The Antibiotic Resistome and Its Association with Bacterial Communities in Raw Camel Milk from Altay Xinjiang
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. 16S rRNA Gene Amplication, Sequencing and Data Processing
2.3. High–Throughput Quantitative PCR (HT–qPCR) and Data Processing
2.4. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Parameters of Camel Milk
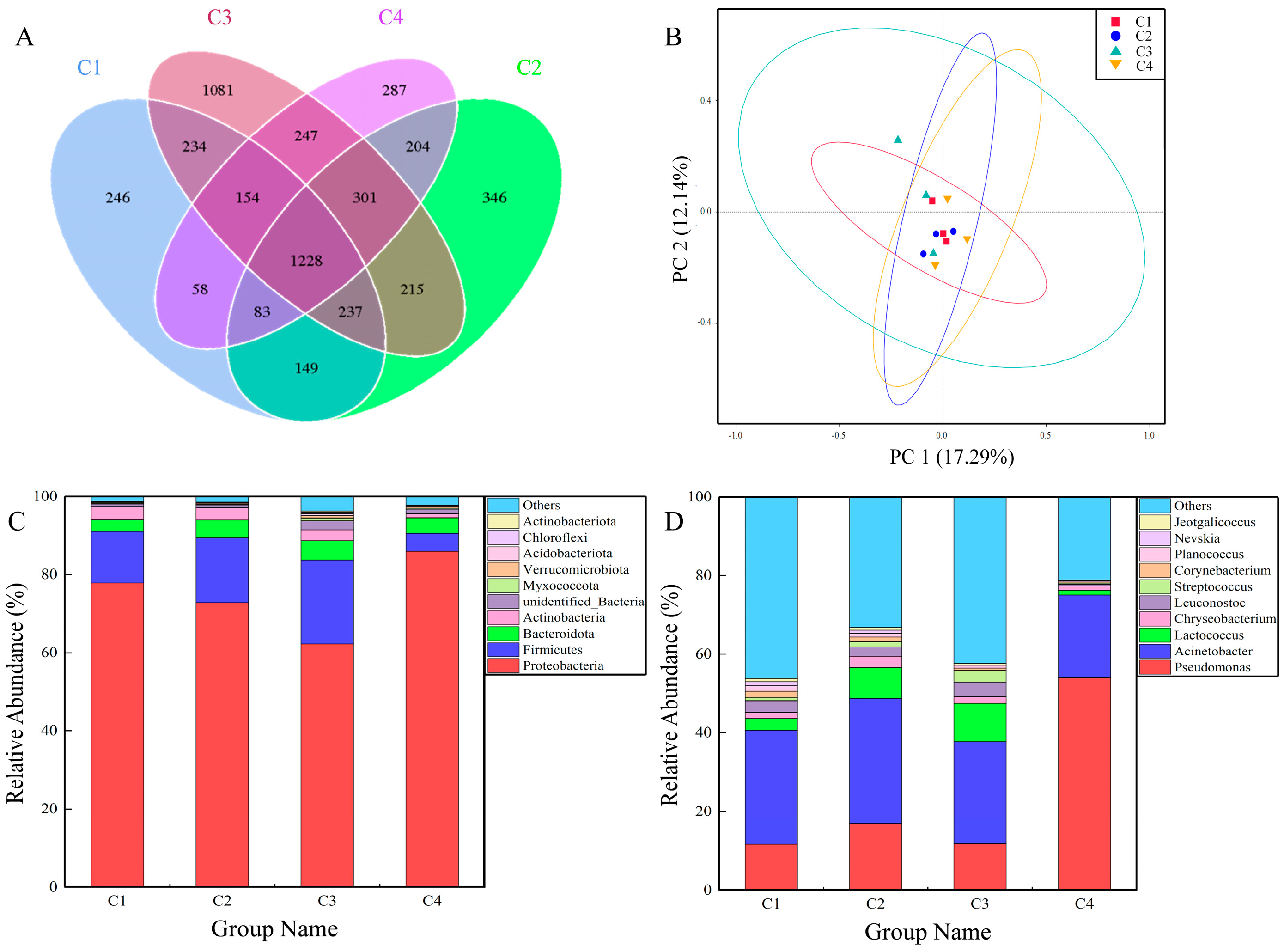
3.2. The Bacterial Community Structure of Camel Milk
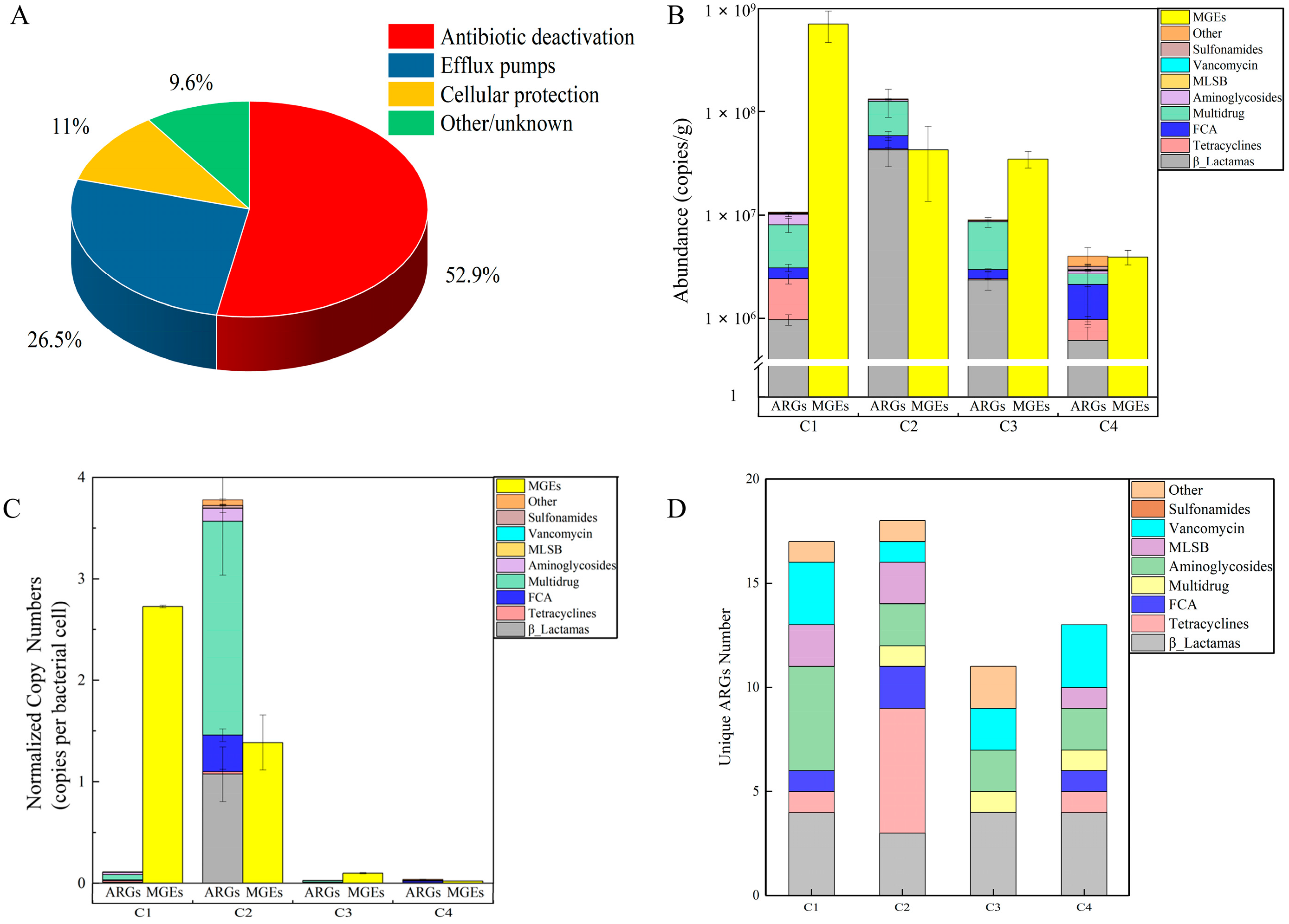
3.3. Antibiotic Resistant Genes in Camel Milk
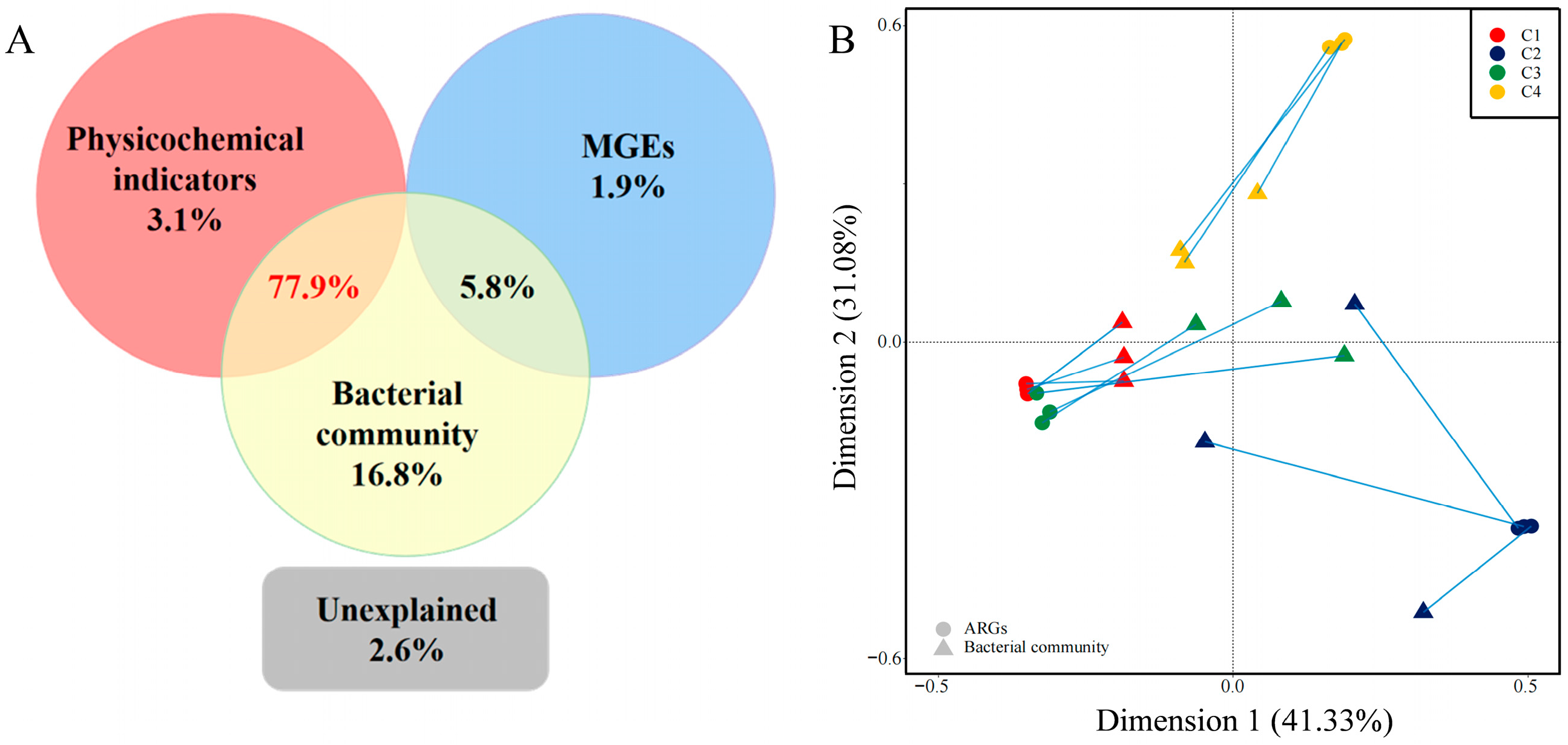
3.4. Relationship between Physicochemical Indicators, ARGs and Bacterial Community Composition
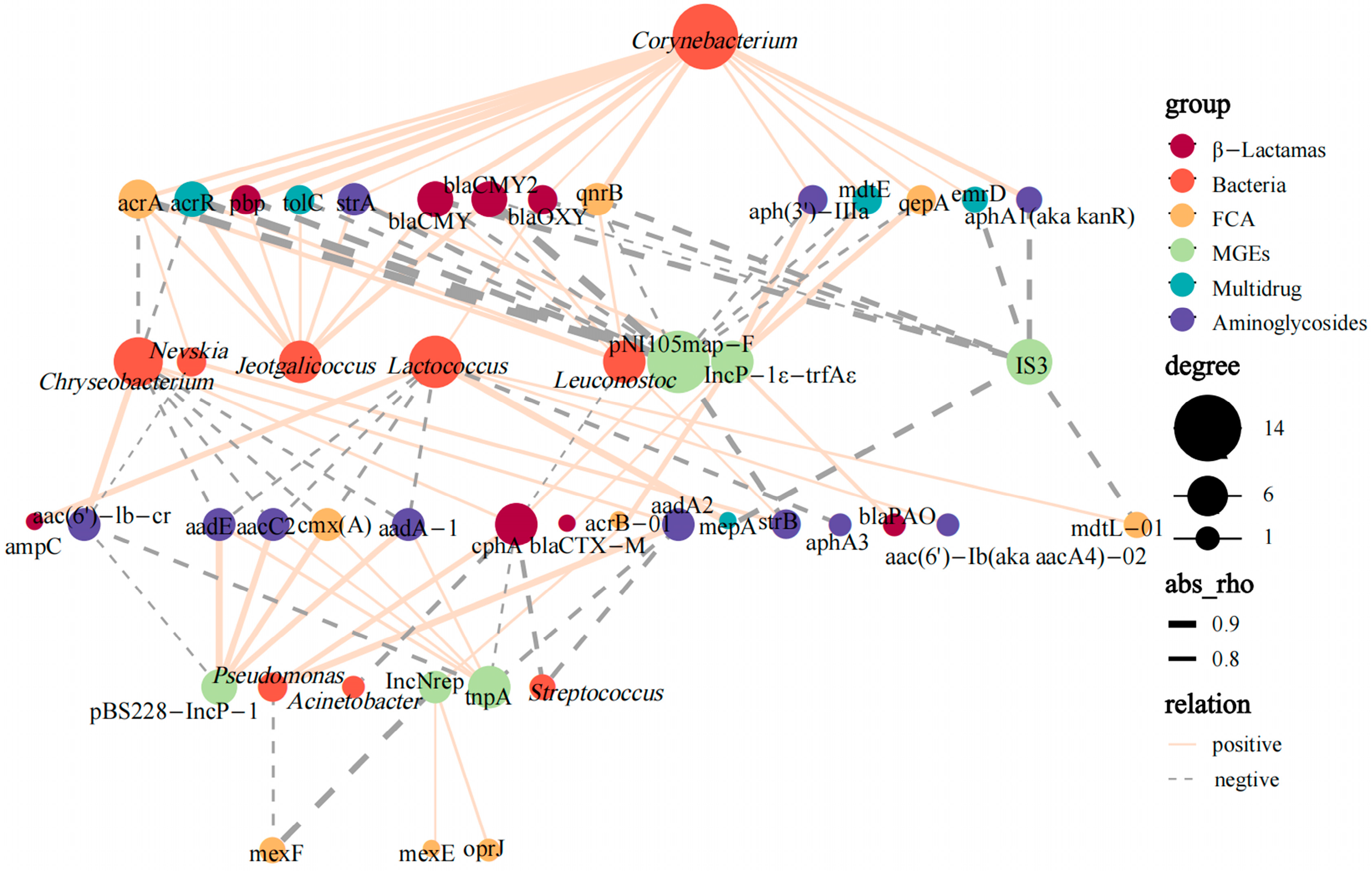
3.5. Transmission of ARGs in Camel Milk
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ji, R.; Cui, P.; Ding, F.; Geng, J.; Gao, H.; Zhang, H.; Yu, J.; Hu, S.; Meng, H. Monophyletic Origin of Domestic Bactrian Camel (Camelus bactrianus) and Its Evolutionary Relationship with the Extant Wild Camel (Camelus bactrianus Ferus). Anim. Genet. 2009, 40, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Chen, H.; Ren, Z.; Yang, X.; Zhang, C. Development of Genomic Resources and Identification of Genetic Diversity and Genetic Structure of the Domestic Bactrian Camel in China by RAD Sequencing. Front. Genet. 2020, 11, 797. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Ren, Z.; Zhao, J.; Zhang, C.; Yang, X. Y-Chromosome Polymorphisms of the Domestic Bactrian Camel in China. J. Genet. 2018, 97, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Adduci, F.; Elshafie, H.S.; Labella, C.; Musto, M.; Freschi, P.; Paolino, R.; Ragni, M.; Cosentino, C. Abatement of the Clostridial Load in the Teats of Lactating Cows with Lysozyme Derived from Donkey Milk. J. Dairy Sci. 2019, 102, 6750–6755. [Google Scholar] [CrossRef] [PubMed]
- Arain, M.A.; Khaskheli, G.B.; Shah, A.H.; Marghazani, I.B.; Barham, G.S.; Shah, Q.A.; Khand, F.M.; Buzdar, J.A.; Soomro, F.; Fazlani, S.A. Nutritional Significance and Promising Therapeutic/Medicinal Application of Camel Milk as a Functional Food in Human and Animals: A Comprehensive Review. Anim. Biotechnol. 2023, 34, 1988–2005. [Google Scholar] [CrossRef]
- Zhao, J.; Fan, H.; Kwok, L.-Y.; Guo, F.; Ji, R.; Ya, M.; Chen, Y. Analyses of Physicochemical Properties, Bacterial Microbiota, and Lactic Acid Bacteria of Fresh Camel Milk Collected in Inner Mongolia. J. Dairy Sci. 2020, 103, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Yehia, H.M.; Al-Masoud, A.H.; Alarjani, K.M.; Alamri, M.S. Prevalence of Methicillin-Resistant (mecA Gene) and Heat-Resistant Staphylococcus aureus Strains in Pasteurized Camel Milk. J. Dairy Sci. 2020, 103, 5947–5963. [Google Scholar] [CrossRef]
- Aljahani, A.H.; Alarjani, K.M.; Hassan, Z.K.; Elkhadragy, M.F.; Ismail, E.A.; Al-Masoud, A.H.; Yehia, H.M. Molecular Detection of Methicillin Heat-Resistant Staphylococcus aureus Strains in Pasteurized Camel Milk in Saudi Arabia. Biosci. Rep. 2020, 40, BSR20193470. [Google Scholar] [CrossRef]
- Du, B.; Lu, M.; Liu, H.; Wu, H.; Zheng, N.; Zhang, Y.; Zhao, S.; Zhao, Y.; Gao, T.; Wang, J. Pseudomonas Isolates from Raw Milk with High Level Proteolytic Activity Display Reduced Carbon Substrate Utilization and Higher Levels of Antibiotic Resistance. LWT 2023, 181, 114766. [Google Scholar] [CrossRef]
- Li, T.; Li, R.; Cao, Y.; Tao, C.; Deng, X.; Ou, Y.; Liu, H.; Shen, Z.; Li, R.; Shen, Q. Soil Antibiotic Abatement Associates with the Manipulation of Soil Microbiome via Long-Term Fertilizer Application. J. Hazard. Mater. 2022, 439, 129704. [Google Scholar] [CrossRef]
- Ellabaan, M.M.H.; Munck, C.; Porse, A.; Imamovic, L.; Sommer, M.O.A. Forecasting the Dissemination of Antibiotic Resistance Genes across Bacterial Genomes. Nat. Commun. 2021, 12, 2435. [Google Scholar] [CrossRef] [PubMed]
- Decimo, M.; Silvetti, T.; Brasca, M. Antibiotic Resistance Patterns of Gram-Negative Psychrotrophic Bacteria from Bulk Tank Milk: Antibiotic-Resistant Psychrotrophs. J. Food Sci. 2016, 81, M944–M951. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, Y.; Jay-Russell, M.; Lemay, D.G.; Mills, D.A. Reservoirs of Antimicrobial Resistance Genes in Retail Raw Milk. Microbiome 2020, 8, 99. [Google Scholar] [CrossRef]
- Taher, E.M.; Hemmatzadeh, F.; Aly, S.A.; Elesswy, H.A.; Petrovski, K.R. Survival of Staphylococci and Transmissibility of Their Antimicrobial Resistance Genes in Milk after Heat Treatments. LWT 2020, 129, 109584. [Google Scholar] [CrossRef]
- Bao, W.; He, Y.; Yu, J.; Yang, X.; Liu, M.; Ji, R. Diversity Analysis and Gene Function Prediction of Bacteria and Fungi of Bactrian Camel Milk and Naturally Fermented Camel Milk from Alxa in Inner Mongolia. LWT 2022, 169, 114001. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly Accurate OTU Sequences from Microbial Amplicon Reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA Ribosomal RNA Gene Database Project: Improved Data Processing and Web-Based Tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef]
- Chen, Q.; An, X.; Li, H.; Su, J.; Ma, Y.; Zhu, Y.-G. Long-Term Field Application of Sewage Sludge Increases the Abundance of Antibiotic Resistance Genes in Soil. Environ. Int. 2016, 92–93, 1–10. [Google Scholar] [CrossRef]
- Ouyang, W.-Y.; Huang, F.-Y.; Zhao, Y.; Li, H.; Su, J.-Q. Increased Levels of Antibiotic Resistance in Urban Stream of Jiulongjiang River, China. Appl. Microbiol. Biotechnol. 2015, 99, 5697–5707. [Google Scholar] [CrossRef]
- He, J.; Xiao, Y.; Orgoldol, K.; Ming, L.; Yi, L.; Ji, R. Effects of Geographic Region on the Composition of Bactrian Camel Milk in Mongolia. Animals 2019, 9, 890. [Google Scholar] [CrossRef]
- Rahmeh, R.; Akbar, A.; Alomirah, H.; Kishk, M.; Al-Ateeqi, A.; Al-Milhm, S.; Shajan, A.; Akbar, B.; Al-Merri, S.; Alotaibi, M.; et al. Camel Milk Microbiota: A Culture-Independent Assessment. Food Res. Int. 2022, 159, 111629. [Google Scholar] [CrossRef] [PubMed]
- Endres, C.M.; Castro, Í.M.S.; Trevisol, L.D.; Severo, J.M.; Mann, M.B.; Varela, A.P.M.; Frazzon, A.P.G.; Mayer, F.Q.; Frazzon, J. Molecular Characterization of the Bacterial Communities Present in Sheep’s Milk and Cheese Produced in South Brazilian Region via 16S rRNA Gene Metabarcoding Sequencing. LWT 2021, 147, 111579. [Google Scholar] [CrossRef]
- Mtshali, K.; Khumalo, Z.T.H.; Kwenda, S.; Arshad, I.; Thekisoe, O.M.M. Exploration and Comparison of Bacterial Communities Present in Bovine Faeces, Milk and Blood Using 16S rRNA Metagenomic Sequencing. PLoS ONE 2022, 17, e0273799. [Google Scholar] [CrossRef] [PubMed]
- Tidona, F.; Meucci, A.; Povolo, M.; Pelizzola, V.; Zago, M.; Contarini, G.; Carminati, D.; Giraffa, G. Applicability of Lactococcus hircilactis and Lactococcus laudensis as Dairy Cultures. Int. J. Food Microbiol. 2018, 271, 1–7. [Google Scholar] [CrossRef]
- Raimondi, S.; Candeliere, F.; Amaretti, A.; Costa, S.; Vertuani, S.; Spampinato, G.; Rossi, M. Phylogenomic Analysis of the Genus Leuconostoc. Front. Microbiol. 2022, 13, 897656. [Google Scholar] [CrossRef]
- Sun, L.; Lundh, Å.; Höjer, A.; Bernes, G.; Nilsson, D.; Johansson, M.; Hetta, M.; Gustafsson, A.H.; Saedén, K.H.; Dicksved, J. Milking System and Premilking Routines Have a Strong Effect on the Microbial Community in Bulk Tank Milk. J. Dairy Sci. 2022, 105, 123–139. [Google Scholar] [CrossRef]
- De Jonghe, V.; Coorevits, A.; Van Hoorde, K.; Messens, W.; Van Landschoot, A.; De Vos, P.; Heyndrickx, M. Influence of Storage Conditions on the Growth of Pseudomonas Species in Refrigerated Raw Milk. Appl. Environ. Microbiol. 2011, 77, 460–470. [Google Scholar] [CrossRef]
- Dehkordi, S.M.H.; Anvar, S.A.; Rahimi, E.; Ahari, H.; Ataee, M. Molecular Investigation of Prevalence, Phenotypic and Genotypic Diversity, Antibiotic Resistance, Frequency of Virulence Genes and Genome Sequencing in Pseudomonas aeruginosa Strains Isolated from Lobster. Int. J. Food Microbiol. 2022, 382, 109901. [Google Scholar] [CrossRef]
- Guo, X.; Akram, S.; Stedtfeld, R.; Johnson, M.; Chabrelie, A.; Yin, D.; Mitchell, J. Distribution of Antimicrobial Resistance across the Overall Environment of Dairy Farms—A Case Study. Sci. Total Environ. 2021, 788, 147489. [Google Scholar] [CrossRef]
- Delannoy, S.; Hoffer, C.; Tran, M.-L.; Madec, J.-Y.; Brisabois, A.; Fach, P.; Haenni, M. High Throughput qPCR Analyses Suggest That Enterobacterales of French Sheep and Cow Cheese Rarely Carry Genes Conferring Resistances to Critically Important Antibiotics for Human Medicine. Int. J. Food Microbiol. 2023, 403, 110303. [Google Scholar] [CrossRef]
- Pascu, C.; Herman, V.; Iancu, I.; Costinar, L. Etiology of Mastitis and Antimicrobial Resistance in Dairy Cattle Farms in the Western Part of Romania. Antibiotics 2022, 11, 57. [Google Scholar] [CrossRef]
- Aqib, A.I.; Muzammil, I.; Naseer, M.A.; Shoaib, M.; Bakht, P.; Zaheer, T.; Khan, Y.R.; Khan, R.L.; Usman, M.; Shafeeq, M.; et al. Pathological Insights into Camel Mastitis. Acta. Tropica. 2022, 231, 106415. [Google Scholar] [CrossRef]
- Liu, B.; Zhang, X.; Ding, X.; Wang, Y.; Zhu, G. Regulatory Mechanisms of Sub-Inhibitory Levels Antibiotics Agent in Bacterial Virulence. Appl. Microbiol. Biotechnol. 2021, 105, 3495–3505. [Google Scholar] [CrossRef]
- Yi, Q.; Zhang, Y.; Gao, Y.; Tian, Z.; Yang, M. Anaerobic Treatment of Antibiotic Production Wastewater Pretreated with Enhanced Hydrolysis: Simultaneous Reduction of COD and ARGs. Water Res. 2017, 110, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Su, J.-Q.; An, X.-L.; Huang, F.-Y.; Rensing, C.; Brandt, K.K.; Zhu, Y.-G. Feed Additives Shift Gut Microbiota and Enrich Antibiotic Resistance in Swine Gut. Sci. Total Environ. 2018, 621, 1224–1232. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Li, L.; Huang, F.; Hu, X.; Cao, X.; Mi, J.; Liao, X.; Xing, S. The Fate of Antibiotic Resistance Genes and Their Association with Bacterial and Archaeal Communities during Advanced Treatment of Pig Farm Wastewater. Sci. Total Environ. 2022, 851, 158364. [Google Scholar] [CrossRef] [PubMed]
- Ji, B.; Qin, J.; Ma, Y.; Liu, X.; Wang, T.; Liu, G.; Li, B.; Wang, G.; Gao, P. Metagenomic Analysis Reveals Patterns and Hosts of Antibiotic Resistance in Different Pig Farms. Environ. Sci. Pollut. Res. 2023, 30, 52087–52106. [Google Scholar] [CrossRef]
- Su, J.-Q.; Wei, B.; Ou-Yang, W.-Y.; Huang, F.-Y.; Zhao, Y.; Xu, H.-J.; Zhu, Y.-G. Antibiotic Resistome and Its Association with Bacterial Communities during Sewage Sludge Composting. Environ. Sci. Technol. 2015, 49, 7356–7363. [Google Scholar] [CrossRef]
- Lin, H.; Zhang, J.; Chen, H.; Wang, J.; Sun, W.; Zhang, X.; Yang, Y.; Wang, Q.; Ma, J. Effect of Temperature on Sulfonamide Antibiotics Degradation, and on Antibiotic Resistance Determinants and Hosts in Animal Manures. Sci. Total Environ. 2017, 607–608, 725–732. [Google Scholar] [CrossRef]
- Ren, Z.; Zhao, Y.; Han, S.; Li, X. Regulatory Strategies for Inhibiting Horizontal Gene Transfer of ARGs in Paddy and Dryland Soil through Computer-Based Methods. Sci. Total Environ. 2023, 856, 159096. [Google Scholar] [CrossRef]
- Zhang, B.; Qin, S.; Guan, X.; Jiang, K.; Jiang, M.; Liu, F. Distribution of Antibiotic Resistance Genes in Karst River and Its Ecological Risk. Water Res. 2021, 203, 117507. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Wang, Y.; Jin, M.; Yuan, Z.; Bond, P.; Guo, J. Both Silver Ions and Silver Nanoparticles Facilitate the Horizontal Transfer of Plasmid-Mediated Antibiotic Resistance Genes. Water Res. 2020, 169, 115229. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Wang, Y.; Du, B.; Li, H.; Dong, L.; Hu, H.; Meng, L.; Zheng, N.; Wang, J. Influence of Dairy Cows Bedding Material on the Microbial Structure and Antibiotic Resistance Genes of Milk. Front. Microbiol. 2022, 13, 830333. [Google Scholar] [CrossRef]
- Nikoloudaki, O.; Lemos Junior, W.J.F.; Campanaro, S.; Di Cagno, R.; Gobbetti, M. Role Prediction of Gram-Negative Species in the Resistome of Raw Cow’s Milk. Int. J. Food Microbiol. 2021, 340, 109045. [Google Scholar] [CrossRef]
- Song, L.; Wang, C.; Jiang, G.; Ma, J.; Li, Y.; Chen, H.; Guo, J. Bioaerosol Is an Important Transmission Route of Antibiotic Resistance Genes in Pig Farms. Environ. Int. 2021, 154, 106559. [Google Scholar] [CrossRef] [PubMed]

| Farms | Protein (%) | Fat (%) | Nonfat Milk Solid (%) | Lactose (%) |
|---|---|---|---|---|
| C1 | 3.82 ± 0.03 | 4.19 ± 0.03 | 9.61 ± 0.03 | 5.07 ± 0.09 |
| C2 | 3.79 ± 0.02 | 5.29 ± 0.18 | 9.48 ± 0.01 | 4.98 ± 0.02 |
| C3 | 3.85 ± 0.07 | 4.74 ± 0.16 | 9.66 ± 0.02 | 5.08 ± 0.07 |
| C4 | 3.82 ± 0.03 | 4.52 ± 0.07 | 9.64 ± 0.01 | 5.09 ± 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, Y.; Huang, W.; Yang, J.; Zhao, Y.; Zhao, M.; Xu, H.; Zhang, M. The Antibiotic Resistome and Its Association with Bacterial Communities in Raw Camel Milk from Altay Xinjiang. Foods 2023, 12, 3928. https://doi.org/10.3390/foods12213928
Qin Y, Huang W, Yang J, Zhao Y, Zhao M, Xu H, Zhang M. The Antibiotic Resistome and Its Association with Bacterial Communities in Raw Camel Milk from Altay Xinjiang. Foods. 2023; 12(21):3928. https://doi.org/10.3390/foods12213928
Chicago/Turabian StyleQin, Yanan, Wanting Huang, Jie Yang, Yan Zhao, Min Zhao, Haotian Xu, and Minwei Zhang. 2023. "The Antibiotic Resistome and Its Association with Bacterial Communities in Raw Camel Milk from Altay Xinjiang" Foods 12, no. 21: 3928. https://doi.org/10.3390/foods12213928







