Stability of the Meat Protein Type I Collagen: Influence of pH, Ionic Strength, and Phenolic Antioxidant
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study of Collagen Stability as Function of pH and Ionic Strength
2.2. Catechin-Treated Collagen
2.3. 1H NMR Time Domain (TD)
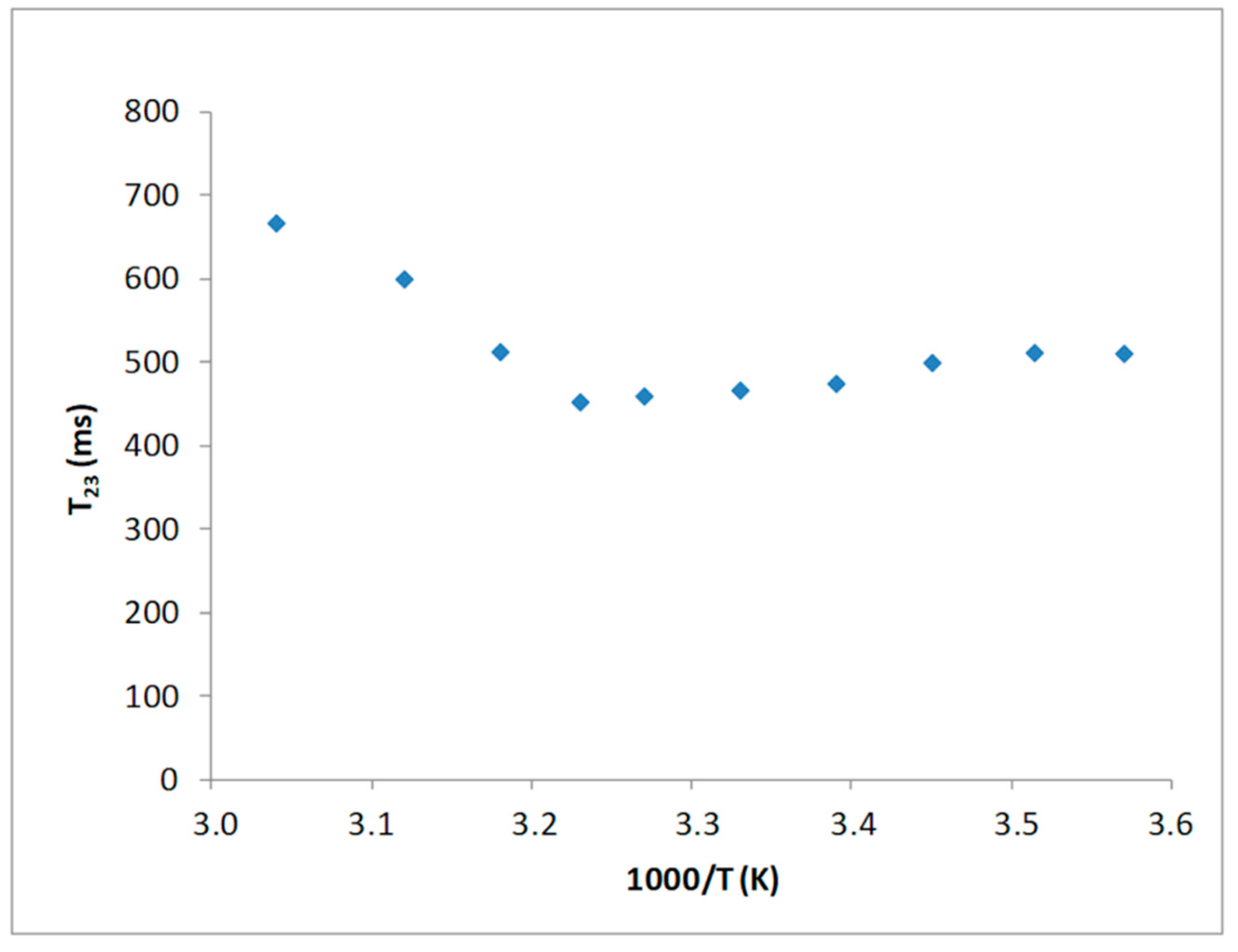
3. Results
3.1. 1H NMR Characterization of Type I Collagen
3.1.1. Time Domain Measurements
3.1.2. Collagen Fibers as Affected by pH and Ionic Strength
3.2. Characterization of Type I Collagen Modified with Catechin.
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cheng, Q.; Sun, D.-W. Factors Affecting the Water Holding Capacity of Red Meat Products: A Review of Recent Research Advances. Crit. Rev. Food Sci. Nutr. 2008, 48, 137–159. [Google Scholar] [CrossRef] [PubMed]
- Warner, R.D. The Eating Quality of Meat—IV Water-Holding Capacity and Juiciness. Chapter 14. In Lawrie’s Meat Science, 8th ed.; Toldrà, F., Ed.; Woodhead Publishing: London, UK, 2017; pp. 419–459. [Google Scholar] [CrossRef]
- Acton, J.C.; Ziegler, G.R.; Burge, D.L.; Froning, G.W. Functionality of muscle constituents in the processing of comminuted meat products. C R C Crit. Rev. Food Sci. Nutr. 1983, 18, 99–121. [Google Scholar] [CrossRef] [PubMed]
- Privalov, P.L. Stability of proteins: Proteins which do not present a single cooperative system. Adv. Protein Chem. 1982, 35, 1–104. [Google Scholar] [PubMed]
- Davies, M.J. Protein oxidation and peroxidation. Biochem. J. 2016, 473, 805–825. [Google Scholar] [CrossRef] [Green Version]
- Gebicki, J.M. Oxidative stress, free radicals and protein peroxides. Arch. Biochem. Biophys. 2016, 595, 33–39. [Google Scholar] [CrossRef]
- Lucarini, M.; Sciubba, F.; Capitani, D.; Di Cocco, M.E.; D’Evoli, L.; Durazzo, A.; Delfini, M.; Ginevra, L.-B. Role of catechin on collagen type I stability upon oxidation: A NMR approach. Nat. Prod. Res. 2019, 34, 53–62. [Google Scholar] [CrossRef]
- Madhan, B.; Subramanian, V.; Rao, J.R.; Nair, B.U.; Ramasami, T. Stabilization of collagen using plant polyphenol: Role of catechin. Int. J. Boil. Macromol. 2005, 37, 47–53. [Google Scholar] [CrossRef]
- Brinckmann, J. Collagens at a Glance. In Collagen. Topics in Current Chemistry; Brinckmann, J., Notbohm, H., Müller, P.K., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; Volume 247. [Google Scholar]
- Di Lullo, G.A.; Sweeney, S.M.; Korkko, J.; Ala-Kokko, L.; San Antonio, J.D. Mapping the ligand-binding sites and disease-associated mutations on the most abundant protein in the human, type I collagen. J. Biol. Chem. 2002, 277, 4223–4231. [Google Scholar] [CrossRef] [Green Version]
- Kadeřávek, P.; Bolik-Coulon, N.; Cousin, S.F.; Marquardsen, T.; Tyburn, J.M.; Dumez, J.N.; Ferrage, F. Protein Dynamics from Accurate Low-Field Site-Specific Longitudinal and Transverse Nuclear Spin Relaxation. J. Phys. Chem. Lett. 2019, 10, 5917–5922. [Google Scholar] [CrossRef]
- Bertram, H.C.; Karlsson, A.H.; Rasmussen, M.; Pedersen, O.D.; Dønstrup, S.; Andersen, H.J. Origin of Multiexponential T2 Relaxation in Muscle Myowater. J. Agric. Food Chem. 2001, 49, 3092–3100. [Google Scholar] [CrossRef]
- Gianferri, R.; Maioli, M.; Delfini, M.; Brosio, E. A low-resolution and high resolution nuclear magnetic resonance integrated approach to investigate the physical structure and metabolic profile of Mozzarella di Bufala Campana cheese. Int. Dairy J. 2007, 17, 167–176. [Google Scholar] [CrossRef]
- Bertram, H.C.; Kristensen, M.; Andersen, H.J. Functionality of myofibrillar proteins as affected by pH, ionic strength and heat-treatments. A low-field NMR study. Meat Sci. 2004, 68, 249–256. [Google Scholar] [CrossRef]
- Meiboom, S.; Gill, D. Modified Spin-Echo Method for Measuring Nuclear Relaxation Times. Rev. Sci. Instrum. 1958, 29, 688–691. [Google Scholar] [CrossRef] [Green Version]
- Marquardt, D.W. An algorithm for least-squares estimation of nonlinear parameters. J. Soc. Ind. Appl. Math. 1963, 11, 431–441. [Google Scholar] [CrossRef]
- Stejskal, E.O.; Tanner, J.E. Spin diffusion measurements: Spin echoes in the presence of a time dependent field gradient. J. Chem. Phys. 1965, 42, 288–292. [Google Scholar] [CrossRef] [Green Version]
- Bjarnason, T.A.; Vavasour, I.M.; Chia, C.L.L.; MacKay, A.L. Characterization of the NMR behavior of white matter in bovine brain. Magn. Reson. Med. 2005, 54, 1072–1081. [Google Scholar] [CrossRef] [PubMed]
- Reiter, D.A.; Lin, P.-C.; Fishbein, K.; Spencer, R.G.S. Multicomponent T2 relaxation analysis in cartilage. Magn. Reson. Med. 2009, 61, 803–809. [Google Scholar] [CrossRef] [Green Version]
- Martini, S.; Bonechi, C.; Foletti, A.; Rossi, C. Water-Protein Interactions: The Secret of Protein Dynamics. Sci. World J. 2013, 2013, 138916. [Google Scholar] [CrossRef] [Green Version]
- Kopp, J.; Bonnet, M.; Renou, J.P. Effect of collagen crosslinking on collagen-water interactions (a DSC investigation). Matrix 1989, 9, 443–450. [Google Scholar] [CrossRef]
- Pineri, M.H.; Escoubes, M.; Roche, G. Water-collagen interactions: Calorimetric and mechanical experiments. Biopolymers 1978, 17, 2799–2815. [Google Scholar] [CrossRef]
- Renou, J.P.; Bonnet, M.; Bielicki, G.; Rochdi, A.; Gatellier, P. NMR study of collagen-water interactions. Biopolymers 1994, 34, 1615–1626. [Google Scholar] [CrossRef] [PubMed]
- Migchelsen, C.; Berendsen, H.J.C. Proton exchange and molecular orientation of water in hydrated collagen fibers. An NMR study of H2O and D2O. J. Chem. Phys. 1973, 59, 296–305. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.H. Theory of the Self-diffusion of Water in Protein Solutions. A New Method for Studying the Hydration and Shape of Protein Molecules. J. Am. Chem. Soc. 1954, 76, 4755–4763. [Google Scholar] [CrossRef]
- Lechert, H.T. Water binding on starch: NMR studies on native and gelatinized starch. In Water Activity: Influences on Food Quality; Rockland, L.B., Stewart, G.F., Eds.; Academic Press: New York, NY, USA, 1981; pp. 223–245. [Google Scholar]
- Goddard, Y.A.; Korb, J.-P.; Bryant, R.G. Water molecule contributions to proton spin–lattice relaxation in rotationally immobilized proteins. J. Magn. Reson. 2009, 199, 68–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diakova, G.; Goddard, Y.; Korb, J.-P.; Bryan, R.G. Water-Proton-Spin-Lattice-Relaxation Dispersion of Paramagnetic Protein Solutions. J. Magn. Reson. 2011, 208, 195–203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bendall, J.R.; Swatland, H.J. A review of the relationships of pH with physical aspects of pork quality. Meat Sci. 1988, 24, 85–126. [Google Scholar] [CrossRef]
- Offer, G.; Trinick, J. On the mechanism of water holding in meat: The swelling and shrinking of myofibrils. Meat Sci. 1993, 8, 245–281. [Google Scholar] [CrossRef]
- Chen, J.Y.; Piva, M.; Labuza, T.P. Evaluation of Water Binding Capacity (WBC) of Food Fiber Sources. J. Food Sci. 1984, 49, 59–63. [Google Scholar] [CrossRef]
- Hamm, R. Functional properties of the myofibrillar system and their measurements. In Muscle as Food; Bechtel, P.J., Ed.; Academic Press: Orlando, FL, USA; Elsevier: Amsterdam, The Netherlands, 1986; pp. 135–199. [Google Scholar]
- Bertram, H.C.; Whittaker, A.K.; Andersen, H.J.; Karlsson, A.H. pH Dependence of the Progression in NMR T2 Relaxation Times in Post-mortem Muscle. J. Agric. Food Chem. 2003, 51, 4072–4078. [Google Scholar] [CrossRef]
- Tornberg, E.; Nerbrink, O. Swelling of whole meat and myofibrils—as measured by pulse-NMR. In Proceedings of the 30th European Meeting of meat Research Workers, Bristol, UK, 9–14 September 1984; pp. 112–113. [Google Scholar]
- Brownstein, K.R.; Tarr, C.E. Importance of classical diffusion in NMR studies of water in biological cells. Physol. Rev. A 1979, 19, 2446–2453. [Google Scholar] [CrossRef]
- Brøndum, J.; Munck, L.; Henckel, P.; Karlsson, A.; Tornberg, E.; Engelsen, S.B. Prediction of waterholding capacity and composition of porcine meat by comparative spectroscopy. Meat Sci. 2000, 55, 177–185. [Google Scholar] [CrossRef]
- Jepsen, S.M.; Pedersen, H.T.; Engelsen, S.B. Application of chemometrics to low-field 1H NMR relaxation data of intact fish flesh. J. Sci. Food Agric. 1999, 79, 1793–1802. [Google Scholar] [CrossRef]
- Bertram, H.C. 1H NMR Relaxometry in Meat Science. In Modern Magnetic Resonance, 2nd ed.; Webb, G., Ed.; Springer: Berlin, Germany, 2016; pp. 1–14. [Google Scholar] [CrossRef]




| Fraction i | |||
|---|---|---|---|
| 1 | 2 | 3 | |
| T2i (ms) | 6 ± 1 | 41 ± 7 | 447 ± 15 |
| fi (%) | 56 ± 5 | 36 ± 4 | 8 ± 2 |
| Di (10−5cm2s−1) | 0.71 | 1.22 | 2.31 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lucarini, M.; Durazzo, A.; Sciubba, F.; Di Cocco, M.E.; Gianferri, R.; Alise, M.; Santini, A.; Delfini, M.; Lombardi-Boccia, G. Stability of the Meat Protein Type I Collagen: Influence of pH, Ionic Strength, and Phenolic Antioxidant. Foods 2020, 9, 480. https://doi.org/10.3390/foods9040480
Lucarini M, Durazzo A, Sciubba F, Di Cocco ME, Gianferri R, Alise M, Santini A, Delfini M, Lombardi-Boccia G. Stability of the Meat Protein Type I Collagen: Influence of pH, Ionic Strength, and Phenolic Antioxidant. Foods. 2020; 9(4):480. https://doi.org/10.3390/foods9040480
Chicago/Turabian StyleLucarini, Massimo, Alessandra Durazzo, Fabio Sciubba, Maria Enrica Di Cocco, Raffaella Gianferri, Mosè Alise, Antonello Santini, Maurizio Delfini, and Ginevra Lombardi-Boccia. 2020. "Stability of the Meat Protein Type I Collagen: Influence of pH, Ionic Strength, and Phenolic Antioxidant" Foods 9, no. 4: 480. https://doi.org/10.3390/foods9040480








