Research Progress on Nanotechnology-Driven Enzyme Biosensors for Electrochemical Detection of Biological Pollution and Food Contaminants
Abstract
1. Introduction
2. Principle of Enzyme Electrode Sensors
3. Classification of Enzyme Electrode Sensors
4. Research and Application of Enzyme Electrode Sensors in Food Safety Detection
4.1. Drug Residue Detection
4.2. Biotoxin Detection
4.3. Food Additive Testing
4.4. Detection of Foodborne Pathogenic Bacteria
4.5. Food Allergen Detection
5. Application of Nanomaterials in Enzyme Electrode Sensors
5.1. Nanomaterials
5.2. Nanomaterials Promote the Performance of Enzyme Electrode Sensors
5.2.1. Carbon Nanomaterials
5.2.2. Other Nanomaterials
6. Conclusions and Outlooks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Park, D.; Han, S.B.; Shin, H.C.; Choi, Y.S.; Kim, Y.R.; Kim, J.Y.; Jang, G.H.; Eom, M. Validation of analytical methods for newly regulated veterinary drug residues in livestock and fishery products with maximum residue limits in Republic of Korea. Food Chem. 2024, 460, 140705. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Yang, Y.; Shen, Q.; Sun, Y.; Kang, Q.; Shen, D. A novel differential ratiometric molecularly imprinted electrochemical sensor for determination of sulfadiazine in food samples. Food Chem. 2024, 434, 137461. [Google Scholar] [PubMed]
- Nilghaz, A.; Mousavi, S.M.; Amiri, A.; Tian, J.; Cao, R.; Wang, X. Surface-Enhanced Raman Spectroscopy Substrates for Food Safety and Quality Analysis. J. Agric. Food Chem. 2022, 70, 5463–5476. [Google Scholar]
- Chen, C.; Tian, T.; Yu, H.; Yuan, H.; Wang, B.; Xu, Z.; Tian, H. Characterisation of the key volatile compounds of commercial Gouda cheeses and their contribution to aromas according to Chinese consumers’ preferences. Food Chem.-X 2022, 15, 100416. [Google Scholar] [CrossRef]
- Zhang, H.; Chang, Q.; Chen, H.; Xie, Y.; Bai, Y.; Wang, X.; Li, L.; Pang, G. Qualitative and quantitative analysis of pesticide isomers based on collision-induced dissociation of high-resolution mass spectrometry. Microchem. J. 2024, 200, 110338. [Google Scholar]
- Cesewski, E.; Johnson, B.N. Electrochemical biosensors for pathogen detection. Biosens. Bioelectron. 2020, 159, 112214. [Google Scholar]
- Zhu, H.; Li, M.; Cheng, C.; Han, Y.; Fu, S.; Li, R.; Cao, G.; Liu, M.; Cui, C.; Liu, J.; et al. Recent Advances in and Applications of Electrochemical Sensors Based on Covalent Organic Frameworks for Food Safety Analysis. Foods 2023, 12, 4274. [Google Scholar] [CrossRef]
- Clark, L.C.; Lyons, C. Electrode systems for continuous monitoring in cardiovascular surgery. Ann. N. Y. Acad. Sci. 1962, 102, 29–45. [Google Scholar]
- Updike, S.J.; Hicks, G.P. The Enzyme Electrode. Nature 1967, 214, 986. [Google Scholar]
- Zhang, J.L.; Bi, C.Y.; Li, J.-L. A New Technology for Detecting L-Glutamic Acid Sodium in Soy Sauce; China Condiment: Harbin, China, 2014. [Google Scholar]
- Zou, B.; Chu, Y.; Xia, J.; Yao, J. Acetylcholinesterase biosensor based on functionalized surface of carbon nanotubes for monocrotophos detection. Anal. Biochem. 2018, 560, 12–18. [Google Scholar]
- Smart, A.; Crew, A.; Pemberton, R.; Hughes, G.; Doran, O.; Hart, J.P. Screen-printed carbon based biosensors and their applications in agri-food safety—ScienceDirect. TrAC Trends Anal. Chem. 2020, 127, 115898. [Google Scholar] [CrossRef]
- Bu, L.; Guo, L.; Xie, J. An in situ assay of nerve agents enabled by a self-assembled bienzymatic electrochemical biosensor. New J. Chem. 2020, 44, 7460–7466. [Google Scholar] [CrossRef]
- Upadhyay, S.; Sharma, M.K.; Rao, G.R.; Bhattacharya, B.K.; Rao, V.K.; Vijayaraghavan, R. Application of bimetallic nanoparticles modified screen printed electrode for the detection of organophosphate compounds using an enzyme inhibition approach. Anal. Methods 2011, 3, 2246–2253. [Google Scholar] [CrossRef]
- Karra, S.; Gorski, W. Signal Amplification in Enzyme-Based Amperometric Biosensors. Anal. Chem. 2013, 85, 10573–10580. [Google Scholar] [CrossRef]
- Xia, J.J.; Bin, Z.; Wang, P.Y.; Qing, L.; Xin, S.; Rui, J. Acetylcholinesterase biosensors based on ionic liquid functionalized carbon nanotubes and horseradish peroxidase for monocrotophos determination. Bioprocess Biosyst. Eng. 2020, 43, 293–301. [Google Scholar]
- Probst, D.; Lee, I.; Sode, K. The development of micro-sized enzyme sensor based on direct electron transfer type open circuit potential sensing principle. Electrochim. Acta 2022, 426, 140798. [Google Scholar] [CrossRef]
- Hatada, M.; Pavlidis, S.; Sode, K. Development of a glycated albumin sensor employing dual aptamer-based extended gate field effect transistors. Biosens. Bioelectron. 2024, 251, 116118. [Google Scholar] [CrossRef]
- Hsu, C.H.; Hsu, Y.W.; Weng, Y.C. A novel potentiometric sensor based on urease/bovine serum albumin-poly(3,4-ethylenedioxythiophene)/Pt for urea detection. Z. Für Naturforschung B 2016, 71, 277–282. [Google Scholar] [CrossRef]
- Shervedani, R.K.; Amini, A. Direct electrochemistry of dopamine on gold-Agaricus bisporus laccase enzyme electrode: Characterization and quantitative detection. Bioelectrochemistry 2012, 84, 25–31. [Google Scholar] [CrossRef]
- Ertek, B.; Akgül, C.; Dilgin, Y. Photoelectrochemical glucose biosensor based on a dehydrogenase enzyme and NAD+/NADH redox couple using a quantum dot modified pencil graphite electrode. RSC Adv. 2016, 6, 20058–20066. [Google Scholar] [CrossRef]
- Cui, J.; Hou, T.; Wang, Q.; Gao, G.; Bi, S.; Zhou, K.; Li, J.; Wu, D. An Enzyme Assisted Electrochemical Detection System of Purine Intracellular Utilizing MWCNTs-IL Modified Glassy Carbon Electrode. Electrochim. Acta 2015, 180, 360–365. [Google Scholar]
- Kang, J.; Hussain, A.T.; Catt, M.; Trenell, M.; Haggett, B.; Yu, E.H. Electrochemical detection of non-esterified fatty acid by layer-by-layer assembled enzyme electrodes. Sens. Actuators B Chem. 2014, 190, 535–541. [Google Scholar]
- Zhao, Y.; Zhang, W.; Lin, Y.; Du, D. The vital function of Fe3O4@Au nanocomposites for hydrolase biosensor design and its application in detection of methyl parathion. Nanoscale 2013, 5, 1121–1126. [Google Scholar] [CrossRef] [PubMed]
- Marx, S.; Jose, M.V.; Andersen, J.D.; Russell, A.J. Electrospun gold nanofiber electrodes for biosensors. Biosens. Bioelectron. 2011, 26, 2981–2986. [Google Scholar] [CrossRef]
- Goebel, G.; Talke, A.; Ahnert, U.; Lisdat, F. Electrochemical Activity Determination of Catechol-O-methyl Transferase by Selective Dopamine Detection. Chemelectrochem 2019, 6, 4533–4540. [Google Scholar] [CrossRef]
- Taghdisi, S.M.; Danesh, N.M.; Ramezani, M.; Alibolandi, M.; Nameghi, M.A.; Gerayelou, G.; Abnous, K. A novel electrochemical aptasensor for ochratoxin a sensing in spiked food using strand-displacement polymerase reaction. Talanta 2021, 223, 121705. [Google Scholar] [CrossRef]
- Chu, Y.-H.; Zou, B.; Chen, X.-S.; Xia, J.-J.; Huo, S.-H. Research progress in electrochemical enymze electrode sensor in detection of food safety. Sci. Technol. Food Ind. 2017, 17, 335–340. [Google Scholar]
- Liu, S.; Meng, S.; Wang, M.; Li, W.; Dong, N.; Liu, D.; Li, Y.; You, T. In-depth interpretation of aptamer-based sensing on electrode: Dual-mode electrochemical-photoelectrochemical sensor for the ratiometric detection of patulin. Food Chem. 2023, 410, 135450. [Google Scholar] [CrossRef]
- Zhu, L.; Dong, X.X.; Gao, C.B.; Gai, Z.; He, Y.X.; Qian, Z.J.; Liu, Y.; Lei, H.T.; Sun, Y.M.; Xu, Z.L. Development of a highly sensitive and selective electrochemical immunosensor for controlling of rhodamine B abuse in food samples. Food Control 2022, 133, 108662. [Google Scholar]
- Mustapha, A.T.; Zhou, C. Novel assisted/unassisted ultrasound treatment: Effect on respiration rate, ethylene production, enzymes activity, volatile composition, and odor of cherry tomato. LWT-Food Sci. Technol. 2021, 149, 111779. [Google Scholar]
- Lu, B.; Han, F.; Aheto, J.H.; Rashed, M.M.A.; Pan, Z. Artificial bionic taste sensors coupled with chemometrics for rapid detection of beef adulteration. Food Sci. Nutr. 2021, 9, 5220–5228. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Sun, Y.; Shi, J.; Zhang, W.; Zhang, X.; Hang, X.; Li, Z.; Zou, X. Convenient self-assembled PDADMAC/PSS/Au@Ag NRs filter paper for swift SERS evaluate of non-systemic pesticides on fruit and vegetable surfaces. Food Chem. 2023, 424, 136232. [Google Scholar] [PubMed]
- Chen, Z.; Sun, Y.; Zhang, X.; Shen, Y.; Huang, X.; Shi, J.; Li, Z.; Zou, X.; Khalifa, S.A.M. Green and sustainable self-cleaning flexible SERS base: Utilized for cyclic-detection of residues on apple surface. Food Chem. 2024, 441, 138345. [Google Scholar] [PubMed]
- Yu, Y.; Wang, Y.; Okonkwo, C.E.; Chen, L.; Zhou, C. Multimode ultrasonic-assisted decontamination of fruits and vegetables: A review. Food Chem. 2024, 450, 139356. [Google Scholar]
- Xu, Y.; Zhang, W.; Shi, J.; Li, Z.; Huang, X.; Zou, X.; Tan, W.; Zhang, X.; Hu, X.; Wang, X.; et al. Impedimetric aptasensor based on highly porous gold for sensitive detection of acetamiprid in fruits and vegetables. Food Chem. 2020, 322, 126762. [Google Scholar]
- Azam, S.R.; Ma, H.; Xu, B.; Devi, S.; Siddique, M.A.B.; Stanley, S.L.; Bhandari, B.; Zhu, J. Efficacy of ultrasound treatment in the removal of pesticide residues from fresh vegetables: A review. Trends Food Sci. Technol. 2020, 97, 417–432. [Google Scholar]
- Zheng, Q.; Yu, Y.; Fan, K.; Ji, F.; Wu, J.; Ying, Y. A nano-silver enzyme electrode for organophosphorus pesticide detection. Anal. Bioanal. Chem. 2016, 408, 5819–5827. [Google Scholar]
- Miao, S.S.; Wu, M.S.; Ma, L.Y.; He, X.J.; Yang, H. Electrochemiluminescence biosensor for determination of organophosphorous pesticides based on bimetallic Pt-Au/multi-walled carbon nanotubes modified electrode. Talanta 2016, 158, 142–151. [Google Scholar]
- Itoh, T.; Shimomura, T.; Hayashi, A.; Yamaguchi, A.; Teramae, N.; Ono, M.; Tsunoda, T.; Mizukami, F.; Stucky, G.D.; Hanaoka, T.A. Electrochemical enzymatic biosensor with long-term stability using hybrid mesoporous membrane. Analyst 2014, 139, 4654–4660. [Google Scholar]
- Zou, B.; Chu, Y.; Xia, J. Monocrotophos detection with a bienzyme biosensor based on ionic-liquid-modified carbon nanotubes. Anal. Bioanal. Chem. 2019, 411, 2905–2914. [Google Scholar]
- Ji, C.; Tang, X.; Wen, R.; Xu, C.; Wei, J.; Han, B.; Wu, L. A Multienzyme Reaction-Mediated Electrochemical Biosensor for Sensitive Detection of Organophosphorus Pesticides. Biosensors 2024, 14, 62. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Liu, Z.; Jing, Y.; Li, J.; Zhan, H. An acetylcholinesterase biosensor based on ionic liquid functionalized graphene–gelatin-modified electrode for sensitive detection of pesticides. Sens. Actuators B Chem. 2015, 210, 389–397. [Google Scholar] [CrossRef]
- Nesakumar, N.; Sethuraman, S.; Krishnan, U.M.; Rayappan, J.B.B. Electrochemical acetylcholinesterase biosensor based on ZnO nanocuboids modified platinum electrode for the detection of carbosulfan in rice. Biosens. Bioelectron. 2016, 77, 1070–1077. [Google Scholar] [PubMed]
- Magar, H.S.; Fayez, M.; Febbraio, F.; Hassan, R.Y.A. Esterase-2 mutant-based nanostructured amperometric biosensors for the selective determination of paraoxon (Neurotoxin). Anal. Biochem. 2025, 698, 115751. [Google Scholar] [CrossRef]
- Wang, D.; Wang, P.; Liu, D.; Zhou, Z. Fluorometric atrazine assay based on the use of nitrogen-doped graphene quantum dots and on inhibition of the activity of tyrosinase. Microchim. Acta 2019, 186, 527. [Google Scholar]
- Han, E.; Chao, P.; Xiao-Mei, C.; Jian-Rong, C. Electrochemical detection of atrazine pesticide residue based on the inhibition of tyrosinase. Food Sci. Technol. 2015, 40, 344–347. [Google Scholar]
- Song, S.H.; Gao, Z.F.; Guo, X.; Chen, G.H. Aptamer-Based Detection Methodology Studies in Food Safety. Food Anal. Methods 2019, 12, 966–990. [Google Scholar]
- Shin, M.C.; Min, K.A.; Cheong, H.; Moon, C.; Huang, Y.; He, H.; Yang, V.C. Preparation and Characterization of Gelonin-Melittin Fusion Biotoxin for Synergistically Enhanced Anti-Tumor Activity. Pharm. Res. 2016, 33, 2218–2228. [Google Scholar]
- Suarez-Ulloa, V.; Fernandez-Tajes, J.; Aguiar-Pulido, V.; Prego-Faraldo, M.V.; Eirin-Lopez, J.M. Unbiased high-throughput characterization of mussel transcriptomic responses to sublethal concentrations of the biotoxin okadaic acid. Peerj 2015, 3, e1429. [Google Scholar]
- You, F.; Wen, Z.; Yuan, R.; Qian, J.; Long, L.; Wang, K. Sensitive and stable detection of deoxynivalenol based on electrochemiluminescence aptasensor enhanced by 0D/2D homojunction effect in food analysis. Food Chem. 2023, 403, 134397. [Google Scholar] [CrossRef]
- Bin, Z.; Feng, L.; Yan, Y. Biomimetic metalloporphyrin oxidase modified carbon nanotubes for highly sensitive and stable quantification of anti-oxidants tert-butylhydroquinone in plant oil. Food Chem. 2022, 388, 132898. [Google Scholar] [CrossRef] [PubMed]
- Han, E.; Li, L.; Gao, T.; Pan, Y.; Cai, J. Nitrite determination in food using electrochemical sensor based on self-assembled MWCNTs/AuNPs/poly-melamine nanocomposite. Food Chem. 2024, 437, 137773. [Google Scholar]
- Zhang, X.; Fang, J.; Zou, L.; Zou, Y.; Lang, L.; Gao, F.; Hu, N.; Wang, P. A novel sensitive cell-based Love Wave biosensor for marine toxin detection. Biosens. Bioelectron. 2016, 77, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Leonardo, S.; Campàs, M. Electrochemical enzyme sensor arrays for the detection of the biogenic amines histamine, putrescine and cadaverine using magnetic beads as immobilisation supports. Microchim. Acta 2016, 183, 1881–1890. [Google Scholar]
- Young, J.A.; Jiang, X.; Kirchhoff, J.R. Amperometric Detection of Histamine with a Pyrroloquinoline-Quinone Modified Electrode. Electroanalysis 2013, 25, 1589–1593. [Google Scholar] [CrossRef]
- Uludag, Y.; Esen, E.; Kokturk, G.; Ozer, H.; Muhammad, T.; Olcer, Z.; Basegmez, H.I.O.; Simsek, S.; Barut, S.; Gok, M.Y.; et al. Lab-on-a-chip based biosensor for the real-time detection of aflatoxin. Talanta 2016, 160, 381–388. [Google Scholar] [CrossRef]
- Chen, J.; Liu, D.; Li, S.; Yao, D. Development of an amperometric enzyme electrode biosensor for sterigmatocystin detection. Enzym. Microb. Technol. 2010, 47, 119–126. [Google Scholar]
- Catanante, G.L.; Espin, L.; Marty, J.L. Sensitive biosensor based on recombinant PP1α for microcystin detection. Biosens. Bioelectron. 2015, 67, 700–707. [Google Scholar] [CrossRef]
- Radulescu, M.C.; Bucur, M.P.; Bucur, B.; Radu, G.L. Biosensor based on inhibition of monoamine oxidases A and B for detection of β-carbolines. Talanta 2015, 137, 94–99. [Google Scholar] [CrossRef]
- Chen, Z.; Lian, X.; Zhou, M.; Zhang, X.; Wang, C. Quantitation of L-cystine in Food Supplements and Additives Using 1 H qNMR: Method Development and Application. Foods 2023, 12, 2421. [Google Scholar] [CrossRef]
- Zhou, C.; Li, C.; Cui, H.; Lin, L. Metabolomics insights into the potential of encapsulated essential oils as multifunctional food additives. Crit. Rev. Food Sci. Nutr. 2022, 64, 5143–5160. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Luo, J.; Zhao, L.; Zhao, M.; Ouyang, Z.; Yang, M. Influences of different storage environments and packaging materials on the quality of the traditional Chinese health food Herba Menthae Haplocalycis. Food Packag. Shelf Life 2017, 15, 52–61. [Google Scholar]
- Mu, R.; Hong, X.; Ni, Y.; Li, Y.; Zheng, Y. Recent trends and applications of cellulose nanocrystals in food industry. Trends Food Sci. Technol. 2019, 93, 136–144. [Google Scholar]
- Hu, D. Monte Carlo: A flexible and accurate technique for modeling light transport in food and agricultural products. Trends Food Sci. Technol. 2020, 102, 280–290. [Google Scholar]
- Sadeghi, S.; Fooladi, E.; Malekaneh, M. A New Amperometric Biosensor Based on Fe3O4/Polyaniline/Laccase/Chitosan Biocomposite-Modified Carbon Paste Electrode for Determination of Catechol in Tea Leaves. Appl. Biochem. Biotechnol. Part A Enzym. Eng. Biotechnol. 2015, 175, 1603–1616. [Google Scholar]
- Qu, J.; Lou, T.; Wang, Y.; Dong, Y.; Xing, H. Determination of Catechol by a Novel Laccase Biosensor Based on Zinc-Oxide Sol-Gel. Anal. Lett. 2015, 48, 1842–1853. [Google Scholar] [CrossRef]
- Wu, L.; Yin, W.; Tang, K.; Li, D.; Han, H. Enzymatic biosensor of horseradish peroxidase immobilized on Au-Pt nanotube/Au-graphene for the simultaneous determination of antioxidants. Anal. Chim. Acta 2016, 933, 89–96. [Google Scholar] [CrossRef]
- Taghdisi, S.M.; Danesh, N.M.; Ramezani, M.; Abnous, K. A novel M-shape electrochemical aptasensor for ultrasensitive detection of tetracyclines. Biosens. Bioelectron. 2016, 85, 509–514. [Google Scholar]
- Song, H.Y.; Kang, T.F.; Li, N.N.; Lu, L.P.; Cheng, S.Y. Highly sensitive voltammetric determination of kanamycin based on aptamer sensor for signal amplification. Anal. Methods 2016, 8, 3366–3372. [Google Scholar]
- Gan, H.; Liang, Y.; Tang, Y.; Huang, M.; Wan, Y. A practical electrochemical method for determining the inhibitory activity of New Delhi Metallo-β-lactamase-1 inhibitors. Microchem. J. 2023, 195, 109418. [Google Scholar]
- Dai, J.; Bai, M.; Li, C.; Cui, H.; Lin, L. Advances in the mechanism of different antibacterial strategies based on ultrasound technique for controlling bacterial contamination in food industry. Trends Food Sci. Technol. 2020, 105, 211–222. [Google Scholar]
- Qian, J.; Zhang, M.; Dai, C.; Huo, S.; Ma, H. Transcriptomic analysis of Listeria monocytogenes under pulsed magnetic field treatment. Food Res. Int. 2020, 133, 109195. [Google Scholar] [PubMed]
- Cui, H.; Chen, Y.; Aziz, T.; Al-Asmari, F.; Alwethaynani, M.S.; Shi, C.; Lin, L. Antibacterial mechanisms of diacetyl on Listeria monocytogenes and its application in Inner Mongolian cheese preservation via gelatin-based edible films. Food Control 2025, 168, 110920. [Google Scholar]
- Alenyorege, E.A.; Ma, H.; Ayim, I.; Aheto, J.H.; Hong, C.; Zhou, C. Reduction of Listeria innocua in fresh-cut Chinese cabbage by a combined washing treatment of sweeping frequency ultrasound and sodium hypochlorite. LWT 2019, 101, 410–418. [Google Scholar]
- Zhang, C.; Li, C.; Abdel-Samie, M.A.; Cui, H.; Lin, L. Unraveling the inhibitory mechanism of clove essential oil against Listeria monocytogenes biofilm and applying it to vegetable surfaces. LWT-Food Sci. Technol. 2020, 134, 110210. [Google Scholar]
- Chen, Q.; Lin, J.; Gan, C.; Wang, Y.; Wang, D.; Xiong, Y.; Lai, W.; Li, Y.; Wang, M. A sensitive impedance biosensor based on immunomagnetic separation and urease catalysis for rapid detection of Listeria monocytogenes using an immobilization-free interdigitated array microelectrode. Biosens. Bioelectron. 2015, 74, 504–511. [Google Scholar] [CrossRef]
- Wang, D.; Wang, Z.; Chen, J.; Kinchla, A.J.; Nugen, S.R. Rapid detection of Salmonella using a redox cycling-based electrochemical method. Food Control 2016, 62, 81–88. [Google Scholar]
- Ölcer, Z.; Esen, E.; Ersoy, A.; Budak, S.; Kaya, D.S.; Gök, M.Y.; Barut, S.; Üstek, D.; Uludag, Y. Microfluidics and nanoparticles based amperometric biosensor for the detection of cyanobacteria (Planktothrix agardhii NIVA-CYA 116) DNA. Biosens. Bioelectron. 2015, 70, 426–432. [Google Scholar]
- Cassotta, M.; Forbes-Hernández, T.Y.; Iglesias, R.C.; Ruiz, R.; Battino, M. Links between Nutrition, Infectious Diseases, and Microbiota: Emerging Technologies and Opportunities for Human-Focused Research. Nutrients 2020, 12, 1827. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Z.H.; Zabed, H.M.; Yun, J.; Zhang, G.; Qi, X. An Insight into the Roles of Dietary Tryptophan and its Metabolites in Intestinal Inflammation and Inflammatory Bowel Disease. Mol. Nutr. Food Res. 2021, 65, 2000461. [Google Scholar]
- Suo, H.; Shishir, M.R.I.; Xiao, J.; Wang, M.; Chen, F.; Cheng, K.W. Red Wine High-Molecular-Weight Polyphenolic Complex: An Emerging Modulator of Human Metabolic Disease Risk and Gut Microbiota. J. Agric. Food Chem. 2021, 69, 10907–10919. [Google Scholar] [PubMed]
- Moradi, S.Z.; Jalili, F.; Farhadian, N.; Joshi, T.; Wang, M.; Zou, L.; Cao, H.; Farzaei, M.H.; Xiao, J. Polyphenols and neurodegenerative diseases: Focus on neuronal regeneration. Crit. Rev. Food Sci. Nutr. 2022, 62, 3421–3436. [Google Scholar] [PubMed]
- Liu, H.; Malhotra, R.; Peczuh, M.W.; Rusling, J.F. Electrochemical immunosensors for antibodies to peanut allergen ara h2 using gold nanoparticle-peptide films. Anal. Chem. 2010, 82, 5865–5871. [Google Scholar] [PubMed]
- Sun, X.; Jia, M.; Ji, J.; Guan, L.; Zhang, Y.; Tang, L.; Li, Z. Enzymatic amplification detection of peanut allergen Ara h1 using a stem-loop DNA biosensor modified with a chitosan-mutiwalled carbon nanotube nanocomposite and spongy gold film. Talanta 2015, 131, 521–527. [Google Scholar]
- Montiel, R.V.; Campuzano, S.; Conzuelo, F.; Torrente-Rodríguez, R.M.; Gamella, M.; Reviejo, A.J.; Pingarrón, J.M. Electrochemical magnetoimmunosensing platform for determination of the milk allergen β-lactoglobulin. Talanta 2015, 131, 156–162. [Google Scholar]
- Amaya-González, S.; de-los-Santos-álvarez, N.; Miranda-Ordieres, A.J.; Lobo-Castañón, M.J. Sensitive gluten determination in gluten-free foods by an electrochemical aptamer-based assay. Anal. Bioanal. Chem. 2015, 407, 6021. [Google Scholar]
- Cheng, C.; Yu, X.; Geng, F.; Wang, L.; Yang, J.; Huang, F.; Deng, Q. Review on the Regulation of Plant Polyphenols on the Stability of Polyunsaturated-Fatty-Acid-Enriched Emulsions: Partitioning Kinetic and Interfacial Engineering. J. Agric. Food Chem. 2022, 70, 3569–3584. [Google Scholar]
- Li, C.; Liu, D.; Huang, M.; Huang, W.; Li, Y.; Feng, J. Interfacial engineering strategy to improve the stabilizing effect of curcumin-loaded nanostructured lipid carriers. Food Hydrocoll. 2022, 127, 107552. [Google Scholar]
- Jin, J.; Ma, H.; Wang, W.; Luo, M.; Wang, B.; Qu, W.; He, R.; Owusu, J.; Li, Y. Effects and mechanism of ultrasound pretreatment on rapeseed protein enzymolysis. J. Sci. Food Agric. 2016, 96, 1159–1166. [Google Scholar]
- Wang, Y.; Li, Z.; Barnych, B.; Huo, J.; Wan, D.; Vasylieva, N.; Xu, J.; Li, P.; Liu, B.; Zhang, C.; et al. Investigation of the Small Size of Nanobodies for a Sensitive Fluorescence Polarization Immunoassay for Small Molecules: 3-Phenoxybenzoic Acid, an Exposure Biomarker of Pyrethroid Insecticides as a Model. J. Agric. Food Chem. 2019, 67, 11536–11541. [Google Scholar] [CrossRef]
- Yang, W.; Jia, W.; Ou, M.; Zhong, W.; Jiang, L.; Wang, X. Effect of Physical Properties of an Emulsion Pesticide on the Atomisation Process and the Spatial Distribution of Droplet Size. Agriculture 2022, 12, 949. [Google Scholar] [CrossRef]
- Zou, Y.; Shi, Y.; Wang, T.; Ji, S.; Zhang, X.; Shen, T.; Huang, X.; Xiao, J.; Farag, M.A.; Shi, J.; et al. Quantum dots as advanced nanomaterials for food quality and safety applications: A comprehensive review and future perspectives. Compr. Rev. Food Sci. Food Saf. 2024, 23, e13339. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.-W.; Zou, X.-M.; Song, S.-H.; Chen, G.-H. Quantum Dots Applied to Methodology on Detection of Pesticide and Veterinary Drug Residues. J. Agric. Food Chem. 2018, 66, 1307–1319. [Google Scholar] [CrossRef] [PubMed]
- Okeke, E.S.; Ezeorba, T.P.C.; Okoye, C.O.; Chen, Y.; Mao, G.; Feng, W.; Wu, X. Analytical detection methods for azo dyes: A focus on comparative limitations and prospects of bio-sensing and electrochemical nano-detection. J. Food Compos. Anal. 2022, 114, 104778. [Google Scholar]
- Liu, G.; Liu, Z.; Sun, Y.; Sun, M.; Duan, J.; Tian, Y.; Du, D.; Li, M. Cascade Amplifying Electrochemical Bioanalysis for Zearalenone Detection in Agricultural Products: Utilizing a Glucose–Fenton–HQ System on Bimetallic–ZIF@CNP Nanocomposites. Foods 2024, 13, 3192. [Google Scholar] [CrossRef]
- Meng, S.; Liu, D.; Li, Y.; Dong, N.; Chen, T.; You, T. Engineering the Signal Transduction between CdTe and CdSe Quantum Dots for in Situ Ratiometric Photoelectrochemical Immunoassay of Cry1Ab Protein. J. Agric. Food Chem. 2022, 70, 13583–13591. [Google Scholar] [CrossRef]
- Li, W.; Shi, Y.; Zhang, X.; Hu, X.; Huang, X.; Liang, N.; Shen, T.; Zou, X.; Shi, J. A DNA tetrahedral scaffolds-based electrochemical biosensor for simultaneous detection of AFB1 and OTA. Food Chem. 2024, 442, 138312. [Google Scholar] [CrossRef]
- Dong, X.; Huang, A.; He, L.; Cai, C.; You, T. Recent advances in foodborne pathogen detection using photoelectrochemical biosensors: From photoactive material to sensing strategy. Front. Sustain. Food Syst. 2024, 8, 1432555. [Google Scholar] [CrossRef]
- Gao, L.; Li, Z.; Wei, X.; Hao, M.; Song, W.; Zou, X.; Huang, X. A cell-based electrochemical biosensor for the detection of capsaicin. J. Food Meas. Charact. 2024, 18, 9341–9352. [Google Scholar] [CrossRef]
- Luo, L.; Liu, X.; Ma, S.; Li, L.; You, T. Quantification of zearalenone in mildewing Cereal crops using an innovative photoelectrochemical aptamer sensing strategy based on ZnO-NGQDs composites. Food Chem. 2020, 322, 126778. [Google Scholar] [CrossRef]
- Rzayev, J. Molecular Bottlebrushes: New Opportunities in Nanomaterials Fabrication. ACS Macro Lett. 2017, 1, 1146–1149. [Google Scholar]
- Ouyang, Q.; Zhang, M.; Yang, Y.; Din, Z.; Chen, Q. Mesoporous silica-modified upconversion biosensor coupled with real-time ion release properties for ultrasensitive detection of Staphylococcus aureus in meat. Food Control 2022, 145, 109444. [Google Scholar]
- Teng, H.; Zheng, Y.; Cao, H.; Huang, Q.; Chen, L. Enhancement of bioavailability and bioactivity of diet-derived flavonoids by application of nanotechnology: A review. Crit. Rev. Food Sci. Nutr. 2021, 63, 378–393. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Sheng, W.; Chen, H.Q. Recent advances in rare earth ion-doped upconversion nanomaterials: From design to their applications in food safety analysis. Compr. Rev. Food Sci. Food Saf. 2023, 22, 3732–3764. [Google Scholar] [CrossRef]
- Patra, J.K.; Shin, H.-S.; Paramithiotis, S. Editorial: Application of Nanotechnology in Food Science and Food Microbiology. Front. Microbiol. 2018, 9, 714. [Google Scholar]
- Zeng, K.; Wei, W.; Jiang, L.; Zhu, F.; Du, D. Use of Carbon Nanotubes as a Solid Support To Establish Quantitative (Centrifugation) and Qualitative (Filtration) Immunoassays To Detect Gentamicin Contamination in Commercial Milk. J. Agric. Food Chem. 2016, 64, 7874–7881. [Google Scholar]
- Li, H.; Liang, J.; Kong, F.; Ren, M.; Mohammed, A.E.A.Y.; Zhou, C. Preparation of lignin-containing cellulose nanofibers from walnut shell using deep eutectic solvent for nanotube conductive film. Ind. Crops Prod. 2024, 207 Pt 1, 117737. [Google Scholar] [CrossRef]
- Sharma, A.S.; Ali, S.; Sabarinathan, D.; Murugavelu, M.; Li, H.; Chen, Q. Recent progress on graphene quantum dots-based fluorescence sensors for food safety and quality assessment applications. Compr. Rev. Food Sci. Food Saf. 2021, 20, 5765–5801. [Google Scholar]
- Zhao, Y.; Ma, Y.; Zhou, R.; He, Y.; Wu, Y.; Yi, Y.; Zhu, G. Highly sensitive electrochemical detection of paraoxon ethyl in water and fruit samples based on defect-engineered graphene nanoribbons modified electrode. J. Food Meas. Charact. 2022, 16, 2596–2603. [Google Scholar]
- Li, W.; Hu, X.; Li, Q.; Shi, Y.; Zhai, X.; Xu, Y.; Li, Z.; Huang, X.; Wang, X.; Shi, J.; et al. Copper nanoclusters @ nitrogen-doped carbon quantum dots-based ratiometric fluorescence probe for lead (II) ions detection in porphyra—ScienceDirect. Food Chem. 2020, 320, 126623. [Google Scholar]
- Zeng, K.; Chen, B.; Li, Y.; Meng, H.; Wu, Q.; Yang, J.; Liang, H. Gold nanoparticle-carbon nanotube nanohybrids with peroxidase-like activity for the highly-sensitive immunoassay of kanamycin in milk. Int. J. Food Sci. Technol. 2022, 57, 6028–6037. [Google Scholar] [CrossRef]
- Luo, R.; Feng, Z.; Shen, G.; Xiu, Y.; Zhou, Y.; Niu, X.; Wang, H. Acetyicholinesterase Biosensor Based On Mesoporous Hollow Carbon Spheres/Core-Shell Magnetic Nanoparticles-Modified Electrode for the Detection of Organophosphorus Pesticides. Sensors 2018, 18, 4429. [Google Scholar] [CrossRef] [PubMed]
- Stankovic, V.; Manojlovic, D.; Roglic, G.M.; Tolstoguzov, D.S.; Zherebtsov, D.A.; Uchaev, D.A.; Avdin, V.V.; Stankovic, D.M. Synthesis and Application of Domestic Glassy Carbon TiO2 Nanocomposite for Electrocatalytic Triclosan Detection. Catalysts 2022, 12, 1571. [Google Scholar] [CrossRef]
- Kang, Y.; Ren, X.; Li, Y.; Yu, Z. Ni-Coated Diamond-like Carbon-Modified TiO2 Nanotube Composite Electrode for Electrocatalytic Glucose Oxidation. Molecules 2022, 27, 5815. [Google Scholar] [CrossRef]
- Daneshvar, L.; Rounaghi, G.H.; Es’ haghi, Z.; Chamsaz, M.; Tarahomi, S. Fabrication a new modified electrochemical sensor based on Au-Pd bimetallic nanoparticle decorated graphene for citalopram determination. Mater. Sci. Eng. C Mater. Biogical Appl. 2016, 69, 653–660. [Google Scholar] [CrossRef]
- Kaur, N.; Thakur, H.; Kumar, R.; Prabhakar, N. An electrochemical sensor modified with poly(3,4-ethylenedioxythiophene)-wrapped multi-walled carbon nanotubes for enzyme inhibition-based determination of organophosphates. Mikrochim. Acta Int. J. Phys. Chem. Methods Anal. 2016, 183, 2307–2315. [Google Scholar] [CrossRef]
- Du, D.; Wang, M.; Cai, J.; Qin, Y.; Zhang, A. One-step synthesis of multiwalled carbon nanotubes-gold nanocomposites for fabricating amperometric acetylcholinesterase biosensor. Sens. Actuators B Chem. 2010, 143, 524–529. [Google Scholar] [CrossRef]
- Wu, Q.; Zhong, Y.; Chen, R.; Ling, G.; Wang, X.; Shen, Y.; Hao, C. Cu-Ag-C@Ni3S4 with core shell structure and rose derived carbon electrode materials: An environmentally friendly supercapacitor with high energy and power density. Ind. Crops Prod. 2024, 222, 119676. [Google Scholar] [CrossRef]
- Murali, M.; Gowtham, H.G.; Singh, S.B.; Shilpa, N.; Aiyaz, M.; Alomary, M.N.; Alshamrani, M.; Salawi, A.; Almoshari, Y.; Ansari, M.A. Fate, bioaccumulation and toxicity of engineered nanomaterials in plants: Current challenges and future prospects. Sci. Total Environ. 2022, 811, 152249. [Google Scholar]
- Fang, Y.; Fan, L.; Bai, H.; Li, B.; Zhang, H.; Xin, F.; Ma, J.; Jiang, M. Bio-based molecules for biosynthesis of nano-metallic materials. Sheng Wu Gong Cheng Xue Bao = Chin. J. Biotechnol. 2021, 37, 541–560. [Google Scholar]
- Wang, B.; Zhang, Y.; Venkitasamy, C.; Wu, B.; Pan, Z.; Ma, H. Effect of pulsed light on activity and structural changes of horseradish peroxidase. Food Chem. 2017, 234, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Zhao, Z.; Zhang, W. Determination of fat content in goat milk using annular photoelectric sensor system. Trans. Inst. Meas. Control 2018, 40, 102–110. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, B.; Tian, Y.; Guo, Q.; Yang, X.; Nie, G. An enhanced photoelectrochemical sensor for aflatoxin B1 detection based on organic-inorganic heterojunction nanomaterial: Poly(5-formylindole)/NiO. Mikrochim. Acta 2020, 187, 467. [Google Scholar] [PubMed]
- Chen, M.M.; Liu, Y.; Zhao, S.; Jiang, J.; Zhang, Q.; Li, P.; Tang, X. Carbon nanospheres bridging in perovskite quantum dots/BiOBr: An efficient heterojunction for high-performance photoelectrochemical sensing of deoxynivalenol. Carbon 2024, 221, 118919. [Google Scholar]
- Zhang, C.; Wang, Q.; Zhong, C. A simple photoelectrochemical aptasensor based on MoS2/rGO for aflatoxin B1 detection in grain crops. Anal. Methods 2024, 16, 1330–1340. [Google Scholar]
- Ding, S.; Lyu, Z.; Li, S.; Ruan, X.; Lin, Y. Molecularly imprinted polypyrrole nanotubes based electrochemical sensor for glyphosate detection. Biosens. Bioelectron. 2021, 191, 113434. [Google Scholar]
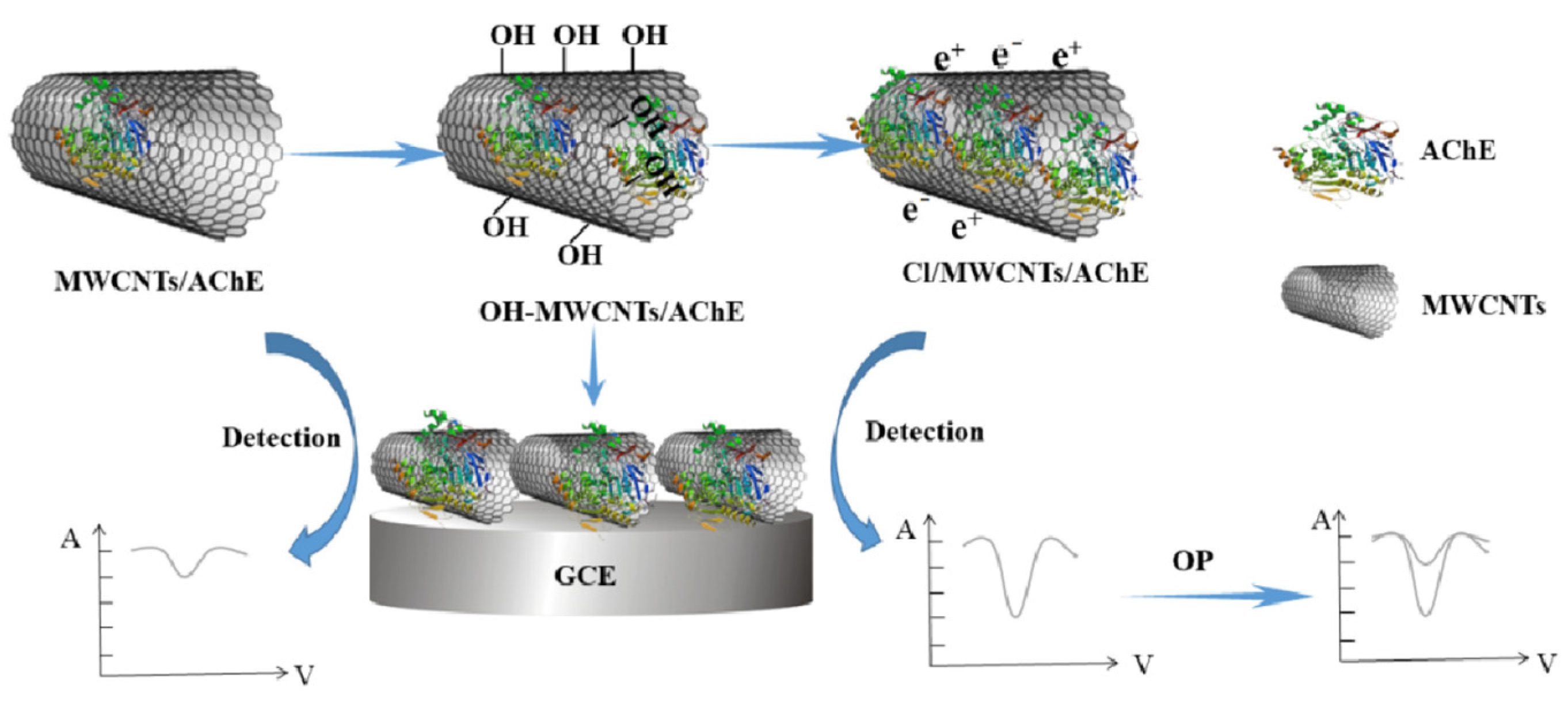
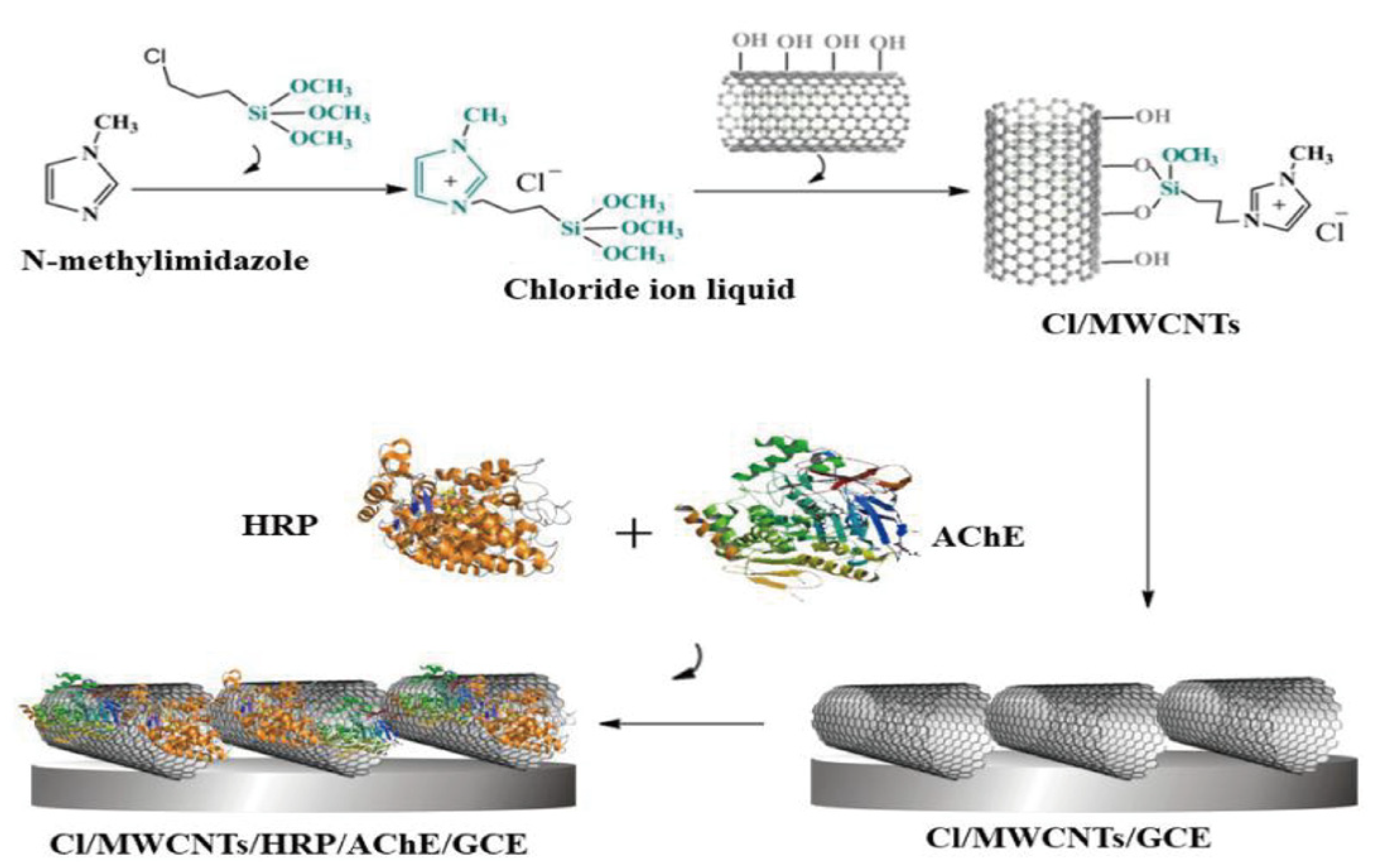
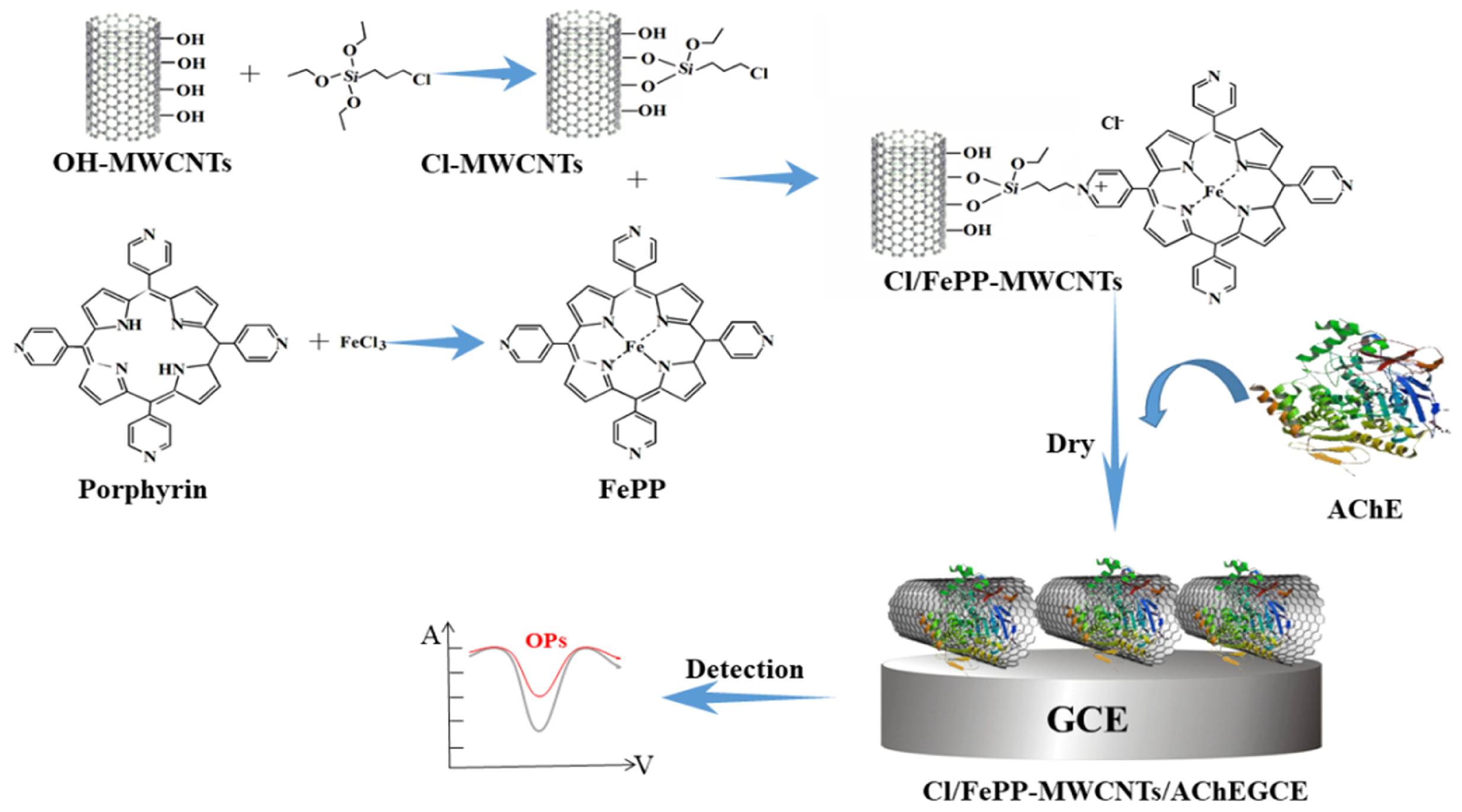
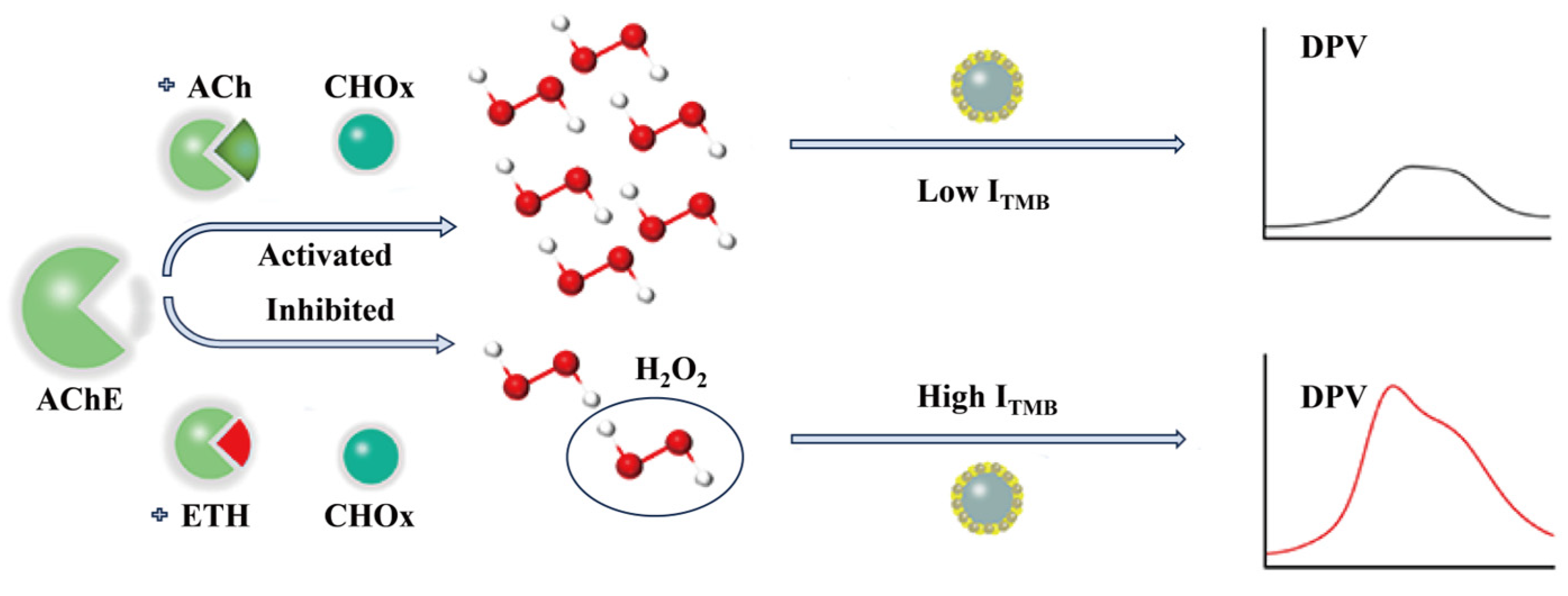
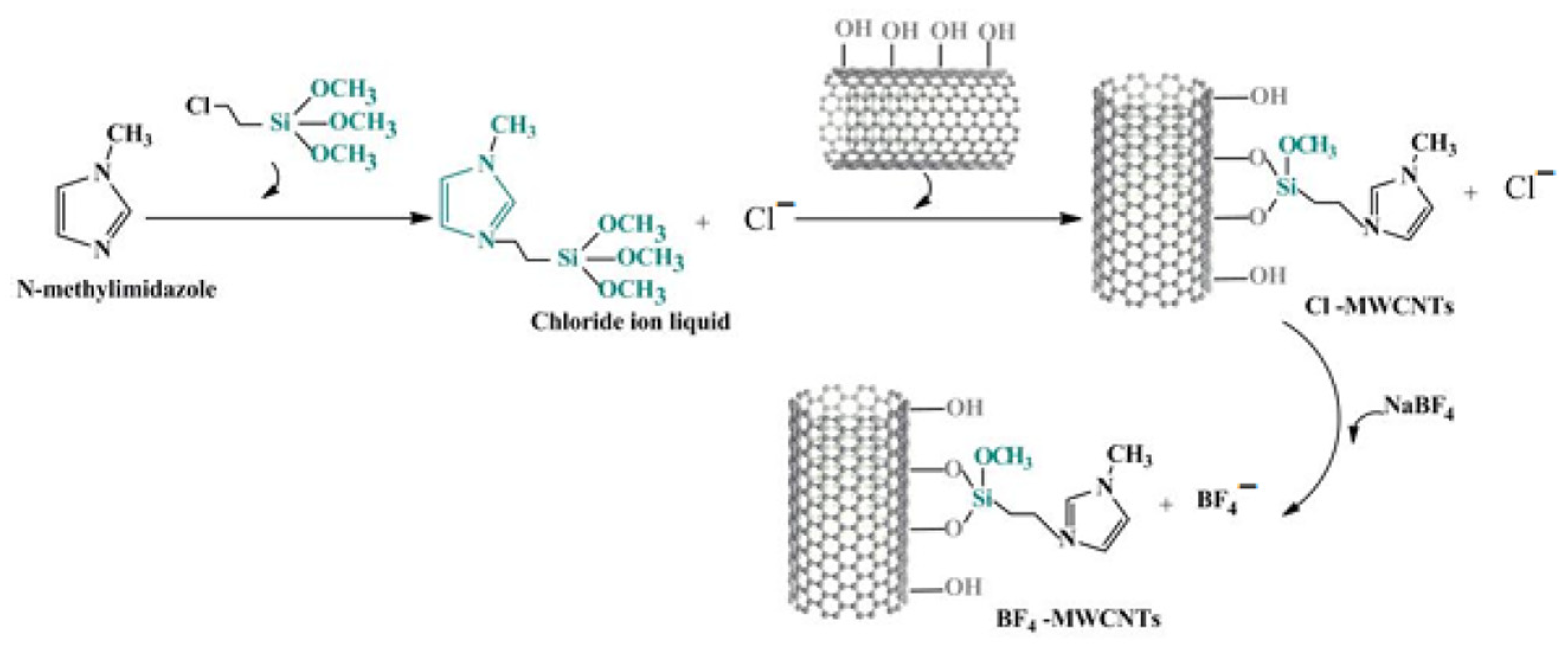
| Type | Detection Object | Enzyme Action Mechanism | Method | Performance Feature | Reference |
|---|---|---|---|---|---|
| Oxidoreductase electrodes | Intracellular purines | Catalyze the purine oxidation reaction | Electrochemical method | High sensitivity, high accuracy, wide application range, real-time monitoring | [22] |
| Hydrolase electrodes | Organophosphorus pesticide | Catalyze the hydrolysis of methyl parathion | Electrochemical method | High selectivity, limited sensitivity, limited application range | [24] |
| Isomerase electrodes | Serum glucose | Catalyze the isomerization of glucose | Electrochemical method | High selectivity, good biocompatibility, limited sensitivity, complex preparation | [25] |
| Transferase electrodes | Dopamine | Catalyze the isomerization of glucose | Electrochemical method | High selectivity, high accuracy, limited application range | [26] |
| Ligase electrodes | Ochratoxin A | Catalytic phosphorylation of OTA aptamer connections | Electrochemical method | High selectivity, high sensitivity, complex operation | [27] |
| Synthase electrodes | Non-esterified fatty acid | Catalyze acyl-coA synthesis reaction | Electrochemical method | High selectivity, high sensitivity, high preparation cost | [23] |
| Material Name | Structural Property | The Role of Sensors | Limit of Detection (LOD) | Reference |
|---|---|---|---|---|
| Carbon nanotubes (CNTs) | Single/multilayer tubular structure, high aspect ratio, excellent electrical conductivity | Enhanced electron transport efficiency, high specific surface area | 0.048 ng/mL | [107] |
| Multi-walled carbon nanotubes (MWCNTs) | Multilayer coaxial tubular structure with defect sites on the surface can be modified functionally | Improve the electrochemical activity of biomolecules, promote electron transfer in proteases, enhanced response to changes in surface conductivity | 11.2 pg/mL | [112] |
| Graphene | Single layer two-dimensional honeycomb structure, high conductivity | Enhance electron transport rate, adsorb target molecules | 4.3 nM | [110] |
| Mesoporous carbon spheres | Porous spherical structure, pore size 2~50 nm, high specific surface area | Provide a large number of enzyme fixation sites, promote molecular diffusion | 0.0182 ppb | [113] |
| Crystalline diamond | Three-dimensional cubic crystal structure, ultra-high hardness and chemical inertness | As electrode base material, provide stable interface | 0.07 μM | [114] |
| Diamond-like carbon | Amorphous carbon material with sp3 and sp2 hybrid bonds, high wear resistance, and chemical inertness | Electrode surface modification, reduce non-specific adsorption | 0.53 μM | [115] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, L.; Zhang, X.; Chu, Y.; Zhang, Y.; Lin, Z.; Kong, F.; Ni, X.; Zhao, Y.; Lu, Q.; Zou, B. Research Progress on Nanotechnology-Driven Enzyme Biosensors for Electrochemical Detection of Biological Pollution and Food Contaminants. Foods 2025, 14, 1254. https://doi.org/10.3390/foods14071254
Qu L, Zhang X, Chu Y, Zhang Y, Lin Z, Kong F, Ni X, Zhao Y, Lu Q, Zou B. Research Progress on Nanotechnology-Driven Enzyme Biosensors for Electrochemical Detection of Biological Pollution and Food Contaminants. Foods. 2025; 14(7):1254. https://doi.org/10.3390/foods14071254
Chicago/Turabian StyleQu, Liang, Xue Zhang, Yanhong Chu, Yuyang Zhang, Zhiyuan Lin, Fanzhuo Kong, Xing Ni, Yani Zhao, Qiongya Lu, and Bin Zou. 2025. "Research Progress on Nanotechnology-Driven Enzyme Biosensors for Electrochemical Detection of Biological Pollution and Food Contaminants" Foods 14, no. 7: 1254. https://doi.org/10.3390/foods14071254
APA StyleQu, L., Zhang, X., Chu, Y., Zhang, Y., Lin, Z., Kong, F., Ni, X., Zhao, Y., Lu, Q., & Zou, B. (2025). Research Progress on Nanotechnology-Driven Enzyme Biosensors for Electrochemical Detection of Biological Pollution and Food Contaminants. Foods, 14(7), 1254. https://doi.org/10.3390/foods14071254







