Impact of LAB from Serpa PDO Cheese in Cheese Models: Towards the Development of an Autochthonous Starter Culture
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Bacterial Isolates
2.3. Cheese-like Model System
2.3.1. Preparation of Models
2.3.2. Microbiological Analysis and pH Monitorization
2.3.3. Determination of Proteolytic Indices
2.3.4. Protein and Peptide Profile
2.3.5. Free Amino Acid (FAA) Profile
2.3.6. Volatiles Fatty Acids (VFA) Profile
2.4. Statistical Analysis
3. Results and Discussion
3.1. Microbiological and pH Monitorization
3.2. Proteolysis
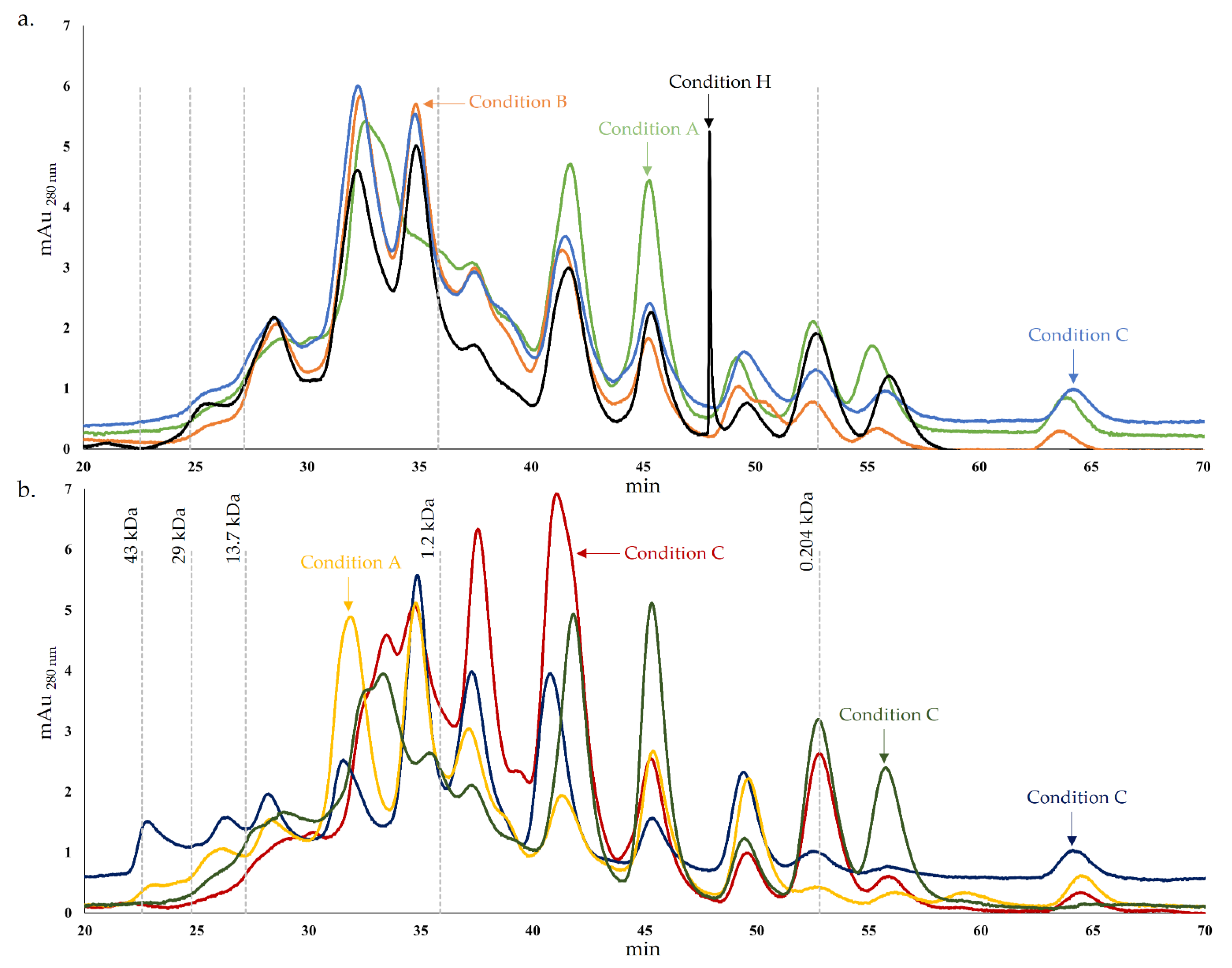
3.2.1. Protein and Peptide Profile
3.2.2. Soluble Nitrogen Assays
3.2.3. Free Amino Acids (FAA) Content
3.3. Volatiles
3.3.1. Volatile Fatty Acids (VFA)
3.3.2. Esters
3.4. Results Integration
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Reis, P.J.M.; Malcata, F.X. Current State of Portuguese Dairy Products from Ovine and Caprine Milks. Small Rumin. Res. 2011, 101, 122–133. [Google Scholar] [CrossRef]
- Dias, J.M.; Lage, P.; Alvarenga, N.; Garcia, J.; Borrega, J.; Santos, M.T.; Lampreia, C.; Coelho, L.; Pássaro, J.; Martins, J.; et al. Impact of Environmental Conditions on the Ripening of Queijo de Évora PDO Cheese. J. Food Sci. Technol. 2021, 58, 3942–3952. [Google Scholar] [CrossRef]
- Guerreiro, O.; Velez, Z.; Alvarenga, N.; Matos, C.; Duarte, M. Molecular Screening of Ovine Mastitis in Different Breeds. J. Dairy Sci. 2013, 96, 752–760. [Google Scholar] [CrossRef]
- Araújo-Rodrigues, H.; Tavaria, F.K.; dos Santos, M.T.P.G.; Alvarenga, N.; Pintado, M.M. A Review on Microbiological and Technological Aspects of Serpa PDO Cheese: An Ovine Raw Milk Cheese. Int. Dairy J. 2020, 100, 104561. [Google Scholar] [CrossRef]
- Roseiro, L.B.; Andrew Wilbey, R.; Barbosa, M. Serpa Cheese: Technological, Biochemical and Microbiological Characterisation of a PDO Ewe’s Milk Cheese Coagulated with Cynara cardunculus L. Le Lait 2003, 83, 469–481. [Google Scholar] [CrossRef]
- Alvarenga, N.; Martins, J.; Caeiro, J.; Garcia, J.; Pássaro, J.; Coelho, L.; Santos, M.T.; Lampreia, C.; Martins, A.; Dias, J. Applying Computational Fluid Dynamics in the Development of Smart Ripening Rooms for Traditional Cheeses. Foods 2021, 10, 1716. [Google Scholar] [CrossRef]
- Ardö, Y.; McSweeney, P.L.H.; Magboul, A.A.A.; Upadhyay, V.K.; Fox, P.F. Biochemistry of Cheese Ripening: Proteolysis. Cheese Chem. Phys. Microbiol. Fourth Ed. 2017, 1, 445–482. [Google Scholar] [CrossRef]
- Alvarenga, N.; Silva, P.; Garcia, J.R.; Sousa, I. Estimation of Serpa Cheese Ripening Time Using Multiple Linear Regression (MLR) Considering Rheological, Physical and Chemical data. J. Dairy Res. 2008, 75, 233–239. [Google Scholar] [CrossRef]
- Freitas, C.; Malcata, F.X. Microbiology and Biochemistry of Cheeses With Appélation d’Origine Protegée and Manufactured in the Iberian Peninsula from Ovine and Caprine Milks. J. Dairy Sci. 2000, 83, 584–602. [Google Scholar] [CrossRef]
- IMAIAA Regras de Produção Do Queijo Serpa. Available online: http://ec.europa.eu/agriculture/quality/door/registeredName.html?denominationId=590 (accessed on 6 March 2022).
- Roseiro, L.B.; Barbosa, M.; Ames, J.M.; Wilbey, R.A. Cheesemaking with Vegetable Coagulants-the Use of Cynara L. for the Production of Ovine Milk Cheeses. Int. J. Dairy Technol. 2003, 56, 76–85. [Google Scholar] [CrossRef]
- Dos Santos, M.T.P.G.; Benito, M.J.; de Guía Córdoba, M.; Alvarenga, N.; de Herrera, S.R.M.S. Yeast Community in Traditional Portuguese Serpa Cheese by Culture-dependent and -Independent DNA Approaches. Int. J. Food Microbiol. 2017, 262, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, M.T.P.; Benito, M.J.; Córdoba, M.D.G.; Egas, C.; Merchán, A.V.; Galván, A.I.; Ruiz-Moyano, S. Bacterial Communities in Serpa Cheese by Culture Dependent Techniques, 16S rRNA Gene Sequencing and High-throughput Sequencing Analysis. J. Food Sci. 2018, 83, 1333–1341. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Moyano, S.; Gonçalves dos Santos, M.T.P.; Galván, A.I.; Merchán, A.V.; González, E.; Córdoba, M.D.G.; Benito, M.J. Screening of Autochthonous Lactic Acid Bacteria Strains from Artisanal Soft Cheese: Probiotic Characteristics and Prebiotic metabolism. LWT-Food Sci. Technol. 2019, 114, 108388. [Google Scholar] [CrossRef]
- RD. Regulatory Decree no 39/87; RD: Lisboa, Portugal, 1987; pp. 2499–2500. [Google Scholar]
- Montel, M.C.; Buchin, S.; Mallet, A.; Delbes-Paus, C.; Vuitton, D.A.; Desmasures, N.; Berthier, F. Traditional cheeses: Rich and diverse microbiota with associated benefits. Int. J. Food Microbiol. 2014, 177, 136–154. [Google Scholar] [CrossRef]
- Silvetti, T.; Capra, E.; Morandi, S.; Cremonesi, P.; Decimo, M.; Gavazzi, F.; Giannico, R.; De Noni, I.; Brasca, M. Microbial Population Profile during Ripening of Protected Designation of Origin (PDO) Silter cheese, Produced with and without Autochthonous Starter Culture. LWT-Food Sci. Technol. 2017, 84, 821–831. [Google Scholar] [CrossRef]
- Chourasia, R.; Abedin, M.M.; Chiring Phukon, L.; Sahoo, D.; Singh, S.P.; Rai, A.K. Biotechnological Approaches for the Production of Designer Cheese with Improved Functionality. Compr. Rev. Food Sci. Food Saf. 2021, 20, 960–979. [Google Scholar] [CrossRef]
- Leroy, F.; De Vuyst, L. Lactic Acid Bacteria as Functional Starter Cultures for the Food Fermentation Industry. Trends Food Sci. Technol. 2004, 15, 67–78. [Google Scholar] [CrossRef]
- Tavaria, F.K.; Tavares, T.G.; Silva-Ferreira, A.C.; Malcata, F.X. Contribution of Coagulant and Native Microflora to the Volatile-Free Fatty Acid Profile of an Artisanal Cheese. Int. Dairy J. 2006, 16, 886–894. [Google Scholar] [CrossRef]
- Câmara, S.P.; Dapkevicius, A.; Riquelme, C.; Elias, R.B.; Silva, C.C.G.; Malcata, F.X.; Dapkevicius, M.L.N.E. Potential of Lactic Acid Bacteria from Pico Cheese for Starter Culture Development. Food Sci. Technol. Int. 2019, 25, 303–317. [Google Scholar] [CrossRef]
- Araújo-Rodrigues, H.; dos Santos, M.T.P.G.; Ruiz-Moyano, S.; Tavaria, F.K.; Martins, A.P.L.; Alvarenga, N.; Pintado, M.E. Technological and Protective Performance of LAB Isolated from Serpa PDO Cheese: Towards Selection and Development of an Autochthonous Starter Culture. LWT-Food Sci. Technol. 2021, 150, 112079. [Google Scholar] [CrossRef]
- Bassi, D.; Puglisi, E.; Cocconcelli, P.S. Comparing Natural and Selected Starter Cultures in Meat and Cheese Fermentations. Curr. Opin. Food Sci. 2015, 2, 118–122. [Google Scholar] [CrossRef]
- Tavaria, F.K.; Dahl, S.; Carballo, F.J.; Malcata, F.X. Amino Acid Catabolism and Generation of Volatiles by Lactic Acid Bacteria. J. Dairy Sci. 2002, 85, 2462–2470. [Google Scholar] [CrossRef]
- Milesi, M.M.; Candioti, M.; Hynes, E. Mini Soft Cheese as a Simple Model for Biochemical Studies on Cheese-Making and Ripening. LWT-Food Sci. Technol. 2007, 40, 1427–1433. [Google Scholar] [CrossRef]
- Pereira, C.I.; Gomes, E.O.; Gomes, A.M.P.; Malcata, F.X. Proteolysis in Model Portuguese Cheeses: Effects of Rennet and Starter Culture. Food Chem. 2008, 108, 862–868. [Google Scholar] [CrossRef]
- De Aguiar e Câmara, S.P.; Maduro Dias, C.; Rocha, L.; Dapkevicius, A.; Duarte Rosa, H.J.; de Borba, A.E.S.; da Graça Silveira, M.; Malcata, F.X.; de Lurdes Enes Dapkevicius, M. Assessment of Autochthonous Lactic Acid Bacteria as Starter Cultures for Improved Manufacture of Pico Cheese using a Cheese Model. Int. Dairy J. 2022, 128, 105294. [Google Scholar] [CrossRef]
- Murtaza, M.A.; Ur-Rehman, S.; Anjum, F.M.; Huma, N.; Hafiz, I. Cheddar Cheese Ripening and Flavor Characterization: A Review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1309–1321. [Google Scholar] [CrossRef]
- Dahl, S.; Tavaria, F.K.; Xavier Malcata, F. Relationships Between Flavour and Microbiological Profiles in Serra da Estrela Cheese Throughout Ripening. Int. Dairy J. 2000, 10, 255–262. [Google Scholar] [CrossRef]
- Tavaria, F.K.; Franco, I.; Javier Carballo, F.; Xavier Malcata, F. Amino Acid and Soluble Nitrogen Evolution Throughout Ripening of Serra da Estrela Cheese. Int. Dairy J. 2003, 13, 537–545. [Google Scholar] [CrossRef]
- Araújo-Rodrigues, H.; Martins, A.P.L.; Tavaria, F.K.; Santos, M.T.G.; Carvalho, M.J.; Dias, J.; Alvarenga, N.B.; Pintado, M.E. Organoleptic Chemical Markers of Serpa PDO Cheese Specificity. Foods 2022, 11, 1898. [Google Scholar] [CrossRef]
- ISO 4833-1; Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms. ISO: Geneva, Switzerland, 2013.
- ISO 6888-1; Microbiology of the Food Chain—Horizontal Method for the Enumeration of Coagulase-Positive Staphylococci (Staphylococcus Aureus and Other Species). ISO: Geneva, Switzerland, 2021.
- Kuchroo, C.N.; Fox, P.F. Soluble Nitrogen in Cheddar Cheese: Comparison of Extraction Procedures. Milk Sci. Int. 1982, 37, 331–335. [Google Scholar]
- Voss, G.B.; Osorio, H.; Valente, L.M.P.; Pintado, M.E. Impact of Thermal Treatment and Hydrolysis by Alcalase and Cynara cardunculus Enzymes on the Functional and Nutritional Value of Okara. Process Biochem. 2019, 83, 137–147. [Google Scholar] [CrossRef]
- Tavares, T.; Contreras, M.D.M.; Amorim, M.; Pintado, M.; Recio, I.; Malcata, F.X. Novel Whey-derived Peptides with Inhibitory Effect Against Angiotensin-Converting Enzyme: In vitro Effect and Stability to Gastrointestinal Enzymes. Peptides 2011, 32, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Alonso, M.L.; Alvarez, A.I.; Zapico, J. Rapid Analysis of Free Amino Acids in Infant Foods. J. Liq. Chromatogr. 1994, 17, 4019–4030. [Google Scholar] [CrossRef]
- Pripi-Nicolau, L.; De Revel, G.; Bertrand, A.; Maujean, A. Formation of Flavor Components by the Reaction of Amino Acid and Carbonyl Compounds in Mild Conditions. J. Agric. Food Chem. 2000, 48, 3761–3766. [Google Scholar] [CrossRef]
- Aquilanti, L.; Babini, V.; Santarelli, S.; Osimani, A.; Petruzzelli, A.; Clementi, F. Bacterial Dynamics in a Raw Cow’s Milk Caciotta Cheese Manufactured with Aqueous Extract of Cynara cardunculus Dried Flowers. Lett. Appl. Microbiol. 2011, 52, 651–659. [Google Scholar] [CrossRef]
- Zheng, X.; Shi, X.; Wang, B. A Review on the General Cheese Processing Technology, Flavor Biochemical Pathways and the Influence of Yeasts in Cheese. Front. Microbiol. 2021, 12, 703284. [Google Scholar] [CrossRef] [PubMed]
- Settanni, L.; Moschetti, G. Non-starter Lactic Acid Bacteria Used to Improve Cheese Quality and Provide Health Benefits. Food Microbiol. 2010, 27, 691–697. [Google Scholar] [CrossRef]
- Fox, P.F.; Law, J.; McSweeney, P.L.H.; Wallace, J. Biochemistry of Cheese Ripening. In Cheese: Chemistry, Physics and Microbiology; Springer: Berlin/Heidelberg, Germany, 1993; pp. 389–438. [Google Scholar]
- Sousa, M.J.; Malcata, F.X. Advances in the Role of a Plant Coagulant (Cynara cardunculus) In Vitro and During Ripening of Cheeses from Several Milk Species. Le Lait 2002, 82, 151–170. [Google Scholar] [CrossRef]
- Conceição, C.; Martins, P.; Alvarenga, N.; Dias, J.; Lamy, E.; Garrido, L.; Gomes, S.; Freitas, S.; Belo, A.; Brás, T.; et al. Cynara cardunculus: Use in Cheesemaking and Pharmaceutical Applications. Technol. Approaches Nov. Appl. Dairy Process. 2018, 73–107. [Google Scholar] [CrossRef]
- Pino, A.; Prados, F.; Galán, E.; Vivo, R.; Fernández-Salguero, J. Amino Acids Evolution During Ripening of Goats’ Milk Cheese Manufactured with Different Coagulants. Int. J. Food Sci. Technol. 2009, 44, 2062–2069. [Google Scholar] [CrossRef]
- Araújo-Rodrigues, H.; Coscueta, E.R.; Pereira, M.F.; Cunha, S.A.; Almeida, A.; Rosa, A.; Martins, R.; Pereira, C.D. Membrane Fractionation of Cynara cardunculus Swine Blood Hydrolysate: Ingredients of High Nutritional and Nutraceutical Value. Food Res. Int. 2022, 158, 111549. [Google Scholar] [CrossRef]
- Ben Amira, A.; Besbes, S.; Attia, H.; Blecker, C. Milk-clotting Properties of Plant Rennets and Their Enzymatic, Rheological, and Sensory Role in Cheesemaking: A Review. Int. J. Food Prop. 2017, 20, S76–S93. [Google Scholar]
- McSweeney, P.L.H.; Fox, P.F. Chemical Methods for the Characterization of Proteolysis in Cheese During Ripening. Le Lait 1997, 77, 41–76. [Google Scholar] [CrossRef]
- Roseiro, L.B.; Garcia-Risco, M.; Barbosa, M.; Ames, J.M.; Wilbey, R.A. Evaluation of Serpa Cheese Proteolysis by Nitrogen Content and Capillary Zone Electrophoresis. Int. J. Dairy Technol. 2003, 56, 99–104. [Google Scholar] [CrossRef]
- Alvarenga, N.; Canada, J.; Sousa, I. Effect of Freezing on the Rheological, Chemical and Colour Properties of Serpa Cheese. J. Dairy Res. 2011, 78, 80–87. [Google Scholar] [CrossRef]
- Macedo, A.C.; Tavares, T.G.; Malcata, F.X. Influence of Native Lactic Acid Bacteria on the Microbiological, Biochemical and Sensory Profiles of Serra da Estrela Cheese. Food Microbiol. 2004, 21, 233–240. [Google Scholar] [CrossRef]
- Niro, S.; Succi, M.; Tremonte, P.; Sorrentino, E.; Coppola, R.; Panfili, G.; Fratianni, A. Evolution of Free Amino Acids During Ripening of Caciocavallo cheeses Made with Different Milks. J. Dairy Sci. 2017, 100, 9521–9531. [Google Scholar] [CrossRef]
- Reis Lima, M.J.; Santos, A.O.; Falcão, S.; Fontes, L.; Teixeira-Lemos, E.; Vilas-Boas, M.; Veloso, A.C.A.; Peres, A.M. Serra da Estrela Cheese’s Free Amino Acids Profiles by UPLC-DAD-MS/MS and Their Application for Cheese Origin Assessment. Food Res. Int. 2019, 126, 108729. [Google Scholar] [CrossRef]
- Ribeiro, S.C.; Coelho, M.C.; Todorov, S.D.; Franco, B.D.G.M.; Dapkevicius, M.L.E.; Silva, C.C.G. Technological Properties of Bacteriocin-producing Lactic Acid Bacteria Isolated from Pico cheese an Artisanal Cow’s Milk Cheese. J. Appl. Microbiol. 2014, 116, 573–585. [Google Scholar] [CrossRef]
- Malacarne, M.; Summer, A.; Formaggioni, P.; Franceschi, P.; Sandri, S.; Pecorari, M.; Vecchia, P.; Mariani, P. Dairy Maturation of Milk Used in the Manufacture of Parmigiano-Reggiano cheese: Effects on Physico-Chemical Characteristics, Rennet-Coagulation Aptitude and Rheological Properties. J. Dairy Res. 2008, 75, 218–224. [Google Scholar] [CrossRef]
- Pinho, O.; Ferreira, I.M.P.L.V.O.; Ferreira, M.A. Solid-Phase Microextraction in Combination with GC/MS for Quantification of the Major Volatile Free Fatty Acids in Ewe Cheese. Anal. Chem. 2002, 74, 5199–5204. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, F.; Ferrocino, I.; Milanović, V.; Belleggia, L.; Corvaglia, M.R.; Garofalo, C.; Foligni, R.; Mannozzi, C.; Mozzon, M.; Cocolin, L.; et al. Microbial Communities and Volatile Profile of Queijo de Azeitão PDO Cheese, a Traditional Mediterranean Thistle-Curdled Cheese from Portugal. Food Res. Int. 2021, 147, 110537. [Google Scholar] [CrossRef] [PubMed]
- Cuffia, F.; Bergamini, C.V.; Wolf, I.V.; Hynes, E.R.; Perotti, M.C. Characterization of Volatile Compounds Produced by Lactobacillus helveticus Strains in a Hard Cheese Model. Food Sci. Technol. Int. 2018, 24, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, I.M.P.L.V.O.; Pinho, O.; Sampaio, P. Volatile Fraction of DOP “Castelo Branco” Cheese: Influence of Breed. Food Chem. 2009, 112, 1053–1059. [Google Scholar] [CrossRef]
- Tavaria, F.K.; Silva Ferreira, A.C.; Xavier Malcata, F. Volatile Free Fatty Acids as Ripening Indicators for Serra da Estrela Cheese. J. Dairy Sci. 2004, 87, 4064–4072. [Google Scholar] [CrossRef] [PubMed]
- Filipczak-Fiutak, M.; Pluta-Kubica, A.; Domagała, J.; Duda, I.; Migdał, W. Nutritional Value and Organoleptic Assessment of Traditionally Smoked Cheeses Made from Goat, Sheep and Cow’s Milk. PLoS ONE 2021, 16, e0254431. [Google Scholar] [CrossRef]
- Liu, S.-Q.; Baker, K.; Bennett, M.; Holland, R.; Norris, G.; Crow, V.L. Characterisation of Esterases of Streptococcus thermophilus ST1 and Lactococcus lactis subsp. cremoris B1079 as Alcohol Acyltransferases. Int. Dairy J. 2004, 14, 865–870. [Google Scholar] [CrossRef]
- Curioni, P.M.G.; Bosset, J.O. Key Odorants in Various Cheese Types as Determined by Gas Chromatography-Olfactometry. Int. Dairy J. 2002, 12, 959–984. [Google Scholar] [CrossRef]
- Guzmán, J.L.; Delgado Pertíñez, M.; Galán Soldevilla, H.; Ruiz Pérez-Cacho, P.; Polvillo Polo, O.; Zarazaga, L.Á.; Avilés Ramírez, C. Effect of Citrus By-product on Physicochemical Parameters, Sensory Analysis and Volatile Composition of Different Kinds of Cheese from Raw Goat Milk. Foods 2020, 9, 1420. [Google Scholar] [CrossRef]
- Gonzalez-Mendoza, L.A.; Diaz-Rodriguez, F. Sensory Analysis Using Sniffing, for Determination of the Aroma of Flor de Guia Cheese. Aliment. Equipos Tecnol. 1993, 12, 43–49. [Google Scholar]
- Castada, H.; Hanas, K.; Barringer, S. Swiss Cheese Flavor Variability Based on Correlations of Volatile Flavor Compounds, Descriptive Sensory Attributes, and Consumer Preference. Foods 2019, 8, 78. [Google Scholar] [CrossRef] [PubMed]




| Species | Probiotic Potential [14] | Biogenic Amine Production and Antibiotic Resistance [14] | Extracellular Proteolytic Activity [22] | Lipolytic Activity [22] | Salt and Temperature Resistance [22] | D(-)-lactic acid Production [22] 1 | Acidification Potential [22] 2 | Antimicrobial Activity [22] 3 |
|---|---|---|---|---|---|---|---|---|
| Lacticaseibacillus paracasei PC | - | - | + | + | + | <1 | 1.8–2.0 | Listeria monocytogenes 934 Salmonella choleraesuis CECT 4395 |
| Lactiplantibacillus plantarum PL1 | + | - | + | + | + | 6–8 | 2.6–2.8 | 3 Listeria spp. strains Bacillus cereus CECT 131 S. choleraesuis CECT 4395 |
| Lactiplantibacillus plantarum PL2 | + | - | - | - | + | 8–10 | 2.6–2.8 | 3 Listeria spp. strains B. cereus CECT 131 S. choleraesuis CECT 4395 |
| Lactobacillus crustorum CR | + | - | + | - | + | 1–4 | 2.2–2.4 | S. choleraesuis CECT 4395 |
| Lactobacillus pentosus PE | + | - | + | - | + | 6–8 | 2.6–2.8 | S. choleraesuis CECT 4395 |
| Levilactobacillus brevis BR1 | + | - | + | - | + | 1–4 | 2.0–2.2 | S. choleraesuis CECT 4395 |
| Ripening Time | |||||
|---|---|---|---|---|---|
| Condition | 0 Days | 7 Days | 15 Days | 30 Days | 45 Days |
| A: L. paracasei PC | 5.81 ± 0.14 a | 5.19 ± 0.05 a | 4.96 ± 0.09 a | 4.70 ± 0.05 a | 4.62 ± 0.07 a |
| B: L. plantarum PL1 | 5.88 ± 0.05 a | 4.80 ± 0.04 b | 4.65 ± 0.05 b | 4.38 ± 0.02 b,c | 4.31 ± 0.03 b,c |
| C: L. paracasei PC and L. plantarum PL1 | 6.12 ± 0.00 b | 5.29 ± 0.03 a | 4.92 ± 0.14 a | 4.63 ± 0.10 a,c | 4.39 ± 0.07 c,d |
| D: L. plantarum PL2 | 5.94 ± 0.13 a | 4.63 ± 0.03 c | 4.52 ± 0.02 b | 4.33 ± 0.02 b | 4.49 ± 0.08 a,d |
| E: L. crustorum CR | 5.73 ± 0.01 a | 4.64 ± 0.01 c | 4.52 ± 0.14 b | 4.42 ± 0.03 b,c | 4.23 ± 0.07 b |
| F: L. pentosus PE | 5.28 ± 0.04 c | 4.18 ± 0.04 d | 4.00 ± 0.03 c | 3.86 ± 0.02 d | 3.84 ± 0.01 e |
| G: L. brevis BR1 | 6.22 ± 0.03 b | 5.77 ± 0.05 g | 5.33 ± 0.06 d | 5.22 ± 0.06 e | 4.98 ± 0.05 f |
| H: Control | 6.55 ± 0.01 d | 6.29 ± 0.01 f | 6.00 ± 0.01 e | 5.60 ± 0.02 f | 5.36 ± 0.03 g |
| Condition | ||||||||
|---|---|---|---|---|---|---|---|---|
| FAA | A | B | C | D | E | F | G | H |
| Asp | 68.10 ± 5.69 a,b | 58.69 ± 5.77 a,d | 73.14 ± 4.57 b | 159.67 ± 7.21 c | 39.10 ± 1.15 e | 49.10 ± 3.01 d,e | 26.08 ± 1.86 g | 13.49 ± 0.21 f |
| Glu | 126.88 ± 1.20 a | 105.57 ± 4.45 b | 150.89 ± 3.82 c | 164.47 ± 4.75 c | 127.52 ± 4.94 a | 80.52 ± 8.95 d | 46.70 ± 4.81 e | 78.55 ± 0.20 d |
| Cys | N.D. a | N.D. a | 1.30 ± 0.35 b | 5.03 ± 0.63 c | 6.25 ± 0.07 d | N.D. a | 0.47 ± 0.02 a | N.D. a |
| Asn | 31.44 ± 6.40 a | 28.39 ± 4.91 a | 31.56 ± 1.56 a | 42.88 ± 2.18 b | 18.12 ± 0.39 c,e | 28.09 ± 5.40 a,c | 13.93 ± 1.91 d | 5.18 ± 0.01 d |
| Ser | 52.22 ± 0.00 a | 7.04 ± 1.31 b | 22.53 ± 3.10 c | 17.72 ± 0.24 d | 8.34 ± 0.42 b,e | 11.51 ± 1.04 e | 15.60 ± 0.23 d | 5.79 ± 0.70 b |
| Gln | 117.14 ± 13.78 a | 30.55 ± 0.00 b | 103.22 ± 6.51 a,e | 11.78 ± 0.94 b,c | N.D. c | 78.78 ± 22.41 e | 38.11 ± 6.55 d | N.D. c |
| Thr | 37.61 ± 1.75 a | 12.97 ± 2.05 b | 27.53 ± 1.85 c | 25.99 ± 0.00 c | 14.83 ± 0.53 b | 18.69 ± 0.50 d | 5.96 ± 0.30 e | N.D. f |
| Arg | 52.92 ± 6.86 a | 76.04 ± 3.33 b | 106.18 ± 5.87 c | 55.84 ± 5.75 a | 39.67 ± 1.37 d | 50.77 ± 5.68 a,d | 38.73 ± 1.12 d | 62.02 ± 0.50 a |
| Ala | 122.48 ± 1.44 a,b | 102.14 ± 0.44 c | 119.91 ± 5.49 a | 129.07 ± 1.35 b | 110.32 ± 0.18 c | 53.27 ± 5.05 d | 27.39 ± 1.93 e | 51.25 ± 2.57 d |
| Tyr | 109.32 ± 8.09 a | 65.03 ± 3.91 b | 105.37 ± 9.45 a | 60.84 ± 3.21 b | 1.50 ± 0.00 c | N.D. c | 70.59 ± 5.97 b | 8.95 ± 0.59 c |
| Val | 80.58 ± 17.11 a | 30.55 ± 1.64 b | 57.01 ± 2.18 c | 108.11 ± 6.48 d | 68.17 ± 0.16 a,c | 65.87 ± 4.74 a,c | 53.48 ± 2.26 c | 9.46 ± 0.45 e |
| Trp | 58.49 ± 0.00 a | N.D. b | 24.90 ± 4.95 c | 7.14 ± 1.11 d | N.D. b | 16.02 ± 0.29 e | 22.28 ± 3.69 c,e | N.D. b |
| Phe | 128.85 ± 11.45 a | 78.99 ± 4.99 d,e | 106.08 ± 6.86 b | 50.30 ± 3.28 f,g | 57.66 ± 0.19 e,f | 82.09 ± 8.89 c,d | 100.78 ± 9.26 b,c | 33.56 ± 3.73 g |
| Ile | 59.40 ± 0.90 a | 7.41 ± 0.94 b | 29.17 ± 2.52 c | 14.82 ± 1.34 d | 13.99 ± 0.38 d | 16.12 ± 3.09 d | 11.21 ± 2.23 b,d | N.D. e |
| Leu | 362.85 ± 11.29 a | 240.14 ± 5.48 b | 259.76 ± 2.93 c | 257.44 ± 5.72 b,c | 121.31 ± 2.89 d | 167.21 ± 5.23 e | 194.98 ± 11.01 f | 49.74 ± 1.08 g |
| T 1 | 1460.57 ± 43.53 a | 843.51 ± 12.84 b | 1218.54 ± 41.87 c | 1111.11 ± 23.76 d | 626.78 ± 4.69 e | 718.02 ± 22.88 f | 666.30 ± 24.32 e,f | 317.98 ± 7.46 g |
| Condition | ||||||||
|---|---|---|---|---|---|---|---|---|
| VFA | A | B | C | D | E | F | G | H |
| iC5 | 5.07 ± 0.45 a,b | 12.40 ± 2.47 c | 5.52 ± 1.29 a | 4.14 ± 0.08 a,b | 11.77 ± 1.26 c | 2.12 ± 0.53 b,d | 0.28 ± 0.02 d | 0.36 ± 0.01 d |
| C5 | 4.74 ± 0.41 a,b | 4.28 ± 0.90 a | 43.94 ± 0.62 c | 7.73 ± 0.40 b,d | 10.58 ± 2.99 d | 0.27 ± 0.02 e | 0.71 ± 0.15 e | 0.35 ± 0.09 e |
| C6 | 679.33 ± 5.85 a,b | 230.47 ± 52.62 c,d | 712.74 ± 14.44 a,b | 826.58 ± 144.78 b | 1689.07 ± 221.27 e | 515.68 ± 126.03 a,d | 8.78 ± 0.23 c | 26.09 ± 2.94 c |
| C7 | 11.94 ± 1.47 a | 10.95 ± 1.69 a | 11.85 ± 0.42 a | 16.53 ± 1.28 a | 28.63 ± 5.52 b | 15.60 ± 0.26 a | 1.15 ± 0.27 c | 0.31 ± 0.05 c |
| C8 | 736.15 ± 78.56 a.b | 555.73 ± 80.34 a | 697.15 ± 6.72 a,b | 666.96 ± 113.86 a,b | 1403.49 ± 185.82 c | 910.45 ± 19.95 b | 90.76 ± 9.08 d | 21.03 ± 0.43 d |
| C9 | 9.41 ± 2.51 a,b,c | 6.78 ± 1.54 c,d | 8.84 ± 0.69 b,c,d | 7.12 ± 2.09 c,d | 15.53 ± 4.31 a | 14. 51 ± 2.42 a,b | 3.12 ± 0.83 d | 2.72 ± 0.16 d |
| C10 | 742.24 ± 8.83 a,b,c | 601.01 ± 153.83 a | 609.91 ± 22.82 a,b | 882.07 ± 133.13 c | 716.96 ± 139.11 a,b,c | 861.11 ± 43.09 b,c | 203.22 ± 43.59 d | 16.57 ± 2.04 d |
| C12 | 77.27 ± 12.43 a,b | 47.24 ± 4.04 a | 61.34 ± 1.79 a,b | 70.40 ± 11.21 a,b | 73.72 ± 18.16 a,b | 89.51 ± 0.58 b | 72.63 ± 17.83 a | 2.43 ± 0.37 c |
| Total 1 | 2266.15 ± 92.85 a | 1468.87 ± 18.48 b | 2151.28 ± 44.13 a,b | 2481.52 ± 402.64 a | 3949.74 ± 578.44 c | 2409.24 ± 145.39 a | 380.65 ± 69.29 d | 69.86 ± 5.60 d |
| Condition | ||||||||
|---|---|---|---|---|---|---|---|---|
| Ester | A | B | C | D | E | F | G | H |
| Ethyl butanoate | 28.86 ± 4.68 a | 7.36 ± 2.05 b | 6.06 ± 0.72 b | 23.27 ± 3.54 a | 29.40 ± 3.05 a | 11.08 ± 1.19 b | 67.31 ± 7.57 c | 6.97 ± 0.86 b |
| Ethyl isovalerate | N.D. a | N.D. a | N.D. a | N.D. a | N.D. a | N.D. a | 0.50 ± 0.07 b | N.D. a |
| Ethyl valerate | 0.36 ± 0.10 a | 0.59 ± 0.02 a | 0.15 ± 0.02 a | 0.26 ± 0.06 a | N.D. a | 0.15 ± 0.01 a | 4.85 ± 0.78 b | N.D. a |
| Ethyl hexanoate | 34.89 ± 9.08 a,b | 10.18 ± 0.46 c,d | 13.65 ± 2.20 c,d | 23.13 ± 2.37 a,c | 35.80 ± 5.42 a,b | 16.24 ± 3.38 c,d | 49.15 ± 8.16 b | 7.70 ± 2.27 d |
| Ethyl Heptanoate | 1.48 ± 0.18 a,b | 0.85 ± 0.13 a,c | 0.98 ± 0.18 a | 0.62 ± 0.04 a,c | 2.15 ± 0.36 b | 1.06 ± 0.27 a | 9.27 ± 0.72 d | N.D. c |
| Ethyl Octanoate | 82.75 ± 24.68 a,b | 28.25 ± 6.44 b,c | 15.16 ± 4.61 c | 18.83 ± 5.19 c | 116.67 ± 30.03 a | 66.90 ± 22.04 a,b,c | 445.79 ± 32.71 d | 18.28 ± 4.51 c |
| Ethyl Nonanoate | 1.17 ± 0.21 a | 0.30 ± 0.06 a | 0.16 ± 0.00 a | 0.73 ± 01 a | 1.28 ± 0.13 a | 0.82 ± 0.21 a | 16.57 ± 2.69 b | 0.24 ± 0.05 a |
| Ethyl decanoate | 166.50 ± 13.11 a | 24.61 ± 5.77 a | 26.34 ± 6.42 a | 27.97 ± 4.01 a | 128.24 ± 35.00 a | 77.55 ± 21.91 a | 1954.83 ± 135.72 b | 27.64 ± 4.06 a |
| Ethly dodecanoate | 68.30 ± 3.56 a | 26.34 ± 7.59 a | 35.93 ± 8.36 a | 22.33 ± 4.00 a | 107.83 ± 10.97 a | 49.04 ± 13.40 a | 878.09 ± 145.55 b | 6.05 ± 0.23 a |
| Total 1 | 384.32 ± 48.46 a | 98.46 ± 22.52 b,c | 98.43 ± 20.70 b,c | 117.15 ± 19.22 b,c | 421.37 ± 84.96 a | 222.83 ± 62.41 b | 3426.36 ± 43.08 d | 66.88 ± 10.15 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araújo-Rodrigues, H.; Martins, A.P.L.; Tavaria, F.K.; Dias, J.; Santos, M.T.; Alvarenga, N.; Pintado, M.E. Impact of LAB from Serpa PDO Cheese in Cheese Models: Towards the Development of an Autochthonous Starter Culture. Foods 2023, 12, 701. https://doi.org/10.3390/foods12040701
Araújo-Rodrigues H, Martins APL, Tavaria FK, Dias J, Santos MT, Alvarenga N, Pintado ME. Impact of LAB from Serpa PDO Cheese in Cheese Models: Towards the Development of an Autochthonous Starter Culture. Foods. 2023; 12(4):701. https://doi.org/10.3390/foods12040701
Chicago/Turabian StyleAraújo-Rodrigues, Helena, António P. L. Martins, Freni K. Tavaria, João Dias, Maria Teresa Santos, Nuno Alvarenga, and Manuela E. Pintado. 2023. "Impact of LAB from Serpa PDO Cheese in Cheese Models: Towards the Development of an Autochthonous Starter Culture" Foods 12, no. 4: 701. https://doi.org/10.3390/foods12040701
APA StyleAraújo-Rodrigues, H., Martins, A. P. L., Tavaria, F. K., Dias, J., Santos, M. T., Alvarenga, N., & Pintado, M. E. (2023). Impact of LAB from Serpa PDO Cheese in Cheese Models: Towards the Development of an Autochthonous Starter Culture. Foods, 12(4), 701. https://doi.org/10.3390/foods12040701







