Ching, S.H.; Prakash, S.; Bhandari, B. Physical and mechanical properties of alginate based composite gels. Trends Food Sci. Technol. 2020, 106, 150–159. [Google Scholar] [CrossRef]
- Chan, G.; Mooney, D.J. Ca2+ released from calcium alginate gels can promote inflammatory responses in vitro and in vivo. Acta Biomater. 2013, 9, 9281–9291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donati, I.; Paoletti, S. Material properties of alginates. In Alginates: Biology and Applications, 1st ed.; Rehm, B.H.A., Ed.; Springer: Berlin/Heidelberg, Germany, 2009; Volume 13, pp. 1–53. [Google Scholar]
- Fujiwara, Y.; Maeda, R.; Takeshita, H.; Komohara, Y. Alginates as food ingredients absorb extra salt in sodium chloride-treated mice. Heliyon 2021, 7, e06551. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Ke, C.-J.; Yen, K.-C.; Hsieh, H.-C.; Sun, J.-S.; Lin, F.-H. 3D porous calcium-alginate scaffolds cell culture system improved human osteoblast cell clusters for cell therapy. Theranostics 2015, 5, 643–655. [Google Scholar] [CrossRef] [Green Version]
- Huang, S.-L.; Lin, Y.-S. The Size Stability of Alginate Beads by Different ionic crosslinkers. Adv. Mater. Sci. Eng. 2017, 2017, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Jain, D.; Bar-Shalom, D. Alginate drug delivery systems: Application in context of pharmaceutical and biomedical research. Drug Dev. Ind. Pharm. 2014, 40, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ponce, B.; Urtuvia, V.; Maturana, N.; Peña, C.; Díaz-Barrera, A. Increases in alginate production and transcription levels of alginate lyase (alyA1) by control of the oxygen transfer rate in Azotobacter vinelandii cultures under diazotrophic conditions. Electron. J. Biotechnol. 2021, 52, 35–44. [Google Scholar] [CrossRef]
- Agulhon, P.; Robitzer, M.; David, L.; Quignard, F. Structural regime identification in ionotropic alginate gels: Influence of the cation nature and alginate structure. Biomacromolecules 2012, 13, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Lu, W.; Mata, A.; Nishinari, K.; Fang, Y. Egg-box model-based gelation of alginate and pectin: A review. Carbohydr. Polym. 2020, 242, 1–15. [Google Scholar] [CrossRef]
- Kuo, C.K.; Ma, P.X. Ionically crosslinked alginate hydrogels as scaffolds for tissue engineering: Part 1. Structure, gelation rate and mechanical properties. Biomaterials 2001, 22, 511–521. [Google Scholar] [CrossRef]
- Lozano-Vazquez, G.; Lobato-Calleros, C.; Escalona-Buendia, H.; Chavez, G.; Alvarez-Ramirez, J.; Vernon-Carter, E.J. Effect of the weight ratio of alginate-modified tapioca starch on the physicochemical properties and release kinetics of chlorogenic acid containing beads. Food Hydrocoll. 2015, 48, 301–311. [Google Scholar] [CrossRef]
- Soon-Shiong, P.; Heintz, R.E.; Merideth, N.; Yao, Q.X.; Yao, Z.; Zheng, T. Insulin independence in a type 1 diabetic patient after encapsulated islet transplantation. Lancet 1994, 343, 950–951. [Google Scholar] [CrossRef]
- Alsberg, E.; Anderson, K.W.; Albeiruti, A.; Franceschi, R.T.; Mooney, D.J. Cell interactive alginate hydrogels for bone tissue engineering. J. Dent. Res. 2001, 80, 2025–2029. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Shen, R.; Komasa, S.; Xue, Y.; Jin, B.; Hou, Y. Drug-loadable calcium alginate hydrogel system for use in oral bone. Int. J. Mol. Sci. 2017, 18, 989. [Google Scholar] [CrossRef] [PubMed]
- Le Goff, G.C.; Srinivas, R.L.; Hill, W.A.; Doyle, P.S. Hydrogel microparticles for biosensing. Eur. Polym. J. 2015, 72, 386–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Z.; Wang, T.; Yan, Q. Building a polysaccharide hydrogel capsule delivery system for control release of ibuprofen. J. Biomater. Sci. 2018, 29, 309–324. [Google Scholar] [CrossRef]
- Chan, L.W.; Lee, H.Y.; Heng, P.W.S. Mechanisms of external and internal gelation and their impact on the functions of alginate as a coat and delivery system. Carbohydr. Polym. 2006, 63, 176–187. [Google Scholar] [CrossRef]
- Lopez-Sanchez, P.; Fredriksson, N.; Larsson, A.; Altskär, A.; Ström, A. High sugar content impacts microstructure, mechanics, and release of calcium-alginate gels. Food Hydrocoll. 2018, 84, 26–33. [Google Scholar] [CrossRef]
- Liu, X.D.; Yu, W.Y.; Zhang, Y.; Xue, W.M.; Yu, W.T.; Xiong, Y. Characterization of structure and diffusion behavior of Ca alginate beads prepared with external or internal calcium sources. J. Microencapsul. 2002, 19, 775–782. [Google Scholar] [CrossRef]
- Choi, B.Y.; Park, H.J.; Hwang, S.J.; Park, J.B. Preparation of alginate beads for floating drug delivery system: Effects of CO2 gas-forming agents. Int. J. Pharm. 2002, 239, 81–91. [Google Scholar] [CrossRef]
- Vanderberg, G.W.; De La Noüe, J. Evaluation of protein release from chitosan-alginate microcapsules produced using external or internal gelation. J. Microencapsul. 2001, 18, 433–441. [Google Scholar] [CrossRef]
- Petit, B.; Masuda, K.; D’Souza, A.L.; Otten, L.; Pietryla, D.; Hartmann, D.J. Characterization of crosslinked collagens synthesized by mature articular chondrocytes cultured in alginate beads: Comparison of two distinct matrix compartments. Exp. Cell Res. 1996, 225, 151–161. [Google Scholar] [CrossRef]
- Bajpai, M.; Shukla, P.; Bajpai, S.K. Ca(II)+Ba(II) ions crosslinked alginate gels prepared by a novel diffusion through dialysis tube (DTDT) approach and preliminary BSA release study. Polym. Degrad. Stab. 2016, 134, 22–29. [Google Scholar] [CrossRef]
- Simonescu, C.M.; Mason, T.J.; Călinescu, I.; Lavric, V.; Vînătoru, M.; Melinescu, A.; Culiţă, D.C. Ultrasound assisted preparation of calcium alginate beads to improve absorption of Pb+2 from water. Ultrason. Sonochem. 2020, 68, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Servillo, L.; Balestrieri, M.L.; Giovane, A.; De Sio, F.; Cannavacciuolo, M.; Squitieri, G. Improving diced tomato firmness by pulsed vacuum calcification. LWT Food Sci. Technol. 2018, 92, 451–457. [Google Scholar] [CrossRef]
- Meiri, A.; Anderson, W.P. Observations on the exchange of salt between the xylem and neighbouring cells in zea mays primary roots. J. Exp. Bot. 1970, 21, 908–914. [Google Scholar] [CrossRef]
- Betoret, E.; Betoret, N.; Rocculi, P.; Rosa, M.D. Strategies to improve food functionality: Structure-property relationships on high pressures homogenization, vacuum impregnation and drying technologies. Trends Food Sci. Technol. 2015, 46, 1–12. [Google Scholar] [CrossRef]
- Corrêa, J.L.G.; Ernesto, D.B.; de Mendonça, K.S. Pulsed vacuum osmotic dehydration of tomatoes: Sodium incorporation reduction and kinetics modeling. LWT Food Sci. Technol. 2016, 71, 17–24. [Google Scholar] [CrossRef]
- De Jesus Junqueira, J.R.; Corrêa, J.L.G.; de Mendonça, K.S.; Resende, N.S.; de Barros Vilas Boas, E.V. Influence of sodium replacement and vacuum pulse on the osmotic dehydration of eggplant slices. Innov. Food Sci. Emerg. Technol. 2017, 41, 10–18. [Google Scholar] [CrossRef]
- Moreno, J.; Echeverria, J.; Silva, A.; Escudero, A.; Petzold, G.; Mella, K.; Escudero, C. Apple snack enriched with L-arginine using vacuum impregnation/ohmic heating technology. Food Sci. Technol. Int. 2017, 23, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Radziejewska-Kubzdela, E.; Biegańska-Marecik, R.; Kidoń, M. Applicability of vacuum impregnation to modify physico-chemical, sensory and nutritive characteristics of plant origin products-A review. Int. J. Mol. Sci. 2014, 15, 16577–16610. [Google Scholar] [CrossRef] [Green Version]
- Chrastil, J. Gelation of calcium alginate. Influence of rice starch or rice flour on the gelation kinetics and on the final gel structure. J. Agric. Food Chem. 1991, 39, 874–876. [Google Scholar] [CrossRef]
- Mohsenin, N.N. Physical Properties of Plant and Animal Materials: Structure, Physical Characteristics and Mechanical Properties, 1st ed.; Routledge, Gordon and Breach: New York, NY, USA, 1970; p. 278. [Google Scholar]
- Diao, X.Q.; Guan, H.N.; Zhao, X.; Diao, X.P.; Kong, B.H. Physicochemical and structural properties of composite gels prepared with myofibrillar protein and lard diacylglycerols. Meat Sci. 2016, 121, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ma, J.; Chen, S.; He, J.; Huang, Y. Characterization of calcium alginate/deacetylated konjac glucomannan blend films prepared by Ca2+ crosslinking and deacetylation. Food Hydrocoll. 2018, 82, 363–369. [Google Scholar] [CrossRef]
- Hajikhani, A.; Wriggers, P.; Marino, M. Chemo-mechanical modelling of swelling and crosslinking reaction kinetics in alginate hydrogels: A novel theory and its numerical implementation. J. Mech. Phys. Solids 2021, 153, 104476. [Google Scholar] [CrossRef]
- Favre, E.; Leonard, M.; Laurent, A.; Dellacherie, E. Diffusion of polyethyleneglycols in calcium alginate hydrogels. Colloids Surf. A Physicochem. Eng. Asp. 2001, 194, 197–206. [Google Scholar] [CrossRef]
- Lee, B.-B.; Bhandari, B.R.; Howes, T. Gelation of an alginate film via spraying of calcium chloride droplets. Chem. Eng. Sci. 2018, 183, 1–12. [Google Scholar] [CrossRef]
- Lupo, B.; Maestro, A.; Gutiérrez, J.M.; González, C. Characterization of alginate beads with encapsulated cocoa extract to prepare functional food: Comparison of two gelation mechanisms. Food Hydrocoll. 2015, 49, 25–34. [Google Scholar] [CrossRef]
- Escriche, I.; García-Pinchi, R.; Andrés, A.; Fito, P. Osmotic dehydration of kiwi fruit (Actinidia chinensis): Fluxes and mass transfer kinetics. J. Food Process. Eng. 2000, 23, 191–205. [Google Scholar] [CrossRef]
- Fito, P.; Chiralt, A.; Barat, J.M.; Andrés, A.; Martínez-Monzó, J.; Martínez-Navarrete, N. Vacuum impregnation for development of new dehydrated products. J. Food Eng. 2001, 49, 297–302. [Google Scholar] [CrossRef]
- Bienaimé, C.; Barbotin, J.-N.; Nava-Saucedo, J.-E. How to build an adapted and bioactive cell microenvironment? A chemical interaction study of the structure of Ca-alginate matrices and their repercussion on confined cells. J. Biomed. Mater. Res. Part A 2003, 67, 376–388. [Google Scholar] [CrossRef]
- Rezende, R.A.; Bártolo, P.J.; Mendes, A.; Filho, R.M. Experimental Characterisation of the Alginate Gelation Process for Rapid Prototyping. In Proceedings of the 8th International Conference on Chemical and Process Engineering, Ischia, Italy, 24–27 June 2007. [Google Scholar]
- Fito, P.; Andrés, A.; Chiralt, A.; Pardo, P. Coupling of hydrodynamic mechanism and deformation-relaxation phenomena during vacuum treatments in solid porous food-liquid systems. J. Food Eng. 1996, 27, 229–240. [Google Scholar] [CrossRef]
- Jeong, C.; Kim, S.; Lee, C.; Cho, S.; Kim, S.-B. Changes in the physical properties of calcium alginate gel beads under a wide range of gelation temperature conditions. Foods 2020, 9, 180. [Google Scholar] [CrossRef] [Green Version]
- Moreno, J.; Simpson, R.; Estrada, D.; Lorenzen, S.; Moraga, D.; Almonacid, S. Effect of pulsed-vacuum and ohmic heating on the osmodehydration kinetics, physical properties and microstructure of apples (cv. Granny Smith). Innov. Food Sci. Emerg. Technol. 2011, 12, 562–568. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, Y. Effects of pulsed-vacuum and ultrasound on the osmodehydration kinetics and microstructure of apples (Fuji). J. Food Eng. 2008, 85, 84–93. [Google Scholar] [CrossRef]
- Nieto, A.B.; Vicente, S.; Hodara, K.; Castro, M.A.; Alzamora, S.M. Osmotic dehydration of apple: Influence of sugar and water activity on tissue structure, rheological properties and water mobility. J. Food Eng. 2013, 119, 104–114. [Google Scholar] [CrossRef]
- Lin, X.; Luo, C.; Chen, Y. Effects of vacuum impregnation with sucrose solution on mango tissue. J. Food Sci. 2016, 81, 1412–1418. [Google Scholar] [CrossRef]
- Puguan, J.M.C.; Yu, X.; Kim, H. Characterization of structure, physico-chemical properties and diffusion behavior of Ca-Alginate gel beads prepared by different gelation methods. J. Colloid Interface Sci. 2014, 432, 109–116. [Google Scholar] [CrossRef]





| Treatment | Pressure (kPa) | Time (Cycle 1) | D (×10−4 cm2/s) | R2 2 | Rate of WR (mg/s) | Rate of Sv (mm3/s) |
|---|---|---|---|---|---|---|
| Control | 101 | 1 | 3.47 ± 0.38 cA | 0.993 | 0.43 ± 0.09 bAB | 0.37 ± 0.08 bA |
| 2 | 0.51 ± 0.39 bA | 0.977 | 0.36 ± 0.06 bB | 0.32 ± 0.03 bB | ||
| 3 | 0.26 ± 0.44 aB | 0.970 | 0.16 ± 0.07 aA | 0.12 ± 0.06 aB | ||
| PVS | 61 | 1 | 9.14 ± 1.73 cC | 0.980 | 0.48 ± 0.06 bAB | 0.38 ± 0.03 bA |
| 2 | 0.83 ± 0.50 bB | 0.990 | 0.20 ± 0.07 aA | 0.31 ± 0.01 bB | ||
| 3 | 0.49 ± 0.82 aC | 0.957 | 0.07 ± 0.12 aA | 0.00 ± 0.06 aA | ||
| 35 | 1 | 5.17 ± 1.15 cB | 0.991 | 0.38 ± 0.05 bA | 0.31 ± 0.03 bA | |
| 2 | 1.86 ± 0.87 bC | 0.984 | 0.29 ± 0.04 bAB | 0.30 ± 0.04 bB | ||
| 3 | 0.03 ± 0.03 aA | 0.975 | 0.12 ± 0.08 aA | 0.08 ± 0.04 aB | ||
| 8 | 1 | 5.01 ± 0.95 cB | 0.964 | 0.54 ± 0.10 bB | 0.49 ± 0.05 cB | |
| 2 | 2.75 ± 1.47 bD | 0.952 | 0.18 ± 0.07 aA | 0.22 ± 0.04 aA | ||
| 3 | 0.00 ± 0.00 aA | 0.976 | 0.39 ± 0.11 bB | 0.37 ± 0.05 bC |
| Treatment | Pressure (kPa) | a | b | R2 |
|---|---|---|---|---|
| Control | 101 | 1.158 * | −3.192 | 0.988 |
| PVS | 61 | 0.861 * | 3.651 | 0.967 |
| 35 | 0.813 * | 5.993 | 0.914 | |
| 8 | 0.851 * | 5.024 | 0.975 |
| Treatment | Pressure (kPa) | Hardness (N) | Breaking Deformation (%) | Young’s Modulus (kPa) | Rupture Strength (N) |
|---|---|---|---|---|---|
| Control | 101 | 36.1 ± 2.3 a | 58.1 ± 0.3 a | 66.2 ± 2.7 a | 7.4 ± 0.6 ab |
| PVS | 61 | 51.0 ± 1.7 c | 63.0 ± 0.9 c | 95.0 ± 2.3 c | 7.0 ± 0.4 a |
| 35 | 42.2 ± 2.1 b | 60.3 ± 1.1 b | 69.6 ± 6.4 b | 7.1 ± 0.5 a | |
| 8 | 64.5 ± 5.5 d | 66.9 ± 0.6 d | 142.7 ± 7.9 d | 8.2 ± 0.2 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jinnoros, J.; Innawong, B.; Udomkun, P.; Parakulsuksatid, P.; Silva, J.L. The Feasibility of Using Pulsed-Vacuum in Stimulating Calcium-Alginate Hydrogel Balls. Foods 2021, 10, 1521. https://doi.org/10.3390/foods10071521
Jinnoros J, Innawong B, Udomkun P, Parakulsuksatid P, Silva JL. The Feasibility of Using Pulsed-Vacuum in Stimulating Calcium-Alginate Hydrogel Balls. Foods. 2021; 10(7):1521. https://doi.org/10.3390/foods10071521
Chicago/Turabian StyleJinnoros, Janjira, Bhundit Innawong, Patchimaporn Udomkun, Pramuk Parakulsuksatid, and Juan L. Silva. 2021. "The Feasibility of Using Pulsed-Vacuum in Stimulating Calcium-Alginate Hydrogel Balls" Foods 10, no. 7: 1521. https://doi.org/10.3390/foods10071521