Emulsifying and Anti-Oxidative Properties of Proteins Extracted from Industrially Cold-Pressed Rapeseed Press-Cake
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sources of Materials and Chemicals
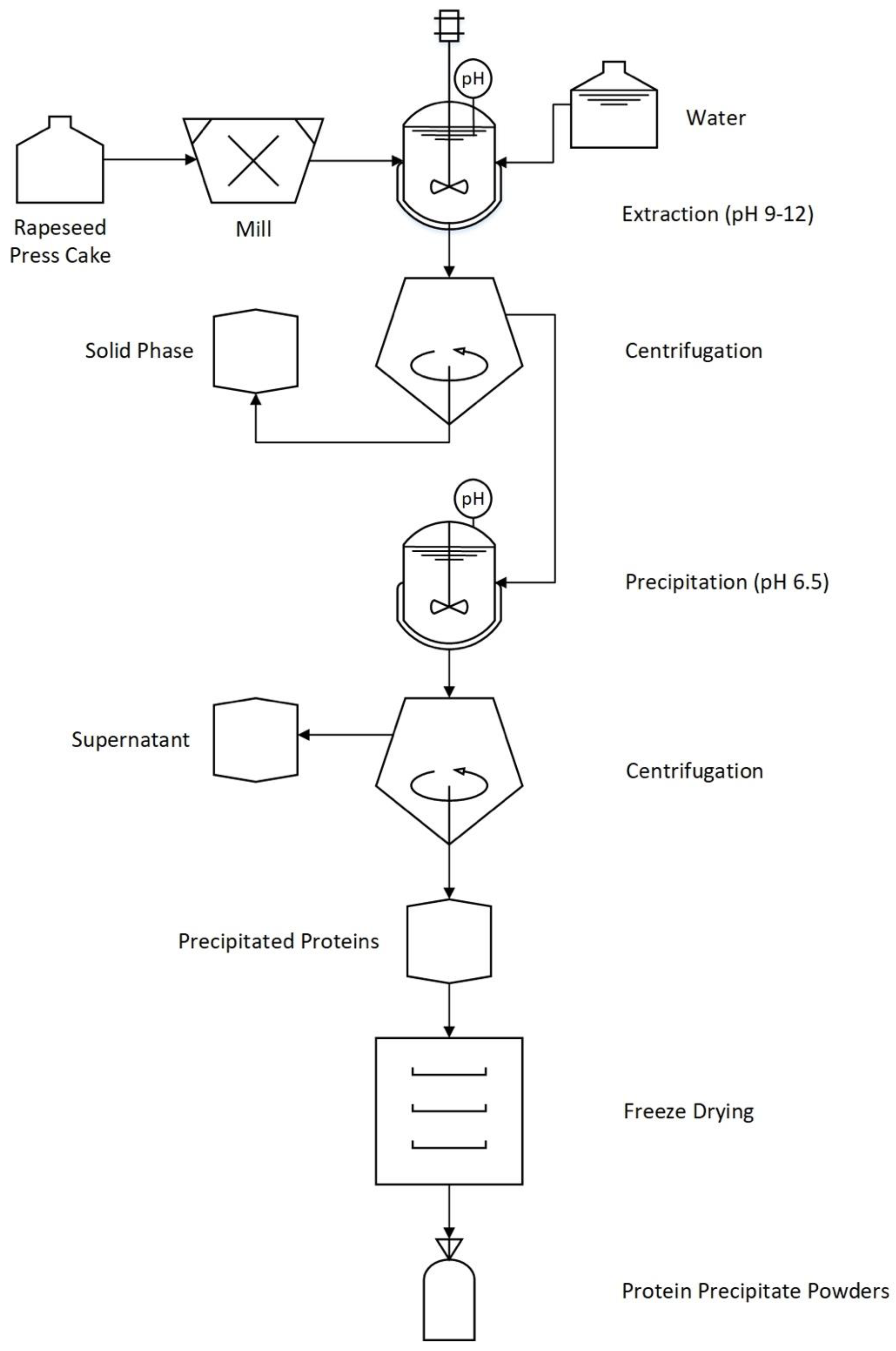
2.2. Protein Extraction and Recovery from Cold-Pressed Rapeseed Press-Cake
2.3. Dry Matter Analysis and Protein Content
2.4. Emulsifying Properties of Emulsions Stabilized by Rapeseed Proteins
2.4.1. Preparation of Emulsions
2.4.2. Particle Size Analysis of Emulsions
2.4.3. Emulsion Stability during Storage
2.5. Oxidative Stability of Emulsions
2.6. Statistical Analysis
3. Results and Discussion
3.1. Effect of Alkali pH on Protein Yield
3.2. Emulsifying Properties
3.2.1. Function of Protein Concentration and Extraction pH
3.2.2. Comparison with Other Emulsifiers during Storage
3.3. Oxidative Stability
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wahlgren, M.; Bergenståhl, B.; Nilsson, L.; Rayner, M. Formulation of emulsions. In Engineering Aspects of Food Emulsification and Homogenization; Rayner, M., Dejmek, P., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 51–124. [Google Scholar]
- Dickinson, E. Stabilising emulsion-based colloidal structures with mixed food ingredients. J. Sci. Food Agric. 2013, 93, 710–721. [Google Scholar] [CrossRef]
- Horne, D.S. Protein-stabilized emulsions. Cur. Opin. Colloid Interface Sci. 1996, 1, 752–758. [Google Scholar]
- Chang, C.; Tu, S.; Ghosh, S.; Nickerson, M.T. Effect of pH on the inter-relationships between the physicochemical, interfacial and emulsifying properties for pea, soy, lentil and canola protein isolates. Food Res. Int. 2015, 77, 360–367. [Google Scholar] [CrossRef]
- De Folter, J.W.J.; Van Ruijven, M.W.M.; Velikov, K.P. Oil-in-water Pickering emulsions stabilized by colloidal particles from the water-insoluble protein zein. Soft Matter 2012, 8, 2807–2815. [Google Scholar] [CrossRef] [Green Version]
- Karaca, A.C.; Low, N.; Nickerson, M. Emulsifying properties of chickpea, faba bean, lentil and pea proteins produced by isoelectric precipitation and salt extraction. Food Res. Int. 2011, 44, 2742–2750. [Google Scholar] [CrossRef]
- Wijesundera, C.; Boiteau, T.; Xu, X.; Shen, Z.; Watkins, P.; Logan, A. Stabilization of fish oil-in-water emulsions with oleosin extracted from canola meal. J. Food Sci. 2013, 78, C1340–C1347. [Google Scholar] [CrossRef] [PubMed]
- Roberts, N.J.; Scott, R.W.; Tzen, J.T.C. Recent biotechnological applications using oleosins. Open Biotechnol. J. 2008, 2, 13–21. [Google Scholar] [CrossRef]
- Maurer, S.; Waschatko, G.; Schach, D.; Zielbauer, B.; Dahl, J.; Weidner, T.; Bonn, M.; Vilgis, T.A. The role of intact oleosin for stabilization and function of oleosomes. J. Phys. Chem. B. 2013, 117, 13872–13883. [Google Scholar] [CrossRef]
- Chen, B.; McClements, D.J.; Gray, D.A.; Decker, E.A. Physical and oxidative stability of pre-emulsified oil bodies extracted from soybeans. Food Chem. 2012, 132, 1514–1520. [Google Scholar] [CrossRef]
- Wanasundara, J.P.D.; Tan, S.H.; Alashi, A.M.; Pudel, F.; Blanchard, C. Proteins from canola/rapeseed: Current status. In Sustainable Protein Sources, 1 ed.; Nadathur, S., Wanasundara, J.P.D., Scanlin, L., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 285–304. [Google Scholar]
- FAOSTAT. Available online: http://www.fao.org/faostat/en/?#data/QC (accessed on 12 February 2020).
- Carré, P.; Pouzet, A. Rapeseed market, worldwide and in Europe. OCL 2014, 21, D102. [Google Scholar] [CrossRef]
- CCC. Canola Council of Canada. Available online: www.canoalacouncil.org (accessed on 12 February 2020).
- Maison, T.; Liu, Y.; Stein, H.H. Digestibility of energy and detergent fiber and digestible and metabolizable energy values in canola meal, 00-rapeseed meal, and 00-rapeseed expellers fed to growing pigs1. J. Anim. Sci. 2015, 93, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Campbell, L.; Rempel, B.C.; Wanasundara, P.J. Canola/Rapeseed protein: Future opportunities and directions—Workshop Proceedings of IRC 2015. Plants 2016, 5, 17. [Google Scholar] [CrossRef]
- Akbari, A.; Wu, J. An integrated method of isolating napin and cruciferin from defatted canola meal. LWT-Food Sci. Technol. 2015, 64, 308–315. [Google Scholar] [CrossRef]
- Wu, J.; Muir, A. Comparative structural, emulsifying, and biological properties of 2 major canola proteins, cruciferin and napin. J. Food Sci 2008, 73, C210–C216. [Google Scholar] [CrossRef] [PubMed]
- Wijesundera, C.; Shen, Z. Mimicking natural oil bodies for stabilising oil-in-water food emulsions. Lipid Technol. 2014, 26, 151–153. [Google Scholar] [CrossRef]
- Nikiforidis, C.V.; Ampatzidis, C.; Lalou, S.; Scholten, E.; Karapantsios, T.D.; Kiosseoglou, V. Purified oleosins at air-water interfaces. Soft Matter 2013, 9, 1354–1363. [Google Scholar] [CrossRef]
- Rayner, M. Current status on novel ways for stabilizing food dispersions by oleosins, particles and microgels. Curr. Opin. Food Sci. 2015, 3, 94–109. [Google Scholar] [CrossRef]
- Mccarthy, J. Recombinant oleosins from cacao and their use as flavoring or emulsifying agents. U.S. Patent 7,126,042, 24 October 2006. [Google Scholar]
- Nikiforidis, C.V.; Biliaderis, C.G.; Kiosseoglou, V. Rheological characteristics and physicochemical stability of dressing-type emulsions made of oil bodies-egg yolk blends. Food Chem. 2012, 134, 64–73. [Google Scholar] [CrossRef]
- Cumby, N.; Ying, Z.; Naczk, M.; Shahidi, F. Antioxidant activity and water-holding capacity of canola protein hydrolysates. Food Chem. 2008, 109, 144–148. [Google Scholar] [CrossRef]
- Deleu, M.; Vaca-Medina, G.; Fabre, J.F.; Roïz, J.; Valentin, R.; Mouloungui, Z. Interfacial properties of oleosins and phospholipids from rapeseed for the stability of oil bodies in aqueous medium. Colloids Surf. B. 2010, 80, 125–132. [Google Scholar] [CrossRef]
- Von Der Haar, D.; Müller, K.; Bader-Mittermaier, S.; Eisner, P. Rapeseed proteins—Production methods and possible application ranges. OCL 2014, 21, D104. [Google Scholar] [CrossRef] [Green Version]
- Tan, S.H.; Mailer, R.J.; Blanchard, C.L.; Agboola, S.O. Emulsifying properties of proteins extracted from Australian canola meal. LWT-Food Sci. Technol. 2014, 57, 376–382. [Google Scholar] [CrossRef]
- Berton-Carabin, C.C.; Ropers, M.H.; Genot, C. Lipid oxidation in oil-in-water emulsions: Involvement of the interfacial layer. Compr. Rev. Food Sci. Food Saf. 2014, 13, 945–977. [Google Scholar] [CrossRef]
- Tzen, J.T.C.; Cao, Y.; Laurent, P.; Ratnayake, C.; Huang, A.H.C. Lipids, proteins, and structure of seed oil bodies from diverse species. Plant. Physiol. 1993, 101, 267–276. [Google Scholar] [CrossRef] [Green Version]
- FDA. Code of Federal Regulations. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=184.1733 (accessed on 15 May 2020).
- Papastergiadis, A.; Mubiru, E.; Van Langenhove, H.; De Meulenaer, B. Malondialdehyde measurement in oxidized foods: Evaluation of the spectrophotometric thiobarbituric acid reactive substances (TBARS) test in various foods. J. Agric. Food Chem. 2012, 60, 9589–9594. [Google Scholar] [CrossRef]
- Aider, M.; Barbana, C. Canola proteins: Composition, extraction, functional properties, bioactivity, applications as a food ingredient and allergenicity-A practical and critical review. Trends Food Sci. Technol. 2011, 22, 21–39. [Google Scholar] [CrossRef]
- Rodrigues, I.M.; Coelho, J.F.J.; Carvalho, M.S. Isolation and valorisation of vegetable proteins from oilseed plants: Methods, limitations and potential. J. Food Eng. 2012, 109, 337–346. [Google Scholar] [CrossRef]
- Fetzer, A.; Herfellner, T.; Stäbler, A.; Menner, M.; Eisner, P. Influence of process conditions during aqueous protein extraction upon yield from pre-pressed and cold-pressed press cake. Ind. Crop. Prod. 2018, 112, 236–246. [Google Scholar] [CrossRef]
- Ghodsvali, A.; Haddad Khodaparast, M.H.; Vosoughi, M.; Diosady, L.L. Preparation of canola protein materials using membrane technology and evaluation of meals functional properties. Food Res. Int. 2005, 38, 223–231. [Google Scholar] [CrossRef]
- Östbring, K.; Malmqvist, E.; Nilsson, K.; Rosenlind, I.; Rayner, M. The effects of oil extraction methods on recovery yield and emulsifying properties of proteins from rapeseed meal and press cake. Foods 2020, 9, 19. [Google Scholar] [CrossRef] [Green Version]
- Jiang, J.; Chen, J.; Xiong, Y. Structural and emulsifying properties of soy protein isolate subjected to acid and alkaline pH-shifting processes. J. Agric. Food Chem. 2009, 57, 7576–7583. [Google Scholar] [CrossRef] [PubMed]
- Gray, D.A.; Payne, G.; McClements, D.J.; Decker, E.A.; Lad, M. Oxidative stability of Echium plantagineum seed oil bodies. Eur. J. Lipid Sci. Technol. 2010, 112, 741–749. [Google Scholar] [CrossRef]

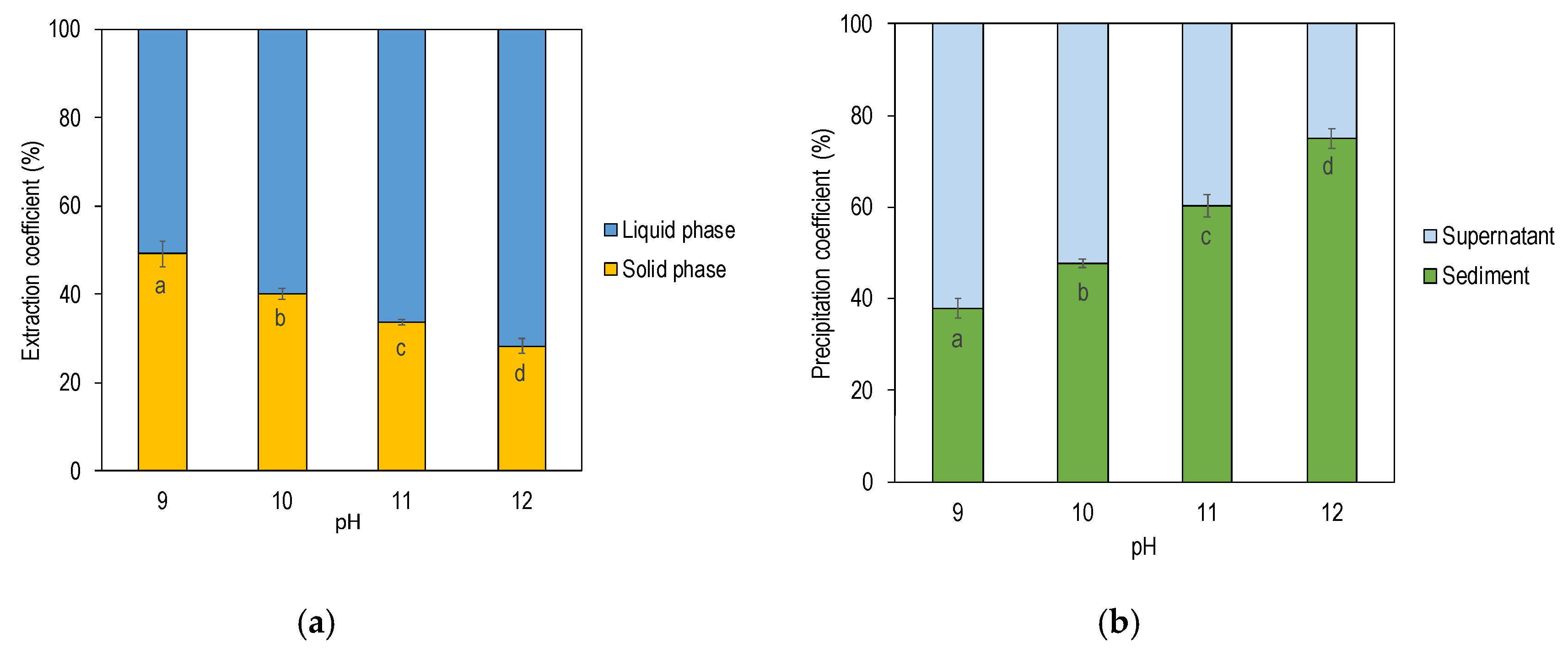



| pH 9 | pH 10 | pH 11 | pH 12 | ||
|---|---|---|---|---|---|
| Centrifugation After Meal Extraction | Liquid phase | ||||
| Mass (g) | 360 ± 1 a | 370 ± 4 b | 370 ± 1 b | 370 ± 1 b | |
| Dry Matter (g) | 18 ± 0 a | 20 ± 1 b | 21 ± 0 b | 22 ± 1 c | |
| Protein (g) | 7.2 ± 0.3 a | 8.5 ± 0.2 b | 9.5 ± 0.1 c | 10 ± 0.2 d | |
| Protein Yield (% of Starting Material) | 51 ± 0 a | 60 ± 0 b | 66 ± 0 c | 72 ± 0 d | |
| Solid-Phase | |||||
| Mass (g) | 134 ± 1 a | 126 ± 2 b | 129 ± 1 b | 129 ± 1 b | |
| Dry Matter (g) | 27 ± 0 a | 25 ± 1 b | 25 ± 0 b | 23 ± 1 c | |
| Protein (g) | 7.0 ± 0.3 a | 5.7 ± 0.2 b | 4.8 ± 0.1 c | 4.0 ± 0.2 d | |
| Protein Yield (% of Starting Material) | 49 ± 0 a | 40 ± 0 b | 34 ± 0 c | 28 ± 0 d | |
| Protein Precipitation After pH Adjustment | Supernatant | ||||
| Mass (g) | 330 ± 1 a | 330 ± 4 a | 320 ± 1 b | 300 ± 2 c | |
| Dry Matter (g) | 12 ± 0 a | 12 ± 1 a | 9.5 ± 0.4 b | 8.9 ± 0.8 b | |
| Protein (g) | 4.5 ± 0.3 a | 4.5 ± 0.1 ab | 3.8 ± 0.2 b | 2.6 ± 0.2 c | |
| Protein Yield (% of Starting Material) | 32 ± 0 a | 31 ± 0 ab | 26 ± 0 b | 18 ± 0 c | |
| Precipitate | |||||
| Mass (g) | 29 ± 1 a | 38 ± 1 b | 52 ± 2 c | 74 ± 3 d | |
| Dry Matter (g) | 6.1 ± 0 a | 8.4 ± 0.1 b | 11 ± 0 c | 13 ± 0 d | |
| Protein (g) | 2.7 ± 0.0 a | 4.1 ± 0.1 b | 5.7 ± 0.2 c | 7.7 ± 0.3 d | |
| Protein Yield (% of Starting Material) | 19 ± 0 a | 29 ± 0 b | 40 ± 0 c | 54 ± 0 d |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Östbring, K.; Nilsson, K.; Ahlström, C.; Fridolfsson, A.; Rayner, M. Emulsifying and Anti-Oxidative Properties of Proteins Extracted from Industrially Cold-Pressed Rapeseed Press-Cake. Foods 2020, 9, 678. https://doi.org/10.3390/foods9050678
Östbring K, Nilsson K, Ahlström C, Fridolfsson A, Rayner M. Emulsifying and Anti-Oxidative Properties of Proteins Extracted from Industrially Cold-Pressed Rapeseed Press-Cake. Foods. 2020; 9(5):678. https://doi.org/10.3390/foods9050678
Chicago/Turabian StyleÖstbring, Karolina, Kajsa Nilsson, Cecilia Ahlström, Anna Fridolfsson, and Marilyn Rayner. 2020. "Emulsifying and Anti-Oxidative Properties of Proteins Extracted from Industrially Cold-Pressed Rapeseed Press-Cake" Foods 9, no. 5: 678. https://doi.org/10.3390/foods9050678






