Compression of Bio-Signals Using Block-Based Haar Wavelet Transform and COVIDOA for IoMT Systems
Abstract
1. Introduction
- The HWT can extract local spectral and temporal information simultaneously.
- Wavelet-based coding allows for progressive data transmission and is more robust to transmission and decoding failures.
- The HWT is conceptually simple and fast.
- The HWT is completely reversible and does not suffer from the edge effects that are an issue with other wavelet transformations.
- Block-based HWT and inverse transform can be performed by applying matrix multiplication.
2. Literature Review
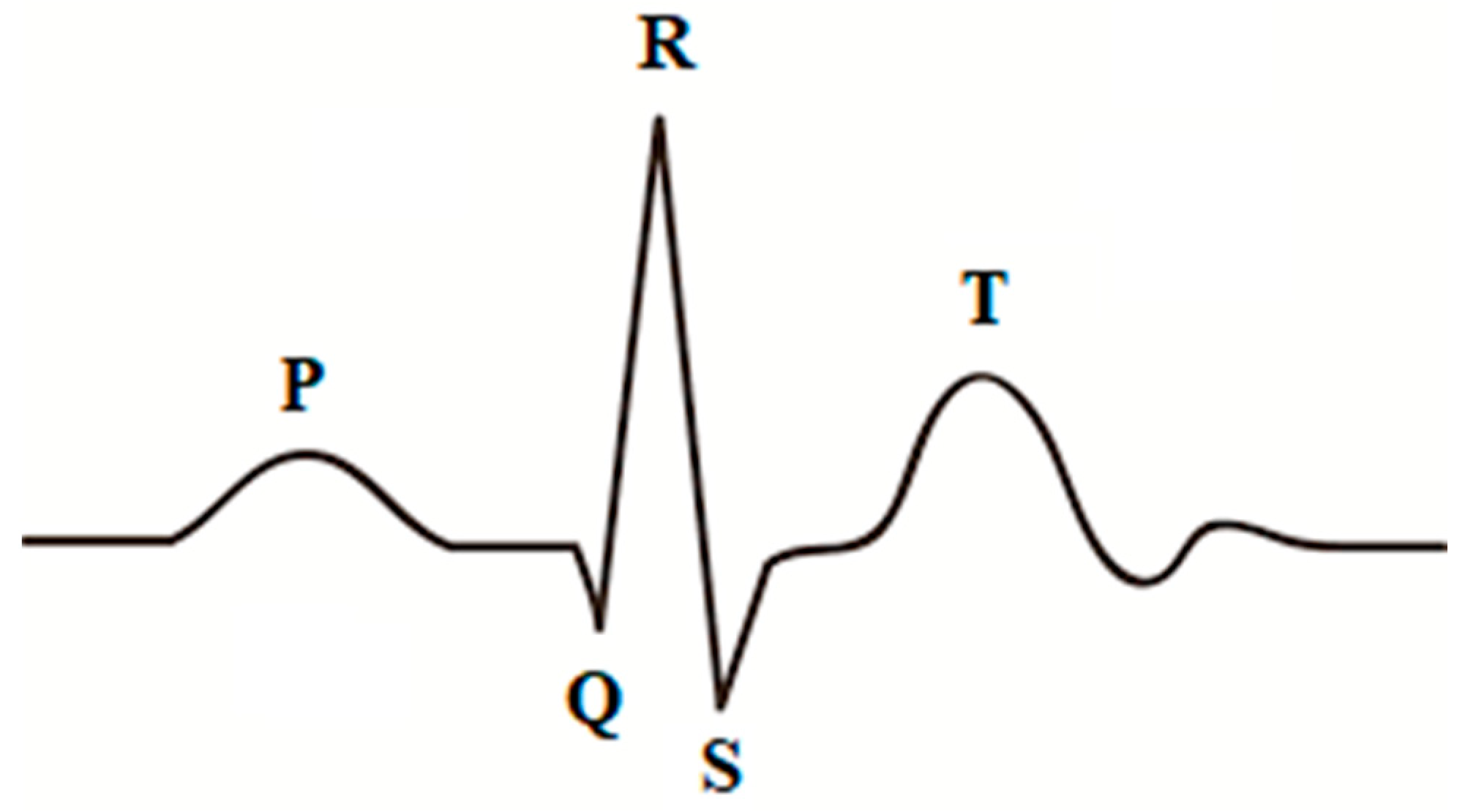
3. HWT
4. COVIDOA
- Virus entry and uncoating
- b.
- Virus replication
- c.
- Virus mutation
- d.
- New virion formation and release
| Algorithm 1 Pseudocode of COVIDOA. |
| Set initial values of the following parameters: Dimension (D), population size (popSize), maximum number of iterations (MaxItr), number of proteins, shifting number, and mutation rate (MR). |
| For (i = 1: I ≤ nPop) do |
| Generate initial random population. |
| Evaluate the fitness function for all solutions in the population. |
| End for |
| Order solutions ascendingly according to fitness function. |
| Set the first solution as the optimum solution. |
| Set t = 1 |
| Repeat |
| For (i = 1: I ≤ nPop) do |
| Select a parent solution P, |
| For (k = 1: I ≤ number of proteins) do |
| Generate protein Vk from parent solution P using Equations (9) and (10). |
| End for |
| Apply uniform crossover between the generated proteins to generate new virion (new solution). |
| if (rand (0,1) < MR) then |
| Mutate the new solution using Equation (11). |
| End if |
| End for |
| Until t ≥ MaxItr |
5. The Proposed Compression/Decompression Algorithm
- The signal is split up into blocks of size 1 × N; N can be 8, 16, 32, or 64.
- The required subset of the size of the coefficients is calculated using Equation (12).
- The Haar wavelet kernel matrix is calculated using Equations (3)–(5).
- The parameters of COVIDOA are set as follows: D = SS; population size (popSize) = 30; the maximum number of iterations (MaxItr) = 50; the number of proteins = 2; shifting number = 1; mutation rate (MR) = 0.5.
- For each signal block
- Calculate the block-based HWT to obtain the Haar coefficients using Equation (2).
- COVIDOA is used to select the optimal coefficients according to the PRD objective function using Equation (1) as follows:
- Generate an initial random population of solutions and compute the objective function for each solution.
- Select parent solution using tournament selection and apply the frameshifting technique to generate several proteins using Equations (9) and (10).
- Apply crossover between the generated proteins to generate a new virion.
- Apply mutation to the previously generated solution to obtain a new mutated solution.
- Replace the new solution with the parent solution if the new solution is fitter than the parent. Otherwise, the parent solution remains.
- Repeat steps ii to v until the MaxItr is reached.
- Select the optimal solution achieved so far.
- From the coefficient obtained in step a, only the coefficients whose positions correspond to the values in the optimum solution are selected, and the remaining coefficients are ignored (set to zero).
- Apply the inverse transform to the optimum coefficients obtained in the previous step to obtain the reconstructed signal block using Equation (8).
- Concatenate the reconstructed blocks to obtain the reconstructed signal.
- Evaluate the algorithm’s performance using CR, PRD, SSIM, and QS metrics.
6. Results
6.1. Datasets
6.2. Evaluation Criteria
- CR
- PRD %
- NCC
- QS
6.3. Numerical Results and Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tian, S.; Yang, W.; Le Grange, J.M.; Wang, P.; Huang, W.; Ye, Z. Smart healthcare: Making medical care more intelligent. Glob. Health J. 2019, 3, 62–65. [Google Scholar] [CrossRef]
- Reaz, M.B.I.; Hussain, M.S.; Mohd-Yasin, F. Techniques of EMG signal analysis: Detection, processing, classification, and applications. Biol. Proced. Online 2006, 8, 11–35. [Google Scholar] [CrossRef] [PubMed]
- Houssein, E.H.; Kilany, M.; Hassanien, A.E. ECG signals classification: A review. Int. J. Intell. Eng. Inform. 2017, 5, 376–396. [Google Scholar] [CrossRef]
- Nagel, S. Towards a Home-Use BCI: Fast Asynchronous Control and Robust Non-Control State Detection. Ph.D. Thesis, Universität Tübingen, Tübingen, Germany, 2019. [Google Scholar]
- Jeong, J.S.; Han, O.; You, Y.Y. A design characteristics of smart healthcare system as the IoT application. Indian J. Sci. Technol. 2016, 9, 52. [Google Scholar] [CrossRef]
- Abdellatif, A.A.; Emam, A.; Chiasserini, C.F.; Mohamed, A.; Jaoua, A.; Ward, R. Edge-based compression and classification for smart healthcare systems: Concept, implementation, and evaluation. Expert Syst. Appl. 2019, 117, 1–14. [Google Scholar] [CrossRef]
- Makbol, N.M.; Khoo, B.E.; Rassem, T.H. Block-based discrete wavelet transform-singular value decomposition image watermarking scheme using human visual system characteristics. IET Image Process. 2016, 10, 34–52. [Google Scholar] [CrossRef]
- Mentzer, F.; Gool, L.V.; Tschannen, M. Learning better lossless compression using lossy compression. In Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition, Seattle, WA, USA, 14–19 June 2020; pp. 6638–6647. [Google Scholar]
- Hosny, K.M.; Khalid, A.M.; Mohamed, E.R. Efficient compression of volumetric medical images using Legendre moments and differential evolution. Soft Comput. 2020, 24, 409–427. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.K.; Mitra, S.; Mitra, M. A lossless ECG data compression technique using ASCII character encoding. Comput. Electr. Eng. 2011, 37, 486–497. [Google Scholar] [CrossRef]
- Chen, S.L.; Wang, J.G. VLSI implementation of low-power cost-efficient lossless ECG encoder design for wireless healthcare monitoring application. Electron. Lett. 2013, 49, 91–93. [Google Scholar] [CrossRef]
- Rzepka, D. Low-complexity lossless multichannel ECG compression based on selective linear prediction. Biomed. Signal Process. Control 2020, 57, 101705. [Google Scholar] [CrossRef]
- Zhou, X.; Bai, Y.; Wang, C. Image compression based on discrete cosine transform and multistage vector quantization. Int. J. Multimed. Ubiquitous Eng. 2015, 10, 347–356. [Google Scholar] [CrossRef]
- Mahmmod, B.M.; Ramli, A.R.B.; Abdulhussain, S.H.; Al-Haddad, S.A.R.; Jassim, W.A. Signal compression and enhancement using a new orthogonal-polynomial-based discrete transform. IET Signal Process. 2018, 12, 129–142. [Google Scholar] [CrossRef]
- Batista, L.V.; Carvalho, L.C.; Melcher, E.U.K. Compression of ECG signals based on optimum quantization of discrete cosine transform coefficients and Golomb-Rice coding. In Proceedings of the 25th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (IEEE Cat. No. 03CH37439), Cancún, Mexico, 17–21 September 2003; Volume 3, pp. 2647–2650. [Google Scholar]
- Jha, C.K.; Kolekar, M.H. Electrocardiogram data compression using DCT-based discrete orthogonal Stockwell transform. Biomed. Signal Process. Control 2018, 46, 174–181. [Google Scholar] [CrossRef]
- Soni, E.; Nagpal, A.; Garg, P.; Pinheiro, P.R. Assessment of Compressed and Decompressed ECG Databases for Telecardiology Applying a Convolution Neural Network. Electronics 2022, 11, 2708. [Google Scholar] [CrossRef]
- Pal, H.S.; Kumar, A.; Vishwakarma, A.; Balyan, L.K. A Hybrid 2D ECG Compression Algorithm using DCT and Embedded Zero Tree Wavelet. In Proceedings of the 2022 IEEE 6th Conference on Information and Communication Technology (CICT), Gwalior, India, 18–20 November 2022; pp. 1–5. [Google Scholar]
- Su, Y.; Lu, X.; Huang, L.; Du, X.; Guizani, M. A novel DCT-based compression scheme for 5G vehicular networks. IEEE Trans. Veh. Technol. 2019, 68, 10872–10881. [Google Scholar] [CrossRef]
- Pandey, A.; Saini, B.S.; Singh, B.; Sood, N. Quality-controlled ECG data compression based on 2D discrete cosine coefficient filtering and iterative JPEG2000 encoding. Measurement 2020, 152, 107252. [Google Scholar] [CrossRef]
- Rajankar, S.O.; Talbar, S.N. An electrocardiogram signal compression techniques: A comprehensive review. Analog Integr. Circuits Signal Process. 2019, 98, 59–74. [Google Scholar] [CrossRef]
- Bencherqui, A.; Daoui, A.; Karmouni, H.; Qjidaa, H.; Alfidi, M.; Sayyouri, M. Optimal reconstruction and compression of signals and images by Hahn moments and artificial bee Colony (ABC) algorithm. Multimed. Tools Appl. 2022, 81, 29753–29783. [Google Scholar] [CrossRef]
- Akkar, H.A.; Hadi, W.A.; Al-Dosari, I.H. A Squared-Chebyshev wavelet thresholding based 1D signal compression. Def. Technol. 2019, 15, 426–431. [Google Scholar] [CrossRef]
- Hosny, K.M.; Khalid, A.M.; Mohamed, E.R. Efficient compression of bio-signals by using Tchebichef moments and Artificial Bee Colony. Biocybern. Biomed. Eng. 2018, 38, 385–398. [Google Scholar] [CrossRef]
- Kumar Jha, C.; Kolekar, M.H. Diagnostic quality assured ECG signal compression with selection of appropriate mother wavelet for minimal distortion. IET Sci. Meas. Technol. 2019, 13, 500–508. [Google Scholar] [CrossRef]
- Jha, C.K.; Kolekar, M.H. Empirical mode decomposition and wavelet transform-based ECG data compression scheme. IRBM 2021, 42, 65–72. [Google Scholar] [CrossRef]
- Singhai, P.; Ateek, A.; Kumar, A.; Ansari, I.A.; Bhalerao, S. ECG Signal Compression based on Wavelet Parameterization and Thresholding using PSO. In Proceedings of the 2020 International Conference on Communication and Signal Processing (ICCSP), Chennai, India, 28–30 July 2020. [Google Scholar]
- Kolekar, M.H.; Jha, C.K.; Kumar, P. ECG Data Compression Using Modified Run Length Encoding of Wavelet Coefficients for Holter Monitoring. IRBM 2022, 43, 325–332. [Google Scholar] [CrossRef]
- Shi, J.; Wang, F.; Qin, M.; Chen, A.; Liu, W.; He, J.; Wang, H.; Chang, S.; Huang, Q. New ECG Compression Method for Portable ECG Monitoring System Merged with Binary Convolutional Auto-Encoder and Residual Error Compensation. Biosensors 2022, 12, 524. [Google Scholar] [CrossRef] [PubMed]
- Singhai, P.; Kumar, A.; Ateek, A.; Ansari, I.A.; Singh, G.K.; Lee, H.N. ECG Signal Compression Based on Optimization of Wavelet Parameters and Threshold Levels Using Evolutionary Techniques. Circuits Syst. Signal Process. 2023, 1–29. [Google Scholar] [CrossRef]
- Aznam, S.M.; Chowdhury, M.S.H. Generalized Haar wavelet operational matrix method for solving hyperbolic heat conduction in thin surface layers. Results Phys. 2018, 11, 243–252. [Google Scholar] [CrossRef]
- Khalid, A.M.; Hosny, K.M.; Mirjalili, S. COVIDOA: A novel evolutionary optimization algorithm based on coronavirus disease replication lifecycle. Neural Comput. Appl. 2022, 34, 22465–22492. [Google Scholar] [CrossRef]
- Mark, R.G.; Schluter, P.S.; Moody, G.B.; Devlin, P.H.; Chernoff, D. An annotated ECG database for evaluating arrhythmia detectors. IEEE Trans. Biomed. Eng. 1982, 29, 600. [Google Scholar]
- Schalk, G.; McFarland, D.J.; Hinterberger, T.; Birbaumer, N.; Wolpaw, J.R. BCI2000: A General-Purpose Brain-Computer Interface (BCI) System. IEEE Trans. Biomed. Eng. 2004, 51, 1034–1043. [Google Scholar] [CrossRef]


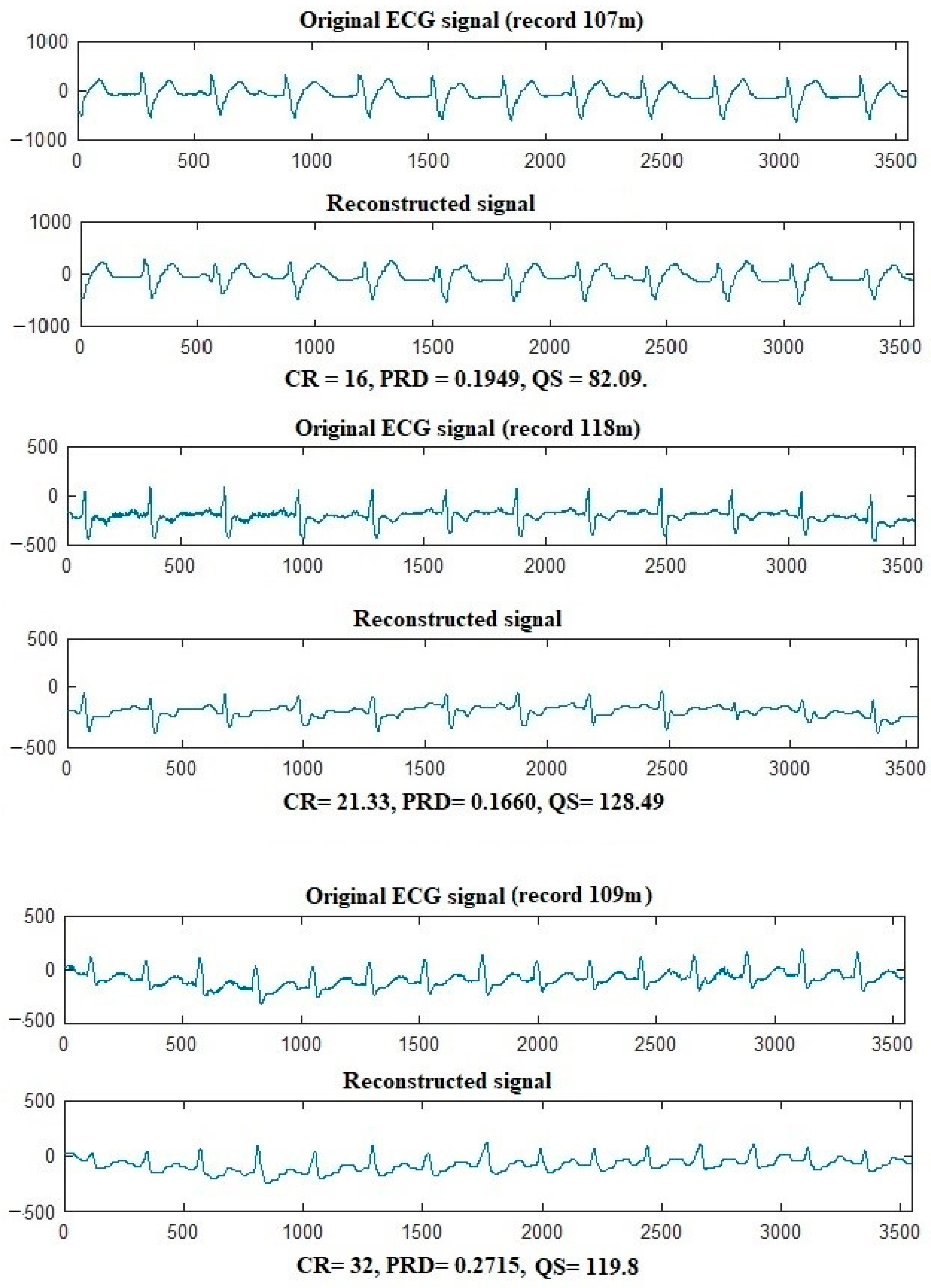

| Signal | Metric | |||
|---|---|---|---|---|
| CR | PRD | NCC | QS | |
| 100 | 25.6000 | 0.2764 | 0.9305 | 92.6194 |
| 101 | 21.3 | 0.2951 | 0.9173 | 72.2034 |
| 102 | 21.3 | 0.3700 | 0.9399 | 57.5676 |
| 103 | 10.6667 | 0.2628 | 0.9734 | 40.7137 |
| 104 | 16 | 0.2730 | 0.9833 | 58.6081 |
| 105 | 16 | 0.3051 | 0.9763 | 52.4590 |
| 106 | 8 | 0.2336 | 0.9890 | 34.2466 |
| 107 | 10.6667 | 0.1832 | 0.9909 | 58.2242 |
| 108 | 21.333 | 0.1790 | 0.9689 | 119.1788 |
| 109 | 32 | 0.2665 | 0.9436 | 119.8951 |
| 111 | 10.6667 | 0.2471 | 0.9805 | 43.1850 |
| 112 | 16 | 0.0968 | 0.9250 | 166.6667 |
| 113 | 10.6667 | 0.2584 | 0.9739 | 41.3438 |
| 114 | 21.3333 | 0.5272 | 0.9238 | 40.4647 |
| 115 | 21.333 | 0.3017 | 0.8993 | 70.8738 |
| 116 | 18.2857 | 0.2845 | 0.9188 | 64.3863 |
| 117 | 16 | 0.1453 | 0.9158 | 110.1170 |
| 118 | 21.333 | 0.1501 | 0.9455 | 142.2222 |
| 119 | 16 | 0.1511 | 0.9480 | 105.8901 |
| 121 | 21.3 | 0.1008 | 0.9452 | 211.3095 |
| 122 | 16 | 0.1660 | 0.9653 | 96.3855 |
| 123 | 21.3 | 0.1765 | 0.8928 | 120.6799 |
| 124 | 21.3 | 0.1856 | 0.9214 | 114.7629 |
| 200 | 21.3 | 0.3895 | 0.9301 | 54.6855 |
| 202 | 16 | 0.3518 | 0.9689 | 45.4804 |
| Average | 18.06 | 0.2470 | 0.9467 | 85.366 |
| Signal | Metric | |||
|---|---|---|---|---|
| CR | PRD | NCC | QS | |
| S001R01 | 8 | 0.4259 | 0.9145 | 18.7838 |
| S001R02 | 10.6667 | 0.4440 | 0.8993 | 24.0241 |
| S001R03 | 10.6667 | 0.4271 | 0.9257 | 24.9747 |
| S001R04 | 16 | 0.3911 | 0.9207 | 40.9103 |
| S001R05 | 10.6667 | 0.4481 | 0.8986 | 23.8027 |
| S001R06 | 16 | 0.4344 | 0.9085 | 36.8324 |
| S001R07 | 8 | 0.3580 | 0.9393 | 22.3464 |
| S001R08 | 10.6667 | 0.4130 | 0.9224 | 25.8274 |
| S001R09 | 10.6667 | 0.4134 | 0.9187 | 25.8024 |
| S001R10 | 16 | 0.4116 | 0.9161 | 38.8727 |
| S001R11 | 16 | 0.3595 | 0.9429 | 44.5063 |
| S001R12 | 10.6667 | 0.4148 | 0.9159 | 25.7153 |
| S001R13 | 8 | 0.3540 | 0.9381 | 22.5989 |
| S001R14 | 16 | 0.3424 | 0.9427 | 46.7290 |
| S002R01 | 16 | 0.4884 | 0.8829 | 32.7600 |
| S002R02 | 10.6667 | 0.4436 | 0.8907 | 24.0458 |
| S002R05 | 16 | 0.4753 | 0.8522 | 33.6629 |
| S003R01 | 16 | 0.4329 | 0.9105 | 36.9600 |
| S003R03 | 16 | 0.2423 | 0.9725 | 66.033 |
| S003R05 | 10.6667 | 0.3098 | 0.9623 | 34.4309 |
| Average | 12.6668 | 0.4014 | 0.9187 | 32.4809 |
| Algorithm | Metric | ||||
|---|---|---|---|---|---|
| CR | PRD | NCC | QS | ||
| 100 m | Proposed | 25.6000 | 0.2821 | 0.9005 | 90.7801 |
| Ref. [24] | 16 | 0.4058 | 0.5068 | 39.4283 | |
| Ref. [22] | 10.6667 | 0.6993 | 0.4181 | 15.2534 | |
| 101 | Proposed | 21.3 | 0.2951 | 0.9173 | 72.2034 |
| Ref. [24] | 21.3 | 0.4411 | 0.5791 | 48.2884 | |
| Ref. [22] | 21.3 | 0.9085 | 0.3474 | 23.4452 | |
| 112 | Proposed | 16 | 0.0968 | 0.9190 | 166.6667 |
| Ref. [24] | 16 | 0.1930 | 0.6022 | 82.9016 | |
| Ref. [22] | 12.8000 | 0.9045 | 0.1599 | 14.1515 | |
| 117 | Proposed | 16 | 0.1453 | 0.8954 | 110.1170 |
| Ref. [24] | 16 | 0.1903 | 0.6946 | 84.0778 | |
| Ref. [22] | 16 | 0.9120 | 0.1919 | 17.5439 | |
| 121 | proposed | 21.3 | 0.1008 | 0.9352 | 211.3095 |
| Ref. [24] | 21.3 | 0.2403 | 0.5872 | 88.6392 | |
| Ref. [22] | 21.3 | 0.9291 | 0.1553 | 22.9254 | |
| S001R14 | Proposed | 16 | 0.3424 | 0.9427 | 46.7290 |
| Ref. [24] | 16 | 0.3817 | 0.9317 | 41.9177 | |
| Ref. [22] | 16 | 0.6630 | 0.7470 | 24.1327 | |
| S003R03 | Proposed | 16 | 0.2423 | 0.9725 | 66.033 |
| Ref. [24] | 16 | 0.3368 | 0.9686 | 47.5059 | |
| Ref. [22] | 16 | 0.6574 | 0.7365 | 24.3383 | |
| Signal | CR | 8 | 10 | 16 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Algorithm | Proposed | Ref. [24] | Ref. [22] | Proposed | Ref. [24] | Ref. [22] | Proposed | Ref. [24] | Ref. [22] | |
| 100 | Time(s) | 16.8569 | 20.50271 | 43.0804 | 5.9904 | 5.2764 | 12.7438 | 5.1200 | 4.9127 | 10.5913 |
| 101 | Time(s) | 9.3482 | 18.44711 | 24.0403 | 5.3523 | 5.31273 | 10.3791 | 5.22571 | 5.9275 | 9.9437 |
| 102 | Time(s) | 20.92532 | 22.2531 | 42.5343 | 14.40836 | 14.8672 | 21.5439 | 12.74184 | 15.4386 | 18.3288 |
| Signal | CR | 8 | 10 | 16 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Algorithm | Proposed | Ref. [24] | Ref. [22] | Proposed | Ref. [24] | Ref. [22] | Proposed | Ref. [24] | Ref. [22] | |
| S003R03 | Time(s) | 19.84490 | 22.29458 | 60.8584 | 10.72566 | 17.24832 | 18.9420 | 6.588784 | 8.447046 | 8.36956 |
| S001R06 | Time(s) | 19.05750 | 32.05123 | 80.70620 | 11.41363 | 27.05330 | 34.86599 | 10.18366 | 17.933577 | 36.61682 |
| S002R01 | Time(s) | 22.52732 | 45.5811 | 91.5449 | 16.21887 | 25.7678 | 44.4210 | 15.2215 | 15.93446 | 32.3308 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khafaga, D.S.; Aldakheel, E.A.; Khalid, A.M.; Hamza, H.M.; Hosny, K.M. Compression of Bio-Signals Using Block-Based Haar Wavelet Transform and COVIDOA for IoMT Systems. Bioengineering 2023, 10, 406. https://doi.org/10.3390/bioengineering10040406
Khafaga DS, Aldakheel EA, Khalid AM, Hamza HM, Hosny KM. Compression of Bio-Signals Using Block-Based Haar Wavelet Transform and COVIDOA for IoMT Systems. Bioengineering. 2023; 10(4):406. https://doi.org/10.3390/bioengineering10040406
Chicago/Turabian StyleKhafaga, Doaa Sami, Eman Abdullah Aldakheel, Asmaa M. Khalid, Hanaa M. Hamza, and Khaid M. Hosny. 2023. "Compression of Bio-Signals Using Block-Based Haar Wavelet Transform and COVIDOA for IoMT Systems" Bioengineering 10, no. 4: 406. https://doi.org/10.3390/bioengineering10040406
APA StyleKhafaga, D. S., Aldakheel, E. A., Khalid, A. M., Hamza, H. M., & Hosny, K. M. (2023). Compression of Bio-Signals Using Block-Based Haar Wavelet Transform and COVIDOA for IoMT Systems. Bioengineering, 10(4), 406. https://doi.org/10.3390/bioengineering10040406







