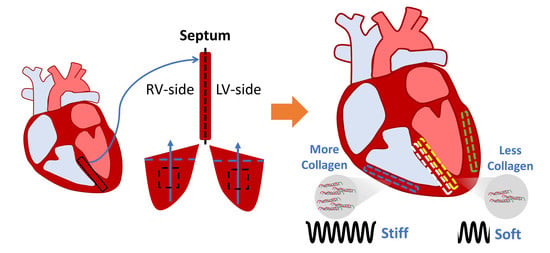
The Interventricular Septum Is Biomechanically Distinct from the Ventricular Free Walls
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
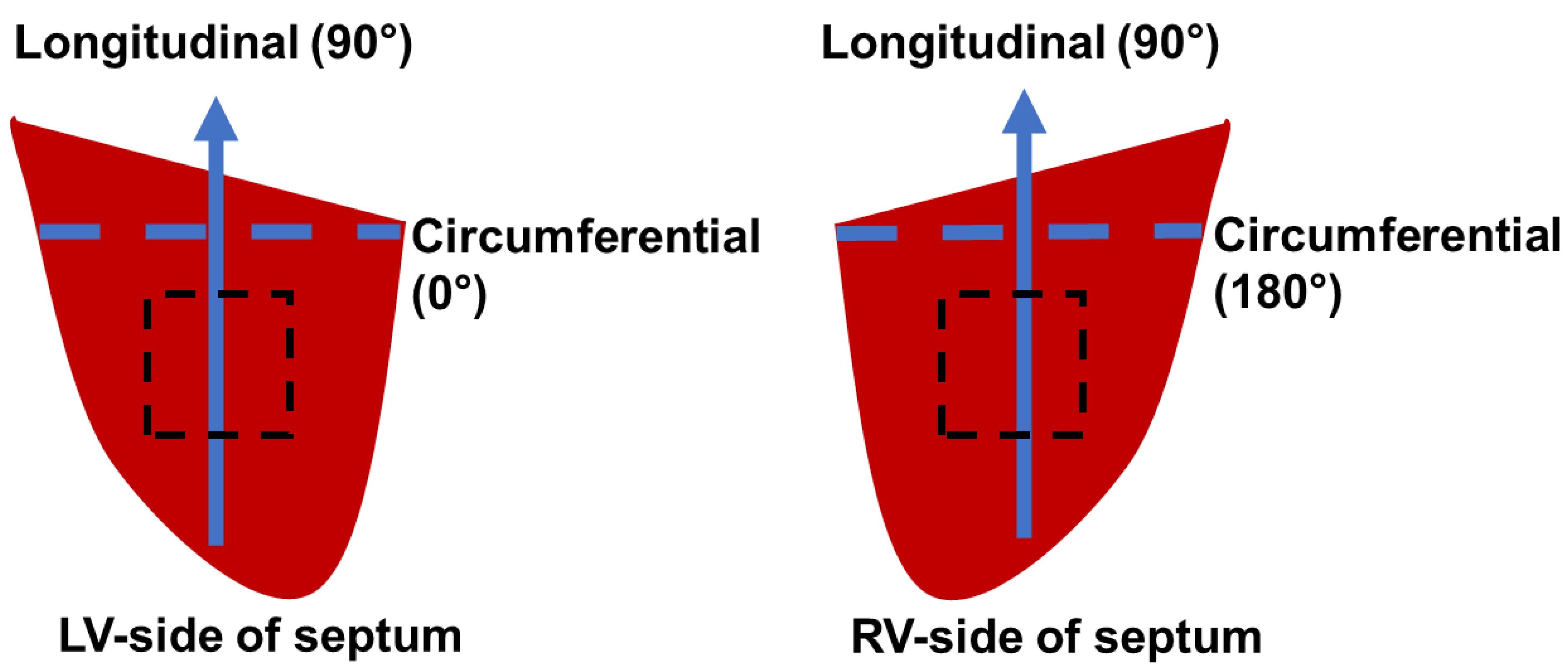
2.2. Biaxial Testing
2.3. Constitutive Modeling
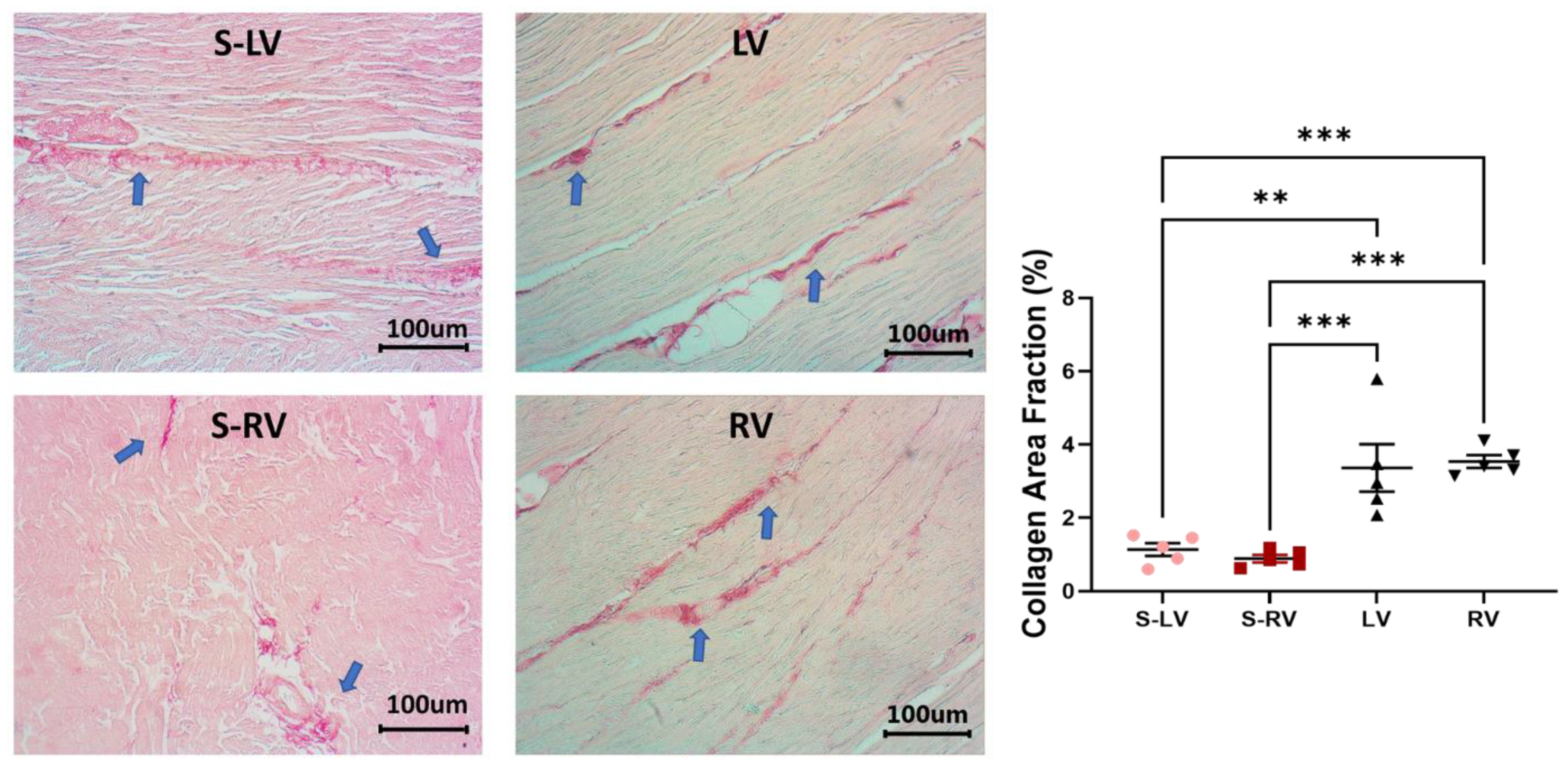
2.4. Histology
2.5. Statistical Analysis
3. Results
3.1. Transmural Change in Septum Myofiber Orientation
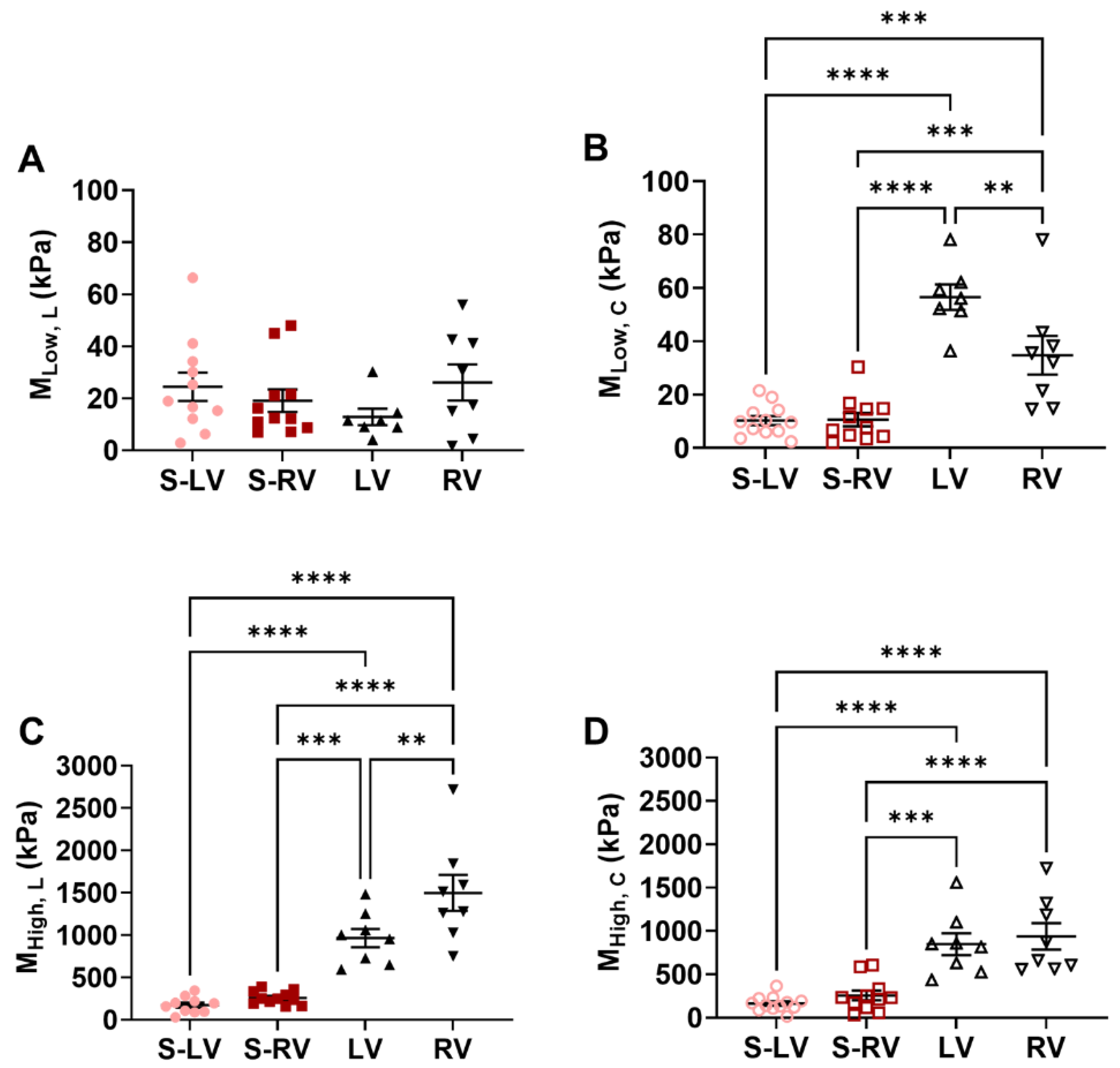
3.2. Softer Septal Side Compared to the Corresponding Side of Ventricular Free Wall
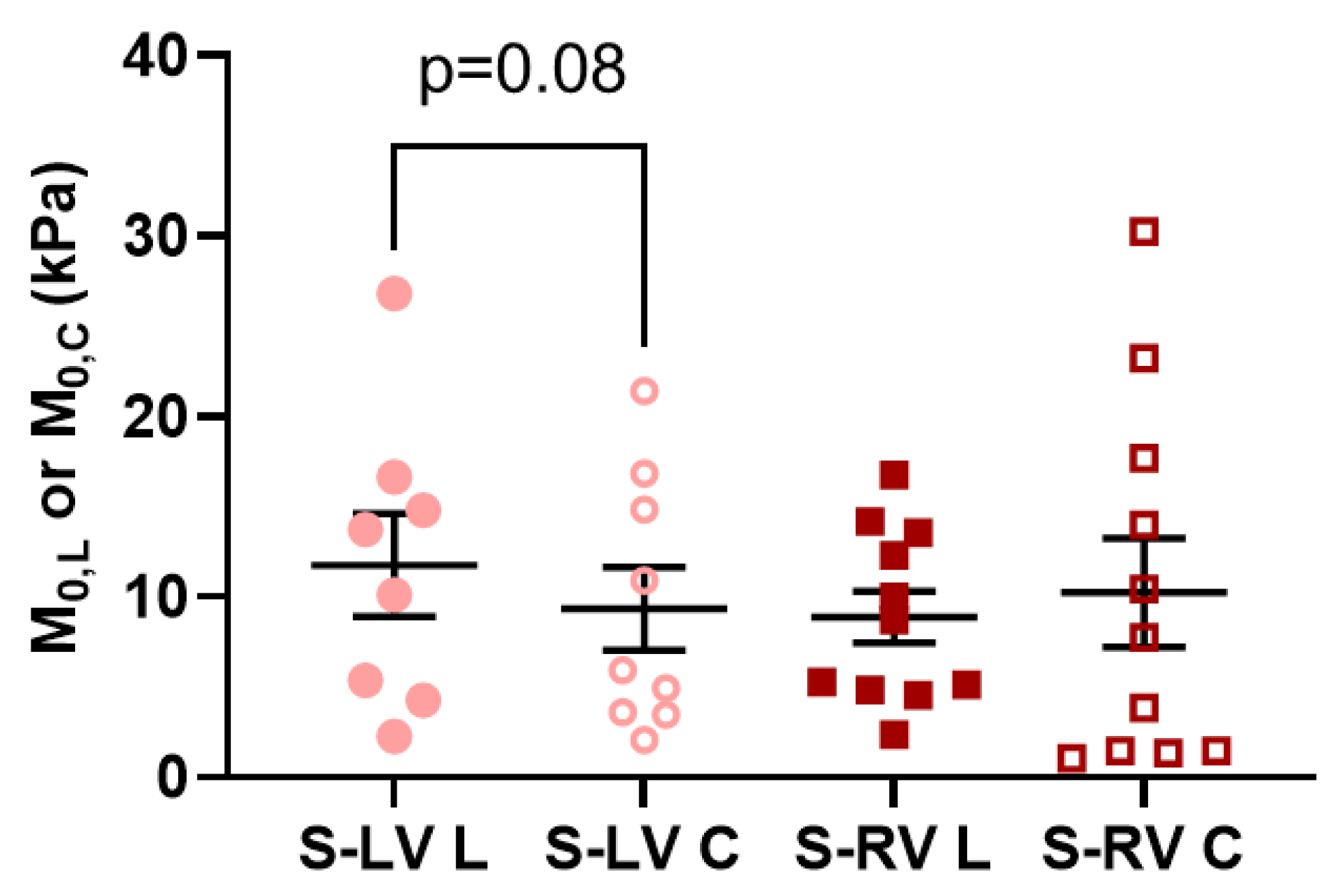
3.3. Transmural Differences in the Septum Obtained from Experimental Data
3.4. Transmural Differences in the Septum Obtained from Computational Modeling
3.5. Difference in Collagen Content between Septum and Ventricular Tissues
4. Discussion
4.1. Different Mechanical Behavior between Ventricle Free Walls and Septum
4.2. Fiber Orientation in the Septum
4.3. Anisotropy Behavior of the Septum
4.4. Transmural Differences between LV-Side and RV-Side of the Septum
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Inamdar, A.A.; Inamdar, A.C. Heart failure: Diagnosis, management and utilization. J. Clin. Med. 2016, 5, 62. [Google Scholar] [CrossRef]
- Flachskampf, F.A.; Voigt, J.U. The interventricular septum is functionally bilayered: A fresh look at a well known structure. Heart 2005, 91, 1260–1261. [Google Scholar] [CrossRef]
- Damiano, R.J., Jr.; La Follette, P., Jr.; Cox, J.L.; Lowe, J.E.; Santamore, W.P. Significant left ventricular contribution to right ventricular systolic function. Am. J. Physiol. 1991, 261 Pt 2, H1514–H1524. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, D.; Sisto, D.; Frater, R.W.; Nikolic, S.D. Left-to-right ventricular interaction with a noncontracting right ventricle. J Thorac. Cardiovasc. Surg. 1994, 107, 1496–1502. [Google Scholar] [CrossRef]
- Sawatani, S.; Mandell, G.; Kusaba, E.; Schraut, W.; Cascade, P.; Wajszczuk, W.J.; Kantrowitz, A. Ventricular performance following ablation and prosthetic replacement of right ventricular myocardium. Trans. Am. Soc. Artif. Intern. Organs 1974, 20, 629–636. [Google Scholar] [PubMed]
- Agarwal, J.B.; Yamazaki, H.; Bodenheimer, M.M.; Banka, V.S.; Helfant, R.H. Effects of isolated interventricular septal ischemia on global and segmental function of the canine right and left ventricle. Am. Heart J. 1981, 102, 654–658. [Google Scholar] [CrossRef]
- Li, K.S.; Santamore, W.P. Contribution of each wall to biventricular function. Cardiovasc. Res. 1993, 27, 792–800. [Google Scholar] [CrossRef]
- Buckberg, G.; Hoffman, J.I. Right ventricular architecture responsible for mechanical performance: Unifying role of ventricular septum. J. Thorac. Cardiovasc. Surg. 2014, 148, 3166–3171.e4. [Google Scholar] [CrossRef]
- Friedberg, M.K.; Redington, A.N. Right versus left ventricular failure: Differences, similarities, and interactions. Circulation 2014, 129, 1033–1044. [Google Scholar] [CrossRef]
- Dwivedi, A.; Axel, L. Abnormal Motion Patterns of the Interventricular Septum. JACC Cardiovasc. Imaging 2017, 10 Pt B, 1281–1284. [Google Scholar] [CrossRef]
- Boettler, P.; Claus, P.; Herbots, L.; McLaughlin, M.; D’Hooge, J.; Bijnens, B.; Ho, S.Y.; Kececioglu, D.; Sutherland, G.R. New aspects of the ventricular septum and its function: An echocardiographic study. Heart 2005, 91, 1343–1348. [Google Scholar] [CrossRef]
- Holland, M.R.; Gibson, A.A.; Bauer, A.Q.; Peterson, L.R.; Schaffer, J.E.; Bach, R.G.; Cresci, S.; Miller, J.G. Echocardiographic tissue characterization demonstrates differences in the left and right sides of the ventricular septum. Ultrasound Med. Biol. 2010, 36, 1653–1661. [Google Scholar] [CrossRef]
- Lindqvist, P.; Mörner, S.; Karp, K.; Waldenström, A. New Aspects of Septal Function by Using 1-Dimensional Strain and Strain Rate Imaging. J. Am. Soc. Echocardiogr. 2006, 19, 1345–1349. [Google Scholar] [CrossRef]
- Novak, V.P.; Yin, F.C.P.; Humphrey, J.D. Regional mechanical properties of passive myocardium. J. Biomech. 1994, 27, 403–412. [Google Scholar] [CrossRef]
- Sommer, G.; Schriefl, A.J.; Andrä, M.; Sacherer, M.; Viertler, C.; Wolinski, H.; Holzapfel, G.A. Biomechanical properties and microstructure of human ventricular myocardium. Acta Biomater. 2015, 24 (Suppl. C), 172–192. [Google Scholar] [CrossRef] [PubMed]
- Camacho, P.; Fan, H.; Liu, Z.; He, J.Q. Large Mammalian Animal Models of Heart Disease. J. Cardiovasc. Dev. Dis. 2016, 3, 30. [Google Scholar] [CrossRef] [PubMed]
- Silva, K.A.S.; Emter, C.A. Large Animal Models of Heart Failure: A Translational Bridge to Clinical Success. JACC Basic Transl. Sci. 2020, 5, 840–856. [Google Scholar] [CrossRef] [PubMed]
- Annabi, M.R.; Kerndt, C.C.; Makaryus, A.N. Embryology, Atrioventricular Septum; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Tan, C.M.J.; Lewandowski, A.J. The Transitional Heart: From Early Embryonic and Fetal Development to Neonatal Life. Fetal Diagn. Ther. 2020, 47, 373–386. [Google Scholar] [CrossRef] [PubMed]
- Meilhac, S.M.; Esner, M.; Kelly, R.G.; Nicolas, J.F.; Buckingham, M.E. The clonal origin of myocardial cells in different regions of the embryonic mouse heart. Dev. Cell 2004, 6, 685–698. [Google Scholar] [CrossRef]
- Hill, M.R.; Simon, M.A.; Valdez-Jasso, D.; Zhang, W.; Champion, H.C.; Sacks, M.S. Structural and mechanical adaptations of right ventricle free wall myocardium to pressure overload. Ann. Biomed. Eng. 2014, 42, 2451–2465. [Google Scholar] [CrossRef]
- Jang, S.; Vanderpool, R.R.; Avazmohammadi, R.; Lapshin, E.; Bachman, T.N.; Sacks, M.; Simon, M.A. Biomechanical and Hemodynamic Measures of Right Ventricular Diastolic Function: Translating Tissue Biomechanics to Clinical Relevance. J. Am. Heart Assoc. 2017, 6, e006084. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.H.; Yin, F.C. A multiaxial constitutive law for mammalian left ventricular myocardium in steady-state barium contracture or tetanus. J. Biomech. Eng. 1998, 120, 504–517. [Google Scholar] [CrossRef] [PubMed]
- Kirton, R.S.; Taberner, A.J.; Nielsen, P.M.; Young, A.A.; Loiselle, D.S. Effects of BDM, [Ca2+]o, and temperature on the dynamic stiffness of quiescent cardiac trabeculae from rat. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, H1662–H1667. [Google Scholar] [CrossRef]
- Park, J.H.; Choi, J.O.; Park, S.W.; Cho, G.Y.; Oh, J.K.; Lee, J.H.; Seong, I.W. Normal references of right ventricular strain values by two-dimensional strain echocardiography according to the age and gender. Int. J. Cardiovasc. Imaging 2018, 34, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Park, J.-H. Strain Analysis of the Right Ventricle Using Two-dimensional Echocardiography. J. Cardiovasc. Imaging 2018, 26, 111–124. [Google Scholar] [CrossRef]
- Liu, W.; Nguyen-Truong, M.; Ahern, M.; Labus, K.M.; Puttlitz, C.M.; Wang, Z. Different Passive Viscoelastic Properties Between the Left and Right Ventricles in Healthy Adult Ovine. J. Biomech. Eng. 2021, 143, 121002. [Google Scholar] [CrossRef]
- Javani, S.; Gordon, M.; Azadani, A.N. Biomechanical properties and microstructure of heart chambers: A paired comparison study in an ovine model. Ann. Biomed. Eng. 2016, 44, 3266–3283. [Google Scholar] [CrossRef]
- Matsumoto, T.; Fukui, T.; Tanaka, T.; Ikuta, N.; Ohashi, T.; Kumagai, K.; Akimoto, H.; Tabayashi, K.; Sato, M. Biaxial Tensile Properties of Thoracic Aortic Aneurysm Tissues. J. Biomech. Sci. Eng. 2009, 4, 518–529. [Google Scholar] [CrossRef]
- Fratzl, P. Collagen: Structure and Mechanics; Springer: New York, NY, USA, 2008; 506p. [Google Scholar]
- Lorenz, C.H.; Walker, E.S.; Graham, T.P.; Powers, T.A. Right Ventricular Performance and Mass by Use of Cine MRI Late After Atrial Repair of Transposition of the Great Arteries. Circulation 1995, 92, 233–239. [Google Scholar] [CrossRef]
- Nguyen-Truong, M.; Liu, W.; Boon, J.; Nelson, B.; Easley, J.; Monnet, E.; Wang, Z. Establishment of adult right ventricle failure in ovine using a graded, animal-specific pulmonary artery constriction model. Anim. Models Exp. Med. 2020, 3, 182–192. [Google Scholar] [CrossRef]
- Ostenfeld, E.; Stephensen, S.S.; Steding-Ehrenborg, K.; Heiberg, E.; Arheden, H.; Rådegran, G.; Holm, J.; Carlsson, M. Regional contribution to ventricular stroke volume is affected on the left side, but not on the right in patients with pulmonary hypertension. Int. J. Cardiovasc. Imaging 2016, 32, 1243–1253. [Google Scholar] [CrossRef]
- Stephensen, S.; Steding-Ehrenborg, K.; Munkhammar, P.; Heiberg, E.; Arheden, H.; Carlsson, M. The relationship between longitudinal, lateral, and septal contribution to stroke volume in patients with pulmonary regurgitation and healthy volunteers. Am. J. Physiol.-Heart Circ. Physiol. 2014, 306, H895–H903. [Google Scholar] [CrossRef]
- Holzapfel, G.A.; Ogden, R.W. Constitutive modelling of passive myocardium: A structurally based framework for material characterization. Philos Trans. A Math. Phys. Eng. Sci. 2009, 367, 3445–3475. [Google Scholar] [CrossRef]
- Streeter Daniel, D.; Spotnitz Henry, M.; Patel Dali, P.; Ross, J.; Sonnenblick Edmund, H. Fiber Orientation in the Canine Left Ventricle during Diastole and Systole. Circ. Res. 1969, 24, 339–347. [Google Scholar] [CrossRef]
- Reese, T.G.; Weisskoff, R.M.; Smith, R.N.; Rosen, B.R.; Dinsmore, R.E.; Wedeen, V.J. Imaging myocardial fiber architecture in vivo with magnetic resonance. Magn. Reson. Med. 1995, 34, 786–791. [Google Scholar] [CrossRef]
- Streeter, D.D., Jr.; Bassett, D.L. An engineering analysis of myocardial fiber orientation in pig’s left ventricle in systole. Anat. Rec. 1966, 155, 503–511. [Google Scholar] [CrossRef]
- Shi, X.; Liu, Y.; Copeland, K.M.; McMahan, S.R.; Zhang, S.; Butler, J.R.; Hong, Y.; Cho, M.; Bajona, P.; Gao, H.; et al. Epicardial prestrained confinement and residual stresses: A newly observed heart ventricle confinement interface. J. R. Soc. Interface 2019, 16, 20190028. [Google Scholar] [CrossRef] [PubMed]
- Avazmohammadi, R.; Hill, M.R.; Simon, M.A.; Zhang, W.; Sacks, M.S. A novel constitutive model for passive right ventricular myocardium: Evidence for myofiber-collagen fiber mechanical coupling. Biomech. Modeling Mechanobiol. 2017, 16, 561–581. [Google Scholar] [CrossRef] [PubMed]


| Group | εL | εC | εLC | εCL |
|---|---|---|---|---|
| LV-side (n = 12) | 0.110.01 | 0.180.02 | 0.030.02 | 0.050.02 |
| RV-side (n = 12) | 0.150.02 | 0.210.03 | 0.010.02 | 0.010.02 |
| LV-Side (n = 4) | Midwall (n = 9) | RV-Side (n = 4) | |
|---|---|---|---|
| Myofiber angle (°) | 78 ± 3 | 9 ± 2 *,# | 108 ± 8 |
| Septum | bL | bC | bLC | B (kPa) | RMS (kPa) |
|---|---|---|---|---|---|
| LV-side | 12.62 | 7.74 | 0.002 | 0.04 | 0.04 |
| RV-side | 7.21 | 7.63 | 0.001 | 0.04 | 0.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen-Truong, M.; Liu, W.; Doherty, C.; LeBar, K.; Labus, K.M.; Puttlitz, C.M.; Easley, J.; Monnet, E.; Chicco, A.; Wang, Z. The Interventricular Septum Is Biomechanically Distinct from the Ventricular Free Walls. Bioengineering 2021, 8, 216. https://doi.org/10.3390/bioengineering8120216
Nguyen-Truong M, Liu W, Doherty C, LeBar K, Labus KM, Puttlitz CM, Easley J, Monnet E, Chicco A, Wang Z. The Interventricular Septum Is Biomechanically Distinct from the Ventricular Free Walls. Bioengineering. 2021; 8(12):216. https://doi.org/10.3390/bioengineering8120216
Chicago/Turabian StyleNguyen-Truong, Michael, Wenqiang Liu, Courtney Doherty, Kristen LeBar, Kevin M. Labus, Christian M. Puttlitz, Jeremiah Easley, Eric Monnet, Adam Chicco, and Zhijie Wang. 2021. "The Interventricular Septum Is Biomechanically Distinct from the Ventricular Free Walls" Bioengineering 8, no. 12: 216. https://doi.org/10.3390/bioengineering8120216
APA StyleNguyen-Truong, M., Liu, W., Doherty, C., LeBar, K., Labus, K. M., Puttlitz, C. M., Easley, J., Monnet, E., Chicco, A., & Wang, Z. (2021). The Interventricular Septum Is Biomechanically Distinct from the Ventricular Free Walls. Bioengineering, 8(12), 216. https://doi.org/10.3390/bioengineering8120216