Taking It Personally: 3D Bioprinting a Patient-Specific Cardiac Patch for the Treatment of Heart Failure
Abstract
:1. Introduction
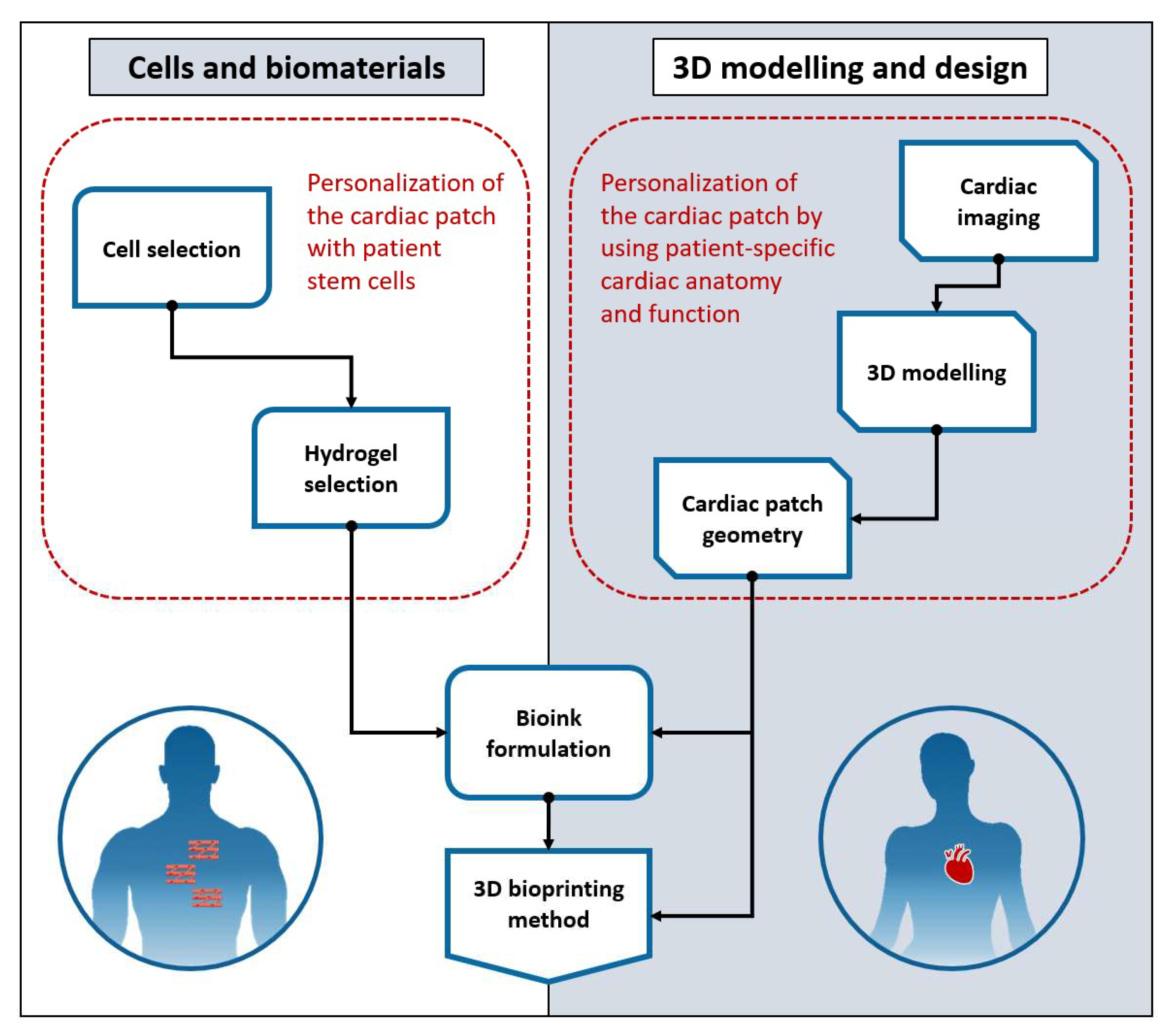
2. Overview
2.1. Bioinks and 3D Bioprinting Methods
2.1.1. Bioink Formulation
2.1.2. Three-Dimensional Bioprinting Methods
2.2. Three-Dimensional Modelling and Design
2.2.1. Cardiac CT
2.2.2. Cardiac MRI
2.2.3. Hybrid Imaging
2.2.4. Three-Dimensional Modelling
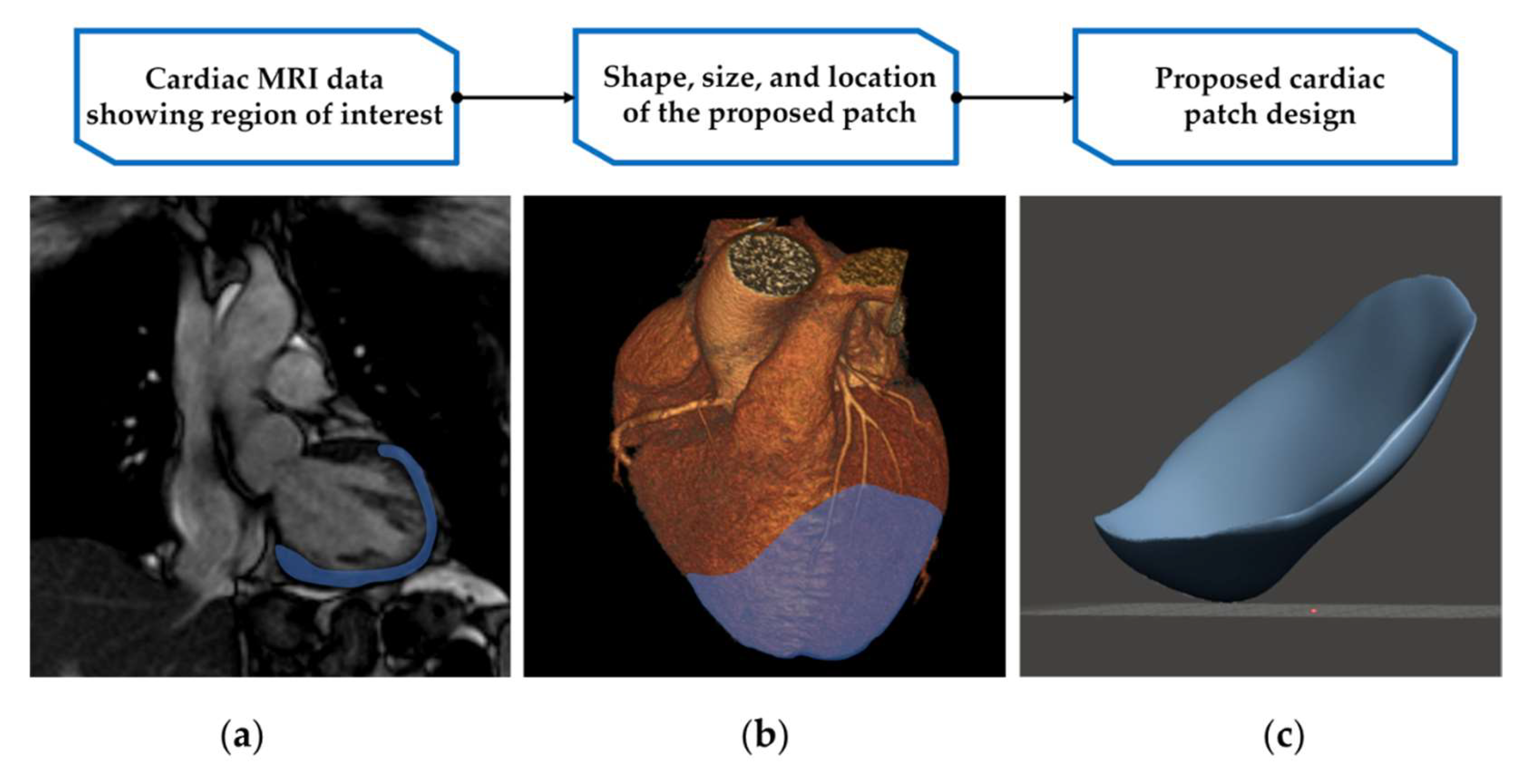
2.2.5. Cardiac Patch Design
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. The Top 10 Causes of Death. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 23 October 2021).
- World Health Statistics 2021: Monitoring Health for the SDGs, Sustainable Development Goals; World Health Organization: Geneva, Switzerland, 2021.
- Zhu, D.; Cheng, K. Cardiac Cell Therapy for Heart Repair: Should the Cells Be Left Out? Cells 2021, 10, 641. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Serpooshan, V.; Zhang, J. Engineering Human Cardiac Muscle Patch Constructs for Prevention of Post-infarction LV Remodeling. Front. Cardiovasc. Med. 2021, 8, 621781. [Google Scholar] [CrossRef] [PubMed]
- Tomasoni, D.; Adamo, M.; Anker, M.S.; von Haehling, S.; Coats, A.J.S.; Metra, M. Heart failure in the last year: Progress and perspective. ESC Heart Fail. 2020, 7, 3505–3530. [Google Scholar] [CrossRef]
- Liu, N.; Ye, X.; Yao, B.; Zhao, M.; Wu, P.; Liu, G.; Zhuang, D.; Jiang, H.; Chen, X.; He, Y.; et al. Advances in 3D bioprinting technology for cardiac tissue engineering and regeneration. Bioact. Mater. 2021, 6, 1388–1401. [Google Scholar] [CrossRef] [PubMed]
- Bowen, R.E.S.; Graetz, T.J.; Emmert, D.A.; Avidan, M.S. Statistics of heart failure and mechanical circulatory support in 2020. Ann. Transl. Med. 2020, 8, 827. [Google Scholar] [CrossRef] [PubMed]
- Khush, K.K.; Cherikh, W.S.; Chambers, D.C.; Harhay, M.O.; Hayes, D., Jr.; Hsich, E.; Meiser, B.; Potena, L.; Robinson, A.; Rossano, J.W.; et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-sixth adult heart transplantation report—2019; focus theme: Donor and recipient size match. J. Heart Lung Transplant. 2019, 38, 1056–1066. [Google Scholar] [CrossRef] [PubMed]
- Roshanbinfar, K.; Esser, T.U.; Engel, F.B. Stem Cells and Their Cardiac Derivatives for Cardiac Tissue Engineering and Regenerative Medicine. Antioxid. Redox Signal. 2021, 35, 143–162. [Google Scholar] [CrossRef]
- Carotenuto, F.; Teodori, L.; Maccari, A.M.; Delbono, L.; Orlando, G.; Di Nardo, P. Turning regenerative technologies into treatment to repair myocardial injuries. J. Cell. Mol. Med. 2020, 24, 2704–2716. [Google Scholar] [CrossRef] [Green Version]
- Sun, Z. Clinical applications of patient-specific 3d printed models in cardiovascular disease: Current status and future directions. Biomolecules 2020, 10, 1577. [Google Scholar] [CrossRef]
- Gardin, C.; Ferroni, L.; Latremouille, C.; Chachques, J.C.; Mitrečić, D.; Zavan, B. Recent Applications of Three Dimensional Printing in Cardiovascular Medicine. Cells 2020, 9, 742. [Google Scholar] [CrossRef] [Green Version]
- Birbara, N.S.; Otton, J.M.; Pather, N. 3D Modelling and Printing Technology to Produce Patient-Specific 3D Models. Hear. Lung Circ. 2019, 28, 302–313. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef]
- Heinrich, M.A.; Liu, W.; Jimenez, A.; Yang, J.; Akpek, A.; Liu, X.; Pi, Q.; Mu, X.; Hu, N.; Schiffelers, R.M.; et al. 3D Bioprinting: From Benches to Translational Applications. Small 2019, 15, e1805510. [Google Scholar] [CrossRef]
- Roche, C.D.; Brereton, R.J.L.; Ashton, A.W.; Jackson, C.; Gentile, C. Current challenges in three-dimensional bioprinting heart tissues for cardiac surgery. Eur. J. Cardiothorac. Surg. 2020, 58, 500–510. [Google Scholar] [CrossRef]
- Noor, N.; Shapira, A.; Edri, R.; Gal, I.; Wertheim, L.; Dvir, T. 3D Printing of Personalized Thick and Perfusable Cardiac Patches and Hearts. Adv. Sci. 2019, 6, 1900344. [Google Scholar] [CrossRef] [Green Version]
- Qin, X.; Riegler, J.; Tiburcy, M.; Zhao, X.; Chour, T.; Ndoye, B.; Nguyen, M.; Adams, J.; Ameen, M.; Denney, T.S.; et al. Magnetic Resonance Imaging of Cardiac Strain Pattern Following Transplantation of Human Tissue Engineered Heart Muscles. Circ. Cardiovasc. Imaging 2016, 9, e004731. [Google Scholar] [CrossRef] [Green Version]
- Menasché, P.; Vanneaux, V.; Hagège, A.; Bel, A.; Cholley, B.; Parouchev, A.; Cacciapuoti, I.; Al-Daccak, R.; Benhamouda, N.; Blons, H.; et al. Transplantation of Human Embryonic Stem Cell-Derived Cardiovascular Progenitors for Severe Ischemic Left Ventricular Dysfunction. J. Am. Coll. Cardiol. 2018, 71, 429–438. [Google Scholar] [CrossRef]
- Sawa, Y.; Yoshikawa, Y.; Toda, K.; Fukushima, S.; Yamazaki, K.; Ono, M.; Sakata, Y.; Hagiwara, N.; Kinugawa, K.; Miyagawa, S. Safety and Efficacy of Autologous Skeletal Myoblast Sheets (TCD-51073) for the Treatment of Severe Chronic Heart Failure Due to Ischemic Heart Disease. Circ. J. 2015, 79, 991–999. [Google Scholar] [CrossRef] [Green Version]
- Yoshikawa, Y.; Miyagawa, S.; Toda, K.; Saito, A.; Sakata, Y.; Sawa, Y. Myocardial regenerative therapy using a scaffold-free skeletal-muscle-derived cell sheet in patients with dilated cardiomyopathy even under a left ventricular assist device: A safety and feasibility study. Surg. Today 2018, 48, 200–210. [Google Scholar] [CrossRef]
- Agarwal, T.; Fortunato, G.M.; Hann, S.Y.; Ayan, B.; Vajanthri, K.Y.; Presutti, D.; Cui, H.; Chan, A.H.P.; Costantini, M.; Onesto, V.; et al. Recent advances in bioprinting technologies for engineering cardiac tissue. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 124, 112057. [Google Scholar] [CrossRef]
- Byrne, N.; Velasco Forte, M.; Tandon, A.; Valverde, I.; Hussain, T. A systematic review of image segmentation methodology, used in the additive manufacture of patient-specific 3D printed models of the cardiovascular system. JRSM Cardiovasc. Dis. 2016, 5, 2048004016645467. [Google Scholar] [CrossRef] [Green Version]
- Ozbolat, I.; Gudapati, H. A review on design for bioprinting. Bioprinting 2016, 3, 1–14. [Google Scholar] [CrossRef]
- Arguchinskaya, N.V.; Beketov, E.E.; Kisel, A.A.; Isaeva, E.V.; Osidak, E.O.; Domogatsky, S.P.; Mikhailovsky, N.V.; Sevryukov, F.E.; Silantyeva, N.K.; Agababyan, T.A.; et al. The Technique of Thyroid Cartilage Scaffold Support Formation for Extrusion-Based Bioprinting. Int. J. Bioprint 2021, 7, 348. [Google Scholar] [CrossRef]
- Mancuso, A.; Barone, A.; Cristiano, M.C.; Cianflone, E.; Fresta, M.; Paolino, D. Cardiac stem cell-loaded delivery systems: A new challenge for myocardial tissue regeneration. Int. J. Mol. Sci. 2020, 21, 7701. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Pretorius, D.; Zhang, J. Cardiac Patch-Based Therapies of Ischemic Heart Injuries. In Cardiovascular Regenerative Medicine: Tissue Engineering and Clinical Applications; Serpooshan, V., Wu, S.M., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 141–171. [Google Scholar]
- Arai, K.; Murata, D.; Takao, S.; Nakayama, K. Fabrication of Cardiac Constructs Using Bio-3D Printer. Methods Mol. Biol. 2021, 2320, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Birla, R.K. A methodological nine-step process to bioengineer heart muscle tissue. Tissue Cell 2020, 67, 101425. [Google Scholar] [CrossRef] [PubMed]
- Datta, P.; Barui, A.; Wu, Y.; Ozbolat, V.; Moncal, K.K.; Ozbolat, I.T. Essential steps in bioprinting: From pre- to post-bioprinting. Biotechnol. Adv. 2018, 36, 1481–1504. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, L.; Zhu, Y.; Woo, Y.J.; Appel, E. Engineered biomaterials for heart disease. Curr. Opin. Biotechnol. 2020, 66, 246–254. [Google Scholar] [CrossRef]
- Kumar, M.; Sharma, V. Additive manufacturing techniques for the fabrication of tissue engineering scaffolds: A review. Rapid Prototyp. J. 2021, 27, 1230–1272. [Google Scholar] [CrossRef]
- Birla, R.K.; Williams, S.K. 3D bioprinting and its potential impact on cardiac failure treatment: An industry perspective. APL Bioeng. 2020, 4, 010903. [Google Scholar] [CrossRef] [Green Version]
- Ng, W.L.; Chua, C.K.; Shen, Y.-F. Print Me An Organ! Why We Are Not There Yet. Prog. Polym. Sci. 2019, 97, 101145. [Google Scholar] [CrossRef]
- Serpooshan, V.; Mahmoudi, M.; Hu, D.A.; Hu, J.B.; Wu, S.M. Bioengineering cardiac constructs using 3D printing. J. 3D Print Med. 2017, 1, 123–139. [Google Scholar] [CrossRef]
- Budharaju, H.; Subramanian, A.; Sethuraman, S. Recent advancements in cardiovascular bioprinting and bioprinted cardiac constructs. Biomater. Sci. 2021, 9, 1974–1994. [Google Scholar] [CrossRef]
- Sedlakova, V.; Ahumada, M.; Suuronen, E.J.; Alarcon, E.I. Building new cardiac vasculature and myocardium: Where are we at? Curr. Opin. Cardiol. 2021, 36, 728–734. [Google Scholar] [CrossRef]
- Shapira, A.; Dvir, T. 3D Tissue and Organ Printing—Hope and Reality. Adv. Sci. (Weinh.) 2021, 8, 2003751. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, L.; Li, T.; Liu, S.; Guo, B.; Huang, W.; Wu, Y. 3D bioprinting in cardiac tissue engineering. Theranostics 2021, 11, 7948–7969. [Google Scholar] [CrossRef]
- Yadid, M.; Oved, H.; Silberman, E.; Dvir, T. Bioengineering approaches to treat the failing heart: From cell biology to 3D printing. Nat. Rev. Cardiol. 2021, 16, 83–99. [Google Scholar] [CrossRef]
- Montero, P.; Flandes-Iparraguirre, M.; Musquiz, S.; Pérez Araluce, M.; Plano, D.; Sanmartín, C.; Orive, G.; Gavira, J.J.; Prosper, F.; Mazo, M.M. Cells, Materials, and Fabrication Processes for Cardiac Tissue Engineering. Front. Bioeng. Biotechnol. 2020, 8, 955. [Google Scholar] [CrossRef]
- Mazzola, M.; Di Pasquale, E. Toward Cardiac Regeneration: Combination of Pluripotent Stem Cell-Based Therapies and Bioengineering Strategies. Front. Bioeng. Biotechnol. 2020, 8, 455. [Google Scholar] [CrossRef]
- Scognamiglio, C.; Soloperto, A.; Ruocco, G.; Cidonio, G. Bioprinting stem cells: Building physiological tissues one cell at a time. Am. J. Physiol. Cell Physiol. 2020, 319, C465–C480. [Google Scholar] [CrossRef]
- Xie, Z.; Gao, M.; Lobo, A.O.; Webster, T.J. 3D Bioprinting in Tissue Engineering for Medical Applications: The Classic and the Hybrid. Polymers 2020, 12, 1717. [Google Scholar] [CrossRef]
- Leberfinger, A.N.; Dinda, S.; Wu, Y.; Koduru, S.V.; Ozbolat, V.; Ravnic, D.J.; Ozbolat, I.T. Bioprinting functional tissues. Acta Biomater. 2019, 95, 32–49. [Google Scholar] [CrossRef]
- Hölzl, K.; Lin, S.; Tytgat, L.; Van Vlierberghe, S.; Gu, L.; Ovsianikov, A. Bioink properties before, during and after 3D bioprinting. Biofabrication 2016, 8, 032002. [Google Scholar] [CrossRef]
- Tadevosyan, K.; Iglesias-García, O.; Mazo, M.M.; Prósper, F.; Raya, A. Engineering and assessing cardiac tissue complexity. Int. J. Mol. Sci. 2021, 22, 1479. [Google Scholar] [CrossRef]
- Yilmaz, B.; Al Rashid, A.; Mou, Y.A.; Evis, Z.; Koç, M. Bioprinting: A review of processes, materials and applications. Bioprinting 2021, 23, e00148. [Google Scholar] [CrossRef]
- Subramaniyan, M.; Eswaran, P.; Appusamy, A.; Srimannarayana Raju, P.; Rahini, V.; Madhumitha, T.R.; Thisha, R. A survey on applications of additive manufacturing techniques in tissue engineering. Mater. Today Proc. 2021, 45, 8036–8040. [Google Scholar] [CrossRef]
- Matai, I.; Kaur, G.; Seyedsalehi, A.; McClinton, A.; Laurencin, C.T. Progress in 3D bioprinting technology for tissue/organ regenerative engineering. Biomaterials 2020, 226, 119536. [Google Scholar] [CrossRef]
- Ozbolat, I.T.; Moncal, K.K.; Gudapati, H. Evaluation of bioprinter technologies. Addit. Manuf. 2017, 13, 179–200. [Google Scholar] [CrossRef] [Green Version]
- Askari, M.; Afzali Naniz, M.; Kouhi, M.; Saberi, A.; Zolfagharian, A.; Bodaghi, M. Recent progress in extrusion 3D bioprinting of hydrogel biomaterials for tissue regeneration: A comprehensive review with focus on advanced fabrication techniques. Biomater. Sci. 2021, 9, 535–573. [Google Scholar] [CrossRef] [PubMed]
- Tomov, M.L.; Gil, C.J.; Cetnar, A.; Theus, A.S.; Lima, B.J.; Nish, J.E.; Bauser-Heaton, H.D.; Serpooshan, V. Engineering Functional Cardiac Tissues for Regenerative Medicine Applications. Curr. Cardiol. Rep. 2019, 21, 105. [Google Scholar] [CrossRef] [PubMed]
- Polonchuk, L.; Surija, L.; Lee, M.; Sharma, P.; Liu Chung Ming, C.; Richter, F.; Ben-Sefer, E.; Rad, M.A.; Mahmodi, H.; Shamery, W.; et al. Towards engineering heart tissues from bioprinted cardiac spheroids. Biofabrication 2021, 13, 045009. [Google Scholar] [CrossRef]
- Roche, C.D.; Sharma, P.; Ashton, A.W.; Jackson, C.; Xue, M.; Gentile, C. Printability, Durability, Contractility and Vascular Network Formation in 3D Bioprinted Cardiac Endothelial Cells Using Alginate–Gelatin Hydrogels. Front. Bioeng. Biotechnol. 2021, 9, 636257. [Google Scholar] [CrossRef]
- Davoodi, E.; Sarikhani, E.; Montazerian, H.; Ahadian, S.; Costantini, M.; Swieszkowski, W.; Willerth, S.; Walus, K.; Mofidfar, M.; Toyserkani, E.; et al. Extrusion and Microfluidic-based Bioprinting to Fabricate Biomimetic Tissues and Organs. Adv. Mater. Technol. 2020, 5, 1901044. [Google Scholar] [CrossRef]
- Maiullari, F.; Costantini, M.; Milan, M.; Pace, V.; Chirivì, M.; Maiullari, S.; Rainer, A.; Baci, D.; Marei, H.E.S.; Seliktar, D.; et al. A multi-cellular 3D bioprinting approach for vascularized heart tissue engineering based on HUVECs and iPSC-derived cardiomyocytes. Sci. Rep. 2018, 8, 13532. [Google Scholar] [CrossRef]
- Ripley, B.; Levin, D.; Kelil, T.; Hermsen, J.L.; Kim, S.; Maki, J.H.; Wilson, G.J. 3D printing from MRI Data: Harnessing strengths and minimizing weaknesses. J. Magn. Reson. Imaging 2017, 45, 635–645. [Google Scholar] [CrossRef]
- Storey, P. Imaging for cardiac disease: A practical guide for general practitioners. Aust. Fam. Phys. 2014, 43, 260–263. [Google Scholar]
- O’Dell, W.G.; McCulloch, A.D. Imaging three-dimensional cardiac function. Annu. Rev. Biomed. Eng. 2000, 2, 431–456. [Google Scholar] [CrossRef]
- Gaemperli, O.; Saraste, A.; Knuuti, J. Cardiac hybrid imaging. Eur. Heart J. Cardiovasc. Imaging 2012, 13, 51–60. [Google Scholar] [CrossRef] [Green Version]
- Otton, J.M.; Birbara, N.S.; Hussain, T.; Greil, G.; Foley, T.A.; Pather, N. 3D printing from cardiovascular CT: A practical guide and review. Cardiovasc. Diagn. Ther. 2017, 7, 507–526. [Google Scholar] [CrossRef] [Green Version]
- Yoo, S.J.; Hussein, N.; Peel, B.; Coles, J.; van Arsdell, G.S.; Honjo, O.; Haller, C.; Lam, C.Z.; Seed, M.; Barron, D. 3D Modeling and Printing in Congenital Heart Surgery: Entering the Stage of Maturation. Front. Pediatr. 2021, 9, 621672. [Google Scholar] [CrossRef]
- Tavakoli, V.; Amini, A.A. A survey of shaped-based registration and segmentation techniques for cardiac images. Comput. Vis. Image Underst. 2013, 117, 966–989. [Google Scholar] [CrossRef]
- Chitiboi, T.; Axel, L. Magnetic resonance imaging of myocardial strain: A review of current approaches. J. Magn. Reson. Imaging 2017, 46, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Saraste, A.; Knuuti, J. ESC 2019 guidelines for the diagnosis and management of chronic coronary syndromes. Herz 2020, 45, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Machida, H.; Tanaka, I.; Fukui, R.; Shen, Y.; Ishikawa, T.; Tate, E.; Ueno, E. Current and Novel Imaging Techniques in Coronary CT. RadioGraphics 2015, 35, 991–1010. [Google Scholar] [CrossRef]
- Nieman, K.; Balla, S. Dynamic CT myocardial perfusion imaging. J. Cardiovasc. Comput. Tomogr. 2020, 14, 303–306. [Google Scholar] [CrossRef]
- Lopez-Perez, A.; Sebastian, R.; Ferrero, J.M. Three-dimensional cardiac computational modelling: Methods, features and applications. Biomed. Eng. Online 2015, 14, 35. [Google Scholar] [CrossRef] [Green Version]
- Ballyns, J.J.; Bonassar, L.J. Image-guided tissue engineering. J. Cell. Mol. Med. 2009, 13, 1428–1436. [Google Scholar] [CrossRef]
- Peng, P.; Lekadir, K.; Gooya, A.; Shao, L.; Petersen, S.E.; Frangi, A.F. A review of heart chamber segmentation for structural and functional analysis using cardiac magnetic resonance imaging. Magma 2016, 29, 155–195. [Google Scholar] [CrossRef] [Green Version]
- Moghari, M.H.; Barthur, A.; Amaral, M.E.; Geva, T.; Powell, A.J. Free-breathing whole-heart 3D cine magnetic resonance imaging with prospective respiratory motion compensation. Magn. Reson. Med. 2018, 80, 181–189. [Google Scholar] [CrossRef]
- Kido, T.; Kido, T.; Nakamura, M.; Watanabe, K.; Schmidt, M.; Forman, C.; Mochizuki, T. Compressed sensing real-time cine cardiovascular magnetic resonance: Accurate assessment of left ventricular function in a single-breath-hold. J. Cardiovasc. Magn. Reson. 2016, 18, 50. [Google Scholar] [CrossRef] [Green Version]
- Almutairi, H.M.; Boubertakh, R.; Miquel, M.E.; Petersen, S.E. Myocardial deformation assessment using cardiovascular magnetic resonance-feature tracking technique. Br. J. Radiol. 2017, 90, 20170072. [Google Scholar] [CrossRef]
- Ibrahim, E.-S.H. Myocardial tagging by cardiovascular magnetic resonance: Evolution of techniques–pulse sequences, analysis algorithms, and applications. J. Cardiovasc. Magn. Reson. 2011, 13, 36. [Google Scholar] [CrossRef] [Green Version]
- Rizvi, A.; Han, D.; Danad, I.; Ó Hartaigh, B.; Lee, J.H.; Gransar, H.; Stuijfzand, W.J.; Roudsari, H.M.; Park, M.W.; Szymonifka, J.; et al. Diagnostic Performance of Hybrid Cardiac Imaging Methods for Assessment of Obstructive Coronary Artery Disease Compared With Stand-Alone Coronary Computed Tomography Angiography: A Meta-Analysis. JACC Cardiovasc. Imaging 2018, 11, 589–599. [Google Scholar] [CrossRef]
- Stolzmann, P.; Alkadhi, H.; Scheffel, H.; Hennemuth, A.; Kuehnel, C.; Baumueller, S.; Kozerke, S.; Falk, V.; Marincek, B.; Donati, O.F. Image fusion of coronary CT angiography and cardiac perfusion MRI: A pilot study. Eur. Radiol. 2010, 20, 1174–1179. [Google Scholar] [CrossRef] [Green Version]
- von Spiczak, J.; Manka, R.; Gotschy, A.; Oebel, S.; Kozerke, S.; Hamada, S.; Alkadhi, H. Fusion of CT coronary angiography and whole-heart dynamic 3D cardiac MR perfusion: Building a framework for comprehensive cardiac imaging. Int. J. Card. Imaging 2018, 34, 649–660. [Google Scholar] [CrossRef]
- Habijan, M.; Babin, D.; Galić, I.; Leventić, H.; Romić, K.; Velicki, L.; Pižurica, A. Overview of the Whole Heart and Heart Chamber Segmentation Methods. Cardiovasc. Eng. Technol. 2020, 11, 725–747. [Google Scholar] [CrossRef]
- Bertolini, M.; Rossoni, M.; Colombo, G. Operative Workflow from CT to 3D Printing of the Heart: Opportunities and Challenges. Bioengineering 2021, 8, 130. [Google Scholar] [CrossRef]
- Abudayyeh, I.; Gordon, B.; Ansari, M.M.; Jutzy, K.; Stoletniy, L.; Hilliard, A. A practical guide to cardiovascular 3D printing in clinical practice: Overview and examples. J. Interv. Cardiol. 2018, 31, 375–383. [Google Scholar] [CrossRef]
- Wang, Z.; Lee, S.J.; Cheng, H.J.; Yoo, J.J.; Atala, A. 3D bioprinted functional and contractile cardiac tissue constructs. Acta Biomater. 2018, 70, 48–56. [Google Scholar] [CrossRef]
- Tian, S.; Zhao, H.; Lewinski, N. Key parameters and applications of extrusion-based bioprinting. Bioprinting 2021, 23, e00156. [Google Scholar] [CrossRef]
- Naveed, M.; Han, L.; Khan, G.J.; Yasmeen, S.; Mikrani, R.; Abbas, M.; Cunyu, L.; Xiaohui, Z. Cardio-supportive devices (VRD & DCC device) and patches for advanced heart failure: A review, summary of state of the art and future directions. Biomed. Pharmacother. 2018, 102, 41–54. [Google Scholar] [CrossRef]
- Bejleri, D.; Streeter, B.W.; Nachlas, A.L.Y.; Brown, M.E.; Gaetani, R.; Christman, K.L.; Davis, M.E. A Bioprinted Cardiac Patch Composed of Cardiac-Specific Extracellular Matrix and Progenitor Cells for Heart Repair. Adv. Healthc. Mater. 2018, 7, e1800672. [Google Scholar] [CrossRef]
- Huang, S.; Lei, D.; Yang, Q.; Yang, Y.; Jiang, C.; Shi, H.; Qian, B.; Long, Q.; Chen, W.; Chen, Y.; et al. A perfusable, multifunctional epicardial device improves cardiac function and tissue repair. Nat. Med. 2021, 27, 480–490. [Google Scholar] [CrossRef]
- Yang, Y.; Lei, D.; Huang, S.; Yang, Q.; Song, B.; Guo, Y.; Shen, A.; Yuan, Z.; Li, S.; Qing, F.L.; et al. Elastic 3D-Printed Hybrid Polymeric Scaffold Improves Cardiac Remodeling after Myocardial Infarction. Adv. Healthc. Mater. 2019, 8, e1900065. [Google Scholar] [CrossRef]
- Baker, R.S.; Zafar, F.; Kimura, N.; Knilans, T.; Osinska, H.; Robbins, J.; Taylor, M.; Morales, D.L.S. In Vivo Remodeling of an Extracellular Matrix Cardiac Patch in an Ovine Model. Asaio J. 2019, 65, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Scully, B.B.; Fan, C.; Grigoryan, B.; Jacot, J.G.; Vick, G.W., 3rd; Kim, J.J.; Fraser, C.D., Jr.; Grande-Allen, K.J.; Morales, D.L. Remodeling of ECM patch into functional myocardium in an ovine model: A pilot study. J. Biomed. Mater. Res. B Appl. Biomater. 2016, 104, 1713–1720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giannopoulos, A.A.; Mitsouras, D.; Yoo, S.J.; Liu, P.P.; Chatzizisis, Y.S.; Rybicki, F.J. Applications of 3D printing in cardiovascular diseases. Nat. Rev. Cardiol. 2016, 13, 701–718. [Google Scholar] [CrossRef] [PubMed]
- Mangion, K.; Gao, H.; Husmeier, D.; Luo, X.; Berry, C. Advances in computational modelling for personalised medicine after myocardial infarction. Heart 2018, 104, 550–557. [Google Scholar] [CrossRef] [Green Version]
- Pixmeo. DICOM Image Library. Available online: http://www.osirix-viewer.com/resources/dicom-image-library/ (accessed on 14 February 2022).
- Zhang, J.; Zhu, W.; Radisic, M.; Vunjak-Novakovic, G. Can We Engineer a Human Cardiac Patch for Therapy? Circ. Res. 2018, 123, 244–265. [Google Scholar] [CrossRef]
- Moore, K.L. Clinically Oriented Anatomy, 8th ed.; Wolters Kluwer: Philadelphia, PA, USA, 2018. [Google Scholar]
- Grabczak, E.M.; Stec, S.; Dabrowska, M.; Plevkova, J.; Krenke, R. Cough as a Cause and Consequence of Heart Dysfunction—Current State of Art. Physiol. Res. 2020, 69, S105–S121. [Google Scholar] [CrossRef]
- LoMauro, A.; Aliverti, A. Blood Shift During Cough: Negligible or Significant? Front. Physiol. 2018, 9, 501. [Google Scholar] [CrossRef]
| Factor | Murphy and Atala, 2014 [14] | Current Literature | Refs. |
|---|---|---|---|
| 3D modelling and design |
|
| [30,33,35] |
|
| [22,36,37,38,39,40] | |
|
| [6,22,24,30,33,41] | |
| Biomaterials |
|
| [34,35,36,39,42,43,44] |
|
| [14,15,34,36,44,45,46] | |
| Cell sources |
|
| [4,15,22,36,40,41,42,47] |
|
| [40,41,42,47] | |
| 3D bioprinting methods |
|
| [15,32,34,35,36,39,43,44,45,48,49,50] |
|
| [32,34,36,44,51] | |
| Challenges and future research |
|
| [15,30,34,38,39,44,51] |
|
| [6,15,22,32,34,36,38,39,41,50,52] | |
|
| [6,15,22,30,32,34,35,36,39,41,42,44,48,50,52] | |
|
| [6,16,30,33,34,35,36,39,40,42,45,47,51,52,53] | |
|
| [16,22,34,39,41,44,45,50,51] | |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matthews, N.; Pandolfo, B.; Moses, D.; Gentile, C. Taking It Personally: 3D Bioprinting a Patient-Specific Cardiac Patch for the Treatment of Heart Failure. Bioengineering 2022, 9, 93. https://doi.org/10.3390/bioengineering9030093
Matthews N, Pandolfo B, Moses D, Gentile C. Taking It Personally: 3D Bioprinting a Patient-Specific Cardiac Patch for the Treatment of Heart Failure. Bioengineering. 2022; 9(3):93. https://doi.org/10.3390/bioengineering9030093
Chicago/Turabian StyleMatthews, Niina, Berto Pandolfo, Daniel Moses, and Carmine Gentile. 2022. "Taking It Personally: 3D Bioprinting a Patient-Specific Cardiac Patch for the Treatment of Heart Failure" Bioengineering 9, no. 3: 93. https://doi.org/10.3390/bioengineering9030093
APA StyleMatthews, N., Pandolfo, B., Moses, D., & Gentile, C. (2022). Taking It Personally: 3D Bioprinting a Patient-Specific Cardiac Patch for the Treatment of Heart Failure. Bioengineering, 9(3), 93. https://doi.org/10.3390/bioengineering9030093







