RETRACTED: Biosynthesized Iron Oxide Nanoparticles from Petroselinum crispum Leaf Extract Mitigate Lead-Acetate-Induced Anemia in Male Albino Rats: Hematological, Biochemical and Histopathological Features
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Plant Extraction
2.2. Preparation of Chemical and Natural Magnetic Iron Oxide Nanoparticles
2.3. Experimental Design
2.4. Collection of Blood and Serum Samples
2.5. Hematological Analysis
2.6. Serum Biochemical Analysis
2.7. Histopathological Analysis
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Van Hove, L.; Schisano, T.; Brace, L. Anemia diagnosis, classification and monitoring using Cell-Dyn technology reviewed for the new millennium. Lab. Hematol. 2000, 6, 93–108. [Google Scholar]
- Tuormaa, T.E. The adverse effects of lead. J. Orthomol. Med. 1995, 10, 149–164. [Google Scholar] [CrossRef]
- Markowitz, M. Lead poisoning. Pediatr. Rev. 2000, 21, 327–335. [Google Scholar] [CrossRef]
- Hershko, C. Lead poisoning by contaminated flour: An unfinished story. Harefuah 2005, 144, 458–462, 528. [Google Scholar]
- Kalia, K.; Flora, S.J. Strategies for safe and effective therapeutic measures for chronic arsenic and lead poisoning. J. Occup. Health 2005, 47, 1–21. [Google Scholar] [CrossRef]
- Casas, J.S.; Sordo, J. (Eds.) Chemistry, analytical aspects, environmental impact and health effects. In Lead; Elsevier: Amsterdam, The Netherlands, 2006; p. 366. [Google Scholar] [CrossRef]
- WHO. Lead Poisoning and Health; World Health Organization (WHO): Geneva, Switzerland, 2019. [Google Scholar]
- Pitot, C.H.; Dragan, P.Y. Chemical carcinogenesis. In Casarett and Doull’s Toxicology: The Basic Science of Poisons, 5th ed.; McGraw-Hill: New York, NY, USA, 1996; pp. 201–260. [Google Scholar]
- Lanphear, B.P.; Dietrich, K.; Auinger, P.; Cox, C. Cognitive deficits associated with blood lead concentrations <10 microg/dL in US children and adolescents. Public Health Rep. 2000, 115, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Klauder, D.S.; Petering, H.G. Anemia of lead intoxication: A role for copper. J. Nutr. 1977, 107, 1779–1785. [Google Scholar] [CrossRef]
- Chmielnicka, J.; Zarȩba, G.; Nasiadek, M. Combined effect of tin and lead on heme biosynthesis in rats. Ecotoxicol. Environ. Saf. 1994, 29, 165–173. [Google Scholar] [CrossRef]
- Peter, S.S.; Herbert, H.S.; Albert, C.L. Experimental and Clinical Neurotoxicology, 2nd ed.; Oxford University Press: New York, NY, USA, 2000. [Google Scholar]
- Mugahi, M.N.; Heidari, Z.; Sagheb, H.M.; Barbarestani, M. Effects of chronic lead acetate intoxication on blood indices of male adult rat. DARU J. Pharm. Sci. 2003, 11, 147–151. [Google Scholar]
- Smith, A. Prescribing iron. Prescr. J. 1997, 37, 82–87. [Google Scholar]
- Wu, H.; Zhu, S.; Zeng, M.; Liu, Z.; Dong, S.; Zhao, Y.; Huang, H.; Lo, Y.M. Enhancement of non-heme iron absorption by anchovy (engraulis japonicus) muscle protein hydrolysate involves a nanoparticle-mediated mechanism. J. Agric. Food Chem. 2014, 62, 8632–8639. [Google Scholar] [CrossRef] [PubMed]
- De Jong, W.H.; Borm, P.J.A. Drug delivery and nanoparticles:applications and hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, Q.; Scotter, M.; Blackburn, J.; Ross, B.; Boxall, A.; Castle, L.; Aitken, R.; Watkins, R. Applications and implications of nanotechnologies for the food sector. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2008, 25, 241–258. [Google Scholar] [CrossRef]
- Erfanian, A.; Mirhosseini, H.; Rasti, B.; Hair-Bejo, M.; Bin Mustafa, S.; Abd Manap, M.Y. Absorption and bioavailability of nano-size reduced calcium citrate fortified milk powder in ovariectomized and ovariectomized-osteoporosis rats. J. Agric. Food Chem. 2015, 63, 5795–5804. [Google Scholar] [CrossRef] [PubMed]
- Sastry, M.; Ahmad, A.; Khan, M.I.; Kumar, R. Microbial nanoparticle production. In Nanobiotechnology; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2004; Chapter 9; pp. 126–135. [Google Scholar] [CrossRef]
- Mohanpuria, P.; Rana, N.K.; Yadav, S.K. Biosynthesis of nanoparticles: Technological concepts and future applications. J. Nanopart. Res. 2008, 10, 507–517. [Google Scholar] [CrossRef]
- Mittal, A.K.; Chisti, Y.; Banerjee, U.C. Synthesis of metallic nanoparticles using plant extracts. Biotechnol. Adv. 2013, 31, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Shahwan, T.; Abu Sirriah, S.; Nairat, M.; Boyacı, E.; Eroğlu, A.E.; Scott, T.B.; Hallam, K.R. Green synthesis of iron nanoparticles and their application as a Fenton-like catalyst for the degradation of aqueous cationic and anionic dyes. Chem. Eng. J. 2011, 172, 258–266. [Google Scholar] [CrossRef]
- Virkutyte, J.; Varma, R.S. CHAPTER 2 Environmentally friendly preparation of metal nanoparticles. In Sustainable Preparation of Metal Nanoparticles: Methods and Applications; The Royal Society of Chemistry, Thomas Graham House: Cambridge, UK, 2013; pp. 7–33. [Google Scholar] [CrossRef]
- Njagi, E.C.; Huang, H.; Stafford, L.; Genuino, H.; Galindo, H.M.; Collins, J.B.; Hoag, G.E.; Suib, S.L. Biosynthesis of iron and silver nanoparticles at room temperature using aqueous sorghum bran extracts. Langmuir 2011, 27, 264–271. [Google Scholar] [CrossRef]
- Bishnoi, S.; Kumar, A.; Selvaraj, R. Facile synthesis of magnetic iron oxide nanoparticles using inedible Cynometra ramiflora fruit extract waste and their photocatalytic degradation of methylene blue dye. Mater. Res. Bull. 2018, 97, 121–127. [Google Scholar] [CrossRef]
- Kamaraj, M.; Kidane, T.; Muluken, K.U.; Aravind, J. Biofabrication of iron oxide nanoparticles as a potential photocatalyst for dye degradation with antimicrobial activity. Int. J. Environ. Sci. Technol. Tehran 2019, 16, 8305–8314. [Google Scholar] [CrossRef]
- Bibi, I.; Nazar, N.; Ata, S.; Sultan, M.; Ali, A.; Abbas, A.; Jilani, K.; Kamal, S.; Sarim, F.M.; Khan, M.I.; et al. Green synthesis of iron oxide nanoparticles using pomegranate seeds extract and photocatalytic activity evaluation for the degradation of textile dye. J. Mater. Res. Technol. 2019, 8, 6115–6124. [Google Scholar] [CrossRef]
- Karpagavinayagam, P.; Vedhi, C. Green synthesis of iron oxide nanoparticles using Avicennia marina flower extract. Vacuum 2019, 160, 286–292. [Google Scholar] [CrossRef]
- Al-Daraji, H.J.; Al-Mashadani, H.; Mirza, H.; Al-Hassani, A.; Al-Hayani, W. The effect of utilization of parsley (Petroselinum crispum) in local Iraqi geese diets on blood biochemistry. J. Am. Sci. 2012, 8, 427–432. [Google Scholar]
- Shan, B.; Cai, Y.Z.; Sun, M.; Corke, H. Antioxidant capacity of 26 spice extracts and characterization of their phenolic constituents. J. Agric. Food Chem. 2005, 53, 7749–7759. [Google Scholar] [CrossRef]
- Davey, M.W.; Bauw, G.; Montagu, M.V. Analysis of ascorbate in plant tissues by high-performance capillary zone electrophoresis. Anal. Biochem. 1996, 239, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Chenard, C.H.; Kopsell, D.A.; Kopsell, D.E. Nitrogen concentration affects nutrient and carotenoid accumulation in parsley. J. Plant Nut. 2005, 28, 285–297. [Google Scholar] [CrossRef]
- Stan, M.; Popa, A.; Toloman, D.; Silipas, T.-D.; Vodnar, D.C.; Katona, G. Enhanced antibacterial activity of zinc oxide nanoparticles synthesized using Petroselinum crispum extracts. AIP Conf. Proc. 2015, 1700, 060004. [Google Scholar] [CrossRef]
- Maity, D.; Agrawal, D.C. Synthesis of iron oxide nanoparticles under oxidizing environment and their stabilization in aqueous and non-aqueous media. J. Magn. Magn. Mater. 2007, 308, 46–55. [Google Scholar] [CrossRef]
- Xu, J.; Sun, J.; Wang, Y.; Sheng, J.; Wang, F.; Sun, M. Application of iron magnetic nanoparticles in protein immobilization. Molecules 2014, 19, 11465–11486. [Google Scholar] [CrossRef]
- Ajinkya, N.; Yu, X.; Kaithal, P.; Luo, H.; Somani, P.; Ramakrishna, S. Magnetic Iron Oxide Nanoparticle (IONP) Synthesis to Applications: Present and Future. Materials 2020, 13, 4644. [Google Scholar] [CrossRef]
- Kubde, M.S.; Khadabadi, S.; Farooqui, I.; Deore, S. Lagenaria siceraria: Phytochemistry, pharmacognosy and pharmacological studies. Rep. Opin. 2010, 2, 91–98. [Google Scholar]
- Okediran, B.; Biobaku, K.; Olaifa, F.H.; Atata, A. Haematological and antioxidant enzyme response to lead toxicity in male Wistar rats. Ceylon J. Sci. 2017, 46, 31. [Google Scholar] [CrossRef]
- Salaheldin, T.; Regheb, E. In-Vivo nutritional and toxicological evaluation of nano iron fortified biscuits as food supplement for iron deficient anemia. J. Nanomed. Res. 2016, 3, 00049. [Google Scholar] [CrossRef]
- Reitman, S.; Frankel, S. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Patton, C.J.; Crouch, S.R. Spectrophotometric and kinetics investigation of the Berthelot reaction for the determination of ammonia. Anal. Chem. 1977, 49, 464–469. [Google Scholar] [CrossRef]
- Jaffé, M. Ueber den niederschlag, welchen pikrinsäure in normalem harn erzeugt und über eine neue reaction des kreatinins. Biol. Chem. 1886, 10, 391–400. [Google Scholar] [CrossRef]
- Linpisarn, S.; Kricka, L.J.; Kennedy, J.H.; Whitehead, T.P. Sensitive sandwich enzyme immunoassay for serum ferritin on microtitre plates. Ann. Clin. Biochem. 1981, 18, 48–53. [Google Scholar] [CrossRef]
- Ceriotti, F.; Ceriotti, G. Improved direct specific determination of serum iron and total iron-binding capacity. Clin. Chem. 1980, 26, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Fischl, J.; Cohen, S. A simple colorimetric method for the determination of total iron-binding-capacity of serum. Clin. Chim. Acta 1962, 7, 121–123. [Google Scholar] [CrossRef]
- Bancroft, J.D.; Gamble, M. Theory and Practice of Histological Techniques; Churchill Livingstone: London, UK; New York, NY, USA, 2002. [Google Scholar]
- Rashid, H.; Mansoor, M.A.; Haider, B.; Nasir, R.; Abd Hamid, S.B.; Abdulrahman, A. Synthesis and characterization of magnetite nano particles with high selectivity using in-situ precipitation method. Sep. Sci. Technol. 2020, 55, 1207–1215. [Google Scholar] [CrossRef]
- Prasad, C.; Gangadhara, S.; Venkateswarlu, P. Bio-inspired green synthesis of Fe3O4 magnetic nanoparticles using watermelon rinds and their catalytic activity. Appl. Nanosci. 2016, 6, 797–802. [Google Scholar] [CrossRef]
- Fahmy, H.M.; Mohamed, F.M.; Marzouq, M.H.; Mustafa, A.B.E.-D.; Alsoudi, A.M.; Ali, O.A.; Mohamed, M.A.; Mahmoud, F.A. Review of green methods of iron nanoparticles synthesis and applications. BioNanoScience 2018, 8, 491–503. [Google Scholar] [CrossRef]
- Demirezen, D.A.; Yilmaz, D.; Yılmaz, Ş. Green synthesis and characterization of iron nanoparticles using Aesculus hippocastanum seed extract. Int. J. Adv. Sci. Eng. Technol. 2018, 6, 2321–8991. [Google Scholar]
- Shi, S.; Jia, J.; Guo, X.; Zhao, Y.; Liu, B.; Chen, D.; Guo, Y.; Zhang, X. Toxicity of iron oxide nanoparticles against osteoblasts. J. Nanoparticle Res. 2012, 14, 1–11. [Google Scholar] [CrossRef]
- Chandrasekar, N.; Kumar, K.; Balasubramnian, K.S.; Karunamurthy, K.; Varadharajan, R. Facile synthesis of iron oxide, iron-cobalt and zero valent iron nanoparticles and evaluation of their anti microbial activity, free radicle scavenginging activity and antioxidant assay. Dig. J. Nanomater. Bios. 2013, 8, 765–775. [Google Scholar]
- Sankaralingam, K.; Kadirvelu, K. Green synthesis of Iron oxide nanoparticles using Lagenaria siceraria and evaluation of its Antimicrobial activity. Def. Life Sci. J. 2017, 2, 422. [Google Scholar] [CrossRef]
- Kavaz, D.; Aİgbe, R.; Augustine, E.E. Detection of pathogens in aqueous media with modified magnetic nanoscaled particles. Fresenius Environ. Bull. 2017, 26, 761–771. [Google Scholar]
- Offor, S.J.; Mbagwu, H.O.C.; Orisakwe, O.E. Lead induced hepato-renal damage in male albino rats and effects of activated charcoal. Front. Pharmacol. 2017, 8, 107. [Google Scholar] [CrossRef]
- Elayat, W.; Bakheetf, M. Effects of chronic lead toxicity on liver and kidney functions. J. Med. Lab. Sci. 2010, 1, 29–36. [Google Scholar]
- Shafie, E.H.; Keshavarz, S.A.; Kefayati, M.E.; Taheri, F.; Sarbakhsh, P.; Vafa, M.R. The effects of nanoparticles containing iron on blood and inflammatory markers in comparison to ferrous sulfate in anemic rats. Int. J. Prev. Med. 2016, 7, 7–17. [Google Scholar] [CrossRef]
- Zariwala, M.G.; Elsaid, N.; Jackson, T.L.; Corral López, F.; Farnaud, S.; Somavarapu, S.; Renshaw, D. A novel approach to oral iron delivery using ferrous sulphate loaded solid lipid nanoparticles. Int. J. Pharm. 2013, 456, 400–407. [Google Scholar] [CrossRef]
- Elshemy, M.A. Iron oxide nanoparticles versus ferrous sulfate in treatment of iron deficiency anemia in rats. Egypt. J. Vet. Sci. 2018, 49, 103–109. [Google Scholar] [CrossRef]
- Rey Vázquez, G.; Guerrero, G.A. Characterization of blood cells and hematological parameters in Cichlasoma dimerus (Teleostei, Perciformes). Tissue Cell 2007, 39, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Piomelli, S. Childhood lead poisoning. Pediatr. Clin. N. Am. 2002, 49, 1285–1304. [Google Scholar] [CrossRef]
- Hu, L.; Tang, X.; Cui, F. Solid lipid nanoparticles (SLNs) to improve oral bioavailability of poorly soluble drugs. J. Pharm. Pharmacol. 2004, 56, 1527–1535. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xie, P.; Li, D.; Shi, Z. Hematological and plasma biochemical responses of crucian carp (Carassius auratus) to intraperitoneal injection of extracted microcystins with the possible mechanisms of anemia. Toxicon 2007, 49, 1150–1157. [Google Scholar] [CrossRef]
- Suradkar, S.; Ghodasara, D.; Vihol, P.; Patel, J.; Jaiswal, V.; Prajapati, K. Haemato-Biochemical Alterations induced by lead acetate toxicity in Wistar Rats. Vet. World 2009, 2, 429–431. [Google Scholar]
- Aly, M.H.; Kim, H.C.; Renner, S.W.; Boyarsky, A.; Kosmin, M.; Paglia, D.E. Hemolytic anemia associated with lead poisoning from shotgun pellets and the response to succimer treatment. Am. J. Hematol. 1993, 44, 280–283. [Google Scholar] [CrossRef]
- Klaassen, C.D. Casarett and Doull’s Toxicology: The Basic Science of Poisons, 6th ed.; McGraw-Hill: New York, NY, USA, 2001. [Google Scholar]
- Ambali, S.F.; Ayo, J.O.; Esievo, K.A.N.; Ojo, S.A. Hemotoxicity induced by chronic chlorpyrifos exposure in wistar rats: Mitigating effect of vitamin C. Vet. Med. Int. 2011, 2011, 945439. [Google Scholar] [CrossRef]
- Yagminas, A.; Franklin, C.; Villeneuve, D.; Gilman, A.; Little, P.; Valli, V. Subchronic oral toxicity of triethyl lead in the male weanling rat. Clinical, biochemical, hematological, and histopathological effects. Toxicol. Sci. 1990, 15, 580–596. [Google Scholar] [CrossRef]
- Dai, J.; Nagai, T.; Wang, X.; Zhang, T.; Meng, M.; Zhang, Q. pH-sensitive nanoparticles for improving the oral bioavailability of cyclosporine A. Int. J. Pharm. 2004, 280, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Hashem, F.; Nasr, M.; Ahmed, Y. Preparation and evaluation of iron oxide nanoparticles for treatment of iron deficiency anemia. Int. J. Pharm. Pharmaceut. Sci. 2018, 10, 142–146. [Google Scholar] [CrossRef]
- Abdou, Z.; Attia, M.; Raafat, M. Protective effect of citric acid and thiol compounds against cadmium and lead toxicity in experimental animals. J. Biol. Chem. Environ. Sci. 2007, 2, 481–497. [Google Scholar]
- Ibrahim, N.M.; Eweis, E.A.; El-Beltagi, H.S.; Abdel-Mobdy, Y.E. Effect of lead acetate toxicity on experimental male albino rat. Asian Pac. J. Trop. Biomed. 2012, 2, 41–46. [Google Scholar] [CrossRef]
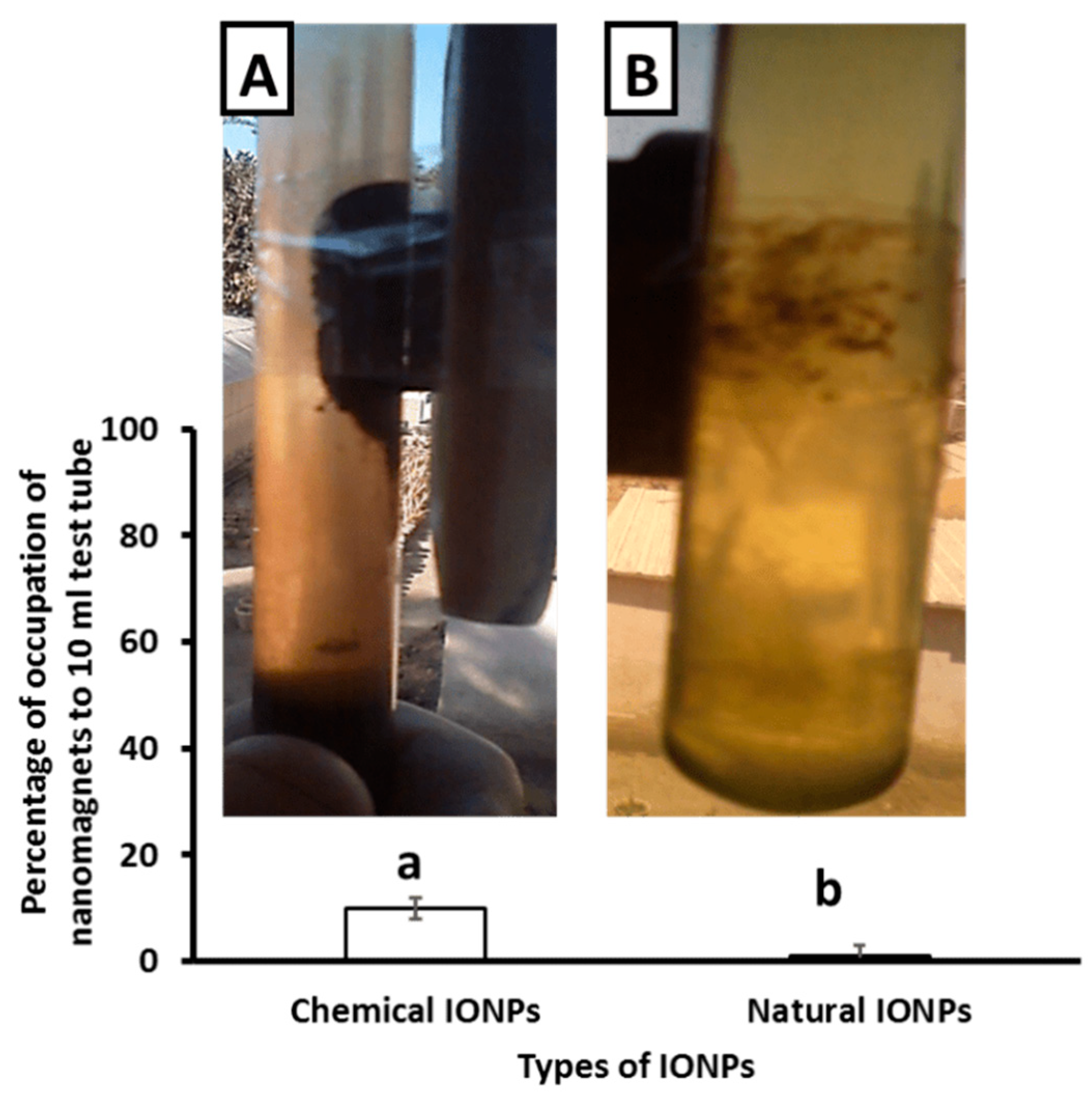
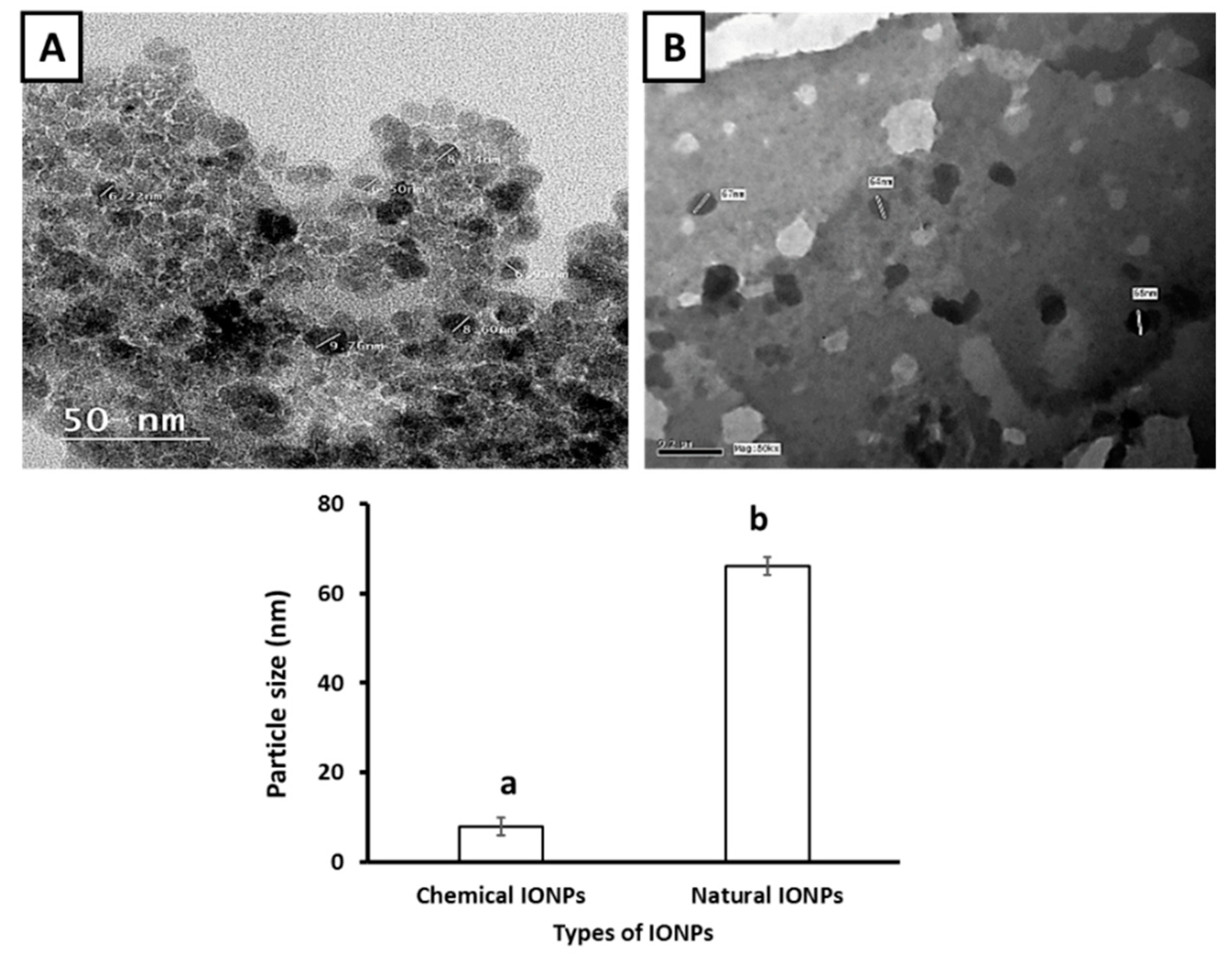

| Groups | Ferritin (ng/dL) | Iron (µg/dL) | TIBC (mcg/dL) |
|---|---|---|---|
| 1 | 0.70 ± 1.5 a | 2.31 ± 1.8 a | 6.01 ± 1.4 b |
| 2 | 0.47 ± 0.2 b | 1.29 ± 0.1 c | 8.53 ± 0.2 a |
| 3 | 0.50 ± 0.1 b | 1.89 ± 0.4 b | 6.26 ± 0.7 b |
| 4 | 0.70 ± 0.3 a | 1.64 ± 0.2 b | 6.96 ± 0.3 b |
| Groups | Hb (mg/dl) | RBCs (×10⁶ µL) | PCV (%) | MCV (fl/cell) | MCH (pg/cell) | MCHC (g/dL) |
|---|---|---|---|---|---|---|
| 1 | 13.8 ± 2.7 a | 6.07 ± 1.6 ab | 36.6 ± 5.5 a | 60.5 ± 3.5 a | 20.43 ± 1.2 a | 33.9 ± 3.1 a |
| 2 | 8.73 ± 2.36 c | 5.48 ± 1.2 c | 28.3 ± 3.4 b | 51.7 ± 1.6 b | 19.07 ± 1.5 b | 30.7 ± 1.5 b |
| 3 | 11.7 ± 1.3 b | 6.09 ± 1.7 ab | 36.83 ± 1.8 a | 60.8 ± 1.5 a | 20.8 ± 1.0 a | 32.67 ± 1.3 a |
| 4 | 12.66 ± 2.5 a | 6.57 ± 1.5 a | 39.53 ± 2.0 a | 60.13 ± 2.8 a | 20.73 ± 1.1 a | 32.5 ± 0.3 a |
| Groups | Urea (mg/dL) | Creatinine (mg/dL) | AST (U/mL) | ALT (U/mL) |
|---|---|---|---|---|
| 1 | 24.89 ± 4.4 d | 0.71 ± 0.1 c | 64.1 ± 6.1 d | 27.9 ± 3.4 d |
| 2 | 125.87 ± 5.1 a | 3.21 ± 0.9 a | 145.7 ± 6.4 a | 60.00 ± 1.7 a |
| 3 | 55.50 ± 3.3 b | 1.61 ± 0.3 b | 111.9 ± 8.8 b | 44.67 ± 3.1 b |
| 4 | 42.98 ± 6.1 c | 0.97 ± 0.3 c | 84.62 ± 9.1 c | 37.34 ± 3.0 c |
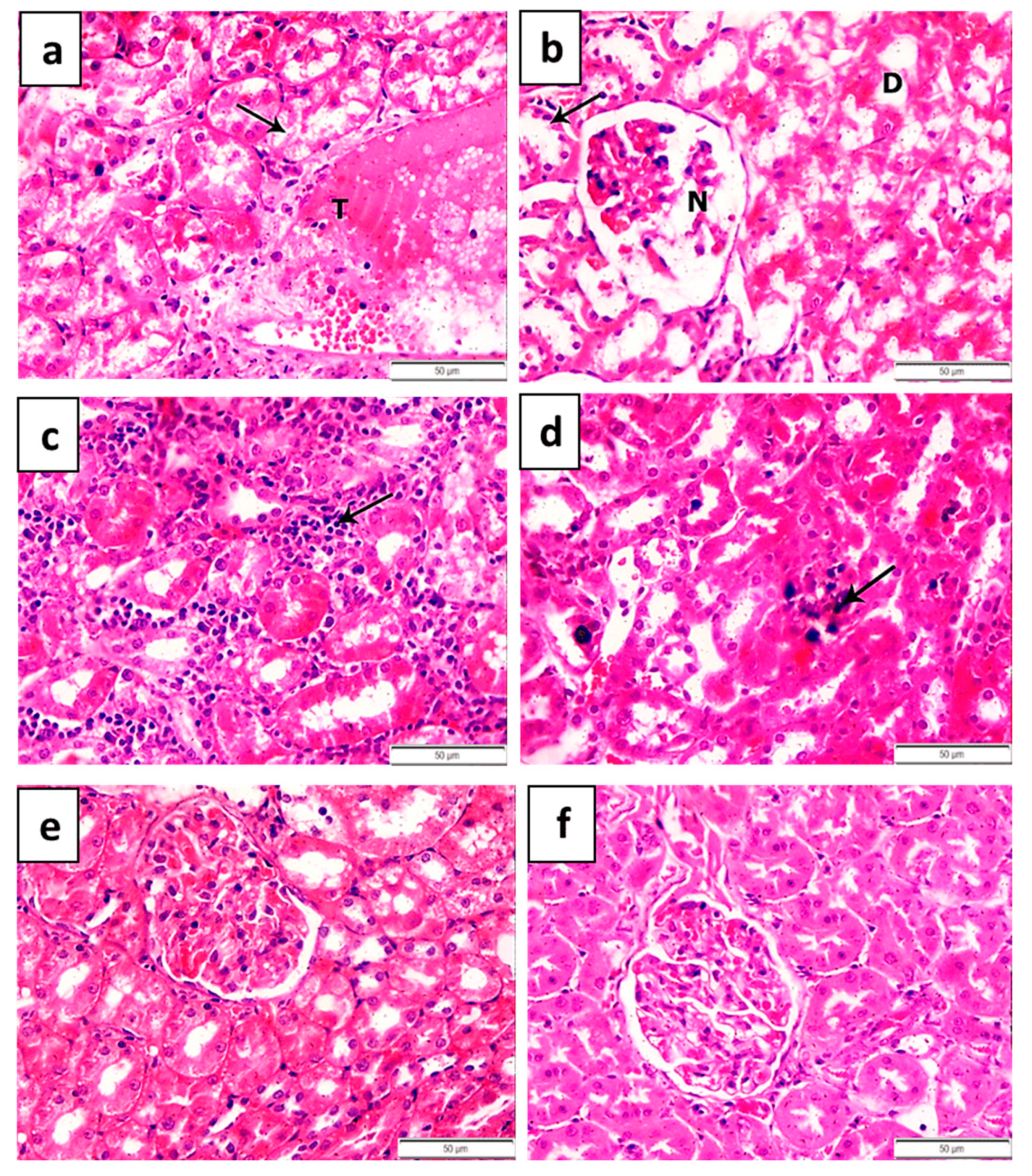
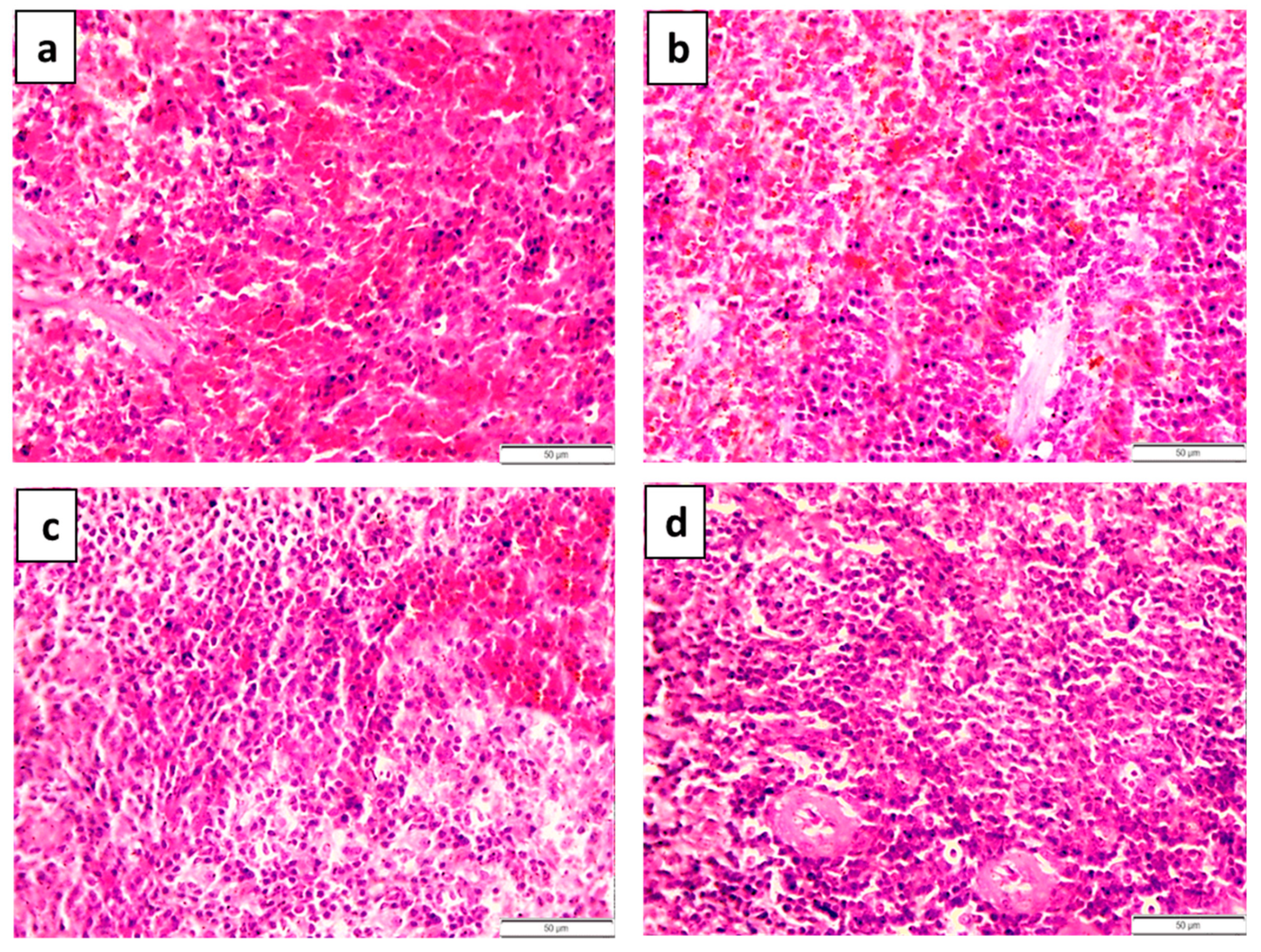
| Groups | Number | Pathological Grading | |||
|---|---|---|---|---|---|
| 0 | I | II | III | ||
| 1 | 5 | 5 | 0 | 0 | 0 |
| 2 | 5 | 0 | 1 | 1 | 3 |
| 3 | 5 | 0 | 1 | 3 | 1 |
| 4 | 5 | 3 | 1 | 1 | 0 |
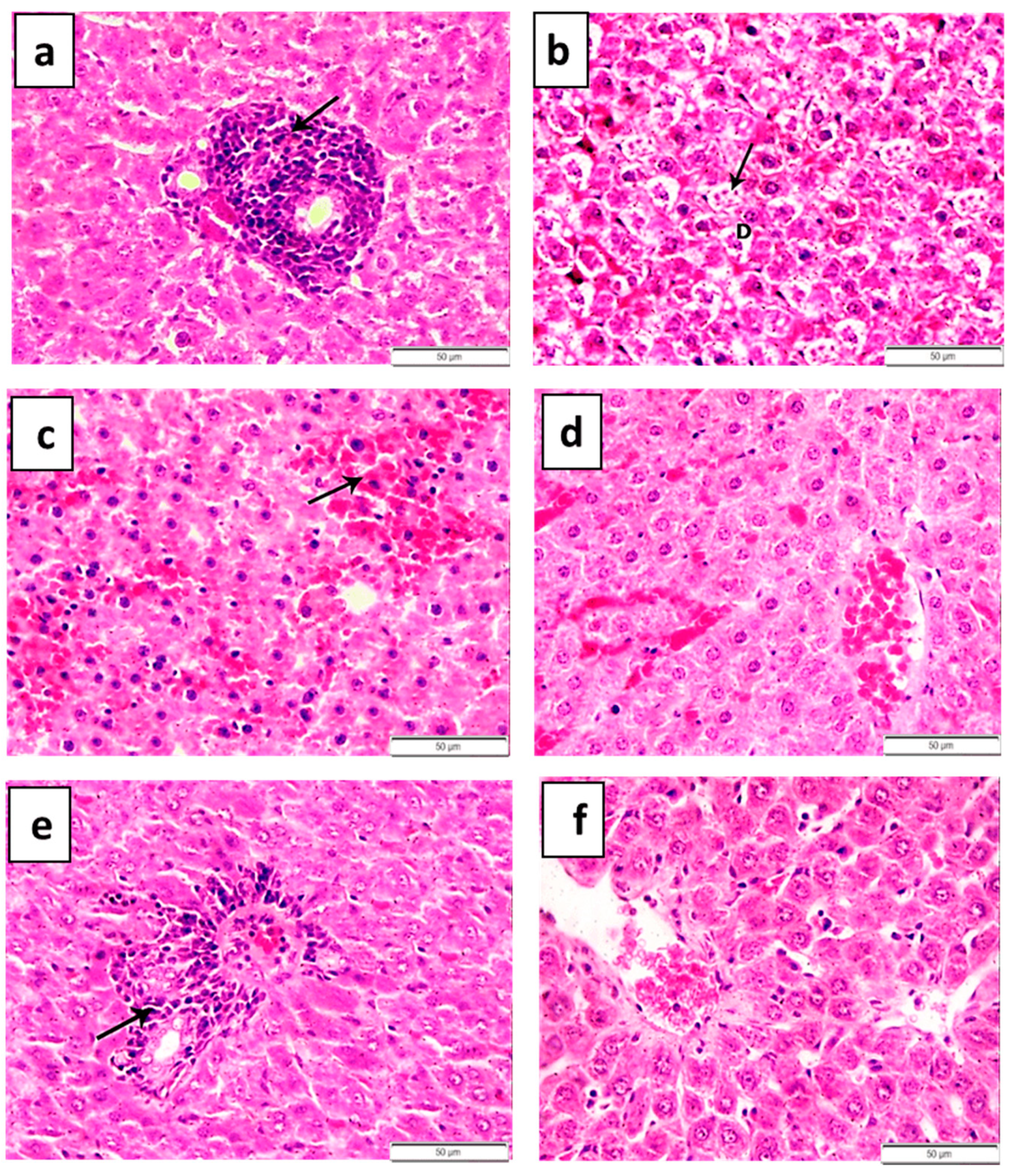
| Groups | Number | Pathological Grading | |||
|---|---|---|---|---|---|
| 0 | I | II | III | ||
| 1 | 5 | 5 | 0 | 0 | 0 |
| 2 | 5 | 0 | 1 | 1 | 3 |
| 3 | 5 | 0 | 2 | 3 | 0 |
| 4 | 5 | 2 | 2 | 1 | 0 |
| Groups | Number | Pathological Grading | |||
|---|---|---|---|---|---|
| 0 | I | II | III | ||
| 1 | 5 | 5 | 0 | 0 | 0 |
| 2 | 5 | 0 | 1 | 2 | 2 |
| 3 | 5 | 0 | 2 | 3 | 0 |
| 4 | 5 | 3 | 1 | 1 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Bahr, S.M.; Elbakery, A.M.; El-Gazzar, N.; Amin, A.A.; Al-Sultan, S.; Alfattah, M.A.; Shousha, S.; Alhojaily, S.; Shathele, M.; Sabeq, I.I.; et al. RETRACTED: Biosynthesized Iron Oxide Nanoparticles from Petroselinum crispum Leaf Extract Mitigate Lead-Acetate-Induced Anemia in Male Albino Rats: Hematological, Biochemical and Histopathological Features. Toxics 2021, 9, 123. https://doi.org/10.3390/toxics9060123
El-Bahr SM, Elbakery AM, El-Gazzar N, Amin AA, Al-Sultan S, Alfattah MA, Shousha S, Alhojaily S, Shathele M, Sabeq II, et al. RETRACTED: Biosynthesized Iron Oxide Nanoparticles from Petroselinum crispum Leaf Extract Mitigate Lead-Acetate-Induced Anemia in Male Albino Rats: Hematological, Biochemical and Histopathological Features. Toxics. 2021; 9(6):123. https://doi.org/10.3390/toxics9060123
Chicago/Turabian StyleEl-Bahr, Sabry M., Amal M. Elbakery, Nashwa El-Gazzar, Aziza A. Amin, Saad Al-Sultan, Mohammed A. Alfattah, Saad Shousha, Sameer Alhojaily, Mohammad Shathele, Islam I. Sabeq, and et al. 2021. "RETRACTED: Biosynthesized Iron Oxide Nanoparticles from Petroselinum crispum Leaf Extract Mitigate Lead-Acetate-Induced Anemia in Male Albino Rats: Hematological, Biochemical and Histopathological Features" Toxics 9, no. 6: 123. https://doi.org/10.3390/toxics9060123
APA StyleEl-Bahr, S. M., Elbakery, A. M., El-Gazzar, N., Amin, A. A., Al-Sultan, S., Alfattah, M. A., Shousha, S., Alhojaily, S., Shathele, M., Sabeq, I. I., & Hamouda, A. F. (2021). RETRACTED: Biosynthesized Iron Oxide Nanoparticles from Petroselinum crispum Leaf Extract Mitigate Lead-Acetate-Induced Anemia in Male Albino Rats: Hematological, Biochemical and Histopathological Features. Toxics, 9(6), 123. https://doi.org/10.3390/toxics9060123






