Improvement of Raw and Pasteurized Milk Quality through the Use of Lactoperoxidase Systems (LPSs) along the Dairy Value Chain, under Real Conditions in Ethiopia
Abstract
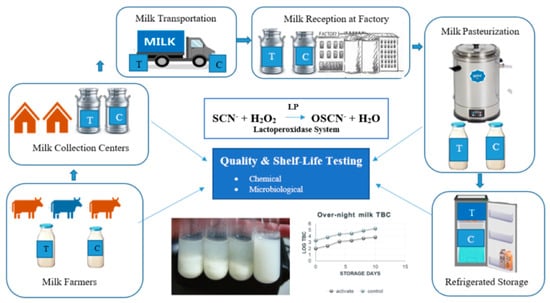
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Study Design, Sample Size, and Sampling Technique
2.3. Collection of Laboratory Sample, Transportation, and Storage
2.4. Activation of LPSs in Raw Milk
2.5. Experimental Trial and Procedure
2.6. Validation of Pasteurization and Shelf Life Evaluation of the Pasteurized Milk
2.7. Assessment of Microbiological Quality of Milk
2.7.1. Total Bacterial Count (TBC)
- V =the volume applied to each plate
- N = the number of plates counted at first dilution
- n2 = the number of plates counted at second dilution
- d = dilution from which first count was obtained
- N = the average plate count.
2.7.2. Enumeration of Total Coliform Count (TCC) and E. coil
- V = the volume of culture plates
- d = the dilution corresponding to the first dilution retained
2.8. Determination of Milk Quality Indicators
2.8.1. Determination of pH
2.8.2. Determination of Titratable Acidity (TTA)
2.8.3. Determination of Thiocyanate Concentration
2.9. Statistical Analysis
3. Results
3.1. Effect of LPSs on the Microbiological Quality of Raw Milk along the Dairy Value Chain
3.2. Effect of LPSs on the Microbiological Shelf Life of Pasteurized Milk
Effect of LPSs on the TBC and TCC of Pasteurized Milk
3.3. Comparison of Microbiological Quality with Ethiopian Standards
3.4. Effect of LPSs on Quality Test of Raw and Pasteurized Milk along the Dairy Value Chain
pH, TTA, and Thiocyanate Concentration
4. Discussion
4.1. Effect of LPSs on the Microbiological Quality of Farmer’s Milk
4.2. Effect of LPSs on the Microbiological Quality of Collector’s Milk
4.3. Effect of LPSs on the Microbiological Shelf Life of Pasteurized Milk
4.4. Effect of LPSs on the Quality of Farmer’s Milk
4.5. Effect of LPSs on the Quality of Collector’s Milk
4.6. Effect of LPSs on the Keeping Quality of Pasteurized Milk
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sarkar, S. Microbiological Considerations: Pasteurized Milk. Int. J. Dairy Sci. 2015, 10, 206–218. [Google Scholar] [CrossRef]
- Seifu, E.; Buys, E.M.; Donkin, E. Significance of the lactoperoxidase system in the dairy industry and its potential applications: A review. Trends Food Sci. Technol. 2005, 16, 137–154. [Google Scholar] [CrossRef]
- Zebib, H.; Abate, D.; Woldegiorgis, A.Z. Nutritional quality and adulterants of raw milk, pasteurized milk and cottage cheese collected along value chain from three regions of Ethiopia. Heliyon 2022, 9, 15922. [Google Scholar] [CrossRef] [PubMed]
- FAO. Guidelines for the preservation of raw milk by use of the lactoperoxidase system. CAC/GL, 13-1991.
- FAO. Manual on the use of the LP-system in milk handling and preservation. In Codex Alimentarius; Food and Agriculture Organization of the United Nations: Rome, Italy, 1999. [Google Scholar]
- Chamberlain, A. Milk Production in the Tropics; Intermediate Tropical Agriculture Series; Longman scientific and technical: London, UK, 1993; p. 178. [Google Scholar]
- Bennet, A. The Lactoperoxidase System (LPs) of milk preservation. In Proceedings of the FAO E-mail Conference on “Small-Scale Milk Collection and Processing in Developing Countries”, Rome, Italy, 6 June–3 August 2000. [Google Scholar]
- Barrett, N.E.; Grandison, A.S.; Lewis, M.J. Contribution of the lactoperoxidase system to the keeping quality of pasteurized milk. J. Dairy Res. 1999, 66, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Marks, N.; Grandison, A.; Lewis, M. Challenge testing of the lactoperoxidase system in pasteurized milk. J. Appl. Microbiol. 2001, 91, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Özer, B. Natural Anti-Microbial Systems|Lactoperoxidase and Lactoferrin. In Encyclopedia of Food Microbiology, 2nd ed.; Elsevier Inc.: Berkeley, CA, USA, 2014. [Google Scholar]
- Gemechu, A.T. Assessment of safety and quality of raw whole cow milk produced and marketed by smallholders in central highlands of Ethiopia. Food Sci. Qual. Manag. 2016, 49, 63–71. [Google Scholar]
- Bille, P.; Haradoeb, B.; Shigwedha, N. Evaluation of chemical and bacteriological quality of raw milk from Neudamm dairy farm in Namibia. Afr. J. Food Agric. Nutr. Dev. 2010, 9, 1511–1523. [Google Scholar] [CrossRef]
- Getachew Felleke. A Review of the Small Scale Dairy Sector - Ethiopia. FAO Prevention of Food Losses Programme: Milk and Dairy Products, Post-harvest Losses and Food Safety in Sub Saharan Africa and the Near East; FAO: Addis Ababa, Ethiopia, 2003. [Google Scholar]
- FAO/WHO. Benefits and Potential Risks of the Lactoperoxidase System of Raw Milk Preservation; Report of FAO/Technical Meeting; FAO/WHO headquarters: Rome, Italy, 2005; p. 73. [Google Scholar]
- FAO. Technical and Investment Guidelines for Milk Cooling Centers; Moffat, F., Khanal, S., Bennett, A., Thapa, T.B., Malakaran, S., Eds.; George: Rome, Italy, 2016. [Google Scholar]
- Kassa, F.; Yilma, Z.; Assefa, G.; Bekele, T.; Gojam, Y.; Nebiyu, R.; Kassa, B. Evaluation of Lactoperoxidase system as raw milk preservative at different storage temperature conditions in the central highlands of Ethiopia. Development 2013, 25. [Google Scholar]
- Fanta, D.G.; Hani, S.; Fufa, D.; Takele, B.T.; Dinka, A. Assessment of the Effect of ActivatedLactoperoxidase System on keeping quality of raw cow milk under different climatic zones of Ethiopia. Dairy Vet. Sci. J. 2019, 10, 555796. [Google Scholar] [CrossRef]
- CSA Livestock Sample Survey (AgSSLV 2010–2011); Central Statistical Agency of Ethiopia (CSA): Addis Ababa, Ethiopia, 2011.
- ES ISO 707; Milk and Milk Products-Guidance on Milk Sampling, 1st ed. Ethiopian Standards Agency: Addis Ababa, Ethiopia, 2012; pp. 1–45.
- Tetra Pack. Dairy Processing Handbook; Tetra Pak Processing Systems AB: Lund, Sweden, 1995; p. 442. [Google Scholar]
- FDA. Center for Food Safety and Applied Nutrition. Bacteriological Analytical Manual; Food and Drug Administration: Washington, DC, USA, 2001.
- 3M Food Safety. Petrifilm E. coli/Coliform Count Plate Interpretation Guide; 3M Food Safety: Minnesota, MN, USA, 2017. [Google Scholar]
- AOAC. Official Methods of Analysis, 19th ed.; Association of Official Analytical Chemists: Gaithersburg, MD, USA, 2016. [Google Scholar]
- East African Standards. Pasteurized Milk-Specification, EAS 69:2006. East African Community Arusha, Tanzania. 2006. Available online: https://law.resource.org/pub/eac/ibr/eas.69.2006.html (accessed on 25 March 2024).
- ES 3462; Pasteurized Liquid Milk-Specification, 2nd ed. Ethiopian Standard: Addis Ababa, Ethiopia, 2009; pp. 1–5.
- Sepúlveda, N.; Muñoz, A.; Jara, R. Evaluacion de un activador del Sistema Lactoperoxidasaen leche sin refrigerar recolectada de pequenos productores. Rev. Científica De La Fac. De Cienc. Vet. 2003, 13, 12–18. [Google Scholar]
- Ponce, P. Lactoperoxidase system under tropical conditions: Use, advantages and limitations in conservation of raw milk and potential applications. Rev. Salud Anim. 2010, 32, 146. [Google Scholar]
- Amenu, B.; Eshetu, M.; Hailu, Y.; Hansen, E.B. Activation of Lactoperoxidase System: Evaluation of the Acidification Rate, Microbial Quality, and Shelf Life Of Camel and Cow Milk. East Afr. J. Sci. 2017, 11, 107–116. [Google Scholar]
- Nigussie, H.; Seifu, E. Effect of the lactoperoxidase system and container smoking on the microbial quality of cows’ milk produced in Kombolcha woreda, eastern Ethiopia. Livest. Res. Rural. Dev. 2007, 19, 157. [Google Scholar]
- Reiter, B. The biological significance of the non-immunoglobulin protective proteins in milk: Lysozyme, lactoferrin, lactoperoxidase. In Developments in Dairy Chemistry—3: Lactose and Minor Constituents; Springer: New York City, NY, USA, 1985; pp. 281–336. [Google Scholar]
- Zapico, P.; Gaya, P.; Nuñez, M.; Medina, M.; Zapico, P.G.P.; Althaus, R.; Torres, A.; Peris, C.; Beltran, M.C.; Fernandez, N.; et al. Goats’ Milk Lactoperoxidase System Against Listeria monocytogenes. J. Food Prot. 1993, 56, 988–990. [Google Scholar] [CrossRef]
- Thomas, E.L.; Aune, T.M. Lactoperoxidase, peroxide, thiocyanate antimicrobial system: Correlation of sulfhydryl oxidation with antimicrobial action. Infect. Immun. 1978, 20, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Seifu, E.; Buys, E.; Donkin, E. Effect of the lactoperoxidase system on the activity of mesophilic cheese starter cultures in goat milk. Int. Dairy J. 2003, 13, 953–959. [Google Scholar] [CrossRef]
- Pruitt, K.M.; Njage, D.N. The lactoperoxidase system of bovine and human milk. In Oxidative Enzymes in Foods; Robinson, D.S., Eskin, N.A.M., Eds.; Elsevier Applied Science: London, UK, 1991; pp. 133–174. [Google Scholar]
- Welearegay, H.; Yilma, Z.; Tekle-Giorgis, Y. Hygienic practices and microbiological quality of raw milk produced under different farm size in Hawassa, southern Ethiopia. Agric. Res. Rev. 2012, 1, 1132–1142. [Google Scholar]
- Campos-Vallejo, M.; Puga-Torres, B.; Núñez-Naranjo, L.; De la Torre-Duque, D.; Morales-Arciniega, S.; Vayas, E. Evaluation of the Use of Sodium Thiocyanate and Sodium Percarbonate in the Activation of the Lactoperoxidase System in the Conservation of Raw Milk without Refrigeration in the Ecuadorian Tropics. Food Nutr. Sci. 2017, 8, 526–534. [Google Scholar] [CrossRef]
- Schlorke, D.; Atosuo, J.; Flemmig, J.; Lilius, E.M.; Arnhold, J. Impact of cyanogen iodide in killing of Escherichia coli by the lactoperoxidase-hydrogen peroxide-(pseudo) halide system. Free. Radic. Res. 2016, 50, 1287–1295. [Google Scholar] [CrossRef]
- Pokhrel, P.; Das, S.K.L. Study on the Extension of Shelf-life by Activation of Inherent Lactoperoxidase System in Raw Cow Milk. J. Food Sci. Technol. Nepal 2012, 7, 57–60. [Google Scholar] [CrossRef]
- Frazier, W.C.; Westhoff, D.C. Food microbiology, 4th ed.; Tata McGraw-Hill Publishing Company Limited: New Delhi, India, 1995. [Google Scholar]
- Mwaikambo, J.J.; Kurwijila, R.L.; Ryoba, R.Z. The effect of activation of lactoperoxide system (LPS) on the quality and shelf life of in-pouch pasteurised milk. Tanzan. J. Agric. Sci. 2003, 6, 1–12. [Google Scholar]
- Trujillo, A.J.; Pozo, P.I.; Guamis, B. Effect of heat treatment on lactoperoxidase activity in caprine milk. Small Rumin. Res. 2007, 67, 243–246. [Google Scholar] [CrossRef]
- Kamau, D.N.; Doores, S.; Pruitt, K.M. Activation of the lactoperoxidase system prior to pasteurization for shelf-life extension of milk. Milchwissenschaft 1991, 46, 213–214. [Google Scholar]
- Kamau, D.N.; Doores, S.; Pruitt, K.M. Antibacterial activity of the lactoperoxidase system against Listeria monocytogenes and Staphylococcus aureus in milk. J. Food Prot. 1990, 53, 1010–1014. [Google Scholar] [CrossRef] [PubMed]
- Fonteh, F.A.; Grandison, A.S.; Lewis, M.J.; Niba, A.T. The keeping quality of LPS-activated milk in the western highlands of Cameroon. Livest. Res. Rural. Dev. 2006, 17, 114. [Google Scholar]
- Njage, K.M.P.; Wangoh, J. Impact of the lactoperoxidase system on activity of selected lactic starter cultures in camel milk. Food 2008, 14, 70–74. [Google Scholar]
- Asaah, N.O.; Fonteh, F.; Kamga, P.; Mendi, S.; Imele, H. Activation of the lactoperoxidase system as a method of preserving raw milk in areas without cooling facilities. Afr. J. Food Agric. Nutr. Dev. 2007, 7, 1–15. [Google Scholar] [CrossRef]
- Korhonen, H. A New Method for Preserving Raw Milk: The Lactoperoxidase Antibacterial System; Revue Mondiale de Zootechnie (FAO)-Revista Mundial de Zootecnia (FAO): Rome, Italy, 1980. [Google Scholar]
- Kumar, S.; Mathur, B.N. Preservation of raw buffalo milk through activation of LP-system. Part I. Under farm conditions. Indian J. Dairy Sci. 1989, 42, 339–341. [Google Scholar]


| Study Site | Treatments | Farmers | Collectors | Factories | Total | |||
|---|---|---|---|---|---|---|---|---|
| MM | OM | MM | OM | MM | OM | |||
| Selale | Activated | 10 | 10 | 5 | 5 | 1 | 1 | 32 |
| Control | 10 | 10 | 5 | 5 | 1 | 1 | 32 | |
| Holeta | Activated | 10 | 10 | 5 | 5 | 1 | 1 | 32 |
| Control | 10 | 10 | 5 | 5 | 1 | 1 | 32 | |
| Asella | Activated | 10 | 9 | 5 | 5 | 1 | 1 | 32 |
| Control | 10 | 9 | 5 | 5 | 1 | 1 | 32 | |
| D/zeyit | Activated | 9 | 9 | 5 | 5 | 1 | 1 | 30 |
| Control | 9 | 9 | 5 | 5 | 1 | 1 | 30 | |
| Total | 78 | 76 | 40 | 40 | 8 | 8 | 250 | |
| Value Chain | Bacteria | Sampling Time | N | Treatment | Mean | Std. Err. | p-Value |
|---|---|---|---|---|---|---|---|
| Farmers | Log10 TBC | Morning | 39 | Control | 6.73 | 0.18 | 0.0003 |
| 39 | Activated | 5.79 | 0.19 | ||||
| Overnight | 38 | Control | 7.31 | 0.16 | 0.001 | ||
| 38 | Activated | 6.55 | 0.17 | ||||
| Log10 TCC | Morning | 39 | Control | 4.70 | 0.17 | 0.0002 | |
| 39 | Activated | 3.77 | 0.18 | ||||
| Overnight | 38 | Control | 5.04 | 0.29 | 0.0196 | ||
| 38 | Activated | 4.19 | 0.29 | ||||
| Log10 E. coli | Morning | 39 | Control | 0.84 | 0.25 | 0.0067 | |
| 39 | Activated | 0.18 | 0.09 | ||||
| Overnight | 38 | Control | 0.52 | 0.18 | 0.0022 | ||
| 38 | Activated | 0.00 | (omitted) | ||||
| Collectors | Log10 TBC | Morning | 20 | Control | 6.99 | 0.12 | 1 × 10−5 |
| 20 | Activated | 6.15 | 0.13 | ||||
| Overnight | 20 | Control | 7.78 | 0.16 | 0.0008 | ||
| 20 | Activated | 6.96 | 0.18 | ||||
| Log10 TCC | Morning | 20 | Control | 5.63 | 0.11 | 1 × 10−5 | |
| 20 | Activated | 4.63 | 0.14 | ||||
| Overnight | 20 | Control | 6.41 | 0.11 | 1 × 10−5 | ||
| 20 | Activated | 5.27 | 0.14 | ||||
| Log10 E. coli | Morning | 20 | Control | 0.93 | 0.31 | 0.0206 | |
| 20 | Activated | 0.20 | 0.16 | ||||
| Overnight | 20 | Control | 0.63 | 0.25 | 0.0085 | ||
| 20 | Activated | 0.00 | (omitted) |
| Quality | Value Chain | Sampling Time | Treatment | N | N (% of Passes) | p-Value |
|---|---|---|---|---|---|---|
| TBC | Farmers | Morning | Activated | 39 | 20 (51%) | 0.04 |
| Control | 39 | 11 (28%) | ||||
| Overnight | Activated | 38 | 15 (40%) | 0.02 | ||
| Control | 38 | 6 (16%) | ||||
| Collectors | Morning | Activated | 20 | 9 (45%) | 0.001 * | |
| Control | 20 | - | ||||
| Overnight | Activated | 20 | 3 (15%) | 0.072 | ||
| Control | 20 | - | ||||
| TCC | Farmers | Morning | Activated | 39 | 32 (82%) | 0.004 * |
| Control | 39 | 20 (51%) | ||||
| Overnight | Activated | 38 | 23 (60.5%) | 0.039 * | ||
| Control | 38 | 14 (36.9%) | ||||
| Collectors | Morning | Activated | 20 | 14 (70%) | 0.0001 | |
| Control | 20 | 1 (5%) | ||||
| Overnight | Activated | 20 | 6 (30%) | 0.008 | ||
| Control | 20 | 0 | ||||
| E-coli | Farmers | Morning | Activated | 39 | 35 (89.7%) | 0.77 |
| Control | 39 | 29 (74.4%) | ||||
| Overnight | Activated | 38 | 38 (100%) | 0.005 * | ||
| Control | 38 | 31 (81.6%) | ||||
| Collectors | Morning | Activated | 20 | 18 (90%) | 0.058 | |
| Control | 20 | 13 (65%) | ||||
| Overnight | Activated | 20 | 20 (100%) | 0.017 * | ||
| Control | 20 | 15 (75%) |
| Value Chain | Quality Test | Sampling Time | N | Treatment | Mean | Std. Err. | p-Value |
|---|---|---|---|---|---|---|---|
| Farmers | pH | Morning | 39 | Control | 6.33 | 0.072 | 0.0359 * |
| 39 | Activated | 6.51 | 0.044 | ||||
| Overnight | 38 | Control | 6.04 | 0.102 | 0.0288 * | ||
| 38 | Activated | 6.31 | 0.062 | ||||
| TTA (%) | Morning | 39 | Control | 0.21 | 0.010 | 0.1087 | |
| 39 | Activated | 0.19 | 0.009 | ||||
| Overnight | 38 | Control | 0.25 | 0.016 | 0.0019 * | ||
| 38 | Activated | 0.20 | 0.008 | ||||
| Thiocyanate (ppm) | Morning | 39 | Control | 11.08 | 1.097 | 0.1238 | |
| 39 | Activated | 13.64 | 1.219 | ||||
| Overnight | 38 | Control | 12.12 | 1.356 | 0.1565 | ||
| 38 | Activated | 15.14 | 1.614 | ||||
| Collectors | pH | Morning | 20 | Control | 6.223 | 0.064 | 0.014 |
| 20 | Activated | 6.404 | 0.046 | ||||
| Overnight | 20 | Control | 6.056 | 0.061 | 0.0024 | ||
| 20 | Activated | 6.304 | 0.055 | ||||
| TTA | Morning | 20 | Control | 0.263 | 0.012 | 0.0143 | |
| 20 | Activated | 0.227 | 0.010 | ||||
| Overnight | 20 | Control | 0.352 | 0.024 | 0.0054 | ||
| 20 | Activated | 0.270 | 0.019 | ||||
| Thiocyanate (ppm) | Morning | 20 | Control | 13.110 | 1.595 | 0.5155 | |
| 20 | Activated | 13.025 | 1.488 | ||||
| Overnight | 20 | Control | 15.487 | 1.745 | 0.6731 | ||
| 20 | Activated | 14.518 | 1.244 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashenafi, T.; Zebib, H.; Woldegiorgis, A.Z. Improvement of Raw and Pasteurized Milk Quality through the Use of Lactoperoxidase Systems (LPSs) along the Dairy Value Chain, under Real Conditions in Ethiopia. Foods 2024, 13, 1272. https://doi.org/10.3390/foods13081272
Ashenafi T, Zebib H, Woldegiorgis AZ. Improvement of Raw and Pasteurized Milk Quality through the Use of Lactoperoxidase Systems (LPSs) along the Dairy Value Chain, under Real Conditions in Ethiopia. Foods. 2024; 13(8):1272. https://doi.org/10.3390/foods13081272
Chicago/Turabian StyleAshenafi, Tigist, Haftom Zebib, and Ashagrie Zewdu Woldegiorgis. 2024. "Improvement of Raw and Pasteurized Milk Quality through the Use of Lactoperoxidase Systems (LPSs) along the Dairy Value Chain, under Real Conditions in Ethiopia" Foods 13, no. 8: 1272. https://doi.org/10.3390/foods13081272
APA StyleAshenafi, T., Zebib, H., & Woldegiorgis, A. Z. (2024). Improvement of Raw and Pasteurized Milk Quality through the Use of Lactoperoxidase Systems (LPSs) along the Dairy Value Chain, under Real Conditions in Ethiopia. Foods, 13(8), 1272. https://doi.org/10.3390/foods13081272