Prosthesis–Patient Mismatch and Aortic Root Enlargement: Indications, Techniques and Outcomes
Abstract
:1. Introduction and Definition of Prosthesis–Patient Mismatch
2. Measuring and Predicting PPM
- Method 1—Using in house TTE data obtained from patients 6 months postoperatively to create “home-grown” iEOA charts (r = 0.62);
- Method 2—Using the geometric orifice area based on static parameters (the valve internal diameter specified by the manufacturer) (r = 0.27);
- Method 3—Using commercial iEOA charts, which are produced by using data obtained through various methods but can include in vitro measurements (varies based on manufacturer; r = 0.27–0.59 for four different valve types);
- Method 4—Using published EOA data from the literature (r = 0.53).
3. Clinical Impact of PPM
4. Management of PPM by Aortic Root Enlargement
- Proceed with the selected prosthesis if the predicted iEOA is >0.85.
- If the predicted iEOA is ≤0.85 then:
- -
- Accept PPM in certain clinical contexts;
- -
- Choose a prosthetic with larger EOA;
- -
- Carry out an aortic root enlargement.
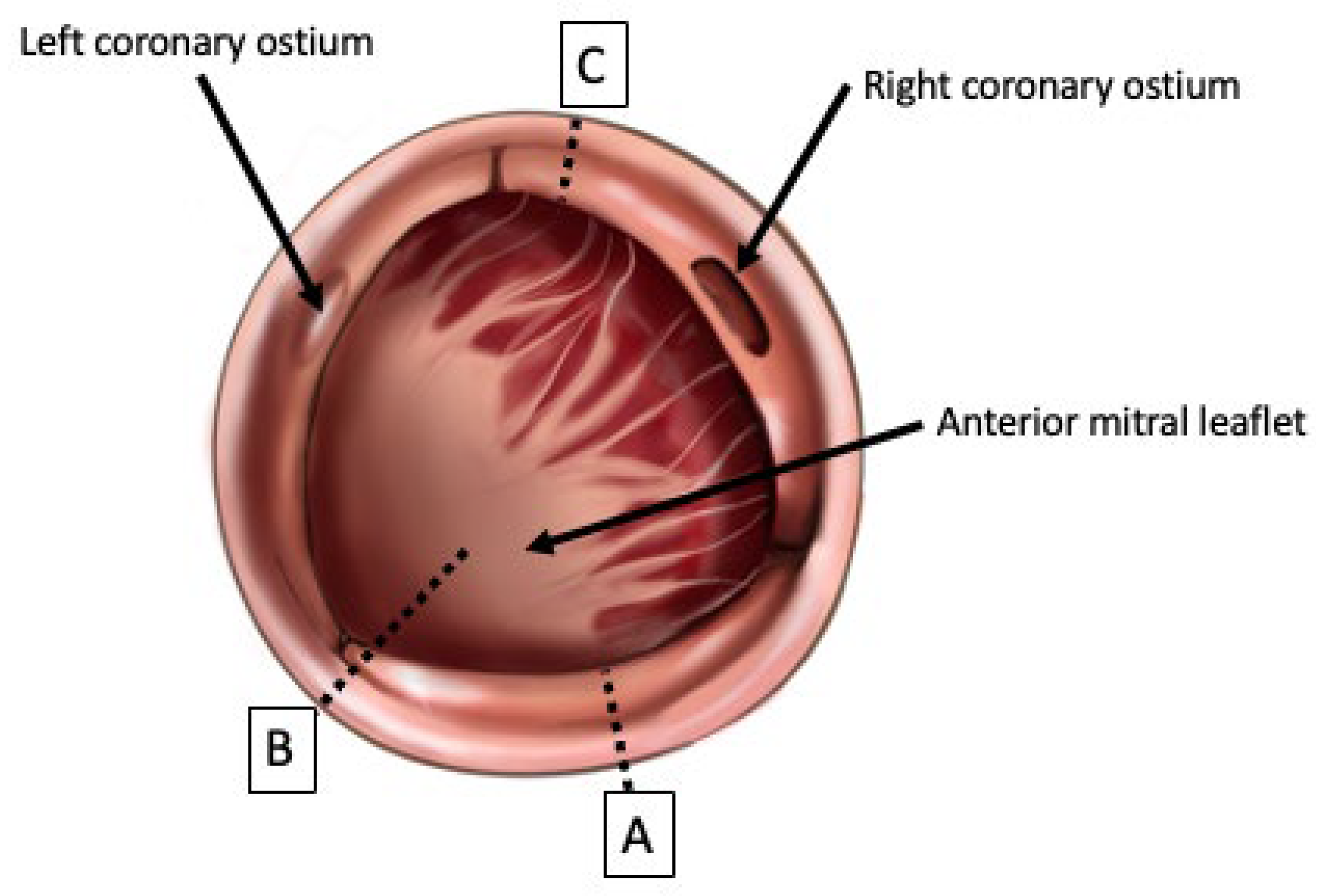
5. Established Techniques of Aortic Root Enlargement
5.1. Techniques of Aortic Root Enlargement
5.2. Outcomes of Aortic Root Enlargement Procedures
6. Contemporary Developments in ARE: The Yang Procedure
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rahimtoola, S.H. The Problem of Valve Prosthesis–patient Mismatch. Circulation 1978, 58, 20–24. [Google Scholar] [CrossRef]
- Pibarot, P.; Dumesnil, J.G. Prosthesis–patient Mismatch: Definition, Clinical Impact, and Prevention. Heart 2006, 92, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- VARC-3 WRITING COMMITTEE; Généreux, P.; Piazza, N.; Alu, M.C.; Nazif, T.; Hahn, R.T.; Pibarot, P.; Bax, J.J.; Leipsic, J.A.; Blanke, P.; et al. Valve Academic Research Consortium 3: Updated Endpoint Definitions for Aortic Valve Clinical Research. Eur. Heart J. 2021, 42, 1825–1857. [Google Scholar] [CrossRef]
- Mack, M.J.; Adams, D.H. Avoidance of Patient Prosthesis Mismatch after Aortic Valve Replacement. J. Am. Coll. Cardiol. 2023, 81, 976–978. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Sá, M.P.B.; de Carvalho, M.M.B.; Sobral Filho, D.C.; Cavalcanti, L.R.P.; Rayol, S.D.C.; Diniz, R.G.S.; Menezes, A.M.; Clavel, M.-A.; Pibarot, P.; Lima, R.C. Surgical Aortic Valve Replacement and Patient-Prosthesis Mismatch: A Meta-Analysis of 108182 Patients. Eur. J. Cardiothorac. Surg. 2019, 56, 44–54. [Google Scholar] [CrossRef]
- Elmahdy, W.; Osman, M.; Farag, M.; Shoaib, A.; Saad, H.; Sullivan, K.; Krishnan, U.; Nashef, S.; Berman, M. Prosthesis–patient Mismatch Increases Early and Late Mortality in Low Risk Aortic Valve Replacement. Semin. Thorac. Cardiovasc. Surg. 2021, 33, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Bleiziffer, S.; Eichinger, W.B.; Hettich, I.; Guenzinger, R.; Ruzicka, D.; Bauernschmitt, R.; Lange, R. Prediction of Valve Prosthesis–patient Mismatch Prior to Aortic Valve Replacement: Which Is the Best Method? Heart 2007, 93, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Ternacle, J.; Guimaraes, L.; Vincent, F.; Côté, N.; Côté, M.; Lachance, D.; Clavel, M.-A.; Abbas, A.E.; Pibarot, P.; Rodés-Cabau, J. Reclassification of Prosthesis–Patient Mismatch after Transcatheter Aortic Valve Replacement Using Predicted vs. Measured Indexed Effective Orifice Area. Eur. Heart J.—Cardiovasc. Imaging 2021, 22, 11–20. [Google Scholar] [CrossRef]
- Ternacle, J.; Pibarot, P.; Herrmann, H.C.; Kodali, S.; Leipsic, J.; Blanke, P.; Jaber, W.; Mack, M.J.; Clavel, M.-A.; Salaun, E.; et al. Prosthesis–patient Mismatch after Aortic Valve Replacement in the PARTNER 2 Trial and Registry. JACC Cardiovasc. Interv. 2021, 14, 1466–1477. [Google Scholar] [CrossRef]
- Vriesendorp, M.D.; De Lind Van Wijngaarden, R.A.F.; Head, S.J.; Kappetein, A.-P.; Hickey, G.L.; Rao, V.; Weissman, N.J.; Reardon, M.J.; Moront, M.G.; Sabik, J.F., III; et al. The Fallacy of Indexed Effective Orifice Area Charts to Predict Prosthesis–Patient Mismatch after Prosthesis Implantation. Eur. Heart J.—Cardiovasc. Imaging 2020, 21, 1116–1122. [Google Scholar] [CrossRef]
- Flameng, W.; Herregods, M.-C.; Vercalsteren, M.; Herijgers, P.; Bogaerts, K.; Meuris, B. Prosthesis–patient Mismatch Predicts Structural Valve Degeneration in Bioprosthetic Heart Valves. Circulation 2010, 121, 2123–2129. [Google Scholar] [CrossRef]
- Zorn, G.L.; Little, S.H.; Tadros, P.; Deeb, G.M.; Gleason, T.G.; Heiser, J.; Kleiman, N.S.; Oh, J.K.; Popma, J.J.; Adams, D.; et al. Prosthesis–patient Mismatch in High-Risk Patients with Severe Aortic Stenosis: A Randomized Trial of a Self-Expanding Prosthesis. J. Thorac. Cardiovasc. Surg. 2016, 151, 1014–1022, 1023.e1–e3. [Google Scholar] [CrossRef] [PubMed]
- Dismorr, M.; Glaser, N.; Franco-Cereceda, A.; Sartipy, U. Effect of Prosthesis–patient Mismatch on Long-Term Clinical Outcomes after Bioprosthetic Aortic Valve Replacement. J. Am. Coll. Cardiol. 2023, 81, 964–975. [Google Scholar] [CrossRef]
- Dayan, V.; Vignolo, G.; Soca, G.; Paganini, J.J.; Brusich, D.; Pibarot, P. Predictors and Outcomes of Prosthesis–patient Mismatch after Aortic Valve Replacement. JACC Cardiovasc. Imaging 2016, 9, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Lin, Y.; Kang, B.; Wang, Z. Indexed Effective Orifice Area Is a Significant Predictor of Higher Mid- and Long-Term Mortality Rates Following Aortic Valve Replacement in Patients with Prosthesis–patient Mismatch. Eur. J. Cardio-Thorac. Surg. 2014, 45, 234–240. [Google Scholar] [CrossRef]
- Takagi, H.; Yamamoto, H.; Iwata, K.; Goto, S.; Umemoto, T. A Meta-Analysis of Effects of Prosthesis–Patient Mismatch after Aortic Valve Replacement on Late Mortality. Int. J. Cardiol. 2012, 159, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Head, S.J.; Mokhles, M.M.; Osnabrugge, R.L.J.; Pibarot, P.; Mack, M.J.; Takkenberg, J.J.M.; Bogers, A.J.J.C.; Kappetein, A.P. The Impact of Prosthesis–patient Mismatch on Long-Term Survival after Aortic Valve Replacement: A Systematic Review and Meta-Analysis of 34 Observational Studies Comprising 27186 Patients with 133141 Patient-Years. Eur. Heart J. 2012, 33, 1518–1529. [Google Scholar] [CrossRef] [PubMed]
- Blais, C.; Dumesnil, J.G.; Baillot, R.; Simard, S.; Doyle, D.; Pibarot, P. Impact of Valve Prosthesis–patient Mismatch on Short-Term Mortality after Aortic Valve Replacement. Circulation 2003, 108, 983–988. [Google Scholar] [CrossRef]
- Vahanian, A.; Beyersdorf, F.; Praz, F.; Milojevic, M.; Baldus, S.; Bauersachs, J.; Capodanno, D.; Conradi, L.; De Bonis, M.; De Paulis, R.; et al. 2021 ESC/EACTS Guidelines for the Management of Valvular Heart Disease: Developed by the Task Force for the Management of Valvular Heart Disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur. Heart J. 2022, 43, 561–632. [Google Scholar] [CrossRef]
- Bilkhu, R.; Jahangiri, M.; Otto, C.M. Patient-Prosthesis Mismatch Following Aortic Valve Replacement. Heart 2019, 105, s28–s33. [Google Scholar] [CrossRef]
- Belluschi, I.; Moriggia, S.; Giacomini, A.; Del Forno, B.; Di Sanzo, S.; Blasio, A.; Scafuri, A.; Alfieri, O. Can Perceval Sutureless Valve Reduce the Rate of Patient-Prosthesis Mismatch?†. Eur. J. Cardio-Thorac. Surg. 2017, 51, 1093–1099. [Google Scholar] [CrossRef] [PubMed]
- Fiore, A.; Gueldich, M.; Folliguet, T. Sutureless Valves Fit/Perform Well in a Small Aortic Annulus. Ann. Cardiothorac. Surg. 2020, 9, 414–416. [Google Scholar] [CrossRef]
- Leone, P.P.; Regazzoli, D.; Pagnesi, M.; Sanz-Sanchez, J.; Chiarito, M.; Cannata, F.; Van Mieghem, N.M.; Barbanti, M.; Tamburino, C.; Teles, R.; et al. Predictors and Clinical Impact of Prosthesis–patient Mismatch after Self-Expandable TAVR in Small Annuli. JACC Cardiovasc. Interv. 2021, 14, 1218–1228. [Google Scholar] [CrossRef] [PubMed]
- Weltert, L.P.; Lucertini, G. Simplification of the Manougian Approach to Treat Small Annuli: Finally a Reproducible Low Risk Approach on the Horizon? J. Card. Surg. 2022, 37, 579–580. [Google Scholar] [CrossRef] [PubMed]
- Leone, P.P.; Fazzari, F.; Cannata, F.; Sanz-Sanchez, J.; Mangieri, A.; Monti, L.; Cozzi, O.; Stefanini, G.G.; Bragato, R.; Colombo, A.; et al. Clinical and Technical Challenges of Prosthesis–Patient Mismatch after Transcatheter Aortic Valve Implantation. Front. Cardiovasc. Med. 2021, 8, 670457. [Google Scholar] [CrossRef] [PubMed]
- Takagi, H.; Umemoto, T. Prosthesis–Patient Mismatch after Transcatheter Aortic Valve Implantation. Ann. Thorac. Surg. 2016, 101, 872–880. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, T.; Makar, M.; Ahmad, Y. Patient-Prosthesis Mismatch after SAVR and TAVR: The Importance of Comparing Apples with Apples. JACC Cardiovasc. Interv. 2021, 14, 1478–1480. [Google Scholar] [CrossRef]
- Pibarot, P.; Dumesnil, J.G. Prosthetic Heart Valves: Selection of the Optimal Prosthesis and Long-Term Management. Circulation 2009, 119, 1034–1048. [Google Scholar] [CrossRef]
- Grubb, K.J. Aortic Root Enlargement during Aortic Valve Replacement: Nicks and Manouguian Techniques. Oper. Tech. Thorac. Cardiovasc. Surg. 2015, 20, 206–218. [Google Scholar] [CrossRef]
- Dhareshwar, J.; Sundt, T.M.; Dearani, J.A.; Schaff, H.V.; Cook, D.J.; Orszulak, T.A. Aortic Root Enlargement: What Are the Operative Risks? J. Thorac. Cardiovasc. Surg. 2007, 134, 916–924. [Google Scholar] [CrossRef]
- Massias, S.A.; Pittams, A.; Mohamed, M.; Ahmed, S.; Younas, H.; Harky, A. Aortic Root Enlargement: When and How. J. Card. Surg. 2021, 36, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Tam, D.Y.; Rocha, R.V.; Wijeysundera, H.C.; Austin, P.C.; Dvir, D.; Fremes, S.E. Surgical Valve Selection in the Era of Transcatheter Aortic Valve Replacement in the Society of Thoracic Surgeons Database. J. Thorac. Cardiovasc. Surg. 2020, 159, 416–427.e8. [Google Scholar] [CrossRef]
- Sá, M.P.; den Eynde, J.V.; Amabile, A.; Malin, J.H.; Jacquemyn, X.; Tasoudis, P.; Sicouri, S.; Schena, S.; Torregrossa, G.; Ramlawi, B. Late Outcomes after Aortic Root Enlargement during Aortic Valve Replacement: Meta-Analysis with Reconstructed Time-to-Event Data. J. Cardiothorac. Vasc. Anesth. 2022, 36, 3065–3073. [Google Scholar] [CrossRef] [PubMed]
- Sá, M.P.B.O.; Carvalho, M.M.B.; Sobral Filho, D.C.; Cavalcanti, L.R.P.; Diniz, R.G.S.; Rayol, S.C.; Soares, A.M.M.N.; Sá, F.B.C.D.A.; Menezes, A.M.; Clavel, M.-A.; et al. Impact of Surgical Aortic Root Enlargement on the Outcomes of Aortic Valve Replacement: A Meta-Analysis of 13 174 Patients. Interact. CardioVascular Thorac. Surg. 2019, 29, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Sá, M.P.B.O.; Zhigalov, K.; Cavalcanti, L.R.P.; Escorel Neto, A.C.; Rayol, S.C.; Weymann, A.; Ruhparwar, A.; Lima, R.C. Impact of Aortic Annulus Enlargement on the Outcomes of Aortic Valve Replacement: A Meta-Analysis. Semin. Thorac. Cardiovasc. Surg. 2021, 33, 316–325. [Google Scholar] [CrossRef] [PubMed]
- Nicks, R.; Cartmill, T.; Bernstein, L. Hypoplasia of the Aortic Root. The Problem of Aortic Valve Replacement. Thorax 1970, 25, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Manouguian, S.; Seybold-Epting, W. Patch Enlargement of the Aortic Valve Ring by Extending the Aortic Incision into the Anterior Mitral Leaflet: New Operative Technique. J. Thorac. Cardiovasc. Surg. 1979, 78, 402–412. [Google Scholar] [CrossRef]
- Roeser, M.E. The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement. Oper. Tech. Thorac. Cardiovasc. Surg. 2015, 20, 219–233. [Google Scholar] [CrossRef]
- Imanaka, K.; Takamoto, S.; Furuse, A. Mitral Regurgitation Late after Manouguian’s Anulus Enlargement and Aortic Valve Replacement. J. Thorac. Cardiovasc. Surg. 1998, 115, 727–729. [Google Scholar] [CrossRef]
- Konno, S.; Imai, Y.; Iida, Y.; Nakajima, M.; Tatsuno, K. A New Method for Prosthetic Valve Replacement in Congenital Aortic Stenosis Associated with Hypoplasia of the Aortic Valve Ring. J. Thorac. Cardiovasc. Surg. 1975, 70, 909–917. [Google Scholar] [CrossRef]
- Losenno, K.L.; Gelinas, J.J.; Johnson, M.; Chu, M.W.A. Defining the Efficacy of Aortic Root Enlargement Procedures: A Comparative Analysis of Surgical Techniques. Can. J. Cardiol. 2013, 29, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Borracci, R.; Rubio, M.; Camargo, R.L.P.; Baldi, J.; Archer, M.; Ingino, C. Aortic Root Enlargement of a Small Annulus Using the Nicks Technique during Aortic Valve Replacement. Rev. Argent. Cardiol. 2014, 82, 504–507. [Google Scholar]
- Yu, W.; Tam, D.Y.; Rocha, R.V.; Makhdoum, A.; Ouzounian, M.; Fremes, S.E. Aortic Root Enlargement Is Safe and Reduces the Incidence of Patient-Prosthesis Mismatch: A Meta-Analysis of Early and Late Outcomes. Can. J. Cardiol. 2019, 35, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Haunschild, J.; Scharnowski, S.; Mende, M.; von Aspern, K.; Misfeld, M.; Mohr, F.-W.; Borger, M.A.; Etz, C.D. Aortic Root Enlargement to Mitigate Patient–Prosthesis Mismatch: Do Early Adverse Events Justify Reluctance?†. Eur. J. Cardio-Thorac. Surg. 2019, 56, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Penaranda, J.G.; Greason, K.L.; Pislaru, S.V.; Schaff, H.V.; Daly, R.C.; Park, S.J.; Suri, R.M.; Burkhart, H.M.; Joyce, L.D.; Stulak, J.M.; et al. Aortic Root Enlargement in Octogenarian Patients Results in Less Patient Prosthesis Mismatch. Ann. Thorac. Surg. 2014, 97, 1533–1538. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, R.B.; Beller, J.P.; Mehaffey, J.H.; Charles, E.J.; Quader, M.A.; Rich, J.B.; Kiser, A.C.; Joseph, M.; Speir, A.M.; Kern, J.A.; et al. Incremental Risk of Annular Enlargement: A Multi-Institutional Cohort Study. Ann. Thorac. Surg. 2019, 108, 1752–1759. [Google Scholar] [CrossRef] [PubMed]
- Mehaffey, J.H.; Hawkins, R.B.; Wegermann, Z.K.; Grau-Sepulveda, M.V.; Fallon, J.M.; Brennan, J.M.; Thourani, V.H.; Badhwar, V.; Ailawadi, G. Aortic Annular Enlargement in the Elderly: Short and Long-Term Outcomes in the United States. Ann. Thorac. Surg. 2021, 112, 1160–1166. [Google Scholar] [CrossRef]
- Yang, B. A Novel Simple Technique to Enlarge the Aortic Annulus by Two Valve Sizes. JTCVS Tech. 2021, 5, 13–16. [Google Scholar] [CrossRef]
- Nyamande, D.; Ramoroko, P.S. The Yang Procedure: Renaming the Recently Described “Y” Incision/Rectangular Patch Aortic Annulus Enlargement Technique. JTCVS Tech. 2023, 18, 43. [Google Scholar] [CrossRef]
- Yang, B.; Burris, N.S.; Prasch, P. Reply from Authors: Not Lemon on a Stick, but Crown (Valve) on a Head (Left Ventricular Outflow Tract). JTCVS Tech. 2022, 16, 22–24. [Google Scholar] [CrossRef]
- Yang, B.; Naeem, A.; Palmer, S. “Roof” Technique—A Modified Aortotomy Closure in Y-Incision Aortic Root Enlargement Upsizing 3–4 Valve Sizes. JTCVS Tech. 2022, 12, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Price, J. Commentary: Enlargement of the Aortic Annulus: Always a Technical Price to Pay. JTCVS Tech. 2021, 5, 17–18. [Google Scholar] [CrossRef] [PubMed]
- Norton, E.L.; Grubb, K.J. Commentary: Aortic Root Enlargement—Optimizing for Today and Preparing for the Future. JTCVS Tech. 2022, 12, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Said, S.M.; Jahanyar, J. Yang Technique for Aortic Annular Enlargement and the “Lemon on a Stick”. JTCVS Tech. 2022, 16, 21. [Google Scholar] [CrossRef] [PubMed]
- Rubens, F.D. Commentary: Expertise in Fitting Square Pegs in Round Holes. JTCVS Tech. 2021, 5, 19. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Naeem, A. A Y Incision and Rectangular Patch to Enlarge the Aortic Annulus by Three Valve Sizes. Ann. Thorac. Surg. 2021, 112, e139–e141. [Google Scholar] [CrossRef] [PubMed]
- Naeem, A.; Yang, B. A “Y” Incision/Rectangular Patch to Enlarge the Aortic Annulus by 4 Valve Sizes in TAV and BAV Patients. Available online: https://ctsnet.figshare.com/articles/media/Untitled_ItemA_Y_Incision_Rectangular_Patch_to_Enlarge_the_Aortic_Annulus_by_4_Valve_Sizes_in_TAV_and_BAV_Patients/14408741/2 (accessed on 3 June 2023).
- Yang, B.; Ghita, C.; Palmer, S. Y-Incision Aortic Root Enlargement with Modified Aortotomy Upsizing the Annulus by 5 Valve Sizes. Ann. Thorac. Surg. 2022, 114, e479–e481. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Ghita, C.; Makkinejad, A.; Green, C.; Wu, X. Early Outcomes of the Y-Incision Technique to Enlarge the Aortic Annulus 3 to 4 Valve Sizes. J. Thorac. Cardiovasc. Surg. 2022. [Google Scholar] [CrossRef]

| Definition | iEOA | BMI Adjustment (>30 kg/m2) |
|---|---|---|
| Moderate PPM | ≤0.85 cm2/m2 | <0.70 cm2/m2 |
| Severe PPM | ≤0.65 cm2/m2 | <0.55 cm2/m2 |
| Valve Size (mm) | EOA (cm2) | Indexed EOA (cm2/m2) | |||
|---|---|---|---|---|---|
| 21 | 1.20 | 0.80 | 0.69 | 0.60 | 0.53 |
| 23 | 1.40 | 0.93 | 0.80 | 0.70 | 0.62 |
| 25 | 1.60 | 1.07 | 0.91 | 0.80 | 0.71 |
| 27 | 1.80 | 1.20 | 1.03 | 0.90 | 0.80 |
| 29 | 2.00 | 1.33 | 1.14 | 1.00 | 0.89 |
| 1.50 | 1.75 | 2.00 | 2.25 | ||
| Patient BSA (m2) | |||||
| Valve Size (mm) | EOA (cm2) | Indexed EOA (cm2/m2) | |||
|---|---|---|---|---|---|
| 19 | 1.58 | 1.05 | 0.90 | 0.79 | 0.70 |
| 21 | 1.90 | 1.27 | 1.09 | 0.95 | 0.84 |
| 23 | 2.07 | 1.38 | 1.18 | 1.04 | 0.92 |
| 25 | 2.33 | 1.55 | 1.33 | 1.17 | 1.04 |
| 27 | 2.38 | 1.59 | 1.36 | 1.19 | 1.06 |
| 29 | 2.84 | 1.89 | 1.62 | 1.42 | 1.26 |
| 1.50 | 1.75 | 2.00 | 2.25 | ||
| Patient BSA (m2) | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fazmin, I.T.; Ali, J.M. Prosthesis–Patient Mismatch and Aortic Root Enlargement: Indications, Techniques and Outcomes. J. Cardiovasc. Dev. Dis. 2023, 10, 373. https://doi.org/10.3390/jcdd10090373
Fazmin IT, Ali JM. Prosthesis–Patient Mismatch and Aortic Root Enlargement: Indications, Techniques and Outcomes. Journal of Cardiovascular Development and Disease. 2023; 10(9):373. https://doi.org/10.3390/jcdd10090373
Chicago/Turabian StyleFazmin, Ibrahim Talal, and Jason M. Ali. 2023. "Prosthesis–Patient Mismatch and Aortic Root Enlargement: Indications, Techniques and Outcomes" Journal of Cardiovascular Development and Disease 10, no. 9: 373. https://doi.org/10.3390/jcdd10090373
APA StyleFazmin, I. T., & Ali, J. M. (2023). Prosthesis–Patient Mismatch and Aortic Root Enlargement: Indications, Techniques and Outcomes. Journal of Cardiovascular Development and Disease, 10(9), 373. https://doi.org/10.3390/jcdd10090373






