Abstract
Background: Left ventricular ejection fraction (LVEF) is a suboptimal indicator of risk stratification for patients with an implantable cardioverter defibrillator (ICD). Studies have shown that left ventricular end-diastolic diameter (LVEDD) was associated with all-cause mortality and ventricular arrhythmias. We examined the quantified prognostic value of LVEF and LVEDD for clinical outcomes, respectively. Method: This study retrospectively enrolled patients with ICD implantation in a single center. The associations between LVEF or LVEDD and all-cause mortality and appropriate shocks were analyzed using Cox regression and Fine-gray competing risk regression, respectively. Result: During a median follow up of 59.6 months, 168/630 (26.7%) patients died. LVEF and LVEDD were strongly associated with all-cause mortality (LVEF per 10%: HR 0.77, 95%CI 0.64–0.93, p = 0.006; LVEDD per 10 mm: HR 1.54, 95%CI 1.27–1.85, p < 0.001). After a median interrogation time of 37.1 months, 156 (24.8%) patients received at least one shock. LVEF was not associated with appropriate shock, whereas larger LVEDD (per 10 mm) was significantly associated with a higher risk of shock (HR: 1.27, 95%CI 1.06–1.52, p = 0.008). The addition of LVEF or LVEDD to clinical factors provided incremental prognostic value and discrimination improvement for all-cause mortality, while only the addition of LVEDD to clinical factors improved prognostic value for shock intervention. Conclusions: Baseline LVEF and LVEDD show a linear relationship with all-cause mortality in patients with ICD. However, whereas LVEF is not associated with shock, a linear relationship exists between LVEDD and appropriate shock. LVEDD adds more predictive value in relation to all-cause mortality and appropriate shocks than LVEF.
1. Introduction
Sudden cardiac death (SCD) accounts for approximately 25% of cardiovascular death, and implantable cardioverter defibrillator (ICD) effectively reduces mortality related to sudden death. Current ventricular arrhythmias (VA) and SCD guidelines recommend primary ICD implantation in symptomatic (New York Heart Association (NYHA) function class II-III) ischemic cardiomyopathy (ICM)/non-ischemic cardiomyopathy (NICM) patients and left ventricular ejection fraction (LVEF) ≤35% after ≥3 months of optimal medical treatment [1,2]. LVEF is the core indicator of risk stratification for patients with a risk of SCD, and the cut-off of 35% is derived from the pivotal clinical trials conducted approximately 20 years ago [3,4,5].
Other echocardiographic characteristics such as LV mass, left atrial dimension and left ventricular end-systolic dimension have been shown to be associated with clinical outcomes [6]. Among these, baseline left ventricular end-diastolic diameter (LVEDD) has been associated with all-cause mortality and SCD in the general population and heart failure patients [7,8,9,10,11]. However, the impact of LVEF and LVEDD on all-cause mortality and SCD has not been thoroughly investigated in patients with ICD.
Therefore, the aim of this study is to quantify the prognostic value of LVEF and LVEDD for clinical outcomes, respectively. We also investigated the incremental value of adding LVEF and LVEDD to the established clinical risk predictors.
2. Methods
2.1. Population
We retrospectively included patients from a maintained ICD database at Fuwai Hospital from March 2010 to September 2020. Eligible patients were age older than 18, had complete echocardiography examination and had a diagnosis of ICM or NICM. For the indication of ICD, primary prevention was defined as in symptomatic ICM/NICM patients with NYHA function class II-III and LVEF ≤ 35% following optimal medical treatment for at least 3 months. The secondary prevention was defined as prophylactic ICD implantation to prevent sudden cardiac death in patients with a history of life-threatening ventricular arrhythmia [1,2]. ICM was defined as significant stenosis (≥50% of left main or ≥70% of major epicardial coronary artery) on coronary angiography or with evidence of myocardial infarction. We excluded patients who were lost of follow-up, without current implantation indication of ICD [1], or who had undergone device replacement. This study was conducted in accordance with the Declaration of Helsinki and approved by the institutional review board and ethics committees. All patients provided written informed consent.
2.2. Echocardiography
Electrocardiogram-triggered two-dimensional echocardiographic examination was performed before the implantation of ICD by experienced sonographers using commercially available equipment (GE Vivid) and interpreted by experienced cardiologists. LVEF was calculated using the biplane summation-of-disks method (modified Simpson’s rule) in the apical four- and two- chamber views. LVEDD was measured at a level just below the mitral valve leaflet tips in the parasternal long-axis view [12].
2.3. Follow-Up and Outcomes
ICD patients underwent routine interrogation and follow up at Fuwai hospital. The primary endpoint of this study was all-cause mortality. The secondary endpoint was the occurrence of appropriate ICD shock as a substitute for SCD. Appropriate ICD shock was defined as shocks for ventricular tachycardia (VT) or ventricular fibrillation (VF). Because this study included patients with secondary prevention, the ICD programming parameters for VT and VF varied among patients.
2.4. Statistical Analysis
Continuous variables were presented as means and standard deviation (SD) or medians and interquartile range (IQR) as appropriate. Categorical variables were presented as counts and percentages. Differences among quintiles were analyzed using one-way ANOVA or Kruskal-Wallis test for continuous variables and Pearson chi-square test or Fisher’s exact test for categorical variables. For variables missing less than 5%, they were considered to be missing at random, and random forest imputation was used. The correlation between potential predictors was performed by calculating Pearson’s correlation coefficient. When predictors demonstrated a high degree of collinearity, only one factor was considered to be included in the regression model. Multivariable Cox regressions were used to investigate the relationship of LVEF and LVEDD to all-cause mortality. Fine-Gray analyses accounting for all-cause mortality as a competing risk were used to investigate their relationships to appropriate ICD shocks. We adjusted models for important clinical predictors as well as for covariates that were significant in univariable analysis. For the Cox regression analysis, our basic model (model 1) started with several clinical predictors based on prior research and univariable Cox regression results: age, sex, BMI, systolic blood pressure, NYHA class >II and indication of ICD. Model 2 was additionally adjusted for comorbidities, including complete right bundle branch block (RBBB), atrial fibrillation (AF), diabetes, and ICM. Model 3 was additionally adjusted for medications including digoxin, diuretic and ACEI/ARB/ARNI compared with model 2. Model 4 further included eGFR, albumin and NT-proBNP. For the analysis of the secondary endpoint, model 1 adjusted for covariates mentioned above in Cox regression model 1, as well as for smoking and alcohol. The additional variables in models 2– 4 were the same as in the Cox regression model.
To evaluate the association of LVEF and LVEDD with the endpoints, we first stratified the population into quintiles according to LVEF and LVEDD, respectively. Then P for linear trend was calculated. The potential non-linearity was tested using penalized spline terms and the association between the echocardiographic parameter and the risk of the endpoint was plotted using penalized splines. Secondly, we treated the echocardiographic parameter continuously if it did not violate the linearity assumption of the Cox regression.
The performance of models was compared using C-statistic improvement. The likelihood ratio test was used to evaluate the fit of the models. Category-free net reclassification index (NRI) and integrated discrimination index (IDI) were assessed to measure the prediction improvement of LVEF and LVEDD. Possible interactions of LVEF or LVEDD with sex, the indication of ICD, ICM, and CLBBB were tested. Furthermore, we performed sensitivity analyses examining the robustness of the results using complete-case analysis. Statistical analysis was performed in R version 4.1.2 (Vienna, Austria). Multivariable Cox regressions were performed using the survival (version 3.2-13) package and competing risk models were fitted using the cmprsk (version 2.2-10) package.
3. Results
3.1. Baseline Characteristics
Among 771 patients, 647 fulfilled the inclusion criteria. Of these, 17 patients were lost to follow-up, without indication of ICD or having undergone device replacement, and 630 patients were finally included for analysis (Figure 1).
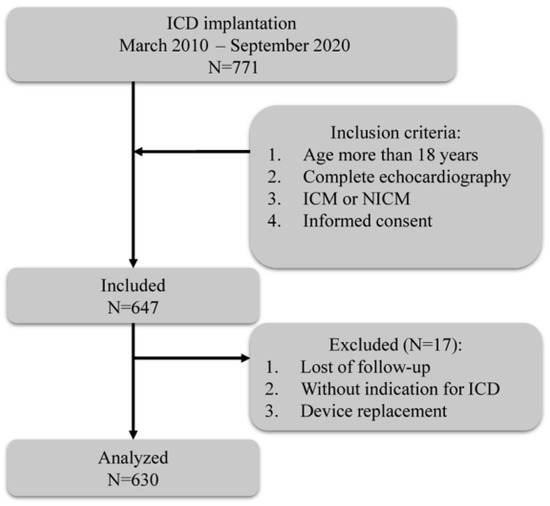
Figure 1.
Flowchart of the study. ICD, implantable cardioverter defibrillation; ICM, ischemic cardiomyopathy; NICM, non-ischemic cardiomyopathy.
The baseline characteristics of the population are summarized in Table 1. The mean age was 60.7 years, 83.7% were men, 54.4% had a diagnosis of ICM, and 23.0% were primary prevention of SCD. The mean LVEF and LVEDD was 37.3% and 64.2 mm, respectively. Patients were stratified into five groups according to LVEF: 126 patients were in Q1 (LVEF < 27%), 122 patients were in Q2 (27–33%), 125 patients were in Q3 (33–38%), 128 patients were in Q4 (38–48%) and 129 patients were in Q5 (LVEF ≥ 48%). Notable differences between the LVEF quintiles were seen in age, systolic blood pressure, NYHA functional class >II, hypertension, AF, ICM, hyperlipidemia, primary indication, use of digoxin, diuretic, amiodarone, eGFR and NT-proBNP (Table S1). LVEDD was also categorized into Q1 (<57 mm, N = 121), Q2 (57–61 mm, N = 109), Q3 (61–66 mm, N = 141), Q4 (66–72 mm, N = 131), and Q5 (≥72 mm, N = 128). The differences between LVEDD quintiles were also included in Table S1. Larger LVEDD was associated with younger age, male sex, lower systolic blood pressure, worse NYHA functional class, less comorbidities of ICM and hyperlipidemia, higher prevalence of complete left bundle branch block (CLBBB) and primary prevention of SCD. We found an excellent reverse correlation between LVEF and LVEDD in this population, with Pearson correlation coefficient = −0.62 (p < 0.001).

Table 1.
Baseline characteristics of the study population, analyzed separately according to the LVEF and LVDD quintiles.
3.2. Follow-Up
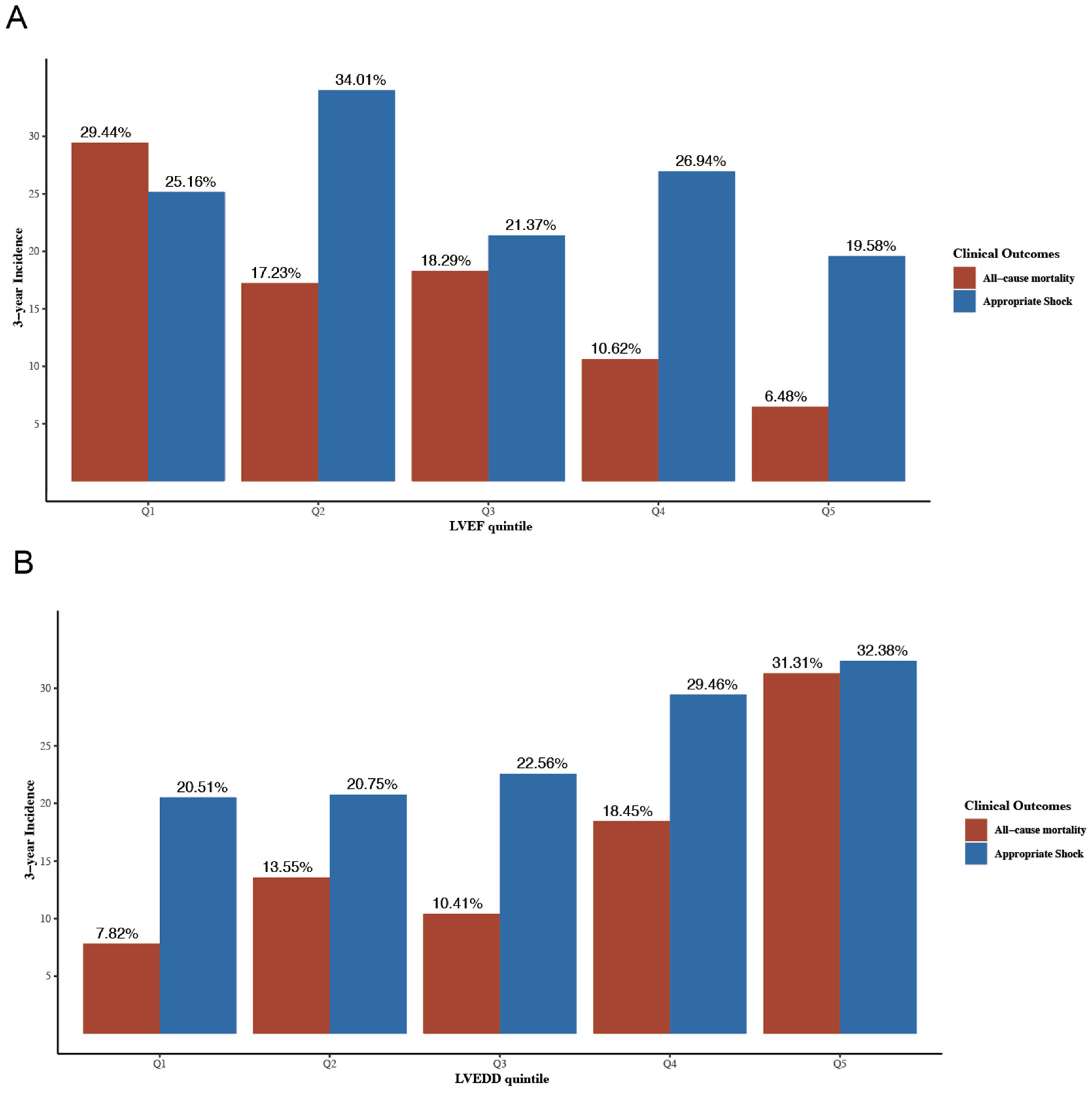
During a median follow-up time of 59.6 months (interquartile range 55.3–63.7), 168 (26.7%) patients reached the primary endpoint. A total of 156 (24.8%) patients received at least one appropriate shock during a median pacemaker interrogation time of 37.1 months (interquartile range 37.1–42.9). Results of univariable cox regression for all-cause mortality and appropriate shock are shown in Table S2. Figure 2 shows the 3-year cumulative incidences of all-cause mortality and appropriate shock according to LVEF (Figure 2A) and LVEDD (Figure 2B) quintiles. The multivariable regression results according to echo parameters quintiles and clinical outcomes in model 4 are shown in Table S3.
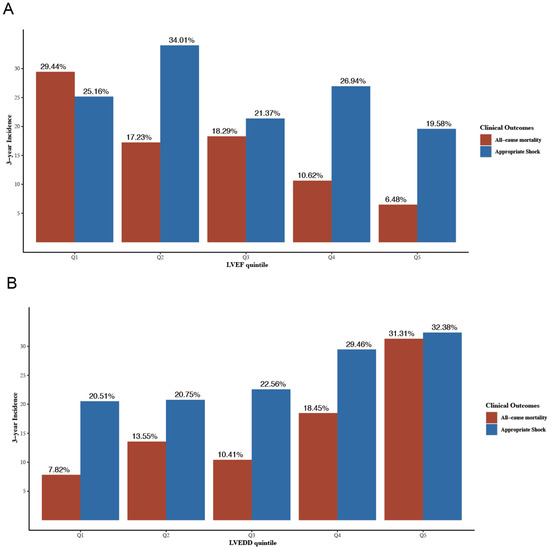
Figure 2.
3-year cumulative incidence of all-cause mortality and appropriate shock according to LVEF quintiles (A) and LVEDD quintiles (B). LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction. LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction.
3.3. Association of LVEF with Mortality
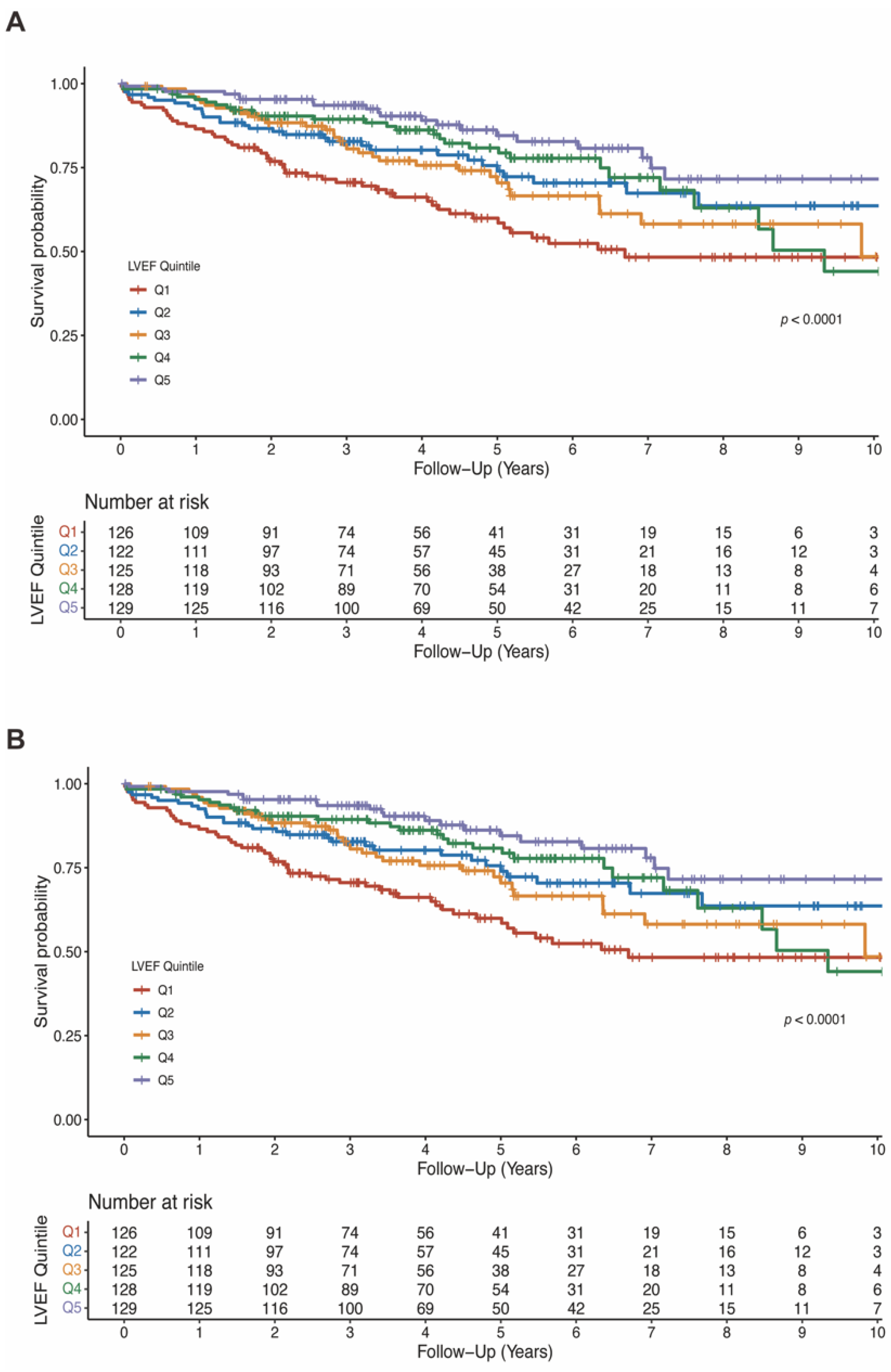
The mean LVEF in the entire population was 37.3 ± 11.7%. LVEF was significantly lower in patients who died than patients who survived (33.7 ± 10.2% vs. 38.6 ± 11.9%, p < 0.001; Table S4). As shown in Figure 3A, a lower baseline LVEF quintile was significantly associated with all-cause death (log-rank test p < 0.0001). In the model 4 adjusting for baseline characteristics, comorbidities, medication and biomarkers, higher LVEF was associated with lower risk of all-cause mortality when compared with Q1 (HR 0.64, 95%CI 0.41–1.01, p = 0.056 for Q2; HR 0.72, 95%CI 0.45–1.15, p = 0.165 for Q3; HR 0.64, 95%CI 0.38–1.07, p = 0.086 for Q4; HR 0.49, 95%CI 0.26–0.92, p = 0.027 for Q5). The summarized results of multivariable cox analyses for LVEF in all four models are shown in Table 2.
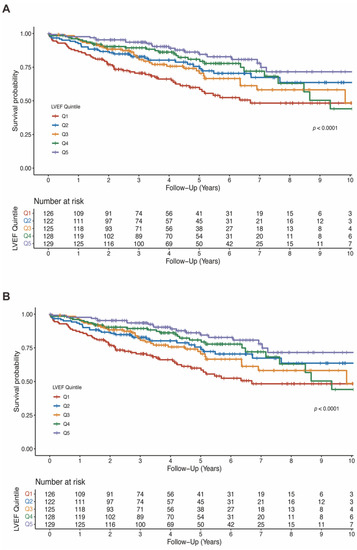
Figure 3.
Kaplan-Meier estimates of survival according to LVEF quintiles (A) and LVEDD quintiles (B). LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction.

Table 2.
Results of multivariable Cox regression for all-cause mortality by LVEF quintiles.
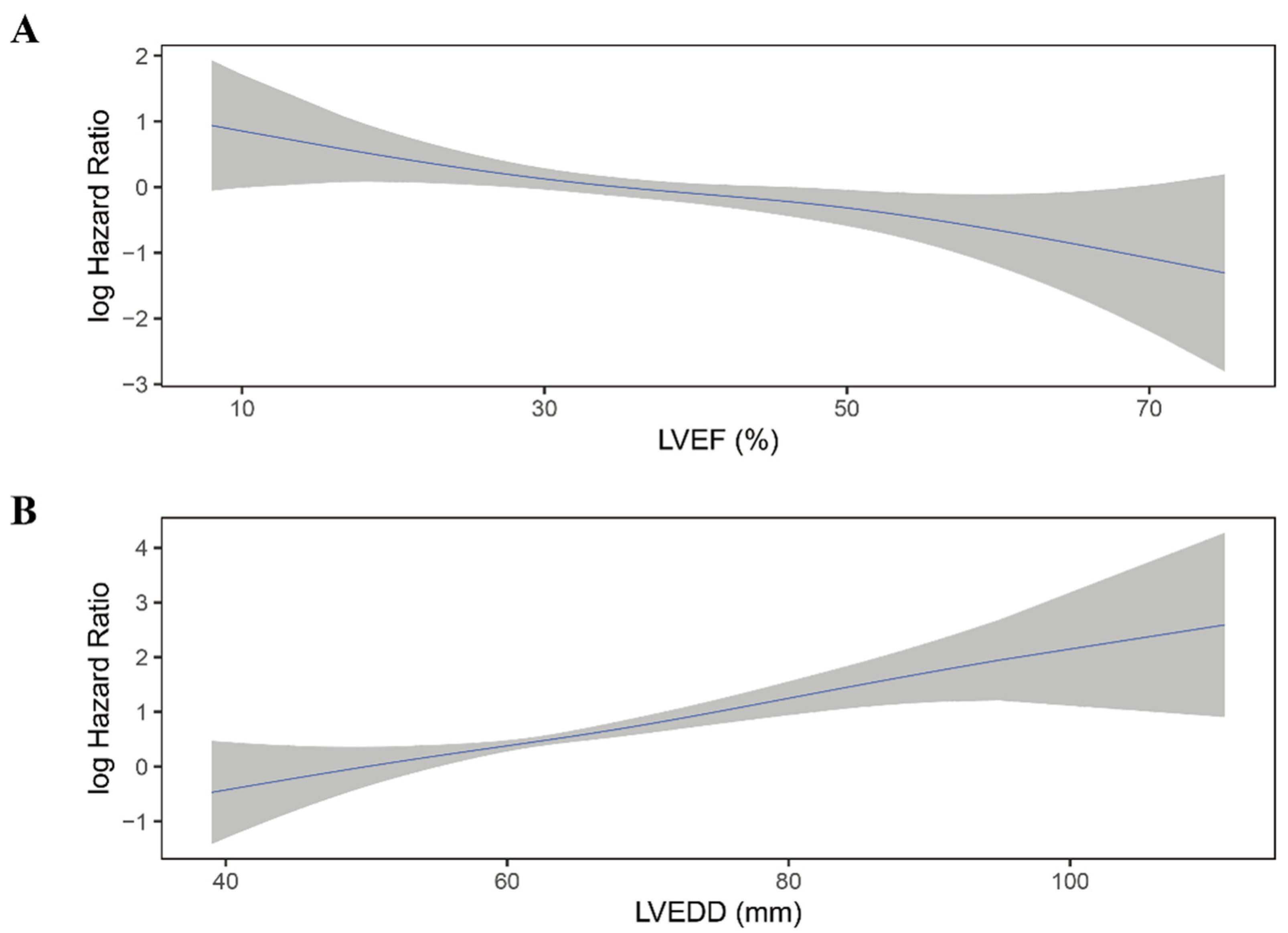
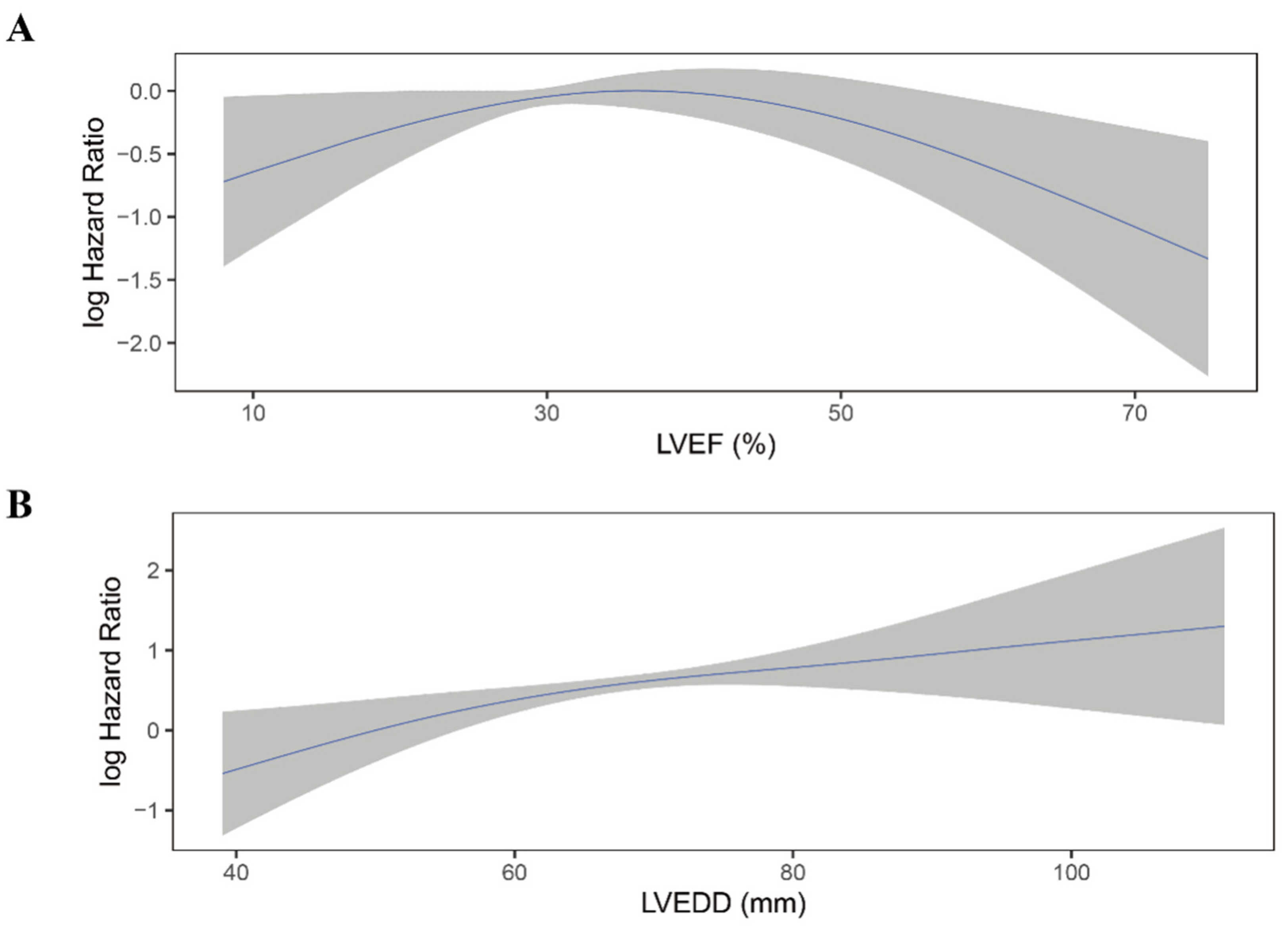
Moreover, LVEF quintiles showed a clear linear trend for all-cause mortality in all four models (P for trend < 0.05). The association between LVEF as a continuous variable and the risk of all-cause mortality was plotted (Figure 4A). As LVEF did not violate the linearity assumption of the Cox regression model (P for non-linearity > 0.05), we then included LVEF as a continuous variable in model 1 to model 4. LVEF was an independent predictor for all-cause mortality in Cox regression analysis and consistent in all models (HR for per 10% increase in LVEF: model 1: 0.76; 95% 0.64–0.89; p = 0.001; model 2: 0.76; 95%CI 0.64–0.90; p = 0.002; model 3: 0.75; 95%CI 0.62–0.89; p = 0.001; model 4: 0.77; 95%CI 0.64–0.93; p = 0.006) (Table 2).
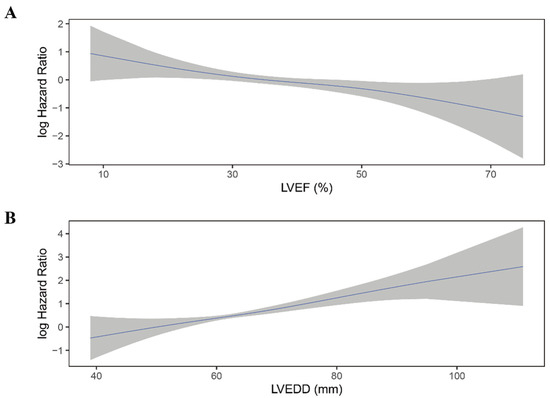
Figure 4.
Continuous log HR plot for the relationship between echocardiographic parameters and all-cause mortality ((A) LVEF; (B) LVEDD). Blue line: log HR; grey area: 95%CI. LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction.
3.4. Association of LVEDD with Mortality
The mean LVEDD was 64.2 ± 9.7 mm and was significantly larger in patients who died than survived (67.3 ± 10.0 mm vs. 63.1 ± 9.4 mm, p < 0.001; Table S4). Similarly, lower LVEDD quintiles were associated with lower risk of all-cause mortality (log-rank test p < 0.001; Figure 3B). In fully adjusted model, LVEDD was independently associated with all-cause mortality (compared to Q5: HR 0.39, 95%CI 0.22–0.70, p = 0.002 for Q1; HR 0.61, 95%CI 0.36–1.02, p = 0.058 for Q2; HR 0.41, 95%CI 0.26–0.66, p < 0.001 for Q3; HR 0.54, 95%CI 0.35–0.85, p = 0.008 for Q4; Table 3).

Table 3.
Results of multivariable Cox regression for all-cause mortality by LVEDD quintiles.
Similar to LVEF, LVEDD quintiles showed a trend toward higher all-cause mortality risk (p < 0.001) and did not violate the linearity assumption of continuous variable (Figure 4B; P for non-linearity > 0.05). When considered as a continuous variable, Larger LVEDD was significantly associated with a higher risk of all-cause mortality (HR for per 10 mm increase in LVEDD: model 1: HR 1.54; 95%CI 1.30–1.82; p < 0.001; mode2: HR 1.56; 95%CI 1.31–1.87; p < 0.001; model 3: HR 1.57; 95%CI 1.31–1.88; p < 0.001; model 4: HR 1.54; 95%CI 1.27–1.85; p < 0.001) (Table 3).
3.5. Association of LVEF and LVEDD with Appropriate Shock
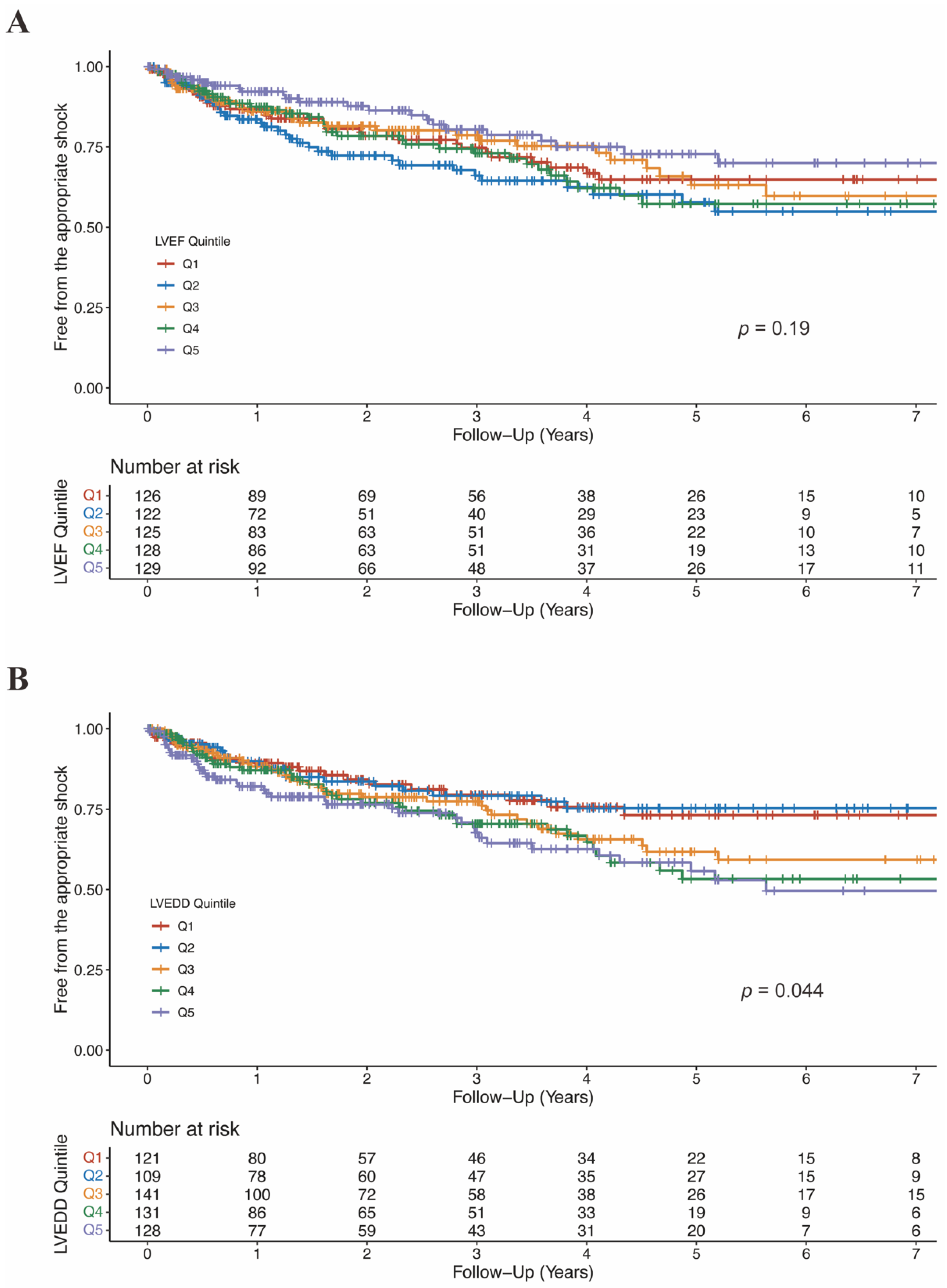
There was no significant difference in LVEF between patients who experienced shock and who not (36.1 ± 10.2% vs. 37.2 ± 12.1%, p = 0.12; Table S4). Moreover, when categorized into quintiles, LVEF was not associated with the risk of appropriate ICD shock (Figure 5A; Table 4). As shown in Figure 6A, the association between LVEF and the risk of appropriate shock violated the linearity assumption; therefore, we did not treat LVEF as a continuous variable.
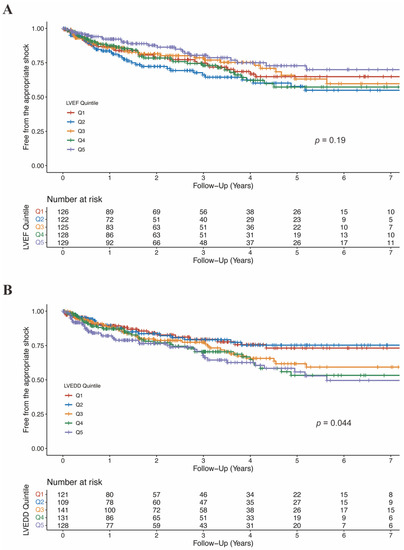
Figure 5.
Free from the appropriate shock by LVEF quintiles (A) and LVEDD quintiles (B). CI, confidence interval; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction.

Table 4.
Results of competing risk analysis for free from appropriate shock by LVEF quintiles.
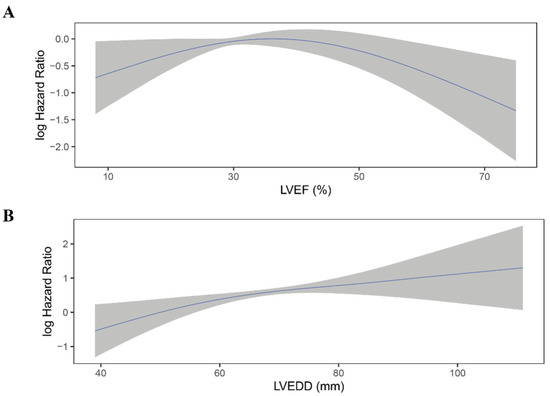
Figure 6.
Continuous log HR plot for the relationship between echocardiographic parameters and appropriate shock ((A) LVEF; (B) LVEDD). Blue line: log HR; grey area: 95%CI. CI, confidence interval; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction.
LVEDD was significantly larger in patients who experienced appropriate shock than those who did not (66.1 ± 9.2 mm vs. 63.6 ± 9.8 mm, p = 0.004; Table S4). When categorized into quintiles, there was an increasing risk of appropriate shock as LVEDD increased (Figure 5B). In the fully adjusted model (model 4), LVEDD was an independent predictor of appropriate shocks (compared to Q5: HR 0.55, 95%CI 0.31–0.99, p = 0.048 for Q1; HR 0.47, 95%CI 0.26–0.85, p = 0.013 for Q2; HR 0.83, 95%CI 0.53–1.29, p = 0.405 for Q3; HR 0.78, 95%CI 0.50–1.20, p = 0.260 for Q4; Table 5). Then LVEDD was used as a continuous variable (P for trend = 0.014, P for non-linearity = 0.446; Figure 6B). Multivariable competing risk analyses demonstrated that the HR for the occurrence of appropriate shock became higher with increasing LVEDD in the fully adjusted model (LVEDD per 10 mm: HR 1.27; 95%CI 1.06–1.52; p = 0.008; Table 5).

Table 5.
Results of competing risk analysis for free from appropriate shock by LVEDD quintiles.
3.6. Incremental Prognostic Value of LVEF and LVEDD in Multivariable Models
To compare the incremental prognostic value of LVEF and LVEDD, we first constructed the corresponding clinical models including all variates in the aforementioned models except for the echocardiographic parameter. For the primary endpoint of all-cause mortality, the addition of LVEF resulted in modest improvement in model fit (χ2 increased by 7.779; p = 0.005) and discrimination improvement (IDI 0.015, 95%CI 0.002–0.038, p = 0.007) in the fully adjusted model (model 4). The addition of LVEDD in model 4 resulted in a significant increase in χ2 by 19.574 (p < 0.0001) and IDI by 0.037 (95%CI 0.011–0.069, p < 0.0001). As for category-free reclassification analysis for all-cause mortality, the addition of LVEF and LVEDD to clinical variables resulted in a significant reclassification improvement of 12.9% (95%CI 0.7%–24.8%) and 24.3% (95%CI 9.1%–32%), respectively (Table S5).
For the secondary endpoint of appropriate shock, LVEDD rather than LVEF was an independent predictor of the shock. The addition of LVEF to a clinical model did not provide incremental prognostic value or discrimination improvement. However, the addition of LVEDD to the fully adjusted model significantly increased the model χ2 (5.814; p = 0.02) and showed a trend towards improved discrimination (IDI 0.013, 95%CI-0.006–0.044, p = 0.140). For NRI, the addition of LVEF and LVEDD resulted in nonsignificant reclassification improvement of 10.2% (95%CI-5.3%–18.5%) and 13.6% (95%CI-0.6%–20.2%) (Table S5).
3.7. Subgroup and Sensitivity Analyses
There were no significant interactions between LVEF/LVEDD and sex, the indication of ICD and cardiomyopathy etiology for the risk of mortality and appropriate shock (Table S6). Analyses of sensitivity using complete-case were included in Table S7. Effect sizes in the fully adjusted model were similar to the entire population. LVEF was predictive for all-cause mortality (HR for per 10%: 0.78, 95%CI 0.63–0.96, p = 0.020) but not appropriate shock (P for trend: 0.230), while LVEDD was predictive for both all-cause mortality (HR for per 10 mm: 1.51, 95%CI 1.23–1.86, p < 0.001) and appropriate shock (HR for per 10 mm: 1.25, 95%CI 1.04–1.51, p = 0.017).
4. Discussion
The main findings of our study including patients with ICD implantation are as follows: (1) patients with lower LVEF or larger LVEDD had a higher risk of all-cause mortality during follow-up. (2) patients with larger LVEDD at baseline had a higher risk of appropriate shock. (3) Moreover, the addition of LVEDD to clinical models provided incremental prognostic information over LVEF for both endpoints. These findings indicate that LVEDD could be a helpful predictor of all-cause mortality and appropriate shock better than LVEF in patients with ICD implantation.
According to current guidelines, LVEF is the core indicator of the selection criterion for primary prevention [2]. LVEF was a strong, independent predictor of SCD and cardiovascular mortality in pivotal trials [5,13]. For HFrEF patients, ICD was associated with 23% relative risk reduction in all-cause mortality in pivotal RCTs [14]. However, the DANISH trial in which the inclusion criteria required LVEF ≤ 35%, failed to show the survival benefit of ICD in NICM patients [15]. Prior studies have shown that LVEF is neither highly sensitive nor specific in regard to SCD prediction [16,17]. New predictors for risk prediction of clinical outcomes are urgently warranted. A suitable marker for patients should therefore predict not only all-cause mortality but also the risk of arrhythmic risk. Our study shows that LVEDD could be a valuable marker to predict both clinical outcomes. Moreover, LVEF cutoff of 35% bypassed the rigorous steps of evaluation to test whether it is sufficient to predict clinical outcomes [17]. Our results provide the first detailed, quantified relationship between echocardiographic parameters (LVEF and LVEDD) and clinical outcomes. Our results suggest a linear relationship between LVEF and LVEDD to all-cause mortality; however, for the appropriate shocks, a linear association was only identified with LVEDD.
Several observational studies demonstrated that LVEDD was associated with the risk of all- cause mortality and ventricular arrhythmias in patients with ICD [7,18,19]. Sataka et al. showed that LVEDD ≥ 65 mm was an independent predictor of fatal arrhythmic events in patients with primary prevention for class I and class IIa indication in Japan [20]. Another study including 853 patients with only Biotronik ICD demonstrated that moderate LVEF dysfunction (35–45%) and left ventricular enlargement (LVEDD ≥ 60 mm) were associated with a higher risk of VA and cardiac death [21]. Moreover, in a sub-study of the Genetic Risk of Assessment of Defibrillator Events (GRADE) study, LVEDD was associated with appropriate shocks (HR for per 10 mm: 1.33, p < 0.001) and the composite endpoint of death, transplant and left ventricular assist device (HR for per 10 mm 1.40 p < 0.001) [7]. Our study supports these results. However, LVEF was found to be both associated with shocks (HR for per 1% 0.95, p = 0.0002) and the composite endpoint (HR for per 1% 0.95, p < 0.001) in this study [7]. This is different from our results. Of note, there were several key differences between the two studies. First, this study ignored the competing risk of death. Therefore, this could overestimate the risk of appropriate shocks and might distort the results [22]. Second, the differences in patients’ characteristics might contribute to the controversial results. The inclusion criterion was LVEF < 30%, and the majority of patients (74.61%) had the primary indication in the sub-study of the GRADE study. However, in our study, the mean LVEF was 37.3 ± 11.7% and 23.1% of patients had the primary indication. Subgroup analyses in our study indicated that there was no interaction between the predictive value of echocardiographic parameters and the indication of ICD.
Larger LVEDD represents dilation of the left ventricular. Left ventricular dilation reflects the alternations in the expression of connexins and stretch-mediated changes in ion channel function and adverse left ventricular remodeling, and could create a favorable milieu for re-entrant ventricular arrhythmias [23,24]. In animal models of dyssynchronous heart failure, LV end- diastolic volume was significantly larger in the heart failure (HF) model. Moreover, remodeling of K+ current and Ca2+ current and prolongation of action potential duration (APD) were shown in the dyssynchronous HF model, which might be cellular triggers and substrate for arrhythmias [23,25]. Another mechanism underlying these might be attributable to myocardial fibrosis. Prior study has demonstrated that NICM patients with fibrosis had higher left ventricular end-diastolic volumes and the myocardial fibrosis was independently associated with all-cause mortality and SCD [26,27].
In this study, calculation of LVEF and measurement of LVEDD were according to current recommendations [12]. However, the accuracy of LVEF calculation was in excess of ±10% for Simpson’s rule with echocardiography [28]. Thus, LVEF might have limited reproducibility, especially in 2D imaging, which is commonly used in clinical practice. In contrast, LVEDD was measured using an electronic caliper and a line at the certain level just below the mitral valve leaflet tips, which enhanced the reproducibility of the results.
For the optimal cut-off of LVEDD, previous studies applied different cut-off values according to the characteristics of patients. In CHART-1 study, LVEDD > 60 mm was associated with SCD in heart failure patients, while in CHART-2 study, LVEDD > 65 mm rather than 60 mm was a significant risk factor [8,20]. In our study, we decided not to define the optimal cut-off using the Youden index in receiver-operating characteristic, as this could lead to a type-I error inflation. Moreover, LVEDD represents a continuum of risk, and the aim of this study is to qualify the relationship of LVEDD and clinical outcomes. Thus, we moved from a dichotomous to a continuous risk stratification.
LVEDD and LVEF in this study were measured at baseline. Prior study has proven that 24% of NICM patients with prophylactic ICD implantation had left ventricular function improvement (LVEF increase ≥5% and LVEDD decrease ≥5 mm) within 1 year [29]. However, our study shows that LVEDD was still associated with clinical outcomes for almost ten years of follow-up, and future studies are warranted to assess the value of dynamic examination of LVEDD.
Limitations
Our study has several limitations. First, the main limitation of the present study is its retrospective observation design. However, it reflects the real-world clinical practice and prospective research is needed in the future. Second, in our study, the appropriate shock was the secondary outcome as the substitute for SCD. We acknowledge that SCD is a more meaningful endpoint. Moreover, a study showed that ICD therapies are more frequent than sudden death, with approximately twice as many shocks as fatal arrhythmias [30]. However, we did not obtain the causes of death, thus we could only use appropriate shock as a surrogate for SCD. Third, echocardiography was measured at baseline, and the association of temporal change of LVEDD and clinical outcomes was not evaluated. In our study, LVEDD was still associated with clinical outcomes after 59.6 months of follow-up. Future studies are needed to assess the relationship between LVEDD temporal change and clinical outcomes. Fourth, we included a mixed population of patients with ischemic and non- ischemic cardiomyopathy and primary and secondary prevention indication, while there were no significant interactions in subgroup analysis. Fifth, we did not have the genetic testing results to perform a subgroup analysis. Finally, as granular CMR data were not available in this study, no further relationship between LVEDD and fibrosis could be investigated.
5. Conclusions
In conclusion, in patients with ICD implantation, both LVEDD and LVEF showed a linear relationship with all-cause mortality. However, LVEF was not associated with appropriate shock. In contrast, LVEDD still showed a linear relationship with appropriate shock. Moreover, LVEDD had a strong incremental prognostic value for outcomes over clinical factors. Thus, further studies are needed to validate the results, and LVEDD may be incorporated into the risk stratification of patients with ICD implantation.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jcdd9120421/s1, Table S1: Baseline characteristics of the study population according to the LVEF and LVDD quintiles; Table S2: Univariable Cox regression for the prediction of all-cause mortality and appropriate shock; Table S3: Multivariable regression results according to echo parameter quintiles and clinical outcomes in model 4; Table S4: Statistical association of baseline variables with all-cause mortality and appropriate shock; Table S5: Incremental prognostic value of LVEF and LVEDD for clinical outcomes; Table S6: Interaction analysis for prediction of outcomes; Table S7: Prediction of all-cause mortality and appropriate shock using complete-case (N = 535).
Author Contributions
Conceptualization, W.H.; methodology, S.C.; software, Y.D.; validation, S.C.; formal analysis, H.H.; investigation, H.N.; resources, X.C.; data curation, Y.D.; writing—original draft preparation, S.C., X.L. and M.G.; writing—review and editing, W.H.; visualization, Y.Y.; supervision, W.H.; project administration, W.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of Fuwai hospital (protocol code 2012-397; Approval date: 17 July 2012) for studies involving humans.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data of this study are available on reasonable request from the corresponding author.
Acknowledgments
The authors thank all patients for their contribution to this study.
Conflicts of Interest
The authors declare no conflict of interest.
Nonstandard Abbreviations and Acronyms
| ACEI | angiotensin-converting enzyme inhibitor |
| AF | atrial fibrillation |
| ARB | angiotensin II receptor blocker |
| ARNI | angiotensin receptor-neprilysin inhibitor |
| BMI | body mass index |
| CI | confidence interval |
| CLBBB | complete left bundle branch block |
| CRBBB | complete right bundle branch block |
| eGFR | estimated glomerular filtration rate |
| HR | hazard ratio |
| ICD | implantable cardioverter defibrillator |
| ICM | ischemic cardiomyopathy |
| IDI | integrated discrimination index |
| LVEDD | left ventricular end-diastolic diameter |
| LVEF | left ventricular ejection fraction |
| NICM | non-ischemic cardiomyopathy |
| NRI | net reclassification index |
| NT-proBNP | N-terminal pro-brain natriuretic peptide |
| NYHA | New York Heart Association |
| SCD | sudden cardiac death |
References
- Zeppenfeld, K.; Tfelt-Hansen, J.; de Riva, M.; Winkel, B.G.; Behr, E.R.; Blom, N.A.; Charron, P.; Corrado, D.; Dagres, N.; de Chillou, C.; et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur. Heart J. 2022, 26, ehac262. [Google Scholar]
- Al-Khatib, S.M.; Stevenson, W.G.; Ackerman, M.J.; Bryant, W.J.; Callans, D.J.; Curtis, A.B.; Deal, B.J.; Dickfeld, T.; Field, M.E.; Fonarow, G.C.; et al. 2017 AHA/ACC/HRS Guideline for Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death. J. Am. Coll. Cardiol. 2018, 72, e91–e220. [Google Scholar] [CrossRef] [PubMed]
- Moss, A.J.; Wilber, D.J.; Brown, M.W. Prophylactic Implantation of a Defibrillator in Patients with Myocardial Infarction and Reduced Ejection Fraction. N. Engl. J. Med 2002, 346, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Kadish, A.; Quigg, R.; Calkins, H.; Shalaby, A.; Levine, J.H. Prophylactic Defibrillator Implantation in Patients with Nonischemic Dilated Cardiomyopathy. N. Engl. J. Med. 2004, 350, 2151–2158. [Google Scholar] [CrossRef] [PubMed]
- Bardy, G.H.; Boineau, R.; Johnson, G.; Davidson-Ray, L.D.; Ip, J.H. Amiodarone or an Implantable Cardioverter–Defibrillator for Congestive Heart Failure. N. Engl. J. Med. 2005, 352, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Quiñones, M.A.; Greenberg, B.H.; Kopelen, H.A.; Koilpillai, C.; Limacher, M.C.; Shindler, D.M.; Shelton, B.J.; Weiner, D.H.; for the SOLVD Investigators. Echocardiographic predictors of clinical outcome in patients with left ventricular dysfunction enrolled in the SOLVD registry and trials: Significance of left ventricular hypertrophy. J. Am. Coll. Cardiol. 2000, 35, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Aleong, R.G.; Mulvahill, M.J.; Halder, I.; Carlson, N.E.; Singh, M.; Bloom, H.L.; Dudley, S.C.; Ellinor, P.T.; Shalaby, A.; Weiss, R.; et al. Left Ventricular Dilatation Increases the Risk of Ventricular Arrhythmias in Patients with Reduced Systolic Function. J. Am. Hear. Assoc. 2015, 4, e001566. [Google Scholar] [CrossRef]
- Watanabe, J.; Shinozaki, T.; Shiba, N.; Fukahori, K.; Koseki, Y.; Karibe, A.; Sakuma, M.; Miura, M.; Kagaya, Y.; Shirato, K. Accumulation of risk markers predicts the incidence of sudden death in patients with chronic heart failure. Eur. J. Heart Fail. 2006, 8, 237–242. [Google Scholar] [CrossRef]
- Kajimoto, K.; Minami, Y.; Otsubo, S.; Sato, N. Sex Differences in Left Ventricular Cavity Dilation and Outcomes in Acute Heart Failure Patients with Left Ventricular Systolic Dysfunction. Can. J. Cardiol. 2018, 34, 477–484. [Google Scholar] [CrossRef]
- Catanzaro, J.N.; Makaryus, J.N.; Makaryus, A.N.; Sison, C.; Vavasis, C.; Fan, D.; Jadonath, R. Echocardiographic Predictors of Ventricular Tachycardia. Clin. Med. Insights Cardiol. 2015, 8 (Suppl. S4), 37–42. [Google Scholar] [CrossRef]
- Narayanan, K.; Reinier, K.; Teodorescu, C.; Uy-Evanado, A.; Aleong, R.; Chugh, H.; Nichols, G.A.; Gunson, K.; London, B.; Jui, J.; et al. Left Ventricular Diameter and Risk Stratification for Sudden Cardiac Death. J. Am. Heart Assoc. 2014, 3, e001193. [Google Scholar] [CrossRef]
- Mitchell, C.; Rahko, P.S.; Blauwet, L.A.; Canaday, B.; Finstuen, J.A.; Foster, M.C.; Horton, K.; Ogunyankin, K.O.; Palma, R.A.; Velazquez, E.J.; et al. Guidelines for Performing a Comprehensive Transthoracic Echocardiographic Examination in Adults: Recommendations from the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2019, 32, 1–64. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.D.; Velazquez, E.; Kober, L.; Pfeffer, M.A. Sudden Death in Patients with Myocardial Infarction and Left Ventricular Dysfunction, Heart Failure, or Both. N. Engl. J. Med. 2005, 352, 2581–2588. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, P.A.; Bozkurt, B.; Aguilar, D.; Allen, L.A.; Byun, J.J.; Colvin, M.M.; Deswal, A.; Drazner, M.H.; Dunlay, S.M.; Evers, L.R.; et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. J. Am. Coll. Cardiol. 2022, 79, e263–e421. [Google Scholar] [PubMed]
- Køber, L.; Thune, J.J.; Nielsen, J.C.; Haarbo, J.; Videbæk, L.; Korup, E.; Jensen, G.; Hildebrandt, P.; Steffensen, F.H.; Bruun, N.E.; et al. Defibrillator Implantation in Patients with Nonischemic Systolic Heart Failure. N. Engl. J. Med. 2016, 375, 1221–1230. [Google Scholar] [CrossRef]
- Di Marco, A.; Brown, P.F.; Bradley, J.; Nucifora, G.; Claver, E.; de Frutos, F.; Dallaglio, P.D.; Comin-Colet, J.; Anguera, I.; Miller, C.A.; et al. Improved Risk Stratification for Ventricular Arrhythmias and Sudden Death in Patients with Nonischemic Dilated Cardiomyopathy. J. Am. Coll. Cardiol. 2021, 77, 2890–2905. [Google Scholar] [CrossRef] [PubMed]
- Marijon, E.; Garcia, R.; Narayanan, K.; Karam, N.; Jouven, X. Fighting against sudden cardiac death: Need for a paradigm shift—Adding near-term prevention and pre-emptive action to long-term prevention. Eur. Heart J. 2022, 43, 1457–1464. [Google Scholar] [CrossRef] [PubMed]
- Zecchin, M. The role of implantable cardioverter defibrillator for primary vs secondary prevention of sudden death in patients with idiopathic dilated cardiomyopathy. Europace 2004, 6, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Sarak, B.; Kaplan, A.J.; Oosthuizen, R.; Beardsall, M.; Wulffhart, Z.; Higenbottam, J.; Khaykin, Y. Predictors of Appropriate Implantable Cardioverter Defibrillator (ICD) Therapy in Primary Prevention Patients with Ischemic and Nonischemic Cardiomyopathy. Pacing Clin. Electrophysiol. 2010, 33, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Satake, H.; Fukuda, K.; Sakata, Y.; Miyata, S.; Nakano, M.; Kondo, M.; Hasebe, Y.; Segawa, M.; Shimokawa, H.; on behalf of the CHART-2 Investigators. Current Status of Primary Prevention of Sudden Cardiac Death with Implantable Cardioverter Defibrillator in Patients with Chronic Heart Failure—A Report From the CHART-2 Study. Circ. J. 2015, 79, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Chen, K.; Su, Y.; Hua, W.; Yang, J.; Chen, S.; Liang, Z.; Xu, W.; Zhang, S. High incidence of ventricular arrhythmias in patients with left ventricular enlargement and moderate left ventricular dysfunction. Clin. Cardiol. 2016, 39, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.C.; Lee, D.S.; Fine, J.P. Introduction to the Analysis of Survival Data in the Presence of Competing Risks. Circulation 2016, 133, 601–609. [Google Scholar] [CrossRef]
- Aiba, T.; Tomaselli, G.F. Electrical remodeling in the failing heart. Curr. Opin. Cardiol. 2010, 25, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Adelstein, E.C.; Schwartzman, D.; Jain, S.; Bazaz, R.; Wang, N.C.; Saba, S. Left ventricular dimensions predict risk of appropriate shocks but not mortality in cardiac resynchronization therapy- defibrillator recipients with left bundle-branch block and non-ischemic cardiomyopathy. Europace 2017, 19, 1689–1694. [Google Scholar] [CrossRef] [PubMed]
- Aiba, T.; Hesketh, G.G.; Barth, A.S.; Liu, T.; Daya, S.; Chakir, K.; Dimaano, V.L.; Abraham, T.P.; O’Rourke, B.; Akar, F.G.; et al. Electrophysiological Consequences of Dyssynchronous Heart Failure and Its Restoration by Resynchronization Therapy. Circulation 2009, 119, 1220–1230. [Google Scholar] [CrossRef] [PubMed]
- Gulati, A.; Jabbour, A.; Ismail, T.F.; Guha, K.; Khwaja, J.; Raza, S.; Morarji, K.; Brown, T.D.H.; Ismail, N.A.; Dweck, M.R.; et al. Association of Fibrosis with Mortality and Sudden Cardiac Death in Patients with Nonischemic Dilated Cardiomyopathy. JAMA 2013, 309, 896. [Google Scholar] [CrossRef] [PubMed]
- Klem, I.; Klein, M.; Khan, M.; Yang, E.Y.; Nabi, F.; Ivanov, A.; Bhatti, L.; Hayes, B.; Graviss, E.A.; Nguyen, D.T.; et al. Relationship of LVEF and Myocardial Scar to Long-Term Mortality Risk and Mode of Death in Patients with Nonischemic Cardiomyopathy. Circulation 2021, 143, 1343–1358. [Google Scholar] [CrossRef] [PubMed]
- Goldberger, J.J.; Cain, M.E.; Hohnloser, S.H.; Kadish, A.H.; Knight, B.P.; Lauer, M.S.; Maron, B.J.; Page, R.L.; Passman, R.S.; Siscovick, D.; et al. American Heart Association/American College of Cardiology Foundation/Heart Rhythm Society Scientific Statement on Noninvasive Risk Stratification Techniques for Identifying Patients at Risk for Sudden Cardiac Death. J. Am. Coll. Cardiol. 2008, 52, 1179–1199. [Google Scholar] [CrossRef]
- Grimm, W.; Timmesfeld, N.; Efimova, E. Left ventricular function improvement after prophylactic implantable cardioverter-defibrillator implantation in patients with non-ischaemic dilated cardiomyopathy. Europace 2013, 15, 1594–1600. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ellenbogen, K.A.; Levine, J.H.; Berger, R.D.; Daubert, J.P.; Winters, S.L.; Greenstein, E.; Shalaby, A.; Schaechter, A.; Subacius, H.; Kadish, A.; et al. Are Implantable Cardioverter Defibrillator Shocks a Surrogate for Sudden Cardiac Death in Patients with Nonischemic Cardiomyopathy? Circulation 2006, 113, 776–782. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).