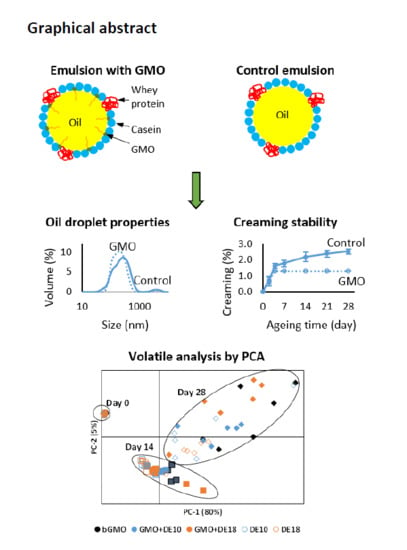
3.2. Droplet Size and Polydispersity Index
Table 3 shows the droplet size and polydispersity index of model emulsions formulated with different emulsifier systems over 28 days of storage at 45 °C. The average droplet size of the control emulsions (DE10 and DE18; without GMO) differed significantly from emulsions containing GMO (bGMO, GMO + DE10 and GMO + DE18) (
p < 0.05). Fresh and aged emulsions with GMO had significantly smaller droplet sizes (172–186 nm) than the controls without GMO (194–200 nm). This observation agrees with previous findings [
8] that emulsions with monoglycerides form smaller droplets than controls. However, emulsion droplet sizes were the same (
p > 0.05) for bulk GMO and instantized GMO powders of different DE values of maltodextrin. The sodium stearate from the GMO powders was very low (around 80 ppm in the emulsions) and did not affect the droplet size. Both controls with either maltodextrin DE10 and DE18 also had the same droplet sizes, illustrating no apparent effect of maltodextrin type on the emulsion properties.
Droplet sizes for all emulsion samples were stable during 28 days of storage at 45 °C. This finding concurs with previous works on protein-stabilized emulsions that indicated droplet sizes were stable during storage, without any significant droplet growth due to flocculation, coalescence, and Ostwald ripening [
8,
18,
19,
20].
Table 3b shows the polydispersity index of emulsions over 28 days of storage at 45 °C. All emulsions with GMO and controls had a polydispersity index of 0.2 or lower, which indicated narrow size-range distributions. The volume-weighted droplet size distribution showed that all fresh and 28-day aged emulsions had a pseudo-monomodal size distribution (
Figure 2), supporting the polydispersity index results. The fine droplets around 20 nm in the 28-day aged DE10 sample were less than 1.5%, hence did not affect its polydispersity index. The polydispersity index values were 18% lower for emulsions formulated with GMO compared to the controls. Regardless of storage time, emulsions with bulk GMO and instantized GMO had smaller average droplet sizes and narrower droplet size distributions than controls. Both control emulsions with maltodextrin with different DE values did not show any significant difference in terms of droplet size and polydispersity index from each other. This observation reaffirmed that maltodextrin at this level had no significant effect on the droplet size and its size-range distribution, which is in line with the findings by Gharsallaoui et al. [
21]. In our previous study, the emulsions with a sodium stearate concentration up to 120 ppm did not show any significant change to the emulsion droplet size when compared to the control emulsions without sodium stearate [
8]. All emulsions had similar protein composition and concentration, thus suggesting that GMO is responsible for the reduction in droplet size of the emulsions.
3.3. ζ-Potential
Table 4 shows the ζ-potential of model emulsions formulated with different emulsifier systems over 28 days of storage at 45 °C. All fresh emulsions (Day 0) formulated with different emulsifier systems had a ζ-potential in the range of −51 to −53 mV. In order to simplify the discussion, as all emulsions in this study exhibited negative ζ-potential values, the negative sign will be ignored and only the magnitude will be discussed. Hence, an increase in ζ-potential means the increase in the negativity of the ζ-potential. The ζ-potential provides information on the repulsive forces at the oil-water interface, which can explain droplet stability in the emulsions [
22]. All emulsions with different emulsifier systems showed statistically significant differences after Day 7 (
p < 0.05) when comparing the samples at each timepoint, but the small differences in ζ-potential (<3 mV) are not useful to distinguish the stability of emulsifier systems, and all samples can be considered stable [
16]. Sodium stearate in the instantized powders increased the ζ-potential of the emulsions, similar to previous findings [
8]. The increase in ζ-potential by sodium stearate was potentially due to the adsorption of anionic molecules at the oil-water interface, thereby increasing the ionic repulsive force among the oil droplets. The similarity in ζ-potential of the different samples at the same storage time indicates that the emulsion droplets are predominantly stabilized by protein [
23] and not much protein displacement by the emulsifier is taking place. It is hypothesized that sodium caseinate contributes to the negative ζ-potential in the soluble form and the slightly lower ζ-potential value after storage might be due to partial precipitation of sodium caseinate [
24]. All emulsions showed a decreasing trend in ζ-potential with storage time (
p < 0.05), with a small reduction of ζ-potential (<5 mV) over 28 days of storage. However, this may not have any practical implication because the oil droplets still retained high repulsive forces (−47 to −50 mV). Ross and Morrison [
25] stated that oil droplets with a ζ-potential below −30 mV have excellent stability against flocculation or coalescence. The stable droplet size distribution during storage also reaffirms the absence of flocculation or coalescence.
3.7. Oxidative Stability of Model Emulsions Measured Using Volatile Analysis
The development of volatile compounds was monitored in all model emulsions with different emulsifier systems at various storage times using headspace SPME with GC-MS as a tool to evaluate changes in secondary oxidation products during storage. Volatile compounds that increased significantly (p < 0.05) during storage and had been reported as markers of lipid oxidation in the literature were reported in this study. Compounds with reduced or no change in concentrations during storage were not reported. Analysis of the volatile compounds detected 36 compounds (including two unknowns) that increased significantly in the model emulsions during storage in the model emulsions, from four chemical classes, namely aldehydes, furans, alcohols, and ketones. Hexanal was present with the highest abundance relative to other identified compounds.
Figure 4 shows the scores and loadings plot of principal component analysis (PCA) for the volatile analysis of the model emulsions at various storage times (Day 0, 14, 28). The first principal component (PC-1) explains most of the variation (80%) in the data and PC-2 explains 5% of the variation. Sample storage time (fresh vs. stored) was the main cause of data variation in the PCA model, and by using the first two principal components, the samples can be grouped into three clusters according to the storage time, namely Day 0, 14, and 28. Storage at 45 °C was the main cause of variation on PC-1 in the scores plot to differentiate fresh and stored emulsions. The discrimination on PC-1 was due to the abundance of alcohols (1-pentanol, 1-heptanol, 1-octanol, and 1 octen-3-ol), ketones (3-octen-2-one, 6-methyl-5-heptene-2-one, 2-heptanone, and 2 octanone), and furans (2-ethyl furan and 2-propyl furan), which were positively loaded on PC-1. PC-2 was able to distinguish the samples stored at 14 and 28 days by aldehydes, such as propanal, pentanal, hexanal, heptanal, octanal, 2-pentenal, and 2,4-heptadienal, which clustered together at the bottom of the loading plot that were associated with higher concentrations at Day 14.
The fresh control emulsions had eight volatile compounds that significantly increased during storage, namely pentanal, hexanal, heptanal, nonanal, 2,4-heptadienal isomer 2, 1-penten-3-ol, and two isomers of 3,5-octadien-2-one. In addition to the compounds that appeared in the controls, the fresh emulsions with bulk and instantized GMO had additional compounds, including 2-heptenal, 1-pentanol, 1-heptanol, 1-octanol, and 1-octen-3-ol. The number of compounds that significantly increased during storage increased to 35 compounds in the control emulsions after 28 days of storage, while the emulsions with GMO had 36 compounds. None of the furan compounds were detected in the fresh emulsions.
To investigate more closely the difference in the degree of oxidation between the different emulsions, three compounds, namely hexanal, octanal, and 1-octen-3-ol, were selected as oxidation markers. These compounds have been commonly used as lipid oxidation markers in past literature [
10,
15,
29,
30]. Hexanal and 1-octen-3-ol are formed by oxidation of linoleic acid, while octanal is derived from oleic acid [
31,
32]. Calibration curves for hexanal (R
2 = 0.998), octanal (R
2 = 0.996), and 1-octen-3-ol (R
2 = 0.999) in the range of 0–900 µg/L were used for semi-quantification.
Table 5 shows the concentration of the calibrated compounds at various storage times. The concentration of hexanal in the 28-day stored emulsions was outside of the calibration curve range and was determined by extrapolating the calibration curve beyond 900 µg/L with the assumption that the response continued to be linear.
All emulsions at Day 0 showed comparable low concentrations of hexanal and octanal, while 1- octen-3-ol was only detected in the emulsions with bulk GMO. This observation indicated that canola oil and GMO used to prepare the emulsions had a low degree of oxidation. The absence of 1-octen-3-ol in control emulsions (DE10 and DE18) indicated that this compound was present in the bulk GMO, while the non-detectable level in instantized GMO may be due to the evaporation during spray-drying. All three oxidation markers increased with the increasing storage time, which aligned with the PCA results, where compounds were positively loaded on PC-1 (
Figure 4). The oxidation markers in the 28-day aged emulsions did not show any effect of the emulsifier system on oxidative stability.
Table 6 shows additional 12 selected lipid oxidation compounds in the 28-day aged emulsions derived from oleic, linoleic, and linolenic acids [
31] that had high loadings on PC-1. In bulk GMO, four compounds, namely propanal, 2,4-heptadienal isomer 2, 3 octen-2-one, and 3,5-octadien-2-one isomer 1, were significantly higher in concentration than in the controls (
p < 0.05). All selected compounds, except propanal, indicated that there was no difference between instantized GMO samples and the controls. This observation was positive because it indicated that instantized GMO at a low concentration did not negatively affect the oxidative stability of protein-stabilized emulsions.
There was no influence of emulsion droplet size on lipid oxidation, which agrees with findings by Osborn and Akoh [
33] and Dimakou et al. [
34]. All emulsions also had very similar ζ-potentials, which indicated that the emulsions had similar protein composition in the aqueous phase and at the emulsion interface. Previous research [
15] on the influence of protein composition on physical stability hypothesized that the formation of a compact multilayer interface comprised of sodium caseinate and whey proteins could improve oxidative stability in lipid emulsions. In this study, the emulsions with bulk and instantized GMO had the same oxidative stability as controls without GMO. The same protein composition at the interface indicated by the ζ-potential could have a bigger influence on oxidative stability compared to GMO at a low concentration and mask any effect by the GMO.







_Loi.jpg)