Pesticide Residue Behavior and Risk Assessment in Celery after Se Nanoparticles Application
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Field Trials and Sample Collection
2.3. Method Validation
2.4. Extraction and Purification
2.5. LC-MS/MS Conditions
2.6. GC-MS/MS Conditions
2.7. Dissipation Kinetics of Pesticides
2.8. Risk Assessment
3. Results and Discussion
3.1. Method Validation
3.2. Residue Levels in Celery
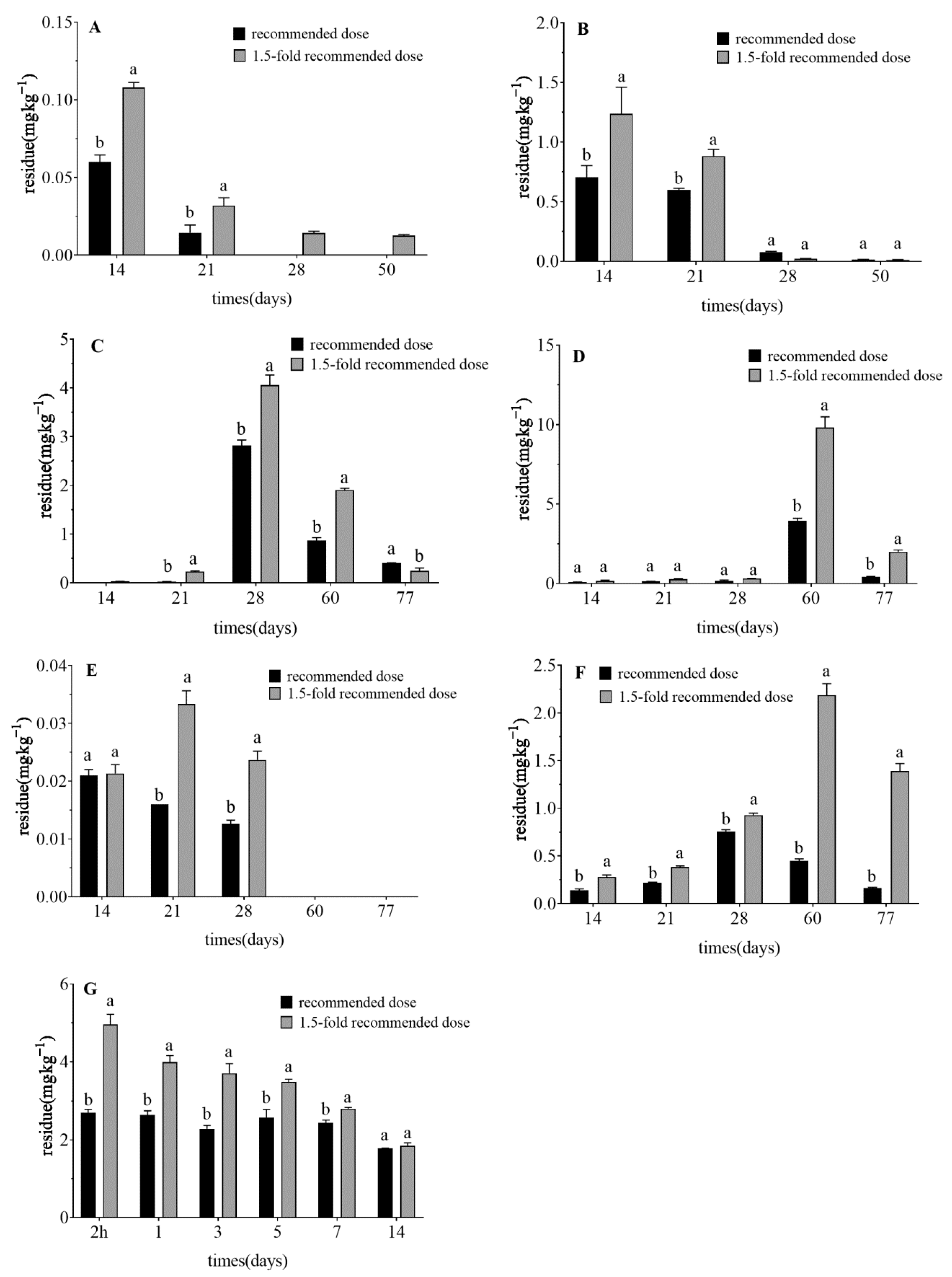
3.3. Dissipation Dynamics of Pesticide Residues in Celery
3.4. Dissipation Dynamics of Five Pesticide Residues in Celery by Se Nanoparticles
3.5. Dietary Intake Risk Assessment for Celery
3.5.1. Chronic Dietary Exposure Risk Assessment for Celery
3.5.2. Acute Dietary Exposure Risk Assessment for Celery
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, M.Y.; Hou, X.L.; Wang, F.; Tan, G.F.; Xu, Z.S.; Xiong, A.S. Advances in the research of celery, an important Apiaceae vegetable crop. Crit. Rev. Biotechnol. 2018, 38, 172–183. [Google Scholar] [CrossRef]
- Kooti, W.; Daraei, N. A Review of the Antioxidant Activity of Celery (Apium graveolens L.). J. Evid. Based Complement. Altern. Med. 2017, 22, 1029–1034. [Google Scholar] [CrossRef] [Green Version]
- Turner, L.; Lignou, S.; Gawthrop, F.; Wagstaff, C. Investigating the factors that influence the aroma profile of Apium graveolens: A review. Food Chem. 2021, 345, 128673. [Google Scholar] [CrossRef]
- Aldemir, O.; Zcan, F.Z.; Karabulut, B. Flavones (Apigenin, Luteolin, Crhysin) and Their Importance for Health. Mellifera 2020, 20, 16–27. [Google Scholar]
- Liu, J.; Feng, K.; Hou, X.; Li, H.; Wang, G.; Xu, Z.; Xiong, A. Transcriptome profiling reveals the association of multiple genes and pathways contributing to hormonal control in celery leaves. Acta Biochim. Biophys. Sin. 2019, 51, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Li, G.; Lin, Q.; Wu, X.; Wang, J. Residual dynamics and dietary exposure risk of dimethoate and its metabolite in greenhouse celery. PeerJ 2021, 9, 10789. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.M.; Zhao, Y.; Yang, M.O.; Yang, Z.H.; Li, J.H. The enantioselective effects and potential risks of paclobutrazol residue during cucumber pickling process. J. Hazard. Mater. 2020, 386, 121882. [Google Scholar] [CrossRef]
- Kermani, M.; Dowlati, M.; Gholami, M.; Sobhi, H.R.; Azari, A.; Esrafili, A.; Yeganeh, M.; Ghaffari, H.R. A global systematic review, meta-analysis and health risk assessment on the quantity of Malathion, Diazinon and Chlorpyrifos in Vegetables. Chemosphere 2021, 270, 129382. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.L.; He, W.; Li, Y.L.; Li, Y.Y.; Qin, Y.F.; Meng, F.Q.; Wang, L.G.; Xu, F.L. Residual concentrations and ecological risks of neonicotinoid insecticides in the soils of tomato and cucumber greenhouses in Shouguang, Shandong Province, East China. Sci. Total Environ. 2020, 738, 140248. [Google Scholar] [CrossRef] [PubMed]
- Deka, A.C.; Sinha, S.K. Mycogenic Silver Nanoparticle Biosynthesis and Its Pesticide Degradation Potentials. Int. J. Technol. Enhanc. Emerg. Eng. Res. 2015, 3, 108–113. [Google Scholar]
- Farre, M.; Gajda-Schrantz, K.; Kantiani, L.; Barcelo, D. Ecotoxicity and analysis of nanomaterials in the aquatic environment. Anal. Bioanal. Chem. 2009, 393, 81–95. [Google Scholar] [CrossRef]
- El-Ramady, H.R.; Domokos-Szabolcsy, É.; Abdalla, N.A.; Alshaal, T.A.; Shalaby, T.A.; Sztrik, A.; Prokisch, J.; Fári, M. Selenium and nano-selenium in agroecosystems. Environ. Chem. Lett. 2014, 12, 495–510. [Google Scholar] [CrossRef]
- Wang, C.; Cheng, T.; Liu, H.; Zhou, F.; Zhang, J.; Zhang, M.; Liu, X.; Shi, W.; Cao, T. Nano-selenium controlled cadmium accumulation and improved photosynthesis in Indica rice cultivated in lead and cadmium combined paddy soils. J. Environ. Sci. 2021, 103, 336–346. [Google Scholar] [CrossRef]
- de Brito Mateus, M.P.; Tavanti RF, R.; Tavanti, T.R.; Santos, E.F.; Jalal, A.; Dos Reis, A.R. Selenium biofortification enhances ROS scavenge system increasing yield of coffee plants. Ecotoxicol. Environ. Saf. 2021, 209, 111772. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhou, C.; Zhang, J.; An, Q.; Wu, Y.; Li, J.Q.; Pan, C.P. Nanoselenium Foliar Applications Enhance the Nutrient Quality of Pepper by Activating the Capsaicinoid Synthetic Pathway. J. Agric. Food Chem. 2020, 68, 9888–9895. [Google Scholar] [CrossRef]
- Liu, N.; Li, J.; Lv, J.; Yu, J.; Xie, J.; Wu, Y.; Tang, Z. Melatonin alleviates imidacloprid phytotoxicity to cucumber (Cucumis sativus L.) through modulating redox homeostasis in plants and promoting its metabolism by enhancing glutathione dependent detoxification. Ecotoxicol. Environ. Saf. 2021, 217, 112248. [Google Scholar] [CrossRef]
- Liu, T.; Li, T.; Zhang, L.; Li, H.; Liu, S.; Yang, S.; An, Q.; Pan, C.; Zou, N. Exogenous salicylic acid alleviates the accumulation of pesticides and mitigates pesticide-induced oxidative stress in cucumber plants (Cucumis sativus L.). Ecotoxicol. Environ. Saf. 2021, 208, 111654. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Yuan, C.; Gao, Y.; Luo, J.; Yang, S.; Liu, S.; Zhang, R.; Zou, N. Exogenous salicylic acid mitigates the accumulation of some pesticides in cucumber seedlings under different cultivation methods. Ecotoxicol. Environ. Saf. 2020, 198, 110680. [Google Scholar] [CrossRef] [PubMed]
- Ordaz-Silva, S.; López-Sánchez, I.V.; Soto-Hernández, M.; Chacón-Hernández, J.C.; Gaona-García, G.; Mora-Ravelo, S.G.; Delgadillo-Ángeles, J.L.; Merino-González, R. Spatial Distribution of Listroderes costirostris and Hypera postica (Curculionidae: Cyclominae, Hyperinae) on a Celery Crop in Mexico’s Northwest Region. Fla. Entomol. 2020, 103, 397–400. [Google Scholar] [CrossRef]
- Zheng, H.; Xie, W.; Fu, B.; Xiao, S.; Tan, X.; Ji, Y.; Cheng, J.X.; Wang, R.; Liu, B.M.; Yang, X.; et al. Annual analysis of field-evolved insecticide resistance in Bemisia tabaci across China. Pest Manag. Sci. 2021, 77, 2990–3001. [Google Scholar] [CrossRef]
- Tao, Y.; Jia, C.; Jing, J.; Zhang, J.; Yu, P.; He, M.; Wu, J.; Chen, L.; Zhao, E. Occurrence and dietary risk assessment of 37 pesticides in wheat fields in the suburbs of Beijing, China. Food Chem. 2021, 350, 129245. [Google Scholar] [CrossRef]
- Cui, K.; Wu, X.; Wei, D.; Zhang, Y.; Cao, J.; Xu, J.; Dong, F.; Liu, X.; Zheng, Y. Health risks to dietary neonicotinoids are low for Chinese residents based on an analysis of 13 daily-consumed foods. Environ. Int. 2021, 149, 106385. [Google Scholar] [CrossRef]
- Riga, M.; Tsakireli, D.; Ilias, A.; Morou, E.; Myridakis, A.; Stephanou, E.G.; Nauen, R.; Dermauw, W.; Leeuwen, T.V.; Paine, M.; et al. Abamectin is metabolized by CYP392A16, a cytochrome P450 associated with high levels of acaricide resistance in Tetranychus urticae. Insect Biochem. Mol. Biol. 2014, 46, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Nie, J.; Wu, Y.; Yan, Z.; Ye, M. The effects of fruit bagging on residue behavior and dietary risk for four pesticides in apple. Sci. Rep. 2018, 8, 14348. [Google Scholar] [CrossRef] [Green Version]
- El-Gendy, K.S.; Radwan, M.A.; Gad, A.F.; Khamis, A.E.; Eshra, E.H. Use of multiple endpoints to investigate the ecotoxicological effects of abamectin and thiamethoxam on Theba pisana snails. Ecotoxicol. Environ. Saf. 2019, 167, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Jayakrishnan, S.; Dikshit, A.K.; Singh, J.P.; Pachauri, D.C. Dissipation of lambda-cyhalothrin on tomato (Lycopersicon esculentum Mill.) and removal of its residues by different washing processes and steaming. Bull. Environ. Contam. Toxicol. 2005, 75, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Stahnke, H.; Kittlaus, S.; Kempe, G.; Hemmerling, C.; Alder, L. The influence of electrospray ion source design on matrix effects. J. Mass Spectrom. 2012, 47, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Ming, X.; Cui, S.; Ma, X.; Zhang, Q.; Li, X. Determination of clothianidin residues in garlic by liquid chromatography-tandem mass spectrometry combined with QuEChERS. J. Food Agric. Environ. 2012, 10, 232–236. [Google Scholar]
- Chen, R.; Xue, X.; Wang, G.; Wang, J. Determination and dietary intake risk assessment of 14 pesticide residues in apples of China. Food Chem. 2021, 351, 129266. [Google Scholar] [CrossRef]
- Fan, X.; Zhao, S.; Hu, J. Dissipation behavior and dietary risk assessment of lambda-cyhalothrin, thiamethoxam and its metabolite clothianidin in apple after open field application. Regul. Toxicol. Pharmacol. 2019, 101, 135–141. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, X.; Li, L.; Chen, Y.; Wang, J.; Shen, Q. Development of a PRiME Cleanup Procedure for the Purification of Avermectins in Grass Carps and Liquid Chromatography-Tandem Mass Spectrometry Analysis. Food Anal. Methods 2019, 12, 1905–1913. [Google Scholar] [CrossRef]
- Li, X.; Chen, X.; Hu, J. Dissipation behavior, residue distribution and dietary risk assessment of chlorfenapyr and clothianidin in leek using RRLC-QqQ-MS/MS technique. Chin. Chem. Lett. 2019, 30, 107–110. [Google Scholar] [CrossRef]
- Ge, G.; Gao, W.; Yan, M.; Song, W.; Xiao, Y.; Zheng, P.; Peng, C.; Cai, H.; Hou, R. Comparation study on the metabolism destination of neonicotinoid and organophosphate insecticides in tea plant (Camellia sinensis L.). Food Chem. 2021, 344, 128579. [Google Scholar] [CrossRef]
- Fu, D.; Zhang, S.; Wang, M.; Liang, X.; Xie, Y.; Zhang, Y.; Zhang, C. Dissipation behavior, residue distribution and dietary risk assessment of cyromazine, acetamiprid and their mixture in cowpea and cowpea field soil. J. Sci. Food Agric. 2020, 100, 4540–4548. [Google Scholar] [CrossRef] [PubMed]
- Pang, N.; Fan, X.; Fantke, P.; Zhao, S.; Hu, J. Dynamics and dietary risk assessment of thiamethoxam in wheat, lettuce and tomato using field experiments and computational simulation. Environ. Pollut. 2020, 256, 113285. [Google Scholar] [CrossRef]
- EC—European Commission. Guidance Document on Analytical Quality Control and Method Validation Procedures for Pesticide Residues and Analysis in Food and Feed, SANTE/12682/2019; European Commission Directorate General for Health and Food Safety: Brussels, Belgium, 2019. [Google Scholar]
- Karlik, J.F. Calculation of pesticide residues on roses using california’s total reporting data and first order chemical kinetics. Acta Hortic. 2010, 870, 121–128. [Google Scholar] [CrossRef]
- Li, C.; Zhou, J.; Yue, N.; Wang, Y.; Wang, J.; Jin, F. Dissipation and dietary risk assessment of tristyrylphenol ethoxylate homologues in cucumber after field application. Food Chem. 2021, 338, 127988. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Zhao, S.; Fan, X.; Ma, Y.; Wu, X.; Su, Y.; Hu, J. Residue behavior and dietary risk assessment of chlorothalonil and its metabolite SDS-3701 in water spinach to propose maximum residue limit (MRL). Regul. Toxicol. Pharmacol. 2019, 107, 104416. [Google Scholar] [CrossRef]
- Bai, A.; Chen, A.; Chen, W.; Luo, X.; Liu, S.; Zhang, M.; Liu, Y.; Zhang, D. Study on degradation behavior, residue distribution, and dietary risk assessment of propiconazole in celery and onion under field application. J. Sci. Food Agric. 2021, 101, 1998–2005. [Google Scholar] [CrossRef]
- Soliman, M.; Khorshid, M.A.; El-Marsafy, A.M.; Abo-Aly, M.M.; Khedr, T. Determination of 10 pesticides, newly registered in Egypt, using modified QuEChERS method in combination with gas and liquid chromatography coupled with tandem mass spectrometric detection. Int. J. Environ. Anal. Chem. 2019, 99, 224–242. [Google Scholar] [CrossRef]
- China, National Food Safety Standard-Determination of 208 Pesticides and Metabolites Residues in Food of Plant Origin-Gas Chro-matography-Tandem Mass Spectrometry Method (GB 23200.113-2018), China Agriculture Press: Beijing, China, 2018.
- Chen, X.; Liu, X.; Dong, B.; Hu, J. Simultaneous determination of pyridaben, dinotefuran, DN and UF in eggplant ecosystem under open-field conditions: Dissipation behavior and residue distribution. Chemosphere 2018, 195, 245–251. [Google Scholar] [CrossRef]
- Wei, D.; Wu, X.; Ji, M.; Xu, J.; Dong, F.; Liu, X.; Zheng, Y. Carboxin and its major metabolites residues in peanuts: Levels, dietary intake and chronic intake risk assessment. Food Chem. 2019, 275, 169–175. [Google Scholar] [CrossRef]
- China, Guideline for the Testing of Pesticide Residues in Crops (NY/T 788-2018), China Agriculture Press: Beijing, China, 2018.
- Song, L.; Zhong, Z.; Han, Y.; Zheng, Q.; Qin, Y.; Wu, Q.; He, X.; Pan, C. Dissipation of sixteen pesticide residues from various applications of commercial formulations on strawberry and their risk assessment under greenhouse conditions. Ecotoxicol. Environ. Saf. 2020, 188, 109842. [Google Scholar] [CrossRef]
- Shalaby, A.A. Residual behavior of abamectin and cyflufenamid in squash plants. Ann. Agric. Sci. Moshtohor. 2016, 54, 955–960. [Google Scholar]
- Song, W.; Jia, C.; Jing, J.; Zhao, E.; He, M.; Chen, L.; Yu, P. Residue behavior and dietary intake risk assessment of carbosulfan and its metabolites in cucumber. Regul. Toxicol. Pharmacol. 2018, 95, 250–253. [Google Scholar] [CrossRef]
- Mahmoud, H.A.; Arief, M.; Nasr, I.N.; Mohammed, I.H. Residues and half-lives of abamectin, diniconazole and methomyl on and in strawberry under the normal field conditions. J. Appl. Ences Res. 2010, 6, 932–936. [Google Scholar]
- Mohapatra, S.; Siddamallaiah, L.; Matadha, N.Y. Behavior of acetamiprid, azoxystrobin, pyraclostrobin, and lambda-cyhalothrin in/on pomegranate tissues. Environ. Sci. Pollut. Res. 2021, 28, 27481–27492. [Google Scholar] [CrossRef] [PubMed]
- China, National Food Safety Standard-Maximum Residue Limits for Pesticides in Food (GB 2763-2019), China Agriculture Press: Beijing, China, 2019.
- European Food Safety Authority. Peer Review of the Pesticide Risk Assessment of the Active Substance Abamectin. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7439425/ (accessed on 20 August 2020).
- Kalefetoglu Macar, T. Investigation of cytotoxicity and genotoxicity of abamectin pesticide in Allium cepa L. Environ. Sci. Pollut. Res. Int. 2021, 28, 2391–2399. [Google Scholar] [CrossRef] [PubMed]

| Time (min) | Flow Velocity (mL/min) | Phase A: Methanol (%) | Phase B: Water (5 mmol/L Ammonium Acetate + 0.1% Formic Acid) (%) |
|---|---|---|---|
| 0.0 | 0.2 | 5 | 95 |
| 3.0 | 0.2 | 5 | 95 |
| 7.0 | 0.2 | 95 | 5 |
| 16.0 | 0.2 | 95 | 5 |
| 18.0 | 0.2 | 5 | 95 |
| 20.0 | 0.2 | 5 | 95 |
| Pesticide | Linear Range (mg·kg−1) | Correlation Coefficient (R2) | LOQ (mg·kg−1) | Recovery (%) ± RSDs (%), Spiked Level (mg·kg−1) | ||
|---|---|---|---|---|---|---|
| 0.01 | 0.05 | 0.1 | ||||
| Clothianidin | 0.01–5.0 | 0.9943 | 0.01 | 72.0 ± 1.7 | 80.0 ± 2.2 | 74.1 ± 3.0 |
| Imidacloprid | 0.01–5.0 | 0.9982 | 0.01 | 101.0 ± 4.8 | 101.2 ± 3.8 | 97.4 ± 3.6 |
| Abamectin | 0.01–5.0 | 0.9975 | 0.01 | 66.3 ± 3.3 | 65.5 ± 2.2 | 69.8 ± 2.4 |
| Chlorantraniliprole | 0.01–5.0 | 0.9983 | 0.01 | 94.1 ± 3.0 | 115.8 ± 3.5 | 112.7 ± 2.8 |
| Thiamethoxam | 0.01–5.0 | 0.9921 | 0.01 | 105.2 ± 3.2 | 97.1 ± 4.0 | 93.6 ± 4.5 |
| Lamda-cyhalothrin | 0.01–5.0 | 0.9954 | 0.01 | 102.4 ± 6.2 | 92.3 ± 8.2 | 105.1 ± 5.6 |
| Acetamiprid | 0.01–5.0 | 0.9961 | 0.01 | 80.2 ± 9.2 | 88.6 ± 10.1 | 90.3 ± 8.5 |
| Pesticide | Linear Range (mg·kg−1) | Correlation Coefficient (R2) | LOQ (mg·kg−1) | Recovery (%) ± RSDs (%), Spiked Level (mg·kg−1) | ||
|---|---|---|---|---|---|---|
| 0.01 | 0.05 | 0.1 | ||||
| Clothianidin | 0.01–10.0 | 0.9965 | 0.01 | 79.2 ± 3.4 | 83.1 ± 4.6 | 82.0 ± 5.2 |
| Imidacloprid | 0.01–10.0 | 0.9959 | 0.01 | 85.2 ± 6.2 | 91.6 ± 2.9 | 106.2 ± 4.3 |
| Abamectin | 0.01–10.0 | 0.9982 | 0.01 | 68.1 ± 2.9 | 69.7 ± 3.0 | 70.2 ± 3.8 |
| Dose | Pesticide | PHI (Days) (mg·kg−1) | MRL (mg·kg−1) | Recommended PHI (Days) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 14 | 21 | 28 | 50 | 60 | 77 | China | CAC | EU | |||
| Recommended dose | Clothianidin (GR) | 0.06 ± 0.00 b | 0.014 ± 0.00 b | <LOQ | <LOQ | - | - | 0.04 | 0.04 | 0.01 | 10 |
| 1.5-fold recommended dose | 0.11 ± 0.00 a | 0.032 ± 0.01 a | 0.014 ± 0.00 | 0.013 ± 0.00 | - | - | 22 | ||||
| Recommended dose | Imidacloprid (GR) | <LOQ | 0.024 ± 0.00 b | 2.81 ± 0.12 b | - | 0.86 ± 0.06 b | 0.4 ± 0.01 a | 5 | 6 | 0.5 | 14 |
| 1.5-fold recommended dose | 0.029 ± 0.00 | 0.23 ± 0.01 a | 4.06 ± 0.21 a | - | 1.9 ± 0.03 a | 0.25 ± 0.06 b | 31 | ||||
| Recommended dose | Abamectin (GR) | 0.021 ± 0.00 a | 0.016 ± 0.00 b | 0.013 ± 0.00 b | - | <LOQ | <LOQ | 0.05 | 0.03 | 0.01 | 9 |
| 1.5-fold recommended dose | 0.021 ± 0.00 a | 0.033 ± 0.00 a | 0.024 ± 0.00 a | - | <LOQ | <LOQ | 9 | ||||
| Dose | Pesticide | PHI (Days) (mg·kg−1) | MRL (mg·kg−1) | Recommended PHI (Days) | |||||||
| 2 h | 1 | 3 | 5 | 7 | 14 | China | CAC | EU | |||
| Recommended dose | Chlorantraniliprole (SC) | 2.70 ± 0.08 b | 2.64 ± 0.11 b | 2.28 ± 0.09 b | 2.58 ± 0.21 b | 2.43 ± 0.07 b | 1.78 ± 0.00 a | 7 * | 7 | 0.06 | 31 |
| 1.5-fold recommended dose | 4.96 ± 0.26 a | 3.99 ± 0.17 a | 3.70 ± 0.25 a | 3.48 ± 0.07 a | 2.80 ± 0.04 a | 1.85 ± 0.08 a | (7) 31 | ||||
| Treatment | Pesticide | PHI (Days) (mg·kg−1) | MRL (mg·kg−1) | Recommended PHI (Days) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2 h | 1 | 3 | 5 | 7 | 14 | China | CAC | EU | |||
| Control | Thiamethoxam (SC) | 0.101 ± 0.01 b | 0.084 ± 0.01 a | 0.044 ± 0.01 a | 0.020 ± 0.00 a | 0.011 ± 0.00 | <LOQ | 1 | 1 | 0.01 | 7 |
| Se nanoparticles | 0.131 ± 0.01 a | 0.073 ± 0.01 a | 0.037 ± 0.00 a | 0.017 ± 0.00 a | <LOQ | <LOQ | 7 | ||||
| Control | Abamectin (EC) | 0.219 ± 0.04 b | 0.139 ± 0.02 a | 0.025 ± 0.00 a | <LOQ | <LOQ | <LOQ | 0.05 | 0.03 | 0.01 | 2 |
| Se nanoparticles | 0.351 ± 0.03 a | 0.166 ± 0.02 a | 0.014 ± 0.00 a | 0.011 ± 0.00 | <LOQ | <LOQ | 3 | ||||
| Control | Imidacloprid (SC) | 4.446 ± 0.30 a | 2.565 ± 0.26 a | 1.263 ± 0.08 a | 0.338 ± 0.03 a | 0.301 ± 0.03 a | 0.034 ± 0.01 a | 5 | 6 | 0.5 | 2 |
| Se nanoparticles | 4.613 ± 0.07 a | 2.154 ± 0.11 b | 0.803 ± 0.06 b | 0.529 ± 0.01 a | 0.281 ± 0.01 a | 0.030 ± 0.01 a | 2 | ||||
| Control | Lambda-cyhalothrin (SC) | 0.687 ± 0.03 a | 0.585 ± 0.01 a | 0.601 ± 0.02 a | 0.151 ± 0.03 a | 0.175 ± 0.03 a | 0.118 ± 0.03 a | 0.5 | / | 0.07 | 2 |
| Se nanoparticles | 0.709 ± 0.06 a | 0.621 ± 0.07 a | 0.578 ± 0.02 a | 0.196 ± 0.02 a | 0.191 ± 0.05 a | 0.035 ± 0.02 b | 3 | ||||
| Control | Acetamiprid (WDG) | 0.042 ± 0.00 a | 0.031 ± 0.01 b | 0.013 ± 0.00 b | 0.016 ± 0.00 a | 0.011 ± 0.00 b | <LOQ | 3 | 1.5 | 0.01 | 1 |
| Se nanoparticles | 0.040 ± 0.00 a | 0.039 ± 0.01 a | 0.023 ± 0.01 a | 0.017 ± 0.00 a | 0.024 ± 0.00 a | <LOQ | 1 | ||||
| Pesticide | Sample Type | Dosage | Initial Residue (mg·kg−1) | Final Residue (mg·kg−1) | Dissipation Kinetics | Correlation Coefficient (R) | Half-Life (Day) |
|---|---|---|---|---|---|---|---|
| Clothianidin (GR) | Celery | Recommended dose | 0.060 ± 0.00 b | <LOQ | Ct = 0.059 × 10−0.04t | 0.78 | 16.9 |
| 1.5-fold recommended dose | 0.108 ± 0.00 a | 0.013 ± 0.00 | Ct = 0.119 × 10−0.05t | 0.81 | 15.8 | ||
| Soil | Recommended dose | 0.705 ± 0.10 b | 0.015 ± 0.00 a | Ct = 3.632 × 10−0.11t | 0.96 | 21.2 | |
| 1.5-fold recommended dose | 1.238 ± 0.22 a | 0.012 ± 0.00 a | Ct = 5.636 × 10−0.13t | 0.86 | 17 | ||
| Imidacloprid (GR) | Celery | Recommended dose | <LOQ | 0.404 ± 0.01 a | Ct = 8.619 × 10−0.04t | 0.99 | 46.5 |
| 1.5-fold recommended dose | 0.029 ± 0.00 | 0.246 ± 0.06 b | Ct = 22.670 × 10−0.05t | 0.91 | 45.5 | ||
| Soil | Recommended dose | 0.083 ± 0.01 a | 0.423 ± 0.03 b | Ct = 23.988 × 10−0.07t | 0.99 | 37.9 | |
| 1.5-fold recommended dose | 0.154 ± 0.05 a | 1.991 ± 0.11 a | Ct = 75.784 × 10−0.07t | 0.98 | 40.3 | ||
| Abamectin (GR) | Celery | Recommended dose | 0.021 ± 0.00 a | <LOQ | Ct = 0.035 × 10−0.04t | 0.99 | 33.1 |
| 1.5-fold recommended dose | 0.021 ± 0.00 a | <LOQ | Ct = 0.041 × 10−0.02t | 0.97 | 39.8 | ||
| Soil | Recommended dose | 0.141 ± 0.01 b | 0.163 ± 0.01 b | Ct = 1.907 × 10−0.03t | 0.94 | 55.9 | |
| 1.5-fold recommended dose | 0.279 ± 0.02a | 1.389 ± 0.08 a | Ct= 3.611 × 10−0.02t | 0.99 | 70.3 | ||
| Chlorantraniliprole (SC) | Celery | Recommended dose | 2.701 ± 0.08b | 1.784 ± 0.00 a | Ct= 2.766 × 10−0.03t | 0.98 | - |
| 1.5-fold recommended dose | 4.958 ± 0.26a | 1.847 ± 0.08 a | Ct= 4.614 × 10−0.07t | 0.97 | - |
| Pesticide | Treatment | Initial Residue (mg·kg−1) | Final Residue (mg·kg−1) | Dissipation Kinetics | Correlation Coefficient (R) | Half-Life (Day) |
|---|---|---|---|---|---|---|
| Thiamethoxam (SC) | Control | 0.101 ± 0.01 b | <LOQ | Ct = 0.109 × 10−0.32t | 0.99 | 2.4 |
| Se nanoparticles | 0.131 ± 0.01 a | <LOQ | Ct = 0.103 × 10−0.33t | 0.99 | 1.4 | |
| Abamectin (EC) | Control | 0.219 ± 0.04 b | <LOQ | Ct = 0.093 × 10−0.34t | 0.86 | 0.5 |
| Se nanoparticles | 0.351 ± 0.03 a | <LOQ | Ct = 0.198 × 10−0.52t | 0.98 | 0.2 | |
| Imidacloprid (SC) | Control | 4.446 ± 0.30 a | 0.034 ± 0.01 a | Ct = 3.365 × 10−0.34t | 0.98 | 1.2 |
| Se nanoparticles | 4.613 ± 0.07 a | 0.030 ± 0.01 a | Ct = 3.158 × 10−0.34t | 0.99 | 0.9 | |
| Lambda-cyhalothrin (SC) | Control | 0.687 ± 0.03 a | 0.118 ± 0.03 a | Ct = 0.595 × 10−0.13t | 0.86 | 4.2 |
| Se nanoparticles | 0.709 ± 0.06 a | 0.035 ± 0.02 b | Ct = 0.797 × 10−0.22t | 0.98 | 3.7 | |
| Acetamiprid (WDG) | Control | 0.042 ± 0.00 a | <LOQ | Ct = 0.036 × 10−0.19t | 0.97 | 2.9 |
| Se nanoparticles | 0.040 ± 0.00 a | <LOQ | Ct = 0.042 × 10−0.14t | 0.95 | 5.3 |
| Pesticide | Treatment | STMR (mg·kg−1) | ADI (mg·kg−1 bw) | ARfD (mg·kg−1 bw) | RQ c% | RQ a% |
|---|---|---|---|---|---|---|
| Clothianidin (GR) | Recommended dose | 0.010 | 0.1 | 0.6 | 9.85 | 1.64 |
| 1.5-fold recommended dose | 0.021 | 9.86 | 1.64 | |||
| Imidacloprid (GR) | Recommended dose | 0.40 | 0.06 | 0.4 | 22.83 | 3.34 |
| 1.5-fold recommended dose | 0.23 | 22.42 | 3.42 | |||
| Abamectin (GR) | Recommended dose | 0.016 | 0.001 | - | 116.47 | - |
| 1.5-fold recommended dose | 0.024 | 117.63 | - | |||
| Chlorantraniliprole (SC) | Recommended dose | 2.46 | 2 | - | 3.83 | - |
| 1.5-fold recommended dose | 3.52 | 3.90 | - |
| Pesticide | Treatment | STMR (mg·kg−1) | ADI (mg·kg−1 bw) | ARfD (mg·kg−1 bw) | RQ c% | RQ a% |
|---|---|---|---|---|---|---|
| Thiamethoxam (SC) | Control | 0.046 | 0.08 | 1.0 | 7.61 | 0.61 |
| Se nanoparticles | 0.050 | 7.62 | 0.61 | |||
| Abamectin (EC) | Control | 0.13 | 0.001 | - | 133.03 | - |
| Se nanoparticles | 0.08 | 125.77 | - | |||
| Imidacloprid (SC) | Control | 0.78 | 0.06 | 0.4 | 23.75 | 3.56 |
| Se nanoparticles | 0.64 | 23.41 | 3.51 | |||
| Lambda-cyhalothrin (SC) | Control | 0.18 | 0.02 | 0.02 | 63.30 | 63.30 |
| Se nanoparticles | 0.40 | 64.90 | 64.90 | |||
| Acetamiprid (WDG) | Control | 0.016 | 0.07 | 0.1 | 32.48 | 22.74 |
| Se nanoparticles | 0.028 | 32.51 | 22.76 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, L.; Liu, H.; Zhao, D.; Pan, C.; Wang, C. Pesticide Residue Behavior and Risk Assessment in Celery after Se Nanoparticles Application. Foods 2021, 10, 1987. https://doi.org/10.3390/foods10091987
Kang L, Liu H, Zhao D, Pan C, Wang C. Pesticide Residue Behavior and Risk Assessment in Celery after Se Nanoparticles Application. Foods. 2021; 10(9):1987. https://doi.org/10.3390/foods10091987
Chicago/Turabian StyleKang, Lu, Hejiang Liu, Duoyong Zhao, Canping Pan, and Cheng Wang. 2021. "Pesticide Residue Behavior and Risk Assessment in Celery after Se Nanoparticles Application" Foods 10, no. 9: 1987. https://doi.org/10.3390/foods10091987







