A Novel Edible Coating Produced from a Wheat Gluten, Pistacia vera L. Resin, and Essential Oil Blend: Antimicrobial Effects and Sensory Properties on Chicken Breast Fillets
Abstract
:1. Introduction
1.1. Edible Films and Coatings
1.2. Pistacia vera L. Resin and Essential Oil
1.3. Scope and Purpose of the Study
2. Materials and Methods
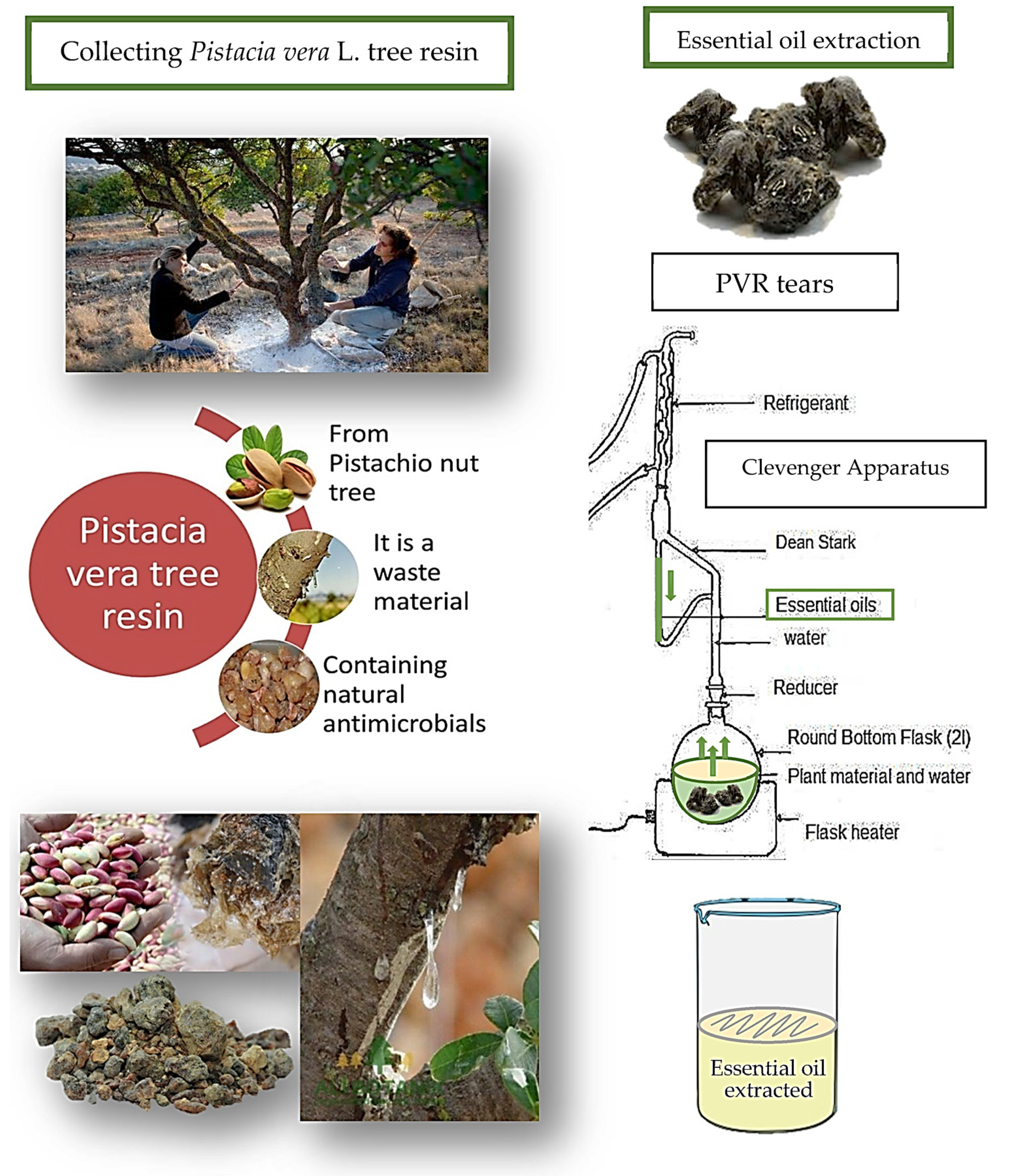
2.1. Obtaining Pistacia vera L. Tree Resin and Essential Oil Extraction
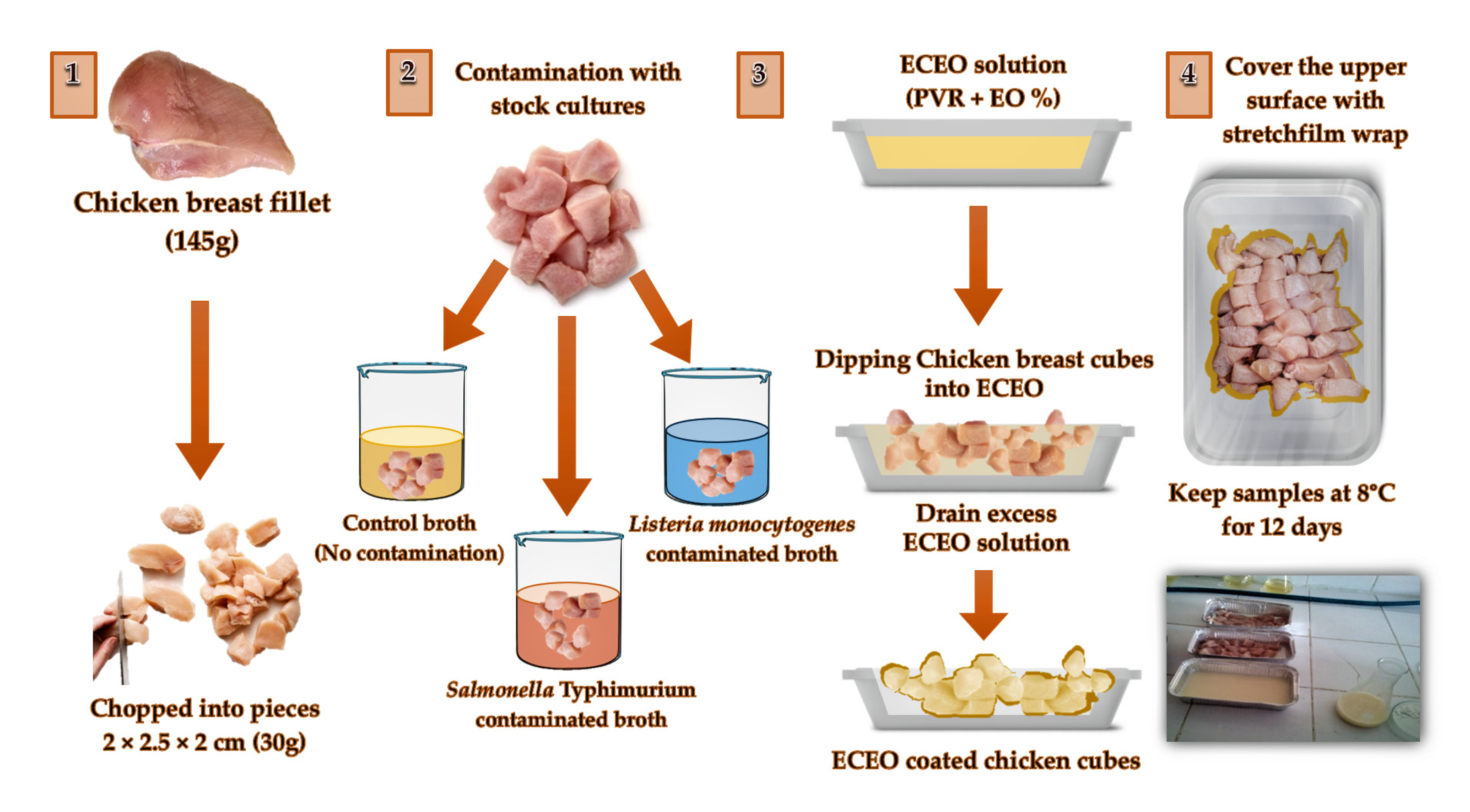
2.2. Preparation of Bacterial Strains and Contamination of Chicken Breast Fillets
2.3. Preparation of Edible Coatings and Application on Chicken Breast Fillets
2.4. Microbiological Analysis
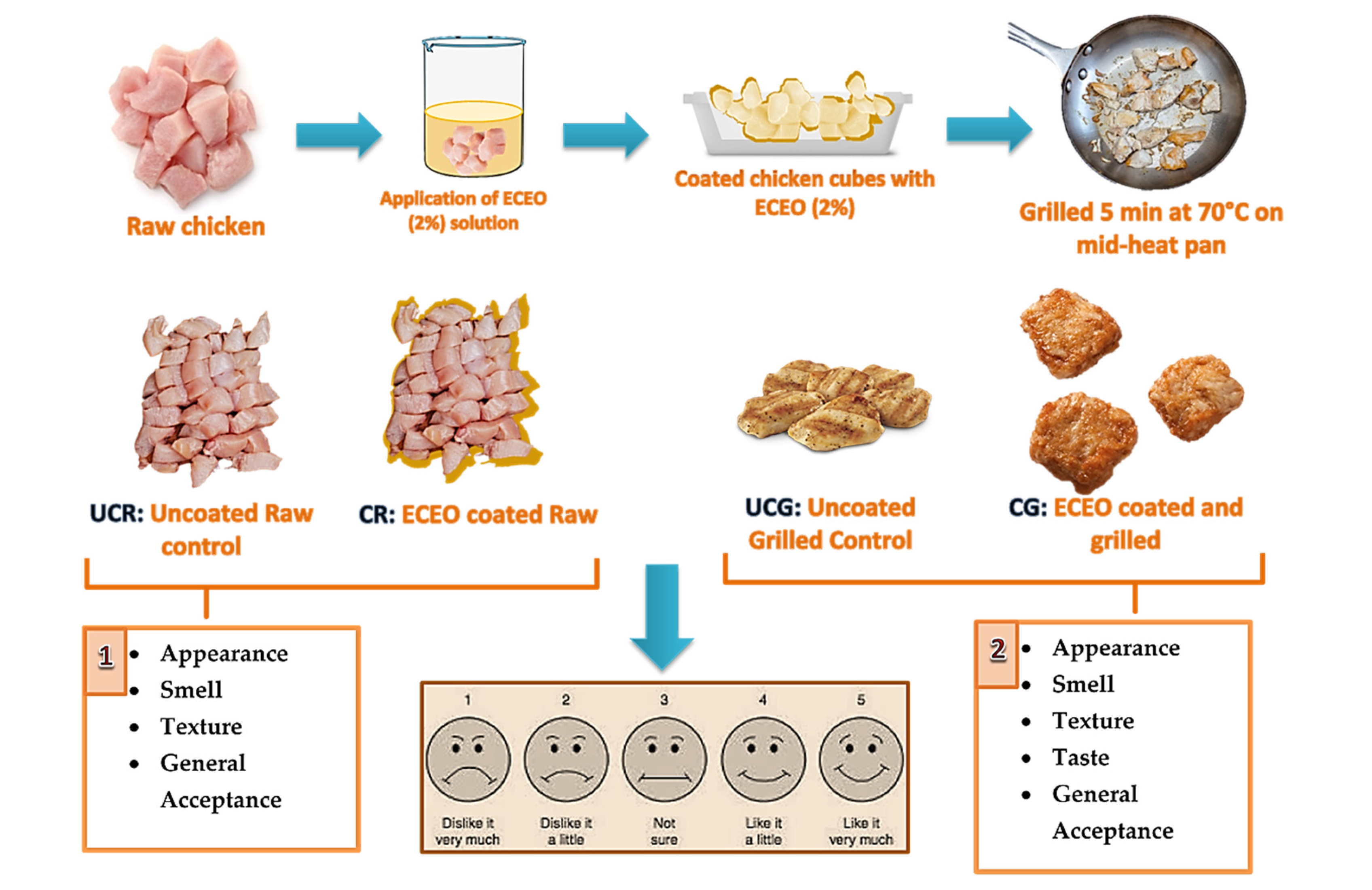
2.5. Sensory Analysis
2.6. Statistical Analysis
3. Results and Discussion
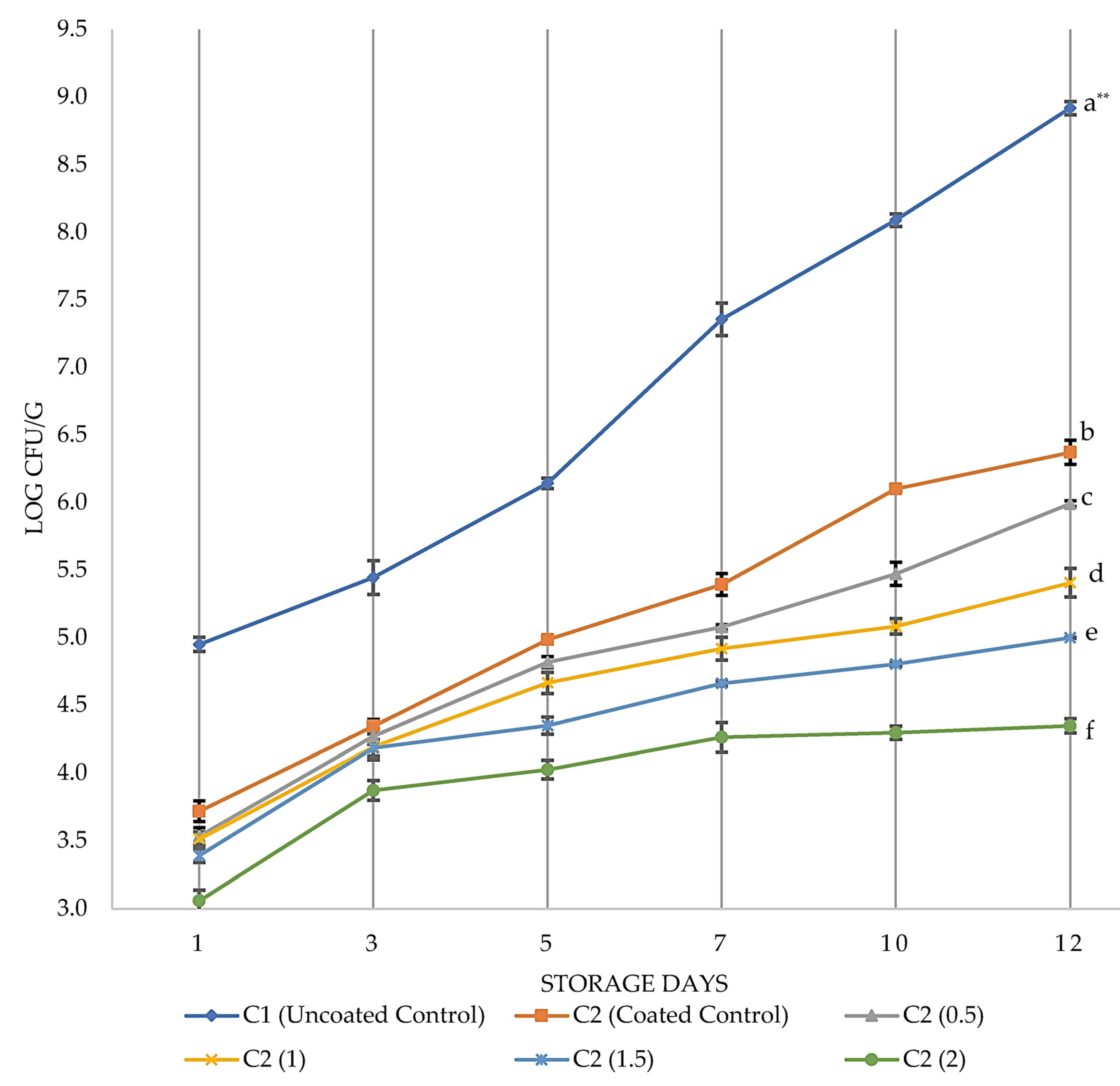
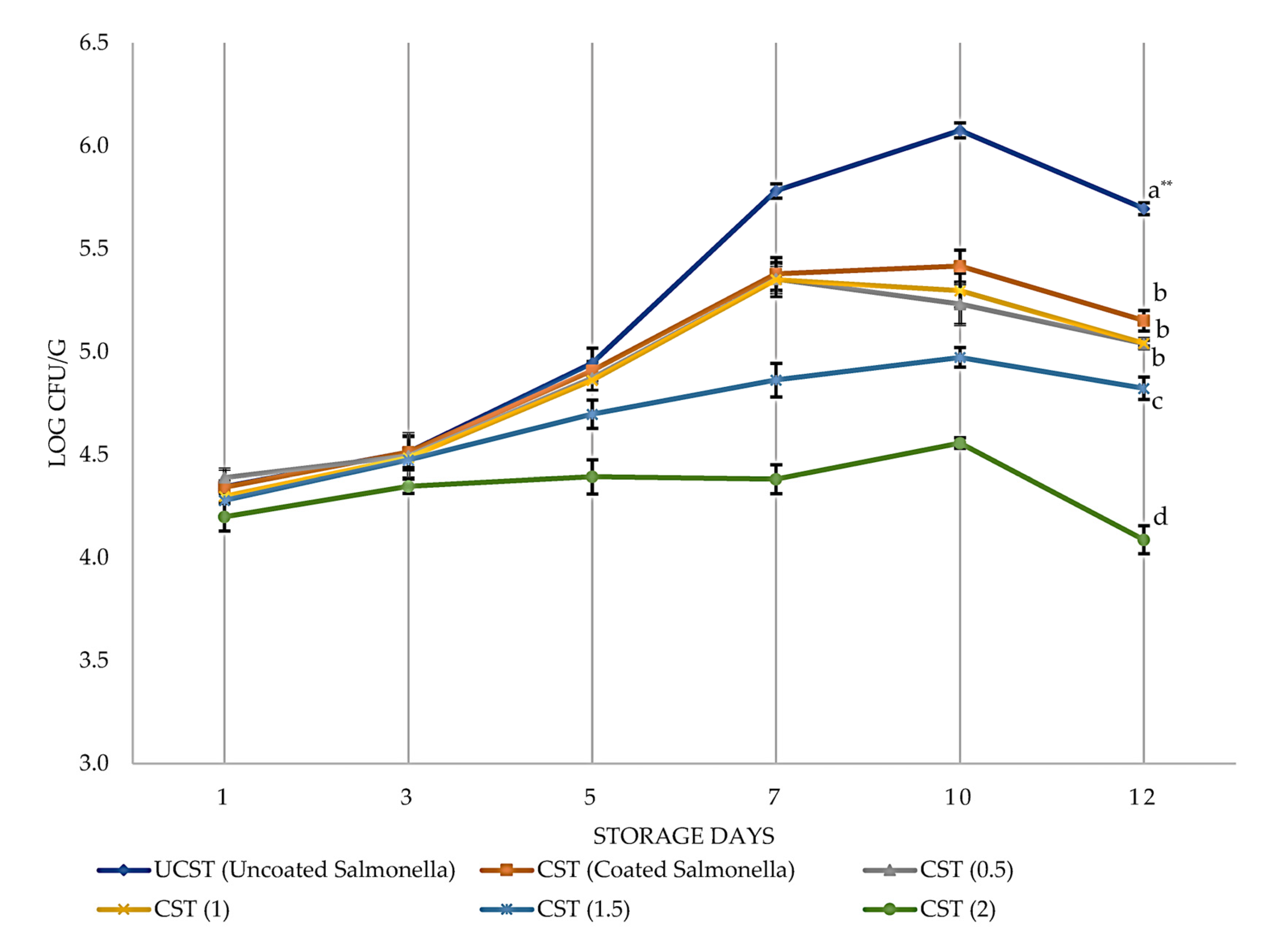
3.1. Antimicrobial Effects of PVR Edible Coating on Chicken Breast Fillets
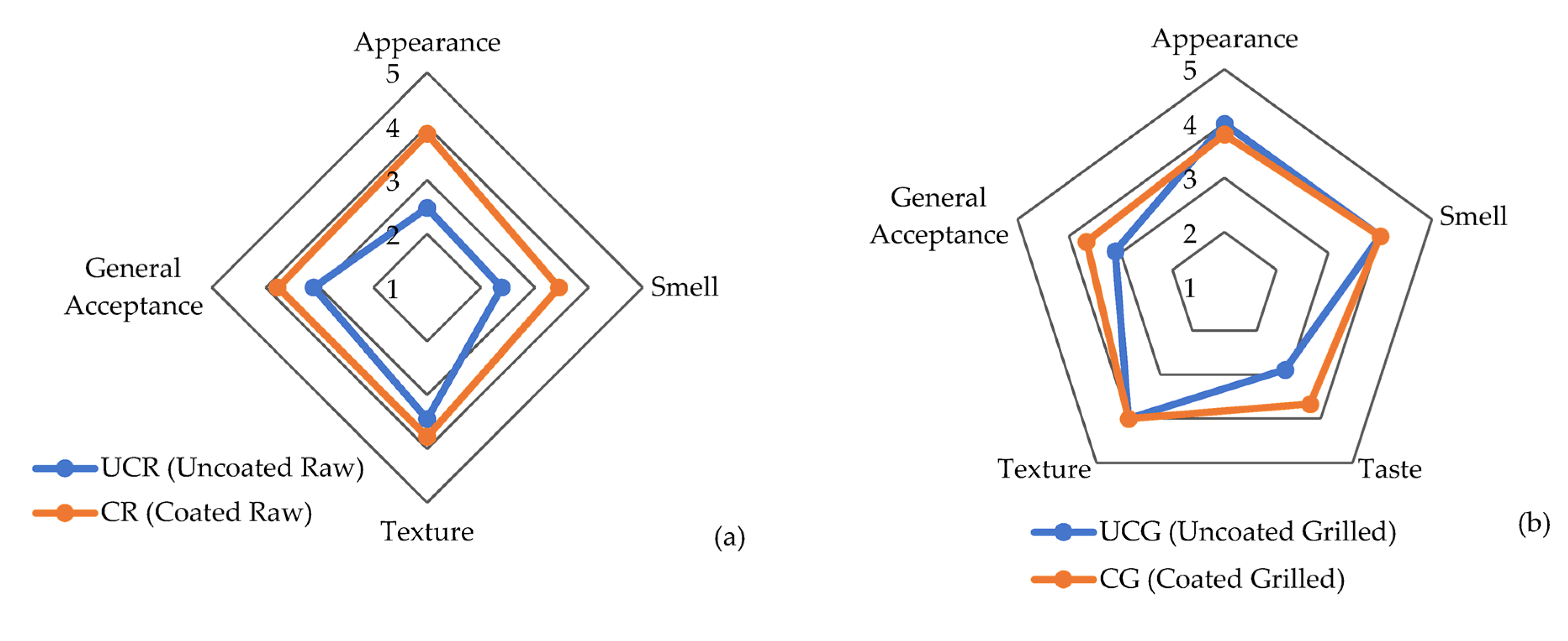
3.2. Sensory Analysis of Chicken Breast Fillets with PVR Edible Coating
4. Conclusions
5. Patents
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sánchez-Ortega, I.; García-Almendárez, B.E.; Santos-López, E.M.; Amaro-Reyes, A.; Barboza-Corona, J.E.; Regalado, C. Antimicrobial Edible Films and Coatings for Meat and Meat Products Preservation. Sci. World J. 2014, 2014, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nychas, G.J.E.; Skandamis, P.N.; Tassou, C.C.; Koutsoumanis, K.P. Meat spoilage during distribution. Meat Sci. 2008, 78, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Pan, I.; Carrión-Granda, X.; Maté, J.I. Antimicrobial efficiency of edible coatings on the preservation of chicken breast fillets. Food Control 2014, 36, 69–75. [Google Scholar] [CrossRef]
- Garavito, J.; Moncayo-Martínez, D.; Castellanos, D.A. Evaluation of antimicrobial coatings on preservation and shelf life of fresh chicken breast fillets under cold storage. Foods 2020, 9, 1203. [Google Scholar] [CrossRef] [PubMed]
- de Moraes Pinto, L.A.; Frizzo, A.; Benito, C.E.; da Silva Júnior, R.C.; Alvares, L.K.; Pinto, A.N.; Tellini, C.; de Oliveira Monteschio, J.; Fernandes, J.I.M. Effect of an antimicrobial photoinactivation approach based on a blend of curcumin and Origanum essential oils on the quality attributes of chilled chicken breast. LWT 2023, 176. [Google Scholar] [CrossRef]
- Tack, D.M.; Marder, E.P.; Griffin, P.M.; Cieslak, P.R.; Dunn, J.; Hurd, S.; Scallan, E.; Lathrop, S.; Muse, A.; Ryan, P.; et al. Preliminary incidence and trends of infections with pathogens transmitted commonly through food—Foodborne Diseases Active Surveillance Network, 10 U.S. sites, 2015–2018. Am. J. Transplant. 2019, 19, 1859–1863. [Google Scholar] [CrossRef] [Green Version]
- ECDC. Surveillance Atlas of Infectious Disease. Available online: https://atlas.ecdc.europa.eu/public/index.aspx (accessed on 19 May 2023).
- Silva, F.; Domingues, F.C.; Nerín, C. Trends in microbial control techniques for poultry products. Crit. Rev. Food Sci. Nutr. 2017, 58, 591–609. [Google Scholar] [CrossRef]
- Fernández-Pan, I.; Mendoza, M.; Maté, J.I. Whey protein isolate edible films with essential oils incorporated to improve the microbial quality of poultry. J. Sci. Food Agric. 2013, 93, 2986–2994. [Google Scholar] [CrossRef]
- Pérez-Santaescolástica, C.; Munekata, P.E.S.; Feng, X.; Liu, Y.; Bastianello Campagnol, P.C.; Lorenzo, J.M. Active edible coatings and films with Mediterranean herbs to improve food shelf-life. Crit. Rev. Food Sci. Nutr. 2022, 62, 2391–2403. [Google Scholar] [CrossRef]
- Abbasi, Z.; Aminzare, M.; Hassanzad Azar, H.; Rostamizadeh, K. Effect of corn starch coating incorporated with nanoemulsion of Zataria multiflora essential oil fortified with cinnamaldehyde on microbial quality of fresh chicken meat and fate of inoculated Listeria monocytogenes. J. Food Sci. Technol. 2021, 58, 2677–2687. [Google Scholar] [CrossRef]
- Janjarasskul, T.; Krochta, J.M. Edible Packaging Materials. Annu. Rev. Food Sci. Technol. 2010, 1, 415–448. [Google Scholar] [CrossRef] [PubMed]
- Bharti, S.K.; Pathak, V.; Alam, T.; Arya, A.; Singh, V.K.; Verma, A.K.; Rajkumar, V. Materialization of novel composite bio-based active edible film functionalized with essential oils on antimicrobial and antioxidative aspect of chicken nuggets during extended storage. J. Food Sci. 2020, 85, 2857–2865. [Google Scholar] [CrossRef] [PubMed]
- Debeaufort, F.; Quezada-Gallo, J.-A.; Voilley, A. Edible Films and Coatings: Tomorrow’s Packagings: A Review. Crit. Rev. Food Sci. Nutr. 1998, 38, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Bourtoom, T. Edible films and coatings: Characteristics and properties. Int. Food Res. J. 2008, 15, 237–248. [Google Scholar]
- Cagri, A.; Ustunol, Z.; Ryser, E.T. Inhibition of three pathogens on bologna and summer sausage using antimicrobial edible films. J. Food Sci. 2002, 67, 2317–2324. [Google Scholar] [CrossRef]
- Falguera, V.; Quintero, J.P.; Jiménez, A.; Muñoz, J.A.; Ibarz, A. Edible films and coatings: Structures, active functions and trends in their use. Trends Food Sci. Technol. 2011, 22, 292–303. [Google Scholar] [CrossRef]
- Guilbert, S.; Gontard, N.; Gorris, L.G.M. Prolongation of the Shelf-life of Perishable Food Products Using Biodegradable Films and Coatings. LWT Food Sci. Technol. 1996, 29, 10–17. [Google Scholar] [CrossRef]
- Han, J.H. Edible Films and Coatings: A Review. In Innovations in Food Packaging: Second Edition; Taylor, S.L., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 213–255. ISBN 9780123946010. [Google Scholar]
- Cagri, A.; Ustunol, Z.; Ryser, E.T. Antimicrobial Edible Films and Coatings. J. Food Prot. 2004, 67, 833–848. [Google Scholar] [CrossRef]
- Campos, C.A.; Gerschenson, L.N.; Flores, S.K. Development of Edible Films and Coatings with Antimicrobial Activity. Food Bioprocess. Technol. 2011, 4, 849–875. [Google Scholar] [CrossRef]
- Abdollahzadeh, E.; Nematollahi, A.; Hosseini, H. Composition of antimicrobial edible films and methods for assessing their antimicrobial activity: A review. Trends Food Sci. Technol. 2021, 110, 291–303. [Google Scholar] [CrossRef]
- Khaledian, Y.; Pajohi-Alamoti, M.; Bazargani-Gilani, B. Development of cellulose nanofibers coating incorporated with ginger essential oil and citric acid to extend the shelf life of ready-to-cook barbecue chicken. J. Food Process. Preserv. 2019, 43, 1–13. [Google Scholar] [CrossRef]
- Khezrian, A.; Shahbazi, Y. Application of nanocompostie chitosan and carboxymethyl cellulose films containing natural preservative compounds in minced camel’s meat. Int. J. Biol. Macromol. 2018, 106, 1146–1158. [Google Scholar] [CrossRef] [PubMed]
- Sallak, N.; Motallebi Moghanjoughi, A.; Ataee, M.; Anvar, S.A.A.; Golestan, L. Evaluation of the effect of corn starch film composed of Ag-TiO2 nanocomposites and Satureja khuzestanica essential oi on the shelf-life of chicken fillet. Food Health 2022, 5, 20–29. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, A.; Wang, W.; Ye, R.; Liu, Y.; Xiao, J.; Wang, K. Characterisation of microemulsion nanofilms based on Tilapia fish skin gelatine and ZnO nanoparticles incorporated with ginger essential oil: Meat packaging application. Int. J. Food Sci. Technol. 2017, 52, 1670–1679. [Google Scholar] [CrossRef]
- Ghalem, B.R.; Mohamed, B. Antimicrobial activity evaluation of the oleoresin oil of Pistacia vera L. Afr. J. Plant Sci. 2010, 4, 300–303. [Google Scholar]
- Digrak, M.; Alma, M.H.; Ilçim, A.; Sen, S. Antibacterial and Antifungal Effects of Various Commercial Plant Extracts. Pharm. Biol. 1999, 37, 216–220. [Google Scholar] [CrossRef]
- Digrak, M.; Alma, M.H.; Ilçim, A. Antibacterial and Antifungal Activities of Turkish Medicinal Plants. Pharm. Biol. 2001, 39, 346–350. [Google Scholar] [CrossRef]
- Alma, M.H.; Nitz, S.; Kollmannsberger, H.; Digrak, M.; Efe, F.T.; Yilmaz, N. Chemical composition and antimicrobial activity of the essential oils from the gum of Turkish Pistachio (Pistacia vera L.). J. Agric. Food Chem. 2004, 52, 3911–3914. [Google Scholar] [CrossRef]
- Erkmen, O. Bacterial inactivation mechanism of SC-CD and TEO combinations in watermelon and melon juices. Food Sci. Technol. 2021, 42. [Google Scholar] [CrossRef]
- Tornuk, F.; Ozturk, I.; Sagdic, O.; Yilmaz, A.; Erkmen, O. Application of Predictive Inactivation Models to Evaluate Survival of Staphylococcus aureus in Fresh-Cut Apples Treated with Different Plant Hydrosols. Int. J. Food Prop. 2013, 17, 587–598. [Google Scholar] [CrossRef] [Green Version]
- Erkmen, O. Practice 10. Counting of mesophilic and thermophilic sporeformers. In Microbiological Analysis of Foods and Food Processing Environments; Elsevier: London, UK, 2021; pp. 77–90. ISBN 978-0-323-91651-6. [Google Scholar]
- Erkmen, O. Practice 15. Isolation and counting of Salmonella. In Microbiological Analysis of Foods and Food Processing Environments; Elsevier: London, UK, 2021; pp. 151–168. ISBN 978-0-323-91651-6. [Google Scholar]
- Erkmen, O. Practice 16. Isolation and counting of Listeria monocytogenes. In Microbiological Analysis of Foods and Food Processing Environments; Elsevier: London, UK, 2021; pp. 169–180. ISBN 978-0-323-91651-6. [Google Scholar]
- Watts, B.M.; Ylimaki, G.; Jeffery, L.H.; Elias, L. Basic Sensory Methods for Food Evaluation; International Development Research Centre: Ottawa, ON, Canada, 1989; ISBN 0-88936-563-6. [Google Scholar]
- Yantis, J.E. ASTM International. The Role of Sensory Analysis in Quality Control; ASTM: Philadelphia, PA, USA, 1992; ISBN 0803114869. [Google Scholar]
- Lawless, H.T.; Heymann, H. Discrimination Testing. In Sensory Evaluation of Food Principles and Practices; Springer: New York, NY, USA, 2010; pp. 79–100. [Google Scholar]
- Economou, T.; Pournis, N.; Ntzimani, A.; Savvaidis, I.N. Nisin–EDTA treatments and modified atmosphere packaging to increase fresh chicken meat shelf-life. Food Chem. 2009, 114, 1470–1476. [Google Scholar] [CrossRef]
- Bazargani-Gilani, B.; Aliakbarlu, J.; Tajik, H. Effect of pomegranate juice dipping and chitosan coating enriched with Zataria multiflora Boiss essential oil on the shelf-life of chicken meat during refrigerated storage. Innov. Food Sci. Emerg. Technol. 2015, 29, 280–287. [Google Scholar] [CrossRef]
- Petrou, S.; Tsiraki, M.; Giatrakou, V.; Savvaidis, I.N. Chitosan dipping or oregano oil treatments, singly or combined on modified atmosphere packaged chicken breast meat. Int. J. Food Microbiol. 2012, 156, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Bolumar, T.; Andersen, M.L.; Orlien, V. Antioxidant active packaging for chicken meat processed by high pressure treatment. Food Chem. 2011, 129, 1406–1412. [Google Scholar] [CrossRef]
- Soni, A.; Kandeepan, G.; Mendiratta, S.K.; Shukla, V.; Kumar, A. Development and characterization of essential oils incorporated carrageenan based edible film for packaging of chicken patties. Nutr. Food Sci. 2016, 46, 82–95. [Google Scholar] [CrossRef]
- Shekarforoush, S.S.; Basiri, S.; Ebrahimnejad, H.; Hosseinzadeh, S. Effect of chitosan on spoilage bacteria, Escherichia coli and Listeria monocytogenes in cured chicken meat. Int. J. Biol. Macromol. 2015, 76, 303–309. [Google Scholar] [CrossRef]
- Patsias, A.; Badeka, A.V.; Savvaidis, I.N.; Kontominas, M.G. Combined effect of freeze chilling and MAP on quality parameters of raw chicken fillets. Food Microbiol. 2008, 25, 575–581. [Google Scholar] [CrossRef]
- Senter, S.D.; Arnold, J.W.; Chew, V. APC values and volatile compounds formed in commercially processed, raw chicken parts during storage at 4 and 13 °C and under simulated temperature abuse conditions. J. Sci. Food Agric. 2000, 80, 1559–1564. [Google Scholar] [CrossRef]
- FAO. General hygiene principles for meat handling. In Guidelines for Slaughtering, Meat Cutting and Further Processing; Heinz, G., Ed.; Food and Agriculture Organization of the United Nations: Rome, Italy, 1991; pp. 1–26. ISBN 92-5-102921-0. [Google Scholar]
- Nouri Ala, M.A.; Shahbazi, Y. The effects of novel bioactive carboxymethyl cellulose coatings on food-borne pathogenic bacteria and shelf life extension of fresh and sauced chicken breast fillets. LWT 2019, 111, 602–611. [Google Scholar] [CrossRef]
- Matiacevich, S.; Acevedo, N.; López, D. Characterization of Edible Active Coating Based on Alginate-Thyme Oil-Propionic Acid for the Preservation of Fresh Chicken Breast Fillets. J. Food Process. Preserv. 2015, 39, 2792–2801. [Google Scholar] [CrossRef]
- Baldwin, E.A.; Hagenmaier, R.D.; Bai, J. (Eds.) Edible Coatings and Films to Improve Food Quality, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012; ISBN 9781138198937. [Google Scholar]
- Khezerlou, A.; Zolfaghari, H.; Banihashemi, S.A.; Forghani, S.; Ehsani, A. Plant gums as the functional compounds for edible films and coatings in the food industry: A review. Polym. Adv. Technol. 2021, 32, 2306–2326. [Google Scholar] [CrossRef]
- Raeisi, M.; Tabaraei, A.; Hashemi, M.; Behnampour, N. Effect of sodium alginate coating incorporated with nisin, Cinnamomum zeylanicum, and rosemary essential oils on microbial quality of chicken meat and fate of Listeria monocytogenes during refrigeration. Int. J. Food Microbiol. 2016, 238, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Noori, S.; Zeynali, F.; Almasi, H. Antimicrobial and antioxidant efficiency of nanoemulsion-based edible coating containing ginger (Zingiber officinale) essential oil and its effect on safety and quality attributes of chicken breast fillets. Food Control 2018, 84, 312–320. [Google Scholar] [CrossRef]
- Aykut, B.; Osman, E. Antimicrobial activity of a novel biodegradable edible film produced from Pistacia vera resin and Origanum vulgare essential oil. Res. J. Biotechnol. 2017, 12, 15–21. [Google Scholar]
- Souza, V.G.L.; Pires, J.R.A.; Vieira, É.T.; Coelhoso, I.M.; Duarte, M.P.; Fernando, A.L. Shelf Life Assessment of Fresh Poultry Meat Packaged in Novel Bionanocomposite of Chitosan/Montmorillonite Incorporated with Ginger Essential Oil. Coatings 2018, 8, 177. [Google Scholar] [CrossRef] [Green Version]
- Janes, M.E.; Kooshesh, S.; Johnson, M.G. Control of Listeria monocytogenes on the surface of refrigerated, ready-to-eat chicken coated with edible zein film coatings containing nisin and/or calcium propionate. J. Food Sci. 2002, 67, 2754–2757. [Google Scholar] [CrossRef]
- İncili, G.K.; Akgöl, M.; Aydemir, M.E.; Alan, S.; Mutlu, M.; İlhak, O.İ.; Öksüztepe, G. Fate of Listeria monocytogenes and Salmonella Typhimurium in homemade marinade and on marinated chicken drumsticks, wings and breast meat. LWT 2020, 134. [Google Scholar] [CrossRef]
- Goswami, N.; Han, J.H.; Holley, R.A. Effectiveness of antimicrobial starch coating containing thyme oil against Salmonella, Listeria, Campylobacter, and Pseudomonas on chicken breast meat. Food Sci. Biotechnol. 2009, 18, 425–431. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Panahi, Z.; Mohsenzadeh, M. Sodium alginate edible coating containing Ferulago angulata (Schlecht.) Boiss essential oil, nisin, and NaCl: Its impact on microbial, chemical, and sensorial properties of refrigerated chicken breast. Int. J. Food Microbiol. 2022, 380, 109883. [Google Scholar] [CrossRef]
- Yousefi, M.; Farshidi, M.; Ehsani, A. Effects of lactoperoxidase system-alginate coating on chemical, microbial, and sensory properties of chicken breast fillets during cold storage. J. Food Saf. 2018, 38. [Google Scholar] [CrossRef]


| Type of CBF | Edible Coating Application | Contamination of Pathogens | Microbial Count |
|---|---|---|---|
| C1 | Uncoated CBF | No contamination | TBC |
| C2 | Coating CBF with EC | ||
| C2(0.5) *, C2(1), C2(1.5), C2(2) | Coating CBF with ECEO | ||
| UCLM | Uncoated CBF | L. monocytogenes contamination | L. monocytogenes count |
| CLM | Coating CBF with EC | ||
| CLM(0.5), CLM(1) CLM(1.5), CLM(2) | Coating CBF with ECEO | ||
| UCST | Uncoated CST | S. Typhimurium contamination | S. Typhimurium count |
| CST | Coating CBF with EC | ||
| CST(0.5), CST(1), CST(1.5), CST(2) | Coating CBF with ECEO |
| Name of the Sample | Treatment | Sensory Analysis |
|---|---|---|
| UCR | Uncoated raw CBF | Appearance, smell, texture, general acceptance, and taste (only for grilled samples) |
| CR | Coating raw CBF with ECEO (containing 2% EO) | |
| UCG | Uncoated CBF grilled for 5 min | |
| CG | Coating CBF with ECEO (2%) + grilled for 5 min |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barazi, A.Ö.; Mehmetoğlu, A.Ç.; Erkmen, O. A Novel Edible Coating Produced from a Wheat Gluten, Pistacia vera L. Resin, and Essential Oil Blend: Antimicrobial Effects and Sensory Properties on Chicken Breast Fillets. Foods 2023, 12, 2276. https://doi.org/10.3390/foods12122276
Barazi AÖ, Mehmetoğlu AÇ, Erkmen O. A Novel Edible Coating Produced from a Wheat Gluten, Pistacia vera L. Resin, and Essential Oil Blend: Antimicrobial Effects and Sensory Properties on Chicken Breast Fillets. Foods. 2023; 12(12):2276. https://doi.org/10.3390/foods12122276
Chicago/Turabian StyleBarazi, Aykut Önder, Arzu Çağrı Mehmetoğlu, and Osman Erkmen. 2023. "A Novel Edible Coating Produced from a Wheat Gluten, Pistacia vera L. Resin, and Essential Oil Blend: Antimicrobial Effects and Sensory Properties on Chicken Breast Fillets" Foods 12, no. 12: 2276. https://doi.org/10.3390/foods12122276
APA StyleBarazi, A. Ö., Mehmetoğlu, A. Ç., & Erkmen, O. (2023). A Novel Edible Coating Produced from a Wheat Gluten, Pistacia vera L. Resin, and Essential Oil Blend: Antimicrobial Effects and Sensory Properties on Chicken Breast Fillets. Foods, 12(12), 2276. https://doi.org/10.3390/foods12122276






