Spectroscopic Techniques for Monitoring Thermal Treatments in Fish and Other Seafood: A Review of Recent Developments and Applications
Abstract
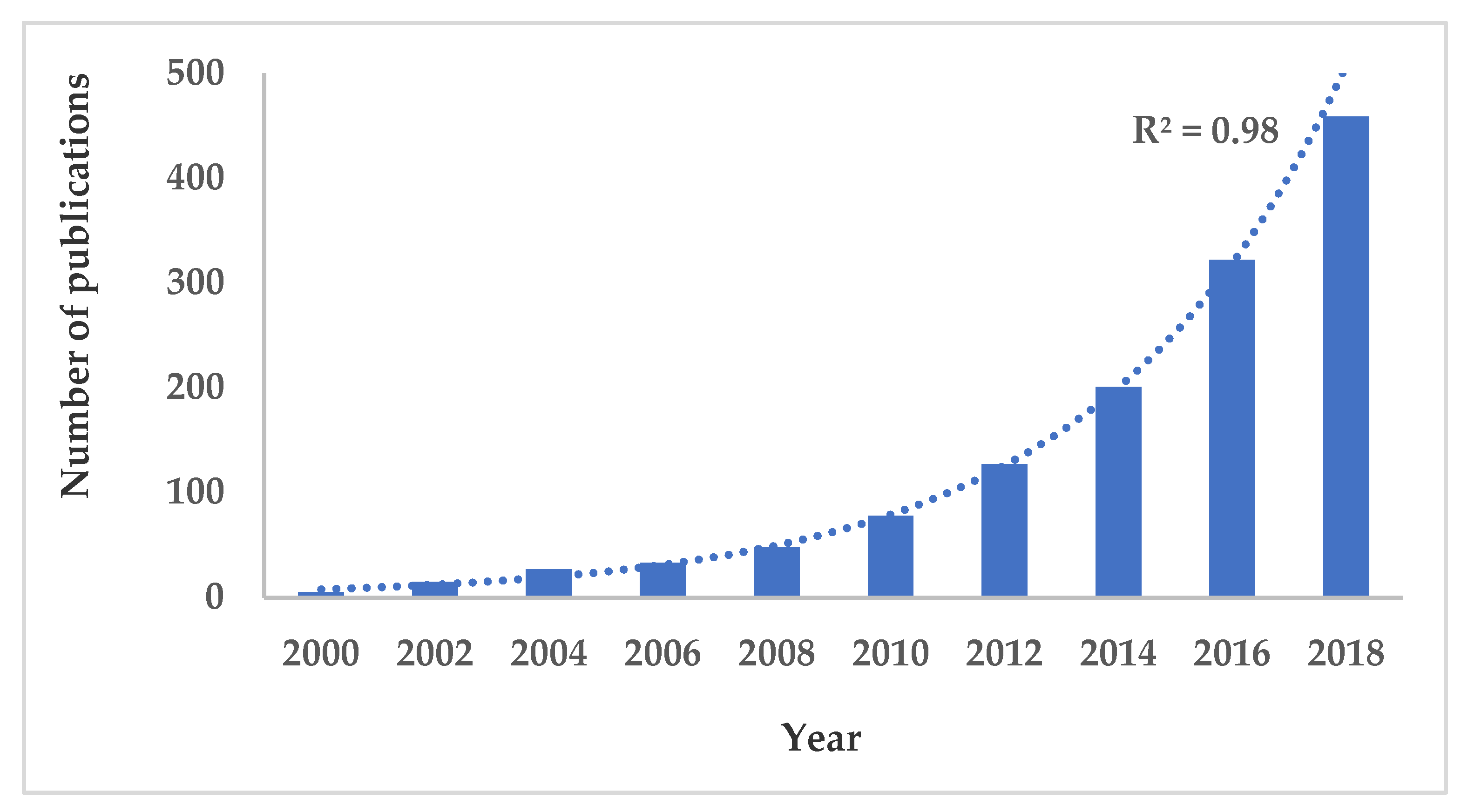
:1. Introduction
- (i)
- To briefly present traditional cooking methods and recent advances in this context.
- (ii)
- To discuss the impact of thermal treatments on different quality parameters of seafood.
- (iii)
- To demonstrate the potential of spectroscopic techniques for monitoring thermal treatments rapidly and non-destructively in comparison with traditional methods.
2. Cooking Methods Used in Households and Seafood Industry
3. Monitoring Thermal Treatments
3.1. Traditional Methods Used to Assess Changes in Quality Induced by Thermal Treatments
3.2. Monitoring Thermal Treatments by Spectroscopic Techniques
3.2.1. Visible and Infrared Spectroscopy
3.2.2. Raman Spectroscopy
3.2.3. NMR Spectroscopy
3.2.4. Fluorescence Spectroscopy
4. Limitations and Future Trends
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sampels, S. The effects of processing technologies and preparation on the final quality of fish products. Trends Food Sci. Technol. 2015, 44, 131–146. [Google Scholar] [CrossRef]
- Liu, Y.; Pu, H.; Sun, D.-W. Hyperspectral imaging technique for evaluating food quality and safety during various processes: A review of recent applications. Trends Food Sci. Technol. 2017, 69, 25–35. [Google Scholar] [CrossRef]
- Zhao, Y.-M.; De Alba, M.; Sun, D.-W.; Tiwari, B. Principles and recent applications of novel non-thermal processing technologies for the fish industry—A review. Crit. Rev. Food Sci. Nutr. 2018, 59, 728–742. [Google Scholar] [CrossRef] [PubMed]
- Elmasry, G.; Nakauchi, S. Noninvasive sensing of thermal treatments of Japanese seafood products using imaging spectroscopy. Int. J. Food Sci. Technol. 2015, 50, 1960–1971. [Google Scholar] [CrossRef]
- Johnsen, S.; Jørgensen, K.; Birkeland, S.; Skipnes, D.; Skåra, T. Effects of Phosphates and Salt in Ground Raw and Cooked Farmed Cod (Gadus morhua) Muscle Studied by the Water Holding Capacity (WHC), and Supported by31P-NMR Measurements. J. Food Sci. 2009, 74, C211–C220. [Google Scholar] [CrossRef]
- Ovissipour, M.; Rasco, B.; Tang, J.; Sablani, S.S. Kinetics of Protein Degradation and Physical Changes in Thermally Processed Atlantic Salmon (Salmo salar). Food Bioprocess Technol. 2017, 10, 1865–1882. [Google Scholar] [CrossRef]
- Wold, J.P. On-line and non-destructive measurement of core temperature in heat treated fish cakes by NIR hyperspectral imaging. Innov. Food Sci. Emerg. Technol. 2016, 33, 431–437. [Google Scholar] [CrossRef] [Green Version]
- Skipnes, D. Heat Processing of Fish. In Seafood Processing: Technology, Quality and Safety; Wiley: Hoboken, NJ, USA, 2014; pp. 61–81. [Google Scholar]
- Skipnes, D.; Johnsen, S.O.; Skåra, T.; Sivertsvik, M.; Lekang, O. Optimization of Heat Processing of Farmed Atlantic Cod (Gadus morhua) Muscle with Respect to Cook Loss, Water Holding Capacity, Color, and Texture. J. Aquat. Food Prod. Technol. 2011, 20, 331–340. [Google Scholar] [CrossRef]
- Tavares, W.P.S.; Dong, S.; Yang, Y.; Zeng, M.; Zhao, Y. Influence of cooking methods on protein modification and in vitro digestibility of hairtail (Thichiurus lepturus) fillets. LWT 2018, 96, 476–481. [Google Scholar] [CrossRef]
- Hu, L.; Ren, S.; Shen, Q.; Ye, X.; Chen, J.; Ling, J. Protein oxidation and proteolysis during roasting and in vitro digestion of fish (Acipenser gueldenstaedtii). J. Sci. Food Agric. 2018, 98, 5344–5351. [Google Scholar] [CrossRef]
- Pasha, I.; Saeed, F.; Sultan, M.T.; Khan, M.R.; Rohi, M. Recent Developments in Minimal Processing: A Tool to Retain Nutritional Quality of Food. Crit. Rev. Food Sci. Nutr. 2013, 54, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Rosnes, J.T.; Skipnes, D. Minimal heat processing applied in fish processing. In Trends in Fish Processing Technologies; Informa UK Limited: London, UK, 2017; pp. 27–70. [Google Scholar]
- Cortés, V.; Blasco, J.; Aleixos, N.; Cubero, S.; Talens, P. Monitoring strategies for quality control of agricultural products using visible and near-infrared spectroscopy: A review. Trends Food Sci. Technol. 2019, 85, 138–148. [Google Scholar] [CrossRef]
- Hassoun, A.; Sahar, A.; Lakhal, L.; Aït-Kaddour, A. Fluorescence spectroscopy as a rapid and non-destructive method for monitoring quality and authenticity of fish and meat products: Impact of different preservation conditions. LWT 2019, 103, 279–292. [Google Scholar] [CrossRef]
- McGrath, T.F.; Haughey, S.A.; Patterson, J.; Fauhl-Hassek, C.; Donarski, J.; Alewijn, M.; Ruthd, S.; Elliott, C.T. What are the scientific challenges in moving from targeted to non-targeted methods for food fraud testing and how can they be addressed?–Spectroscopy case study. Trends Food Sci. Technol. 2018, 76, 38–55. [Google Scholar] [CrossRef]
- Cropotova, J.; Mozuraityte, R.; Standal, I.B.; Rustad, T. A non-invasive approach to assess texture changes in sous-vide cooked Atlantic mackerel during chilled storage by fluorescence imaging. Food Control 2018, 92, 216–224. [Google Scholar] [CrossRef]
- Skåra, T.; Stormo, S.K.; Skipnes, D.; Kondjoyan, A.; Sivertsen, A.; Gins, G.; Van Derlinden, E.; Valdramidis, V.; Van Impe, J.F. Estimation of surface temperature and thermal load in short-time heat treatment of surimi through reflectance spectroscopy and heat transfer modeling. J. Food Eng. 2014, 120, 75–80. [Google Scholar] [CrossRef]
- Stormo, S.K.; Sivertsen, A.H.; Heia, K.; Skipnes, D. Endpoint temperature of heat-treated surimi can be measured by visible spectroscopy. Food Control 2012, 26, 92–97. [Google Scholar] [CrossRef]
- Cropotova, J.; Mozuraityte, R.; Standal, I.B.; Rustad, T. Assessment of lipid oxidation in Atlantic mackerel (Scomber scombrus) subjected to different antioxidant and sous-vide cooking treatments by conventional and fluorescence microscopy methods. Food Control 2019, 104, 1–8. [Google Scholar] [CrossRef]
- Barham, P.; Skibsted, L.H.; Bredie, W.L.; Frøst, M.; Møller, P.; Risbo, J.; Snitkjaer, P.; Mortensen, L.M. Molecular Gastronomy: A New Emerging Scientific Discipline. Chem. Rev. 2010, 110, 2313–2365. [Google Scholar] [CrossRef]
- Sobral, M.M.; Cunha, S.C.; Faria, M.; Ferreira, I. Domestic Cooking of Muscle Foods: Impact on Composition of Nutrients and Contaminants. Compr. Rev. Food Sci. Food Saf. 2018, 17, 309–333. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.; Liu, Y.; Lai, S.; Cao, H.; Guan, Y.; Cheang, W.S.; Liu, B.; Zhao, K.; Miao, S.; Riviere, C.; et al. Effects of domestic cooking process on the chemical and biological properties of dietary phytochemicals. Trends Food Sci. Technol. 2019, 85, 55–66. [Google Scholar] [CrossRef] [Green Version]
- Rosnes, J.T.; Skåra, T.; Skipnes, D. Recent Advances in Minimal Heat Processing of Fish: Effects on Microbiological Activity and Safety. Food Bioprocess Technol. 2011, 4, 833–848. [Google Scholar] [CrossRef]
- Stormo, S.K.; Skåra, T.; Skipnes, D.; Sone, I.; Carlehög, M.; Heia, K.; Skjelvareid, M.H. In-Pack Surface Pasteurization of Capture-Based, Pre-Rigor Filleted Atlantic Cod (Gadus morhua). J. Aquat. Food Prod. Technol. 2018, 7, 1–12. [Google Scholar] [CrossRef]
- Erdogdu, F.; Karatas, O.; Sarghini, F. A short update on heat transfer modelling for computational food processing in conventional and innovative processing. Curr. Opin. Food Sci. 2018, 23, 113–119. [Google Scholar] [CrossRef]
- Aguilera, J.M. Relating Food Engineering to Cooking and Gastronomy. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1021–1039. [Google Scholar] [CrossRef] [Green Version]
- Lerfall, J.; Jakobsen, A.N.; Skipnes, D.; Waldenstrøm, L.; Hoel, S.; Rotabakk, B.T. Comparative Evaluation on the Quality and Shelf life of Atlantic Salmon (Salmo salar L.) Filets Using Microwave and Conventional Pasteurization in Combination with Novel Packaging Methods. J. Food Sci. 2018, 83, 3099–3109. [Google Scholar] [CrossRef]
- Liu, Y.; Sun, D.-W.; Cheng, J.-H.; Han, Z. Hyperspectral Imaging Sensing of Changes in Moisture Content and Color of Beef during Microwave Heating Process. Food Anal. Methods 2018, 11, 2472–2484. [Google Scholar] [CrossRef]
- Cao, H.; Fan, D.; Jiao, X.; Huang, J.; Zhao, J.; Yan, B.; Zhou, W.; Zhang, W.; Ye, W.; Zhang, H. Importance of thickness in electromagnetic properties and gel characteristics of surimi during microwave heating. J. Food Eng. 2019, 248, 80–88. [Google Scholar] [CrossRef]
- Guo, C.; Mujumdar, A.S.; Zhang, M. New Development in Radio Frequency Heating for Fresh Food Processing: A Review. Food Eng. Rev. 2019, 11, 29–43. [Google Scholar] [CrossRef]
- Jiang, H.; Liu, Z.; Wang, S. Microwave processing: Effects and impacts on food components. Crit. Rev. Food Sci. Nutr. 2017, 58, 2476–2489. [Google Scholar] [CrossRef]
- Ekezie, F.-G.C.; Sun, D.-W.; Han, Z.; Cheng, J.-H. Microwave-assisted food processing technologies for enhancing product quality and process efficiency: A review of recent developments. Trends Food Sci. Technol. 2017, 67, 58–69. [Google Scholar] [CrossRef]
- Guo, Q.; Sun, D.-W.; Cheng, J.-H.; Han, Z. Microwave processing techniques and their recent applications in the food industry. Trends Food Sci. Technol. 2017, 67, 236–247. [Google Scholar] [CrossRef]
- Jaeger, H.; Roth, A.; Toepfl, S.; Holzhauser, T.; Engel, K.H.; Knorr, D.; Vogel, R.F.; Bandick, N.; Kulling, S.; Steinberg, P.; et al. Opinion on the use of ohmic heating for the treatment of foods. Trends Food Sci. Technol. 2016, 55, 84–97. [Google Scholar] [CrossRef]
- Tadpitchayangkoon, P.; Park, J.W.; Yongsawatdigul, J. Gelation characteristics of tropical surimi under water bath and ohmic heating. LWT 2012, 46, 97–103. [Google Scholar] [CrossRef] [Green Version]
- Stormo, S.K.; Skipnes, D.; Sone, I.; Skuland, A.; Heia, K.; Skåra, T. Modeling-assisted minimal heat processing of Atlantic cod (Gadus morhua). J. Food Process. Eng. 2017, 40, e12555. [Google Scholar] [CrossRef]
- Dong, X.-P.; Fan, X.; Wang, Y.; Qi, L.; Liang, S.; Qin, L.; Yu, C.; Zhu, B. The effect of different pretreatments on the quality of ready-to-eat jellyfish Rhopilema esculentum Kishinouye products. Fish. Sci. 2018, 84, 413–422. [Google Scholar] [CrossRef]
- Cao, H.; Fan, D.; Jiao, X.; Huang, J.; Zhao, J.; Yan, B.; Zhou, W.; Zhang, W.; Zhang, H. Heating surimi products using microwave combined with steam methods: Study on energy saving and quality. Innov. Food Sci. Emerg. Technol. 2018, 47, 231–240. [Google Scholar] [CrossRef]
- Lin, X.; Yang, W.; Xu, D.; Wang, L. Effect of electron irradiation and heat on the structure of hairtail surimi. Radiat. Phys. Chem. 2015, 114, 50–54. [Google Scholar] [CrossRef]
- Cao, H.; Fan, D.; Jiao, X.; Huang, J.; Zhao, J.; Yan, B.; Zhou, W.; Zhang, W.; Zhang, H. Effects of microwave combined with conduction heating on surimi quality and morphology. J. Food Eng. 2018, 228, 1–11. [Google Scholar] [CrossRef]
- Kong, F.; Tang, J.; Rasco, B.; Crapo, C. Kinetics of salmon quality changes during thermal processing. J. Food Eng. 2007, 83, 510–520. [Google Scholar] [CrossRef]
- Ovissipour, M.; Rasco, B.; Tang, J.; Sablani, S.S. Kinetics of quality changes in whole blue mussel (Mytilus edulis) during pasteurization. Food Res. Int. 2013, 53, 141–148. [Google Scholar] [CrossRef]
- Hu, L.; Ren, S.; Shen, Q.; Chen, J.; Ye, X.; Ling, J. Proteomic study of the effect of different cooking methods on protein oxidation in fish fillets. RSC Adv. 2017, 7, 27496–27505. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Tang, J.; Rasco, B.; Sablani, S.S.; Ovissipour, M.; Qu, Z. Kinetics of Quality Changes of Shrimp (Litopenaeus setiferus) During Pasteurization. Food Bioprocess Technol. 2018, 11, 1027–1038. [Google Scholar] [CrossRef]
- Cai, L.; Feng, J.; Cao, A.; Zhang, Y.; Lv, Y.; Li, J. Denaturation Kinetics and Aggregation Mechanism of the Sarcoplasmic and Myofibril Proteins from Grass Carp During Microwave Processing. Food Bioprocess Technol. 2017, 11, 417–426. [Google Scholar] [CrossRef]
- Abel, N.; Rotabakk, B.T.; Lerfall, J. Effect of heat treatment and packaging technology on the microbial load of lightly processed seafood. LWT 2019, 101, 123–129. [Google Scholar] [CrossRef] [Green Version]
- Blikra, M.J.; Skipnes, D.; Feyissa, A.H. Model for heat and mass transport during cooking of cod loin in a convection oven. Food Control 2019, 102, 29–37. [Google Scholar] [CrossRef]
- He, S.; Sun, X.; Du, M.; Chen, H.; Tan, M.; Sun, H.; Zhu, B. Effects of muscle protein denaturation and water distribution on the quality of false abalone (Volutharpa ampullacea perryi) during wet heating. J. Food Process. Eng. 2018, 42, e12932. [Google Scholar] [CrossRef] [Green Version]
- Nieva-Echevarría, B.; Goicoechea, E.; Manzanos, M.J.; Guillén, M.D. Effects of different cooking methods on the lipids and volatile components of farmed and wild European sea bass (Dicentrarchus labrax). Food Res. Int. 2018, 103, 48–58. [Google Scholar] [CrossRef]
- Cropotova, J.; Mozuraityte, R.; Standal, I.B.; Rustad, T. The Influence of Cooking Parameters and Chilled Storage Time on Quality of Sous-Vide Atlantic Mackerel (Scomber scombrus). J. Aquat. Food Prod. Technol. 2019, 28, 505–518. [Google Scholar] [CrossRef]
- Bland, J.; Bett-Garber, K.; Li, C.H.; Brashear, S.S.; Lea, J.M.; Bechtel, P.J. Comparison of sensory and instrumental methods for the analysis of texture of cooked individually quick frozen and fresh-frozen catfish fillets. Food Sci. Nutr. 2018, 6, 1692–1705. [Google Scholar] [CrossRef]
- Xia, K.; Xu, W.; Huang, L.; Song, Y.; Zhu, B.-W.; Tan, M. Water dynamics of turbot flesh during frying, boiling, and stewing processes and its relationship with color and texture properties: Low-field NMR and MRI studies. J. Food Process. Preserv. 2017, 42, e13338. [Google Scholar] [CrossRef] [Green Version]
- Yuan, L.; Dang, Q.; Mu, J.; Feng, X.; Gao, R. Mobility and redistribution of waters within bighead carp (Aristichthys nobilis) heat-induced myosin gels. Int. J. Food Prop. 2018, 21, 835–849. [Google Scholar] [CrossRef] [Green Version]
- Grönqvist, S.; Skipnes, D.; Ohlsson, T.; Rosnes, J.T. Use of α-amylase TTI and Heat Transfer Modeling to Assess the Thermal Pasteurization of Fried Fish Burgers. J. Food Process. Preserv. 2013, 38, 1547–1555. [Google Scholar] [CrossRef]
- Verhaeghe, T.; Van Poucke, C.; Vlaemynck, G.; De Block, J.; Hendrickx, M. Kinetics of drosopterin release as indicator pigment for heat-induced color changes of brown shrimp (Crangon crangon). Food Chem. 2018, 254, 359–366. [Google Scholar] [CrossRef]
- Blikra, M.J.; Jessen, F.; Feyissa, A.H.; Vaka, M.R.; Skipnes, D. Low-concentration salting of cod loins: The effect on biochemical properties and predicted water retention during heating. LWT 2020, 118, 108702. [Google Scholar] [CrossRef]
- Llave, Y.; Shibata-Ishiwatari, N.; Watanabe, M.; Fukuoka, M.; Hamada-Sato, N.; Sakai, N. Analysis of the effects of thermal protein denaturation on the quality attributes of sous-vide cooked tuna. J. Food Process. Preserv. 2017, 42, e13347. [Google Scholar] [CrossRef] [Green Version]
- Skjelvareid, M.; Heia, K.; Olsen, S.-H.; Stormo, S.K. Detection of blood in fish muscle by constrained spectral unmixing of hyperspectral images. J. Food Eng. 2017, 212, 252–261. [Google Scholar] [CrossRef]
- Washburn, K.E.; Stormo, S.K.; Skjelvareid, M.; Heia, K. Non-invasive assessment of packaged cod freeze-thaw history by hyperspectral imaging. J. Food Eng. 2017, 205, 64–73. [Google Scholar] [CrossRef]
- Ma, J.; Sun, D.-W.; Pu, H.; Cheng, J.-H.; Wei, Q. Advanced Techniques for Hyperspectral Imaging in the Food Industry: Principles and Recent Applications. Annu. Rev. Food Sci. Technol. 2019, 10, 197–220. [Google Scholar] [CrossRef]
- Baiano, A. Applications of hyperspectral imaging for quality assessment of liquid based and semi-liquid food products: A review. J. Food Eng. 2017, 214, 10–15. [Google Scholar] [CrossRef]
- Hassoun, A.; Karoui, R. Quality Evaluation of Fish and Other Seafood by Traditional and Nondestructive Instrumental Methods: Advantages and Limitations. Crit. Rev. Food Sci. Nutr. 2015, 57. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Pu, H.; Sun, D.-W. Novel techniques for evaluating freshness quality attributes of fish: A review of recent developments. Trends Food Sci. Technol. 2019, 83, 259–273. [Google Scholar] [CrossRef]
- Hassoun, A.; Cropotova, J.; Rustad, T.; Heia, K.; Lindberg, S.-K.; Nilsen, H. Use of Spectroscopic Techniques for a Rapid and Non-Destructive Monitoring of Thermal Treatments and Storage Time of Sous-Vide Cooked Cod Fillets. Sensors 2020, 20, 2410. [Google Scholar] [CrossRef]
- Abbas, O.; Zadravec, M.; Baeten, V.; Mikuš, T.; Lešić, T.; Vulić, A.; Prpić, J.; Jemeršić, L.; Pleadin, J. Analytical methods used for the authentication of food of animal origin. Food Chem. 2018, 246, 6–17. [Google Scholar] [CrossRef]
- Uddin, M.; Okazaki, E.; Ahmad, M.U.; Fukuda, Y.; Tanaka, M. NIR spectroscopy: A non-destructive fast technique to verify heat treatment of fish-meat gel. Food Control 2006, 17, 660–664. [Google Scholar] [CrossRef]
- Gao, R.; Feng, X.; Li, W.; Yuan, L.; Ge, J.; Lu, D.; Chen, B.; Yu, G. Changes in properties of white shrimp (Litopenaeus vannamei) protein during thermal denaturation. Food Sci. Biotechnol. 2016, 25, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhu, Y.; Chen, S.; Xu, C.; Yu, Y.; Wang, X.; Shi, W. Determination of the effects of different high-temperature treatments on texture and aroma characteristics in Alaska pollock surimi. Food Sci. Nutr. 2018, 6, 2079–2091. [Google Scholar] [CrossRef] [PubMed]
- Ovissipour, M.; Shiroodi, S.G.; Rasco, B.; Tang, J.; Sablani, S.S. Electrolyzed water and mild-thermal processing of Atlantic salmon (Salmo salar): Reduction of Listeria monocytogenes and changes in protein structure. Int. J. Food Microbiol. 2018, 276, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Tavares, W.P.S.; Dong, S.; Jin, W.; Yang, Y.; Han, K.; Zha, F.; Zhao, Y.; Zeng, M. Effect of different cooking conditions on the profiles of Maillard reaction products and nutrient composition of hairtail (Thichiurus lepturus) fillets. Food Res. Int. 2018, 103, 390–397. [Google Scholar] [CrossRef]
- Argyri, A.A.; Jarvis, R.M.; Wedge, D.; Xu, Y.; Panagou, E.Z.; Goodacre, R.; Nychas, G.-J.E. A comparison of Raman and FT-IR spectroscopy for the prediction of meat spoilage. Food Control 2013, 29, 461–470. [Google Scholar] [CrossRef]
- Blümich, B.; Blümich, B. Introduction to compact NMR: A review of methods. TrAC Trends Anal. Chem. 2016, 83, 2–11. [Google Scholar] [CrossRef]
- Riegel, S.D.; Leskowitz, G.M. Benchtop NMR spectrometers in academic teaching. TrAC Trends Anal. Chem. 2016, 83, 27–38. [Google Scholar] [CrossRef]
- Shaikh, S.; O’Donnell, C. Applications of fluorescence spectroscopy in dairy processing: A review. Curr. Opin. Food Sci. 2017, 17, 16–24. [Google Scholar] [CrossRef]
- Sikorska, E.; Khmelinskii, I.; Sikorski, M. Fluorescence spectroscopy and imaging instruments for food quality evaluation. Eval. Technol. Food Qual. 2019, 491–533. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Nomi, Y.; Homma, T.; Kasai, M.; Otsuka, Y. Determination of Furosine and Fluorescence as Markers of the Maillard Reaction for the Evaluation of Meat Products during Actual Cooking Conditions. Food Sci. Technol. Res. 2012, 18, 67–76. [Google Scholar] [CrossRef] [Green Version]
- Sahar, A.; Rahman, U.U.; Kondjoyan, A.; Portanguen, S.; Dufour, É. Monitoring of thermal changes in meat by synchronous fluorescence spectroscopy. J. Food Eng. 2016, 168, 160–165. [Google Scholar] [CrossRef]
- Trevisan, A.J.B.; Lima, D.D.A.; Sampaio, G.R.; Soares, R.A.M.; Bastos, D.H.M. Influence of home cooking conditions on Maillard reaction products in beef. Food Chem. 2016, 196, 161–169. [Google Scholar] [CrossRef]
- Liu, J.; Zamora, A.; Castillo, M.; Saldo, J. Using front-face fluorescence spectroscopy for prediction of retinol loss in milk during thermal processing. LWT 2018, 87, 151–157. [Google Scholar] [CrossRef]
- Mungkarndee, R.; Techakriengkrai, I.; Tumcharern, G.; Sukwattanasinitt, M. Fluorescence sensor array for identification of commercial milk samples according to their thermal treatments. Food Chem. 2016, 197, 198–204. [Google Scholar] [CrossRef]
- Ahmad, N.; Saleem, M. Studying heating effects on desi ghee obtained from buffalo milk using fluorescence spectroscopy. PLoS ONE 2018, 13, e0197340. [Google Scholar] [CrossRef] [Green Version]
- Tan, J.; Li, R.; Jiang, Z.-T.; Tang, S.-H.; Wang, Y.; Shi, M.; Xiao, Y.-Q.; Jia, B.; Lu, T.-X.; Wang, H. Synchronous front-face fluorescence spectroscopy for authentication of the adulteration of edible vegetable oil with refined used frying oil. Food Chem. 2017, 217, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Bui, M.V.; Rahman, M.; Nakazawa, N.; Okazaki, E.; Nakauchi, S. Visualize the quality of frozen fish using fluorescence imaging aided with excitation-emission matrix. Opt. Express 2018, 26, 22954–22964. [Google Scholar] [CrossRef] [PubMed]



| Fish or Other Seafood | Cooking Method (s) | Quality Parameter (s) | Main Results | References |
|---|---|---|---|---|
| Hairtail | Boiling, baking, and frying | Nutritional quality: digestibility | Boiled fillets exhibited higher digestibility compared to the fillets cooked by baking or frying | Tavares et al. 2018 [10] |
| Salmon | Heating in an oil bath | Physico-chemical parameters: cooking loss, area shrinkage, color, texture | Most of the cooking loss and area shrinkage occurred within the first half hour of cooking, then approached an equilibrium. Color tended to be whiter, then browner, and texture gradually became soft as heating progressed | Kong et al. 2007 [42] |
| Blue mussel | Heating in an oil bath | Physico-chemical parameters: cooking loss, protein shrinkage, and texture | Cooking loss increased with increasing cooking time and temperature and correlated positively with area shrinkage and compression force | Ovissipour et al. 2013 [43] |
| Sea bream surimi | Microwave and water bath | Physico-chemical parameters: gel strength, WHC, microstructure morphology | Microwave- and water bath-treated samples demonstrated better gel properties (stronger gel with finer texture and increased WHC) than samples heated only with microwave | Cao et al. 2018 [39] |
| Sea bream surimi | Microwave and water bath | Gel texture properties, dielectric properties, WHC, cooking loss, color | Surimi paste with a thickness of 2 cm of surimi gave the highest WHC, lowest cooking loss, uniformity of temperature distribution, higher tensile force and whiteness | Cao et al. 2019 [30] |
| Sturgeon | Boiling, steaming, microwaving, roasting, and deep-frying | Physico-chemical parameters: protein (carbonyls, Schiff bases, and free thiols) and lipid (TBARS) oxidation | Higher proteins and lipid oxidation in roasted and fried samples than those cooked under less intense cooking conditions (boiling, steaming and microwaving) | Hu et al. 2017 [44] |
| Atlantic salmon | Microwave and conventional pasteurization | Sensory, microbial growth, protein denaturation, color, and liquid loss | Combination of CO2 with heating increased shelf life compared to heating (with microwave or conventional pasteurization) without presence of CO2 in vacuum-package | Lerfall et al. 2018 [28] |
| Shrimp | Water bath | Cooking loss, shrinkage, texture, and color | Hardness, cooking loss, area shrinkage, and overall color change increased with thermal load | Wang et al. 2018 [45] |
| Grass carp | Microwave | Denaturation and aggregation of sarcoplasmic and myofibrillar proteins | Increased microwave power and treatment time induced a decrease in solubility and an increase in turbidity, indicating a more aggregation of proteins | Cai et al. 2017 [46] |
| Atlantic cod | Water bath | Microbial and sensory analysis, pH, and liquid loss | Mild heat treatment with high temperature and short cooking time decreased liquid loss and inactivated bacteria of the muscle surface, but did not extend shelf life significantly compared to samples cooked at lower temperature for longer time | Stormo et al. 2018 [19] |
| Fish patties: silver smelt | Water bath | Microbial growth, water content, lipid content, pH | Deceased bacterial load with increasing cooking temperature and time. Increased shelf life for samples packed with high CO2 levels compared to regular modified atmosphere packaging or vacuum | Abel et al. 2019 [47] |
| Cod | Convection oven | Rheological analysis, WHC, mass loss | Development of a model to predict temperature and moisture concentration based on both heat and mass transfer during cooking | Blikra et al. 2019 [48] |
| False abalone | Boiling | Sensory, texture properties, protein denaturation, water distribution | Increasing cooking time resulted in a decrease in shear force and sensory acceptability, with a rapid denaturation of proteins (denaturation at the first minute). Development a model to predict myosin and actin denaturation | He et al. 2018 [49] |
| European sea bass | Microwave and conventional oven | Lipid content and composition, and volatile compounds | Conventional oven backing gave higher abundances of volatile compounds than microwave cooking and salt-crusted oven baking | Nieva-Echevarría et al. 2018 [50] |
| Atlantic cod | Water bath | Cooking loss, WHC, color, and texture | Optimal cooking conditions by cooking with heat that is equivalent to 2 min at 70 °C, thus keeping cooking loss below 5.6% and WHC above 66%, along with low hardness and whiteness values | Skipnes et al. 2011 [9] |
| Atlantic Mackerel | Water bath | Protein solubility, lipid oxidation, color, texture, cooking loss, and pH | Increase in lipid oxidation products, lightness, yellowness with increasing storage time. Liquid loss increased with increasing cooking temperature but decreased with storage time. Heating caused a reduction in protein solubility | Cropotova et al. 2019 [51] |
| Fish or Other Seafood | Applied Technique | Wavelength or Wavenumber Range | Model | Key Issues-Outcomes | References |
|---|---|---|---|---|---|
| Walleye pollack and horse mackerel gel | VIS/NIR | 650–1100 nm | PLSR, MLR | High accuracy of prediction of heating temperature (R = 0.98 with prediction error of 1.85 °C). Spectral changes were attributed to protein denaturation and changes in the state of water, induced by the heat | Uddin et al. 2006 [67] |
| Kamaboko | NIR | 900–2500 nm | PLSR, LAD | Reasonable level of accuracy for prediction of core temperature (R2 = 0.86, with prediction error 3.9 °C) and thermal history (R2 = 0.83 with prediction error 0.29 min) and high classification accuracy | Elmasry et al. 2015 [4] |
| Surimi | VIS/NIR | 400–2500 nm | PLSR | Best results for prediction of cooking surface temperature in the visible range 400–550 nm (R2 ≥ 0.9) with prediction errors lower than 3 °C | Skåra et al. 2014 [18] |
| Surimi | VIS/NIR | 400–2500 nm | PLSR | Endpoint temperature was best predicted in the visible range 400–700 nm (R2 = 0.96) with a small prediction error (<2 °C) | Stormo et al. 2012 [19] |
| Fish cakes | NIR | 760–1040 nm | PLSR | Prediction core temperature in the fish cakes with prediction errors of 2.3 °C (NIR point system) and 4.5 °C (imaging system) | Wold et al. 2016 [7] |
| Bighead carp | Raman NMR | 400–3500 cm−1 22.6 MHz | PCA | Decrease in α-helix structures, reflecting changes in myosin secondary structures with increasing heat treatment. Water distribution and mobility of water change with thermal treatments | Yuan et al. 2018 [54] |
| False abalone | NMR, MRI | 21.3 MHz | PLSR | Providing quantitative characterizations of actin and myosin protein denaturation and water distribution on the quality of the product | He et al. 2018 [49] |
| Turbot | NMR, MRI | 21.2 MHz | PCA | Different water dynamics according to cooking method. Good correlation between NMR relaxation parameters and texture, color measurements. Internal structure visualized by MRI | Xia et al. 2017 [53] |
| White Shrimp | Raman | 1600–1700 cm−1 2200–3500 cm−1 | Univariate analysis | Various bands of Raman spectra demonstrated changes in hydrogen bonding and protein denaturation | Gao et al. 2016 [68] |
| Hairtail surimi | FTIR, Raman | 400–3500 cm−1 300–1800 cm−1 | Univariate analysis | Changes in secondary structures of protein, reflected in a decrease in α-helix and an increase in β-sheet structures, indicating protein denaturation induced by irradiation and heat treatment | Lin et al. 2015 [40] |
| Alaska pollock surimi | FTIR | 4000–400 cm−1 | PCA | Increasing cooking temperature reduced the gel strength of the surimi as a result of changes in protein secondary structures | Zhang et al. 2018 [69] |
| Atlantic salmon | FTIR | 4000–400 cm−1 | PCA | Amid I region revealed increased denaturation and aggregation of proteins with increasing cooking temperature and cooking time | Ovissipour et al. 2017 [6] |
| Atlantic salmon | FTIR | 4000–400 cm−1 | PCA | Cooking combined with electrolyzed water decreased strongly bacterial growth of Listeria monocytogenes. Higher protein denaturation and unfolding with increasing cooking temperature | Ovissipour et al. 2018 [70] |
| Hairtail | Fluorescence | Ex; 347 nm Em; 415 nm | Univariate analysis | Increased fluorescence was obtained in cooked fish as compared to raw samples. More fluorescence was noticed from baked and fried fillets as compared to boiled ones | Tavares et al. 2018 [71] |
| Sturgeon | Fluorescence | Ex; 360 nm Em; 380–600 nm | Univariate analysis | Roasting and frying cooking methods induced significant increases in fluorescence, as a result of formation of Schiff bases compounds, compared to boiling and steaming cooking methods | Hu et al. 2017 [44] |
| Sturgeon | Fluorescence | Ex; 360 nm Em; 380–600 nm | Univariate analysis | Fluorescence was increased after digestion (especially after gastrointestinal digestion) as compared to fluorescence of samples before digestion. Changes in spectral patters (shape and intensity) were observed with different roasting times | Hu et al. 2018 [11] |
| Atlantic mackerel | Fluorescence | Ex; 365 nm Em; 445 nm | Univariate analysis | Direct relationship between total collagen content and hardness, and total fluorescence intensity of collagenous tissue | Cropotova et al. 2018 [17] |
| Atlantic mackerel | Fluorescence | Ex; 475 nm Em; 530 nm | Univariate analysis | High correlation between fluorescence and lipid oxidation products (primary and secondary indicators of lipid oxidation) | Cropotova et al. 2019 [20] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hassoun, A.; Heia, K.; Lindberg, S.-K.; Nilsen, H. Spectroscopic Techniques for Monitoring Thermal Treatments in Fish and Other Seafood: A Review of Recent Developments and Applications. Foods 2020, 9, 767. https://doi.org/10.3390/foods9060767
Hassoun A, Heia K, Lindberg S-K, Nilsen H. Spectroscopic Techniques for Monitoring Thermal Treatments in Fish and Other Seafood: A Review of Recent Developments and Applications. Foods. 2020; 9(6):767. https://doi.org/10.3390/foods9060767
Chicago/Turabian StyleHassoun, Abdo, Karsten Heia, Stein-Kato Lindberg, and Heidi Nilsen. 2020. "Spectroscopic Techniques for Monitoring Thermal Treatments in Fish and Other Seafood: A Review of Recent Developments and Applications" Foods 9, no. 6: 767. https://doi.org/10.3390/foods9060767





