In Vivo Evaluation of the Oral Toxicity of the Chlorobutanol
Abstract
:1. Introduction
2. Materials and Methods
2.1. Test Substance and Vehicle
2.2. Test Animals
2.3. Single-Dose Toxicity Study Design
2.4. Repeated-Dose 28-Day Oral Toxicity Design
2.5. Hematology and Clinical Biochemistry
2.6. Gross Necropsy
2.7. Histopathology
2.8. Statistical Analysis
3. Results
3.1. The Single-Dose Toxicity Study
3.2. The 28-Day Repeated Oral Toxicity Study
3.2.1. Clinical Signs, Necropsy Findings, and Food and Water Consumption
3.2.2. Relative Organ Weights
3.2.3. Hematological Changes
3.2.4. Biochemical Changes
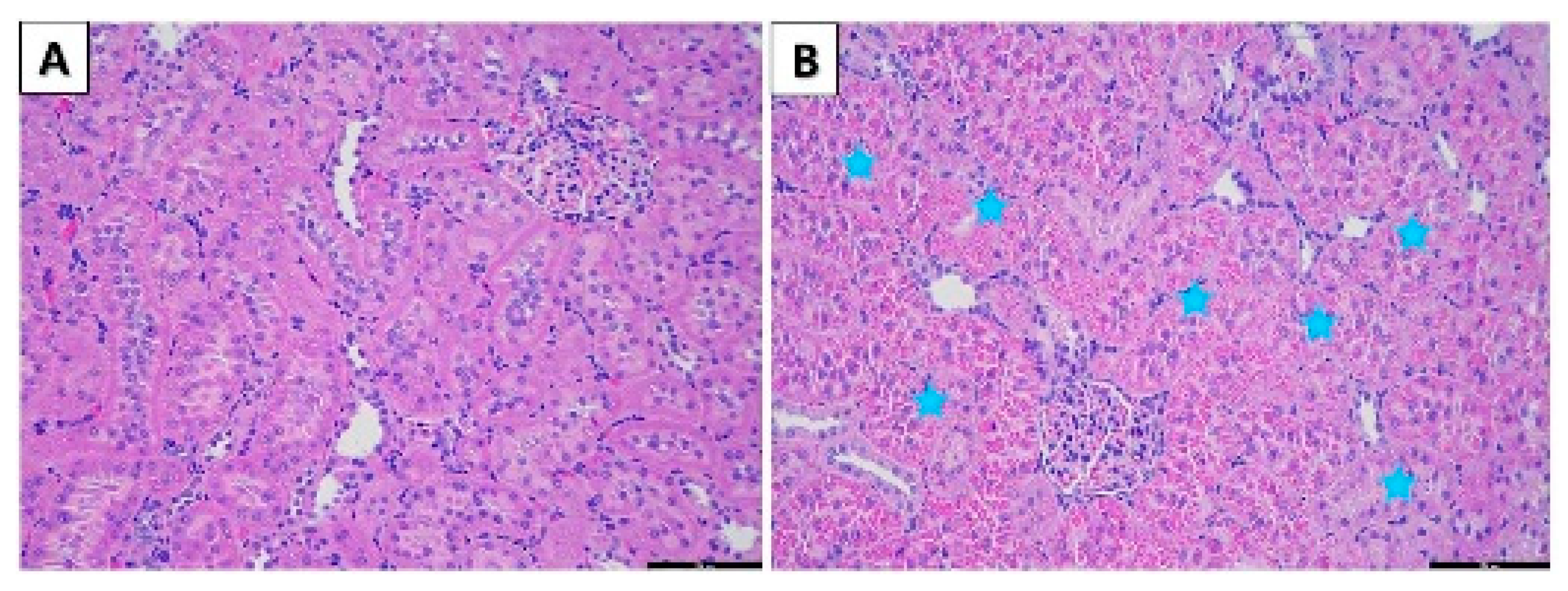
3.2.5. Histopathological Examination
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kreiner, C. Biochemical Aspects of Ophthalmic Preservatives. Contacto 1979, 23, 10–14. [Google Scholar]
- Epstein, S.P.; Ahdoot, M.; Marcus, E.; Asbell, P.A. Comparative toxicity of preservatives on immortalized corneal and conjunctival epithelial cells. Journal of ocular pharmacology and therapeutics. J. Ocul. Pharmacol. Ther. 2009, 25, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Ayaki, M.; Yaguchi, S.; Iwasawa, A.; Koide, R. Cytotoxicity of ophthalmic solutions with and without preservatives to human corneal endothelial cells, epithelial cells and conjunctival epithelial cells. Clin. Experiment. Ophthalmol. 2008, 36, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Smoak, I.W. Embryotoxic effects of chlorobutanol in cultured mouse embryos. Teratology 1993, 47, 203–208. [Google Scholar] [CrossRef] [PubMed]
- DailyMed. US National Library of Medicine. 2019. Available online: https://dailymed.nlm.nih.gov/dailymed/search.cfm?adv=1&labeltype=all&pagesize=200&page=1&query=%28chlorobutanol%29+ (accessed on 15 October 2017).
- Brun, P.M.; Querellou, E.; Leyral, J.; Barberis, C.; Levy, D.; Puidupin, A. Intoxication médicamenteuse volontaire à l’Alodont(®): À propos d’un cas [Chlorobutanol poisoning: About a case]. Ann. Fr. Anesth. Reanim. 2010, 29, 741–742. [Google Scholar] [CrossRef] [PubMed]
- Halla, N.; Fernandes, I.P.; Heleno, S.A.; Costa, P.; Boucherit-Otmani, Z.; Boucherit, K.; Rodrigues, A.E.; Ferreira, I.; Barreiro, M.F. Cosmetics Preservation: A Review on Present Strategies. Molecules 2018, 23, 1571. [Google Scholar] [CrossRef] [Green Version]
- Gupta, S.; Kaisheva, E. Development of a Multidose Formulation for a Humanized Monoclonal Antibody Using Experimental Design Techniques. AAPS PharmSci 2003, 5, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Nair, A.D.; Lach, J.L. The kinetics of degradation of chlorobutanol. J. Am. Pharm. Assoc. 1959, 48, 390–395. [Google Scholar] [CrossRef] [PubMed]
- Hagen, N.; Bizimana, T.; Kayumba, P.C.; Khuluza, F.; Heide, L. Stability of Oxytocin Preparations in Malawi and Rwanda: Stabilizing Effect of Chlorobutanol. Am. J. Trop. Med. 2020, 103, 2129–2141. [Google Scholar] [CrossRef]
- Tripathi, B.J.; Tripathi, R.C. Cytotoxic effects of benzalkonium chloride and chlorobutanol on human corneal epithelial cells in vitro. Lens Eye Toxic. Res. 1989, 6, 395–403. [Google Scholar]
- Fassihi, A.R.; Naidoo, N.T. Irritation associated with tear-replacement ophthalmic drops. A pharmaceutical and subjective investigation. S. Afr. Med. J. 1989, 75, 233–235. [Google Scholar] [PubMed]
- Tomlinson, A.; Trees, G.R. Effect of preservatives in artificial tear solutions on tear film evaporation. Ophthalmic Physiol. Opt. 1991, 11, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Valentour, J.C.; Sunshine, I. Chlorobutanol poisoning. Report of a fatal case. Z. Rechtsmed. 1975, 77, 61–63. [Google Scholar] [CrossRef] [PubMed]
- Nordt, S.P. Chlorobutanol toxicity. Ann. Pharmacother. 1996, 30, 1179–1180. [Google Scholar] [PubMed]
- Gokulan, K.; Kumar, A.; Lahiani, M.H.; Sutherland, V.L.; Cerniglia, C.E.; Khare, S. Differential Toxicological Outcome of Corn Oil Exposure in Rats and Mice as Assessed by Microbial Composition, Epithelial Permeability, and Ileal Mucosa-Associated Immune Status. Toxicol. Sci. 2021, 180, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Organisation for Economic Cooperation and Development. OECD Guideline for the Testing of Chemicals: Acute Oral Toxicity-Acute Toxic Class Method. Test Guideline No. 423. Available online: https://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=env/jm/mono(2001)4&doclanguage=en (accessed on 17 December 2001).
- The Ministry of Food and Drug Safety. Toxicity Test Standards of Medicine and Medical Supplies; The Ministry of Food and Drug Safety: Osong, Korea, 2017.
- Organisation for Economic Cooperation and Development. OECD Guideline for the Testing of Chemicals: Repeated Dose 28-Day Oral Toxicity Study in Rodents; Test Guideline No. 407 (Adopted on 3 October 2008); OECD Publishing: Paris, France, 2008. [Google Scholar] [CrossRef]
- The Ministry of Food and Drug Safety. Guideline for Risk Assessment of Cosmetic Products; The Ministry of Food and Drug Safety: Osong, Korea, 2017.
- National Center for Biotechnology Information PubChem Compound Summary for CID 7132, Lauryl Isoquinolinium Bromide. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Lauryl-isoquinolinium-bromide (accessed on 14 May 2021).
- Read, N.G. The role of lysosomes in hyaline droplet nephropathy induced by a variety of pharmacological agents in the male rat. Histochem. J. 1991, 23, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shi, S.; Zhao, X.J.; Wang, J.K.; Liu, Z.W.; Liu, F.Q.; Pan, S. Association between the lipid profile and renal dysfunction in the heart failure patients. Kidney Blood Press. Res. 2019, 44, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Asahina, M.; Shimizu, F.; Ohta, M.; Takeyama, M.; Tozawa, R. Human apolipoprotein B transgenic SHR/NDmcr-cp rats show exacerbated kidney dysfunction. Exp. Anim. 2015, 64, 313–321. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.H.; Lee, H.S.; Cho, A.R.; Lee, Y.J.; Kwon, Y.J. Non-Alcoholic Fatty Liver Disease Is an Independent Risk Factor for LDL Cholesterol Target Level. Int. J. Environ. Res. Public Health 2021, 18, 3442. [Google Scholar] [CrossRef]
- Kim, S.; Qualls, C.W., Jr.; Reddy, G.; Stair, E.L. 1,3,5-Trinitrobenzene-induced alpha-2u-globulin nephropathy. Toxicol. Pathol. 1997, 25, 195–201. [Google Scholar] [CrossRef] [Green Version]
- Kanerva, R.L.; Ridder, G.M.; Stone, L.C.; Alden, C.L. Characterization of spontaneous and decalin-induced hyaline droplets in kidneys of adult male rats. Food Chem. Toxicol. 1987, 25, 63–82. [Google Scholar]
- Fine, L. The biology of renal hypertrophy. Kidney Int. 1986, 29, 619–634. [Google Scholar] [CrossRef] [Green Version]
- Zimmer, D.P.; Shea, C.M.; Tobin, J.V.; Tchernychev, B.; Germano, P.; Sykes, K.; Masferrer, J.L. Olinciguat, an oral sGC stimulator, exhibits diverse pharmacology across preclinical models of cardiovascular, metabolic, renal, and inflammatory disease. Front. Pharmacol. 2020, 11, 419. [Google Scholar] [CrossRef]
- Brott, D.A.; Bentley, P.; Nadella, M.V.; Thurman, D.; Fikes, J.; Cheatham, L.; Kinter, L.B. Renal biomarker changes associated with hyaline droplet nephropathy in rats are time and potentially compound dependent. Toxicology 2013, 303, 133–138. [Google Scholar] [CrossRef]
- Hard, G.C.; Rodgers, I.S.; Baetcke, K.P.; Richards, W.L.; McGaughy, R.E.; Valcovic, L.R. Hazard evaluation of chemicals that cause accumulation of alpha 2u-globulin, hyaline droplet nephropathy, and tubule neoplasia in the kidneys. Environ. Health Perspect. 1993, 99, 313–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, A.K.; Nath, T.S.; Motwani, N.M.; Chatterjee, B. Age-dependent regulation of the polymorphic forms of a2u-globulin. J. Biol. Chem. 1983, 258, 10123–10127. [Google Scholar] [CrossRef]
- Williams, K.D.; Dunnick, J.; Horton, J.; Greenwell, A.; Eldridge, S.R.; Elwell, M.; Sills, R.C. P-Nitrobenzoic acid alpha2u nephropathy in 13-week studies is not associated with renal carcinogenesis in 2-year feed studies. Toxicol. Pathol. 2001, 29, 507–513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borghoff, S.J.; Miller, A.B.; Bowen, J.P.; Swenberg, J.A. Characteristics of chemical binding to alpha 2u-globulin in vitro—Evaluating structure-activity relationships. Toxicol. Appl. Pharmacol. 1991, 107, 228–238. [Google Scholar] [CrossRef]
- Dietrich, D.R.; Swenberg, J.A. The presence of alpha 2u-globulin is necessary for d-limonene promotion of male rat kidney tumors. Cancer Res. 1991, 51, 3512–3521. [Google Scholar]
- Saillenfait, A.M.; Marquet, F.; Sabaté, J.P.; Ndiaye, D.; Lambert-Xolin, A.M. 4-Week repeated dose oral toxicity study of N-ethyl-2-pyrrolidone in Sprague Dawley rats. Regul. Toxicol. Pharmacol. 2016, 81, 275–283. [Google Scholar] [CrossRef]
- Baetcke, K.P.; Hard, G.C.; Rodgers, I.S.; McGaughy, R.E.; Tahan, L.M. a2u- Globulin: Association with chemically induced renal toxicity and neoplasia in the male rat. In US Environmental Protection Agency, Risk Assessment Forum; Washington, DC, USA, 1991; Volume 20460. Available online: https://hero.epa.gov/hero/index.cfm/reference/details/reference_id/635839 (accessed on 15 November 2021).

| Body Weight (g) | ||||||
|---|---|---|---|---|---|---|
| Sex | Dose (mg/kg) | Number Dosed | Day 0 | Day 7 | Day 14 | Mortality (Dead/Total) |
| Male | 0 | 5 | 176.34 ± 5.06 | 229.41 ± 6.94 | 275.50 ± 6.05 | 0% (0/5) |
| 62.5 | 5 | 176.02 ± 2.66 | 221.15 ± 8.16 | 272.97 ± 11.83 | 0% (0/5) | |
| 125 | 5 | 176.49 ± 2.51 | 220.38 ± 4.23 | 274.30 ± 5.24 | 0% (0/5) | |
| 250 | 5 | 176.95 ± 4.92 | 203.08 ± 9.15 | 266.49 ± 10.31 | 0% (0/5) | |
| Female | 0 | 5 | 131.99 ± 4.83 | 157.25 ± 7.37 | 180.22 ± 9.81 | 0% (0/5) |
| 62.5 | 5 | 131.76 ± 3.17 | 160.48 ± 5.99 | 180.22 ± 9.11 | 0% (0/5) | |
| 125 | 5 | 131.66 ± 3.14 | 159.92 ± 9.75 | 182.97 ± 10.46 | 0% (0/5) | |
| 250 | 5 | 131.57 ± 3.76 | 145.31 ± 4.56 | 171.35 ± 6.38 | 20% (1/5) | |
| Body Weight (g) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sex | Dose (mg/kg) | Number Dosed | Day 0 | Day 7 | Day 14 | Day 21 | Day 28 | Mortality (Dead/Total) |
| Male | 0 | 5 | 175.5 ± 5.8 | 224.9 ± 8.3 | 269.7 ± 13.1 | 305.3 ± 16.4 | 330.6 ± 16.4 | 0% (0/5) |
| 12.5 | 7 | 175.5 ± 7.5 | 225.9 ± 9.7 | 270.1 ± 12.9 | 302.1 ± 13.2 | 328.8 ± 12.1 | 0% (0/5) | |
| 25 | 7 | 175.8 ± 6.0 | 227.3 ± 9.1 | 270.7 ± 10.5 | 302.5 ± 17.4 | 325.7 ± 20.6 | 0% (0/7) | |
| 50 | 7 | 175.6 ± 4.7 | 226.4 ± 6.4 | 268.2 ± 11.4 | 302.0 ± 13.5 | 327.0 ± 14.6 | 0% (0/7) | |
| 100 | 7 | 176.3 ± 4.0 | 221.9 ± 9.2 | 263.1 ± 14.3 | 296.7 ± 24.1 | 324.1 ± 28.9 | 0% (0/7) | |
| Female | 0 | 5 | 137.1 ± 4.2 | 167.0 ± 4.0 | 185.7 ± 5.9 | 199.3 ± 8.0 | 209.9 ± 10.4 | 0% (0/5) |
| 12.5 | 7 | 137.7 ± 4.4 | 167.8 ± 8.5 | 186.5 ± 17.4 | 203.4 ± 21.4 | 210.3 ± 20.2 | 0% (0/5) | |
| 25 | 7 | 138.0 ± 4.9 | 170.3 ± 11.8 | 189.0 ± 14.8 | 205.0 ± 17.1 | 213.5 ± 18.3 | 0% (0/7) | |
| 50 | 7 | 137.4 ± 4.6 | 171.0 ± 9.3 | 187.2 ± 11.9 | 199.6 ± 12.9 | 210.2 ± 15.1 | 0% (0/7) | |
| 100 | 7 | 138.8 ± 5.7 | 172.4 ± 9.9 | 195.8 ± 11.6 | 212.8 ± 7.7 | 202.8 ± 28.1 | 14% (1/7) | |
| Feed Intake (g) | ||||||
|---|---|---|---|---|---|---|
| Sex | Dose (mg/kg) | Number Dosed | 1st Week | 2nd Week | 3rd Week | 4th Week |
| Male | 0 | 5 | 18.3 ± 2.1 | 17.1 ± 2.5 | 18.2 ± 2.1 | 15.9 ± 3.1 |
| 12.5 | 7 | 19.0 ± 0.7 | 17.8 ± 0.2 | 17.1 ± 1.6 | 16.4 ± 1.5 | |
| 25 | 7 | 18.2 ± 3.1 | 18.7 ± 1.4 | 17.9 ± 2.0 | 15.3 ± 1.6 | |
| 50 | 7 | 18.9 ± 0.8 | 18.7 ± 1.6 | 17.2 ± 1.3 | 16.8 ± 1.3 | |
| 100 | 7 | 14.6 ± 2.1 * | 22.8 ± 4.5 * | 19.8 ± 2.0 | 17.8 ± 3.4 | |
| Female | 0 | 5 | 12.0 ± 1.0 | 11.4 ± 1.6 | 9.8 ± 1.5 | 10.2 ± 1.1 |
| 12.5 | 7 | 11.7 ± 1.5 | 11.7 ± 1.0 | 11.0 ± 1.7 | 10.1 ± 1.3 | |
| 25 | 7 | 12.4 ± 2.1 | 12.2 ± 0.7 | 11.5 ± 1.5 | 12.2 ± 1.5 * | |
| 50 | 7 | 13.1 ± 0.8 | 11.6 ± 1.1 | 10.8 ± 0.4 | 13.6 ± 0.4 * | |
| 100 | 7 | 10.4 ± 1.7 | 12.8 ± 0.9 * | 12.0 ± 2.2 * | 8.6 ± 2.0 | |
| Water Consumption (g) | ||||||
|---|---|---|---|---|---|---|
| Sex | Dose (mg/kg) | Number Dosed | 1st Week | 2nd Week | 3rd Week | 4th Week |
| Male | 0 | 5 | 33.3 ± 4.9 | 33.9 ± 3.3 | 36.0 ± 4.9 | 30.3 ± 5.7 |
| 12.5 | 7 | 33.2 ± 1.9 | 32.7 ± 3.7 | 36.6 ± 6.2 | 33.2 ± 5.2 | |
| 25 | 7 | 36.9 ± 5.2 | 36.9 ± 5.0 | 36.4 ± 5.1 | 34.8 ± 6.8 | |
| 50 | 7 | 34.6 ± 6.6 | 34.5 ± 5.2 | 32.9 ± 6.8 | 31.0 ± 4.6 | |
| 100 | 7 | 23.3 ± 4.6 * | 37.4 ± 6.3 | 30.6 ± 2.6 | 29.9 ± 4.4 | |
| Female | 0 | 5 | 23.8 ± 1.8 | 23.5 ± 3.4 | 25.9 ± 1.6 | 24.3 ± 1.7 |
| 12.5 | 7 | 24.5 ± 1.1 | 22.4 ± 2.5 | 24.3 ± 2.8 | 24.4 ± 5.6 | |
| 25 | 7 | 26.1 ± 2.3 | 22.4 ± 4.1 | 25.1 ± 8.4 | 25.6 ± 6.9 | |
| 50 | 7 | 25.0 ± 1.6 | 23.2 ± 4.8 | 23.8 ± 2.1 | 23.7 ± 1.9 | |
| 100 | 7 | 28.6 ± 8.2 | 24.6 ± 3.0 | 23.1 ± 2.9 | 15.2 ± 5.6 * | |
| Organ (g/100 g Body Weight) | Groups (mg/kg/day) | ||||
|---|---|---|---|---|---|
| 0 | 12.5 | 25 | 50 | 100 | |
| Male | |||||
| Liver | 2.78 ± 0.07 | 2.86 ± 0.14 | 3.00 ± 0.13 * | 3.09 ± 0.09 * | 3.51 ± 0.15 * |
| Kidney-R | 0.34 ± 0.01 | 0.37 ± 0.09 | 0.35 ± 0.02 | 0.38 ± 0.01 | 0.44 ± 0.04 * |
| Kidney-L | 0.33 ± 0.01 | 0.34 ± 0.02 | 0.35 ± 0.03 | 0.39 ± 0.02 * | 0.43 ± 0.04 * |
| Adrenal gland-R (mg) | 7.73 ± 0.19 | 7.93 ± 0.90 | 7.91 ± 1.04 | 7.68 ± 0.77 | 8.64 ± 1.41 |
| Adrenal gland-L (mg) | 8.00 ± 0.98 | 8.06 ± 1.05 | 7.83 ± 0.85 | 7.68 ± 0.73 | 8.08 ± 1.04 |
| Heart | 0.40 ± 0.02 | 0.39 ± 0.04 | 0.39 ± 0.02 | 0.40 ± 0.02 | 0.43 ± 0.02 |
| Lung | 0.47 ± 0.04 | 0.45 ± 0.02 | 0.48 ± 0.03 | 0.46 ± 0.04 | 0.46 ± 0.03 |
| Brain | 0.57 ± 0.03 | 0.57 ± 0.02 | 0.57 ± 0.03 | 0.57 ± 0.03 | 0.56 ± 0.05 |
| Pituitary gland | 3.16 ± 0.20 | 3.45 ± 0.50 | 3.16 ± 0.25 | 3.55 ± 0.30 | 3.27 ± 0.39 |
| Spleen | 0.20 ± 0.02 | 0.22 ± 0.01 | 0.22 ± 0.02 | 0.23 ± 0.02 * | 0.22 ± 0.02 |
| Thymus | 0.14 ± 0.01 | 0.15 ± 0.02 | 0.16 ± 0.01 | 0.15 ± 0.01 | 0.14 ± 0.02 |
| Testis-R | 0.60 ± 0.04 | 0.59 ± 0.03 | 0.57 ± 0.03 | 0.57 ± 0.05 | 0.57 ± 0.05 |
| Testis-L | 0.59 ± 0.05 | 0.60 ± 0.03 | 0.57 ± 0.03 | 0.56 ± 0.05 | 0.56 ± 0.05 |
| Epididymides-R | 0.15 ± 0.01 | 0.15 ± 0.01 | 0.15 ± 0.01 | 0.14 ± 0.01 | 0.15 ± 0.01 |
| Epididymides-L | 0.15 ± 0.01 | 0.15 ± 0.01 | 0.15 ± 0.01 | 0.14 ± 0.01 | 0.15 ± 0.02 |
| Prostate | 0.11 ± 0.02 | 0.11 ± 0.01 | 0.11 ± 0.03 | 0.10 ± 0.01 | 0.10 ± 0.02 |
| Seminal vesicle | 0.45 ± 0.07 | 0.45 ± 0.04 | 0.43 ± 0.07 | 0.42 ± 0.05 | 0.42 ± 0.04 |
| Salivary gland | 0.19 ± 0.02 | 0.19 ± 0.01 | 0.19 ± 0.01 | 0.19 ± 0.03 | 0.16 ± 0.01 |
| Thyroid gland-R (mg) | 3.02 ± 0.62 | 3.10 ± 0.61 | 2.61 ± 0.60 | 2.99 ± 0.37 | 2.78 ± 0.29 |
| Thyroid gland-L (mg) | 3.30 ± 1.18 | 2.94 ± 0.30 | 2.67 ± 0.42 | 2.64 ± 0.70 | 2.84 ± 0.59 |
| Female | |||||
| Liver | 2.82 ± 0.15 | 2.88 ± 0.21 | 2.93 ± 0.17 | 2.94 ± 0.27 | 4.49 ± 0.99 * |
| Kidney-R | 0.34 ± 0.02 | 0.35 ± 0.02 | 0.36 ± 0.03 | 0.37 ± 0.02 | 0.43 ± 0.10 * |
| Kidney-L | 0.34 ± 0.02 | 0.36 ± 0.03 | 0.34 ± 0.02 | 0.36 ± 0.02 | 0.44 ± 0.12 * |
| Adrenal gland-R (mg) | 14.48 ± 1.44 | 15.26 ± 2.51 | 15.50 ± 1.62 | 16.42 ± 2.64 | 18.35 ± 7.76 |
| Adrenal gland-L (mg) | 15.07 ± 1.97 | 15.07 ± 2.22 | 14.70 ± 2.52 | 16.04 ± 1.67 | 17.39 ± 7.25 |
| Heart | 0.42 ± 0.01 | 0.42 ± 0.03 | 0.41 ± 0.02 | 0.44 ± 0.03 | 0.50 ± 0.04 * |
| Lung | 0.55 ± 0.02 | 0.57 ± 0.03 | 0.56 ± 0.04 | 0.56 ± 0.04 | 0.58 ± 0.06 |
| Brain | 0.82 ± 0.07 | 0.86 ± 0.08 | 0.81 ± 0.07 | 0.82 ± 0.08 | 0.84 ± 0.11 |
| Pituitary gland | 5.30 ± 0.66 | 5.65 ± 1.10 | 5.24 ± 1.07 | 5.53 ± 0.79 | 5.80 ± 0.75 |
| Spleen | 0.28 ± 0.02 | 0.28 ± 0.03 | 0.28 ± 0.02 | 0.27 ± 0.02 | 0.22 ± 0.07 |
| Thymus | 0.16 ± 0.01 | 0.17 ± 0.03 | 0.16 ± 0.02 | 0.16 ± 0.01 | 0.12 ± 0.06 |
| Ovary-R | 21.68 ± 1.89 | 23.06 ± 5.77 | 23.37 ± 4.16 | 23.89 ± 4.23 | 22.11 ± 3.78 |
| Ovary-L | 23.21 ± 2.78 | 22.07 ± 2.56 | 22.79 ± 1.92 | 21.39 ± 2.27 | 22.12 ± 5.20 |
| Uterus | 0.21 ± 0.05 | 0.24 ± 0.06 | 0.34 ± 0.20 | 0.26 ± 0.11 | 0.20 ± 0.08 |
| Salivary gland | 0.20 ± 0.02 | 0.21 ± 0.02 | 0.21 ± 0.02 | 0.22 ± 0.01 | 0.17 ± 0.02 * |
| Thyroid gland-R (mg) | 4.52 ± 1.45 | 4.20 ± 1.17 | 4.77 ± 0.52 | 4.69 ± 1.85 | 5.00 ± 1.40 |
| Thyroid gland-L (mg) | 4.67 ± 0.50 | 4.01 ± 0.76 | 4.20 ± 0.82 | 4.01 ± 1.30 | 4.49 ± 1.87 |
| Groups (mg/kg/day) | |||||
|---|---|---|---|---|---|
| Parameter | 0 | 12.5 | 25 | 50 | 100 |
| Male | |||||
| WBC (×103 cells/μL) | 8.1 ± 2.3 | 6.6 ± 1.8 | 9.1 ± 2.1 | 7.4 ± 2.3 | 8.0 ± 1.5 |
| RBC (×106 cells/μL) | 8.2 ± 0.2 | 8.3 ± 0.4 | 8.2 ± 0.2 | 8.0 ± 0.8 | 8.1 ± 0.3 |
| HGB (g/dL) | 16.2 ± 0.5 | 15.9 ± 0.7 | 15.9 ± 0.4 | 15.2 ± 1.4 | 15.8 ± 0.6 |
| HCT (%) | 48.6 ± 1.4 | 48.7 ± 1.9 | 48.9 ± 1.6 | 47.1 ± 5.2 | 49.3 ± 2.0 |
| MCV (fL) | 59.1 ± 1.1 | 59.0 ± 1.3 | 59.8 ± 1.3 | 58.7 ± 2.5 | 60.7 ± 1.6 |
| MCH (pg) | 19.7 ± 0.5 | 19.2 ± 0.5 | 19.4 ± 0.6 | 19.0 ± 0.7 | 19.5 ± 0.4 |
| MCHC (g/dL) | 33.3 ± 0.3 | 32.6 ± 0.5 | 32.6 ± 0.5 | 32.5 ± 0.8 * | 32.0 ± 0.4 * |
| PLT (×103 cells/μL) | 1083.8 ± 128.1 | 993.0 ± 48.8 | 1001.7 ± 44.8 | 999.4 ± 84.0 | 984.0 ± 106.1 |
| NEUT (% of WBC) | 11.6 ± 3.5 | 11.4 ± 2.1 | 11.6 ± 3.9 | 15.2 ± 2.5 | 15.6 ± 5.5 |
| LYM (% of WBC) | 84.0 ± 3.8 | 85.0 ± 2.1 | 83.4 ± 4.2 | 80.9 ± 2.8 | 80.0 ± 5.5 |
| MONO (% of WBC) | 2.1 ± 0.8 | 1.8 ± 0.4 | 2.4 ± 0.9 | 2.1 ± 0.6 | 2.4 ± 0.3 |
| EOS (% of WBC) | 1.0 ± 0.2 | 0.7 ± 0.4 | 0.7 ± 0.2 | 0.7 ± 0.3 | 0.7 ± 0.1 |
| BASO (% of WBC) | 0.6 ± 0.3 | 0.5 ± 0.2 | 0.7 ± 0.3 | 0.5 ± 0.2 | 0.6 ± 0.1 |
| Retic (%) | 2.7 ± 0.4 | 2.5 ± 0.3 | 2.6 ± 0.3 | 2.6 ± 0.3 | 3.0 ± 0.5 |
| Female | |||||
| WBC (×103 cells/μL) | 5.1 ± 2.1 | 5.1 ± 1.6 | 5.3 ± 1.4 | 4.8 ± 1.2 | 4.7 ± 0.7 |
| RBC (×106 cells/μL) | 6.2 ± 2.8 | 7.6 ± 0.3 | 7.8 ± 0.3 | 7.9 ± 0.2 * | 7.4 ± 0.2 |
| HGB (g/dL) | 14.7 ± 1.1 | 14.5 ± 0.5 | 14.9 ± 0.4 | 15.2 ± 0.8 | 14.3 ± 0.5 |
| HCT (%) | 36.4 ± 16.6 | 43.8 ± 1.5 | 45.1 ± 1.7 | 45.9 ± 1.9 | 43.6 ± 1.6 |
| MCV (fL) | 57.9 ± 1.0 | 57.9 ± 1.2 | 58.1 ± 0.9 | 57.9 ± 1.5 | 59.2 ± 1.4 |
| MCH (pg) | 18.7 ± 2.2 | 19.2 ± 0.3 | 19.2 ± 0.6 | 19.2 ± 0.6 | 19.4 ± 0.4 |
| MCHC (g/dL) | 32.2 ± 3.3 | 33.2 ± 0.6 | 33.0 ± 0.7 | 33.2 ± 0.7 | 32.8 ± 0.3 |
| PLT (×103 cells/μL) | 1039.8 ± 89.8 | 1057.3 ± 149.4 | 1064.6 ± 98.3 | 1111.7 ± 40.4 | 1063.7 ± 75.0 |
| NEUT (% of WBC) | 12.1 ± 5.2 | 12.9 ± 4.8 | 9.5 ± 1.9 | 9.7 ± 1.8 | 15.2 ± 2.8 |
| LYM (% of WBC) | 83.6 ± 4.4 | 84.1 ± 5.0 | 85.4 ± 4.2 | 85.7 ± 2.5 | 75.9 ± 12.4 |
| MONO (% of WBC) | 2.2 ± 0.8 | 1.6 ± 0.6 | 1.8 ± 0.5 | 2.5 ± 1.0 | 2.3 ± 1.0 |
| EOS (% of WBC) | 0.9 ± 0.5 | 0.4 ± 0.3 * | 0.6 ± 0.2 | 0.7 ± 0.3 | 0.5 ± 0.2 |
| BASO (% of WBC) | 0.4 ± 0.1 | 0.4 ± 0.2 | 0.4 ± 0.1 | 0.5 ± 0.2 | 0.3 ± 0.1 |
| Retic (%) | 2.0 ± 0.2 | 2.2 ± 0.5 | 2.3 ± 0.3 | 2.1 ± 0.4 | 1.9 ± 1.0 |
| Groups (mg/kg/day) | |||||
|---|---|---|---|---|---|
| Parameter | 0 | 12.5 | 25 | 50 | 100 |
| Male | |||||
| ALT (U/L) | 43.0 ± 8.5 | 45.0 ± 13.0 | 47.6 ± 11.3 | 43.3 ± 9.5 | 52.3 ± 5.9 |
| AST (U/L) | 109.2 ± 17.2 | 105.3 ± 20.1 | 98.7 ± 17.6 | 103.2 ± 21.8 | 104.4 ± 15.8 |
| ALP (U/L) | 177.4 ± 40.4 | 205.4 ± 18.9 | 208.4 ± 22.9 | 215.7 ± 23.9 * | 186.6 ± 10.6 |
| GGT (U/L) | 2.2 ± 0.8 | 2.2 ± 1.0 | 2.5 ± 0.5 | 2.2 ± 0.4 | 2.5 ± 0.5 |
| BUN (mg/dL) | 15.5 ± 4.2 | 12.7 ± 3.0 | 12.8 ± 2.4 | 13.5 ± 4.2 | 15.0 ± 2.9 |
| CREA (mg/dL) | 0.7 ± 0.0 | 0.7 ± 0.0 | 0.7 ± 0.0 | 0.8 ± 0.1 | 0.7 ± 0.1 |
| TP (g/L) | 5.9 ± 0.3 | 5.9 ± 0.3 | 6.0 ± 0.3 | 6.1 ± 0.5 | 6.2 ± 0.4 |
| ALB (g/dL) | 3.4 ± 0.1 | 3.4 ± 0.1 | 3.4 ± 0.1 | 3.4 ± 0.1 | 3.4 ± 0.1 |
| T-CHO (mg/dL) | 87.0 ± 10.7 | 93.3 ± 20.0 | 105.3 ± 12.1 | 104.0 ± 16.8 | 117.1 ± 16.5 * |
| GLU (mg/dL) | 132.0 ± 19.5 | 120.4 ± 17.6 | 118.6 ± 20.1 | 132.8 ± 15.5 | 78.7 ± 14.4 * |
| TG (mmol/L) | 64.4 ± 18.0 | 64.9 ± 23.2 | 68.3 ± 22.9 | 65.3 ± 24.8 | 64.3 ± 20.7 |
| T-BIL (mg/dL) | 0.2 ± 0.1 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.1 | 0.2 ± 0.0 |
| D-BIL (mg/dL) | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| LDH (U/L) | 968.2 ± 207.1 | 861.3 ± 387.3 | 645.9 ± 348.8 | 989.0 ± 262.4 | 912.0 ± 273.3 |
| CK (U/L) | 454.0 ± 107.7 | 460.9 ± 207.0 | 374.1 ± 192.4 | 446.5 ± 243.0 | 390.3 ± 128.4 |
| UA (mg/dL) | 1.8 ± 0.2 | 1.6 ± 0.4 | 1.4 ± 0.4 | 1.6 ± 0.4 | 1.4 ± 0.2 |
| CA (mg/dL) | 9.5 ± 0.2 | 10.7 ± 1.5 | 10.0 ± 0.8 | 10.3 ± 1.2 | 11.0 ± 2.5 |
| IP (mg/dL) | 8.9 ± 0.8 | 9.2 ± 0.9 | 8.4 ± 0.9 | 9.5 ± 1.5 | 9.8 ± 1.1 |
| HDL (mg/dL) | 77.4 ± 7.9 | 78.7 ± 14.0 | 85.4 ± 7.9 | 85.3 ± 10.4 | 93.4 ± 10.5 |
| LDL (mg/dL) | 19.8 ± 5.0 | 20.9 ± 6.6 | 26.9 ± 3.8 | 26.5 ± 5.3 | 27.6 ± 5.3 |
| Female | |||||
| ALT (U/L) | 39.6 ± 6.5 | 34.7 ± 9.9 | 32.7 ± 5.6 | 33.0 ± 6.5 | 41.5 ± 5.9 |
| AST (U/L) | 107.0 ± 9.9 | 103.3 ± 13.6 | 101.3 ± 18.2 | 92.9 ± 17.0 | 83.7 ± 17.2 |
| ALP (U/L) | 148.8 ± 32.9 | 147.4 ± 27.1 | 142.9 ± 53.0 | 107.4 ± 20.3 | 116.5 ± 21.0 |
| GGT (U/L) | 3.0 ± 0.7 | 2.4 ± 0.5 | 2.9 ± 0.4 | 3.0 ± 0.0 | 2.6 ± 0.5 |
| BUN (mg/dL) | 11.8 ± 1.2 | 13.0 ± 3.2 | 12.8 ± 2.9 | 16.4 ± 1.6 * | 14.8 ± 4.1 |
| CREA (mg/dL) | 0.6 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.9 ± 0.6 |
| TP (g/L) | 5.7 ± 0.2 | 6.0 ± 0.4 | 6.1 ± 0.3 | 6.2 ± 0.3 | 6.8 ± 0.6 * |
| ALB (g/dL) | 3.4 ± 0.1 | 3.5 ± 0.2 | 3.5 ± 0.1 | 3.5 ± 0.1 | 3.6 ± 0.1 |
| T-CHO (mg/dL) | 79.4 ± 11.6 | 92.1 ± 21.5 | 86.6 ± 13.3 | 101.0 ± 22.7 | 163.2 ± 53.3 * |
| GLU (mg/dL) | 110.6 ± 26.9 | 109.1 ± 15.1 | 118.9 ± 14.1 | 112.0 ± 10.0 | 86.0 ± 34.3 |
| TG (mmol/L) | 29.4 ± 9.0 | 33.6 ± 6.6 | 33.0 ± 11.8 | 35.9 ± 14.0 | 49.6 ± 27.8 |
| T-BIL (mg/dL) | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| D-BIL (mg/dL) | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.1 |
| LDH (U/L) | 1492.0 ± 481.2 | 1488.1 ± 503.5 | 1379.7 ± 604.7 | 1598.4 ± 628.0 | 1403.6 ± 830.7 |
| CK (U/L) | 375.0 ± 64.4 | 397.3 ± 61.3 | 394.6 ± 131.9 | 357.0 ± 119.1 | 326.7 ± 85.4 |
| UA (mg/dL) | 1.4 ± 0.2 | 1.6 ± 0.3 | 1.4 ± 0.3 | 1.4 ± 0.2 | 1.1 ± 0.3 |
| CA (mg/dL) | 9.0 ± 0.2 | 9.7 ± 1.2 | 9.7 ± 1.1 | 9.7 ± 1.2 | 10.7 ± 1.6 |
| IP (mg/dL) | 7.3 ± 1.0 | 7.5 ± 0.5 | 7.6 ± 0.9 | 8.3 ± 1.1 | 8.9 ± 3.1 |
| HDL (mg/dL) | 71.6 ± 7.7 | 78.9 ± 16.5 | 76.0 ± 10.0 | 88.1 ± 15.8 | 136.2 ± 29.8 * |
| LDL (mg/dL) | 13.2 ± 4.4 | 16.6 ± 3.8 | 16.3 ± 3.7 | 16.4 ± 6.9 | 31.8 ± 12.6 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, D.; Shin, H.; Lee, J.; Yang, J.; Jung, K.; Jeong, J.; Won, H.; Oh, J. In Vivo Evaluation of the Oral Toxicity of the Chlorobutanol. Toxics 2022, 10, 24. https://doi.org/10.3390/toxics10010024
Jeong D, Shin H, Lee J, Yang J, Jung K, Jeong J, Won H, Oh J. In Vivo Evaluation of the Oral Toxicity of the Chlorobutanol. Toxics. 2022; 10(1):24. https://doi.org/10.3390/toxics10010024
Chicago/Turabian StyleJeong, Dahye, Hyosook Shin, Jinhee Lee, Junyoung Yang, Kikyung Jung, Jayoung Jeong, Hansol Won, and Jaeho Oh. 2022. "In Vivo Evaluation of the Oral Toxicity of the Chlorobutanol" Toxics 10, no. 1: 24. https://doi.org/10.3390/toxics10010024






