Coumarin-Encapsulated Solid Lipid Nanoparticles as an Effective Therapy against Methicillin-Resistant Staphylococcus aureus
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Tween® 20 Stabilized Coumarin-Loaded SLNs
2.3. Characterization of Prepared Nanoparticles
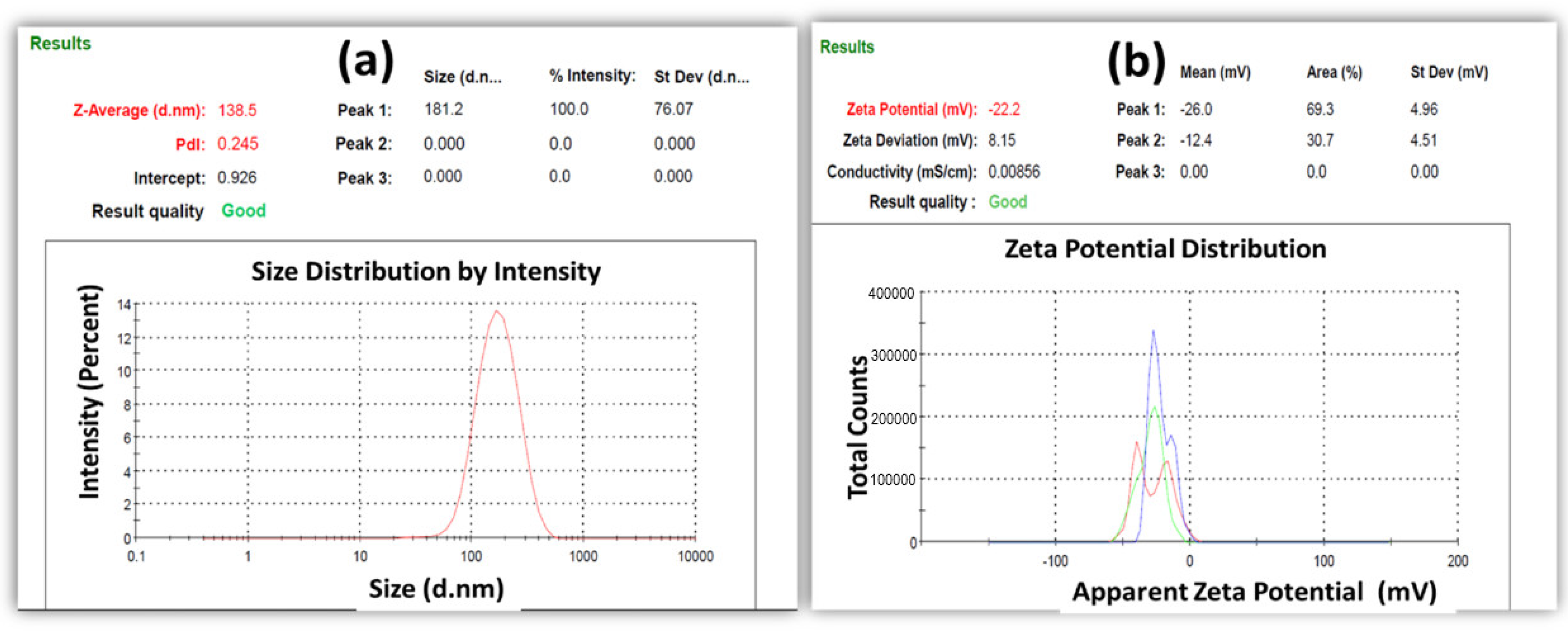
2.3.1. Particle Size, Polydispersity Index, and Zeta Potential Determination
2.3.2. Estimation of Encapsulation Efficiency (% EE)
2.3.3. Morphology
2.3.4. Cell Toxicity Assay
2.3.5. In Vitro Drug Release
2.3.6. In Vitro Antibacterial Activity
2.3.7. Bactericidal Time-Kill Kinetics
3. Results
3.1. Particle Size, Polydispersity Index (PDI), and Zeta Potential Determination
3.2. Estimation of Encapsulation Efficiency (% EE) and Morphology
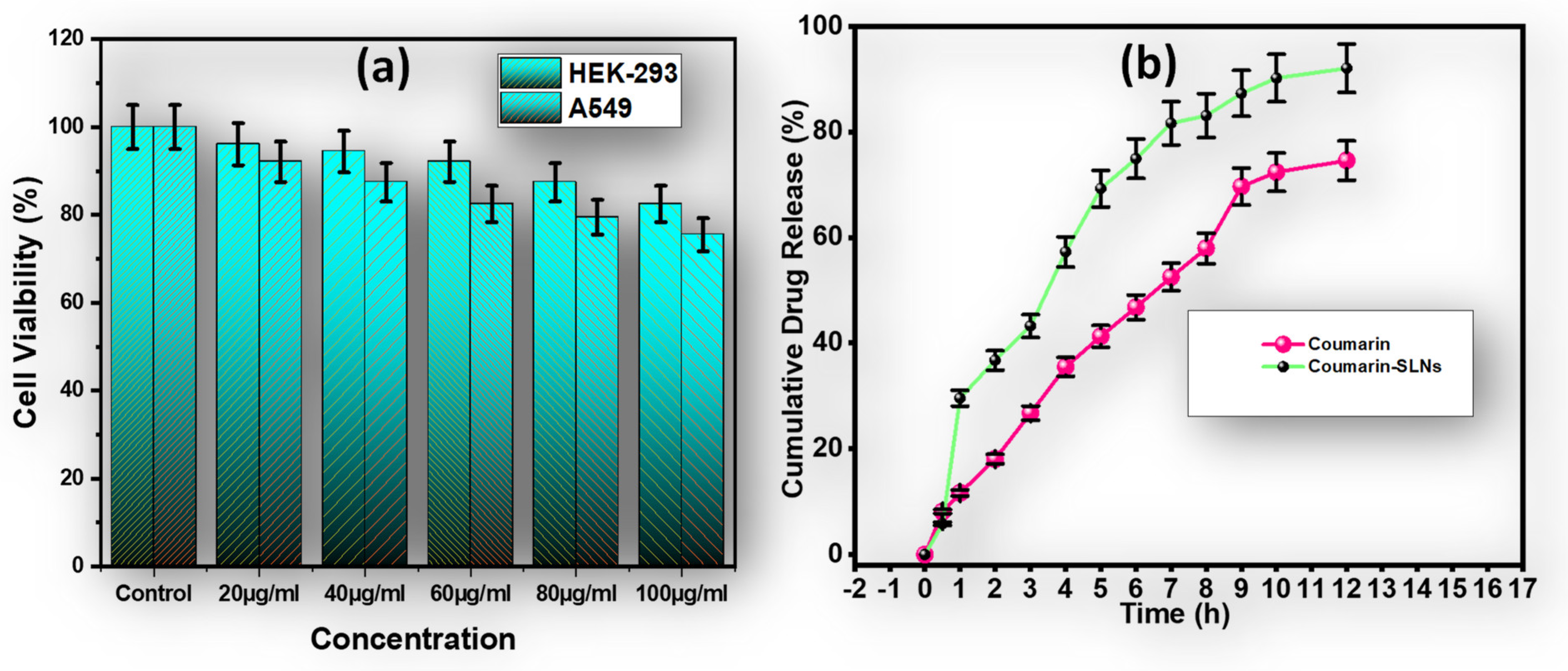
3.3. Cell Toxicity Assay
3.4. In Vitro Drug Release
3.5. In Vitro Antibacterial Activity
3.5.1. Minimum Inhibitory Concentration
3.5.2. Bactericidal Time-Kill Kinetics
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Singh, S.; Numan, A.; Cinti, S. Point-of-Care for Evaluating Antimicrobial Resistance through the Adoption of Functional Materials. Anal. Chem. 2022, 94, 26–40. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Alrobaian, M.M.; Molugulu, N.; Agrawal, N.; Numan, A.; Kesharwani, P. Pyramid-Shaped PEG-PCL-PEG Polymeric-Based Model Systems for Site-Specific Drug Delivery of Vancomycin with Enhance Antibacterial Efficacy. ACS Omega. 2020, 5, 11935–11945. [Google Scholar] [CrossRef] [PubMed]
- Høiby, N.; Ciofu, O.; Johansen, H.K.; Song, Z.J.; Moser, C.; Jensen, P.Ø.; Molin, S.; Givskov, M.; Tolker-Nielsen, T.; Bjarnsholt, T. The clinical impact of bacterial biofilms. Proc. Int. J. Oral Sci. 2011, 3, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Sahli, C.; Moya, S.E.; Lomas, J.S.; Gravier-Pelletier, C.; Briandet, R.; Hémadi, M. Recent advances in nanotechnology for eradicating bacterial biofilm. Theranostics 2022, 12, 2383–2405. [Google Scholar] [CrossRef]
- Kaur, H.; Kaur, A.; Soni, S.K.; Rishi, P. Microbially-derived cocktail of carbohydrases as an anti-biofouling agents: A ‘green approach. Biofouling 2022, 38, 455–481. [Google Scholar] [CrossRef]
- Singh, P.; Pandit, S.; Beshay, M.; Mokkapati, V.R.S.S.; Garnaes, J.; Olsson, M.E.; Sultan, A.; Mackevica, A.; Mateiu, R.V.; Lütken, H.; et al. Anti-biofilm effects of gold and silver nanoparticles synthesized by the Rhodiola rosea rhizome extracts. Artif Cells Nanomedicine Biotechnol. 2018, 46, S886–S899. [Google Scholar] [CrossRef]
- Ansari, M.A.; Khan, H.M.; Khan, A.A.; Cameotra, S.S.; Alzohairy, M.A. Anti-biofi lm effi cacy of silver nanoparticles against MRSA and MRSE isolated from wounds in a tertiary care hospital. Indian J. Med. Microbiol. 2015, 33, 101–109. [Google Scholar] [CrossRef]
- Xu, Z. 1,2,3-Triazole-containing hybrids with potential antibacterial activity against methicillin-resistant Staphylococcus aureus (MRSA). Eur. J. Med. Chem. 2020, 354, e2000223. [Google Scholar] [CrossRef]
- Gao, Y.; Chen, Y.; Cao, Y.; Mo, A.; Peng, Q. Potentials of nanotechnology in treatment of methicillin-resistant Staphylococcus aureus. Eur. J. Med. Chem. 2021, 213, 113056. [Google Scholar] [CrossRef]
- Singh, S.; Numan, A.; Somaily, H.H.; Gorain, B.; Ranjan, S.; Rilla, K.; Siddique, H.R.; Kesharwani, P. Nano-enabled strategies to combat methicillin-resistant Staphylococcus aureus. Mater. Sci. Eng. C. 2021, 129, 112384. [Google Scholar] [CrossRef]
- Singh, S.; Aldawsari, H.M.; Alam, A.; Alqarni, M.H.S.; Ranjan, S.; Kesharwani, P. Synthesis and antimicrobial activity of vancomycin–conjugated zinc coordination polymer nanoparticles against methicillin-resistant staphylococcus aureus. J. Drug Deliv. Sci Technol. 2022, 70, 103255. [Google Scholar] [CrossRef]
- Stokes, J.M.; Macnair, C.R.; Ilyas, B.; French, S.; Côté, J.P.; Bouwman, C.; Farha, M.A.; Sieron, A.O.; Whitfield, C.; Coombes, B.K.; et al. Pentamidine sensitizes Gram-negative pathogens to antibiotics and overcomes acquired colistin resistance. Nat. Microbiol. 2017, 2, 17028. [Google Scholar] [CrossRef] [PubMed]
- Yerer, M.B.; Dayan, S.; Han, M.I.; Sharma, A.; Tuli, H.S.; Sak, K. Nanoformulations of Coumarins and the Hybrid Molecules of Coumarins with Potential Anticancer Effects. Anticancer Agents Med. Chem. 2020, 20, 1797–1816. [Google Scholar] [CrossRef] [PubMed]
- Smyth, T.; Ramachandran, V.N.; Smyth, W.F. A study of the antimicrobial activity of selected naturally occurring and synthetic coumarins. Int. J. Antimicrob. Agents 2009, 33, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.F.; Zhang, P.L.; Sui, Y.F.; Lv, J.S.; Ansari, M.F.; Battini, N.; Li, S.; Zhou, C.H.; Geng, R.X. Ethylenic conjugated coumarin thiazolidinediones as new efficient antimicrobial modulators against clinical methicillin-resistant Staphylococcus aureus. Bioorg. Chem. 2020, 94, 103434. [Google Scholar] [CrossRef] [PubMed]
- Alnufaie, R.; Kc, H.R.; Alsup, N.; Whitt, J.; Chambers, S.A.; Gilmore, D.; Alam, M.A. Synthesis and Antimicrobial Studies of Coumarin-Substituted Pyrazole Derivatives as Potent Anti-Staphylococcus aureus Agents. Molecules 2020, 25, 2758. [Google Scholar] [CrossRef]
- Qu, D.; Hou, Z.; Li, J.; Luo, L.; Su, S.; Ye, Z.; Bai, Y.; Zhang, X.; Chen, G.; Li, Z.; et al. A new coumarin compound DCH combats methicillin-resistant Staphylococcus aureus biofilm by targeting arginine repressor. Sci. Adv. 2020, 6, eaay9597. [Google Scholar] [CrossRef]
- De Araújo, R.S.A.; Barbosa-Filho, J.M.; Scotti, M.T.; Scotti, L.; Da Cruz, R.M.D.; Falcão-Silva, V.D.S.; De Siqueira-Júnior, J.P.; Mendonça-Junior, F.J.B. Modulation of Drug Resistance in Staphylococcus aureus with Coumarin Derivatives. Scientifica 2016, 2016, 6894758. [Google Scholar] [CrossRef]
- Sastri, K.T.; Radha, G.V.; Pidikiti, S.; Vajjhala, P. Solid lipid nanoparticles: Preparation techniques, their characterization, and an update on recent studies. J. Appl. Pharm. Sci. 2020, 10, 126–141. [Google Scholar] [CrossRef]
- Dawoud, M. Chitosan coated solid lipid nanoparticles as promising carriers for docetaxel. J. Drug Deliv. Sci. Technol. 2021, 62, 102409. [Google Scholar] [CrossRef]
- Ban, C.; Jo, M.; Park, Y.H.; Kim, J.H.; Han, J.Y.; Lee, K.W.; Kweon, D.H.; Choi, Y.J. Enhancing the oral bioavailability of curcumin using solid lipid nanoparticles. Food Chem. 2020, 302, 125328. [Google Scholar] [CrossRef] [PubMed]
- Grillone, A.; Battaglini, M.; Moscato, S.; Mattii, L.; De Julián Fernández, C.; Scarpellini, A.; Giorgi, M.; Sinibaldi, E.; Ciofani, G. Nutlin-loaded magnetic solid lipid nanoparticles for targeted glioblastoma treatment. Nanomedicine 2019, 14, 727–752. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.R.; San Martin-Gonzalez, M.F. Characterization of ergocalciferol loaded solid lipid nanoparticles. J. Food Sci. 2012, 77, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Affram, K.O.; Smith, T.; Ofori, E.; Krishnan, S.; Underwood, P.; Trevino, J.G.; Agyare, E. Cytotoxic effects of gemcitabine-loaded solid lipid nanoparticles in pancreatic cancer cells. J. Drug Deliv. Sci. Technol. 2020, 55, 101374. [Google Scholar] [CrossRef]
- Shah, R.M.; Eldridge, D.S.; Palombo, E.A.; Harding, I.H. Stability mechanisms for microwave-produced solid lipid nanoparticles. Colloids Surfaces A Physicochem. Eng. Asp. 2022, 643, 128774. [Google Scholar] [CrossRef]
- Shah, R.M.; Eldridge, D.S.; Palombo, E.A.; Harding, I.H. Microwave-assisted formulation of solid lipid nanoparticles loaded with non-steroidal anti-inflammatory drugs. Int. J. Pharm. 2016, 515, 543–554. [Google Scholar] [CrossRef]
- Kalhapure, R.S.; Sonawane, S.J.; Sikwal, D.R.; Jadhav, M.; Rambharose, S.; Mocktar, C.; Govender, T. Solid lipid nanoparticles of clotrimazole silver complex: An efficient nano antibacterial against Staphylococcus aureus and MRSA. Colloids Surf. B Biointerfaces 2015, 136, 651–658. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. CLSI M26-A Methods for Determining Bactericidal Activity of Antimicrobial Agents; Approved Guideline; CLSI document M26-A; Clinical and Laboratory Standards Institute: Malvern, PA, USA, 1999. [Google Scholar]
- Chung, S.H.; Cho, S.; Kim, K.; Lim, B.S.; Ahn, S.J. Antimicrobial and physical characteristics of orthodontic primers containing antimicrobial agents. Angle Orthod. 2017, 87, 307–312. [Google Scholar] [CrossRef]


| Compound | MIC (µg/mL) | |
|---|---|---|
| 24 h | 48 h | |
| Vancomycin HCl | 2.04 | NA |
| SLN suspension | NA | NA |
| Coumarin | 3.92 | 4.93 |
| Coumarin-loaded SLNs | 1.08 | 2.13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alqarni, M.H.; Foudah, A.I.; Alam, A.; Salkini, M.A.; Muharram, M.M.; Labrou, N.E.; Rawat, P. Coumarin-Encapsulated Solid Lipid Nanoparticles as an Effective Therapy against Methicillin-Resistant Staphylococcus aureus. Bioengineering 2022, 9, 484. https://doi.org/10.3390/bioengineering9100484
Alqarni MH, Foudah AI, Alam A, Salkini MA, Muharram MM, Labrou NE, Rawat P. Coumarin-Encapsulated Solid Lipid Nanoparticles as an Effective Therapy against Methicillin-Resistant Staphylococcus aureus. Bioengineering. 2022; 9(10):484. https://doi.org/10.3390/bioengineering9100484
Chicago/Turabian StyleAlqarni, Mohammed H., Ahmed I. Foudah, Aftab Alam, Mohammad A. Salkini, Magdy M. Muharram, Nikolaos E. Labrou, and Pinki Rawat. 2022. "Coumarin-Encapsulated Solid Lipid Nanoparticles as an Effective Therapy against Methicillin-Resistant Staphylococcus aureus" Bioengineering 9, no. 10: 484. https://doi.org/10.3390/bioengineering9100484
APA StyleAlqarni, M. H., Foudah, A. I., Alam, A., Salkini, M. A., Muharram, M. M., Labrou, N. E., & Rawat, P. (2022). Coumarin-Encapsulated Solid Lipid Nanoparticles as an Effective Therapy against Methicillin-Resistant Staphylococcus aureus. Bioengineering, 9(10), 484. https://doi.org/10.3390/bioengineering9100484








