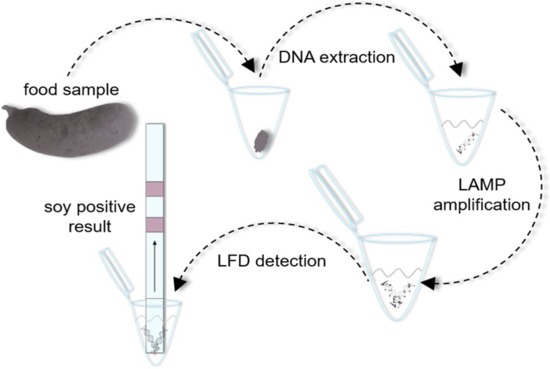
LAMP-LFD Based on Isothermal Amplification of Multicopy Gene ORF160b: Applicability for Highly Sensitive Low-Tech Screening of Allergenic Soybean (Glycine max) in Food
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials and Retail Foods
2.2. Soybean Incurred Food Matrices
2.2.1. Sausages
2.2.2. Chocolate
2.2.3. Instant Tomato Soup
2.3. Commercial Protein-Based LFD
2.4. DNA Extraction and Purification
2.5. Oligonucleotides
2.6. Eukaryotic qPCR
2.7. Generation of Plasmid Standards for Lectin qPCR
2.8. Lectin qPCR
2.9. ORF160b LAMP-LFD
2.10. Replicate Analysis of Food Samples
2.11. Statistical Analysis
3. Results
3.1. Specificity of the Methods
3.2. Detectability of Soy Products
3.3. Determination of Method Sensitvity in Soybean-Incurred Food Matrices
3.3.1. Verification of Various Levels of Incurred Soybean
3.3.2. Comparison of Sensitivity of ORF160b LAMP-LFD and Commercial Protein-Based LFDs
3.4. Analyses of Retail Food Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on the evaluation of allergenic foods and food ingredients for labelling purposes. EFSA J. 2014, 12, 3894. [Google Scholar]
- Burney, P.; Summers, C.; Chinn, S.; Hooper, R.; van Ree, R.; Lidholm, J. Prevalence and distribution of sensitization to foods in the European Community Respiratory Health Survey: A EuroPrevall analysis. Allergy 2010, 65, 1182–1188. [Google Scholar] [CrossRef]
- Worm, M.; Eckermann, O.; Dölle, S.; Aberer, W.; Beyer, K.; Hawranek, T.; Hompes, S.; Koehli, A.; Mahler, V.; Nemat, K.; et al. Triggers and Treatment of Anaphylaxis: An Analysis of 4000 Cases from Germany, Austria and Switzerland. Dtsch. Arztebl. Int. 2014, 111, 367–375. [Google Scholar] [PubMed] [Green Version]
- Ballmer-Weber, B.K.; Holzhauser, T.; Scibilia, J.; Mittag, D.; Zisa, G.; Ortolani, C.; Oesterballe, M.; Poulsen, L.K.; Vieths, S.; Bindslev-Jensen, C. Clinical characteristics of soybean allergy in Europe: A double-blind, placebo-controlled food challenge study. J. Allergy Clin. Immunol. 2007, 119, 1489–1496. [Google Scholar] [CrossRef] [PubMed]
- Gendel, S.M. Comparison of international food allergen labeling regulations. Regul. Toxicol. Pharmacol. 2012, 63, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Añíbarro, B.; Seoane, F.J.; Múgica, M.V. Involvement of hidden allergens in food allergic reactions. J. Investig. Allergol. Clin. Immunol. 2007, 17, 168–172. [Google Scholar] [PubMed]
- Remington, B.C.; Westerhout, J.; Meima, M.Y.; Blom, W.M.; Kruizinga, A.G.; Wheeler, M.W.; Taylor, S.L.; Houben, G.F.; Baumert, J.L. Updated population minimal eliciting dose distributions for use in risk assessment of 14 priority food allergens. Food Chem. Toxicol. 2020, 130, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Holzhauser, T.; Johnson, P.; Hindley, J.P.; O′Connor, G.; Chan, C.H.; Costa, J.; Fæste, C.K.; Hirst, B.J.; Lambertini, F.; Miani, M.; et al. Are current analytical methods suitable to verify VITAL® 2.0/3.0 allergen reference doses for EU allergens in foods? Food Chem. Toxicol. 2020, 145, 111709. [Google Scholar] [CrossRef] [PubMed]
- Scharf, A.; Kasel, U.; Wichmann, G.; Besler, M. Performance of ELISA and PCR Methods for the Determination of Allergens in Food: An Evaluation of Six Years of Proficiency Testing for Soy (Glycine max L.) and Wheat Gluten (Triticum aestivum L.). J. Agric. Food Chem. 2013, 61, 10261–10272. [Google Scholar] [CrossRef] [PubMed]
- Notomi, T.; Mori, Y.; Tomita, N.; Kanda, H. Loop-mediated isothermal amplification (LAMP): Principle, features, and future prospects. J. Microbiol. 2015, 53, 1–5. [Google Scholar] [CrossRef]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanebe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, e63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheu, S.C.; Tsou, P.C.; Lien, Y.Y.; Lee, M.S. Development of loop-mediated isothermal amplification (LAMP) assays for the rapid detection of allergic peanut in processed food. Food Chem. 2018, 257, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Zahradnik, C.; Martzy, R.; Mach, R.L.; Krska, R.; Farnleitner, A.H.; Brunner, K. Detection of the food allergen celery via loop-mediated isothermal amplification technique. Anal. Bioanal. Chem. 2014, 406, 6827–6833. [Google Scholar] [CrossRef]
- Focke, F.; Haase, I.; Fischer, M. Loop-mediated isothermal amplification (LAMP): Methods for plant species identification in food. J. Agric. Food Chem. 2013, 61, 2943–2949. [Google Scholar] [CrossRef] [PubMed]
- Yuan, D.; Kong, J.; Li, X.; Fang, X.; Chen, Q. Colorimetric LAMP microfluidic chip for detecting three allergens: Peanut, sesame and soybean. Sci. Rep. 2018, 8, 8682. [Google Scholar] [CrossRef]
- Di, H.; Shi, L.; Shen, H.; Yan, H.; Meng, H.; Li, L.; Alam, M.J.; Yamasaki, S.; Ye, L. Rapid Detection of Genetically Modified Ingredients in Soybean Products by Real-Time Loop-Mediated Isothermal Amplification. J. Food Nutr. Res. 2014, 2, 363–368. [Google Scholar] [CrossRef] [Green Version]
- Vaagt, F.; Haase, I.; Fischer, M. Loop-mediated isothermal amplification (LAMP)-based method for rapid mushroom species identification. J. Agric. Food Chem. 2013, 61, 1833–1840. [Google Scholar] [CrossRef]
- Allgöwer, S.M.; Hartmann, C.A.; Holzhauser, T. Sensitive detection of allergenic soybean (Glycine max) using multicopy gene loop-mediated isothermal amplification combined with lateral flow dipstick (LAMP-LFD). Foods 2020, 9, 423. [Google Scholar] [CrossRef] [Green Version]
- Röder, M.; Vieths, S.; Holzhauser, T. Commercial lateral flow devices for rapid detection of peanut (Arachis hypogaea) and hazelnut (Corylus avellana) cross-contamination in the industrial production of cookies. Anal. Bioanal. Chem. 2009, 395, 103–109. [Google Scholar] [CrossRef]
- Federal Office of Consumer Protection and Food Safety. BVL Official collection of Test methods. Detection and quantification of mustard (Sinapis alba) and soybean (Glycine max) in boiled sausages with real-time PCR. Ger. Food Feed Law Food Anal. 2012, 64, 1–15. [Google Scholar]
- Allmann, M.; Candrian, U.; Höfelein, C.; Lüthy, J. Polymerase chain reaction (PCR): A possible alternative to immunochemical methods assuring safety and quality of food, Detection of wheat contamination in non-wheat food products. Z Lebensm Unters Forsch 1993, 196, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Kalčáková, L.; Tremlová, B.; Pospiech, M.; Hostovský, M.; Dordević, D.; Javůrková, Z.; Bĕhalová, H.; Bartlová, M. Use of IHF-QD Microscopic Analysis for the Detection of Food Allergenic Components: Peanuts and Wheat Protein. Foods 2020, 9, 239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Golbitz, P. Traditional soyfoods: Processing and products. J. Nutr. 1995, 125 (Suppl. S3), 570–572. [Google Scholar]
- Lusas, E.W.; Riaz, M.N. Soy protein products: Processing and use. J. Nutr. 1995, 125 (Suppl. S3), 573–580. [Google Scholar]
- Belitz, H.; Grosch, W.; Schieberle, P. Lehrbuch der Lebensmittelchemie, 6th ed.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 769–794. [Google Scholar]
- Verhoeckx, K.C.M.; Vissers, Y.M.; Baumert, J.L.; Faludi, R.; Feys, M.; Flanagan, S.; Herouet-Guicheney, C.; Holzhauser, T.; Shimojo, R.; van der Bolt, N.; et al. Food processing and allergenicity. Food Chem. Toxicol. 2015, 80, 223–240. [Google Scholar] [CrossRef] [PubMed]
- Rossen, L.; Nørskov, P.; Holmstrøm, K.; Rasmussen, O.F. Inhibition of PCR by components of food samples, microbial diagnostic assays and DNA-extraction solutions. Int. J. Food Microbiol. 1992, 17, 37–45. [Google Scholar] [CrossRef]
- Young, C.C.; Burghoff, R.L.; Keim, L.G.; Minak-Bernero, V.; Lute, J.R.; Hinton, S.M. Polyvinylpyrrolidone-agarose gel electrophoresis purification of polymerase chain reaction-amplifiable DNA from soils. Appl. Environ. Microbiol. 1993, 59, 1972–1974. [Google Scholar] [CrossRef] [Green Version]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [Green Version]
- Palm, M.; Moneret-Vautrin, D.A.; Kanny, G.; Denery-Papini, S.; Frémont, S. Food allergy to egg and soy lecithins. Allergy 1999, 54, 1116–1117. [Google Scholar] [CrossRef] [Green Version]
- Holck, A.L.; Diaz-Amigo, C.; Kerbach, S.; Popping, B.; Mustorp, S.; Engdahl Axelsson, C. Detection of allergens in food. In Current Topics on Food Authentication; Oliveira, M.B.P.P., Mafra, I., Amaral, J.S., Eds.; Transworld Research Network: Kerala, India, 2011; pp. 173–210. [Google Scholar]
- Taylor, S.L.; Baumert, J.L.; Kruizinga, A.G.; Remington, B.C.; Crevel, R.W.R.; Brooke-Taylor, S.; Allen, K.J.; The Allergen Bureau of Australia & New Zealand; Houben, G. Establishment of Reference Doses for residues of allergenic foods: Report of the VITAL Expert Panel. Food Chem. Toxicol. 2014, 63, 9–17. [Google Scholar] [CrossRef]



| Investigation | ORF160b LAMP-LFD | Lectin qPCR | Commercial LFD 1 |
|---|---|---|---|
| Specificity | Allgöwer et al. 2020 [18] | n.d. 2 | 1 rxn 3, 1:10 dilution (1 extract × 1 rxn) |
| Processed soy products | 4 rxns, 1:104 dilution (2 extracts × 2 rxns) | 4 rxns, 1:104 dilution (2 extracts × 2 rxns) | 1 rxn, 1:104 dilution (1 extract × 1 rxn) |
| Sensitivity in incurred food matrices | 8 rxns, undiluted (2 extracts × 4 rxns) | 8 rxns, undiluted (2 extracts × 4 rxns) | 1 rxn, undiluted (1 extract × 1 rxn) |
| Retail foods | 4 rxns, undiluted (2 extracts × 2 rxns) | 4 rxns, undiluted (2 extracts × 2 rxns) | 1 rxn, undiluted (1 extract × 1 rxn) |
| Sample No. | Soy Product | Protein Content (%) | ORF160b LAMP-LFD | lectin qPCR | LFD1 | LFD2 |
|---|---|---|---|---|---|---|
| P-1 | TVP 1 | 56.7 | + | + | + | − |
| P-2 | TVP 1 | 67.4 | (+) | (+) | + | − |
| P-3 | soy flakes | ~50 2 | + | + | + | − |
| P-4 | soy flakes | ~50 2 | + | + | + | + |
| P-5 | soy protein concentrate | 71.8 | + | + | + | + |
| P-6 | defatted soy flour | ~50 2 | + | + | + | + |
| P-7 | soy semolina | ~50 2 | + | + | + | + |
| P-8 | tofu | 13.5 | + | (+) | + | − |
| 18 | Yellow soybeans Schoenenberger® Hensel® | 38 | + | + | + | + |
| Method | Food Matrix | Level of Incurred Soybean per Matrix (mg/kg) | ||||||
|---|---|---|---|---|---|---|---|---|
| 105 | 104 | 103 | 102 | 101 | 100 | 0 1 | ||
| lectin qPCR | sausage | + | + | + | + | + | (+) | − |
| chocolate | + | + | + | + | + | + | − | |
| tomato soup | + | + | + | + | + | (+) | − | |
| ORF160b LAMP-LFD | sausage | + | + | + | + | + | (+) | − |
| chocolate | + | + | + | + | + | + | − | |
| tomato soup | + | + | + | + | + | (+) | − | |
| LFD1 | sausage | n.d. 2 | n.d. | + | + | (+) | − | − |
| chocolate | n.d. | n.d. | + | + | + | (+) | − | |
| tomato soup | n.d. | n.d. | + | + | + | (+) | (+) | |
| LFD2 | sausage | n.d. | n.d. | + | + | + | − | − |
| chocolate | n.d. | n.d. | + | + | − | − | − | |
| tomato soup | n.d. | n.d. | + | + | + | − | − | |
| Sample No. | Product Description | Soy: Ingredient Labeling (IL)/Precautionary Allergen Labeling (PAL) | Interpretation of IL/PAL | Lectin qPCR | ORF160b LAMP-LFD | LFD1 | LFD2 |
|---|---|---|---|---|---|---|---|
| R-1 | fine biscuit assortment | IL: soy flour, emulsifier: soy lecithin/PAL: none | contains soy | + | + | + | + |
| R-2 | Asia style rice crackers | IL: soy sauce (soybeans, …)/PAL: contains soy | contains soy | − | − | − | − |
| R-3 | sponge cake vanilla and raspberry taste with cocoa compound coating | IL: soybean, soy flour, emulsifier: soy lecithin/PAL: contains soy | contains soy | + | + | + | + |
| R-4 | vegetable balls | IL: 10% plant protein (soy protein, wheat protein), hydrolyzed soy protein/PAL: none | contains soy | + | + | + | + |
| R-5 | meat free, lactose free sausage-type bar for barbecue | IL: 12% soy protein/PAL: none | contains soy | + | + | ± 1 | + |
| R-6 | cereal bar with chocolate | IL: none/PAL: none | no indication of soy presence | + | + | (+) | − |
| R-7 | milk chocolate with puffed rice | IL: emulsifier: soy lecithin/PAL: none | contains soy | (+) | + | (+) | − |
| R-8 | shortbread with milk chocolate and chopped almonds | IL: soy flour, emulsifier: soy lecithin/PAL: none | contains soy | + | + | + | + |
| R-9 | hazelnut cookies | IL: none/PAL: produced in a facility, where soy is also processed | may contain soy | (+) | (+) | − | − |
| R-10 | oat porridge | IL: none/PAL: may contain traces of soy | may contain soy | − | − | − | − |
| R-11 | cashews roasted and salted | IL: none/PAL: none | no indication of soy presence | − | − | − | − |
| R-12 | Cereal-potato-snack Western style flavor | IL: none/PAL: none | no indication of soy presence | (+) | − | (+) | − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allgöwer, S.M.; Hartmann, C.A.; Lipinski, C.; Mahler, V.; Randow, S.; Völker, E.; Holzhauser, T. LAMP-LFD Based on Isothermal Amplification of Multicopy Gene ORF160b: Applicability for Highly Sensitive Low-Tech Screening of Allergenic Soybean (Glycine max) in Food. Foods 2020, 9, 1741. https://doi.org/10.3390/foods9121741
Allgöwer SM, Hartmann CA, Lipinski C, Mahler V, Randow S, Völker E, Holzhauser T. LAMP-LFD Based on Isothermal Amplification of Multicopy Gene ORF160b: Applicability for Highly Sensitive Low-Tech Screening of Allergenic Soybean (Glycine max) in Food. Foods. 2020; 9(12):1741. https://doi.org/10.3390/foods9121741
Chicago/Turabian StyleAllgöwer, Stefanie M., Chris A. Hartmann, Clarissa Lipinski, Vera Mahler, Stefanie Randow, Elke Völker, and Thomas Holzhauser. 2020. "LAMP-LFD Based on Isothermal Amplification of Multicopy Gene ORF160b: Applicability for Highly Sensitive Low-Tech Screening of Allergenic Soybean (Glycine max) in Food" Foods 9, no. 12: 1741. https://doi.org/10.3390/foods9121741