An Effective Chemical Permeabilization of Silkworm Embryos
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Eggs and Embryonic Stages
2.2. Dechorionation and Permeabilization Processes
2.3. Rhodamine B Staining
2.4. Osmotic Response of Embryos to Sucrose and Ethylene Glycol (EG), and Data Analysis
2.5. EG Treatment in Viability Assay
2.6. Assessment of Development and Viability
2.7. Statistical Analysis
3. Results
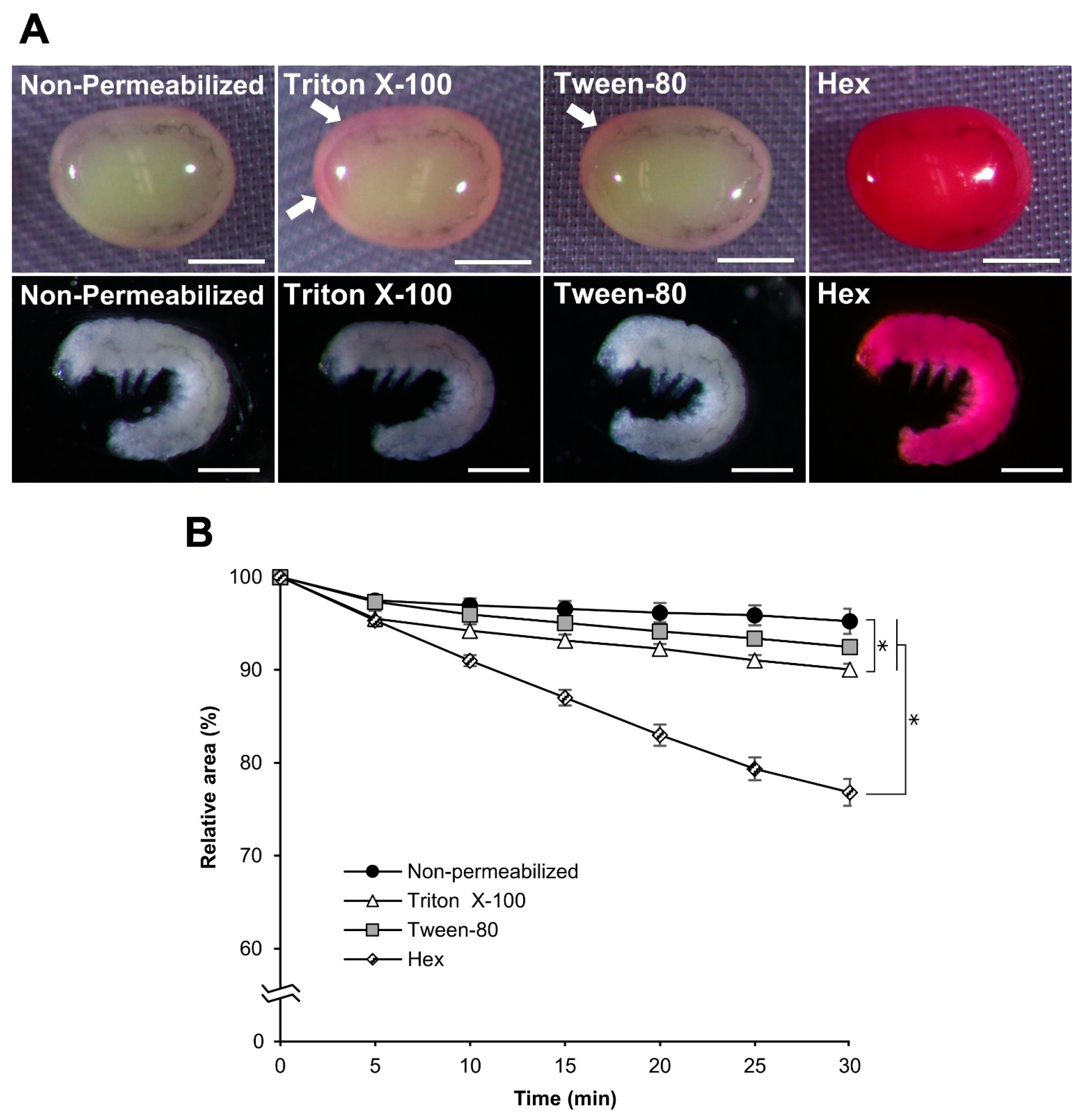
3.1. Improvements in the Permeabilization Process
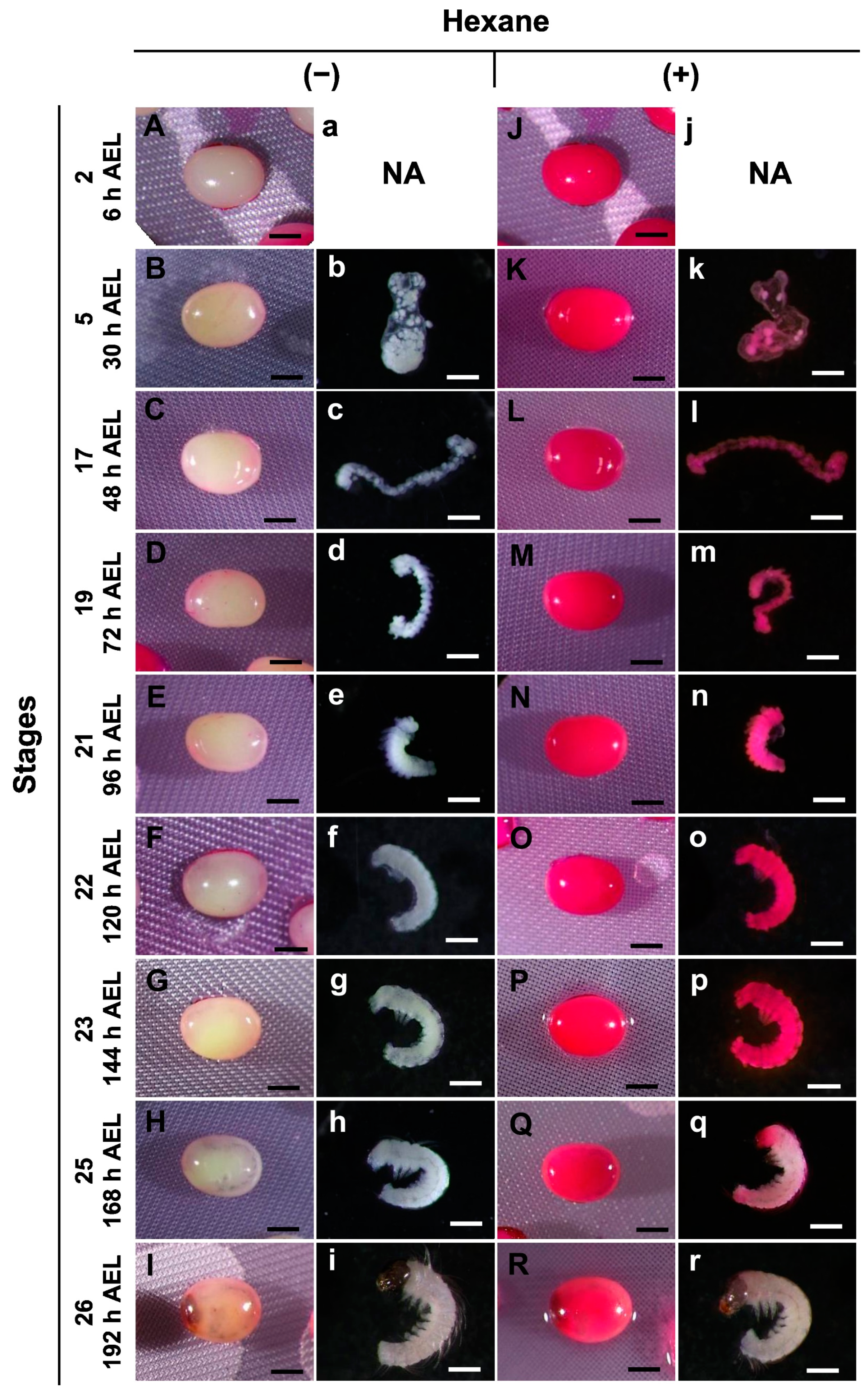
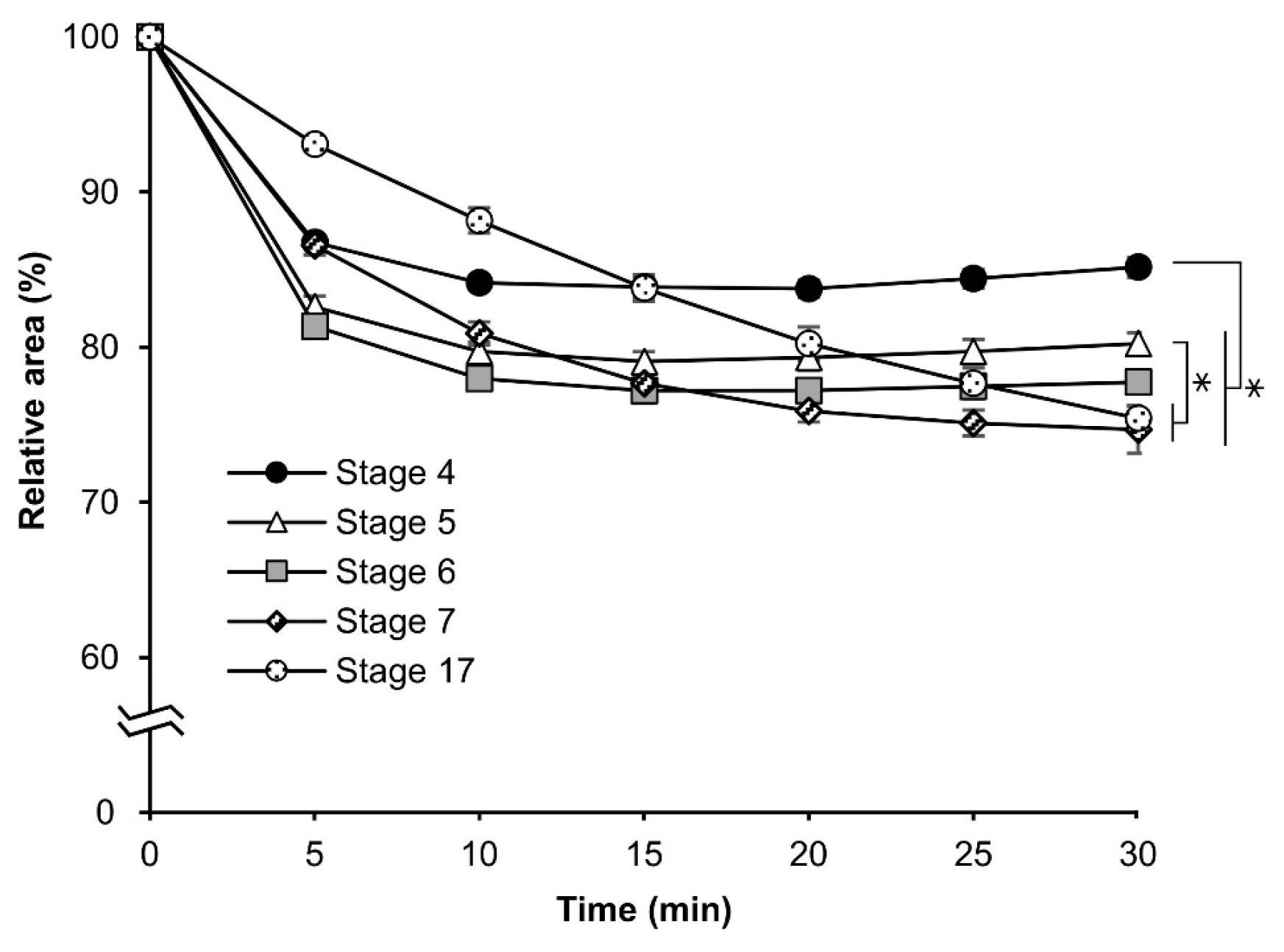
3.2. Permeability at Different Embryonic Stages
3.3. Osmotic Response of Embryos to Sucrose
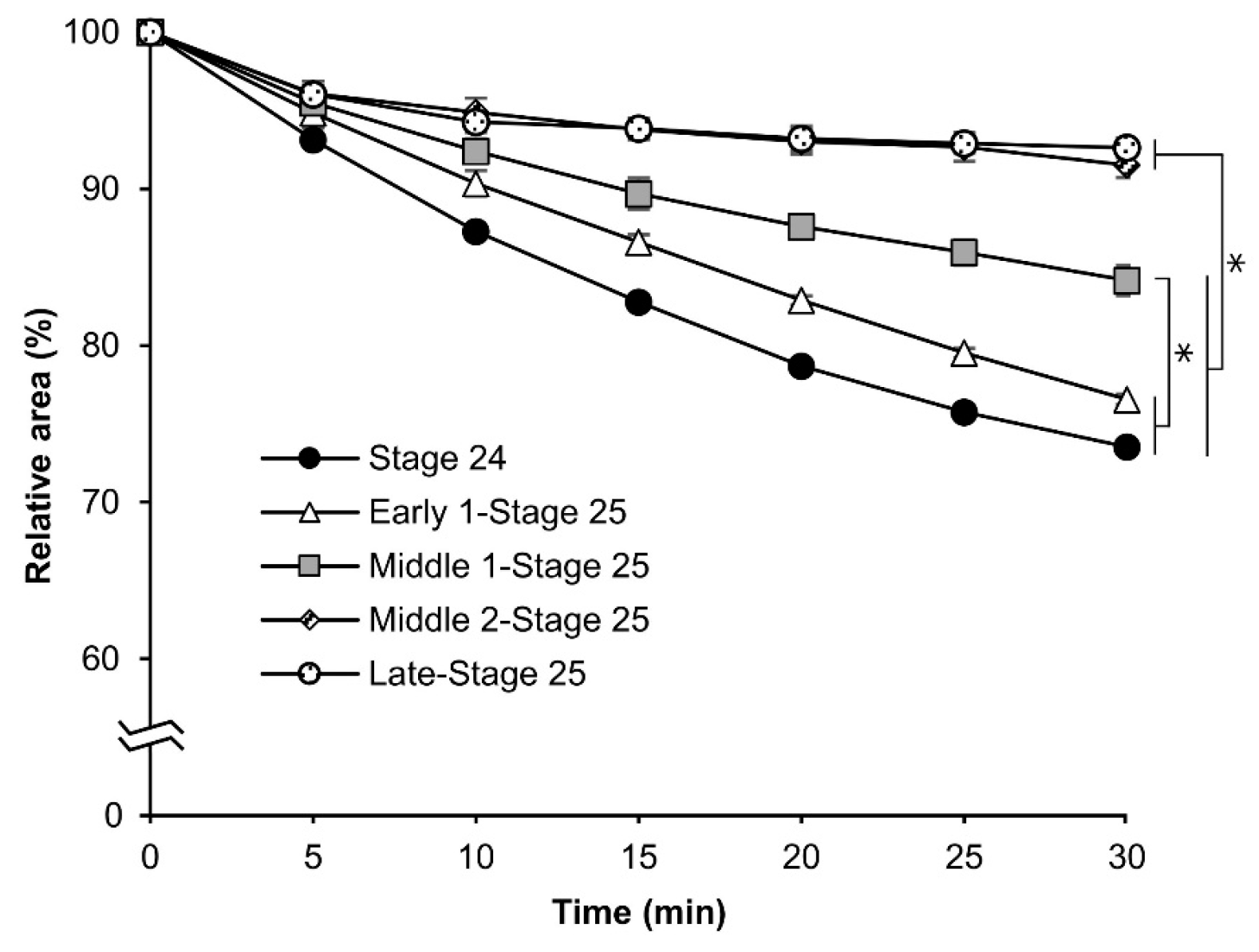
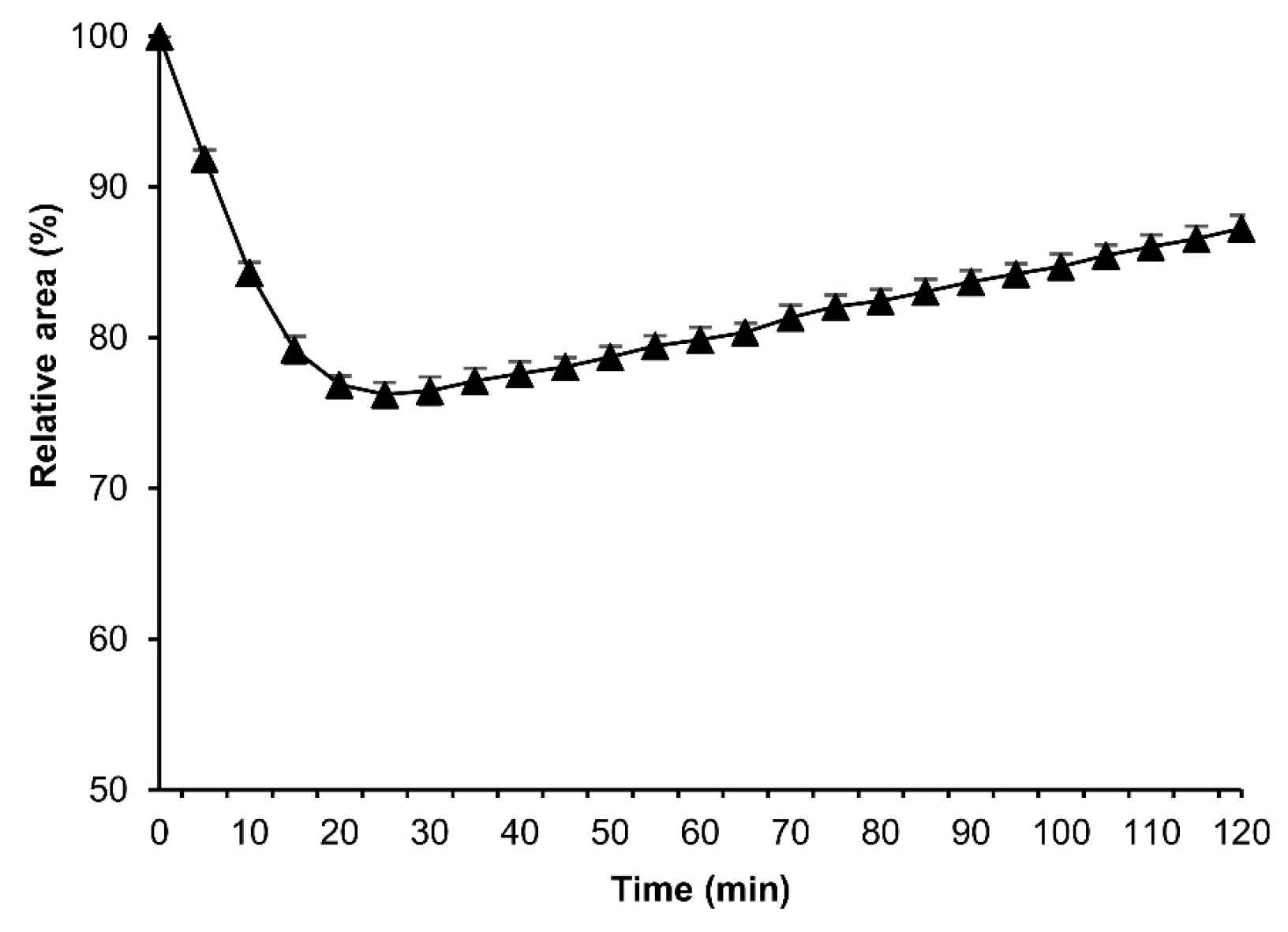
3.4. More Detailed Analysis of the Osmotic Response during Embryonic Developmental Stages
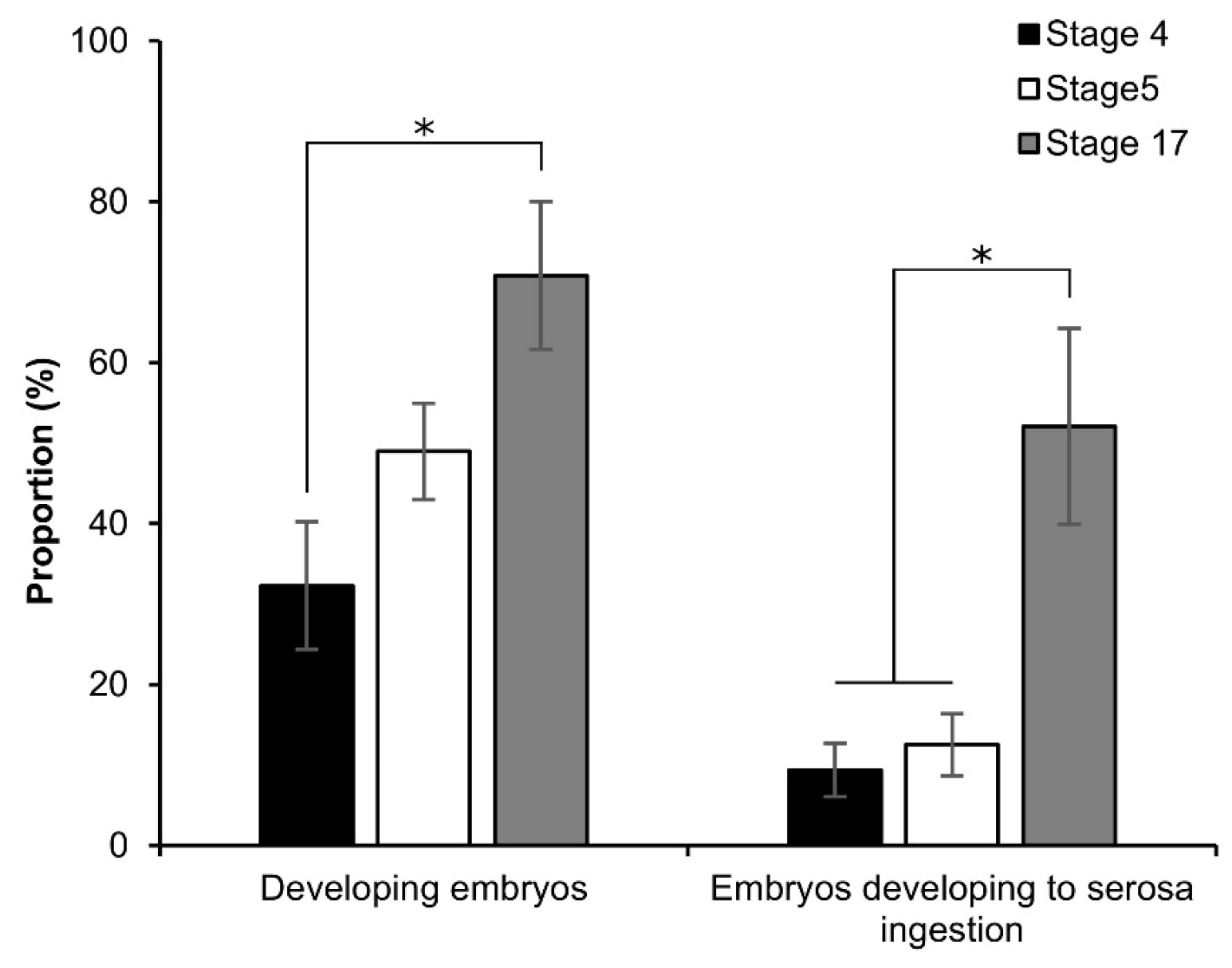
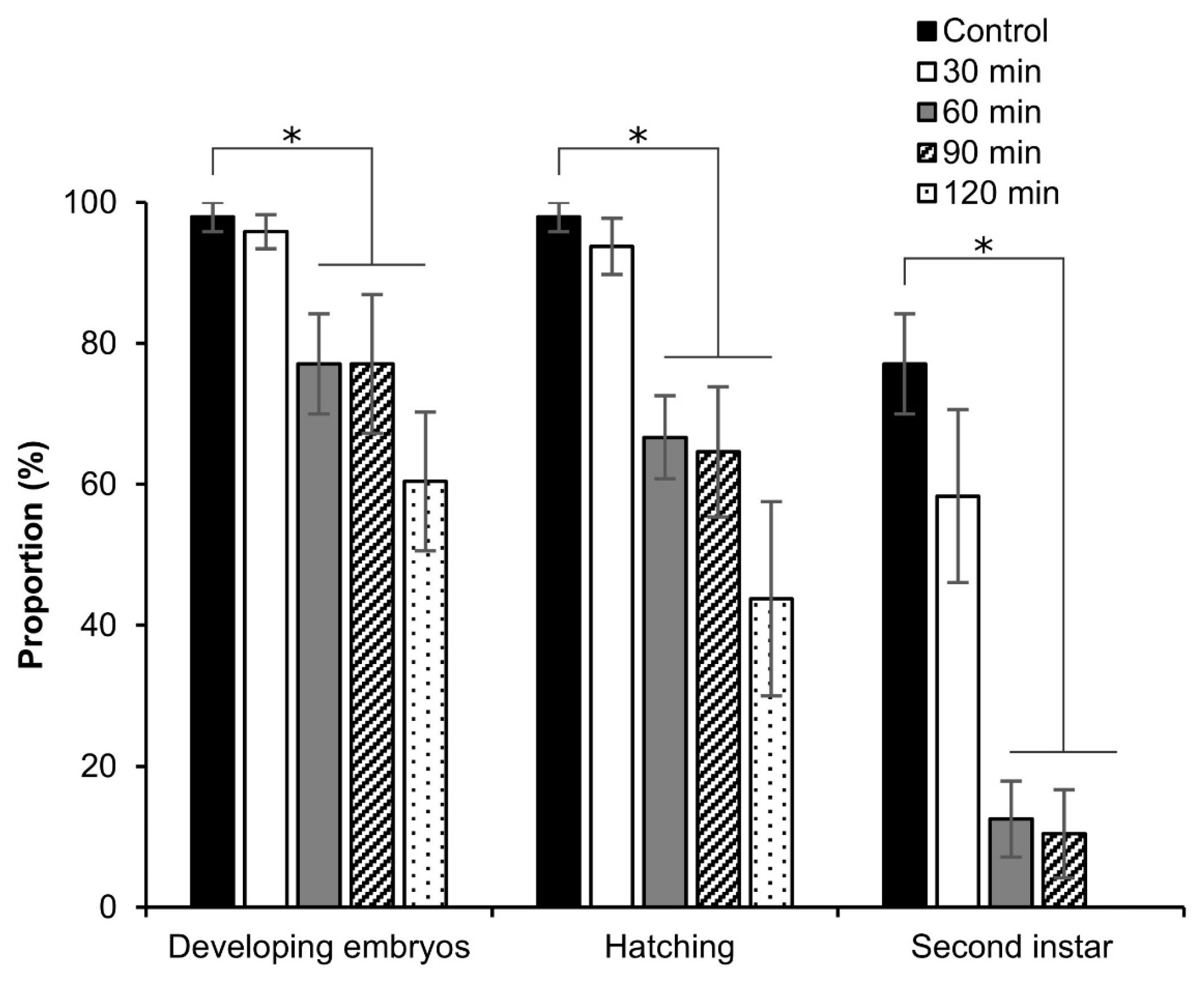
3.5. Viability of Embryos after Permeabilization
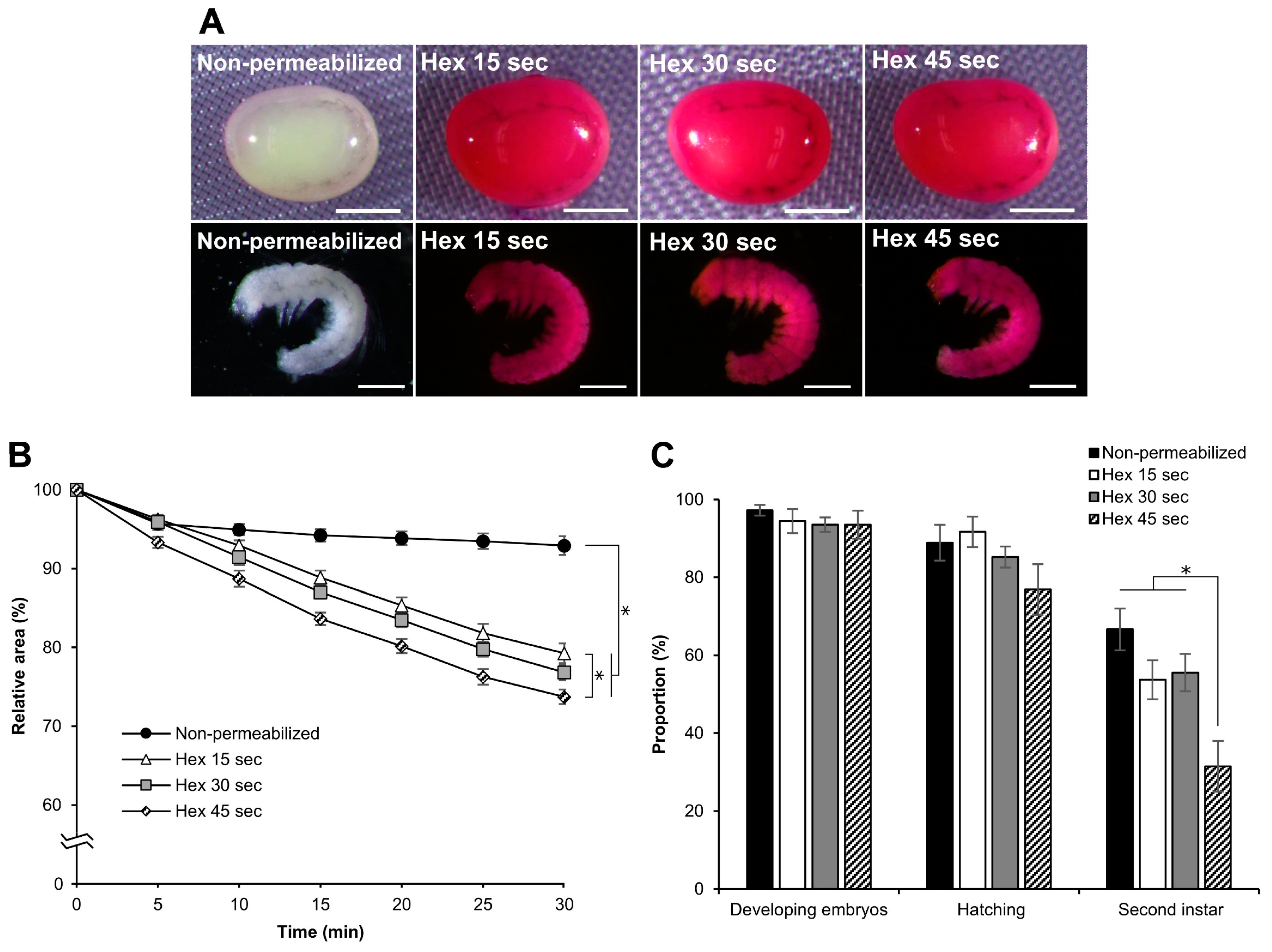
3.6. Effect of Hexane Exposure Time
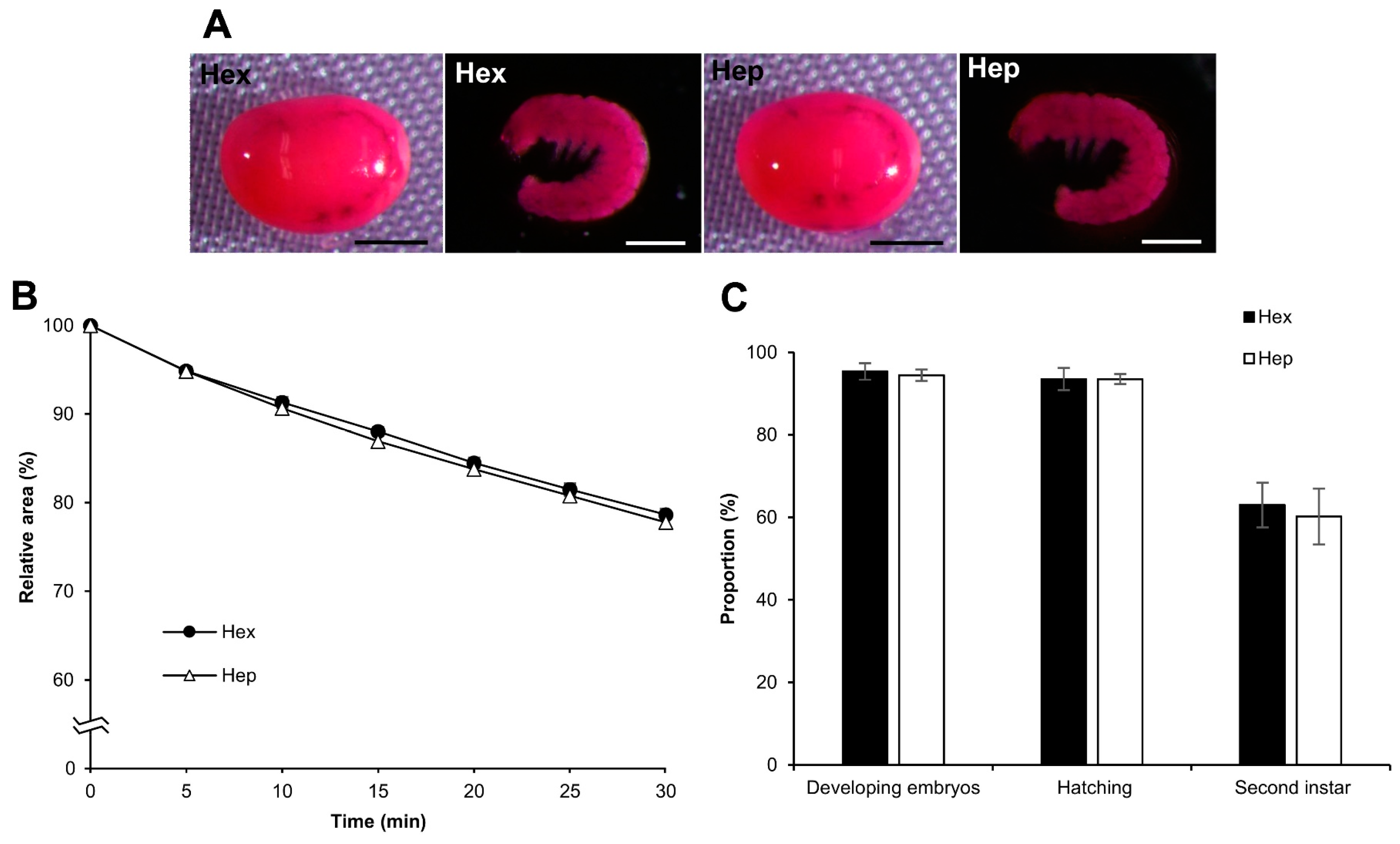
3.7. Effect of Permeabilization with Chemicals Other Than Hexane
3.8. Effect on Different Strains: Diapausing
3.9. Osmotic Response of Embryos to EG and Viability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Atsumi, S.; Miyamoto, K.; Yamamoto, K.; Narukawa, J.; Kawai, S.; Sezutsu, H.; Kobayashi, I.; Uchino, K.; Tamura, T.; Mita, K.; et al. Single amino acid mutation in ATP-binding cassette transporter gene causes resistance to Bt toxin Cry1Ab in the silkworm, Bombyx mori. Proc. Natl. Acad. Sci. USA 2012, 109, E1591–E1598. [Google Scholar] [CrossRef] [PubMed]
- Sajwan, S.; Takasu, Y.; Tamura, T.; Uchino, K.; Sezutsu, H.; Zurovec, M. Efficient disruption of endogenous Bombyx gene by TAL effector nucleases. Insect Biochem. Mol. Biol. 2013, 43, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Sezutsu, H.; Uchino, K.; Kobayashi, I.; Tatematsu, K.I.; Iizuka, T.; Yonemura, N.; Tamura, T. Conservation of fibroin gene promoter function between the domesticated silkworm Bombyx mori and the wild silkmoth Antheraea yamamai. J. Insect Biotechnol. Sericol. 2009, 78, 1_1–1_10. [Google Scholar]
- Tamura, T.; Thibert, C.; Royer, C.; Kanda, T.; Eappen, A.; Kamba, M.; Kômoto, N.; Thomas, J.L.; Mauchamp, B.; Chavancy, G.; et al. Germline transformation of the silkworm Bombyx mori L. using a piggyBac transposon-derived vector. Nat. Biotechnol. 2000, 18, 81–84. [Google Scholar] [CrossRef] [PubMed]
- Uchino, K.; Sezutsu, H.; Imamura, M.; Kobayashi, I.; Tatematsu, K.I.; Iizuka, T.; Yonemura, N.; Mita, K.; Tamura, T. Construction of a piggyBac-based enhancer trap system for the analysis of gene function in silkworm Bombyx mori. Insect Biochem. Mol. Biol. 2008, 38, 1165–1173. [Google Scholar] [CrossRef]
- Yonemura, N.; Tamura, T.; Uchino, K.; Kobayashi, I.; Tatematsu, K.; Iizuka, T.; Sezutsu, H.; Muthulakshmi, M.; Nagaraju, J.; Kusakabe, T. PhiC31 integrase-mediated cassette exchange in silkworm embryos. Mol. Genet. Genom. 2012, 287, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Mochida, Y.; Takemura, Y.; Kanda, T.; Horie, Y. Oviposition by female moth transplanted ovary frozen in combination with artificial insemination with frozen sperm in the silkworm, Bombyx mori and their offspring. J. Seric. Sci. Jpn. 1999, 68, 139–144. (In Japanese) [Google Scholar]
- Mochida, Y.; Takemura, Y.; Kanda, T.; Horie, Y. Fertilized eggs obtained from transplantation of frozen ovaries and parthenogenesis in combination with artificial insemination of frozen semen of the silkworm, Bombyx mori. Cryobiology 2003, 46, 153–160. [Google Scholar] [CrossRef]
- Mochida, Y.; Takemura, Y.; Ohnuma, A.; Kanekatsu, R.; Kiguchi, K. Fertilization of eggs developed from the cryopreserved and transplanted ovaries by artificial insemination with cryopreserved semen in the silkworm, Bombyx mori. J. Insect Biotechnol. Sericol. 2007, 76, 97–100. [Google Scholar]
- Takemura, Y.; Kanda, T.; Horie, Y. Artificial insemination using cryopreserved sperm in the silkworm, Bombyx mori. J. Insect Physiol. 2000, 46, 491–497. [Google Scholar] [CrossRef]
- Banno, Y.; Nagasaki, K.; Tsukada, M.; Minohara, Y.; Banno, J.; Nishikawa, K.; Yamamoto, K.; Tamura, K.; Fujii, T. Development of a method for long-term preservation of Bombyx mori silkworm strains using frozen ovaries. Cryobiology 2013, 66, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Beament, J.W.L. The waterproofing process in eggs of Rhodnius prolixus Stähl. Proc. R. Soc. Lond. B Biol. Sci. 1946, 133, 407–418. [Google Scholar]
- Abidalla, M.; Roversi, P.F. First estimation of Drosophila EPS solution for permeabilizing Lepidoptera Galleria mellonella embryos. Adv. Entomol. 2018, 6, 213–225. [Google Scholar]
- Barbier, R.; Chauvin, G. Ultrastructure et rôle des aéropyles et des enveloppes de l’œuf de Galleria mellonella. J. Insect Physiol. 1974, 20, 809–820. [Google Scholar] [CrossRef]
- Cruickshank, W.J. Ultrastructural modifications in the follicle cells and egg membranes during development of flour moth oöcytes. J. Insect Physiol. 1972, 18, 485–498. [Google Scholar] [CrossRef]
- Margaritis, L.H.; Kafatos, F.C.; Petri, W.H. The eggshell of Drosophila melanogaster. I. Fine structure of the layers and regions of the wild-type eggshell. J. Cell Sci. 1980, 43, 1–35. [Google Scholar] [CrossRef]
- Margaritis, L.H. Structure and physiology of the eggshell. In Comprehensive Insect Physiology, Biochemistry and Pharmacology, 1st ed.; Kerkut, G.A., Gilbert, L.I., Eds.; Pergamon Press: Oxford, UK, 1985; Volume 1, pp. 153–230. [Google Scholar]
- Nelson, D.R.; Leopold, R.A. Composition of the surface hydrocarbons from the vitelline membranes of dipteran embryos. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2003, 136, 295–308. [Google Scholar] [CrossRef]
- Papassideri, I.; Margaritis, L.H.; Gulik-Krzywicki, T. The egg-shell of Drosophila melanogaster VI, structural analysis of the wax layer in laid eggs. Tissue Cell 1991, 23, 567–575. [Google Scholar] [CrossRef]
- Papassideri, I.S.; Margaritis, L.H.; Gulik-Krzywicki, T. The eggshell of Drosophila melanogaster. VIII. Morphogenesis of the wax layer during oogenesis. Tissue Cell 1993, 25, 929–936. [Google Scholar] [CrossRef]
- Telfer, W.H. Egg formation in Lepidoptera. J. Insect Sci. 2009, 9, 1–21. [Google Scholar] [CrossRef]
- Woods, H.A.; Bonnecaze, R.T.; Zrubek, B. Oxygen and water flux across eggshells of Manduca sexta. J. Exp. Biol. 2005, 208, 1297–1308. [Google Scholar] [CrossRef] [PubMed]
- Mazur, P.; Cole, K.W.; Mahowald, A.P. Critical factors affecting the permeabilization of Drosophila embryos by alkanes. Cryobiology 1992, 29, 210–239. [Google Scholar] [CrossRef]
- Mazur, P.; Cole, K.W.; Hall, J.W.; Schreuders, P.D.; Mahowald, A.P. Cryobiological preservation of Drosophila embryos. Science 1992, 258, 1932–1935. [Google Scholar] [CrossRef] [PubMed]
- Rajamohan, A.; Leopold, R.A. Cryopreservation of Mexican fruit flies by vitrification: Stage selection and avoidance of thermal stress. Cryobiology 2007, 54, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Rajamohan, A.; Rinehart, J.P.; Leopold, R.A. Stage selection and restricted oviposition period improves cryopreservation of dipteran embryos. Cryobiology 2015, 70, 143–149. [Google Scholar] [CrossRef]
- Zhan, L.; Li, M.; Hays, T.; Bischof, J. Cryopreservation method for Drosophila melanogaster embryos. Nat. Commun. 2021, 12, 2412. [Google Scholar] [CrossRef]
- Rajamohan, A.; Rinehart, J.P.; Foster, S.P.; Leopold, R.A. Permeability barriers to embryo cryopreservation of Pectinophora gossypiella (Lepidoptera: Gelechiidae). J. Econ. Entomol. 2013, 106, 855–861. [Google Scholar] [CrossRef]
- Roversi, P.F.; Cosi, E.; Irdani, T. Chill sensitivity and cryopreservation of eggs of the greater wax moth Galleria mellonella (Lepidoptera: Pyralidae). Cryobiology 2008, 56, 1–7. [Google Scholar] [CrossRef]
- Cosi, E.; Abidalla, M.T.; Roversi, P.F. The effect of Tween 80 on eggshell permeabilization in Galleria mellonella (L.) (Lepidoptera, Pyralidae). CryoLetters 2010, 31, 291–300. [Google Scholar]
- Urbán-Duarte, D.; Tomita, S.; Sakai, H.; Sezutsu, H.; De La Torre-Sánchez, J.F.; Kainoh, Y.; Furukawa, S.; Uchino, K. Effect of chemical dechorionation on silkworm embryo viability. J. Insect Physiol. 2022, 137, 104327. [Google Scholar] [CrossRef]
- Okada, M. Role of the chorion as a barrier to oxygen in the diapause of the silkworm, Bombyx mori L. Experientia 1971, 27, 658–660. [Google Scholar] [CrossRef] [PubMed]
- Rand, M.D.; Kearney, A.L.; Dao, J.; Clason, T. Permeabilization of Drosophila embryos for introduction of small molecules. Insect Biochem. Mol. Biol. 2010, 40, 792–804. [Google Scholar] [CrossRef] [PubMed]
- Abidalla, M.; Cosi, E.; Cappellozza, S.; Saviane, A.; Pagano, J.; Battaglia, D.; Roversi, P.F. Preliminary studies on the cryopreservation of silkworm (Bombyx mori) eggs. Bull. Insectology 2019, 72, 287–296. [Google Scholar]
- Miya, K. The Early Embryonic Development of Bombyx mori—An Ultrastructural Point of View; Gendaitosho: Sagamihara, Japan, 2003; pp. 5–47. [Google Scholar]
- Rajamohan, A.; Rinehart, J.P.; Leopold, R.A. Cryopreservation of embryos of Lucilia sericata (Diptera: Calliphoridae). J. Med. Entomol. 2014, 51, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.; Kawai, Y.; Hara, T.; Takeda, S.; Seki, S.; Nakata, Y.I.; Matsukawa, K.; Koshimoto, C.; Kasai, M.; Edashige, K. Pathway for the movement of water and cryoprotectants in bovine oocytes and embryos. Biol. Reprod. 2011, 85, 834–847. [Google Scholar] [CrossRef]
- Jin, B.; Higashiyama, R.I.; Nakata, Y.I.; Yonezawa, J.I.; Xu, S.; Miyake, M.; Takahashi, S.; Kikuchi, K.; Yazawa, K.; Mizobuchi, S.; et al. Rapid movement of water and cryoprotectants in pig expanded blastocysts via channel processes: Its relevance to their higher tolerance to cryopreservation. Biol. Reprod. 2013, 89, 87. [Google Scholar] [CrossRef] [PubMed]
- Pedro, P.B.; Yokoyama, E.; Zhu, S.E.; Yoshida, N.; Valdez Jr, D.M.; Tanaka, M.; Edashige, K.; Kasai, M. Permeability of mouse oocytes and embryos at various developmental stages to five cryoprotectants. J. Reprod. Dev. 2005, 51, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Sonobe, H.; Matsumoto, A.; Fukuzaki, Y.; Fujiwara, S. Carbohydrate metabolism and restricted oxygen supply in the eggs of the silkworm, Bombyx mori. J. Insect Physiol. 1979, 25, 381–388. [Google Scholar] [CrossRef]
- Lin, T.T.; Pitt, R.E.; Steponkus, P.L. Osmometric behavior of Drosophila melanogaster embryos. Cryobiology 1989, 26, 453–471. [Google Scholar] [CrossRef]
- Zhang, T.; Isayeva, A.; Adams, S.L.; Rawson, D.M. Studies on membrane permeability of zebrafish (Danio rerio) oocytes in the presence of different cryoprotectants. Cryobiology 2005, 50, 285–293. [Google Scholar] [CrossRef]
- Shu, Z.; Hughes, S.M.; Fang, C.; Huang, J.; Fu, B.; Zhao, G.; Fialkow, M.; Lentz, G.; Hladik, F.; Gao, D. A study of the osmotic characteristics, water permeability, and cryoprotectant permeability of human vaginal immune cells. Cryobiology 2016, 72, 93–99. [Google Scholar] [CrossRef]
- Van den Abbeel, E.; Schneider, U.; Liu, J.; Agca, Y.; Critser, J.K.; Van Steirteghem, A. Osmotic responses and tolerance limits to changes in external osmolalities, and oolemma permeability characteristics, of human in vitro matured MII oocytes. Hum. Reprod. 2007, 22, 1959–1972. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbán-Duarte, D.; Tomita, S.; Sakai, H.; Sezutsu, H.; De La Torre-Sánchez, J.F.; Kainoh, Y.; Furukawa, S.; Uchino, K. An Effective Chemical Permeabilization of Silkworm Embryos. Bioengineering 2023, 10, 563. https://doi.org/10.3390/bioengineering10050563
Urbán-Duarte D, Tomita S, Sakai H, Sezutsu H, De La Torre-Sánchez JF, Kainoh Y, Furukawa S, Uchino K. An Effective Chemical Permeabilization of Silkworm Embryos. Bioengineering. 2023; 10(5):563. https://doi.org/10.3390/bioengineering10050563
Chicago/Turabian StyleUrbán-Duarte, David, Shuichiro Tomita, Hiroki Sakai, Hideki Sezutsu, José Fernando De La Torre-Sánchez, Yooichi Kainoh, Seiichi Furukawa, and Keiro Uchino. 2023. "An Effective Chemical Permeabilization of Silkworm Embryos" Bioengineering 10, no. 5: 563. https://doi.org/10.3390/bioengineering10050563
APA StyleUrbán-Duarte, D., Tomita, S., Sakai, H., Sezutsu, H., De La Torre-Sánchez, J. F., Kainoh, Y., Furukawa, S., & Uchino, K. (2023). An Effective Chemical Permeabilization of Silkworm Embryos. Bioengineering, 10(5), 563. https://doi.org/10.3390/bioengineering10050563






