Use of Oleogels to Replace Margarine in Steamed and Baked Buns
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ingredients
2.2. Oleogel Preparation
2.3. Bun Making
2.4. Analysis of the Crumb Structure
2.5. Specific Volume and Height
2.6. Texture Measurements
2.7. Sensory Analysis
2.8. In Vitro Digestion
2.9. Free Fatty Acid Release
2.10. Statistical Analysis
3. Results and Discussion
3.1. Analysis of the Crumb Structure
3.2. Specific Volume
3.3. Texture
3.4. Sensory Analysis
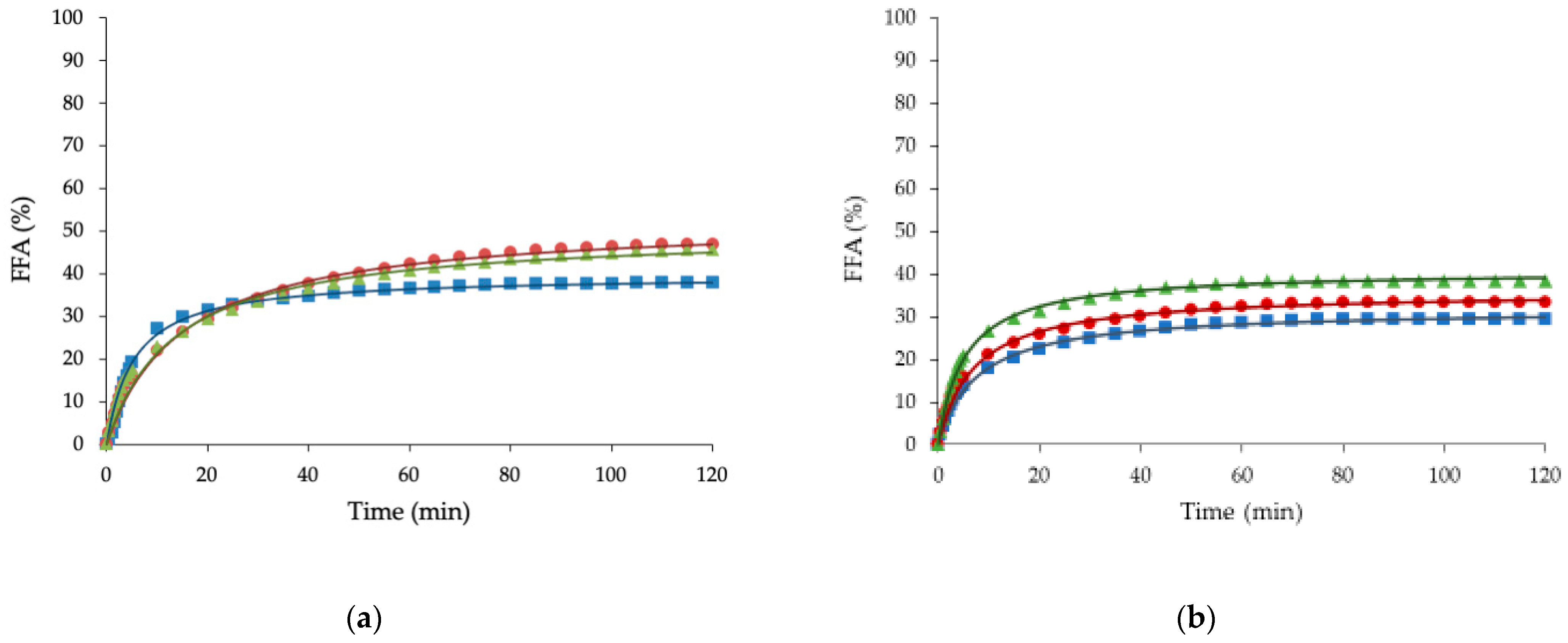
3.5. Free Fatty Acid Release
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Demirkesen, I.; Mert, I.D. Recent developments of oleogel utilizations in bakery products. Crit. Rev. Food Sci. Nutr. 2019, 60, 2460–2479. [Google Scholar] [CrossRef]
- Huang, S.; Miskelly, D. Steamed bread—A review of manufacturing, flour quality requirements, and quality evaluation. Cereal Chem. J. 2019, 96, 8–22. [Google Scholar] [CrossRef] [Green Version]
- Tsai, S.; Yang, J.; Tseng, Y.; Lee, C.; Mau, J.-L. Quality of Silver Ear Steamed Bun. J. Food Process. Preserv. 2010, 34, 649–663. [Google Scholar] [CrossRef]
- Agengo, F.B.; Onyango, A.N.; Serrem, C.A.; Okoth, J. Effect of Fortification with Snail Meat Powder on Physicochemical Properties and Shelf-life of Sorghum-wheat Buns. Curr. Nutr. Food Sci. 2020, 16, 749–756. [Google Scholar] [CrossRef]
- Graça, C.; Raymundo, A.; Sousa, I. Wheat Bread with Dairy Products—Technology, Nutritional, and Sensory Properties. Appl. Sci. 2019, 9, 4101. [Google Scholar] [CrossRef] [Green Version]
- Kenny, S.; Wehrle, K.; Stanton, C.; Arendt, E.K. Incorporation of dairy ingredients into wheat bread: Effects on dough rheology and bread quality. Eur. Food Res. Technol. 2000, 210, 391–396. [Google Scholar] [CrossRef]
- Shon, J.; Yun, Y.; Shin, M.; Chin, K.B.; Eun, J.-B. Effects of milk proteins and gums on quality of bread made from frozen dough. J. Sci. Food Agric. 2009, 89, 1407–1415. [Google Scholar] [CrossRef]
- Nurshahbani, S.; Azrina, A. Trans Fatty Acids in Selected Bakery Products and Its Potential Dietary Exposure. Int. Food Res. J. 2014, 21, 2175–2181. [Google Scholar]
- Van Erp-baart, M.A.; Couet, C.; Cuadrado, C.; Kafatos, A.; Stanley, J.; van Poppel, G. Trans Fatty Acids in Bakery Products from 14 European Countries: The TRANSFAIR Study. J. Food Compos. Anal. 1998, 169, 161–169. [Google Scholar] [CrossRef]
- Gebauer, S.K.; Psota, T.L.; Kris-Etherton, P.M. The Diversity of Health Effects of Individual trans Fatty Acid Isomers. Lipids 2007, 42, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Morenga, L.T.; Montez, J.M. Health effects of saturated and trans-fatty acid intake in children and adolescents: Systematic review and meta-analysis. PLoS ONE 2017, 12, e0186672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pareyt, B.; Finnie, S.M.; Putseys, J.A.; Delcour, J. Lipids in bread making: Sources, interactions, and impact on bread quality. J. Cereal Sci. 2011, 54, 266–279. [Google Scholar] [CrossRef]
- Patel, A.R.; Nicholson, R.A.; Marangoni, A.G. Applications of fat mimetics for the replacement of saturated and hydrogenated fat in food products. Curr. Opin. Food Sci. 2020, 33, 61–68. [Google Scholar] [CrossRef]
- Mensink, R.P.; Zock, P.; Kester, A.D.M.; Katan, M.B. Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: A meta-analysis of 60 controlled trials. Am. J. Clin. Nutr. 2003, 77, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Diet, Nutrition, and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; World Health Organization Technical Report Series; World Health Organization: Geneva, Switzerland, 2003; p. 916. [Google Scholar]
- Martins, A.J.; Vicente, A.A.; Pastrana, L.; Cerqueira, M. Oleogels for development of health-promoting food products. Food Sci. Hum. Wellness 2020, 9, 31–39. [Google Scholar] [CrossRef]
- Oh, I.K.; Lee, S. Utilization of foam structured hydroxypropyl methylcellulose for oleogels and their application as a solid fat replacer in muffins. Food Hydrocoll. 2018, 77, 796–802. [Google Scholar] [CrossRef]
- Patel, A.R.; Cludts, N.; Bin Sintang, M.D.; Lesaffer, A.; Dewettinck, K. Edible oleogels based on water soluble food polymers: Preparation, characterization and potential application. Food Funct. 2014, 5, 2833–2841. [Google Scholar] [CrossRef] [Green Version]
- Bascuas, S.; Hernando, I.; Moraga, G.; Quiles, A. Structure and stability of edible oleogels prepared with different unsaturated oils and hydrocolloids. Int. J. Food Sci. Technol. 2019, 55, 1458–1467. [Google Scholar] [CrossRef]
- Bascuas, S.; Salvador, A.; Hernando, I.; Quiles, A. Designing Hydrocolloid-Based Oleogels with High Physical, Chemical, and Structural Stability. Front. Sustain. Food Syst. 2020, 4, 111. [Google Scholar] [CrossRef]
- AACC. I.M. 10–05. 01 Approved Methods of Analysis, 10th ed.; Method 10-05.01. Guidelines for Measurement of Volume by Rapeseed Displacement; Cereals & Grains Association: St. Paul, MN, USA, 2003; pp. 1–10. [Google Scholar]
- UNE-EN ISO 4120. Análisis Sensorial. Metodología. Prueba Triangular (ISO 4120: 2004); Asociación Española de Normalización y Certificación (AENOR): Madrid, Spain, 2004. [Google Scholar]
- Diez-Sánchez, E.; Quiles, A.; Hernando, I. Interactions between Blackcurrant Polyphenols and Food Macronutrients in Model Systems: In Vitro Digestion Studies. Foods 2021, 10, 847. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Mascaraque, L.G.; Perez-Masiá, R.; Gonzalez-Barrio, R.; Periago, M.J.; López-Rubio, A. Potential of microencapsulation through emulsion-electrospraying to improve the bioaccesibility of β-carotene. Food Hydrocoll. 2017, 73, 1–12. [Google Scholar] [CrossRef]
- Li, Y.; Hu, M.; McClements, D.J. Factors affecting lipase digestibility of emulsified lipids using an in vitro digestion model: Proposal for a standardised pH-stat method. Food Chem. 2011, 126, 498–505. [Google Scholar] [CrossRef]
- Bellesi, F.A.; Martinez, M.J.; Ruiz-Henestrosa, V.M.P.; Pilosof, A.M. Comparative behavior of protein or polysaccharide stabilized emulsion under in vitro gastrointestinal conditions. Food Hydrocoll. 2016, 52, 47–56. [Google Scholar] [CrossRef]
- Garcia, J.R.; Puig, A.; Salvador, A.; Hernando, I. Optimization of a Sponge Cake Formulation with Inulin as Fat Replacer: Structure, Physicochemical, and Sensory Properties. J. Food Sci. 2012, 77, C189–C197. [Google Scholar] [CrossRef]
- Monteiro, J.; Farage, P.; Zandonadi, R.; Botelho, R.; de Oliveira, L.; Raposo, A.; Shakeel, F.; Alshehri, S.; Mahdi, W.; Araújo, W. A Systematic Review on Gluten-Free Bread Formulations Using Specific Volume as a Quality Indicator. Foods 2021, 10, 614. [Google Scholar] [CrossRef]
- Demirkesen, I.; Mert, B. Utilization of Beeswax Oleogel-Shortening Mixtures in Gluten-Free Bakery Products. J. Am. Oil Chem. Soc. 2019, 96, 545–554. [Google Scholar] [CrossRef]
- Liu, X.; Mu, T.; Sun, H.; Zhang, M.; Chen, J.; Fauconnier, M.L. Influence of different hydrocolloids on dough thermo-mechanical properties and in vitro starch digestibility of gluten-free steamed bread based on potato flour. Food Chem. 2018, 239, 1064–1074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohammadi, M.; Sadeghnia, N.; Azizi, M.H.; Neyestani, T.R.; Mortazavian, A. Development of gluten-free flat bread using hydrocolloids: Xanthan and CMC. J. Ind. Eng. Chem. 2014, 20, 1812–1818. [Google Scholar] [CrossRef]
- Jung, D.; Oh, I.; Lee, J.; Lee, S. Utilization of butter and oleogel blends in sweet pan bread for saturated fat reduction: Dough rheology and baking performance. LWT 2020, 125, 109194. [Google Scholar] [CrossRef]
- Diez-Sánchez, E.; Llorca, E.; Tarrega, A.; Fiszman, S.; Hernando, I. Changing chemical leavening to improve the structural, textural and sensory properties of functional cakes with blackcurrant pomace. LWT 2020, 127, 109378. [Google Scholar] [CrossRef]
- Rodriguez-Garcia, J.; Puig, A.; Salvador, A.; Hernando, I. Funcionality of several cake ingredients: A comprehensive approach. Czech J. Food Sci. 2013, 31, 355–360. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Cervera, S.; Salvador, A.; Sanz, T. Cellulose ether emulsions as fat replacers in muffins: Rheological, thermal and textural properties. LWT 2015, 63, 1083–1090. [Google Scholar] [CrossRef]
- Rathnayake, H.A.; Navaratne, S.B.; Navaratne, C.M. Porous Crumb Structure of Leavened Baked Products. Int. J. Food Sci. 2018, 2018, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Barbieri, S.; Bendini, A.; Balestra, F.; Palagano, R.; Rocculi, P.; Toschi, T.G. Sensory and instrumental study of Taralli, a typical Italian bakery product. Eur. Food Res. Technol. 2017, 244, 73–82. [Google Scholar] [CrossRef]
- Torcello-Gómez, A.; Foster, T.J. Influence of interfacial and bulk properties of cellulose ethers on lipolysis of oil-in-water emulsions. Carbohydr. Polym. 2016, 144, 495–503. [Google Scholar] [CrossRef]
- Ruiz-Henestrosa, V.M.P.; Bellesi, F.A.; Camino, N.A.; Pilosof, A.M. The impact of HPMC structure in the modulation of in vitro lipolysis: The role of bile salts. Food Hydrocoll. 2017, 62, 251–261. [Google Scholar] [CrossRef]
- Bellesi, F.A.; Ruiz-Henestrosa, V.M.P.; Maldonado-Valderrama, J.; Santaella, T.D.C.; Pilosof, A.M. Comparative interfacial in vitro digestion of protein and polysaccharide oil/water films. Colloids Surf. B Biointerfaces 2018, 161, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Borreani, J.; Leonardi, C.; Moraga, G.; Quiles, A.; Hernando, I. How do Different Types of Emulsifiers/Stabilizers Affect the In Vitro Intestinal Digestion of O/W Emulsions? Food Biophys. 2019, 14, 313–325. [Google Scholar] [CrossRef]
- Devraj, R.; Williams, H.D.; Warren, D.B.; Müllertz, A.; Porter, C.; Pouton, C. In vitro digestion testing of lipid-based delivery systems: Calcium ions combine with fatty acids liberated from triglyceride rich lipid solutions to form soaps and reduce the solubilization capacity of colloidal digestion products. Int. J. Pharm. 2013, 441, 323–333. [Google Scholar] [CrossRef]
- Torcello-Gómez, A.; Boudard, C.; Mackie, A.R. Calcium Alters the Interfacial Organization of Hydrolyzed Lipids during Intestinal Digestion. Langmuir 2018, 34, 7536–7544. [Google Scholar] [CrossRef] [PubMed]
- Ashkar, A.; Laufer, S.; Rosen-Kligvasser, J.; Lesmes, U.; Davidovich-Pinhas, M. Impact of different oil gelators and oleogelation mechanisms on digestive lipolysis of canola oil oleogels. Food Hydrocoll. 2019, 97, 105218. [Google Scholar] [CrossRef]

| Parameters | BC | BO | BS | |
|---|---|---|---|---|
| Crumb Cell Structure | Cell density Cell area (mm2) Cell circularity Total cell area (%) | 1235 a (113) | 1045 a (140) | 1064 a (100) |
| 0.27 a (0.06) 0.85 b (0.02) 45 b (3) | 0.33 a (0.06) 0.83 a,b (0.01) 39.2 b (0.4) | 0.30 a (0.01) 0.82 a (0.01) 36.6 a (0.7) | ||
| Specific volume (cm3/g) Height (cm) | 2.7 a (0.1) | 2.54 a (0.09) | 2.7 a (0.02) | |
| 4.75 a (0.03) | 4.7 a (0.1) | 4.70 a (0.06) | ||
| Texture | Hardness (n) Springiness Cohesiveness Chewiness (n) | 0.9 a (0.1) | 1.2 b (0.2) | 1.1 b (0.3) |
| 0.92 b (0.02) | 0.90 a,b (0.03) | 0.8 a (0.2) | ||
| 0.74 a (0.02) | 0.73 a (0.02) | 0.74 a (0.06) | ||
| 0.64 a (0.09) | 0.8 b (0.1) | 0.7 a,b (0.2) | ||
| Parameters | SC | SO | SS | |
|---|---|---|---|---|
| Crumb Cell Structure | Cell density Cell area (mm2) Cell circularity Total cell area (%) | 1329 a (87) | 1101 a (161) | 1137 a (105) |
| 0.27 a (0.06) 0.85 a (0.01) 44 a (4) | 0.27 a (0.06) 0.82 a (0.02) 37 a (6) | 0.30 a (0.01) 0.84 a (0.01) 43 a (2) | ||
| Specific volume (cm3/g) Height (cm) | 2.50 a (0.05) | 2.7 a (0.1) | 2.6 a (0.2) | |
| 3.9 a (0.3) | 4.0 a (0.1) | 3.8 a (0.01) | ||
| Texture | Hardness (n) Springiness Cohesiveness Chewiness (n) | 0.7 a (0.1) | 0.75 a (0.08) | 0.7 a (0.1) |
| 0.90 a (0.03) | 0.91 a (0.03) | 0.9 a (0.1) | ||
| 0.72 a (0.02) | 0.72 a (0.03) | 0.74 a (0.05) | ||
| 0.48 a (0.08) | 0.49 a (0.05) | 0.5 a (0.1) | ||
| FFAmax (%) | K (1/min) | R2 | |
|---|---|---|---|
| BC | 38 a (2) | 6.5 b (1.3) | 0.99 |
| BO | 48 a (3) | 4.2 a,b (0.7) | 0.99 |
| BS | 45 a (7) | 3.7 a (0.1) | 0.98 |
| FFAmax (%) | K (1/min) | R2 | |
|---|---|---|---|
| SC | 31 a (8) | 3.8 b (0.6) | 0.99 |
| SO | 35.7 a (0.4) | 5.5 a,b (0.6) | 0.99 |
| SS | 39 a (4) | 6.8 b (1.4) | 0.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bascuas, S.; Morell, P.; Quiles, A.; Salvador, A.; Hernando, I. Use of Oleogels to Replace Margarine in Steamed and Baked Buns. Foods 2021, 10, 1781. https://doi.org/10.3390/foods10081781
Bascuas S, Morell P, Quiles A, Salvador A, Hernando I. Use of Oleogels to Replace Margarine in Steamed and Baked Buns. Foods. 2021; 10(8):1781. https://doi.org/10.3390/foods10081781
Chicago/Turabian StyleBascuas, Santiago, Pere Morell, Amparo Quiles, Ana Salvador, and Isabel Hernando. 2021. "Use of Oleogels to Replace Margarine in Steamed and Baked Buns" Foods 10, no. 8: 1781. https://doi.org/10.3390/foods10081781
APA StyleBascuas, S., Morell, P., Quiles, A., Salvador, A., & Hernando, I. (2021). Use of Oleogels to Replace Margarine in Steamed and Baked Buns. Foods, 10(8), 1781. https://doi.org/10.3390/foods10081781







