Abstract
Bioethanol from sugarcane bagasse is a promising second-generation biofuel due to its abundance as a sugar industry by-product. Herein, enzymatic hydrolysate obtained from sugarcane bagasse pretreated with optimized hydrothermal alkaline sulfite (HAS) was evaluated for its fermentability using Saccharomyces cerevisiae PE-2 and Scheffersomyces stipitis CBS 5773. The HAS pretreatment achieved a high delignification rate (63%), resulting in a cellulose- and hemicellulose-enriched substrate (55% and 27%, respectively). While the cellulose content remained relatively constant, hemicellulose content was reduced by 25%, with significant removal of acetyl groups (80%) and arabinan groups (39%). The pretreated bagasse exhibited high digestibility, applying 10 FPU (filter paper unit) cellulase together with 10 CBU (cellobiose unit) β-glucosidase per gram of dry bagasse in the hydrolysis step, yielding 72% glucan and 66% xylan conversion within 72 h. The resulting hydrolysate was efficiently fermented by S. cerevisiae and S. stipitis, achieving ethanol yields of 0.51 and 0.43 g/g of sugars, respectively. The fermentation kinetics were comparable to those observed in a synthetic medium containing pure sugars, demonstrating the effectiveness of HAS pretreatment in generating readily fermentable, carbohydrate-rich substrates. HAS pretreatment enabled improved conversion of sugarcane bagasse into fermentation-ready sugars, constituting a potential resource for bioethanol synthesis applying both S. cerevisiae and S. stipitis in the future.
1. Introduction
Bioethanol from sugarcane bagasse has been singled out among the leading candidates for second-generation biofuels with the potential to replace fossil fuels given bagasse’s large availability as a by-product of the sugarcane industry [1,2,3]. However, obtaining fermentable sugars from sugarcane bagasse at a viable yield is still technically problematic because of its recalcitrance which prevents its enzymatic digestibility unless it undergoes an appropriate pretreatment process [4,5,6,7]. Besides that, for profitability, cost-competitiveness, and sustainability in a biorefinery context, an integral conversion of all sugars is indispensable. Thus, the xylan in sugarcane bagasse is an issue, as its monomeric sugars are not metabolized by wild-type Saccharomyces cerevisiae strains, the most widely employed microorganism for ethanol generation [8,9,10,11]. In this context, xylose-utilizing pathways have been incorporated into S. cerevisiae by metabolic engineering to obtain microorganisms capable of integral fermentation of lignocellulosic hydrolysate [12,13,14,15]. However, the low stability of these genetically modified microorganisms, together with ethical issues, has been hindering its industrial application [16,17]. In order to avoid such issues, several wild species of fungi and bacteria have been exploited to ferment lignocellulosic hydrolysates, comprising both five-carbon (pentoses) and six-carbon (hexoses) sugars [18,19].
The yeast, Scheffersomyces stipitis (formerly Pichia stipitis), commonly recognized for its capacity to ferment five-carbon sugars, is regarded as highly suitable for industrial implementation because of its ability to achieve high ethanol yields [20]. Beyond its well-documented ability to ferment xylose and glucose, S. stipitis has also been shown to utilize cellobiose, galactose, and mannose, as well as mannan- and xylan-derived oligomers [21]. However, the fermentability of the enzymatic hydrolysate is strongly influenced by the presence of microbial inhibitors formed during lignocellulosic pretreatment, particularly aliphatic acids, furan aldehydes, and aromatic compounds [22,23]. Hence, the suitability of any pretreatment aiming to improve lignocellulosic ethanol production should also be evaluated by the fermentability of its enzymatic hydrolysate by traditionally used microorganisms.
In our previous work [24], we proposed hydrothermal alkaline sulfite (HAS) pretreatment as a new approach to fractionate sugarcane bagasse and maximize the fermentable sugar recovery by enzymatic hydrolysis. The major advantage of HAS pretreatment is its ability to efficiently and selectively solubilize lignin from sugarcane bagasse while preserving carbohydrates, thereby enhancing enzymatic hydrolysis and producing fewer fermentation inhibitors compared to traditional pretreatment methods [9]. The most used traditional methods like dilute acid pretreatment and steam explosion require high energy and cause sugar degradation and inhibitors formation, while alkaline sulfite enables efficient delignification. The hydrosulfite and hydroxide generated in the aqueous media lead to phenolic groups deprotonation and forming of intermediates that undergo sulfonation and cleavage, dissolving lignin and reducing inhibitor formation [25], which is expected to positively impact the fermentability of the hydrolyzed product.
Herein, we investigated the fermentation potential of enzymatically digested sugarcane bagasse subjected to optimized HAS pretreatment conditions (10% sulfite, 150 °C, and 45 min) employing S. cerevisiae PE-2 and S. stipitis CBS 5773.
2. Materials and Methods
2.1. Reagents and Materials
Analytical-grade chemicals, including sodium hydroxide, sodium sulfite, and sulfuric acid, were obtained from Vetec (Duque de Caxias, Brazil) and Sigma-Aldrich Brazil (São Paulo, Brazil). Glass fiber filters (GF-3, 0.45 μm), Millex-HV membrane cartridges (0.45 μm), and Sep-Pak C18 cartridges (Waters) were purchased from Carvalhaes (São Paulo, Brazil). Celluclast 1.5 L (cellulase) and Novozym 188 (β-glucosidase) used in the enzymatic hydrolysis were kindly supplied by Embrapa Agroenergia (Brasília, Brazil).
2.2. Biomass and Pretreatment
The sugarcane bagasse employed for the experiments was kindly supplied by Sugar Mill FURLAN S/A (Santa Bárbara D’Oeste, SP, Brazil). The thoroughly mixed starting bagasse (≅50% moisture) was air-dried to 10–15% moisture. The hydrothermal pretreatment processes were conducted in a 700 mL cylindrical niobium autoclave equipped with temperature and pressure gauges. In operation, the system incorporates an electric heating mantle and is coupled to an oscillating table providing stirring of approximately 1 Hz. The heating was carried out with a temperature increase of 3.6 °C per minute. For the pretreatment trials, 40 g of bagasse (d.w.) and 500 mL of a basic sulfite reagent (4 g Na2SO3 and 2 g NaOH) were added to the reactor (8% (m/v) solid loading) which was then hermetically closed and placed on heating to 150 °C and maintained for 30 min. Upon completion of each pretreatment, the reactor was cooled in an ice-water bath to halt the reactions before being opened. The liquid fraction was separated from the solid pretreated bagasse by filtration, passing it three times to retain fine particles. The pretreated bagasse was washed in the Buchner funnel with 1.6 L of warm water (60 °C) over three recirculation cycles. The spent liquor was combined with the wash water, adjusted to 2 L, and kept for analysis of solubilized lignin and sugars. After air-drying to roughly 10–15% moisture content, the pretreatment yield was calculated as the ratio of the dry weight of pretreated bagasse to the initial dry weight of raw bagasse. The pretreated bagasse was kept in polyethylene zipper-lock bags until further chemical analysis and enzymatic hydrolysis.
2.3. Chemical Characterization of Bagasse Samples and Spent Liquor
Air-dried samples of both the raw and pretreated bagasse were ground to pass a 20-mesh screen using a bench scale Thomas 4 Wiley mill (Thomas Scientific, Swedesboro, NJ, USA), and their chemical constituents were quantified. Ash content across all bagasse samples was carried out at 800 °C for 2 h according to Silva (1995) [26]. For the raw bagasse, roughly 3 g of ground sample placed in filter paper bags was subjected to an 8-hour Soxhlet extraction using 95% ethanol. Extracted and pretreated bagasse samples were hydrolyzed with 72% (w/w) sulfuric acid at 30 °C for 1 h (300 mg sample in 3 mL acid), followed by dilution of the acid to ≅3% with 79 mL of distilled water. The resulting mixture was subsequently autoclaved at 121 °C and 1 bar for 1 h. The hydrolyzed mixture was allowed to cool to room temperature and then filtered through glass microfiber filters. The samples were dried at 105 °C to constant weight and quantified as the insoluble lignin, after subtraction of the lignin ash obtained by calcining the insoluble in muffle (ProLab, São Paulo, Brazil) at 800 °C for 2 h. The soluble lignin in the filtrate was measured in a standard quartz UV cuvette (1 cm path length) (Sigma, St. Louis, MI, USA) at 205 nm using a Halo DB-20 Spectrophotometer (Dynamica, São Paulo, Brazil). An absorptivity (extinction coefficient) value of 105 L/g·cm was used to calculate the amount of acid-soluble lignin present in the hydrolysate [27]. The monomeric sugar contents in the hydrolysate were analyzed by high-performance liquid chromatography (HPLC) (Waters Corporation, Milford, MA, USA) comprising a Waters 1515 system equipped with an Waters 2414 RI detector at 35 °C, and a BIO-RAD HPX87H column (Bio-Rad, Hercules, CA, USA) at 45 °C eluting at a rate of 0.6 mL/min with 5 mM sulfuric acid [28]. Triplicate runs were performed for all assays.
After being separated from the pretreated bagasse, aliquots of the soluble fraction were appropriately diluted and analyzed spectrophotometrically (Halo DB-20, Dynamica, São Paulo, Brazil) for soluble lignin content using a reference quartz UV cuvette (1 cm path length) (Sigma, St. Louis, MI, USA) at 205 nm and using a specific absorptivity of 105 L/g·cm [27]. Other samples of the liquid fraction were stored frozen. On carbohydrate analysis, the samples were thawed, and aliquots were acidified with 1M H2SO4 to pH ≅ 3 and the flasks were closed and autoclaved at 1 bar for 30 min to ensure complete hydrolysis of the remaining oligosaccharides. Thereafter, the hydrolysis mixture was left to cool to room temperature and subsequently filtered, first using 0.45 µm Millex-HV membrane filters (Millipore, Burlington, MA, USA) and subsequently through Sep-Pak C18 cartridges (Waters Corporation, Milford, MA, USA), and analyzed by HPLC for sugar content as described previously [28].
2.4. Enzymatic Hydrolysis
Enzymatic hydrolysis was performed in a shaking incubator (SOLAB, model 420, Solab Equipamentos, Piracicaba, Brazil) at 100 rpm and 45 °C. The substrate concentration was 5% (w/v), corresponding to 8 g of pretreated bagasse in 160 mL of 50 mM sodium citrate buffer (pH 4.8) in 500 mL Erlenmeyer flasks. The hydrolysis tests were carried out under sterile conditions and no antibiotics were added to control microorganisms when hydrolysate was intended for subsequent fermentation. Enzyme dosages were 10 FPU (filter paper unit) cellulase (Celluclast 1.5 L) and 10 CBU (cellobiose unit) β-glucosidase (Novozym188) per gram (d.w.) of pretreated bagasse. Enzymatic hydrolysis was performed over a period of 72 h and then the hydrolysate was filtered from the slurry. Sugars released during the enzymatic hydrolysis were quantified using HPLC (as described above) [28]. To study the hydrolysis kinetics the assays were carried out under similar conditions, except for the working volume which was 40 mL. In these latter assays, 0.02% (w/v) sodium azide was added to the buffer to inhibit microbial growth, and samples were periodically collected and heated in a boiling water bath for 5 min, then centrifuged at 4600 g for 15 min. Sugars released were quantified by HPLC as described above [28]. Glucan and xylan conversion yields during enzymatic hydrolysis were determined as the percentage of glucose or xylose released relative to the initial glucan or xylan content in the pretreated bagasse, using the appropriate stoichiometric conversion factors, as described in Tavares et al. [24].
2.5. Fermentation
The hydrolysate fermentation was carried out using S. cerevisiae PE-2 and S. stipitis CBS 5773. For the inoculum, S. cerevisiae was pre-grown in a medium composed of sucrose (20 g/L), yeast extract (2.5 g/L), urea (0.6 g/L), K2HPO4 (0.87 g/L), MgSO4·7H2O (0.49 g/L), ZnSO4·7H2O (0.029 g/L), MnSO4·H2O (0.017 g/L), and pH = 5.5. S. stipitis was pre-cultured in the same culture medium except for sucrose which was replaced by xylose. Both yeasts were pre-cultured for 72 h at 30 °C after which they were recovered through centrifugation (Hettich ROTANTA 460, Tuttlingen, Germany) at 3000 rpm, 4 °C, 10 min, and washed once with sterile buffer. The fermentation was carried out taking as control a semisynthetic medium containing glucose and xylose at amounts equivalent to those found in the hydrolysate. Both the hydrolysate and the control medium were enriched with the components present in the preculture medium at the same concentrations, except for fermentable sugars, and then autoclaved at 0.5 bar for 10 min. The inoculum cell pellets were resuspended in the fermentation media and transferred into 125 mL Erlenmeyer flasks with a working volume of 30 mL with a starting concentration of about 3 to 4 g/L yeasts. For each sampling time, three independent assays were prepared for each yeast, which were kept in an orbital incubator for 24 h at 100 rpm. Samples for analysis, retrieved after 0, 2, 4, 8, and 24 h, were cooled in the ice bath and centrifuged (HT 80-2B, Huanghua Faithful Instrument, Huanghua, China) for 10 min at 3000 rpm. The obtained supernatant was frozen for further analysis of ethanol, glycerol, acetic acid, and glucose by HPLC as described above in Section 2.2 [28]. The cell pellet was rinsed twice with sterile buffer, resuspended in a defined buffer volume, and then used for biomass quantification by measuring optical density at 600 nm, based on a previously established standard curve.
2.6. Error and Statistical Analyses
For all pretreatment experiments, the standard uncertainty (µ) was evaluated. A mass uncertainty, u (m), of 0.1 mg was attributed to each weighing. The hydrothermal alkaline sulfite pretreatment was conducted with a temperature uncertainty, µ (T), of 2.5 °C, whereas for enzymatic hydrolysis and fermentation, a µ (T) of 1.0 °C was assumed. Experimental errors depended on the calibration method; consequently, a conservative error of 10% of the measured value was applied to all analyses. Each measurement was performed at least in triplicate, and results are presented as mean ± standard deviation. Descriptive statistical analyses were carried out using the Data Analysis tool in Microsoft Excel.
3. Results and Discussion
3.1. Raw and Pretreated Bagasse Chemical Characterization
Regarding the cost-effectiveness of lignocellulosic bioethanol production, the as-received bagasse from sugar-alcohol mills offers a size distribution that often eliminates the need for further milling. Herein, raw bagasse particles measured up to 10 cm and exhibited substantial irregularity. Table 1 summarizes the bagasse chemical profile before and after HAS pretreatment. In the untreated material, cellulose and hemicelluloses together comprised approximately 62% of raw bagasse.

Table 1.
Chemical profiles of raw and HAS-pretreated bagasse.
As detailed in the mass balance of Table 2, HAS pretreatment preserved nearly all the cellulose fraction while removing roughly 63% of the lignin. Consequently, the solid residue was enriched in cellulose and hemicellulose, with respective increases of 31.8% and 11.3%. This apparent carbohydrate enrichment also reflects the solubilization of extractive and ash, as well as a partial removal of hemicelluloses, 24.7% of which was solubilized, including xylan (15%), acetyl groups (81%), and arabinan (39%).

Table 2.
Mass balances (%) of main components for HAS pretreatment at the optimal conditions.
Compared to analogous methods described in the literature [29,30,31], the pretreatment applied here yielded superior results since, in addition to an extensive delignification, it contributed to carbohydrate preservation in the pretreated bagasse, allowing greater fermentable sugars recovery with a considerably lower energy investment. The highly selective delignification observed herein was attributed to the β-O-4 linkages breakdown and sulfonation at β-carbons promoted by both active reagents (NaOH and Na2SO3), thereby smoothing lignin depolymerization and dissolution [32,33,34,35].
3.2. Hydrolysis Kinetics and Hydrolysate Chemical Analysis
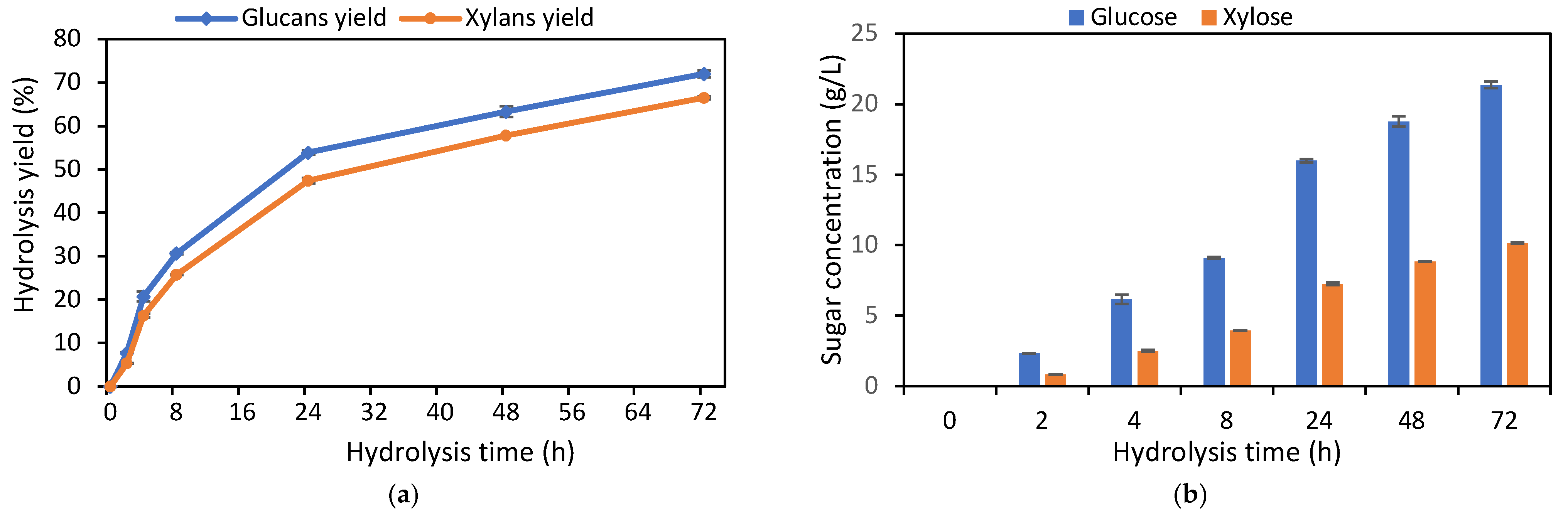
Figure 1a,b illustrates the enzymatic hydrolysis kinetics. As shown, glucose and xylose released from the solid substrate increased rapidly during the first 8 h, after which the hydrolysis rate progressively slowed. By 24 h, the glucose and xylose concentrations reached approximately 75% and 71% of the final values (21.4 and 10.2 g/L, respectively) observed after 72 h of hydrolysis. The saccharification yields of glucans and xylans were 72% and 66%, respectively, after 72 h. End-product inhibition is a widely recognized factor affecting the holocellulose enzymatic hydrolysis rate, especially in batch processes and at higher substrate concentrations [35,36,37]. In this study, we used a 5% (w/v) solids loading, leading to a significant release of sugars during the early stages. This may explain the marked decrease in the hydrolysis rate after 8 h. Another factor is residual lignin, which can act both to block and to bind irreversibly to enzymes, reducing their effectiveness [38,39]. The bagasse’s rheological changes, such as increased cellulose crystallinity, may have further contributed to the decreased hydrolysis rate at later stages, as the amorphous cellulose regions are more readily hydrolyzed in the early phase [40].
Figure 1.
Kinetics of hydrolysis yield (a) and fermentable sugars (b) released from pretreated bagasse (with antibiotics addition). Error bars indicate the standard deviation from three independent replicates.
The conversion yield and composition of the enzymatic hydrolysate for subsequent fermentation are shown in Table 3. As reflected in Figure 1a cellulose and xylan conversion from pretreated bagasse reached about 73% and 63%, respectively. However, since only cellulases and β-glucosidase were used without xylanase or hemicellulase supplementation, xylan hydrolysis was about 10% less efficient. Nevertheless, the result was considerable, suggesting that the enzyme mixture may contain small amounts of xylanases or that xylose was released by lignocellulosic matrix disruption caused by the cellulase and β-glucosidase enzymes. The total fermentable sugars concentration in the enzymatic hydrolysate (Table 3), including cellobiose, glucose, and xylose, was 31.9 g/L. While this concentration is satisfactory for the 5% solids loading and enzyme dosage used, it is below the threshold required for industrial application, as direct fermentation would result in a low ethanol concentration, reducing the economic attractiveness of separation and recovery. Thus, integrating lignocellulosic bioethanol production with first-generation ethanol processes seems to be the most feasible approach, where the hydrolysate could dilute industrial molasses for conventional fermentation without modifying existing plants.

Table 3.
Cellulose and xylan conversion yields and fermentable sugars concentration after the enzymatic hydrolysis for subsequent fermentation.
3.3. Fermentation Kinetics and Products
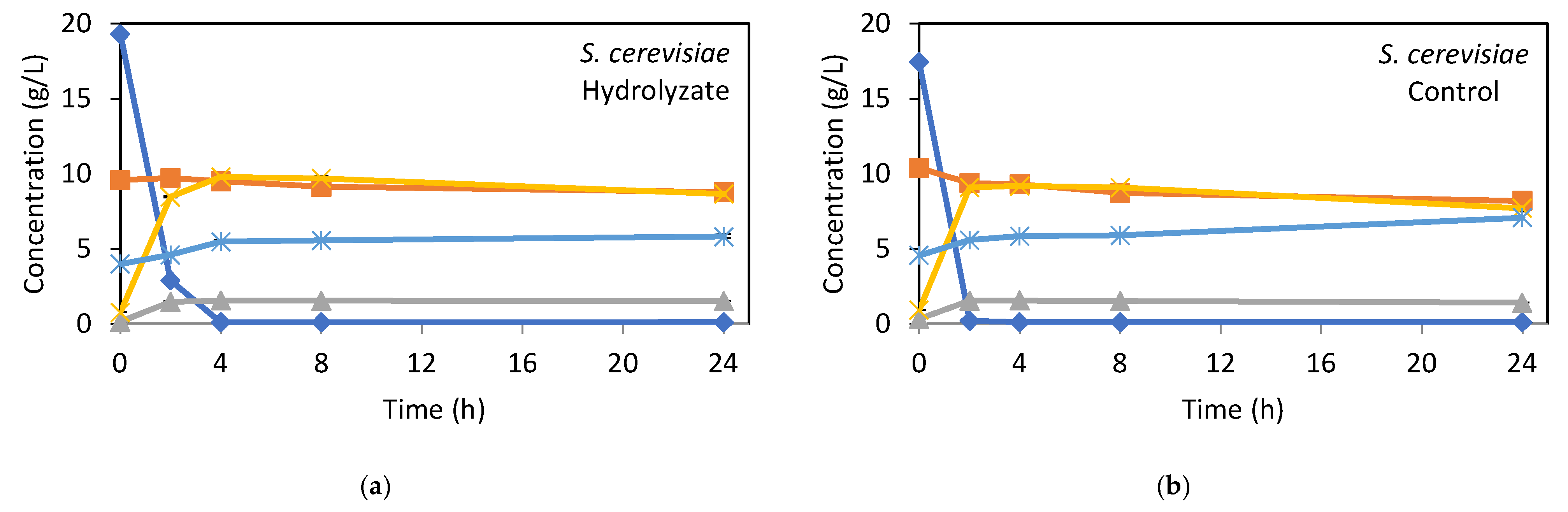
The patterns of sugar consumption and bioethanol production over time are illustrated in Figure 2. As observed, for S. cerevisiae PE-2 (Figure 2a,b), glucose concentration dropped sharply in the first 2 h, while ethanol concentration significantly increased. A maximum ethanol concentration of about 9.8 g/L in the hydrolysate and 9.2 g/L in the synthetic medium was achieved after 4 h of fermentation. No significant differences in fermentability were found between the hydrolysate and synthetic medium, indicating that the hydrolysate obtained from bagasse pretreated by optimized HAS did not contain significant inhibitors for S. cerevisiae PE-2. After 24 h of fermentation, only 8.4% of xylose was consumed in the hydrolysate, versus 21.2% in the synthetic medium. Wild-type S. cerevisiae lacks the enzymes required for the natural assimilation of pentoses. Nevertheless, it can uptake xylose through low-affinity native transporters once glucose is depleted [41]. However, the uptake rates are negligible, xylose is not effectively fermented, and the process is not industrially relevant. The inability of S. cerevisiae to assimilate and metabolize five-carbon sugars like xylose and arabinose remains a key challenge in lignocellulosic hydrolysate fermentation [12,42]. However, due to its robustness in industrial fermentations, significant research, particularly through genetic engineering, has been devoted to enhancing its pentose fermentation capacity and improving its resistance to inhibitors in lignocellulosic hydrolysates [43,44].
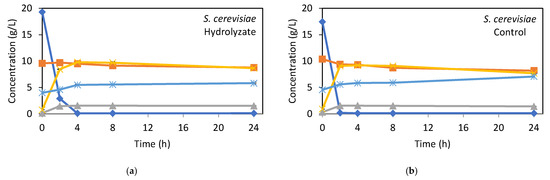
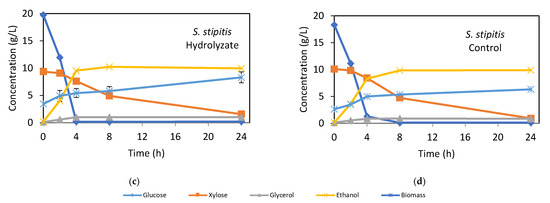
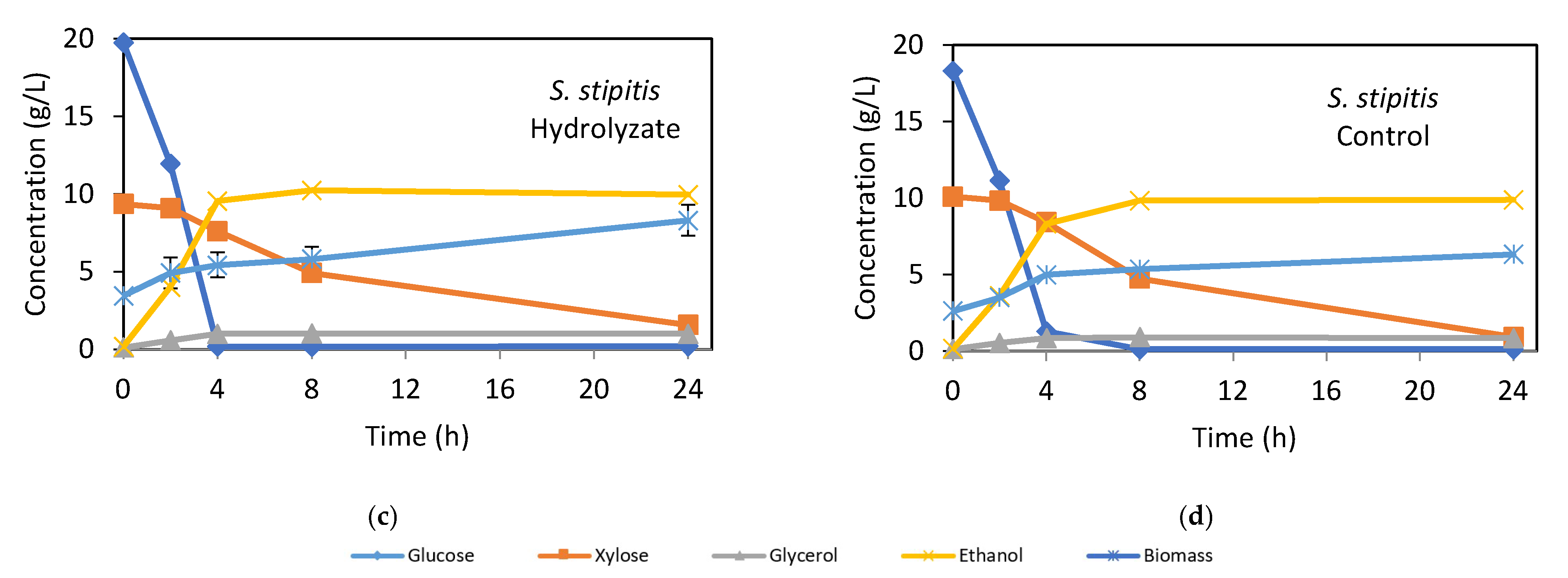
Figure 2.
Kinetics of fermentable sugars consumption and ethanol and biomass production of S. cerevisiae PE-2 (a,b) and S. stipitis CBS 5773 (c,d) cultivated in bagasse hydrolysate and synthetic medium as control. Error bars indicate the standard deviation from three independent replicates.
In the first 2 h, 1.6 g/L of glycerol was generated in both the hydrolysate and synthetic medium, remaining stable throughout fermentation. This may be linked to fermentative stress [45] caused by inhibitory compounds, suggesting that inhibitor levels in the hydrolysate were comparable to those in the synthetic medium. This is further supported by the kinetic parameters in Table 4(A), which show similar yields in both media, with nearly 100% of consumed sugars being converted into ethanol.

Table 4.
Kinetic parameters for S. cerevisiae PE-2 (A) and S. stipitis CBS 5773 (B) cultivated in bagasse hydrolysate and synthetic medium as control.
The fermentation kinetics of S. stipitis CBS 5773 using hydrolysate and synthetic medium are shown in Figure 2c,d, with kinetic parameters in Table 4(B). The maximum ethanol concentration, approximately 10.2 g/L in the hydrolysate and 9.8 g/L in the synthetic medium, was reached after 8 h of fermentation. Glucose consumption was rapid during the first 4 h, reaching 99.5% in the hydrolysate and 93.0% in the synthetic medium. In contrast to S. cerevisiae, S. stipitis consumed all the glucose and the majority of xylose in both media, albeit at different speeds. During the first 4 h, glucose consumption rates were 4.9 g/L·h in the hydrolysate and 4.3 g/L·h in the synthetic medium, while xylose consumption rates were slower, at 0.4 g/L·h in both media until glucose was depleted. Thereafter, the xylose uptake rate rose to 0.7 g/L·h in the hydrolysate and 0.9 g/L·h in the synthetic medium. Though the sugar consumption kinetics were not fully diauxic, xylose uptake occurred slowly (0.1 g/L·h) during the first 2 h, a behavior observed in several pentose-fermenting microorganisms, including Candida shehatae, Candida utilis [46,47], Candida maltosa [48], and Spathaspora arborariae [8]. This phenomenon has been attributed to glucose-mediated repression of xylose-metabolizing enzymes or suppression of high-affinity xylose transporters [42,49].
As shown in Figure 2c,d after glucose depletion, S. stipitis fermented xylose, leading to an increase in ethanol concentrations up to 8 h, though the conversion was less efficient than glucose fermentation. This could be due to a metabolic shift from ethanol or xylitol synthesis toward cell growth, as reported for C. guilliermondii when xylose concentrations dropped below 50 g/L [50]. Silva et al. [28] also demonstrated that S. stipitis requires tight oxygen control to maintain xylose conversion to ethanol or xylitol, which was not achievable in the shaker flask used in this study. The continuous increase in S. stipitis biomass likely resulted from this metabolic shift. Additionally, after glucose depletion, ethanol was metabolized while xylose was still being consumed, as also noted by Guo et al. [48] and Schirmer-Michel et al. [51] in other pentose-metabolizing yeasts.
As shown in Table 4(B), the efficiencies of glucose and xylose conversion to ethanol by S. stipitis CBS 5773 in the hydrolysate and synthetic medium were 80% and 85%, respectively, primarily due to lower xylose conversion efficiency. However, both S. stipitis and S. cerevisiae PE-2 were able to ferment enzymatic hydrolysates from bagasse pretreated by HAS as efficiently as they fermented synthetic media. This highlights the biotechnological potential of this pretreatment approach for lignocellulosic ethanol production.
While S. cerevisiae is extensively employed in industry because of its robustness, high ethanol tolerance, yield, and efficient hexose fermentation, S. stipitis offers an advantage for second-generation bioethanol production by its superior ability to ferment xylose into ethanol. However, its theoretical ethanol yield (0.35–0.44 g/g) remains beneath the level observed in S. cerevisiae (0.51 g/g), a drawback that can be mitigated by employing co-culture strategies involving both species, as well demonstrated by Wu et al. [52], or by adopting continuous or fed-batch fermentations to better mimic the operational environment of large-scale production systems where the oxygen transfer rate can be tightly adjusted [28].
In addition to being one of the most efficient natural xylose fermenters, S. stipitis has been gaining attention in the context of second-generation bioethanol production, especially in light of significant advances in elucidating its metabolic pathways. These studies have identified key enzymes that enhance sugar uptake and fermentation efficiency [53], as well as mechanisms that improve tolerance to inhibitors present in lignocellulosic hydrolysates [54]. Such traits are essential for scaling up industrial bioethanol production and for optimizing fermentation processes so that yields approach those achieved with first-generation biofuels.
Bioethanol obtained from agro-industrial wastes or byproduct streams is regarded as one of the most promising alternatives to conventional fossil fuels. In Brazil, sugarcane bagasse stands out as an abundant agro-industrial by-product that can be effectively exploited for the production of second-generation bioethanol [1,24].
Even though Brazil is among the largest global producers of first-generation bioethanol, the growing demand will require further development of second-generation bioethanol. Thus, both in Brazil and internationally, governments, funding bodies, and researchers have promoted and investigated strategies to address the challenges of producing bioethanol from lignocellulosic agro-industrial residues by implementing biorefinery concepts and adapting existing first-generation bioethanol facilities to also support and integrate second-generation ethanol production [1,5,6,55].
The challenges mentioned are related to the operationalization of fermentable carbohydrate extraction from the complex and recalcitrant structure of lignocellulosic biomass [6,7,23]. The sulfite process, traditionally used in pulping for decades, is well known for its effectiveness in lignin removal from lignocellulosic materials. Although less commonly applied, the alkaline sulfite process offers certain advantages, such as reduced equipment corrosion and lower sulfur emissions, making it a more environmentally friendly alternative [9,24,29]. Considering these aspects, this study employed hydrothermal alkaline sulfite pretreatment to selectively delignify sugarcane bagasse and produce a carbohydrate-rich substrate with high enzymatic digestibility and significant fermentability.
The optimized HAS pretreatment applied in this study demonstrated a high selective delignification rate (63.2%) while preserving most of the cellulose (97.8%) and hemicellulose (84.7%), resulting in efficient enzymatic sugar conversion with yields of 72% glucan and 66% xylan. As opposed to pretreatments like dilute acid or steam explosion, which typically require higher energy inputs and cause greater sugar degradation, HAS offers a more efficient and cost-effective alternative [24,56]. Furthermore, the high delignification efficiency allows enzymatic hydrolysis to be performed using relatively low enzyme loadings (10 FPU cellulase and 10 CBU β-glucosidase per gram of bagasse), providing significant cost savings in enzyme consumption [57]. Besides being less energy intensive, HAS pretreatment of sugarcane bagasse can be integrated with existing first-generation ethanol plants, for example, by diluting molasses with enzymatic hydrolysates. This integration could substantially reduce operational costs and enhance economic feasibility, making HAS a sustainable, cost-effective, and efficient pretreatment option for second-generation bioethanol production. However, the 8% solids loading used in this bench-scale work should be increased in view of the scale-up requirements for any industrial process, where minimizing cost, time, and labor and the efficient use of resources are essential. Combining HAS pretreatment with an extrusion process could be a promising approach, given extrusion’s widespread industrial adoption, strong mechanical action on lignocellulosic biomass, and ability to operate at high solid-to-liquid ratios [58,59].
As shown in the results of this study, both main sugars, glucose and xylose, produced in the process herein employed, can be well-suited for use in the production of bioethanol, with efficiency identical to that verified in synthetic culture medium. This reinforces the conviction defended in several studies [19,21,22,42,43,44] that fermentation of xylose, a major sugar component of hemicellulose, is one of the key factors defining the economic viability of lignocellulosic bioethanol.
Despite the robustness of the results obtained within this research, the enzymatic hydrolysis step, one of the three main stages of the process (pretreatment, hydrolysis, and fermentation), requires further investigation to enhance the potential economic feasibility of the system. High solids loading, typically above 15% w/w (approximately 176.5 g/L), has been identified as a critical parameter for increasing fermentable sugar concentrations while simultaneously reducing downstream processing costs and energy consumption, without significantly compromising yield [60,61]. This aspect should be addressed in future studies, along with the evaluation of in-house hemicellulase supplementation, as proposed by Montiel et al. [62], to assess its synergistic effect when combined with commercial cellulase cocktails and thereby improve the technical sustainability of the overall biotechnological process.
4. Conclusions
Findings from this work confirm that enzymatic hydrolysates from hydrothermal alkaline sulfite (HAS)-pretreated sugarcane bagasse exhibited minimal toxicity toward S. cerevisiae PE-2 and S. stipitis CBS 5773, enabling fermentation performances comparable to those in synthetic media. The optimized HAS pretreatment achieved extensive delignification (63.2%) while preserving most of the holocellulose (97.8% cellulose and 84.7% hemicellulose), producing a highly digestible substrate even at low enzyme loadings. Despite these promising fermentation efficiencies, the 5% solids loading used in enzymatic hydrolysis resulted in ethanol concentrations below the threshold required for industrial-scale applications. For industrial deployment, higher solids loadings during hydrolysis are essential. Alternatively, integrating second-generation ethanol production with first-generation processes, such as using the hydrolysate to dilute molasses in existing bioethanol plants, offers a more practical pathway. Such integration could reduce operational costs, increase overall bioethanol yields, and enhance the sustainability and economic feasibility of lignocellulosic ethanol production.
Author Contributions
J.T.: Conceptualization, Investigation, Methodology, Formal analysis, Visualization, Writing—Original draft preparation and Reviewing and Editing. A.R.: Investigation and Reviewing and Editing. T.d.P.: Writing—Reviewing and Editing, Funding acquisition. F.d.S.: Supervision, Writing—Reviewing and Funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors would like to thank CAPES (Coordination for Improvement of Higher Education Personnel) for the scholarship granted to the author JEBT under the Postgraduate Student-covenant Program (PEC-PG, CAPES/CNPq—Brasil).
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Chandel, A.K.; Silva, S.S.; Carvalho, W.; Singh, O.V. Sugarcane bagasse and leaves: Foreseeable biomass of biofuel and bio-products. J. Chem. Technol. Biotechnol. 2012, 87, 11–20. [Google Scholar] [CrossRef]
- Yadav, P.; Anu, S.K.T.; Kumar, V.; Singh, D.; Kumar, S.; Manisha, V.M.; Singh, B. Sugarcane bagasse: An important lignocellulosic substrate for production of enzymes and biofuels. Biomass Convers. Biorefin. 2022, 14, 6111–6142. [Google Scholar] [CrossRef]
- Tilley, C. Sugarcane as a biofuel and its impact on the climate. Nat. Sci. Educ. 2025, 54, e70010. [Google Scholar] [CrossRef]
- Cardona, C.A.; Quintero, J.A.; Paz, I.C. Production of bioethanol from sugarcane bagasse: Status and perspectives. Bioresour. Technol. 2010, 101, 4754–4766. [Google Scholar] [CrossRef]
- Hiranobe, C.T.; Gomes, A.S.; Paiva, F.F.G.; Tolosa, G.R.; Paim, L.L.; Dognani, G.; Cardim, G.P.; Cardim, H.P.; dos Santos, R.J.; Cabrera, F.C. Sugarcane bagasse: Challenges and opportunities for waste recycling. Clean Technol. 2024, 6, 662–699. [Google Scholar] [CrossRef]
- Prado, C.A.; Loureiro, B.M.S.; Arruda, G.L.; Santos, J.C.; Chandel, A.K. Hydrodynamic cavitation assisted pretreatment of sugarcane bagasse in the presence of yeast cell mass for the production of sugars and their use for biopigments production by Monascus ruber. Biomass Bioenergy 2024, 190, 107434. [Google Scholar] [CrossRef]
- Song, G.; Bai, Y.; Pan, Z.; Liu, D.; Qin, Y.; Zhang, Y.; Fan, Z.; Li, Y.; Madadi, M. Enhancing fermentable sugar production from sugarcane bagasse through surfactant-assisted ethylene glycol pretreatment and enzymatic hydrolysis: Reduced temperature and enzyme loading. Renew. Energy 2024, 227, 120515. [Google Scholar] [CrossRef]
- Cunha-Pereira, F.; Hickert, L.R.; Senhem, N.T.; Rosa, C.A.; Souza-Cruz, P.B.; Ayub, M.A.Z. Conversion of sugars present in rice hull hydrolysates into ethanol by Spathaspora arborariae, Saccharomyces cerevisiae, and their co-fermentations. Bioresour. Technol. 2011, 102, 4218–4225. [Google Scholar] [CrossRef]
- Tharunkumar, J.; Arosha, V.K.; Bajhaiya, A.K.; Rakesh, S. Optimizing alkaline pretreatment for delignification of paddy straw and sugarcane bagasse to enhance bioethanol production. Biomass Convers. Biorefin. 2024, 15, 16409–16419. [Google Scholar] [CrossRef]
- Brandt, B.A.; Teke, G.M.; van Zyl, W.H.; Görgens, J.F. Development of a hardened industrial strain of S. cerevisiae for bioethanol production from sugarcane bagasse hydrolysates. Waste Biomass Valorization 2025, 16, 3605–3614. [Google Scholar] [CrossRef]
- Tadesse, H.M.; Atnafu, T.; Kassahun, E.; Tessema, I.; Abewaa, M.; Tibebu, S. Optimization of bioethanol production from a brewers’ spent grain and sugarcane molasses mixture utilizing Saccharomyces cerevisiae. Biomass Convers. Biorefin. 2025, 15, 20765–20788. [Google Scholar] [CrossRef]
- Hahn Hägerdal, B.; Karhumaa, K.; Jeppsson, M.; Gorwa Grauslund, M.F. Metabolic engineering for pentose utilization in Saccharomyces cerevisiae. Adv. Biochem. Eng. Biotechnol. 2007, 108, 147–177. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Skerker, J.M.; Kang, W.; Lesmana, A.; Wei, N.; Arkin, A.P.; Jin, Y.S. Rational and evolutionary engineering approaches uncover a small set of genetic changes efficient for rapid xylose fermentation in Saccharomyces cerevisiae. PLoS ONE 2013, 8, e57048. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, L.V.; Neitzel, T.; Lima, C.S.; de Carvalho, L.M.; de Lima, T.B.; Ienczak, J.L.; Pereira, G.A.G. Engineering cellular redox homeostasis to optimize ethanol production in xylose-fermenting Saccharomyces cerevisiae strains. Microbiol. Res. 2025, 290, 127955. [Google Scholar] [CrossRef]
- Zhang, Y.W.; Yang, J.J.; Qian, F.H.; Sutton, K.B.; Hjort, C.; Wu, W.P.; Jiang, Y.; Yang, S. Engineering a xylose fermenting yeast for lignocellulosic ethanol production. Nat. Chem. Biol. 2025, 21, 443–450. [Google Scholar] [CrossRef]
- Engwa, G.A. Genetic engineering on microorganism: The ecological and bioethical implications. Eur. J. Biotechnol. Biosci. 2013, 1, 27–33. [Google Scholar]
- Arbel-Groissman, M.; Menuhin-Gruman, I.; Naki, D.; Bergman, S.; Tuller, T. Fighting the battle against evolution: Designing genetically modified organisms for evolutionary stability. Trends Biotechnol. 2023, 41, 1518–1531. [Google Scholar] [CrossRef]
- Olsson, L.; Hahn-Hägerdal, B. Fermentation of lignocellulosic hydrolysates for ethanol production. Enzyme Microb. Technol. 1996, 18, 312–331. [Google Scholar] [CrossRef]
- Gírio, F.M.; Fonseca, C.; Carvalheiro, F.; Duarte, L.C.; Marques, S.; Bogel-Łukasik, R. Hemicelluloses for fuel ethanol: A review. Bioresour. Technol. 2010, 101, 4775–4800. [Google Scholar] [CrossRef]
- Vasylyshyn, R.; Dmytruk, K.; Ruchala, J.; Sibirny, A.A. Construction of the improved producers of first- and second-generation ethanol in conventional and non-conventional yeasts. In Biotechnology of Yeasts and Filamentous Fungi; Sibirny, A.A., Ed.; Springer Nature Switzerland AG: Cham, Switzerland, 2025; pp. 1–48. [Google Scholar] [CrossRef]
- Jeffries, T.W.; Grigoriev, I.V.; Grimwood, J.; Laplaza, J.M.; Aerts, A.; Salamov, A.; Schmutz, J.; Lindquist, E.; Dehal, P.; Shapiro, H.; et al. Genome sequence of the lignocellulose bioconverting and xylose fermenting yeast Pichia stipitis. Nat. Biotechnol. 2007, 25, 319–326. [Google Scholar] [CrossRef]
- Jönsson, L.J.; Alriksson, B.; Nilvebrant, N.O. Bioconversion of lignocellulose: Inhibitors and detoxification. Biotechnol. Biofuels 2013, 6, 16. [Google Scholar] [CrossRef]
- Fan, J.; Lu, Y.; An, N.; Zhu, W.; Li, M.; Gao, M.; Wang, X.; Wu, C.; Wang, Y. Pretreatment technologies for lignocellulosic biomass: Research progress, mechanisms, and prospects. BioResources 2025, 20, 4897–4924. [Google Scholar] [CrossRef]
- Tavares, J.; Łukasik, R.M.; de Paiva, T.; da Silva, F. Hydrothermal alkaline sulfite pretreatment in the delivery of fermentable sugars from sugarcane bagasse. New J. Chem. 2018, 42, 4474–4484. [Google Scholar] [CrossRef]
- Paz-Cedeno, F.R.; Henares, L.R.; Solorzano-Chavez, E.G.; Scontri, M.; Picheli, F.P.; Roldán, I.U.M.; Monti, R.; de Oliveira, S.C.; Masarin, F. Evaluation of the Effects of Different Chemical Pretreatments in Sugarcane Bagasse on the Response of Enzymatic Hydrolysis in Batch Systems Subject to High Mass Loads. Renew. Energy 2021, 165, 1–13. [Google Scholar] [CrossRef]
- Silva, F.T. Obtenção de Insumos Químicos a Partir do Aproveitamento Integral do Bagaço de Cana (Tese de Doutorado, Universidade Estadual de Campinas, Instituto de Química). Repositório da UNICAMP, 1995. Available online: https://repositorio.unicamp.br/acervo/detalhe/90068 (accessed on 23 July 2025).
- Mendes, F.M.; Siqueira, G.; Carvalho, W.; Ferraz, A.; Milagres, A.M.F. Enzymatic hydrolysis of chemithermomechanically pretreated sugarcane bagasse and samples with reduced initial lignin content. Biotechnol. Prog. 2011, 27, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Ferraz, A.; Baeza, J.; Rodriguez, J.; Freer, J. Estimating the chemical composition of biodegraded pine and eucalyptus wood by DRIFT spectroscopy and multivariate analysis. Bioresour. Technol. 2000, 74, 201–212. [Google Scholar] [CrossRef]
- Silva, V.F.N.; Arruda, P.V.; Felipe, M.G.A.; Gonçalves, A.R.; Rocha, G.J.M. Fermentation of cellulosic hydrolysates obtained by enzymatic saccharification of sugarcane bagasse pretreated by hydrothermal processing. J. Ind. Microbiol. Biotechnol. 2011, 38, 809–817. [Google Scholar] [CrossRef]
- Mendes, F.M.; Laurito, D.F.; Bazzeggio, M.; Ferraz, A.; Milagres, A.M.F. Enzymatic digestion of alkaline-sulfite pretreated sugar cane bagasse and its correlation with the chemical and structural changes occurring during the pretreatment step. Biotechnol. Prog. 2013, 29, 890–895. [Google Scholar] [CrossRef]
- Guilherme, A.A.; Dantas, P.V.F.; Santos, E.S.; Fernandes, F.A.N.; Macedo, G.R. Evaluation of composition, characterization and enzymatic hydrolysis of pretreated sugar cane bagasse. Braz. J. Chem. Eng. 2015, 32, 23–33. [Google Scholar] [CrossRef]
- Gellerstedt, G. Chemistry of chemical pulping. In Pulp and Paper Chemistry and Technology; Monica, E.K., Gellerstedt, G., Henriksson, G., Eds.; Walter de Gruyter: Weinheim, Germany, 2009; Volume 2, pp. 121–136. [Google Scholar]
- Yang, M.; Zhang, X.; Cheng, G. A two-stage pretreatment using dilute sodium hydroxide solution followed by an ionic liquid at low temperatures: Toward construction of lignin-first biomass pretreatment. Bioresour. Technol. Rep. 2019, 7, 100286. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, Z.; Li, Y.; Wang, W.; Liu, X.; Shu, H.; Jiang, J. Comparison of the effects of NaOH and deep eutectic solvent catalyzed tobacco stock lignin isolation: Chemical structure and thermal characteristics. Catalysts 2024, 14, 744. [Google Scholar] [CrossRef]
- Holtzapple, M.; Cognata, M.; Shu, Y.; Hendrickson, C. Inhibition of Trichoderma reesei cellulase by sugars and solvents. Biotechnol. Bioeng. 1990, 36, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Gregg, D.J.; Saddler, J.N. Factors affecting cellulose hydrolysis and the potential of enzyme recycle to enhance the efficiency of an integrated wood to ethanol process. Biotechnol. Bioeng. 1996, 51, 375–383. [Google Scholar] [CrossRef]
- Yu, Z. Fundamental Factors Affecting Enzymatic Hydrolysis of Lignocellulosic Biomass. Ph.D. Dissertation, North Carolina State University, Raleigh, NC, USA, 2013. [Google Scholar]
- Mussatto, S.I.; Fernandes, M.; Milagres, A.M.F.; Roberto, I.C. Effect of hemicellulose and lignin on enzymatic hydrolysis of cellulose from brewer’s spent grain. Enzym. Microb. Technol. 2008, 43, 124–129. [Google Scholar] [CrossRef]
- Tu, M.; Pan, X.; Saddler, J.N. Adsorption of cellulase on cellulolytic enzyme lignin from lodgepole pine. J. Agric. Food Chem. 2009, 57, 7771–7778. [Google Scholar] [CrossRef]
- Hall, M.; Bansal, P.; Lee, J.H.; Realff, M.J.; Bommarius, A.S. Cellulose crystallinity—A key predictor of the enzymatic hydrolysis rate. FEBS J. 2010, 277, 1571–1582. [Google Scholar] [CrossRef]
- Donzella, L.; Sousa, M.J.; Morrissey, J.P. Evolution and functional diversification of yeast sugar transporters. Essays Biochem. 2023, 67, 811–827. [Google Scholar] [CrossRef]
- Brink, D.P.; Borgström, C.; Persson, V.C.; Ofuji Osiro, K.; Gorwa-Grauslund, M.F. D-xylose sensing in Saccharomyces cerevisiae: Insights from D-glucose signaling and native D-xylose utilizers. Int. J. Mol. Sci. 2021, 22, 12410. [Google Scholar] [CrossRef]
- Laluce, C.; Schenberg, A.C.G.; Gallardo, J.C.M.; Coradello, L.F.C.; Pombeiro-Sponchiado, S.R. Advances and developments in strategies to improve strains of Saccharomyces cerevisiae and processes to obtain lignocellulosic ethanol: A review. Appl. Biochem. Biotechnol. 2012, 166, 1908–1926. [Google Scholar] [CrossRef]
- Wagner, E.R.; Gasch, A.P. Advances in S. cerevisiae engineering for xylose fermentation and biofuel production: Balancing growth, metabolism, and defense. J. Fungi 2023, 9, 786. [Google Scholar] [CrossRef]
- Gibson, B.R.; Lawrence, S.J.; Leclaire, J.P.; Powell, C.D.; Smart, K.A. Yeast responses to stresses associated with industrial brewery handling. FEMS Microbiol. Rev. 2007, 31, 535–569. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.; van Uden, N. Transport of hemicellulose monomers in the xylose-fermenting yeast Candida shehatae. Appl. Microbiol. Biotechnol. 1986, 23, 491–495. [Google Scholar] [CrossRef]
- Kilian, S.G.; Prior, B.A.; du Preez, J.C. The kinetics and regulation of d xylose transport in Candida utilis. World J. Microbiol. Biotechnol. 1993, 9, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; He, P.; Lu, D.; Shen, A.; Jiang, N. Cloning and molecular characterization of a gene coding D-xylulokinase (CmXYL3) from Candida maltosa. J. Appl. Microbiol. 2006, 101, 139–150. [Google Scholar] [CrossRef]
- Preziosi-Belloy, L.; Nolleau, V.; Navarro, J.M. Fermentation of hemicellulosic sugars and sugar mixtures to xylitol by Candida parapsilosis. Enzyme Microb. Technol. 1997, 21, 124–129. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Dragone, G.; Roberto, I.C. Influence of the toxic compounds present in brewer’s spent grain hemicellulosic hydrolysate on xylose-to-xylitol bioconversion by Candida guilliermondii. Process Biochem. 2005, 40, 3801–3806. [Google Scholar] [CrossRef]
- Schirmer-Michel, Â.C.; Flôres, S.H.; Hertz, P.F.; Ayub, M.A.Z. Effect of oxygen transfer rates on alcohols production by Candida guilliermondii cultivated on soybean hull hydrolysate. J. Chem. Technol. Biotechnol. 2009, 84, 223–228. [Google Scholar] [CrossRef]
- Wu, Y.; Wen, J.; Wang, K.; Su, C.; Chen, C.; Cui, Z.; Cai, D.; Cheng, S.; Cao, H.; Qin, P. Understanding the Dynamics of the Saccharomyces cerevisiae and Scheffersomyces stipitis Abundance in Co-Culturing Process for Bioethanol Production from Corn Stover. Waste Biomass Valor. 2023, 14, 43–55. [Google Scholar] [CrossRef]
- Ochoa-Chacón, A.; Martinez, A.; Poggi-Varaldo, H.M.; Villa-Tanaca, L.; Ramos-Valdivia, A.C.; Ponce-Noyola, T. Xylose Metabolism in Bioethanol Production: Saccharomyces cerevisiae vs. Non-Saccharomyces Yeasts. Bioenerg. Res. 2022, 15, 905–923. [Google Scholar] [CrossRef]
- Biazi, L.E.; Santos, S.C.; Kaupert Neto, A.A.; Sousa, A.S.; Soares, L.B.; Renzano, E.; Velasco, J.; Rabelo, S.C.; Costa, A.C.; Ienczak, J.L. Adaptation Strategy to Increase the Tolerance of Scheffersomyces stipitis NRRL Y-7124 to Inhibitors of Sugarcane Bagasse Hemicellulosic Hydrolysate through Comparative Studies of Proteomics and Fermentation. Bioenerg. Res. 2022, 15, 479–492. [Google Scholar] [CrossRef]
- Rossi, L.M.; Gallo, J.M.R.; Mattoso, L.H.C.; Buckeridge, M.S.; Licence, P.; Allen, D.T. Ethanol from Sugarcane and the Brazilian Biomass-Based Energy and Chemicals Sector. ACS Sustain. Chem. Eng. 2021, 9, 4293–4295. [Google Scholar] [CrossRef]
- Dai, N.H.; Huynh, K.T.T.; Nguyen, T.A.D.; Do, V.V.T.; Van Tran, M. Hydrothermal and Steam Explosion Pretreatment of Bambusa stenostachya Bamboo. Waste Biomass Valor. 2021, 12, 4103–4112. [Google Scholar] [CrossRef]
- Paul, M.; Shroti, G.K.; Mohapatra, S.; DasMohapatra, P.K.; Thatoi, H. A Comparative Study on Pretreatment of Rice Straw and Saccharification by Commercial and Isolated Cellulase–Xylanase Cocktails Towards Enhanced Bioethanol Production. Syst. Microbiol. Biomanuf. 2024, 4, 731–749. [Google Scholar] [CrossRef]
- José, Á.H.M.; Moura, E.A.B.; Rodrigues, D., Jr.; Kleingesinds, E.K.; Rodrigues, R.C.L.B. A Residue-Free and Effective Corncob Extrusion Pretreatment for the Enhancement of High Solids Loading Enzymatic Hydrolysis to Produce Sugars. Ind. Crops Prod. 2022, 188, 115655. [Google Scholar] [CrossRef]
- Cavailles, J.; Vaca-Medina, G.; Wu-Tiu-Yen, J.; Labonne, L.; Evon, P.; Peydecastaing, J.; Pontalier, P.-Y. Impact of thermomechanical pretreatment by twin-screw extrusion on the properties of bio-based materials from sugarcane bagasse obtained by thermocompression. Bioresour. Technol. 2024, 414, 131642. [Google Scholar] [CrossRef]
- Modenbach, A.A.; Nokes, S.E. Enzymatic hydrolysis of biomass at high-solids loadings–A review. Biomass Bioenergy 2013, 56, 526–544. [Google Scholar] [CrossRef]
- Qiao, H.; Ma, Z.; Wang, Y.; Zheng, Z.; Ouyang, J. Achieving efficient and rapid high-solids enzymatic hydrolysis for producing high titer ethanol with the assistance of di-rhamnolipids. Bioresour. Technol. 2024, 394, 130189. [Google Scholar] [CrossRef]
- Montiel, C.; Hernández-Meléndez, O.; Marques, S.; Gírio, F.; Tavares, J.; Ontañon, O.; Campos, E.; Bárzana, E. Application of In-House Xylanases as an Addition to a Commercial Cellulase Cocktail for the Sustainable Saccharification of Pretreated Blue Agave Bagasse Used for Bioethanol Production. Sustainability 2024, 16, 6722. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).