Comparative Analysis of Physicochemical Characteristics, Nutritional and Functional Components and Antioxidant Capacity of Fifteen Kiwifruit (Actinidia) Cultivars—Comparative Analysis of Fifteen Kiwifruit (Actinidia) Cultivars
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples and Chemicals
2.2. Total Soluble Solids (TSS), Titratable Acid (TA), pH
2.3. Ascorbic Acid Content (AAC), Total Polyphenol Content (TPC), Total Flavonoids Content (TFC), Total Anthocyanins Content (TAC)
2.3.1. Extracts
2.3.2. Determination
2.4. Analysis of Antioxidant Capacity
2.5. Protein and Free Amino Acids
2.6. Analysis of Mineral Elements
2.7. Analysis of Folic Acid (FA) and L-5-Methyltetrahydrofolate (5MTHF)
2.8. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Characteristics
3.1.1. Total Soluble Solid (TSS), Titratable Acid (TA) and pH
3.1.2. Sugar-Acid Ratio
3.2. AAC, TPC, TFC, TAC
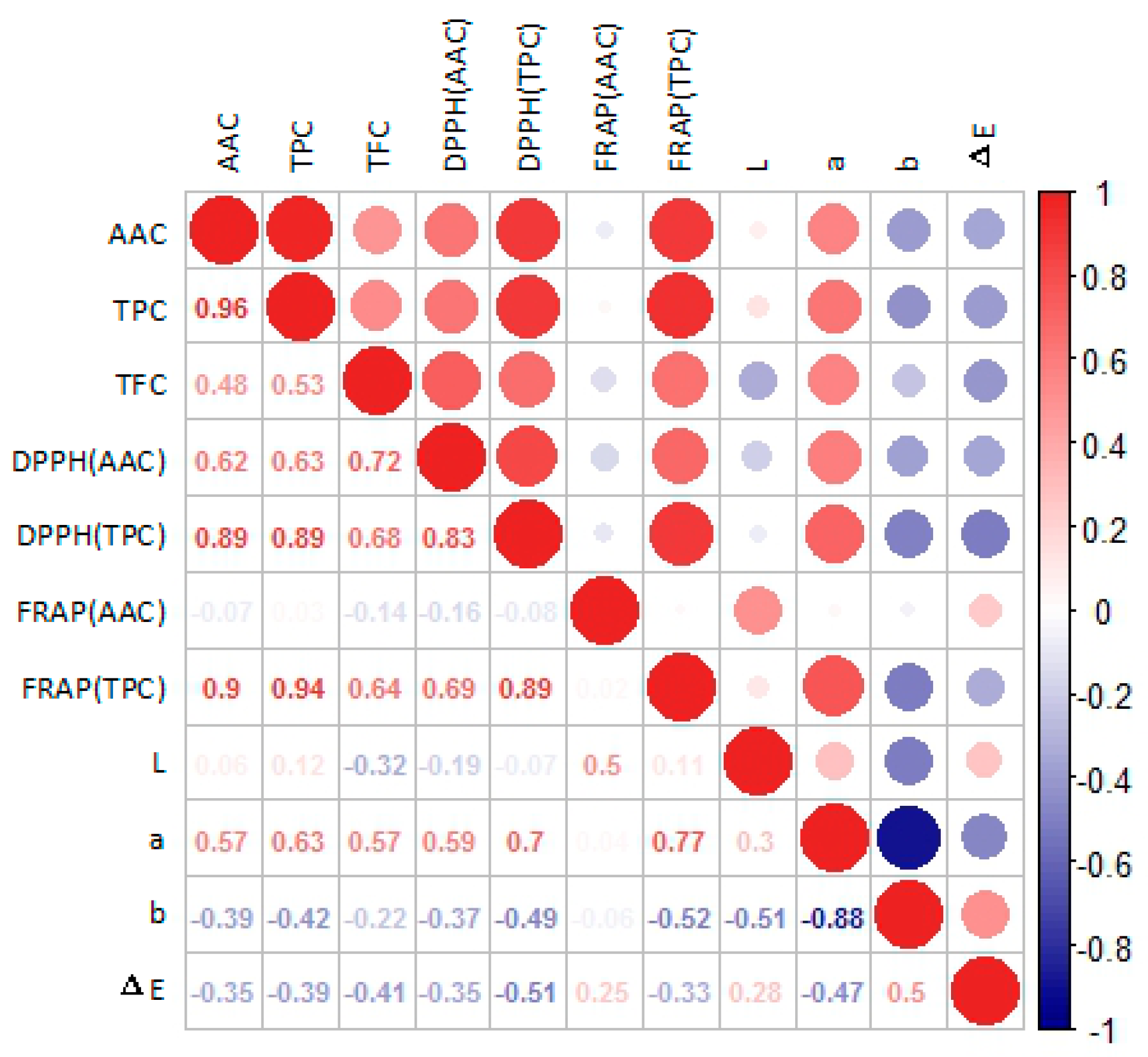
3.3. Antioxidant Activities
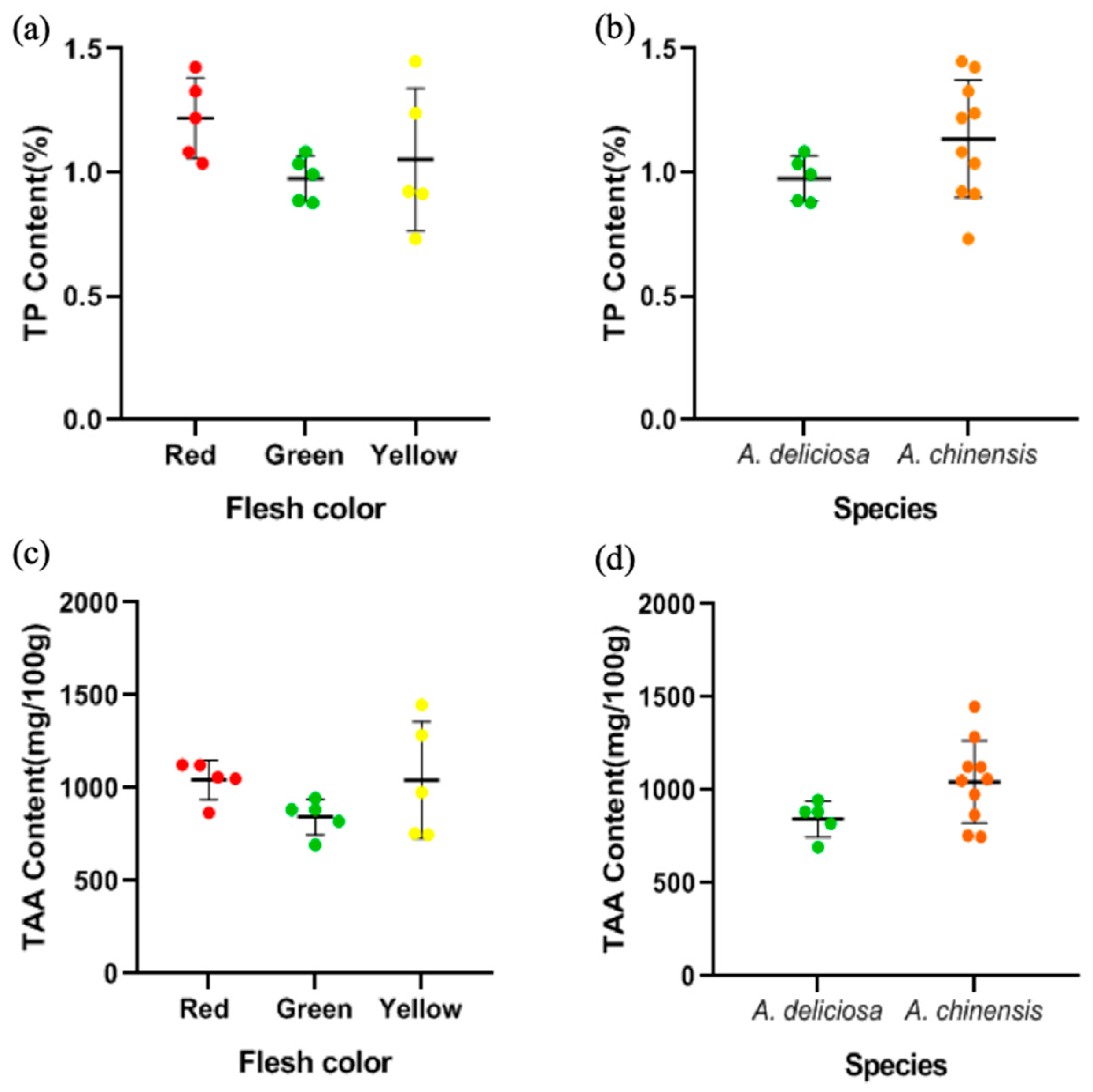
3.4. Total Protein (TP) Content and Total Free Amino Acids (TAA) Content
3.5. Mineral Elements
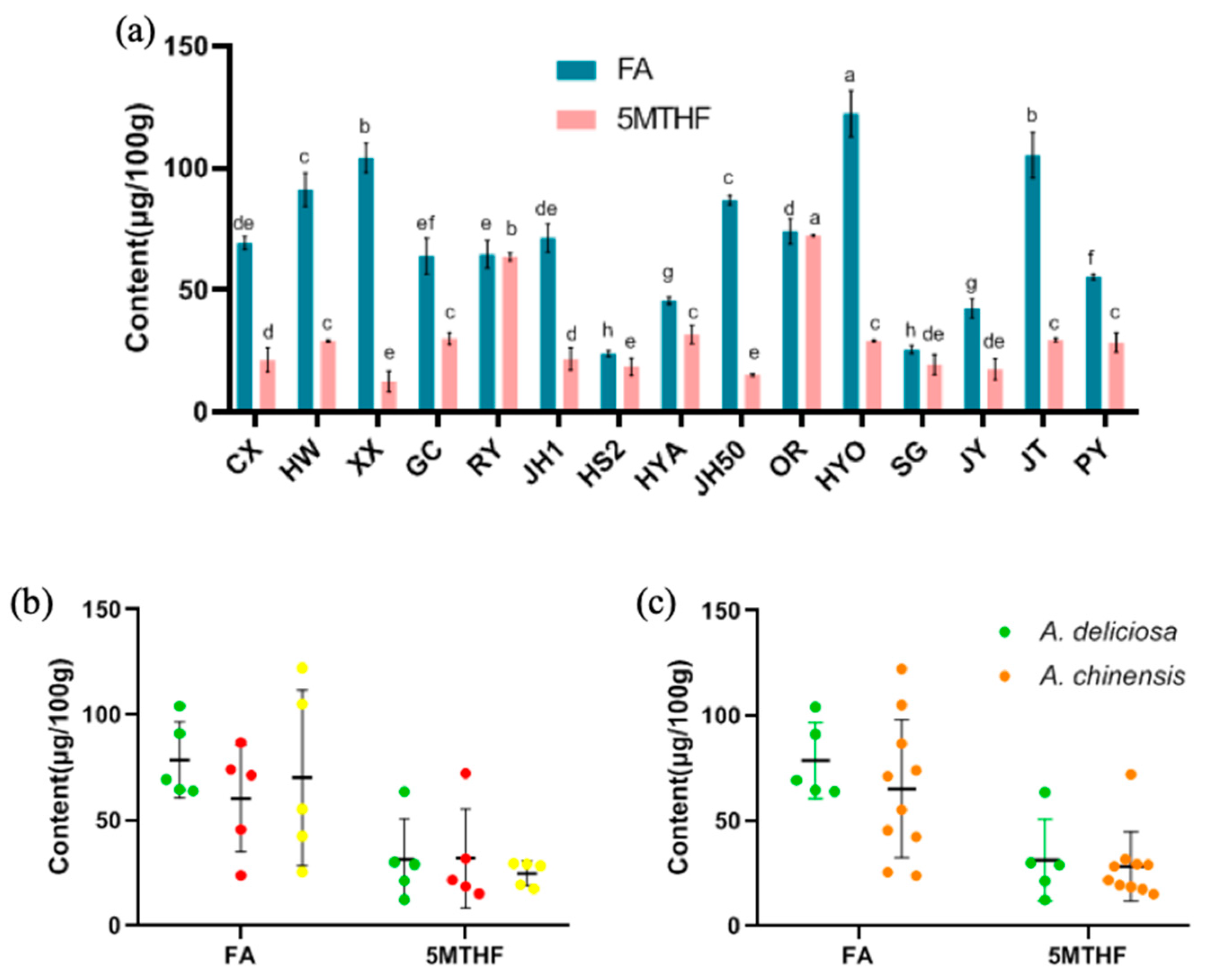
3.6. FA and 5MTHF
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yue, J.; Liu, J.; Ban, R.; Tang, W.; Deng, L.; Fei, Z.; Liu, Y. Kiwifruit Information Resource (KIR): A comparative platform for kiwifruit genomics. Database 2015, 2015. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, Q.; Zhong, C.; Guo, M. Volatile fingerprints and biomarkers of three representative kiwifruit cultivars obtained by headspace solid-phase microextraction gas chromatography mass spectrometry and chemometrics. Food Chem. 2019, 271, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.; Quek, S.; Stevenson, R.; Winz, R. Kiwifruit flavour: A review. Trends Food Sci. Technol. 2012, 24, 82–91. [Google Scholar] [CrossRef]
- Flores, G.; Wu, S.; Negrin, A.; Kennelly, E. Chemical composition and antioxidant activity of seven cultivars of guava (Psidium guajava) fruits. Food Chem. 2015, 170, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Montefiori, M.; McGhie, T.K.; Hallett, I.C.; Costa, G. Changes in pigments and plastid ultrastructure during ripening of green-fleshed and yellow-fleshed kiwifruit. Sci. Hortic. 2009, 119, 377–387. [Google Scholar] [CrossRef]
- Harker, F.; Carr, B.; Lenjo, M.; MacRae, E.; Wismer, W.; Marsh, K.; Williams, M.; White, A.; Lund, C.; Walker, S.; et al. Consumer liking for kiwifruit flavour: A meta-analysis of five studies on fruit quality. Food Qual. Prefer. 2009, 20, 30–41. [Google Scholar] [CrossRef]
- Cozzolino, R.; De Giulio, B.; Petriccione, M.; Martignetti, A.; Malorni, L.; Zampella, L.; Laurino, C.; Pellicano, M. Comparative analysis of volatile metabolites, quality and sensory attributes of Actinidia chinensis fruit. Food Chem. 2020, 316, 126340. [Google Scholar] [CrossRef]
- Richardson, D.; Ansell, J.; Drummond, L. The nutritional and health attributes of kiwifruit: A review. Eur. J. Nutr. 2018, 57, 2659–2676. [Google Scholar] [CrossRef]
- Ma, T.; Lan, T.; Ju, Y.; Cheng, G.; Que, Z.; Geng, T.; Fang, Y.; Sun, X. Comparison of the nutritional properties and biological activities of kiwifruit (Actinidia) and their different forms of products: Towards making kiwifruit more nutritious and functional. Food Funct. 2019, 10, 1317–1329. [Google Scholar] [CrossRef]
- Peng, Y.; Cordiner, S.; Sawyer, G.; McGhie, T.; Espley, R.; Allan, A.; Hurst, R. Kiwifruit with high anthocyanin content modulates NF-κB activation and reduces CCL11 secretion in human alveolar epithelial cells. J. Funct. Foods 2020, 65, 103734. [Google Scholar] [CrossRef]
- Saeed, K.; You, L.; Chen, C.; Zhao, Z.; Fu, X.; Liu, R. Comparative assessment of phytochemical profiles and antioxidant and antiproliferative activities of kiwifruit (Actinidia deliciosa) cultivars. J. Food Biochem. 2019, 43. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Yuan, Q.; Yang, Y.; Han, Q.; He, J.; Zhao, L.; Zhang, Q.; Liu, S.; Lin, D.; Wu, D.; et al. Phenolic profiles, antioxidant capacities, and inhibitory effects on digestive enzymes of different kiwifruits. Molecules 2018, 23, 2957. [Google Scholar] [CrossRef] [PubMed]
- Lizcano, L.J.; Bakkali, F.; Begoña Ruiz-Larrea, M.; Ignacio Ruiz-Sanz, J. Antioxidant activity and polyphenol content of aqueous extracts from Colombian Amazonian plants with medicinal use. Food Chem. 2010, 119, 1566–1570. [Google Scholar] [CrossRef]
- Zuo, L.-L.; Wang, Z.-Y.; Fan, Z.-L.; Tian, S.-Q.; Liu, J.-R. Evaluation of antioxidant and antiproliferative properties of three actinidia (Actinidia kolomikta, Actinidia arguta, Actinidia chinensis) extracts in vitro. Int. J. Mol. Sci. 2012, 13, 5506–5518. [Google Scholar] [CrossRef]
- Silva, A.M.; Pinto, D.; Fernandes, I.; Gonçalves Albuquerque, T.; Costa, H.S.; Freitas, V.; Rodrigues, F.; Oliveira, M.B.P.P. Infusions and decoctions of dehydrated fruits of Actinidia arguta and Actinidia deliciosa: Bioactivity, radical scavenging activity and effects on cells viability. Food Chem. 2019, 289, 625–634. [Google Scholar] [CrossRef]
- Gorinstein, S.; Haruenkit, R.; Poovarodom, S.; Park, Y.-S.; Vearasilp, S.; Suhaj, M.; Ham, K.-S.; Heo, B.-G.; Cho, J.-Y.; Jang, H.G. The comparative characteristics of snake and kiwi fruits. Food Chem. Toxicol. 2009, 47, 1884–1891. [Google Scholar] [CrossRef]
- Paulauskienė, A.; Tarasevičienė, Ž.; Žebrauskienė, A. Amino acid composition of kolomikta actinidia (Actinidia kolomikta (Maxim. and Rupr.) Maxim) fruits of Lithuanian origin. Zemdirbyste 2014, 101, 79–84. [Google Scholar] [CrossRef][Green Version]
- Salzano, A.M.; Sobolev, A.; Carbone, V.; Petriccione, M.; Renzone, G.; Capitani, D.; Vitale, M.; Minasi, P.; Pasquariello, M.S.; Novi, G.; et al. A proteometabolomic study of Actinidia deliciosa fruit development. J. Proteom. 2018, 172, 11–24. [Google Scholar] [CrossRef]
- Sivakumaran, S.; Huffman, L.; Sivakumaran, S.; Drummond, L. The nutritional composition of Zespri® SunGold Kiwifruit and Zespri® Sweet Green Kiwifruit. Food Chem. 2018, 238, 195–202. [Google Scholar] [CrossRef]
- Ma, T.; Lan, T.; Geng, T.; Ju, Y.; Cheng, G.; Que, Z.; Gao, G.; Fang, Y.; Sun, X. Nutritional properties and biological activities of kiwifruit (Actinidia) and kiwifruit products under simulated gastrointestinal in vitro digestion. Food Nutr. Res. 2019, 63. [Google Scholar] [CrossRef]
- Ma, T.; Sun, X.; Zhao, J.; You, Y.; Lei, Y.; Gao, G.; Zhan, J. Nutrient compositions and antioxidant capacity of kiwifruit (Actinidia) and their relationship with flesh color and commercial value. Food Chem. 2017, 218, 294–304. [Google Scholar] [CrossRef] [PubMed]
- The National Standard of China. Refractometric Method for Determination of Total Soluble Solid in Fruits and Vegetables; NY/T 2637-2014; CN-NY: Beijing, China, 2014. [Google Scholar]
- The National Standard of China. GB/T 12456-2008. Determination of Total Acid in Food; CN-GB: Beijing, China, 2008. [Google Scholar]
- The National Standard of China. GB 10468-1989. Determination of pH in Fruits and Vegetable Product; CN-GB: Beijing, China, 1989. [Google Scholar]
- Sun, X.; Wei, X.; Zhang, J.; Ge, Q.; Liang, Y.; Ju, Y.; Zhang, A.; Ma, T.; Fang, Y. Biomass estimation and physicochemical characterization of winter vine prunings in the Chinese and global grape and wine industries. Waste Manag. 2020, 104, 119–129. [Google Scholar] [CrossRef]
- The National Standard of China. GB/T 6324.6-2014. Determination of Color for Liquids; CN-GB: Beijing, China, 2014. [Google Scholar]
- The National Standard of China. GB 5009.86-2016. Determination of Amino acid in Food; CN-GB: Beijing, China, 2016. [Google Scholar]
- Du, G.; Li, M.; Ma, F.; Liang, D. Antioxidant capacity and the relationship with polyphenol and Vitamin C in Actinidia fruits. Food Chem. 2009, 113, 557–562. [Google Scholar] [CrossRef]
- Sun, X.; Cheng, X.; Zhang, J.; Ju, Y.; Que, Z.; Liao, X.; Lao, F.; Fang, Y.; Ma, T. Letting wine polyphenols functional: Estimation of wine polyphenols bioaccessibility under different drinking amount and drinking patterns. Food Res. Int. 2020, 127, 108704. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Ma, T.; Han, L.; Huang, W.; Zhan, J. Effects of copper pollution on the phenolic compound content, color, and antioxidant activity of wine. Molecules 2017, 22, 726. [Google Scholar] [CrossRef]
- The National Standard of China. GB 5009.5-2016. Determination of Protein in Food; CN-GB: Beijing, China, 2016. [Google Scholar]
- The National Standard of China. GB 5009.124-2016. Determination of Amino Acid in Food; CN-GB: Beijing, China, 2016. [Google Scholar]
- The National Standard of China. GB 5009.92-2016. Determination of Calcium in Food; CN-GB: Beijing, China, 2016. [Google Scholar]
- The National Standard of China. GB 5009.91-2017. Determination of Potassium and Sodium in Food; CN-GB: Beijing, China, 2017. [Google Scholar]
- The National Standard of China. GB 5009.13-2017. Determination of Copper in Food; CN-GB: Beijing, China, 2017. [Google Scholar]
- The National Standard of China. GB 5009.14-2017. Determination of Zinc in Food; CN-GB: Beijing, China, 2017. [Google Scholar]
- The National Standard of China. GB 5009.241-2017. Determination of Magnesium in Food; CN-GB: Beijing, China, 2017. [Google Scholar]
- The National Standard of China. GB 5009.87-2016. Determination of Phosphorus in Food; CN-GB: Beijing, China, 2016. [Google Scholar]
- The National Standard of China. GB 5009.241-2016. Determination of Ferrum in Food; CN-GB: Beijing, China, 2016. [Google Scholar]
- Czarnowska-Kujawska, M.; Gujska, E.; Michalak, J. Testing of different extraction procedures for folate HPLC determination in fresh fruits and vegetables. J. Food Compos. Anal. 2017, 57, 64–72. [Google Scholar] [CrossRef]
- Wojdyło, A.; Nowicka, P.; Oszmiański, J.; Golis, T. Phytochemical compounds and biological effects of Actinidia fruits. J. Funct. Foods 2017, 30, 194–202. [Google Scholar] [CrossRef]
- Zaouay, F.; Mena, P.; Garcia-Viguera, C.; Mars, M. Antioxidant activity and physico-chemical properties of Tunisian grown pomegranate (Punica granatum L.) cultivars. Ind. Crop Prod. 2012, 40, 81–89. [Google Scholar] [CrossRef]
- Drewnowski, A. The Nutrient Rich Foods Index helps to identify healthy, affordable foods. Am. J. Clin. Nutr. 2010, 91, 1095S–1101S. [Google Scholar] [CrossRef]
- Li, D.; Zhong, C.; Liu, Y.; Huang, H. Correlation between ploidy level and fruit characters of the main kiwifruit cultivars in China: Implication for selection and improvement. N. Z. J. Crop Hort. 2010, 38, 137–145. [Google Scholar] [CrossRef]
- Latocha, P.; Krupa, T.; Wołosiak, R.; Worobiej, E.; Wilczak, J. Antioxidant activity and chemical difference in fruit of different Actinidiasp. Int. J. Food Sci. Nutr. 2010, 61, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Boland, M. Kiwifruit proteins and enzymes: Actinidin and other significant proteins. Adv. Food Nutr. Res. 2013, 68, 59–80. [Google Scholar] [CrossRef]
- Mizéhoun-Adissoda, C.; Houinato, D.; Houehanou, C.; Chianea, T.; Dalmay, F.; Bigot, A.; Aboyans, V.; Preux, P.-M.; Bovet, P.; Desport, J.-C. Dietary sodium and potassium intakes: Data from urban and rural areas. Nutrition 2017, 33, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Czech, A.; Zarycka, E.; Yanovych, D.; Zasadna, Z.; Grzegorczyk, I.; Kłys, S. Mineral content of the pulp and peel of various citrus fruit cultivars. Biol. Trace Elem. Res. 2019, 193, 555–563. [Google Scholar] [CrossRef] [PubMed]
- De Souza, V.R.; Pereira, P.A.P.; Da Silva, T.L.T.; De Oliveira Lima, L.C.; Pio, R.; Queiroz, F. Determination of the bioactive compounds, antioxidant activity and chemical composition of Brazilian blackberry, red raspberry, strawberry, blueberry and sweet cherry fruits. Food Chem. 2014, 156, 362–368. [Google Scholar] [CrossRef]
- Delchier, N.; Herbig, A.-L.; Rychlik, M.; Renard, C.M.G.C. Folates in fruits and vegetables: Contents, processing, and stability. Compr. Rev. Food Sci. Food Saf. 2016, 15, 506–528. [Google Scholar] [CrossRef]
- Hanamura, T.; Uchida, E.; Aoki, H. Changes of the composition in acerola (Malpighia emarginata DC.) fruit in relation to cultivar, growing region and maturity. J. Sci. Food Agric. 2008, 88, 1813–1820. [Google Scholar] [CrossRef]






| Cultivar | Abbr. | Species | Flesh Color | Region |
|---|---|---|---|---|
| Cuixiang | CX | A. deliciosa | Green | Shaanxi, China |
| Hayward | HW | A. deliciosa | Green | Shaanxi, China |
| Xuxiang | XX | A. deliciosa | Green | Shaanxi, China |
| Guichang | GC | A. deliciosa | Green | Guizhou, China |
| Ruiyu | RY | A. deliciosa | Green | Shaanxi, China |
| Jinhong No.1 | JH1 | A. chinensis | Red * | Jiangsu, China |
| Hongshi No.2 | HS2 | A. chinensis | Red * | Sichuan, China |
| Hongyang | HYA | A. chinensis | Red * | Shaanxi, China |
| Jinhong 50 | JH50 | A. chinensis | Red * | Sichuan, China |
| Oriental Red | OR | A. chinensis | Red * | Shaanxi, China |
| Huayou | HYO | A. chinensis | Yellow | Shaanxi, China |
| Sungold | SG | A. chinensis | Yellow | New zealand |
| Jinyan | JY | A. chinensis | Yellow | Henan, China |
| Jintao | JT | A. chinensis | Yellow | Henan, China |
| Puyu | PY | A. chinensis | Yellow | Shaanxi, China |
| Cultivar | AAC (mg/100 g) | TPC (mg GAE/100 g) | TFC (mg CTE/100 g) | TAC (mg CGE/100 g) |
|---|---|---|---|---|
| Cuixiang | 145.25 ± 0.75 h | 131.03 ± 2.84 e | 20.39 ± 3.55 de | - |
| Hayward | 92.75 ± 1.75 l | 78.04 ± 3.27 h | 10.25 ± 2.13 f | - |
| Xuxiang | 105.42 ± 0.63 k | 85.49 ± 2.55 g | 12.56 ± 3.51 ef | - |
| Guichang | 165.27 ± 3.25 e | 133.94 ± 3.52 e | 5.69 ± 2.68 f | - |
| Ruiyu | 124.49 ± 0.89 j | 110.57 ± 3.46 f | 12.00 ± 1.47 ef | - |
| Jinhong No.1 | 52.39 ± 1.88 n | 80.09 ± 1.91 h | 22.19 ± 3.29 d | 0.52 ± 0.07 c |
| Hongshi No.2 | 248.16 ± 2.67 a | 216.37 ± 0.73 a | 50.89 ± 3.02 a | 3.65 ± 0.27 a |
| Hongyang | 154.57 ± 1.72 g | 129.75 ± 1.59 e | 40.69 ± 3.01 b | 1.02 ± 0.02 b |
| Jinhong 50 | 161.04 ± 1.70 f | 142.89 ± 3.78 c | 31.64 ± 3.69 c | 0.72 ± 0.00 c |
| Oriental Red | 181.07 ± 2.10 d | 144.53 ± 0.77 c | 23.72 ± 5.71 d | 1.58 ± 0.13 b |
| Huayou | 187.33 ± 1.88 c | 146.46 ± 2.43 c | 23.58 ± 1.44 d | - |
| Sungold | 137.42 ± 1.38 i | 113.09 ± 3.22 f | 16.33 ± 3.06 e | - |
| Jinyan | 73.83 ± 1.28 m | 74.06 ± 0.97 h | 15.17 ± 1.12 ef | - |
| Jintao | 151.74 ± 1.61 g | 141.7 ± 2.19 d | 14.69 ± 4.52 ef | - |
| Puyu | 233.99 ± 1.61 b | 158.66 ± 2.35 b | 19.61 ± 3.26 d | - |
| Cultivar | DPPH(AAC) (µmol/g) | DPPH(TPC) (µmol/g) | FRAP(AAC) (µmol/g) | FRAP(TPC) (µmol/g) |
|---|---|---|---|---|
| Cuixiang | 3.56 ± 0.3 b | 7.88 ± 0.37 c | 16.85 ± 0.6 b | 9.14 ± 0.46 f |
| Hayward | 2.01 ± 0.09 d | 4.87 ± 0.62 f | 5.73 ± 0.29 f | 7.08 ± 0.91 h |
| Xuxiang | 1.61 ± 0.29 de | 5.37 ± 0.44 ef | 8.12 ± 0.59 ef | 7.62 ± 0.27 g |
| Guichang | 1.29 ± 0.34 e | 5.64 ± 0.16 e | 11.36 ± 1.49 d | 9.7 ± 0.67 ef |
| Ruiyu | 1.7 ± 0.13 de | 4.11 ± 0.24 g | 13.99 ± 1.63 c | 7.8 ± 0.16 gh |
| Jinhong No.1 | 1.69 ± 0.06 de | 3.69 ± 0.56 g | 9.96 ± 1.33 de | 8.36 ± 0.12 g |
| Hongshi No.2 | 5.02 ± 1.1 a | 11.96 ± 0.26 a | 8.36 ± 1.1 ef | 17.69 ± 0.36 a |
| Hongyang | 2.34 ± 0.29 cd | 7.69 ± 0.17 c | 7.02 ± 0.38 f | 9.81 ± 0.22 ef |
| Jinhong 50 | 3.51 ± 0.14 b | 8.56 ± 0.48 b | 11.05 ± 1.93 de | 15.04 ± 0.24 c |
| Oriental Red | 3.87 ± 0.03 b | 9.29 ± 0.26 b | 9.7 ± 0.61 de | 17.41 ± 0.25 a |
| Huayou | 4.05 ± 0.08 b | 9.14 ± 0.12 b | 10.54 ± 0.27 de | 16.38 ± 0.09 b |
| Sungold | 2.65 ± 0.26 c | 6.99 ± 0.17 d | 25.92 ± 1.81 a | 11.71 ± 0.1 d |
| Jinyan | 1.72 ± 0.16 de | 4.31 ± 0.13 fg | 9.15 ± 1.83 e | 6.22 ± 0.27 i |
| Jintao | 1.32 ± 0.32 e | 5.86 ± 0.36 e | 12.5 ± 1.92 cd | 10.05 ± 0.71 e |
| Puyu | 3.64 ± 0.16 b | 8.62 ± 0.22 b | 6.44 ± 0.19 f | 17.66 ± 0.69 a |
| Cultivar | Asp | Thr | Ser | Glu | Gly | Ala | Val | Met | Ile | Leu | Tyr | Phe | Lys | His | Arg | Pro |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cuixiang | 57.57 ± 5.41 g | 62.64 ± 5.63 d | 46.15 ± 0.7 fg | 132.85 ± 2.73 g | 37.32 ± 0.07 e | 48.06 ± 0.87 g | 41.31 ± 0.36 ef | 4.38 ± 0.66 b | 34.81 ± 0.87 ef | 46.66 ± 1.04 e | 20.39 ± 1.12 ef | 28.24 ± 1.24 e | 58.62 ± 0.71 f | 17.41 ± 1.99 e | 43.39 ± 2.23 n | 13.6 ± 4.99 b |
| Hayward | 94.67 ± 2.94 cd | 57.65 ± 1.53 d | 39.25 ± 1.16 h | 155.47 ± 8.97 e | 22.41 ± 1.1 g | 49.28 ± 1.5 g | 24.55 ± 0.98 h | 1.21 ± 0.13 c | 19.31 ± 0.29 h | 27.06 ± 0.12 g | 13.14 ± 1.28 f | 17.86 ± 1.02 f | 34.36 ± 0.39 g | 14.99 ± 0.21 e | 117.37 ± 1.85 j | 7.91 ± 0.61 c |
| Xuxiang | 74.37 ± 3.53 ef | 65.32 ± 3.32 d | 43.72 ± 0.7 g | 128.53 ± 1.04 g | 46.44 ± 0.17 d | 53.49 ± 0.04 fg | 42.41 ± 0.16 ef | 3.54 ± 0.66 b | 39.98 ± 0.17 d | 51.37 ± 0.99 d | 30.41 ± 0.72 d | 34.9 ± 1.75 d | 60.12 ± 3.88 ef | 16.83 ± 3.5 de | 55.74 ± 2 m | 9.24 ± 0.36 c |
| Guichang | 79.07 ± 10.76 e | 82.48 ± 6.42 c | 53.27 ± 0.56 e | 157.81 ± 3.32 de | 46.33 ± 2.12 d | 66.63 ± 2.8 de | 43.55 ± 0.72 e | 5.73 ± 0.7 3 ab | 40.75 ± 0.58 d | 48.59 ± 0.41 d | 29.11 ± 2.88 d | 31.54 ± 3.14 de | 61.22 ± 4.26 ef | 19.64 ± 3.36 de | 111.49 ± 3.86 k | 6.4 ± 1.42 c |
| Ruiyu | 90.01 ± 1.65 d | 82.1 ± 0.63 c | 59.05 ± 0.37 d | 146.36 ± 2.47 f | 55.53 ± 0.66 b | 70.14 ± 0.67 d | 52.91 ± 0.21 bc | 4.85 ± 0.26 b | 49.65 ± 0.29 b | 60.6 ± 0.23 c | 35.33 ± 1.28 c | 39.75 ± 0.88 c | 77.81 ± 1.62 c | 21.63 ± 1.1 de | 90.97 ± 2.08 l | 9.42 ± 0.31 c |
| Jinhong NO.1 | 86.02 ± 0.94 de | 104.06 ± 18 ab | 70.91 ± 2.68 b | 171.28 ± 2.52 c | 54.12 ± 3.43 b | 81.42 ± 4.33 c | 54.78 ± 2.43 b | 6.15 ± 0.92 ab | 47.6 ± 2.67 bc | 63.8 ± 2.81 b | 28.09 ± 3.68 d | 30.25 ± 1.42 e | 75.93 ± 2.58 c | 82.69 ± 5.56 a | 152.97 ± 2.01 f | 13.74 ± 4.92 b |
| Hongshi NO.2 | 68.05 ± 0.47 f | 89.85 ± 0 bc | 62.53 ± 1.58 c | 161.9 ± 4.55 d | 45.44 ± 2.66 d | 65.21 ± 4.57 e | 50.17 ± 3.47 c | 5.97 ± 0.92 ab | 41.86 ± 2.26 d | 63.67 ± 2.96 b | 29.9 ± 2.56 d | 43.62 ± 3.14 bc | 84.61 ± 4.33 b | 52.87 ± 2.46 b | 252.92 ± 2.7 d | 8.06 ± 3.92 c |
| Hongyang | 62.64 ± 0.47 fg | 75.37 ± 0.47 c | 47 ± 0.79 f | 237.24 ± 3.58 a | 32.29 ± 0.46 f | 55.74 ± 1.1 f | 37.76 ± 1.76 f | 5.87 ± 2.24 ab | 29.64 ± 3.65 f | 46.21 ± 2.96 e | 16.87 ± 1.28 f | 29.79 ± 2.7 e | 60.17 ± 1.1 ef | 52.72 ± 9.67 b | 260.37 ± 1.77 c | 9.06 ± 2.34 c |
| Jinhong 50 | 97.99 ± 3.41 c | 59.66 ± 3.11 d | 55.24 ± 1.3 e | 165.81 ± 2.15 cd | 50.39 ± 0.3 c | 79.27 ± 2.48 c | 47.68 ± 0.26 d | 5.73 ± 0.86 ab | 41.98 ± 0.35 d | 59.2 ± 0.58 c | 28.77 ± 1.28 d | 40.78 ± 0.58 c | 70.08 ± 2.33 d | 22.41 ± 0.69 de | 184.54 ± 0.15 e | 41.8 ± 1.83 a |
| Oriental Red | 97.25 ± 4.71 cd | 64.17 ± 0 d | 47.98 ± 4.51 f | 128.53 ± 1.14 g | 37.5 ± 1.93 e | 65.07 ± 5.71 e | 39.89 ± 3.31 f | 4.57 ± 0.13 b | 32.23 ± 2.32 f | 49.45 ± 3.59 d | 20.89 ± 1.68 e | 33.25 ± 1.9 de | 63.96 ± 3.75 e | 24.06 ± 1.78 d | 147.74 ± 1.32 g | 10.32 ± 0.76 bc |
| Huayou | 88.68 ± 0.24 d | 74.92 ± 4.58 c | 57.08 ± 2.88 de | 148.85 ± 2.67 f | 55.15 ± 2.45 b | 68.94 ± 1.65 de | 49.66 ± 2.23 cd | 5.27 ± 0.59 ab | 46 ± 2.55 c | 61.3 ± 4.12 c | 31.43 ± 3.12 d | 40.58 ± 3.65 c | 77.58 ± 6.2 c | 21.92 ± 1.23 de | 139.63 ± 3.47 h | 10.25 ± 1.27 bc |
| Sungold | 58.9 ± 5.88 g | 56.31 ± 0.37 d | 40.5 ± 1.35 h | 149.4 ± 1.82 f | 30.04 ± 0.73 f | 61.98 ± 0.24 e | 32.53 ± 0.05 g | 4.62 ± 0.07 b | 27.63 ± 0.81 g | 37.31 ± 1.39 f | 21.29 ± 2.72 e | 27 ± 2.56 e | 26.18 ± 2.01 h | 16.98 ± 0.14 e | 219.55 ± 0.85 d | 10.86 ± 1.22 bc |
| Jinyan | 58.11 ± 4.06 g | 57.54 ± 1.68 d | 38.56 ± 1.39 h | 109.82 ± 3.06 f | 38.35 ± 1.19 e | 49.81 ± 0.43 g | 37.36 ± 0.98 f | 3.82 ± 0.53 b | 36.49 ± 0.7 e | 44.65 ± 0.99 e | 29.05 ± 1.04 d | 30.3 ± 0.51 e | 56.24 ± 1.74 f | 19.06 ± 4.6 de | 133.86 ± 1 i | 5.9 ± 1.73 c |
| Jintao | 125.45 ± 1.88 a | 111.58 ± 1.68 a | 77.97 ± 0.74 a | 178.27 ± 2.14 b | 68.46 ± 1.23 a | 111.6 ± 0 a | 65.72 ± 0.93 a | 6.43 ± 0.26 a | 60.48 ± 1.1 a | 77.33 ± 2.2 a | 45.53 ± 0.16 b | 54.31 ± 2.63 a | 103.89 ± 3.49 a | 34.44 ± 3.7 c | 318.57 ± 2.77 a | 9.03 ± 0.05 c |
| Puyu | 118.21 ± 6 a | 98.26 ± 0.32 b | 65.39 ± 0.7 c | 175.14 ± 0.52 bc | 54.66 ± 0.63 b | 98.68 ± 0.47 b | 51.19 ± 0.16 c | 6.62 ± 0.53 a | 45.72 ± 1.8 c | 66.79 ± 2.03 b | 60.87 ± 4.53 a | 45.28 ± 2.7 b | 87.81 ± 2.84 b | 34.44 ± 0.82 c | 268.21 ± 1 b | 8.16 ± 0.76 c |
| Cultivar | Ca (mg/100 g) | Fe (mg/100 g) | K (mg/100 g) | Mg (mg/100 g) | Na (mg/100 g) | P (mg/100 g) | Zn (mg/100 g) | Cu (mg/100 g) |
|---|---|---|---|---|---|---|---|---|
| Cuixiang | 22.37 ± 2.44 cd | 4.27 ± 0.05 cd | 300 ± 8.33 h | 29.61 ± 0.08 g | 6.95 ± 0.1 bc | 18.71 ± 0.04 cd | 0.3 ± 0.14 ab | 1.3 ± 0.13 a |
| Hayward | 19.42 ± 0.36 d | 4.8 ± 1.94 cd | 262.5 ± 4.17 j | 29.55 ± 0.3 g | 8.71 ± 1.37 b | 10.5 ± 0.42 e | 0.36 ± 0.02 ab | 0.18 ± 0.02 g |
| Xuxiang | 23.74 ± 3 c | 3.83 ± 0.2 d | 320.83 ± 12.5 g | 33.39 ± 1.16 d | 7.33 ± 2.48 bc | 21.75 ± 0 c | 0.4 ± 0.19 ab | 0.98 ± 0.01 b |
| Guichang | 14.95 ± 0.65 e | 6.05 ± 1.6 c | 358.33 ± 0 f | 31.36 ± 0.02 ef | 5.62 ± 0.18 c | 18.54 ± 1.13 cd | 0.2 ± 0.07 ab | 0.36 ± 0.01 f |
| Ruiyu | 15.93 ± 0.46 e | 3.27 ± 0.02 d | 445.83 ± 12.5 c | 36.74 ± 0.8 b | 5.29 ± 0.16 c | 34.83 ± 1 a | 0.2 ± 0.1 b | 0.47 ± 0.03 e |
| Jinhong No.1 | 8.74 ± 0.73 f | 3.48 ± 0.06 d | 475 ± 8.33 b | 36.21 ± 0.14 bc | 6.64 ± 0.12 bc | 38.29 ± 2.13 a | 0.24 ± 0.1 b | 0.45 ± 0 ef |
| Hongshi No.2 | 9.18 ± 0.48 f | 5.78 ± 0.6 c | 450 ± 0 c | 36.6 ± 0.31 b | 11.22 ± 2.86 a | 23.67 ± 0.25 bc | 0.35 ± 0.19 a | 0.48 ± 0.05 e |
| Hongyang | 11.31 ± 0.22 f | 4.19 ± 0.42 d | 320.83 ± 4.17 g | 32.74 ± 0.07 de | 6.87 ± 1.46 bc | 19.63 ± 4.79 cd | 0.26 ± 0.02 a | 0.18 ± 0.01 g |
| Jinhong 50 | 27.02 ± 1.17 b | 13.75 ± 0.88 a | 212.5 ± 4.17 l | 30.87 ± 0.03 f | 6.58 ± 0.66 bc | 21.79 ± 0.79 c | 0.22 ± 0.04 b | 0.23 ± 0.01 g |
| Oriental Red | 31.27 ± 2.65 a | 4.53 ± 0.16 cd | 325 ± 0 g | 32.1 ± 0.15 e | 5.62 ± 0.58 c | 25.42 ± 5 b | 0.27 ± 0.06 ab | 0.25 ± 0.02 g |
| Huayou | 19.86 ± 1.68 d | 10.87 ± 2.41 b | 425 ± 8.33 d | 31.46 ± 0.16 ef | 8.03 ± 2.05 b | 24.83 ± 1 bc | 0.43 ± 0.2 a | 0.19 ± 0.04 g |
| Sungold | 15.22 ± 1.64 e | 2.71 ± 0.03 d | 241.67 ± 0 k | 30.63 ± 0.07 f | 4.73 ± 0.97 c | 13.54 ± 0.71 e | 0.25 ± 0.09 ab | 0.72 ± 0.07 c |
| Jinyan | 21.26 ± 2.5 cd | 13.12 ± 2.17 a | 275 ± 0 i | 31.2 ± 0.1 ef | 4.81 ± 0.12 c | 12.08 ± 2.67 e | 0.31 ± 0.07 ab | 0.78 ± 0.05 c |
| Jintao | 24.04 ± 1.42 c | 4.23 ± 0.14 cd | 395.83 ± 4.17 e | 35.44 ± 0.57 c | 4.68 ± 0.97 c | 17.46 ± 0.96 d | 0.21 ± 0.02 b | 0.63 ± 0.05 d |
| Puyu | 17.33 ± 2.56 de | 6.07 ± 0.52 c | 516.67 ± 8.33 a | 60.4 ± 1.36 a | 5.01 ± 0.5 c | 24.17 ± 0.67 bc | 0.26 ± 0.04 a | 0.92 ± 0.11 b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Zhao, Q.; Lan, T.; Geng, T.; Gao, C.; Yuan, Q.; Zhang, Q.; Xu, P.; Sun, X.; Liu, X.; et al. Comparative Analysis of Physicochemical Characteristics, Nutritional and Functional Components and Antioxidant Capacity of Fifteen Kiwifruit (Actinidia) Cultivars—Comparative Analysis of Fifteen Kiwifruit (Actinidia) Cultivars. Foods 2020, 9, 1267. https://doi.org/10.3390/foods9091267
Zhang H, Zhao Q, Lan T, Geng T, Gao C, Yuan Q, Zhang Q, Xu P, Sun X, Liu X, et al. Comparative Analysis of Physicochemical Characteristics, Nutritional and Functional Components and Antioxidant Capacity of Fifteen Kiwifruit (Actinidia) Cultivars—Comparative Analysis of Fifteen Kiwifruit (Actinidia) Cultivars. Foods. 2020; 9(9):1267. https://doi.org/10.3390/foods9091267
Chicago/Turabian StyleZhang, Hexin, Qinyu Zhao, Tian Lan, Tonghui Geng, Chenxu Gao, Quyu Yuan, Qianwen Zhang, Pingkang Xu, Xiangyu Sun, Xuebo Liu, and et al. 2020. "Comparative Analysis of Physicochemical Characteristics, Nutritional and Functional Components and Antioxidant Capacity of Fifteen Kiwifruit (Actinidia) Cultivars—Comparative Analysis of Fifteen Kiwifruit (Actinidia) Cultivars" Foods 9, no. 9: 1267. https://doi.org/10.3390/foods9091267
APA StyleZhang, H., Zhao, Q., Lan, T., Geng, T., Gao, C., Yuan, Q., Zhang, Q., Xu, P., Sun, X., Liu, X., & Ma, T. (2020). Comparative Analysis of Physicochemical Characteristics, Nutritional and Functional Components and Antioxidant Capacity of Fifteen Kiwifruit (Actinidia) Cultivars—Comparative Analysis of Fifteen Kiwifruit (Actinidia) Cultivars. Foods, 9(9), 1267. https://doi.org/10.3390/foods9091267



.jpg)