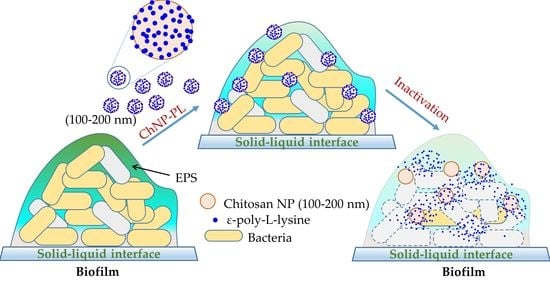
Inactivation of Polymicrobial Biofilms of Foodborne Pathogens Using Epsilon Poly-L-Lysin Conjugated Chitosan Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial and Mammalian Cell Lines Used in This Study
2.2. Synthesis of Chitosan Nanoparticles and Conjugation with ε-poly-L-lysine
2.3. Antibacterial Activity Testing of Chitosan Nanoparticles
2.4. Cell Proliferation and Cytotoxicity Tests of ChNP-PL
2.5. Single and Mixed Culture Biofilm Formation
2.6. Prevention of Biofilm Formation and Inactivation of Preformed Biofilm by ChNP-PL
3. Results
3.1. Synthesis of Chitosan Nanoparticles Conjugated with ε-poly-L-lysine (ChNP-PL)
3.2. ChNP and PL Exhibited Synergistic Antimicrobial Activity
3.3. ChNP-PL Is Nontoxic to Intestinal Epithelial Cells
3.4. Shifting of Population Dynamics within Mixed Culture Biofilms
3.5. ChNP-PL Effectively Prevented Biofilm Formation by Mono- or Mixed-Cultures
3.6. ChNP-PL Inactivated Preformed Biofilms by All Tested Bacteria except P. aeruginosa
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kirk, M.D.; Pires, S.M.; Black, R.E.; Caipo, M.; Crump, J.A.; Devleesschauwer, B.; Doepfer, D.; Fazil, A.; Fischer-Walker, C.L.; Hald, T.; et al. World Health Organization estimates of the global and regional disease burden of 22 foodborne bacterial, protozoal, and viral diseases, 2010: A data synthesis. PLoS Med. 2015, 12, e1001921. [Google Scholar] [CrossRef] [Green Version]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States—major pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Scharff, R. Economic burden from health losses due to foodborne illness in the United States. J. Food Prot. 2012, 75, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, V.; Wiedmann, M.; Teixeira, P.; Stasiewicz, M.J. Listeria monocytogenes persistence in food-associated environments: Epidemiology, strain characteristics, and implications for public health. J. Food Prot. 2014, 77, 150–170. [Google Scholar] [CrossRef]
- Carpentier, B.; Cerf, O. Review-Persistence of Listeria monocytogenes in food industry equipment and premises. Int. J. Food Microbiol. 2011, 145, 1–8. [Google Scholar] [CrossRef]
- Heir, E.; Møretrø, T.; Simensen, A.; Langsrud, S. Listeria monocytogenes strains show large variations in competitive growth in mixed culture biofilms and suspensions with bacteria from food processing environments. Int. J. Food Microbiol. 2018, 275, 46–55. [Google Scholar] [CrossRef]
- Chitlapilly Dass, S.; Bosilevac, J.M.; Weinroth, M.; Elowsky, C.G.; Zhou, Y.; Anandappa, A.; Wang, R. Impact of mixed biofilm formation with environmental microorganisms on E. coli O157:H7 survival against sanitization. NPJ Sci. Food 2020, 4, 16. [Google Scholar] [CrossRef]
- Bonsaglia, E.C.R.; Silva, N.C.C.; Fernades, A.; Araujo, J.P.; Tsunemi, M.H.; Rall, V.L.M. Production of biofilm by Listeria monocytogenes in different materials and temperatures. Food Control 2014, 35, 386–391. [Google Scholar] [CrossRef]
- Fu, Y.; Deering, A.J.; Bhunia, A.K.; Yao, Y. Biofilm of Escherichia coli O157:H7 on cantaloupe surface is resistant to lauroyl arginate ethyl and sodium hypochlorite. Int. J. Food Microbiol. 2017, 260, 11–16. [Google Scholar] [CrossRef]
- Bai, X.; Nakatsu, C.H.; Bhunia, A.K. Bacterial biofilms and their implications in pathogenesis and food safety. Foods 2021, 10, 2117. [Google Scholar] [CrossRef]
- Bhunia, A.K. Food safety concerns of biofilm-forming zoonotic bacterial pathogens. Indian J. Anim. Health 2021, 60, 44–51. [Google Scholar] [CrossRef]
- Srey, S.; Jahid, I.K.; Ha, S.-D. Biofilm formation in food industries: A food safety concern. Food Control 2013, 31, 572–585. [Google Scholar] [CrossRef]
- Fu, Y.; Deering, A.J.; Bhunia, A.K.; Yao, Y. Pathogen biofilm formation on cantaloupe surface and its impact on the antibacterial effect of lauroyl arginate ethyl. Food Microbiol. 2017, 64, 139–144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Bridier, A.; Briandet, R.; Thomas, V.; Dubois-Brissonnet, F. Resistance of bacterial biofilms to disinfectants: A review. Biofouling 2011, 27, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Rawool, D.B.; Doijad, S.; Poharkar, K.; Mohan, V.; Barbuddhe, S.B.; Kolhe, R.; Kurkure, N.V.; Kumar, A.; Malik, S.V.S. Biofilm formation and genetic diversity of Salmonella isolates recovered from clinical, food, poultry and environmental sources. Infect. Genet. Evol. 2015, 36, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Ma, R.; Grimm, M.C.; Riordan, S.M.; Lan, R.; Zhong, L.; Raftery, M.; Zhang, L. Examination of the anaerobic growth of Campylobacter concisus strains. Int. J. Microbiol. 2014, 2014, 476047. [Google Scholar] [CrossRef] [Green Version]
- Bridier, A.; del Pilar Sanchez-Vizuete, M.; Le Coq, D.; Aymerich, S.; Meylheuc, T.; Maillard, J.-Y.; Thomas, V.; Dubois-Brissonnet, F.; Briandet, R. Biofilms of a Bacillus subtilis hospital isolate protect Staphylococcus aureus from biocide action. PLoS ONE 2012, 7, e44506. [Google Scholar] [CrossRef]
- Van der Veen, S.; Abee, T. Mixed species biofilms of Listeria monocytogenes and Lactobacillus plantarum show enhanced resistance to benzalkonium chloride and peracetic acid. Int. J. Food Microbiol. 2011, 144, 421–431. [Google Scholar] [CrossRef] [Green Version]
- Friedlander, A.; Nir, S.; Reches, M.; Shemesh, M. Preventing biofilm formation by dairy-associated bacteria using peptide-coated surfaces. Front. Microbiol. 2019, 10, 1405. [Google Scholar] [CrossRef] [Green Version]
- Meireles, A.; Borges, A.; Giaouris, E.; Simões, M. The current knowledge on the application of anti-biofilm enzymes in the food industry. Food Res. Int. 2016, 86, 140–146. [Google Scholar] [CrossRef] [Green Version]
- Panebianco, F.; Rubiola, S.; Chiesa, F.; Civera, T.; Di Ciccio, P.A. Effect of gaseous ozone on Listeria monocytogenes planktonic cells and biofilm: An in vitro study. Foods 2021, 10, 1484. [Google Scholar] [CrossRef] [PubMed]
- Motelica, L.; Ficai, D.; Ficai, A.; Truşcă, R.-D.; Ilie, C.-I.; Oprea, O.-C.; Andronescu, E. Innovative antimicrobial chitosan/ZnO/Ag NPs/citronella essential oil nanocomposite—Potential coating for grapes. Foods 2020, 9, 1801. [Google Scholar] [CrossRef] [PubMed]
- Simões, M.; Simões, L.C.; Vieira, M.J. A review of current and emergent biofilm control strategies. LWT-Food Sci. Technol. 2010, 43, 573–583. [Google Scholar] [CrossRef] [Green Version]
- Aryal, M.; Muriana, P.M. Efficacy of Commercial Sanitizers Used in Food Processing Facilities for Inactivation of Listeria monocytogenes, E. coli O157:H7, and Salmonella Biofilms. Foods 2019, 8, 639. [Google Scholar] [CrossRef] [Green Version]
- Delshadi, R.; Bahrami, A.; Assadpour, E.; Williams, L.; Jafari, S.M. Nano/microencapsulated natural antimicrobials to control the spoilage microorganisms and pathogens in different food products. Food Control 2021, 128, 108180. [Google Scholar] [CrossRef]
- Zorraquín-Peña, I.; Cueva, C.; Bartolomé, B.; Moreno-Arribas, M.V. Silver Nanoparticles against Foodborne Bacteria. Effects at Intestinal Level and Health Limitations. Microorganisms 2020, 8, 132. [Google Scholar] [CrossRef] [Green Version]
- Singh, A.K.; Bai, X.; Amalaradjou, M.A.R.; Bhunia, A.K. Antilisterial and antibiofilm activities of pediocin and LAP functionalized gold nanoparticles. Front. Sustain. Food Syst. 2018, 2, 74. [Google Scholar] [CrossRef] [Green Version]
- Rampino, A.; Borgogna, M.; Blasi, P.; Bellich, B.; Cesaro, A. Chitosan nanoparticles: Preparation, size evolution and stability. Int. J. Pharm. 2013, 455, 219–228. [Google Scholar] [CrossRef]
- Wu, T.; Zivanovic, S.; Draughon, F.A.; Sams, C.E. Chitin and chitosan value-added products from mushroom waste. J. Agric. Food Chem. 2004, 52, 7905–7910. [Google Scholar] [CrossRef]
- Rabea, E.I.; Badawy, M.E.T.; Stevens, C.V.; Smagghe, G.; Steurbaut, W. Chitosan as antimicrobial agent: Applications and mode of action. Biomacromolecules 2003, 4, 1457–1465. [Google Scholar] [CrossRef] [PubMed]
- Raafat, D.; Sahl, H.G. Chitosan and its antimicrobial potential–a critical literature survey. Microb. Biotechnol. 2009, 2, 186–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chavez de Paz, L.E.; Resin, A.; Howard, K.A.; Sutherland, D.S.; Wejse, P.L. Antimicrobial effect of chitosan nanoparticles on streptococcus mutans biofilms. Appl. Environ. Microbiol. 2011, 77, 3892–3895. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radulescu, M.; Ficai, D.; Oprea, O.; Ficai, A.; Andronescu, E.; Holban, A.M. Antimicrobial Chitosan based formulations with impact on different biomedical applications. Curr. Pharm. Biotechnol. 2015, 16, 128–136. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, Q. Recent advances of chitosan and its derivatives for novel applications in food science. J. Food Processing Beverages 2013, 1, 1–13. [Google Scholar]
- Mu, H.; Guo, F.; Niu, H.; Liu, Q.; Wang, S.; Duan, J. Chitosan improves anti-biofilm efficacy of gentamicin through facilitating antibiotic penetration. Int. J. Mol. Sci. 2014, 15, 22296–22308. [Google Scholar] [CrossRef] [Green Version]
- Fang, L.; Wolmarans, B.; Kang, M.; Jeong, K.C.; Wright, A.C. Application of chitosan microparticles for reduction of Vibrio species in seawater and live oysters (Crassostrea virginica). Appl. Environ. Microbiol. 2015, 81, 640–647. [Google Scholar] [CrossRef] [Green Version]
- Ye, R.; Xu, H.; Wan, C.; Peng, S.; Wang, L.; Xu, H.; Aguilar, Z.P.; Xiong, Y.; Zeng, Z.; Wei, H. Antibacterial activity and mechanism of action of ε-poly-l-lysine. Biochem. Biophys. Res. Commun. 2013, 439, 148–153. [Google Scholar] [CrossRef]
- Yoshida, T.; Nagasawa, T. epsilon-Poly-L-lysine: Microbial production, biodegradation and application potential. Appl. Microbiol. Biotechnol. 2003, 62, 21–26. [Google Scholar] [CrossRef]
- Hyldgaard, M.; Mygind, T.; Vad, B.S.; Stenvang, M.; Otzen, D.E.; Meyer, R.L. The antimicrobial mechanism of action of Epsilon- Poly-L-Lysine. Appl. Environ. Microbiol. 2014, 80, 7758–7770. [Google Scholar] [CrossRef] [Green Version]
- Na, S.; Kim, J.-H.; Jang, H.-J.; Park, H.J.; Oh, S.-W. Shelf life extension of Pacific white shrimp (Litopenaeus vannamei) using chitosan and ε-polylysine during cold storage. Int. J. Biol. Macromol. 2018, 115, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Li, F.; Guan, H.; Xu, Y.; Fu, Q.; Li, D. Combination of nisin and ε-polylysine with chitosan coating inhibits the white blush of fresh-cut carrots. Food Control 2017, 74, 34–44. [Google Scholar] [CrossRef]
- Xu, Z.; Xie, J.; Soteyome, T.; Peters, B.M.; Shirtliff, M.E.; Liu, J.; Harro, J.M. Polymicrobial interaction and biofilms between Staphylococcus aureus and Pseudomonas aeruginosa: An underestimated concern in food safety. Curr. Opin. Food Sci. 2019, 26, 57–64. [Google Scholar] [CrossRef]
- Langsrud, S.; Moen, B.; Møretrø, T.; Løype, M.; Heir, E. Microbial dynamics in mixed culture biofilms of bacteria surviving sanitation of conveyor belts in salmon-processing plants. J. Appl. Microbiol. 2016, 120, 366–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sullivan, D.J.; Cruz-Romero, M.; Collins, T.; Cummins, E.; Kerry, J.P.; Morris, M.A. Synthesis of monodisperse chitosan nanoparticles. Food Hydrocoll. 2018, 83, 355–364. [Google Scholar] [CrossRef]
- Zhu, X.; Liu, D.; Singh, A.K.; Drolia, R.; Bai, X.; Tenguria, S.; Bhunia, A.K. Tunicamycin mediated inhibition of wall teichoic acid affect Staphylococcus aureus and Listeria monocytogenes cell morphology, biofilm formation and virulence. Front. Microbiol. 2018, 9, 1352. [Google Scholar] [CrossRef]
- Zameer, F.; Kreft, J.; Gopal, S. Interaction of Listeria monocytogenes and Staphylococcus epidermidis in dual species biofilms. J. Food Saf. 2010, 30, 954–968. [Google Scholar] [CrossRef] [Green Version]
- Cherifi, T.; Carrillo, C.; Lambert, D.; Miniaï, I.; Quessy, S.; Larivière-Gauthier, G.; Blais, B.; Fravalo, P. Genomic characterization of Listeria monocytogenes isolates reveals that their persistence in a pig slaughterhouse is linked to the presence of benzalkonium chloride resistance genes. BMC Microbiol. 2018, 18, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, D.; Wiedmann, M.; McLandsborough, L.A. Microtiter plate assay for assessment of Listeria monocytogenes biofilm formation. Appl. Environ. Microbiol. 2002, 68, 2950–2958. [Google Scholar] [CrossRef] [Green Version]
- Bai, X.; Liu, D.; Xu, L.; Tenguria, S.; Drolia, R.; Gallina, N.L.F.; Cox, A.D.; Koo, O.-K.; Bhunia, A.K. Biofilm-isolated Listeria monocytogenes exhibits reduced systemic dissemination at the early (12–24 h) stage of infection in a mouse model. NPJ Biofilms Microbiomes 2021, 7, 18. [Google Scholar] [CrossRef]
- Carvalho, E.L.S.; Grenha, A.; Remuñán-López, C.; Alonso, M.J.; Seijo, B. Chapter 15 Mucosal Delivery of Liposome-Chitosan Nanoparticle Complexes. Methods Enzymol. 2009, 465, 289–312. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, B.; Heir, E.; Møretrø, T.; Skaar, I.; Langsrud, S. Microbial background flora in small-scale cheese production facilities does not inhibit growth and surface attachment of Listeria monocytogenes. J. Dairy Sci. 2013, 96, 6161–6171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frank, J.F.; Gillett, R.A.N.; Ware, G.O. Association of Listeria spp. contamination in the dairy processing plant environment with the presence of staphylococci. J. Food Prot. 1990, 53, 928–932. [Google Scholar] [CrossRef] [PubMed]
- Carpentier, B.; Chassaing, D. Interactions in biofilms between Listeria monocytogenes and resident microorganisms from food industry premises. Int. J. Food Microbiol. 2004, 97, 111–122. [Google Scholar] [CrossRef]
- Cotter, P.A.; Stibitz, S. c-di-GMP-mediated regulation of virulence and biofilm formation. Curr. Opin. Microbiol. 2007, 10, 17–23. [Google Scholar] [CrossRef]
- Römling, U.; Galperin, M.Y.; Gomelsky, M. Cyclic di-GMP: The first 25 years of a universal bacterial second messenger. Microbiol. Mol. Biol. Rev. 2013, 77, 1–52. [Google Scholar] [CrossRef] [Green Version]
- Landini, P.; Antoniani, D.; Burgess, J.G.; Nijland, R. Molecular mechanisms of compounds affecting bacterial biofilm formation and dispersal. Appl. Microbiol. Biotechnol. 2010, 86, 813–823. [Google Scholar] [CrossRef]
- Batt, C.A.; Tortorello, M.-L. Encyclopedia of Food Microbiology; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- NRC. National Research Council: Acute Exposure Guideline Levels for Selected Airborne Chemicals; National Academies Press (US): Washington, DC, USA, 2010; Volume 8. [Google Scholar]
- To, M.S.; Favrin, S.; Romanova, N.; Griffiths, M.W. Postadaptational resistance to benzalkonium chloride and subsequent physicochemical modifications of Listeria monocytogenes. Appl. Environ. Microbiol. 2002, 68, 5258–5264. [Google Scholar] [CrossRef] [Green Version]
- Yu, T.; Jiang, X.; Zhang, Y.; Ji, S.; Gao, W.; Shi, L. Effect of benzalkonium chloride adaptation on sensitivity to antimicrobial agents and tolerance to environmental stresses in Listeria monocytogenes. Front. Microbiol. 2018, 9, 2906. [Google Scholar] [CrossRef] [Green Version]
- Knezevic, P.; Aleksic, V.; Simin, N.; Svircev, E.; Petrovic, A.; Mimica-Dukic, N. Antimicrobial activity of Eucalyptus camaldulensis essential oils and their interactions with conventional antimicrobial agents against multi-drug resistant Acinetobacter baumannii. J. Ethnopharmacol. 2016, 178, 125–136. [Google Scholar] [CrossRef]
- Fournomiti, M.; Kimbaris, A.; Mantzourani, I.; Plessas, S.; Theodoridou, I.; Papaemmanouil, V.; Kapsiotis, I.; Panopoulou, M.; Stavropoulou, E.; Bezirtzoglou, E.E. Antimicrobial activity of essential oils of cultivated oregano (Origanum vulgare), sage (Salvia officinalis), and thyme (Thymus vulgaris) against clinical isolates of Escherichia coli, Klebsiella oxytoca, and Klebsiella pneumoniae. Microb. Ecol. Health Dis. 2015, 26, 23289. [Google Scholar] [CrossRef] [PubMed]
- Mekonnen, A.; Yitayew, B.; Tesema, A.; Taddese, S. In vitro antimicrobial activity of essential oil of Thymus schimperi, Matricaria chamomilla, Eucalyptus globulus, and Rosmarinus officinalis. Int. J. Microbiol. 2016, 2016, 9545693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duarte, A.E.; De Menezes, I.R.A.; Bezerra Morais Braga, M.F.; Leite, N.F.; Barros, L.M.; Waczuk, E.P.; Pessoa da Silva, M.A.; Boligon, A.; Teixeira Rocha, J.B.; Souza, D.O. Antimicrobial activity and Modulatory effect of essential oil from the leaf of Rhaphiodon echinus (Nees & Mart) Schauer on some antimicrobial drugs. Molecules 2016, 21, 743. [Google Scholar] [CrossRef]
- Esmaeili, A.; Asgari, A. In vitro release and biological activities of Carum copticum essential oil (CEO) loaded chitosan nanoparticles. Int. J. Biol. Macromol. 2015, 81, 283–290. [Google Scholar] [CrossRef]
- No, H.; Meyers, S.P.; Prinyawiwatkul, W.; Xu, Z. Applications of chitosan for improvement of quality and shelf life of foods: A review. J. Food Sci. 2007, 72, R87–R100. [Google Scholar] [CrossRef]
- Sudarshan, N.R.; Hoover, D.G.; Knorr, D. Antibacterial action of chitosan. Food Biotechnol. 1992, 6, 257–272. [Google Scholar] [CrossRef]
- Duan, C.; Meng, X.; Meng, J.; Khan, M.I.H.; Dai, L.; Khan, A.; An, X.; Zhang, J.; Huq, T.; Ni, Y. Chitosan as a preservative for fruits and vegetables: A review on chemistry and antimicrobial properties. J. Bioresour. Bioprod. 2019, 4, 11–21. [Google Scholar] [CrossRef]
- FDA. GRAS Notices: Shrimp-Derived Chitosan. Available online: https://www.cfsanappsexternal.fda.gov/scripts/fdcc/index.cfm?set=GRASNotices&id=443&sort=GRN_No&order=DESC&startrow=1&type=basic&search=chitosan (accessed on 22 December 2021).
- FDA. GRAS Notice 000135: ε-Polylysine. 2004. Available online: https://www.cfsanappsexternal.fda.gov/scripts/fdcc/?set=GRASNotices (accessed on 22 December 2021).
- Geornaras, I.; Yoon, Y.; Belk, K.; Smith, G.; Sofos, J. Antimicrobial activity of ɛ-polylysine against Escherichia coli O157: H7, Salmonella typhimurium, and Listeria monocytogenes in various food extracts. J. Food Sci. 2007, 72, M330–M334. [Google Scholar] [CrossRef]
- You, X.; Einson, J.E.; Lopez-Pena, C.L.; Song, M.; Xiao, H.; McClements, D.J.; Sela, D.A. Food-grade cationic antimicrobial ε-polylysine transiently alters the gut microbial community and predicted metagenome function in CD-1 mice. NPJ Sci. Food 2017, 1, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Sanguansri, P.; Augustin, M.A. Nanoscale materials development–a food industry perspective. Trends Food Sci. Technol. 2006, 17, 547–556. [Google Scholar] [CrossRef]
- Sawtarie, N.; Cai, Y.; Lapitsky, Y. Preparation of chitosan/tripolyphosphate nanoparticles with highly tunable size and low polydispersity. Colloids Surf. B Biointerfaces 2017, 157, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Janes, K.A.; Fresneau, M.P.; Marazuela, A.; Fabra, A.; Alonso, M.a.J. Chitosan nanoparticles as delivery systems for doxorubicin. J. Control. Release 2001, 73, 255–267. [Google Scholar] [CrossRef]
- Mao, H.-Q.; Roy, K.; Troung-Le, V.L.; Janes, K.A.; Lin, K.Y.; Wang, Y.; August, J.T.; Leong, K.W. Chitosan-DNA nanoparticles as gene carriers: Synthesis, characterization and transfection efficiency. J. Control. Release 2001, 70, 399–421. [Google Scholar] [CrossRef]
- Illum, L.; Jabbal-Gill, I.; Hinchcliffe, M.; Fisher, A.; Davis, S. Chitosan as a novel nasal delivery system for vaccines. Adv. Drug Deliv. Rev. 2001, 51, 81–96. [Google Scholar] [CrossRef]
- Huang, M.; Khor, E.; Lim, L.-Y. Uptake and cytotoxicity of chitosan molecules and nanoparticles: Effects of molecular weight and degree of deacetylation. Pharm. Res. 2004, 21, 344–353. [Google Scholar] [CrossRef]
- Hiraki, J.; Ichikawa, T.; Ninomiya, S.-I.; Seki, H.; Uohama, K.; Seki, H.; Kimura, S.; Yanagimoto, Y.; Barnett, J.W., Jr. Use of ADME studies to confirm the safety of ε-polylysine as a preservative in food. Regul. Toxicol. Pharmacol. 2003, 37, 328–340. [Google Scholar] [CrossRef]
- Rabin, N.; Zheng, Y.; Opoku-Temeng, C.; Du, Y.; Bonsu, E.; Sintim, H.O. Biofilm formation mechanisms and targets for developing antibiofilm agents. Future Med. Chem. 2015, 7, 493–512. [Google Scholar] [CrossRef]
- Limoli, D.H.; Jones, C.J.; Wozniak, D.J. Bacterial extracellular polysaccharides in biofilm formation and function. Microbiol. Spectr. 2015, 3, 223–247. [Google Scholar] [CrossRef] [Green Version]
- Fong, J.N.; Yildiz, F.H. Biofilm matrix proteins. Microbiol. Spectr. 2015, 3, 201–222. [Google Scholar] [CrossRef] [Green Version]
- Høiby, N.; Bjarnsholt, T.; Givskov, M.; Molin, S.; Ciofu, O. Antibiotic resistance of bacterial biofilms. Int. J. Antimicrob. Agents 2010, 35, 322–332. [Google Scholar] [CrossRef] [Green Version]
- Athavale, R.; Sapre, N.; Rale, V.; Tongaonkar, S.; Manna, G.; Kulkarni, A.; Shirolkar, M.M. Tuning the surface charge properties of chitosan nanoparticles. Mater. Lett. 2022, 308, 131114. [Google Scholar] [CrossRef]
- Harmsen, M.; Yang, L.; Pamp, S.J.; Tolker-Nielsen, T. An update on Pseudomonas aeruginosa biofilm formation, tolerance, and dispersal. FEMS Immunol. Med. Microbiol. 2010, 59, 253–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tielen, P.; Rosenau, F.; Wilhelm, S.; Jaeger, K.-E.; Flemming, H.-C.; Wingender, J. Extracellular enzymes affect biofilm formation of mucoid Pseudomonas aeruginosa. Microbiology 2010, 156, 2239–2252. [Google Scholar] [CrossRef] [PubMed] [Green Version]







| Bacteria | MIC (µg/mL) | |
|---|---|---|
| ChNP | ChNP-PL | |
| Pseudomonas aeruginosa ATCC10145 | >37.5 | 12.5–25 |
| P. putida PRI107 | >37.5 | 12.5–25 |
| P. aeruginosa PRI99 | 25–37.5 | 2.5–3.75 |
| Listeria ivanovii ATCC19119 | 25–37.5 | 2.5–3.75 |
| L. seeligeri ATCC 35967 | 25–37.5 | 2.5–3.75 |
| L. marthii ATCC BAA-1595 | 25–37.5 | 2.5–3.75 |
| L. monocytogenes F40 | 25–37.5 | 2.5–3.75 |
| L. monocytogenes F4244 | 25–37.5 | 1.25–2.5 |
| Salmonella enterica serovar Enteritidis PT21 | >37.5 | 3.75–12.5 |
| S. enterica ser. Typhimurium ST1 | >37.5 | 12.5–25 |
| S. enterica ser. Heidelberg 18ENT1418 | >37.5 | 12.5–25 |
| S. enterica ser. Enteritidis 18ENT1344 | >37.5 | 3.75–12.5 |
| Staphylococcus aureus NRRL B767 | >37.5 | 3.75–12.5 |
| S. aureus ATCC25923 | 25–37.5 | 2.5–3.75 |
| S. aureus ATCC29213 | 25–37.5 | 2.5–3.75 |
| Escherichia coli K12 | >37.5 | 2.5–3.75 |
| E. coli O157:H7 SEA13A72 | >37.5 | 2.5–3.75 |
| E. coli O157:H7 PT23 | >37.5 | 1.25–2.5 |
| E. coli O157:H7 EDL933 | >37.5 | 2.5–3.75 |
| Bacteria | Avg CFU/mL | Fold-Change * | ||
|---|---|---|---|---|
| Initial Inoculum | Monoculture Biofilm | Mixed Culture Biofilm | ||
| L. monocytogenes F4244 | 3.5 × 106 | 4.1 × 108 | 2.1 × 109 | 5-fold ↑ |
| S. aureus ATCC25923 | 7.4 × 106 | 4.7 × 109 | 4.2 × 108 | 10-fold ↓ |
| L. monocytogenes F4244 | 3.5 × 106 | 4.1 × 108 | 4.7 × 109 | 11-fold ↑ |
| P. aeruginosa PRI99 | 7.0 × 106 | 7.3 × 109 | 4.6 × 109 | 1.6-fold ↓ |
| L. monocytogenes F4244 | 3.3 × 106 | 3.2 × 108 | 1.1 × 108 | 2.9-fold ↓ |
| S. aureus 2747 | 1.2 × 107 | 3.0 × 108 | 2.1 × 108 | 1.4-fold ↓ |
| L. monocytogenes F4244 | 3.6 × 106 | 4.2 × 108 | 5.1 × 107 | 8.4-fold ↓ |
| S. enterica serovar Enteritidis 1344 | 6.4 × 106 | 7.5 × 107 | 4.0 × 107 | 1.9-fold ↓ |
| L. monocytogenes F4244 | 3.6 × 106 | 4.2 × 108 | 1.4 × 107 | 30-fold ↓ |
| E. coli O157:H7 EDL933 | 3.4 × 106 | 7.6 × 107 | 3.4 × 108 | 4.5-fold ↑ |
| Bacteria | Avg CFU/cm2 | ||
|---|---|---|---|
| Untreated | ChNP * | ChNP-PL * | |
| L. monocytogenes | 9.2 × 106 | <50 (>184,000-fold ↓) | <50 (>184,000-fold ↓) |
| S. aureus | 8.7 × 107 | 8.9 × 106 (9.8-fold ↓) | 3.6 × 104 (2400-fold ↓) |
| P. aeruginosa | 1.7 × 107 | 5.0 × 105 (34-fold ↓) | <50 (>184,000-fold ↓) |
| S. enterica ser. Enteritidis | 1.1 × 107 | 4.4 × 107 (4-fold ↑) | 103 (110,000-fold ↓) |
| E. coli O157:H7 | 2.7 × 107 | 1.2 × 108 (4.4-fold ↑) | <50 (>184,000-fold ↓) |
| Lm + Sa mixed biofilms | |||
| L. monocytogenes | 1.9 × 107 | <50 (>184,000-fold ↓) | <50 (>184,000-fold ↓) |
| S. aureus | 4.2 × 106 | 3.8 × 106 (1.1-fold ↓) | 1.0 × 104 (420-fold ↓) |
| Lm + Pa mixed biofilms | |||
| L. monocytogenes | 8.2 × 107 | <50 (>184,000-fold ↓) | <50 (>184,000-fold ↓) |
| P. aeruginosa | 4.0 × 107 | 1.0 × 103 (40,000-fold ↓) | <50 (>184,000-fold ↓) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bai, X.; Xu, L.; Singh, A.K.; Qiu, X.; Liu, M.; Abuzeid, A.; El-Khateib, T.; Bhunia, A.K. Inactivation of Polymicrobial Biofilms of Foodborne Pathogens Using Epsilon Poly-L-Lysin Conjugated Chitosan Nanoparticles. Foods 2022, 11, 569. https://doi.org/10.3390/foods11040569
Bai X, Xu L, Singh AK, Qiu X, Liu M, Abuzeid A, El-Khateib T, Bhunia AK. Inactivation of Polymicrobial Biofilms of Foodborne Pathogens Using Epsilon Poly-L-Lysin Conjugated Chitosan Nanoparticles. Foods. 2022; 11(4):569. https://doi.org/10.3390/foods11040569
Chicago/Turabian StyleBai, Xingjian, Luping Xu, Atul Kumar Singh, Xiaoling Qiu, Mai Liu, Ahmed Abuzeid, Talaat El-Khateib, and Arun K. Bhunia. 2022. "Inactivation of Polymicrobial Biofilms of Foodborne Pathogens Using Epsilon Poly-L-Lysin Conjugated Chitosan Nanoparticles" Foods 11, no. 4: 569. https://doi.org/10.3390/foods11040569