1. Introduction
Hippophae rhamnoides L., commonly known as sea buckthorn or seaberry, boasts a widespread distribution across Asia, Europe, and North America [
1]. It belongs to the
Elaeagnaceae family and has been traditionally used in a wide range of fields, such as foods, pharmaceuticals, and cosmetics [
2]. The diverse components of sea buckthorn offer a plethora of bioactive compounds, making them prized for medicinal and nutritional applications [
3,
4]. Of noteworthy importance, sea buckthorn seed oil has been subject to extensive investigation due to its exceptional properties, including wound healing, antioxidant, anti-inflammatory, anticancer, antimicrobial, and emollient activities [
5,
6,
7]. Nevertheless, the residues left behind after sea buckthorn seed oil extraction are often discarded or underutilized. However, recent research has highlighted the presence of valuable natural compounds, including flavones, polyphenols, and unsaturated fatty acids, in sea buckthorn seed residues (SBSR) [
8,
9,
10,
11].
Despite the promising potential of SBSR for various applications, a comprehensive exploration of its practical utility in raw material development is lacking. Furthermore, the choice of extraction methods can lead to variations in the composition and efficacy of the resulting extracts. Commonly used solvents for SBSR extraction include water and ethanol. Previous studies indicated that aqueous SBSR extracts exhibit hypoglycemic and hypolipidemic effects in type 2 diabetic rats induced with streptozotocin and a high-fat diet [
12]. In addition, investigations into different concentrations of organic solvents for sea buckthorn seed extraction have demonstrated varying antioxidative capacities, with ethyl acetate extracts displaying the highest and isopropyl extracts displaying the lowest antioxidative potential [
13]. It is also worth noting that SBSR retains a significant amount of liposoluble substances even post-oil recovery. Advanced extraction methods, such as supercritical carbon dioxide, pressurized ethanol, and enzyme-assisted extraction, have been used to isolate valuable components, including tocopherols and monosaccharides, from sea buckthorn pomace and seeds [
14].
Numerous in vitro studies have extensively validated the antioxidant capacities of SBSR. These investigations encompass a range of assays, including the estimation of reactive oxygen species generation, measurement of enzymatic/non-enzymatic antioxidant activities/levels, evaluation of peroxisome proliferator-activated receptors levels, and assessment of 3-ethylbenzothiazoline-6-sulfonic acid (ABTS) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activities [
1,
7,
15]. However, under conditions of oxidative stress, the body initiates a protective stress response to counteract free radicals. This response entails not only direct free radical elimination but also augmentation of antioxidant enzyme activity (e.g., glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and catalase (CAT)) along with a reduction in the accumulation of peroxidation products like malondialdehyde (MDA) and lipofuscin (LP), thus conferring a protective role [
16,
17,
18]. While existing research on SBSR has predominantly focused on a single level of impact, a comprehensive investigation utilizing animal experiments is needed to elucidate the mechanisms by which HYD-SBSR functions within the body. Therefore, a thorough exploration of the antioxidative mechanism of SBSR is warranted.
In recent years, flow cytometry has undergone significant advancements, enabling the identification of multiple phenotypic subsets, the selection of individual cells, and even cell isolation using sorting. This technology plays a pivotal role in clinical settings by identifying aberrant cells, quantifying excessive or reduced populations of specific cells, and monitoring changes in tracked cell populations [
19]. In this study, in conjunction with mouse experiments, the levels of antioxidant enzymes and peroxides were also analyzed in vivo. A proteomics analysis was conducted to scrutinize changes in differentially expressed proteins (DEPs) post-HYD-SBSR administration. Subsequent Gene Ontology (GO) analysis and Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis were performed to provide further insights into the impact of DEPs.
B16F10 cells, a melanoma cell line derived from mice, possess high metabolic activity leading to reactive oxygen species (ROS) production, potential stress response activation, rapid growth facilitating timely observations, and relevance to simulating disease-related conditions [
20,
21]. These attributes collectively position B16F10 cells as a valuable model for investigating oxidative stress and cellular responses. The D-galactose-induced mouse aging model offers simplicity and ease of operation, enabling rapid inter-group comparisons. It has been demonstrated as an effective simulation of aging effects and finds wide application in antioxidant research [
22,
23].
Prior research has demonstrated that sea buckthorn seed products can contribute to reducing blood glucose and lipid levels in obese mice [
12,
24]. Using them as supplementary food additives holds the potential to regulate lipid metabolism in the body. This approach could also mitigate certain suboptimal health conditions arising from high-sugar diets. The primary objective of this study was to assess the effects of the hydroalcoholic extract from sea buckthorn (
Hippophae rhamnoides L.) seed residues (HYD-SBSR) at the cellular and physiological levels while establishing a correlation between in vitro and in vivo experimental findings. This not only supports the broader application of sea buckthorn seed by-products in food but also addresses environmental concerns linked to sea buckthorn production, thereby contributing to sustainable and eco-friendly industrial development.
2. Materials and Methods
2.1. Preparation of HYD-SBSR
HYD-SBSR was prepared following the methodology outlined in [
25]. Ground powder of sea buckthorn seed residues (provided by Qinghai Kompu Biotechnology Co., Ltd., Xining, China, dried, crushed, and sifted) was mixed with an 80% ethanol aqueous solution at a ratio of 8:1 (liquid to solid ratio, mL/g). The mixture was then extracted for 1.5 h. After extraction, centrifugation was carried out at 400×
g and 4 °C for 15 min. The resulting supernatant was collected, and the extraction process was repeated twice to enhance the yield and purity of the extracted components. The collected supernatants from each extraction were combined to create a consolidated sample. The ethanol in the sample was subsequently removed using rotary evaporation under vacuum conditions at a controlled temperature of 40 °C. This evaporation process facilitated solvent removal, concentration of the extracted substances, and conversion into a fine powder. The collected HYD-SBSR was stored securely for future investigations. The flavonoid, procyanidin, and total phenolic contents reached up to 354.00 ± 21.00 mg RE per g DW, 319.31 ± 11.70 mg CE per g DW, and 271.24 ± 91.30 mg GAE per g DW, respectively [
25].
2.2. Cell Culture
B16F10 cells (obtained from the Cell Resource Center, Institute of Basic Medicine, Chinese Academy of Medical Sciences) were cultured in Dulbecco’s Modified Eagle Medium (DMEM, Grand Island Biological Company, New York, NY, USA), supplemented with 10% heat-inactivated fetal calf serum(FBS, GIBCO, Darmstadt, Germany), and a 1% antibiotic–antimycin solution consisting of 100 units/mL penicillin /streptomycin and 100 U/mL amphotericin (GIBCO, Darmstadt, Germany) at 37 °C in a humidified incubator (Shanghai Shengke, Shanghai, China) under 5% CO2.
2.3. Cell Viability
Cell viability was determined using a dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Sigma, Welwyn Garden City, UK) assay. B16F10 cells were seeded into a 96-well plate at a concentration of 5000 cells per well at 37 °C and were incubated in a cell incubator with 5% CO
2 for 12 h. The cells were treated with H
2O
2 or HYD-SBSR at different concentrations for a period. Details of the concentration values are provided in
Section 2.6 and
Section 2.7.
The MTT assay involved the addition of a 100 μL mixture of MTT solution (5 mg/mL) and DMEM at a volumetric ratio of 1:5. This treatment was carried out for 4 h, followed by the addition of 150 μL of DMSO to dissolve the resulting products. After a 10-minute incubation at 37 °C, the absorbance was measured at a wavelength of 490 nm to determine the results.
Cell viability was calculated following the equation below.
where OD
t represents the experimental group absorbance minus zeroing group absorbance and OD
0 represents the control group absorbance minus zeroing group absorbance.
2.4. Establishment of the H2O2-Induced B16F10 Cell Oxidative Stress Model
A model of acute ROS-induced B16F10 cells was established using a 4-hour treatment with hydrogen peroxide(H2O2).
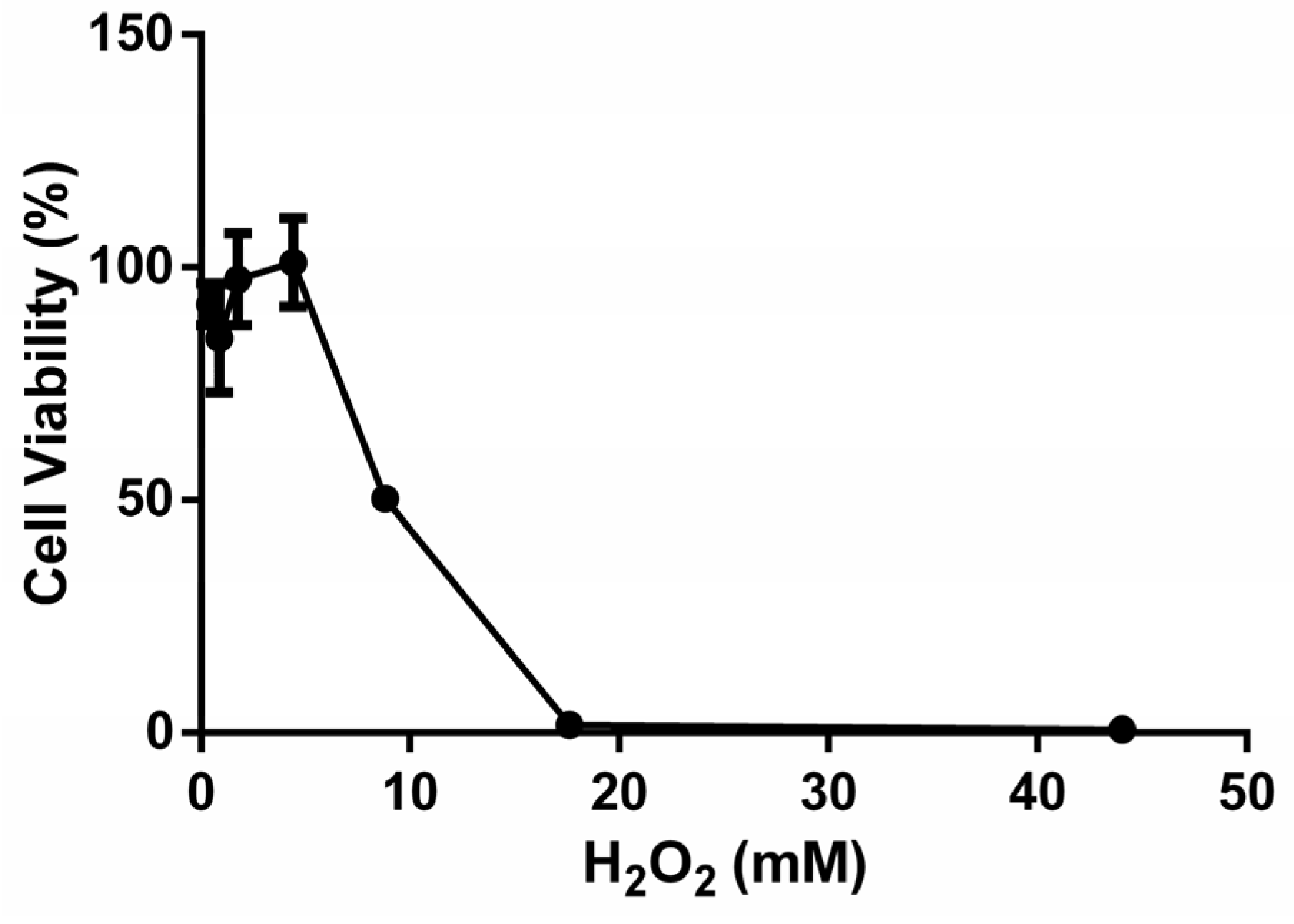
Different concentrations of H2O2 ranging from 0.4 to 44.1 mM were utilized for treatment, each lasting for 4 h. At least 6 parallel wells were used for each group. Cell viability was measured. IC50 parameters were selected to establish the oxidative stress model, striking a balance between eliciting significant effects within a reasonable timeframe and ensuring reproducibility.
2.5. Protective and Repair Effects of HYD-SBSR on the H2O2-Induced B16F10 Cell Oxidative Stress Model
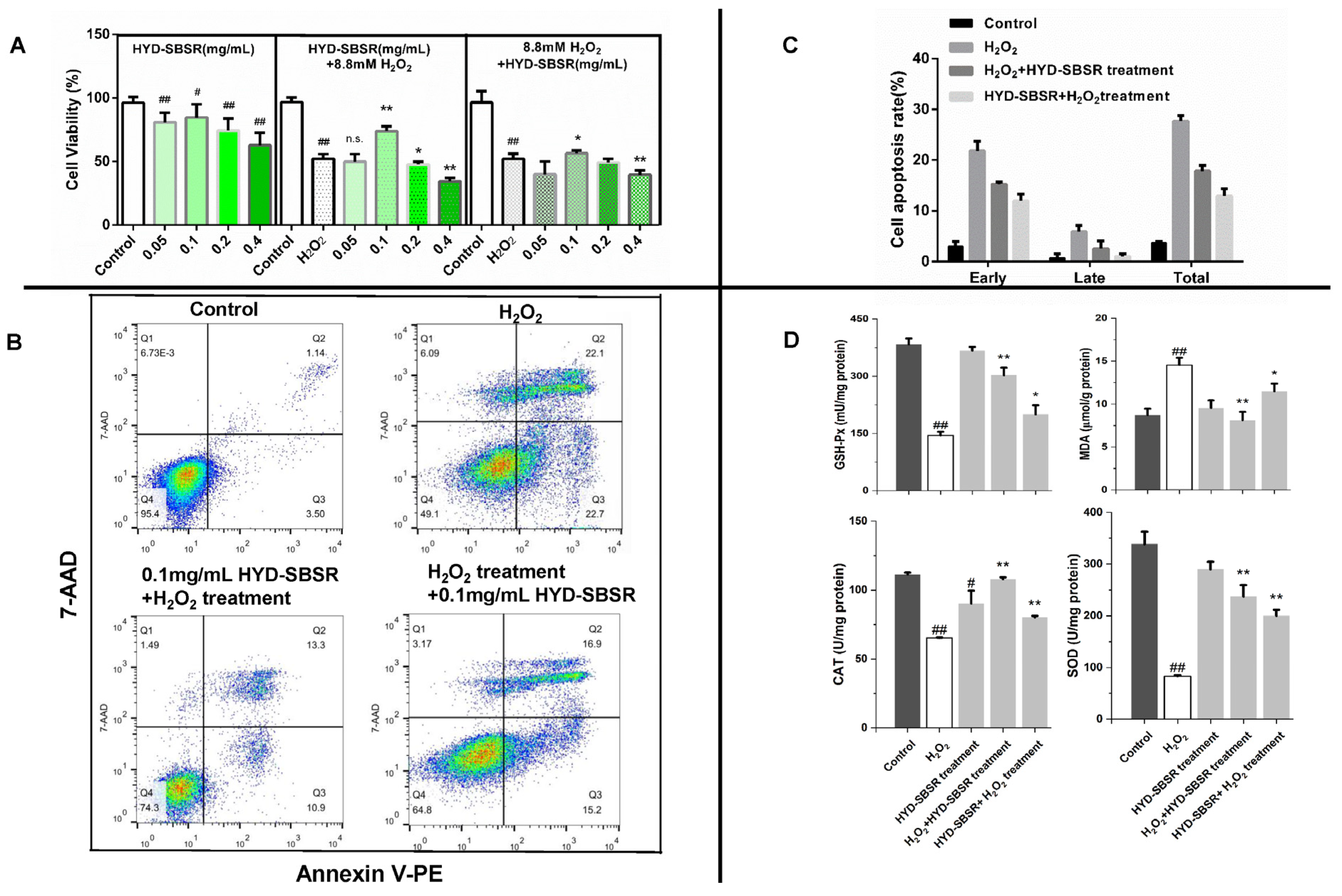
To explore the protective effects of HYD-SBSR on cells, B16F10 cells seeded in a 96-well plate were treated with 0.05, 0.1, 0.2, and 0.4 mg/mL of HYD-SBSR for 24 h, cleaned twice with PBS, and treated with 8.8 mM H2O2 for 4 h. Cell viabilities were detected using an MTT assay.
Additionally, the repair effects of HYD-SBSR were investigated. B16F10 cells were first exposed to H2O2 for 4 h to establish the oxidative stress model. Subsequently, the cells were treated with different concentrations of HYD-SBSR for 24 h. Cell viability was assessed using an MTT assay.
2.6. GSH-Px, CAT, SOD, and MDA in B16F10 Cells
The experimental procedure involved the treatment of B16F10 cells with H2O2 (8.8 mM) for a duration of 4 h to establish the H2O2-induced cell oxidative stress model. Subsequently, the cells were subjected to treatment with varying concentrations of HYD-SBSR (0, 0.05, 0.10, 0.20, and 0.40 mg/mL) for a period of 24 h. The group of cells without any HYD-SBSR treatment served as the model group for comparison.
Cells at a density of 1 × 105 were planted in a six-well plate and incubated overnight. Cells were treated with HYD-SBSR (0, 0.05, 0.10, 0.20, and 0.40 mg/mL) for 24 h and then treated with H2O2 (8.8 mM) for 4 h. Only cells treated with HYD-SBSR were used as the HYD-SBSR control. Cultured cells were washed with PBS. The cells were lysed using 200 µL Western and IP cell lysis buffer (Beyotime, Nanjing, China), followed by a 12,000× g centrifugation for 10 min to collect the supernatant. The contents of GSH-Px, CAT, SOD, and MDA were then determined in a 96-well plate (Corning, Corning, NY, USA), according to the instructions of the GSH-Px, CAT, SOD, and MDA detection kit (Beyotime, Nanjing, China). Protein content was measured using a BCA protein kit from Beyotime. The contents of these parameters were calibrated using the protein content and expressed as micrograms per milligram of protein.
2.7. Flow Cytometry Analysis for Cell Cycle Distribution and Apoptosis
Annexin V-PE is a fluorescently labeled protein that binds to calcium-dependent phospholipids with a strong affinity for phosphatidylserine (PS) binding sites, similar to Annexin V-FITC. During the early stages of cell apoptosis, the loss of membrane symmetry exposes PS on the outer surface of the cell membrane. In both the early and late stages of apoptosis, PS is present on the outer surface of the cell membrane. Importantly, early apoptotic cells maintain membrane integrity, preventing the entry of 7-AAD. In contrast, late apoptotic cells exhibit compromised membrane integrity and can be co-stained with Annexin V-PE or V-FITC and 7-AAD. This staining method allows for the differentiation and identification of cells at different stages of apoptosis based on variations in phospholipid exposure and membrane integrity [
26,
27].
In this study, cell death was detected and analyzed with flow cytometry using FACS Calibur (Becton Dickinson Biosciences, San Jose, CA, USA).
Seeding was performed with a cell density of 1.5 × 105 cells per well in a 6-well plate, with 3 wells per group. The model group was initially exposed to 8.8 mM H2O2 for 4 h, followed by treatment with 0.1 mg/mL HYD-SBSR for 24 h. Conversely, the HYD-SBSR group underwent the opposite treatment sequence. Then, cells from each group were harvested using trypsin digestion for subsequent analyses. After treatment, approximately 10,000 cells were obtained from each sample. Staining of cell samples was performed using Annexin V-PE and 7-AAD (Annexin V-PE Apoptosis Detection Kit I, BD Bioscience, San Jose, CA, USA). Experimental data were obtained and analyzed using CellQuest (Becton Dickinson Immunocytometry Systems, San Jose, CA, USA). Annexin V-PE and 7-AAD fluorescence (Becton, Dickinson, ND, USA) were used for two-parameter point plots.
Cell populations were separated as follows: viable cells-Annexin V-PE-negative and 7-AAD-negative (Q4, Annexin V-PE-/7-AAD−); early apoptotic cells-Annexin V-PE-positive and 7-AAD-negative (Q3, Annexin V-PE+/7-AAD−); late-apoptotic cells or dead cells- Annexin V-PE-positive and 7-AAD-positive (Q1, Annexin V-PE+/7-AAD+); and cells debris-Annexin V-PE-negative and 7-AAD-positive (Q2, Annexin V-PE-/7-AAD+). The proportion of 4 groups was calculated using FlowJo software (version 10, FlowJo, LLC, Ashland, OR, USA). The total apoptosis rate was calculated by summing the rate of early apoptotic cells and late apoptotic cells.
2.8. Animals and Treatment
A D-galactose-induced aging mice model was established to investigate the protective effect of HYD-SBSR. The experiment was conducted using specific pathogen-free (SPF)-grade Institute of Cancer Research (ICR) male mice (Mus musculus).
Fifty male SPF ICR mice with an average body weight of (23 ± 3) g were ordered from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China) and acclimated for one week. They were then randomly divided into 5 groups (n = 10): control group, model group, HYD-SBSR high-dose group (HYD-SBSR-H), HYD-SBSR medium-dose group (HYD-SBSR-M), and HYD-SBSR low-dose group (HYD-SBSR-L). Each group contained 10 mice.
Except for the control group, the remaining groups were subjected to daily intraperitoneal injections of 10% D-galactose solution (100 mg/kg body weight). Control-group mice received an equivalent volume of 0.9% physiological saline. The low-dose, medium-dose, and high-dose groups were orally administered 100, 300, and 600 mg/(kg body weight) of HYD-SBSR, respectively. The control and model groups were administered an equivalent volume of physiological saline.
The treatment duration lasted 42 days. Throughout the intragastric administration period, the mice were maintained in an environment with a temperature of 22 to 25 °C, relative humidity of 50% to 60%, and a 12-hour light (08:00–20:00) and 12-hour dark cycle under fluorescent illumination. Bedding was changed every 3 days. All mice received a standard diet and had access to water.
No mice experienced mortality over the course of the entire experiment, and all subjects were included in this study. The Guide for the Care and Use of Laboratory Animals (National Institutes of Health, Stapleton, NY, USA) was strictly followed in designing all animal experimental procedures. Ethical approval for all animal experiment procedures was granted by the Experimental Animal Welfare Ethics Committee of Beijing Experimental Animal Research Center (BLARC-2017-A015).
2.9. Collection of Mouse Experimental Samples
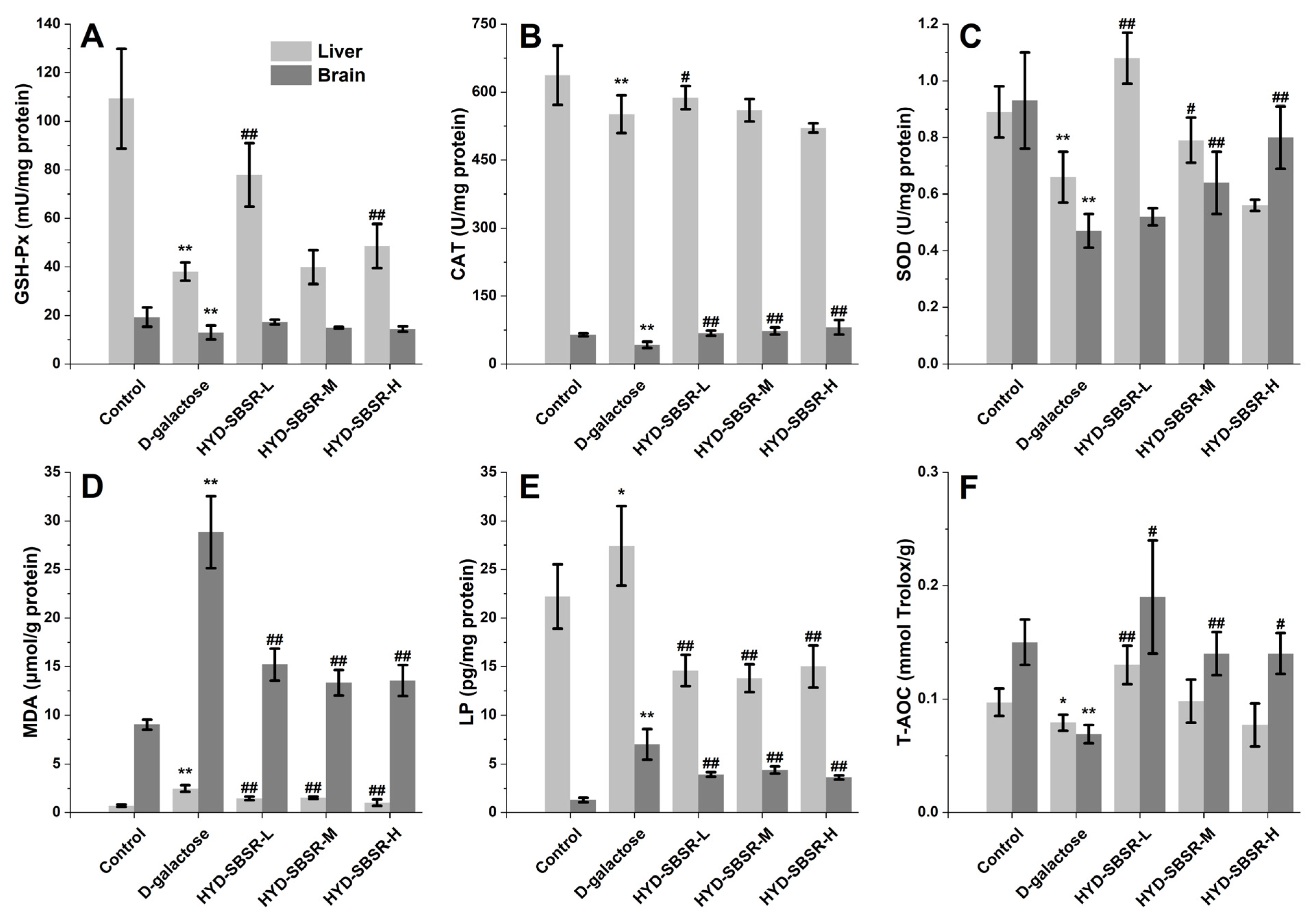
Following euthanization with cervical dislocation, segments of liver and brain tissues were frozen in liquid nitrogen first and then homogenized under an ice bath to prepare 10% homogenate in a 1:9 (w/v) ratio with pre-chilled physiological saline, which was centrifuged at 3 000 r/min and 4 °C for 15 min. The supernatants were removed to determine tissue biochemical indexes (GSH-Px, CAT, SOD, MDA, lipofuscin, and total antioxidant capacity). All indexes were determined following the manufacturer’s instructions.
Additionally, a proteomics assay was conducted on liver samples from the low-dose group, using TMT labeling quantitative proteomics technology (CapitalBio Technology. Beijing, China). The whole process included protein sample preparation, TMT labeling, high-performance liquid chromatography (HPLC) fractionation, liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis, and proteomics data analysis. A detailed protocol for this analysis is provided in the
Supplemental Materials.
2.10. Differentially Expressed Proteins (DEPs) and GO Enrichment and KEGG Pathway Enrichment Analyses
The DEPs satisfied the following conditions: average ratio-fold change >1.1 (up-regulation) and <0.9 (down-regulation) and a p-value < 0.05.
Functional classification of the DEPs was performed according to the Gene Ontology (GO) annotation and enrichment analysis. The Encyclopedia of Genes and Genomes (KEGG) Orthology-Based Annotation System (KOBAS) v2.0 was used. The enrichment analysis of GO function significance unveiled functional categories that exhibited significant enrichment in the pool of differential proteins in comparison with the broader genomic background. This analysis entailed the submission of all differential proteins to the GO database (
http://www.geneontology.org/ Map each term of org/, accessed on 20 July 2023), where the count of proteins for each term was calculated. Subsequently, hypergeometric tests were applied to pinpoint GO entries that exhibited substantial enrichment among the differential proteins, relative to the genome background. After multiple tests and corrections, GO terms with a
p-value of ≤0.05 were considered significantly enriched in the set of differential proteins. These DEPs were then categorized into three primary classifications, namely, biological process (BP), cell component (CC), and molecular function (MF). The KEGG database was used to identify enriched pathways. A two-tailed Fisher’s exact test was used to evaluate the enrichment of DEPs relative to all the identified proteins within specific pathways. A pathway achieving a corrected
p-value of < 0.05 was deemed to be significant. These pathways were subsequently classified into hierarchical categories in accordance with the KEGG website’s structure.
2.11. Statistical Analysis
The experimental data was analyzed using SPSS 19.0 (SPSS, Chicago, IL, USA) and GraphPad Prism 9.0 (GraphPad Software, La Jolla, CA, USA) software. Single factor ANOVA analysis was used for comparisons between groups, and a t-test was used for pairwise comparisons. A p < 0.05 was considered statistically significant, and the results were shown as mean ± standard deviation.
4. Discussion
H
2O
2 is known to readily diffuse into nuclear tissue, leading to the onset of various oxidative stress conditions. Due to this property, exogenous H
2O
2 has often been used in studies to induce oxidative stress damage and apoptosis. This approach helps researchers investigate the cellular protective and repair effects of bioactive substances. Cellular damage often results in a decline in the body’s ability to eliminate H
2O
2, causing an accumulation of ROS and the initiation of lipid peroxidation reactions. In turn, this leads to the formation of products such as MDA, which can damage vital biological molecules like proteins and lipids. To maintain cellular homeostasis, the body relies on its antioxidant enzyme system, which includes key enzymes like GSH-Px, CAT, and SOD. These enzymes play a crucial role in breaking down hydrogen peroxide generated during metabolism and neutralizing ROS and other free radicals that arise during oxidative stress. In this study, an H
2O
2-induced B16F10 model was established to evaluate the protective and repair abilities of HYD-SBSR at the cellular level in vitro. The results indicated that HYD-SBSR exhibited superior protective capabilities, as evidenced by its tendency to enhance cell viability and reduce H
2O
2-induced apoptosis (
Figure 2A–C). However, the cellular antioxidant assays hinted that while HYD-SBSR demonstrated a protective effect, its repair effect showed even greater promise (
Figure 2D). Although the findings did not align completely when comparing the two treatment types, it was evident that HYD-SBSR held the potential to counteract oxidative stress. This aligned with the findings from our laboratory’s analysis of flavonoid, procyanidin, and total phenolic contents present in HYD-SBSR [
25].
To address the limited reports available on the impact of HYD-SBSR in vivo, a more in-depth investigation into its potential repair effects on antioxidant enzymes and peroxides in mice was carried out. The administration of three different levels of HYD-SBSR demonstrated the ability to elevate antioxidant enzyme levels in the liver and brain. These results suggested that the liver, known for its detoxification and metabolism functions, played a crucial role in maintaining cellular homeostasis.
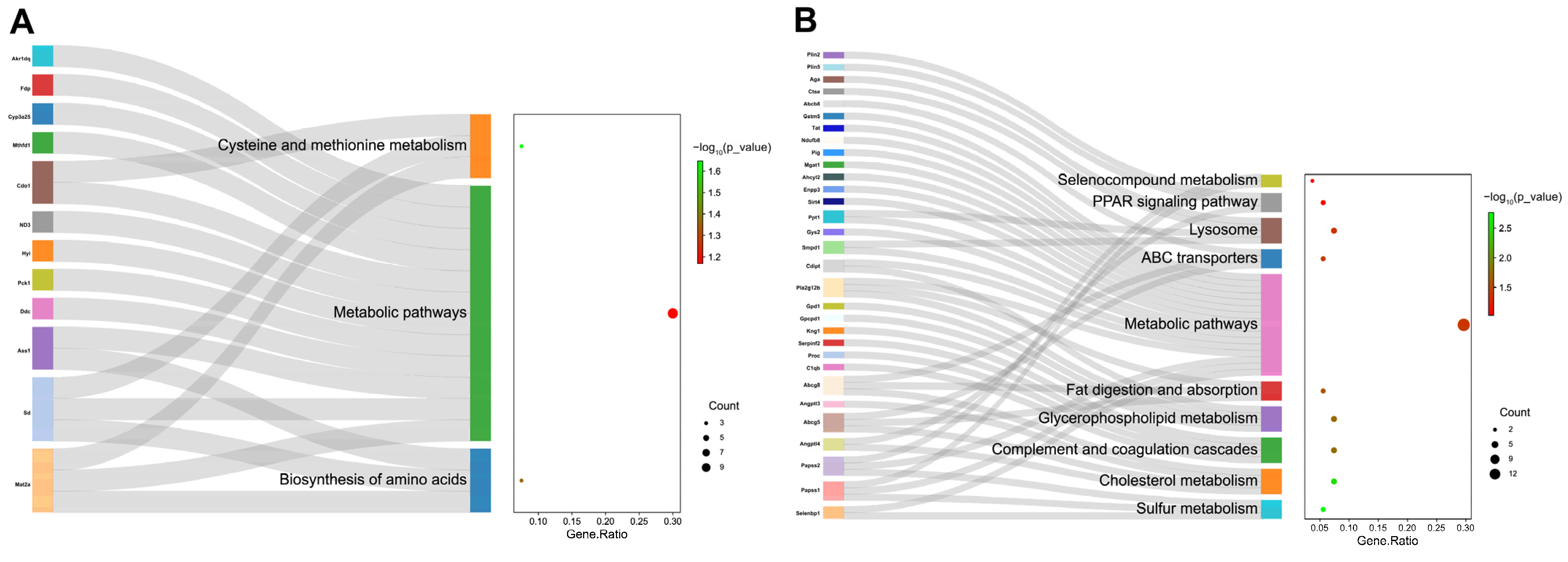
To delve further into the molecular pathways underlying the antioxidant effects of HYD-SBSR in vivo, a proteomics approach was used. Importantly, three DEPs, namely, Eef1e1, Farp1, and Aga, were screened, which were upregulated in the model group and decreased after the treatment of HYD-SBSR. Conversely, one DEP (Pigr) demonstrated the opposite expression pattern.
Of significance among the DEPs is the protein Aga, also referred to as aspartylglucosaminidase, a lysosomal enzyme that starts as an inactive precursor molecule and is swiftly activated within the endoplasmic reticulum [
28]. An Aga deficiency leads to aspartylglycosaminuria, a lysosomal disorder causing impaired glycoprotein degradation [
29]. Remarkably, research by Ulla Dunder et al. illustrated that a 10% increase in Aga activity resulted in a 20% reduction in aspartylglycosaminuria accumulation [
30]. The increased expression of Aga was detected in the model group, probably indicating a self-regulation of the body against disordered glucose metabolism.
Eef1e1, also known as eukaryotic translation elongation factor 1 ε 1, plays a role in protein synthesis and cell differentiation [
31,
32]. It positively modulates the ATM response to DNA damage [
33] and can be induced by DNA-damaging agents like UV, Adriamycin, actinomycin D, and cisplatin. The increased expression of Eef1e1 in the model group is understandable, given its connection to the DNA damage response. The decrease in Eef1e1 protein levels after HYD-SBSR treatment suggested a potential reparative effect.
Moreover, Farp1 was identified as one of the guanine nucleotide exchange factors, which belongs to a family of regulatory proteins for Rho GTPases, influencing various cellular processes [
34]. Farp1 interaction with cell surface proteins regulates neuronal development [
35,
36], and high expression is associated with lymphatic invasion and metastasis [
37]. In the context of D-galactose-induced aging mice with disrupted glucose metabolism and ROS accumulation, upregulated Farp1 likely combats oxidative stress and inflammation in the model group. Conversely, the decreased Farp1 expression upon HYD-SBSR administration implies a beneficial effect.
Pigr (polymeric immunoglobulin receptor precursor) is a single transmembrane protein. Its expression was subsequently upregulated after HYD-SBSR treatment. The impact on the MEK/ERK pathway suggests potential attenuation of liver injury in mice [
38].
Regarding the GO and KEGG pathway analyses, our findings revealed that HYD-SBSR significantly influences GO terms related to mitochondria, lipid storage, triglyceride homeostasis, ATP-binding cassette (ABC) transporter complex, and more.
Enriched KEGG pathways in the HYD-SBSR group compared with the model group encompass the PPAR signaling pathway, fat digestion and absorption, glycerophospholipid metabolism, and cholesterol metabolism. These pathways are closely associated with the body’s antioxidant status and changes in MDA and LP indicators. PPARs, in particular, play a pivotal role in lipid metabolism, mitochondrial function, and antioxidant defense, helping mitigate oxidative stress [
39]. Enhanced expression of ABCG5 and ABCG8 transporters following HYD-SBSR suggests a potential mechanism for reducing oxidative stress by promoting cholesterol excretion and metabolic balance [
40,
41]. This antioxidant effect is indicated by restored SOD, CAT, and GSH-Px activities and decreased MDA levels, which contributes to the reduction in obesity and hepatic steatosis in the liver. The proteomics analysis provides initial insights into the mechanisms underlying HYD-SBSR’s in vivo actions, paving the way for comprehensive research on its promising applications in oxidative stress-related conditions.
5. Conclusions
In this study, we initiated our research by creating an oxidative stress model using H2O2 on B16F10 cells, aiming to assess the potential cytoprotective and repair effects of HYD-SBSR. The results we obtained pointed toward a decrease in apoptosis rates and an improvement in resistance to oxidative stress upon treatment with HYD-SBSR. Additionally, our findings suggested that HYD-SBSR exhibited significant properties in terms of facilitating cell repair.
By incorporating the outcomes of antioxidant enzyme and peroxide analyses at the cellular level, further investigations were conducted to evaluate the impact of HYD-SBSR on antioxidant enzymes and peroxides in mice. The results exhibited significant antioxidant performance, particularly at lower concentrations of the extract, suggesting a potent ability to enhance antioxidant capacity.
To delve into the underlying mechanisms, we carried out a comprehensive proteomics analysis to identify proteins that were differentially expressed upon HYD-SBSR treatment. Subsequently, GO and KEGG pathway analysis were used to reveal the mechanism pathways associated with HYD-SBSR. Noteworthy pathways included PPAR signaling, fat digestion and absorption, glycerophospholipid metabolism, and cholesterol metabolism. These insights provide a deeper understanding of the cellular and molecular processes that underlie the observed cytoprotective and antioxidant effects of HYD-SBSR.
A comparison of in vitro and in vivo research findings demonstrated a consistent alignment. The inclusion of HYD-SBSR into food products not only addresses sustainability concerns in sea buckthorn production but also has the potential to enhance lipid metabolism within the organism. Furthermore, we speculate that it might even contribute to healthy weight management for individuals.