The Effects of Essential and Non-Essential Metal Toxicity in the Drosophila melanogaster Insect Model: A Review
Abstract
:1. Introduction
2. Essential and Non-Essential Metals
3. Metal Homeostasis in Drosophila melanogaster
4. MTF-1 in Drosophila melanogaster
4.1. dMTF-1 Activation and Mechanism
4.2. dMTF-1 and Homeostasis during Metal Stress
4.3. dMTF-1 and Essential Metals
4.4. dMTF-1 and Non-Essential Metals
5. Metallothioneins in Drosophila melanogaster
5.1. Metallothionein Structure and Binding
5.2. Metallothionein Function
5.3. Conservation of Mtn between Species
5.4. Drosophila melanogaster Mtn and Essential Metal Stress
5.5. Sexual Dimorphism in Metallothionein Expression
5.6. Mtn and Non-Essential Stress
6. Metal Transport Systems in Drosophila melanogaster
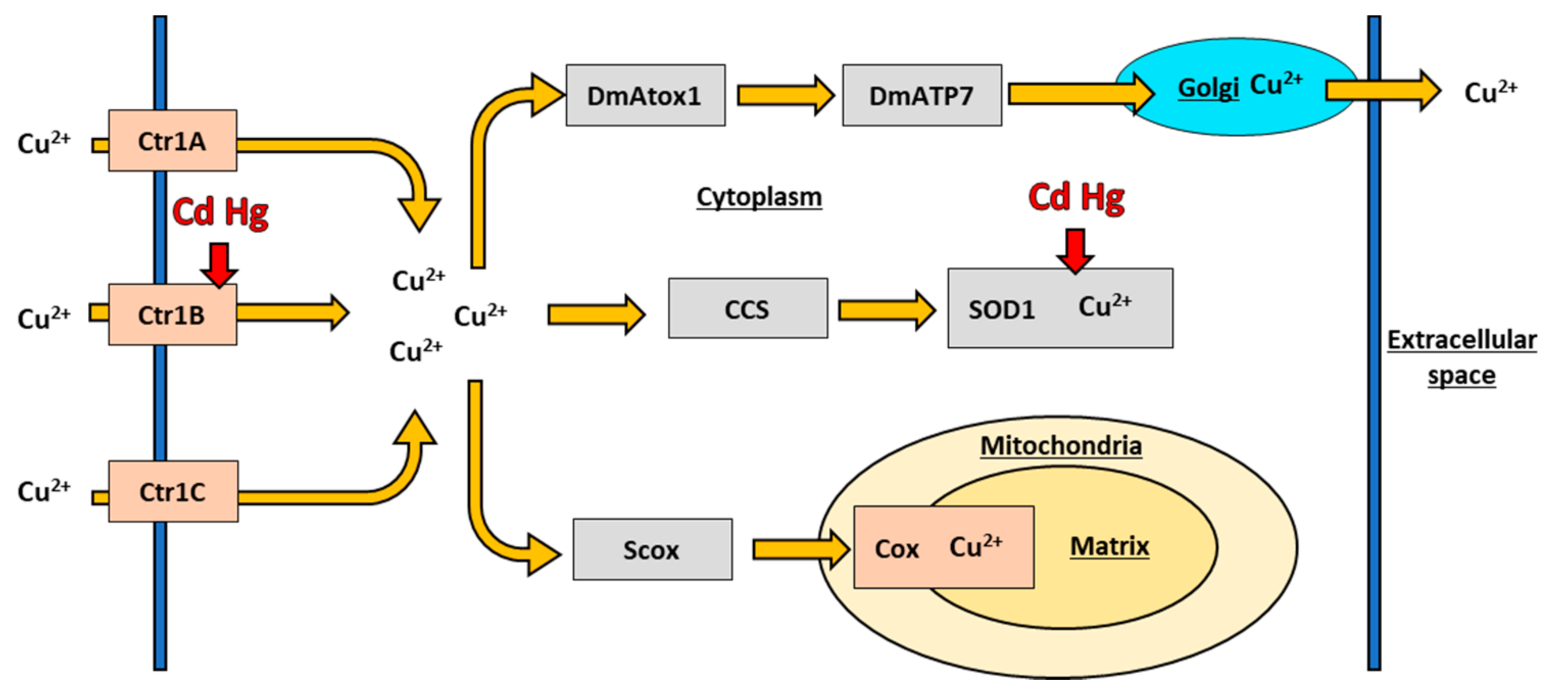
6.1. Copper Transport in Drosophila melanogaster
6.2. Copper Chaperones in Drosophila melanogaster
6.3. Zinc Transport in Drosophila melanogaster
6.4. Zinc Import in Drosophila melanogaster
6.5. Zinc Export in Drosophila melanogaster
6.6. Iron Transport in Drosophila melanogaster
6.7. Iron Import in Drosophila melanogaster
6.8. Iron Involvement in Drosophila melanogaster Spermatogenesis
6.9. Iron Storage and Export in Drosophila melanogaster
7. Non-Essential Metals Interacting with Essential Metal Pathways
8. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Broderick, J.B. Coenzymes and Cofactors. In eLS; American Cancer Society: Atlanta, GA, USA, 2001; ISBN 978-0-470-01590-2. [Google Scholar]
- Keller, W.; Heneberry, J.; Edwards, B.A. Recovery of Acidified Sudbury, Ontario, Canada, Lakes: A Multi-Decade Synthesis and Update. Environ. Rev. 2018, 27, 1–16. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy Metal Toxicity and the Environment. In Molecular, Clinical and Environmental Toxicology: Volume 3: Environmental Toxicology; Luch, A., Ed.; Experientia Supplementum; Springer: Basel, Switzerland, 2012; pp. 133–164. ISBN 978-3-7643-8340-4. [Google Scholar]
- Valko, M.; Morris, H.; Cronin, M.T.D. Metals, Toxicity and Oxidative Stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajasekar, M.; Sreedaran, S.; Prabu, R.; Narayanan, V.; Jegadeesh, R.; Raaman, N.; Rahiman, A.K. Synthesis, Characterization, and Antimicrobial Activities of Nickel(II) and Copper(II) Schiff-Base Complexes. J. Coord. Chem. 2010, 63, 136–146. [Google Scholar] [CrossRef]
- Posthuma, L.; Van Straalen, N.M. Heavy-Metal Adaptation in Terrestrial Invertebrates: A Review of Occurrence, Genetics, Physiology and Ecological Consequences. Comp. Biochem. Physiol. Part C Pharmacol. Toxicol. Endocrinol. 1993, 106, 11–38. [Google Scholar] [CrossRef]
- Sarkar, S.; Duttagupta, A.K.; Mal, T.K. Effects of Heavy Metals on Population Growth and Metallothionein Gene Expression in the Mosquito Culex Quinquefasciatus, from Calcutta, India. Environ. Pollut. 2004, 127, 183–193. [Google Scholar] [CrossRef]
- Nguyen, A.H.; Altomare, L.E.; McElwain, M.C. Decreased Accumulation of Cadmium in Drosophila Selected for Resistance Suggests a Mechanism Independent of Metallothionein. Biol. Trace Elem. Res. 2014, 160, 245–249. [Google Scholar] [CrossRef]
- Kenig, B.; Stamenković-Radak, M.; Andelković, M. Population Specific Fitness Response of Drosophila subobscura to Lead Pollution. Insect Sci. 2013, 20, 245–253. [Google Scholar] [CrossRef]
- Kenig, B.; Patenković, A.; Anđelković, M.; Stamenković-Rada, M. Life-History Variation of Drosophila Subobscura under Lead Pollution Depends on Population History. Genetika 2014, 46, 693–703. [Google Scholar] [CrossRef]
- Kenig, B.; Novičić, Z.K.; Patenković, A.; Stamenković-Radak, M.; Anđelković, M. Adaptive Role of Inversion Polymorphism of Drosophila Subobscura in Lead Stressed Environment. PLoS ONE 2015, 10, e0131270. [Google Scholar] [CrossRef] [Green Version]
- Zhou, S.; Luoma, S.E.; Armour, G.E.S.; Thakkar, E.; Mackay, T.F.C.; Anholt, R.R.H. A Drosophila Model for Toxicogenomics: Genetic Variation in Susceptibility to Heavy Metal Exposure. PLoS Genet. 2017, 13, e1006907. [Google Scholar] [CrossRef] [Green Version]
- Jennings, B.H. Drosophila—A Versatile Model in Biology & Medicine. Mater. Today 2011, 14, 190–195. [Google Scholar] [CrossRef]
- Poteat, M.D.; Buchwalter, D.B. Four Reasons Why Traditional Metal Toxicity Testing with Aquatic Insects Is Irrelevant. Environ. Sci. Technol. 2014, 48, 887–888. [Google Scholar] [CrossRef] [PubMed]
- Skaldina, O.; Sorvari, J. Ecotoxicological Effects of Heavy Metal Pollution on Economically Important Terrestrial Insects. In Networking of Mutagens in Environmental Toxicology; Kesari, K.K., Ed.; Environmental Science and Engineering; Springer International Publishing: Cham, Switzerland, 2019; pp. 137–144. ISBN 978-3-319-96511-6. [Google Scholar]
- Dar, M.I.; Green, I.D.; Khan, F.A. Trace Metal Contamination: Transfer and Fate in Food Chains of Terrestrial Invertebrates. Food Webs 2019, 20, e00116. [Google Scholar] [CrossRef]
- Mogren, C.L.; Trumble, J.T. The Impacts of Metals and Metalloids on Insect Behavior. Entomol. Exp. Appl. 2010, 135, 1–17. [Google Scholar] [CrossRef]
- Maroni, G.; Watson, D. Uptake and Binding of Cadmium, Copper and Zinc by Drosophila melanogaster Larvae. Insect Biochem. 1985, 15, 55–63. [Google Scholar] [CrossRef]
- Machado, H.E.; Bergland, A.O.; Taylor, R.; Tilk, S.; Behrman, E.; Dyer, K.; Fabian, D.K.; Flatt, T.; González, J.; Karasov, T.L.; et al. Broad Geographic Sampling Reveals the Shared Basis and Environmental Correlates of Seasonal Adaptation in Drosophila. eLife 2021, 10, e67577. [Google Scholar] [CrossRef]
- Andreani, G.; Ferlizza, E.; Cabbri, R.; Fabbri, M.; Bellei, E.; Isani, G. Essential (Mg, Fe, Zn and Cu) and Non-Essential (Cd and Pb) Elements in Predatory Insects (Vespa Crabro and Vespa Velutina): A Molecular Perspective. Int. J. Mol. Sci. 2020, 22, 228. [Google Scholar] [CrossRef] [PubMed]
- Rulíšek, L.; Vondrášek, J. Coordination Geometries of Selected Transition Metal Ions (Co2+, Ni2+, Cu2+, Zn2+, Cd2+, and Hg2+) in Metalloproteins. J. Inorg. Biochem. 1998, 71, 115–127. [Google Scholar] [CrossRef]
- Dudev, T.; Lim, C. Competition among Metal Ions for Protein Binding Sites: Determinants of Metal Ion Selectivity in Proteins. Chem. Rev. 2014, 114, 538–556. [Google Scholar] [CrossRef]
- Gutiérrez, L.; Zubow, K.; Nield, J.; Gambis, A.; Mollereau, B.; Lázaro, F.J.; Missirlis, F. Biophysical and Genetic Analysis of Iron Partitioning and Ferritin Function in Drosophila melanogaster. Metallomics 2013, 5, 997–1005. [Google Scholar] [CrossRef] [Green Version]
- Matés, J.M. Effects of Antioxidant Enzymes in the Molecular Control of Reactive Oxygen Species Toxicology. Toxicology 2000, 153, 83–104. [Google Scholar] [CrossRef]
- Wang, S.; Cao, J.; Jia, W.; Guo, W.; Yan, S.; Wang, Y.; Zhang, P.; Chen, H.-Y.; Huang, S. Single Molecule Observation of Hard–Soft-Acid–Base (HSAB) Interaction in Engineered Mycobacterium Smegmatis Porin A (MspA) Nanopores. Chem. Sci. 2020, 11, 879–887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, M. Chemical Softness and Acute Metal Toxicity in Mice and Drosophila. Toxicol. Appl. Pharmacol. 1982, 63, 461–469. [Google Scholar] [CrossRef]
- Remko, M.; Fitz, D.; Rode, B.M. Effect of Metal Ions (Li+, Na+, K+, Mg2+, Ca2+, Ni2+, Cu2+ and Zn2+) and Water Coordination on the Structure and Properties of L-Histidine and Zwitterionic L-Histidine. Amino Acids 2010, 39, 1309–1319. [Google Scholar] [CrossRef]
- Tamás, M.J.; Sharma, S.K.; Ibstedt, S.; Jacobson, T.; Christen, P. Heavy Metals and Metalloids As a Cause for Protein Misfolding and Aggregation. Biomolecules 2014, 4, 252–267. [Google Scholar] [CrossRef] [Green Version]
- Klein Couto, G.; Rodrigues, J.C.; Pacheco, B.S.; Damé Simões, L.; Paschoal, J.D.; Seixas, F.K.; Acunha, T.V.; Iglesias, B.A.; Collares, T. Zinc(II), Copper(II) and Nickel(II) Ions Improve the Selectivity of Tetra-Cationic Platinum(II) Porphyrins in Photodynamic Therapy and Stimulate Antioxidant Defenses in the Metastatic Melanoma Lineage (A375). Photodiagnosis Photodyn. Ther. 2020, 31, 101942. [Google Scholar] [CrossRef]
- Navarro, J.A.; Schneuwly, S. Copper and Zinc Homeostasis: Lessons from Drosophila melanogaster. Front. Genet. 2017, 8, 223. [Google Scholar] [CrossRef] [PubMed]
- Calap-Quintana, P.; González-Fernández, J.; Sebastiá-Ortega, N.; Llorens, J.V.; Moltó, M.D. Drosophila melanogaster Models of Metal-Related Human Diseases and Metal Toxicity. Int. J. Mol. Sci. 2017, 18, 1456. [Google Scholar] [CrossRef] [Green Version]
- Laity, J.H.; Andrews, G.K. Understanding the Mechanisms of Zinc-Sensing by Metal-Response Element Binding Transcription Factor-1 (MTF-1). Arch. Biochem. Biophys. 2007, 463, 201–210. [Google Scholar] [CrossRef]
- Marr, S.K.; Pennington, K.L.; Marr, M.T. Efficient Metal-Specific Transcription Activation by Drosophila MTF-1 Requires Conserved Cysteine Residues in the Carboxy-Terminal Domain. Biochim. Biophys. Acta Gene Regul. Mech. 2012, 1819, 902–912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.; Hua, H.; Balamurugan, K.; Kong, X.; Zhang, L.; George, G.N.; Georgiev, O.; Schaffner, W.; Giedroc, D.P. Copper Sensing Function of Drosophila Metal-Responsive Transcription Factor-1 Is Mediated by a Tetranuclear Cu(I) Cluster. Nucleic Acids Res. 2008, 36, 3128–3138. [Google Scholar] [CrossRef]
- Yepiskoposyan, H.; Egli, D.; Fergestad, T.; Selvaraj, A.; Treiber, C.; Multhaup, G.; Georgiev, O.; Schaffner, W. Transcriptome Response to Heavy Metal Stress in Drosophila Reveals a New Zinc Transporter That Confers Resistance to Zinc. Nucleic Acids Res. 2006, 34, 4866–4877. [Google Scholar] [CrossRef]
- Andrews, G.K. Regulation of Metallothionein Gene Expression by Oxidative Stress and Metal Ions. Biochem. Pharmacol. 2000, 59, 95–104. [Google Scholar] [CrossRef]
- LaRochelle, O.; Gagné, V.; Charron, J.; Soh, J.-W.; Séguin, C. Phosphorylation Is Involved in the Activation of Metal-Regulatory Transcription Factor 1 in Response to Metal Ions. J. Biol. Chem. 2001, 276, 41879–41888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Missirlis, F. Regulation and Biological Function of Metal Ions in Drosophila. Curr. Opin. Insect Sci. 2021, 47, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Giedroc, D.P.; Chen, X.; Pennella, M.A.; LiWang, A.C. Conformational Heterogeneity in the C-Terminal Zinc Fingers of Human MTF-1 AN NMR AND ZINC-BINDING STUDY. J. Biol. Chem. 2001, 276, 42322–42332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, M.; Finet, C.; Cong, H.; Wei, H.; Chung, H. The Evolution of Insect Metallothioneins. Proc. R. Soc. B Biol. Sci. 2020, 287, 20202189. [Google Scholar] [CrossRef]
- Moleirinho, A.; Carneiro, J.; Matthiesen, R.; Silva, R.M.; Amorim, A.; Azevedo, L. Gains, Losses and Changes of Function after Gene Duplication: Study of the Metallothionein Family. PLoS ONE 2011, 6, e18487. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.; Egli, D.; Georgiev, O.; Schaffner, W. The Drosophila Homolog of Mammalian Zinc Finger Factor MTF-1 Activates Transcription in Response to Heavy Metals. Mol. Cell. Biol. 2001, 21, 4505–4514. [Google Scholar] [CrossRef] [Green Version]
- Giedroc, D.P.; Chen, X.; Apuy, J.L. Metal Response Element (MRE)-Binding Transcription Factor-1 (MTF-1): Structure, Function, and Regulation. Antioxid. Redox Signal. 2001, 3, 577–596. [Google Scholar] [CrossRef]
- Larochelle, O.; Stewart, G.; Moffatt, P.; Tremblay, V.; Séguin, C. Characterization of the Mouse Metal-Regulatory-Element-Binding Proteins, Metal Element Protein-1 and Metal Regulatory Transcription Factor-1. Biochem. J. 2001, 353, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Saydam, N.; Adams, T.K.; Steiner, F.; Schaffner, W.; Freedman, J.H. Regulation of Metallothionein Transcription by the Metal-Responsive Transcription Factor MTF-1: Identification of signal transduction cascades that control metal-inducible transcription. J. Biol. Chem. 2002, 277, 20438–20445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Günther, V.; Davis, A.M.; Georgiev, O.; Schaffner, W. A Conserved Cysteine Cluster, Essential for Transcriptional Activity, Mediates Homodimerization of Human Metal-Responsive Transcription Factor-1 (MTF-1). Biochim. Biophys. Acta Mol. Cell Res. 2012, 1823, 476–483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smirnova, I.V.; Bittel, D.C.; Ravindra, R.; Jiang, H.; Andrews, G.K. Zinc and Cadmium Can Promote Rapid Nuclear Translocation of Metal Response Element-Binding Transcription Factor-1. J. Biol. Chem. 2000, 275, 9377–9384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahadorani, S.; Mukai, S.; Egli, D.; Hilliker, A.J. Overexpression of Metal-Responsive Transcription Factor (MTF-1) in Drosophila melanogaster Ameliorates Life-Span Reductions Associated with Oxidative Stress and Metal Toxicity. Neurobiol. Aging 2010, 31, 1215–1226. [Google Scholar] [CrossRef] [PubMed]
- Dalton, T.P.; Li, Q.; Bittel, D.; Liang, L.; Andrews, G.K. Oxidative Stress Activates Metal-Responsive Transcription Factor-1 Binding Activity: Occupancy in vivo of metal response elements in the metallothionein-i gene promoter. J. Biol. Chem. 1996, 271, 26233–26241. [Google Scholar] [CrossRef] [Green Version]
- Selvaraj, A.; Balamurugan, K.; Yepiskoposyan, H.; Zhou, H.; Egli, D.; Georgiev, O.; Thiele, D.J.; Schaffner, W. Metal-Responsive Transcription Factor (MTF-1) Handles Both Extremes, Copper Load and Copper Starvation, by Activating Different Genes. Genes Dev. 2005, 19, 891–896. [Google Scholar] [CrossRef] [Green Version]
- Balamurugan, K.; Egli, D.; Selvaraj, A.; Zhang, B.; Georgiev, O.; Schaffner, W. Metal-Responsive Transcription Factor (MTF-1) and Heavy Metal Stress Response in Drosophila and Mammalian Cells: A Functional Comparison. Biol. Chem. 2004, 385, 597–603. [Google Scholar] [CrossRef]
- Egli, D.; Yepiskoposyan, H.; Selvaraj, A.; Balamurugan, K.; Rajaram, R.; Simons, A.; Multhaup, G.; Mettler, S.; Vardanyan, A.; Georgiev, O.; et al. A Family Knockout of All Four Drosophila Metallothioneins Reveals a Central Role in Copper Homeostasis and Detoxification. Mol. Cell. Biol. 2006, 26, 2286–2296. [Google Scholar] [CrossRef] [Green Version]
- Günther, V.; Lindert, U.; Schaffner, W. The Taste of Heavy Metals: Gene Regulation by MTF-1. Biochim. Biophys. Acta Mol. Cell Res. 2012, 1823, 1416–1425. [Google Scholar] [CrossRef] [Green Version]
- Egli, D.; Domènech, J.; Selvaraj, A.; Balamurugan, K.; Hua, H.; Capdevila, M.; Georgiev, O.; Schaffner, W.; Atrian, S. The Four Members of the Drosophila Metallothionein Family Exhibit Distinct yet Overlapping Roles in Heavy Metal Homeostasis and Detoxification. Genes Cells 2006, 11, 647–658. [Google Scholar] [CrossRef]
- Sims, H.I.; Chirn, G.-W.; Marr, M.T. Single Nucleotide in the MTF-1 Binding Site Can Determine Metal-Specific Transcription Activation. Proc. Natl. Acad. Sci. USA 2012, 109, 16516–16521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Günther, V.; Waldvogel, D.; Nosswitz, M.; Georgiev, O.; Schaffner, W. Dissection of Drosophila MTF-1 Reveals a Domain for Differential Target Gene Activation upon Copper Overload vs. Copper Starvation. Int. J. Biochem. Cell Biol. 2012, 44, 404–411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Norgate, M.; Lee, E.; Southon, A.; Farlow, A.; Batterham, P.; Camakaris, J.; Burke, R. Essential Roles in Development and Pigmentation for the Drosophila Copper Transporter DmATP7. Mol. Biol. Cell 2005, 17, 475–484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burke, R.; Commons, E.; Camakaris, J. Expression and Localisation of the Essential Copper Transporter DmATP7 in Drosophila Neuronal and Intestinal Tissues. Int. J. Biochem. Cell Biol. 2008, 40, 1850–1860. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Cadigan, K.M.; Thiele, D.J. A Copper-Regulated Transporter Required for Copper Acquisition, Pigmentation, and Specific Stages of Development in Drosophila melanogaster. J. Biol. Chem. 2003, 278, 48210–48218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Egli, D.; Selvaraj, A.; Yepiskoposyan, H.; Zhang, B.; Hafen, E.; Georgiev, O.; Schaffner, W. Knockout of ‘Metal-Responsive Transcription Factor’ MTF-1 in Drosophila by Homologous Recombination Reveals Its Central Role in Heavy Metal Homeostasis. EMBO J. 2003, 22, 100–108. [Google Scholar] [CrossRef] [Green Version]
- Southon, A.; Burke, R.; Norgate, M.; Batterham, P.; Camakaris, J. Copper Homoeostasis in Drosophila melanogaster S2 Cells. Biochem. J. 2004, 383, 303–309. [Google Scholar] [CrossRef] [Green Version]
- Richards, C.D.; Burke, R. A Fly’s Eye View of Zinc Homeostasis: Novel Insights into the Genetic Control of Zinc Metabolism from Drosophila. Arch. Biochem. Biophys. 2016, 611, 142–149. [Google Scholar] [CrossRef]
- Xiao, G.; Zhou, B. What Can Flies Tell Us about Zinc Homeostasis? Arch. Biochem. Biophys. 2016, 611, 134–141. [Google Scholar] [CrossRef] [Green Version]
- Balamurugan, K.; Hua, H.; Georgiev, O.; Schaffner, W. Mercury and Cadmium Trigger Expression of the Copper Importer Ctr1B, Which Enables Drosophila to Thrive on Heavy Metal-Loaded Food. Biol. Chem. 2009, 390, 109–113. [Google Scholar] [CrossRef] [Green Version]
- Ziller, A.; Fraissinet-Tachet, L. Metallothionein Diversity and Distribution in the Tree of Life: A Multifunctional Protein. Metallomics 2018, 10, 1549–1559. [Google Scholar] [CrossRef]
- Binz, P.-A.; Kägi, J.H.R. Metallothionein: Molecular evolution and classification. In Metallothionein IV; Klaassen, C.D., Ed.; Advances in Life Sciences; Birkhäuser: Basel, Switzerland, 1999; pp. 7–13. ISBN 978-3-0348-8847-9. [Google Scholar]
- Enescu, M.; Renault, J.-P.; Pommeret, S.; Mialocq, J.-C.; Pin, S. Ab Initio Study of Cd– Thiol Complexes: Application to the Modelling of the Metallothionein Active Site. Phys. Chem. Chem. Phys. 2003, 5, 3762–3767. [Google Scholar] [CrossRef]
- Valls, M.; Bofill, R.; Romero-Isart, N.; Gonzàlez-Duarte, R.; Abián, J.; Carrascal, M.; Gonzàlez-Duarte, P.; Capdevila, M.; Atrian, S. Drosophila MTN: A Metazoan Copper-Thionein Related to Fungal Forms. FEBS Lett. 2000, 467, 189–194. [Google Scholar] [CrossRef] [Green Version]
- Atanesyan, L.; Günther, V.; Celniker, S.E.; Georgiev, O.; Schaffner, W. Characterization of MtnE, the Fifth Metallothionein Member in Drosophila. JBIC J. Biol. Inorg. Chem. 2011, 16, 1047. [Google Scholar] [CrossRef] [Green Version]
- Balamurugan, K.; Egli, D.; Hua, H.; Rajaram, R.; Seisenbacher, G.; Georgiev, O.; Schaffner, W. Copper Homeostasis in Drosophila by Complex Interplay of Import, Storage and Behavioral Avoidance. EMBO J. 2007, 26, 1035–1044. [Google Scholar] [CrossRef] [PubMed]
- Willis, M.S.; Monaghan, S.A.; Miller, M.L.; McKenna, R.W.; Perkins, W.D.; Levinson, B.S.; Bhushan, V.; Kroft, S.H. Zinc-Induced Copper Deficiency: A Report of Three Cases Initially Recognized on Bone Marrow Examination. Am. J. Clin. Pathol. 2005, 123, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, H.; Miyakoshi, Y.; Kobayashi, K.; Sakae, K.; Kawasaki, I.; Suzuki, Y.; Tamura, J. Long-Term Intake of a High Zinc Diet Causes Iron Deficiency Anemia Accompanied by Reticulocytosis and Extra-Medullary Erythropoiesis. Toxicol. Lett. 2009, 191, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Lastowski-Perry, D.; Otto, E.; Maroni, G. Nucleotide Sequence and Expression of a Drosophila Metallothionein. J. Biol. Chem. 1985, 260, 1527–1530. [Google Scholar] [CrossRef]
- Capasso, C.; Carginale, V.; Scudiero, R.; Crescenzi, O.; Spadaccini, R.; Temussi, P.A.; Parisi, E. Phylogenetic Divergence of Fish and Mammalian Metallothionein: Relationships with Structural Diversification and Organismal Temperature. J. Mol. Evol. 2003, 57 (Suppl. 1), S250–S257. [Google Scholar] [CrossRef]
- Guirola, M.; Naranjo, Y.; Capdevila, M.; Atrian, S. Comparative genomics analysis of metallothioneins in twelve Drosophila species. J. Inorg. Biochem. 2011, 105, 1050–1059. [Google Scholar] [CrossRef]
- Maroni, G.; Wise, J.; Young, J.E.; Otto, E. Metallothionein Gene Duplications and Metal Tolerance in Natural Populations of Drosophila melanogaster. Genetics 1987, 117, 739–744. [Google Scholar] [CrossRef]
- Merritt, T.J.S.; Bewick, A.J. Genetic Diversity in Insect Metal Tolerance. Front. Genet. 2017, 8. [Google Scholar] [CrossRef] [Green Version]
- Maghdooni Bagheri, P.; Rahman, M.T.; Van Soest, S.; De Ley, M. Differential Quantitative Zinc-Induced Expression of Human Metallothionein Isogenes in Haematopoietic Precursor Cell Lines. J. Trace Elem. Med. Biol. 2009, 23, 124–131. [Google Scholar] [CrossRef]
- Norgate, M.; Southon, A.; Zou, S.; Zhan, M.; Sun, Y.; Batterham, P.; Camakaris, J. Copper Homeostasis Gene Discovery in Drosophila melanogaster. BioMetals 2007, 20, 683. [Google Scholar] [CrossRef]
- Qiang, W.; Huang, Y.; Wan, Z.; Zhou, B. Metal-Metal Interaction Mediates the Iron Induction of Drosophila MtnB. Biochem. Biophys. Res. Commun. 2017, 487, 646–652. [Google Scholar] [CrossRef]
- Pérez-Rafael, S.; Kurz, A.; Guirola, M.; Capdevila, M.; Palacios, Ò.; Atrian, S. Is MtnE, the Fifth Drosophila Metallothionein, Functionally Distinct from the Other Members of This Polymorphic Protein Family? Metallomics 2012, 4, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Halmenschelager, P.T.; da Rocha, J.B.T. Biochemical CuSO4 Toxicity in Drosophila melanogaster Depends on Sex and Developmental Stage of Exposure. Biol. Trace Elem. Res. 2019, 189, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Balinski, M.A.; Woodruff, R.C. Differential Sexual Survival of Drosophila melanogaster on Copper Sulfate. Genetica 2017, 145, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Frat, L.; Chertemps, T.; Pesce, E.; Bozzolan, F.; Dacher, M.; Planelló, R.; Herrero, O.; Llorente, L.; Moers, D.; Siaussat, D. Single and Mixed Exposure to Cadmium and Mercury in Drosophila melanogaster: Molecular Responses and Impact on Post-Embryonic Development. Ecotoxicol. Environ. Saf. 2021, 220, 112377. [Google Scholar] [CrossRef] [PubMed]
- Yasutake, A.; Nakamura, M. Induction by Mercury Compounds of Metallothioneins in Mouse Tissues: Inorganic Mercury Accumulation Is Not a Dominant Factor for Metallothionein Induction in the Liver. J. Toxicol. Sci. 2011, 36, 365–372. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Wu, H.; Kou, L.; Liu, X.; Zhang, J.; Guo, Y.; Ma, E. Two Metallothionein Genes in Oxya Chinensis: Molecular Characteristics, Expression Patterns and Roles in Heavy Metal Stress. PLoS ONE 2014, 9, e112759. [Google Scholar] [CrossRef]
- Attig, H.; Kamel, N.; Sforzini, S.; Dagnino, A.; Jamel, J.; Boussetta, H.; Viarengo, A.; Banni, M. Effects of Thermal Stress and Nickel Exposure on Biomarkers Responses in Mytilus Galloprovincialis (Lam). Mar. Environ. Res. 2014, 94, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Nemec, A.A.; Leikauf, G.D.; Pitt, B.R.; Wasserloos, K.J.; Barchowsky, A. Nickel Mobilizes Intracellular Zinc to Induce Metallothionein in Human Airway Epithelial Cells. Am. J. Respir. Cell Mol. Biol. 2009, 41, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Festa, R.A.; Thiele, D.J. Copper: An Essential Metal in Biology. Curr. Biol. 2011, 21, R877–R883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hatori, Y.; Lutsenko, S. The Role of Copper Chaperone Atox1 in Coupling Redox Homeostasis to Intracellular Copper Distribution. Antioxidants 2016, 5, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hua, H.; Georgiev, O.; Schaffner, W.; Steiger, D. Human Copper Transporter Ctr1 Is Functional in Drosophila, Revealing a High Degree of Conservation between Mammals and Insects. JBIC J. Biol. Inorg. Chem. 2009, 15, 107. [Google Scholar] [CrossRef] [Green Version]
- Turski, M.L.; Thiele, D.J. Drosophila Ctr1A Functions as a Copper Transporter Essential for Development. J. Biol. Chem. 2007, 282, 24017–24026. [Google Scholar] [CrossRef] [Green Version]
- Balamurugan, K.; Schaffner, W. Copper Homeostasis in Eukaryotes: Teetering on a Tightrope. Biochim. Biophys. Acta Mol. Cell Res. 2006, 1763, 737–746. [Google Scholar] [CrossRef] [Green Version]
- Steiger, D.; Fetchko, M.; Vardanyan, A.; Atanesyan, L.; Steiner, K.; Turski, M.L.; Thiele, D.J.; Georgiev, O.; Schaffner, W. The Drosophila Copper Transporter Ctr1C Functions in Male Fertility. J. Biol. Chem. 2010, 285, 17089–17097. [Google Scholar] [CrossRef] [Green Version]
- Hua, H.; Günther, V.; Georgiev, O.; Schaffner, W. Distorted Copper Homeostasis with Decreased Sensitivity to Cisplatin upon Chaperone Atox1 Deletion in Drosophila. BioMetals 2011, 24, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Binks, T.; Lye, J.C.; Camakaris, J.; Burke, R. Tissue-Specific Interplay between Copper Uptake and Efflux in Drosophila. JBIC J. Biol. Inorg. Chem. 2010, 15, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Kirby, K.; Jensen, L.T.; Binnington, J.; Hilliker, A.J.; Ulloa, J.; Culotta, V.C.; Phillips, J.P. Instability of Superoxide Dismutase 1 of Drosophila in Mutants Deficient for Its Cognate Copper Chaperone. J. Biol. Chem. 2008, 283, 35393–35401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theotoki, E.I.; Velentzas, A.D.; Katarachia, S.A.; Papandreou, N.C.; Kalavros, N.I.; Pasadaki, S.N.; Giannopoulou, A.F.; Giannios, P.; Iconomidou, V.A.; Konstantakou, E.G.; et al. Targeting of Copper-Trafficking Chaperones Causes Gene-Specific Systemic Pathology in Drosophila melanogaster: Prospective Expansion of Mutational Landscapes That Regulate Tumor Resistance to Cisplatin. Biol. Open 2019, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porcelli, D.; Oliva, M.; Duchi, S.; Latorre, D.; Cavaliere, V.; Barsanti, P.; Villani, G.; Gargiulo, G.; Caggese, C. Genetic, Functional and Evolutionary Characterization of Scox, the Drosophila melanogaster Ortholog of the Human SCO1 Gene. Mitochondrion 2010, 10, 433–448. [Google Scholar] [CrossRef] [PubMed]
- Oteiza, P.I. Zinc and the Modulation of Redox Homeostasis. Free Radic. Biol. Med. 2012, 53, 1748–1759. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andreini, C.; Banci, L.; Bertini, I.; Rosato, A. Counting the Zinc-Proteins Encoded in the Human Genome. J. Proteome Res. 2006, 5, 196–201. [Google Scholar] [CrossRef]
- Maret, W. Zinc and the Zinc Proteome. In Metallomics and the Cell; Banci, L., Ed.; Metal Ions in Life Sciences; Springer: Dordrecht, The Netherlands, 2013; pp. 479–501. ISBN 978-94-007-5561-1. [Google Scholar]
- Andreini, C.; Banci, L.; Bertini, I.; Rosato, A. Zinc through the Three Domains of Life. J. Proteome Res. 2006, 5, 3173–3178. [Google Scholar] [CrossRef]
- Yin, S.; Qin, Q.; Zhou, B. Functional Studies of Drosophila Zinc Transporters Reveal the Mechanism for Zinc Excretion in Malpighian Tubules. BMC Biol. 2017, 15, 12. [Google Scholar] [CrossRef] [Green Version]
- Lye, J.C.; Richards, C.D.; Dechen, K.; Paterson, D.; de Jonge, M.D.; Howard, D.L.; Warr, C.G.; Burke, R. Systematic Functional Characterization of Putative Zinc Transport Genes and Identification of Zinc Toxicosis Phenotypes in Drosophila melanogaster. J. Exp. Biol. 2012, 215, 3254–3265. [Google Scholar] [CrossRef] [Green Version]
- Lye, J.C.; Richards, C.D.; Dechen, K.; Warr, C.G.; Burke, R. In Vivo Zinc Toxicity Phenotypes Provide a Sensitized Background That Suggests Zinc Transport Activities for Most of the DrosophilaZip and ZnT Genes. JBIC J. Biol. Inorg. Chem. 2013, 18, 323–332. [Google Scholar] [CrossRef]
- Richards, C.D.; Warr, C.G.; Burke, R. A Role for DZIP89B in Drosophila Dietary Zinc Uptake Reveals Additional Complexity in the Zinc Absorption Process. Int. J. Biochem. Cell Biol. 2015, 69, 11–19. [Google Scholar] [CrossRef]
- Qin, Q.; Wang, X.; Zhou, B. Functional Studies of Drosophilazinc Transporters Reveal the Mechanism for Dietary Zinc Absorption and Regulation. BMC Biol. 2013, 11, 101. [Google Scholar] [CrossRef] [Green Version]
- Mathews, W.R.; Ong, D.; Milutinovich, A.B.; Doren, M.V. Zinc Transport Activity of Fear of Intimacy Is Essential for Proper Gonad Morphogenesis and DE-Cadherin Expression. Development 2006, 133, 1143–1153. [Google Scholar] [CrossRef] [Green Version]
- Mathews, W.R.; Wang, F.; Eide, D.J.; Doren, M.V. Drosophila Fear of Intimacy Encodes a Zrt/IRT-like Protein (ZIP) Family Zinc Transporter Functionally Related to Mammalian ZIP Proteins. J. Biol. Chem. 2005, 280, 787–795. [Google Scholar] [CrossRef] [Green Version]
- Van Doren, M.; Mathews, W.R.; Samuels, M.; Moore, L.A.; Broihier, H.T.; Lehmann, R. Fear of Intimacy Encodes a Novel Transmembrane Protein Required for Gonad Morphogenesis in Drosophila. Development 2003, 130, 2355–2364. [Google Scholar] [CrossRef] [Green Version]
- Carrasco-Rando, M.; Atienza-Manuel, A.; Martín, P.; Burke, R.; Ruiz-Gómez, M. Fear-of-Intimacy-Mediated Zinc Transport Controls the Function of Zinc-Finger Transcription Factors Involved in Myogenesis. Development 2016, 143, 1948–1957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dechen, K.; Richards, C.D.; Lye, J.C.; Hwang, J.E.C.; Burke, R. Compartmentalized Zinc Deficiency and Toxicities Caused by ZnT and Zip Gene over Expression Result in Specific Phenotypes in Drosophila. Int. J. Biochem. Cell Biol. 2015, 60, 23–33. [Google Scholar] [CrossRef]
- Groth, C.; Sasamura, T.; Khanna, M.R.; Whitley, M.; Fortini, M.E. Protein Trafficking Abnormalities in Drosophila Tissues with Impaired Activity of the ZIP7 Zinc Transporter Catsup. Development 2013, 140, 3018–3027. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, J.; Wan, Z.; Zhou, B. Drosophila ZIP13 Is Posttranslationally Regulated by Iron-Mediated Stabilization. Biochim. Biophys. Acta Mol. Cell Res. 2019, 1866, 1487–1497. [Google Scholar] [CrossRef] [PubMed]
- Xiao, G.; Wan, Z.; Fan, Q.; Tang, X.; Zhou, B. The Metal Transporter ZIP13 Supplies Iron into the Secretory Pathway in Drosophila melanogaster. eLife 2014, 3, e03191. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wu, Y.; Zhou, B. Dietary Zinc Absorption Is Mediated by ZnT1 in Drosophila melanogaster. FASEB J. 2009, 23, 2650–2661. [Google Scholar] [CrossRef] [PubMed]
- Tejeda-Guzmán, C.; Rosas-Arellano, A.; Kroll, T.; Webb, S.M.; Barajas-Aceves, M.; Osorio, B.; Missirlis, F. Biogenesis of Zinc Storage Granules in Drosophila melanogaster. J. Exp. Biol. 2018, 221, jeb168419. [Google Scholar] [CrossRef] [Green Version]
- Bird, A.J.; Wilson, S. Zinc Homeostasis in the Secretory Pathway in Yeast. Curr. Opin. Chem. Biol. 2020, 55, 145–150. [Google Scholar] [CrossRef]
- Xiao, G.; Zhou, B. ZIP13: A Study of Drosophila Offers an Alternative Explanation for the Corresponding Human Disease. Front. Genet. 2018, 8, 234. [Google Scholar] [CrossRef] [Green Version]
- Bettedi, L.; Aslam, M.F.; Szular, J.; Mandilaras, K.; Missirlis, F. Iron Depletion in the Intestines of Malvolio Mutant Flies Does Not Occur in the Absence of a Multicopper Oxidase. J. Exp. Biol. 2011, 214, 971–978. [Google Scholar] [CrossRef] [Green Version]
- Orgad, S.; Nelson, H.; Segal, D.; Nelson, N. Metal Ions Suppress the Abnormal Taste Behavior of the Drosophila Mutant Malvolio. J. Exp. Biol. 1998, 201, 115–120. [Google Scholar] [CrossRef]
- Folwell, J.L.; Barton, C.H.; Shepherd, D. Immunolocalisation of the D. melanogaster Nramp Homologue Malvolio to Gut and Malpighian Tubules Provides Evidence That Malvolio and Nramp2 Are Orthologous. J. Exp. Biol. 2006, 209, 1988–1995. [Google Scholar] [CrossRef] [Green Version]
- Southon, A.; Farlow, A.; Norgate, M.; Burke, R.; Camakaris, J. Malvolio Is a Copper Transporter in Drosophila melanogaster. J. Exp. Biol. 2008, 211, 709–716. [Google Scholar] [CrossRef] [Green Version]
- Rouault, T.A. The Role of Iron Regulatory Proteins in Mammalian Iron Homeostasis and Disease. Nat. Chem. Biol. 2006, 2, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Muckenthaler, M.; Gunkel, N.; Frishman, D.; Cyrklaff, A.; Tomancak, P.; Hentze, M.W. Iron-Regulatory Protein-1 (IRP-1) Is Highly Conserved in Two Invertebrate Species. Eur. J. Biochem. 1998, 254, 230–237. [Google Scholar] [CrossRef] [Green Version]
- Lind, M.I.; Missirlis, F.; Melefors, Ö.; Uhrigshardt, H.; Kirby, K.; Phillips, J.P.; Söderhäll, K.; Rouault, T.A. Of Two Cytosolic Aconitases Expressed in Drosophila, Only One Functions as an Iron-Regulatory Protein. J. Biol. Chem. 2006, 281, 18707–18714. [Google Scholar] [CrossRef] [Green Version]
- Huynh, N.; Ou, Q.; Cox, P.; Lill, R.; King-Jones, K. Glycogen Branching Enzyme Controls Cellular Iron Homeostasis via Iron Regulatory Protein 1 and MitoNEET. Nat. Commun. 2019, 10, 5463. [Google Scholar] [CrossRef]
- Metzendorf, C.; Lind, M.I. Drosophila Mitoferrinis Essential for Male Fertility: Evidence for a Role of Mitochondrial Iron Metabolism during Spermatogenesis. BMC Dev. Biol. 2010, 10, 68. [Google Scholar] [CrossRef] [Green Version]
- Anderson, P.R.; Kirby, K.; Orr, W.C.; Hilliker, A.J.; Phillips, J.P. Hydrogen Peroxide Scavenging Rescues Frataxin Deficiency in a Drosophila Model of Friedreich’s Ataxia. Proc. Natl. Acad. Sci. USA 2008, 105, 611–616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Runko, A.P.; Griswold, A.J.; Min, K.-T. Overexpression of Frataxin in the Mitochondria Increases Resistance to Oxidative Stress and Extends Lifespan in Drosophila. FEBS Lett. 2008, 582, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Navarro, J.A.; Botella, J.A.; Metzendorf, C.; Lind, M.I.; Schneuwly, S. Mitoferrin Modulates Iron Toxicity in a Drosophila Model of Friedreich’s Ataxia. Free Radic. Biol. Med. 2015, 85, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Prohaska, J.R. Impact of Copper Limitation on Expression and Function of Multicopper Oxidases (Ferroxidases). Adv. Nutr. 2011, 2, 89–95. [Google Scholar] [CrossRef] [Green Version]
- Vashchenko, G.; MacGillivray, R.T.A. Multi-Copper Oxidases and Human Iron Metabolism. Nutrients 2013, 5, 2289–2313. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Yin, S.; Yang, Z.; Zhou, B. Drosophila Multicopper Oxidase 3 Is a Potential Ferroxidase Involved in Iron Homeostasis. Biochim. Biophys. Acta Gen. Subj. 2018, 1862, 1826–1834. [Google Scholar] [CrossRef]
- Lang, M.; Braun, C.L.; Kanost, M.R.; Gorman, M.J. Multicopper Oxidase-1 Is a Ferroxidase Essential for Iron Homeostasis in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2012, 109, 13337–13342. [Google Scholar] [CrossRef] [Green Version]
- Missirlis, F.; Holmberg, S.; Georgieva, T.; Dunkov, B.C.; Rouault, T.A.; Law, J.H. Characterization of Mitochondrial Ferritin in Drosophila. Proc. Natl. Acad. Sci. USA 2006, 103, 5893–5898. [Google Scholar] [CrossRef] [Green Version]
- Yang, Q.; Liu, W.; Zhang, S.; Liu, S. The Cardinal Roles of Ferroportin and Its Partners in Controlling Cellular Iron in and Out. Life Sci. 2020, 258, 118135. [Google Scholar] [CrossRef]
- Xiao, G.; Liu, Z.-H.; Zhao, M.; Wang, H.-L.; Zhou, B. Transferrin 1 Functions in Iron Trafficking and Genetically Interacts with Ferritin in Drosophila melanogaster. Cell Rep. 2019, 26, 748–758. [Google Scholar] [CrossRef] [Green Version]
- Tang, X.; Zhou, B. Iron Homeostasis in Insects: Insights from Drosophila Studies. IUBMB Life 2013, 65, 863–872. [Google Scholar] [CrossRef]
- Missirlis, F.; Kosmidis, S.; Brody, T.; Mavrakis, M.; Holmberg, S.; Odenwald, W.F.; Skoulakis, E.M.C.; Rouault, T.A. Homeostatic Mechanisms for Iron Storage Revealed by Genetic Manipulations and Live Imaging of Drosophila Ferritin. Genetics 2007, 177, 89–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimitrijevic, Z.M.; Salinger-Martinovic, S.S.; Jankovic, R.J.; Mitic, B.P. Elevated Serum Ferritin Levels Are Predictive of Renal Function Recovery among Patients with Acute Kidney Injury. Tohoku J. Exp. Med. 2019, 248, 63–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.-H.; Shang, J.; Yan, L.; Wei, T.; Xiang, L.; Wang, H.-L.; Cheng, J.; Xiao, G. Oxidative Stress Caused by Lead (Pb) Induces Iron Deficiency in Drosophila melanogaster. Chemosphere 2020, 243, 125428. [Google Scholar] [CrossRef] [PubMed]
- Pead, S.; Durrant, E.; Webb, B.; Larsen, C.; Heaton, D.; Johnson, J.; Watt, G.D. Metal Ion Binding to Apo, Holo, and Reconstituted Horse Spleen Ferritin. J. Inorg. Biochem. 1995, 59, 15–27. [Google Scholar] [CrossRef]
- Vásquez-Procopio, J.; Osorio, B.; Cortés-Martínez, L.; Hernández-Hernández, F.; Medina-Contreras, O.; Ríos-Castro, E.; Comjean, A.; Li, F.; Hu, Y.; Mohr, S.; et al. Intestinal Response to Dietary Manganese Depletion in Drosophila. Metallomics 2020, 12, 218–240. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, H.; Hong, H.; Wang, W.; Liu, F. Can Zinc Protect Cells from the Cytotoxic Effects of Cobalt Ions and Nanoparticles Derived from Metal-on-Metal Joint Arthroplasties? Bone Jt. Res. 2017, 6, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Marelja, Z.; Leimkühler, S.; Missirlis, F. Iron Sulfur and Molybdenum Cofactor Enzymes Regulate the Drosophila Life Cycle by Controlling Cell Metabolism. Front. Physiol. 2018, 9, 50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bahadorani, S.; Hilliker, A.J. Biological and Behavioral Effects of Heavy Metals in Drosophila melanogaster Adults and Larvae. J. Insect Behav. 2009, 22, 399–411. [Google Scholar] [CrossRef]



| System | Gene | Interactions with Metal Ions | References |
|---|---|---|---|
| General Metal Response System | dMTF-1 | Cu, Zn, Fe, and Cd | [35,42,48,50,51,58,59,60] |
| MtnA-F | Cu, Zn, Fe, Cd, Pb, and Hg | [35,40,50,52,54,64,69,70,78,81,84] | |
| Copper Transport System | Ctr1B | Cu, Cd, and Hg | [64] |
| SOD1 | |||
| Zinc Transport System | Catsup | Zn and Fe | [38] |
| dZnt35C | Zn and Cd | [118] | |
| Iron Transport System | Malvolio | Fe, Cu, Cd, and Pb | [124,143] |
| Ferritin | Fe, Cd, and Pb | [142,143,144] | |
| dZip13 | Fe, Zn, Cd, and Pb | [115,143] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slobodian, M.R.; Petahtegoose, J.D.; Wallis, A.L.; Levesque, D.C.; Merritt, T.J.S. The Effects of Essential and Non-Essential Metal Toxicity in the Drosophila melanogaster Insect Model: A Review. Toxics 2021, 9, 269. https://doi.org/10.3390/toxics9100269
Slobodian MR, Petahtegoose JD, Wallis AL, Levesque DC, Merritt TJS. The Effects of Essential and Non-Essential Metal Toxicity in the Drosophila melanogaster Insect Model: A Review. Toxics. 2021; 9(10):269. https://doi.org/10.3390/toxics9100269
Chicago/Turabian StyleSlobodian, Mitchell R., Jesse D. Petahtegoose, Athena L. Wallis, Danica C. Levesque, and Thomas J. S. Merritt. 2021. "The Effects of Essential and Non-Essential Metal Toxicity in the Drosophila melanogaster Insect Model: A Review" Toxics 9, no. 10: 269. https://doi.org/10.3390/toxics9100269






