Zr-Based Metal-Organic Frameworks for Green Biodiesel Synthesis: A Minireview
Abstract
:1. Introduction
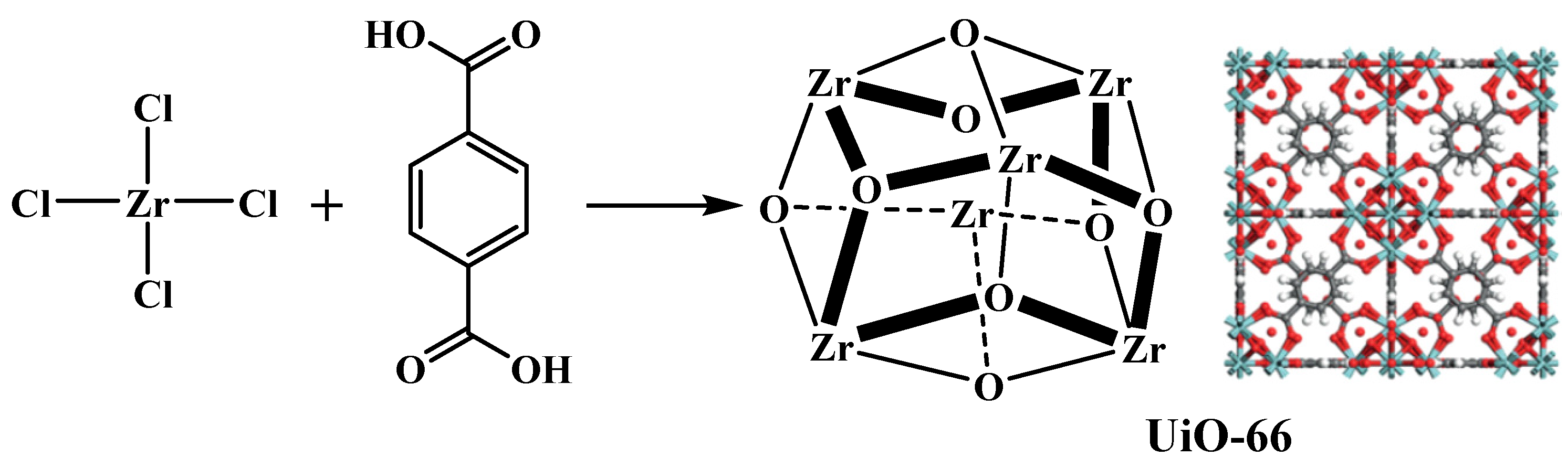
2. Zr-Based MOF Catalysts
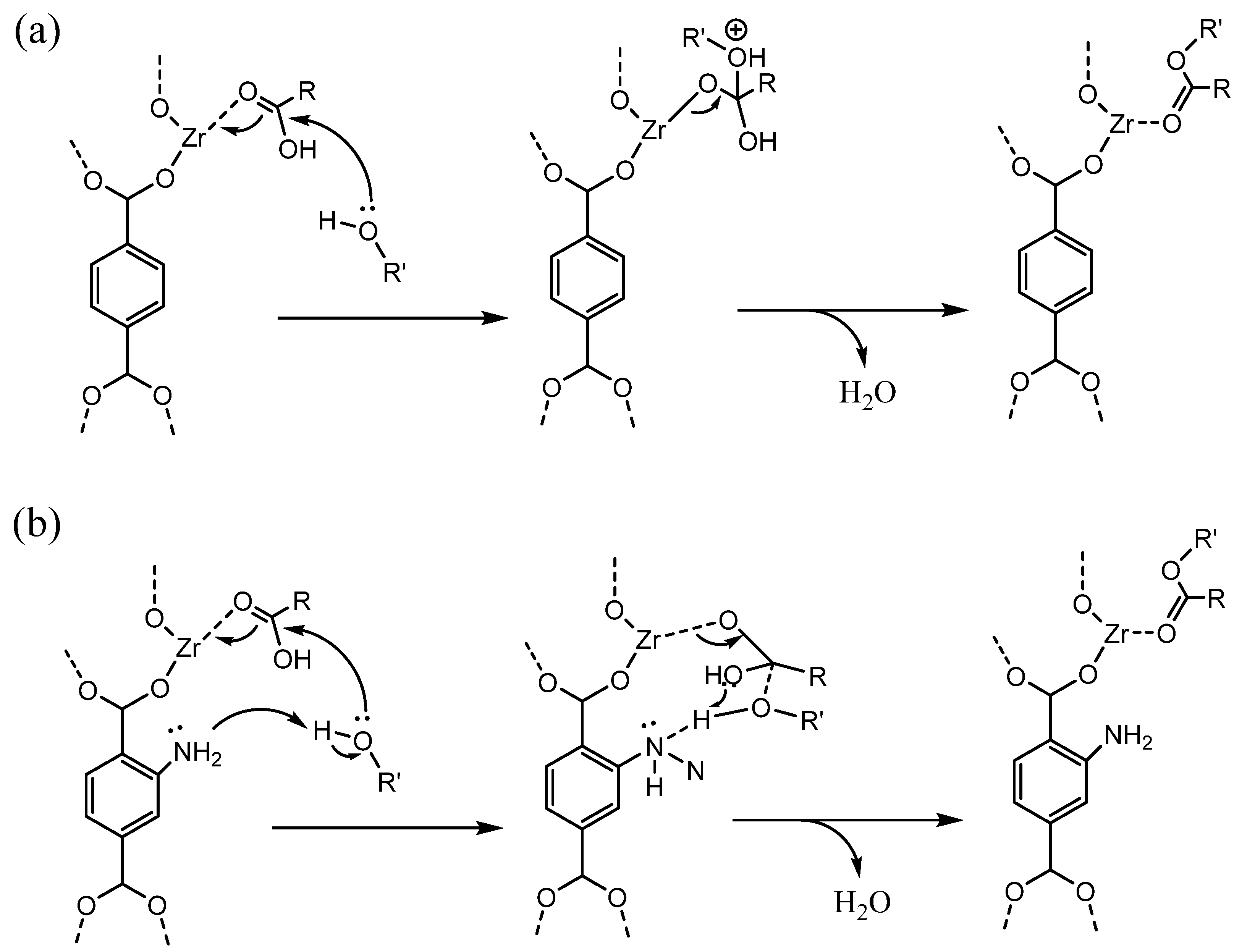
2.1. Single Zr-Based MOF Catalysts
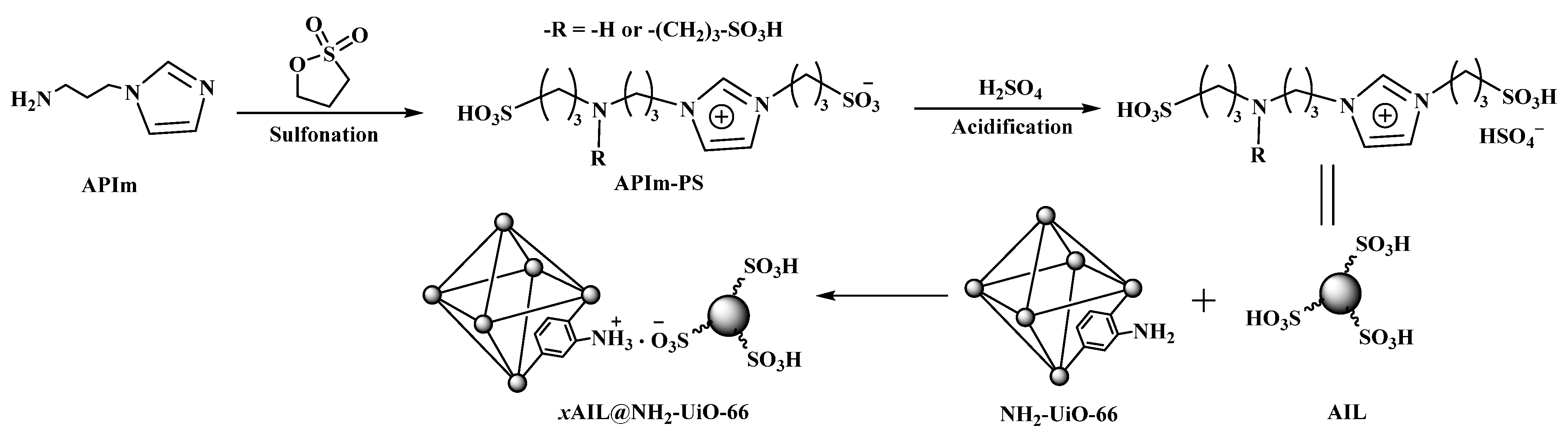
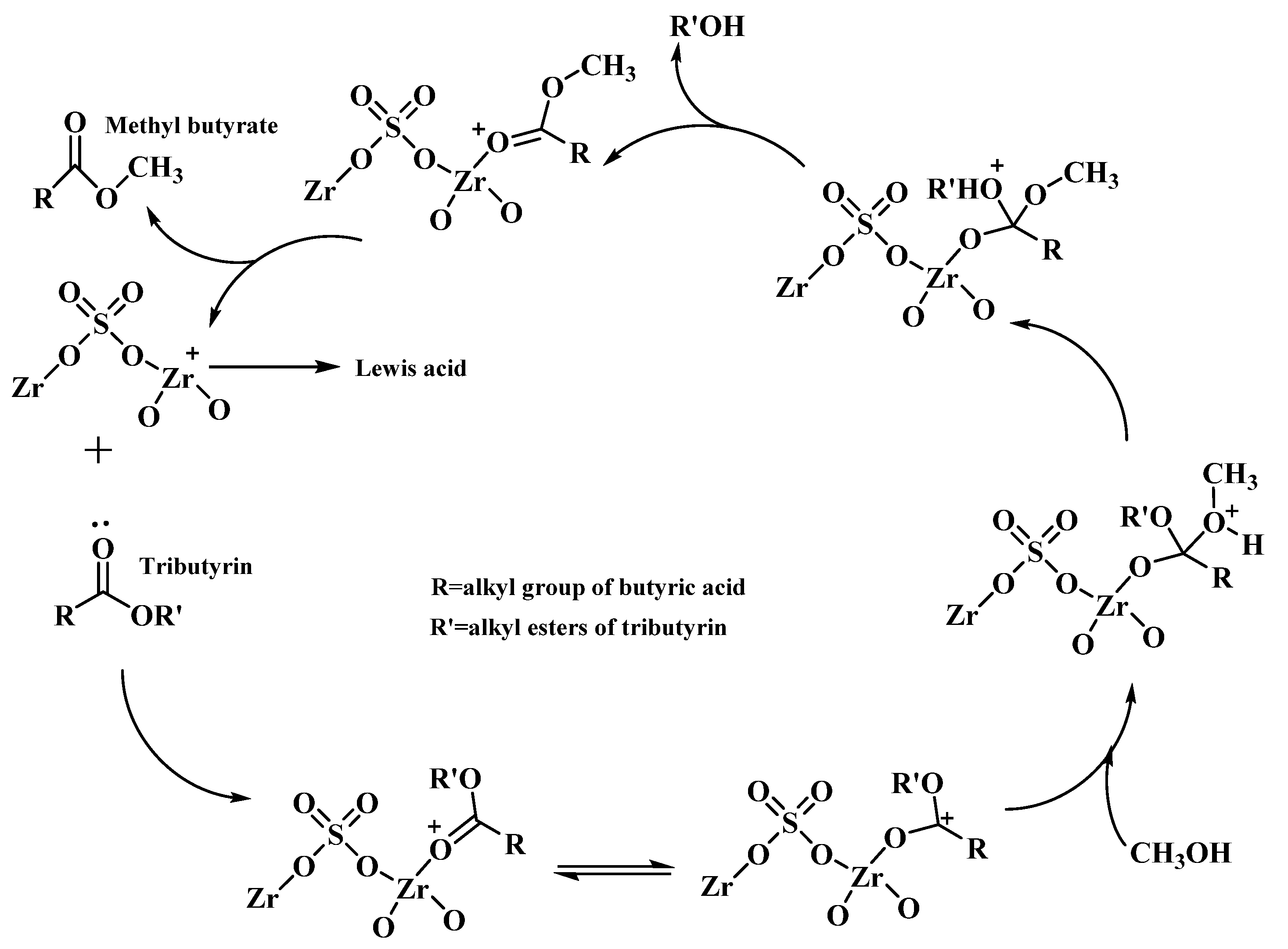
2.2. Modified Zr-Based MOF Catalysts
| Entry | Raw Material | Catalyst | Reaction Conditions (Time, Temperature, Catalyst Amount, Molar Ratio (Acid(Oil):Alcohol)) | Yield (Y/%) or Conversion (C/%) | Reusability | Ea (KJ/mol) | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | Lauric acid + Methanol | UiO-66-NH2 | 2 h, 60 °C, 8%, 1:26 | Y > 99 | Not reported | \ | [34] |
| 2 | Levulinic acid + n-butanol | UiO-66-NH2 | 5 h, 120 °C, 1.8%, 1:6 | Y = 99% | 3 cycles, no significant loss | \ | [35] |
| 3 | Levulinic acid + Ethanol | UiO-66-(COOH)2 | 24 h, 78 °C, 0.39%, 1:20 | Y = 97% | 5 cycles, Y = 93.9% | \ | [36] |
| 4 | Oleic acid + Methanol | UiO-66(Zr)-NH2 | 4 h, 60 °C, 6%, 1:39 | C = 97% | 4 cycles, C > 50% | 15.13 | [37] |
| 5 | Oleic acid + Methanol | 10SA/UiO-66(Zr) | 4 h, 25 °C, 6%, 1:39 | C = 94.5% | 6 cycles, C = 83% | 32.53 | [38] |
| 6 | Levulinic acid + Ethanol | UiO66-SO3H(100) | 6 h, 80 °C, 0.4%, 1:10 | Y = 87% | 4 cycles, Y = 84% | \ | [39] |
| 7 | Ricinus communis oil + Methanol | Lipase/Zr-MOF/PVP | 12 h, 50 °C, 2 mg, 1:3 | C = 83% | 7 cycles, C = 66% | \ | [40] |
| 8 | Oleic acid + Methanol | UiO-G | 2 h, 70 °C, 8%, 1:12 | C = 91.3% | 4 cycles, C = 66.6% | 28.61 | [41] |
| 9 | Acetic acid + Isooctyl alcohol | UiO-67-CF3SO3 | 18 h, 90 °C, 0.2 g, 6:1 | C = 98.6% | 5 cycles, C = 95.9% | \ | [42] |
| 10 | Tripalmitin + Methanol | UiO-66-[C3NH2] [SO3CF3] | 12 h, 85 °C, 0.025 g, 1:121.5 | Y = 86.6–98.4% | Not reported | 38.9 | [43] |
| 11 | Jatropha oil + Methanol | PSH/UiO-66-NO2 | 4 h, 70 °C, 4%, 1:25 | C= 97.57% | 3 cycles, C= 77.14% | \ | [44] |
| 12 | Oleic acid + Methanol | AIL@NH2-UiO-66 | 6 h, 75 °C, 5%, 1:14 | C = 95.22% | 6 cycles, C = 90.42% | \ | [45] |
| 13 | Oleic acid + Methanol | Ca2+/UiO-66(Zr) | 4 h, 60 °C, 6%, 1:39 | Y = 98% | 5 cycles, Y = 84% | 36.73 | [46] |
| 14 | Oleic acid + Methanol | K-PW12@UIO-66(Zr) | 4 h, 75 °C, 5%, 1:20 | C = 90% | 10 cycles, no significant loss | \ | [47] |
| 15 | Acetic acid + n-butanol | HPW@UiO-66 | 3 h, 120 °C, 3%, 1:2 | C = 80.2% | 4 cycles, C = 63% | \ | [48] |
| 16 | Soybean oil + C8 + C10 | Cs2.5H0.5PW12O40@UiO-66 | 10 h, 150 °C, 7%,1:5:5 | FA incorporation =20.3% | 5 cycles, no significant loss | \ | [49] |
| 17 | Soybean oil + Methanol | AILs/HPW/UiO-66-2COOH | 6 h, 110 °C, 10%, 1:35 | C = 95.8% | 5 cycles, C > 80% | \ | [50] |
| 18 | Euphorbia Lathyris L. oil + Methanol | HPW/UiO-66-NH2 | 8 h, 180 °C, 3.5%, 1:40 | Y = 91.2% | 4 cycles, no significant loss | 31.0 | [51] |
| 19 | Oleic acid + Methanol | FDCA/SA-UiO-66(Zr) | 24 h, 60 °C, 6%, 1:40 | Y = 98.4% | 6 cycles, Y > 90% | \ | [52] |
| 20 | Soybean oil + Methanol | PW12@UIO-66 | 4 h, 75 °C, 0.2 g, 1 g:5.5 ml | C = 91.1% | 4 cycles, no significant loss | \ | [53] |
| 21 | Lauric acid + Methanol | HSiW-UiO-66 | 4 h, 160 °C, 7%, 1:20 | C = 80.5% | 4 cycles, C = 70.2% | 27.5 | [54] |
| 22 | Oleic acid + Methanol | ZrSiW/UiO-66 | 4 h, 150 °C, 8%, 1:20 | C = 98.0% | 4 cycles, C = 88.9% | \ | [55] |
| 23 | Lauric acid + Methanol | Ag1(NH4)2PW12O40/UiO-66 | 3 h, 150 °C, 10%, 1:15 | C = 75.6% | 4 cycles, C = 70.6% | 35.2 | [56] |
| 24 | Oleic acid + Methanol | Ce-BDC@HSiW@UiO-66 | 4 h, 130 °C, 0.2 g, 1:30 | C = 81.5% | 6 cycles, C = 76.9% | \ | [57] |
2.3. Zr-Based MOF-Derived Catalysts
3. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hoang, A.T.; Tabatabaei, M.; Aghbashlo, M.; Carlucci, A.P.; Ölçer, A.I.; Le, A.T.; Ghassemi, A. Rice bran oil-based biodiesel as a promising renewable fuel alternative to petrodiesel: A review. Renew. Sustain. Energy Rev. 2020, 135, 110204. [Google Scholar] [CrossRef]
- Bekhradinassab, E.; Tavakoli, A.; Haghighi, M.; Shabani, M. Catalytic biofuel production over 3D macro-structured cheese-like Mn-promoted TiO2 isotype: Mn-catalyzed microwave-combustion design. Energy Convers. Manag. 2021, 251, 114916. [Google Scholar] [CrossRef]
- Rezania, S.; Mahdinia, S.; Oryani, B.; Cho, J.; E Kwon, E.; Bozorgian, A.; Nodeh, H.R.; Darajeh, N.; Mehranzamir, K. Biodiesel production from wild mustard (Sinapis Arvensis) seed oil using a novel heterogeneous catalyst of LaTiO3 nanoparticles. Fuel 2022, 307, 121759. [Google Scholar] [CrossRef]
- Zhang, Q.; Ling, D.; Lei, D.; Wang, J.; Liu, X.; Zhang, Y.; Ma, P. Green and Facile Synthesis of Metal-Organic Framework Cu-BTC-Supported Sn (II)-Substituted Keggin Heteropoly Composites as an Esterification Nanocatalyst for Biodiesel Production. Front. Chem. 2020, 8, 129. [Google Scholar] [CrossRef] [PubMed]
- Cholapandian, K.; Gurunathan, B.; Rajendran, N. Investigation of CaO nanocatalyst synthesized from Acalypha indica leaves and its application in biodiesel production using waste cooking oil. Fuel 2022, 312, 122958. [Google Scholar] [CrossRef]
- Rozina; Ahmad, M.; Elnaggar, A.Y.; Teong, L.K.; Sultana, S.; Zafar, M.; Munir, M.; Hussein, E.E.; Abidin, S.Z.U. Sustainable and eco-friendly synthesis of biodiesel from novel and non-edible seed oil of Monotheca buxifolia using green nano-catalyst of calcium oxide. Energy Convers. Manag. X 2022, 13, 100142. [Google Scholar] [CrossRef]
- Pan, H.; Xia, Q.; Li, H.; Wang, Y.; Shen, Z.; Wang, Y.; Li, L.; Li, X.; Xu, H.; Zhou, Z.; et al. Direct production of biodiesel from crude Euphorbia lathyris L. Oil catalyzed by multifunctional mesoporous composite materials. Fuel 2021, 309, 122172. [Google Scholar] [CrossRef]
- Pan, H.; Xia, Q.; Wang, Y.; Shen, Z.; Huang, H.; Ge, Z.; Li, X.; He, J.; Wang, X.; Li, L.; et al. Recent advances in biodiesel production using functional carbon materials as acid/base catalysts. Fuel Process. Technol. 2022, 237, 107421. [Google Scholar] [CrossRef]
- Zhang, Q.; Luo, Q.; Wu, Y.; Yu, R.; Cheng, J.; Zhang, Y. Construction of a Keggin heteropolyacid/Ni-MOF catalyst for esterification of fatty acids. RSC Adv. 2021, 11, 33416–33424. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Zhang, D.; Sun, Y.; Zhou, Z.; Du, Y.; Du, Y.; Li, Y.; Liu, M.; Zhang, Y.; Shen, J.; et al. Structural sensitivity of heterogeneous catalysts for sustainable chemical synthesis of gluconic acid from glucose. Chin. J. Catal. 2020, 41, 1320–1336. [Google Scholar] [CrossRef]
- Chen, B.; Yan, G.; Chen, G.; Feng, Y.; Zeng, X.; Sun, Y.; Tang, X.; Lei, T.; Lin, L. Recent progress in the development of advanced biofuel 5-ethoxymethylfurfural. BMC Energy 2020, 2, 2. [Google Scholar] [CrossRef]
- Ji, J.; Bao, Y.; Liu, X.; Zhang, J.; Xing, M. Molybdenum-based heterogeneous catalysts for the control of environmental pollutants. EcoMat 2021, 3, e12155. [Google Scholar] [CrossRef]
- Sahar, J.; Farooq, M.; Ramli, A.; Naeem, A.; Khattak, N.S. Biodiesel production from Mazari palm (Nannorrhops ritchiana) seeds oil using Tungstophosphoric acid decorated SnO2@Mn-ZIF bifunctional heterogeneous catalyst. Appl. Catal. A Gen. 2022, 643, 118740. [Google Scholar] [CrossRef]
- Fang, R.; Dhakshinamoorthy, A.; Li, Y.; Garcia, H. Metal organic frameworks for biomass conversion. Chem. Soc. Rev. 2020, 49, 3638–3687. [Google Scholar] [CrossRef]
- Yadav, S.; Dixit, R.; Sharma, S.; Dutta, S.; Solanki, K.; Sharma, R.K. Magnetic metal–organic framework composites: Structurally advanced catalytic materials for organic transformations. Mater. Adv. 2021, 2, 2153–2187. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, Y.; Cheng, J.; Li, H.; Ma, P. An Overview of Metal-organic Frameworks-based Acid/Base Catalysts for Biofuel Synthesis. Curr. Org. Chem. 2020, 24, 1876–1891. [Google Scholar] [CrossRef]
- Wei, Y.; Zhang, Y.; Li, B.; Guan, W.; Yan, C.; Li, X.; Yan, Y. Facile synthesis of metal-organic frameworks embedded in interconnected macroporous polymer as a dual acid-base bifunctional catalyst for efficient conversion of cellulose to 5-hydroxymethylfurfural. Chin. J. Chem. Eng. 2022, 44, 169–181. [Google Scholar] [CrossRef]
- Li, Y.; Meng, X.; Luo, R.; Zhou, H.; Lu, S.; Yu, S.; Bai, P.; Guo, X.; Lyu, J. Aluminum/Tin-doped UiO-66 as Lewis acid catalysts for enhanced glucose isomerization to fructose. Appl. Catal. A Gen. 2022, 632, 118501. [Google Scholar] [CrossRef]
- Ling, L.; Yang, W.; Yan, P.; Wang, M.; Jiang, H. Light-Assisted CO2 Hydrogenation over Pd3Cu@UiO-66 Promoted by Active Sites in Close Proximity. Angew. Chem. Int. Ed. 2022, 61, e202116396. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Dou, Y.; Xie, L.-H.; Rutledge, W.; Li, J.-R.; Zhou, H.-C. Zr-based metal–organic frameworks: Design, synthesis, structure, and applications. Chem. Soc. Rev. 2016, 45, 2327–2367. [Google Scholar] [CrossRef] [PubMed]
- Dhakshinamoorthy, A.; Santiago-Portillo, A.; Asiri, A.M.; Garcia, H. Engineering UiO-66 Metal Organic Framework for Heterogeneous Catalysis. ChemCatChem 2019, 11, 899–923. [Google Scholar] [CrossRef]
- Cavka, J.H.; Jakobsen, S.; Olsbye, U.; Guillou, N.; Lamberti, C.; Bordiga, S.; Lillerud, K.P. A New Zirconium Inorganic Building Brick Forming Metal Organic Frameworks with Exceptional Stability. J. Am. Chem. Soc. 2008, 130, 13850–13851. [Google Scholar] [CrossRef] [PubMed]
- Schelling, M.; Kim, M.; Otal, E.; Hinestroza, J. Decoration of Cotton Fibers with a Water-Stable Metal-Organic Framework (UiO-66) for the Decomposition and Enhanced Adsorption of Micropollutants in Water. Bioengineering 2018, 5, 14. [Google Scholar] [CrossRef] [Green Version]
- Cheng, J.; Qian, L.; Guo, H.; Mao, Y.; Shao, Y.; Yang, W. A new aminobenzoate-substituted s-triazin-based Zr metal organic frameworks as efficient catalyst for biodiesel production from microalgal lipids. Fuel Process. Technol. 2022, 238, 107487. [Google Scholar] [CrossRef]
- Zhou, F.; Lu, N.; Fan, B.; Wang, H.; Li, R. Zirconium-containing UiO-66 as an efficient and reusable catalyst for transesterification of triglyceride with methanol. J. Energy Chem. 2016, 25, 874–879. [Google Scholar] [CrossRef] [Green Version]
- Caratelli, C.; Hajek, J.; Cirujano, F.G.; Waroquier, M.; i Xamena, F.X.L.; Van Speybroeck, V. Nature of active sites on UiO-66 and beneficial influence of water in the catalysis of Fischer esterification. J. Catal. 2017, 352, 401–414. [Google Scholar] [CrossRef] [Green Version]
- Jrad, A.; Abu Tarboush, B.J.; Hmadeh, M.; Ahmad, M. Tuning acidity in zirconium-based metal organic frameworks catalysts for enhanced production of butyl butyrate. Appl. Catal. A Gen. 2019, 570, 31–41. [Google Scholar] [CrossRef]
- Wei, R.; Fan, J.; Qu, X.; Gao, L.; Wu, Y.; Zhang, Z.; Hu, F.; Xiao, G. Tuning the Catalytic Activity of UiO-66 via Modulated Synthesis: Esterification of Levulinic Acid as a Test Reaction. Eur. J. Inorg. Chem. 2020, 2020, 833–840. [Google Scholar] [CrossRef]
- Chaemchuen, S.; Heynderickx, P.M.; Verpoort, F. Kinetic modeling of oleic acid esterification with UiO-66: From intrinsic experimental data to kinetics via elementary reaction steps. Chem. Eng. J. 2020, 394, 124816. [Google Scholar] [CrossRef]
- Desidery, L.; Chaemcheun, S.; Yusubov, M.; Verpoort, F. Di-methyl carbonate transesterification with EtOH over MOFs: Basicity and synergic effect of basic and acid active sites. Catal. Commun. 2018, 104, 82–85. [Google Scholar] [CrossRef]
- Shaik, M.R.; Adil, S.F.; Alothman, Z.A.; Alduhaish, O.M. Fumarate Based Metal-Organic Framework: An Effective Catalyst for the Transesterification of Used Vegetable Oil. Crystals 2022, 12, 151. [Google Scholar] [CrossRef]
- de la Flor, D.; López-Aguado, C.; Paniagua, M.; Morales, G.; Mariscal, R.; Melero, J.A. Defective UiO-66(Zr) as an efficient catalyst for the synthesis of bio jet-fuel precursors via aldol condensation of furfural and MIBK. J. Catal. 2021, 401, 27–39. [Google Scholar] [CrossRef]
- Rapeyko, A.; Rodenas, M.; i Xamena, F.X.L. Zr-Containing UiO-66 Metal-Organic Frameworks as Highly Selective Heterogeneous Acid Catalysts for the Direct Ketalization of Levulinic Acid. Adv. Sustain. Syst. 2022, 6, 2100451. [Google Scholar] [CrossRef]
- Cirujano, F.; Corma, A.; i Xamena, F.L. Zirconium-containing metal organic frameworks as solid acid catalysts for the esterification of free fatty acids: Synthesis of biodiesel and other compounds of interest. Catal. Today 2015, 257, 213–220. [Google Scholar] [CrossRef]
- Cirujano, F.; Corma, A.; i Xamena, F.L. Conversion of levulinic acid into chemicals: Synthesis of biomass derived levulinate esters over Zr-containing MOFs. Chem. Eng. Sci. 2015, 124, 52–60. [Google Scholar] [CrossRef]
- Wang, F.; Chen, Z.; Chen, H.; Goetjen, T.A.; Li, P.; Wang, X.; Alayoglu, S.; Ma, K.; Chen, Y.; Wang, T.; et al. Interplay of Lewis and Brønsted Acid Sites in Zr-Based Metal-Organic Frameworks for Efficient Esterification of Biomass-Derived Levulinic Acid. ACS Appl. Mater. Interfaces 2019, 11, 32090–32096. [Google Scholar] [CrossRef]
- Abou-Elyazed, A.S.; Ye, G.; Sun, Y.; El-Nahas, A.M. A Series of UiO-66(Zr)-Structured Materials with Defects as Heterogeneous Catalysts for Biodiesel Production. Ind. Eng. Chem. Res. 2019, 58, 21961–21971. [Google Scholar] [CrossRef]
- Abou-Elyazed, A.S.; Sun, Y.; El-Nahas, A.M.; Yousif, A.M. A green approach for enhancing the hydrophobicity of UiO-66(Zr) catalysts for biodiesel production at 298 K. RSC Adv. 2020, 10, 41283–41295. [Google Scholar] [CrossRef]
- Desidery, L.; Yusubov, M.S.; Zhuiykov, S.; Verpoort, F. Fully-sulfonated hydrated UiO66 as efficient catalyst for ethyl levulinate production by esterification. Catal. Commun. 2018, 117, 33–37. [Google Scholar] [CrossRef]
- Badoei-Dalfard, A.; Shahba, A.; Zaare, F.; Sargazi, G.; Seyedalipour, B.; Karami, Z. Lipase immobilization on a novel class of Zr-MOF/electrospun nanofibrous polymers: Biochemical characterization and efficient biodiesel production. Int. J. Biol. Macromol. 2021, 192, 1292–1303. [Google Scholar] [CrossRef]
- Li, H.; Han, Z.; Liu, F.; Li, G.; Guo, M.; Cui, P.; Zhou, S.; Yu, M. Esterification catalyzed by an efficient solid acid synthesized from PTSA and UiO-66(Zr) for biodiesel production. Faraday Discuss. 2021, 231, 342–355. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhao, G.; Ullah, L.; Wang, M.; Wang, A.; Zhang, Y.; Zhang, S. Acidic ionic liquid based UiO-67 type MOFs: A stable and efficient heterogeneous catalyst for esterification. RSC Adv. 2018, 8, 10009–10016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, W.-L.; Mi, J.; Liu, F.; Xiao, Y.; Chen, W.; Liu, Z.; Yi, X.; Liu, W.; Zheng, A. Accelerating Biodiesel Catalytic Production by Confined Activation of Methanol over High-Concentration Ionic Liquid-Grafted UiO-66 Solid Superacids. ACS Catal. 2020, 10, 11848–11856. [Google Scholar] [CrossRef]
- Dai, Q.; Yang, Z.; Li, J.; Cao, Y.; Tang, H.; Wei, X. Zirconium-based MOFs-loaded ionic liquid-catalyzed preparation of biodiesel from Jatropha oil. Renew. Energy 2021, 163, 1588–1594. [Google Scholar] [CrossRef]
- Lu, P.; Li, H.; Li, M.; Chen, J.; Ye, C.; Wang, H.; Qiu, T. Ionic liquid grafted NH2-UiO-66 as heterogeneous solid acid catalyst for biodiesel production. Fuel 2022, 324, 124537. [Google Scholar] [CrossRef]
- Abou-Elyazed, A.S.; Sun, Y.; El-Nahas, A.; Abdel-Azeim, S.; Sharara, T.; Yousif, A. Solvent-free synthesis and characterization of Ca2+-doped UiO-66(Zr) as heterogeneous catalyst for esterification of oleic acid with methanol: A joint experimental and computational study. Mater. Today Sustain. 2022, 18, 100110. [Google Scholar] [CrossRef]
- Zhu, J.; Wang, Z.; Song, X.; Zhao, B.; Li, Y.; Wang, Y. Encapsulating Keggin-H3PW12O40 into UIO-66(Zr) for manufacturing the biodiesel. Micro Nano Lett. 2021, 16, 90–96. [Google Scholar] [CrossRef]
- Ma, T.; Liu, D.; Liu, Z.; Xu, J.; Dong, Y.; Chen, G.; Yun, Z. 12-Tungstophosphoric acid-encapsulated metal-organic framework UiO-66: A promising catalyst for the esterification of acetic acid with n-butanol. J. Taiwan Inst. Chem. Eng. 2022, 133, 104277. [Google Scholar] [CrossRef]
- Xie, W.; Hu, P.; Yang, X. Cs2.5H0.5PW12O40 Encapsulated in Metal-Organic Framework UiO-66 as Heterogeneous Catalysts for Acidolysis of Soybean Oil. Catal. Lett. 2017, 147, 2772–2782. [Google Scholar] [CrossRef]
- Xie, W.; Wan, F. Immobilization of polyoxometalate-based sulfonated ionic liquids on UiO-66-2COOH metal-organic frameworks for biodiesel production via one-pot transesterification-esterification of acidic vegetable oils. Chem. Eng. J. 2019, 365, 40–50. [Google Scholar] [CrossRef]
- Tan, X.; Zhang, H.; Li, H.; Yang, S. Electrovalent bifunctional acid enables heterogeneously catalytic production of biodiesel by (trans)esterification of non-edible oils. Fuel 2022, 310, 122273. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, S.; Li, Z.; Zhang, H.; Li, H.; Yang, S. Green synthesis of heterogeneous polymeric bio-based acid decorated with hydrophobic regulator for efficient catalytic production of biodiesel at low temperatures. Fuel 2022, 329, 125467. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, X.; Li, S.; Zhao, B.; Tong, L.; Wang, Y.; Li, Y. Two-step preparation of Keggin-PW12@UIO-66 composite showing high-activity and long-life conversion of soybean oil into biodiesel. RSC Adv. 2021, 11, 38016–38025. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yang, T.; Liu, X.; Yue, C.; Ao, L.; Deng, T.; Zhang, Y. Heteropoly acid-encapsulated metal–organic framework as a stable and highly efficient nanocatalyst for esterification reaction. RSC Adv. 2019, 9, 16357–16365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Lei, D.; Luo, Q.; Wang, J.; Deng, T.; Zhang, Y.; Ma, P. Efficient biodiesel production from oleic acid using metal–organic framework encapsulated Zr-doped polyoxometalate nano-hybrids. RSC Adv. 2020, 10, 8766–8772. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, T.; Lei, D.; Wang, J.; Zhang, Y. Efficient Production of Biodiesel from Esterification of Lauric Acid Catalyzed by Ammonium and Silver Co-Doped Phosphotungstic Acid Embedded in a Zirconium Metal-Organic Framework Nanocomposite. ACS Omega 2020, 5, 12760–12767. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, B.; Tian, Y.; Yang, X.; Yu, R.; Wang, J.; Deng, T.; Zhang, Y. Fabrication of silicotungstic acid immobilized on Ce-based MOF and embedded in Zr-based MOF matrix for green fatty acid esterification. Green Process. Synth. 2022, 11, 184–194. [Google Scholar] [CrossRef]
- Liu, B.; Shioyama, H.; Akita, T.; Xu, Q. Metal-Organic Framework as a Template for Porous Carbon Synthesis. J. Am. Chem. Soc. 2008, 130, 5390–5391. [Google Scholar] [CrossRef]
- Liu, B.; Zhang, X.; Shioyama, H.; Mukai, T.; Sakai, T.; Xu, Q. Converting cobalt oxide subunits in cobalt metal-organic framework into agglomerated Co3O4 nanoparticles as an electrode material for lithium ion battery. J. Power Sources 2010, 195, 857–861. [Google Scholar] [CrossRef]
- Lu, N.; Zhang, X.; Yan, X.; Pan, D.; Fan, B.; Li, R. Synthesis of novel mesoporous sulfated zirconia nanosheets derived from Zr-based metal–organic frameworks. CrystEngComm 2020, 22, 44–51. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; Ma, X.; Guo, M.; Li, Y.; Li, G.; Cui, P.; Zhou, S.; Yu, M. Synthesis of CaO/ZrO2 based catalyst by using UiO–66(Zr) and calcium acetate for biodiesel production. Renew. Energy 2022, 185, 970–977. [Google Scholar] [CrossRef]
- Zhang, Q.; Lei, D.; Luo, Q.; Yang, X.; Wu, Y.; Wang, J.; Zhang, Y. MOF-derived zirconia-supported Keggin heteropoly acid nanoporous hybrids as a reusable catalyst for methyl oleate production. RSC Adv. 2021, 11, 8117–8123. [Google Scholar] [CrossRef] [PubMed]
- Goda, M.N.; Abdelhamid, H.N.; Said, A.E.-A.A. Zirconium Oxide Sulfate-Carbon (ZrOSO4@C) Derived from Carbonized UiO-66 for Selective Production of Dimethyl Ether. ACS Appl. Mater. Interfaces 2019, 12, 646–653. [Google Scholar] [CrossRef]
- Liu, T.; Hong, X.; Liu, G. In Situ Generation of the Cu@3D-ZrOx Framework Catalyst for Selective Methanol Synthesis from CO2/H2. ACS Catal. 2019, 10, 93–102. [Google Scholar] [CrossRef]
- Zeng, L.; Wang, Y.; Li, Z.; Song, Y.; Zhang, J.; Wang, J.; He, X.; Wang, C.; Lin, W. Highly Dispersed Ni Catalyst on Metal-Organic Framework-Derived Porous Hydrous Zirconia for CO2 Methanation. ACS Appl. Mater. Interfaces 2020, 12, 17436–17442. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Q.; Wang, J.; Zhang, S.; Ma, J.; Cheng, J.; Zhang, Y. Zr-Based Metal-Organic Frameworks for Green Biodiesel Synthesis: A Minireview. Bioengineering 2022, 9, 700. https://doi.org/10.3390/bioengineering9110700
Zhang Q, Wang J, Zhang S, Ma J, Cheng J, Zhang Y. Zr-Based Metal-Organic Frameworks for Green Biodiesel Synthesis: A Minireview. Bioengineering. 2022; 9(11):700. https://doi.org/10.3390/bioengineering9110700
Chicago/Turabian StyleZhang, Qiuyun, Jialu Wang, Shuya Zhang, Juan Ma, Jingsong Cheng, and Yutao Zhang. 2022. "Zr-Based Metal-Organic Frameworks for Green Biodiesel Synthesis: A Minireview" Bioengineering 9, no. 11: 700. https://doi.org/10.3390/bioengineering9110700
APA StyleZhang, Q., Wang, J., Zhang, S., Ma, J., Cheng, J., & Zhang, Y. (2022). Zr-Based Metal-Organic Frameworks for Green Biodiesel Synthesis: A Minireview. Bioengineering, 9(11), 700. https://doi.org/10.3390/bioengineering9110700









