Abstract
Agrocybe chaxingu is a widely cultivated edible fungus in China, which is rich in nutrients and medicinal compounds. However, the lack of a high-quality genome hinders further research. In this study, we assembled the telomere-to-telomere genomes of two sexually compatible monokaryons (CchA and CchB) derived from a primarily cultivated strain AS-5. The genomes of CchA and CchB were 50.60 Mb and 51.66 Mb with contig N50 values of 3.95 Mb and 3.97 Mb, respectively. Each contained 13 complete chromosomes with telomeres at both ends. The high mapping rate, uniform genome coverage, high LAI score, all BUSCOs with 98.5%, and all base accuracy exceeding 99.999% indicated the high level of integrity and quality of these two assembled genomes. Comparison of the two genomes revealed that approximately 30% of the nucleotide sequences between homologous chromosomes were non-syntenic, including 19 translocations, 36 inversions, and 15 duplications. An additional gene CchA_000467 was identified at the Mat A locus of CchA, which was observed exclusively in the Cyclocybe cylindracea species complex. A total of 613 (4.26%) and 483 (3.4%) unique genes were identified in CchA and CchB, respectively, with over 80% of these being hypothetical proteins. Transcriptomic analysis revealed that the expression levels of unique genes in CchB were significantly higher than those in CchA, and both CchA and CchB had unique genes specifically expressed at stages of mycelium and fruiting body. It was indicated that the growth and development of the A. chaxingu strain AS-5 required the coordinated action of two different nuclei, with CchB potentially playing a more significant role. These findings contributed to a more profound comprehension of the growth and developmental processes of basidiomycetes.
1. Introduction
Agrocybe chaxingu is a widely cultivated edible mushroom in Asia that belongs to the Strophariaceae family. It is a member of the Cyclocybe cylindracea species complex, which includes Cyclocybe cylindracea, Cyclocybe aegerita, and Cyclocybe salicaceicola [1]. In 2022, the production of A. chaxingu in China reached 882,300 tonnes, ranking 8th among all the cultivated mushrooms, as reported by the China Edible Fungi Association (https://www.cefa.org.cn/web/index.html, accessed on 28 December 2023).
Consumers have a heightened appreciation for the A. chaxingu due to its unique flavor and nutritional value [2]. In addition, A. chaxingu was reported to have various pharmacological effects, such as anti-inflammatory [3], hypoglycemic [4], and anti-tumor activities [5]. Furthermore, the ribotoxin “Ageritin” was discovered in C. aegerita [6] and reported to exhibit ribonucleolytic activity on ribosomes, ribonuclease activity on tobacco mosaic virus RNA, endonuclease activity on plasmid and genomic DNAs, as well as antiproliferative and defense properties [7]. In 2004, the fungal heme-thiolate enzyme subfamily of unspecific peroxygenases were first described in C. aegerita [8], which were versatile biocatalysts oxidizing non-activated C-H bonds in a regiospecific and stereospecific manner under mild reaction conditions [9]. The plethora of bioactive compounds produced by the C. cylindracea species complex requires further comprehensive research. The availability of high-quality genomes will facilitate the exploration of secondary metabolism.
In addition, several species within the C. cylindracea species complex are morphologically similar. The phylogenetic relationships among A. chaxingu, C. cylindracea, C. aegerita, and C. salicaceicola were complex [10]. Phylogenomic analysis can facilitate the elucidation of the phylogenetic relationships, evolutionary histories, and population structures of these species. It is crucial to clarify the phylogenetic relationships between A. chaxingu and other taxa within the C. cylindracea species complex for the future breeding of A. chaxingu. The genome of the C. aegerita strain AAE-3 was published in 2018, unveiling a repertoire of fruiting-related genes and biopolymer-degrading enzymes [11]. Subsequently, the genome of C. cylindracea was assembled, and detailed pathways for the synthesis of nutritional and flavor-related compounds were analyzed [12]. These genomes within the C. cylindracea species complex comprised of 122 and 3790 scaffolds. In 2023, a haploid genome draft of the A. chaxingu strain MP-N11 was reported to be assembled (Submitted GenBank assembly: GCA_027627235.1, https://www.ncbi.nlm.nih.gov/datasets/genome/GCA_027627235.1/, accessed on 22 May 2023). However, the assembly consisted of 4634 contigs with an N50 of 18.1 Kb, indicating that there is room for improvement in the assembly quality.
A. chaxingu is a tetrapolar heterothallic basidiomycete fungus [13]. During most of its lifecycle, each cell contains two nuclei with different mating types, and these two nuclei share the same cytoplasm. The current incomplete haploid genome of A. chaxingu obviously cannot fully represent the entire genetic information of the species. In addition, the chromosomal structural variances between these two nuclei and their interactions are critical for understanding the growth and development mechanisms of mushroom-forming fungi.
In this study, whole-genome sequencing of two sexually compatible monokaryons (CchA and CchB) derived from the dominant cultivator A. chaxingu strain AS-5 was performed. The telomere-to-telomere (T2T) chromosome-level genomes of CchA and CchB were generated by incorporating data from the Illumina, PacBio high fidelity (HiFi), and High-throughput Chromatin Conformation Capture (Hi-C) sequencing platforms. Subsequently, comparative genomic analysis revealed differences between the reference genomes of the two monokaryotic strains with compatible mating types in terms of chromosomal structural variations, mating loci, and gene family evolution. Finally, an analysis was conducted on the gene expression profiles at the mycelial and fruiting body stages of the A. chaxingu AS-5, with a particular focus on the unique genes of the strains CchA and CchB. This study contributed to understanding the genetic differences between the two nuclei of A. chaxingu and their roles in fruiting body development through T2T genome comparisons, mating type loci analysis, and transcriptional profiling at two stages.
2. Materials and Methods
2.1. A. chaxingu Strain, Culture Conditions, and Nucleic Acid Preparation
The strain AS-5 is the commercially dominant cultivar of A. chaxingu in China. It was preserved at the China General Microbiological Culture Collection Center (CGMCC No. 40980). The monokaryotic strains of the dikaryotic strain AS-5 were isolated through protoplast monokaryotization following our previously reported method [14]. The detailed procedure was as follows: Seven fungal plugs (4 mm in diameter) of A. chaxingu AS-5 were inoculated onto a Potato Dextrose Agar (PDA) plate (9 cm in diameter) that had been covered with cellophane. The plate was incubated in the dark at 25 °C until the fungal mycelia of the colonies came into contact with one another. Next, the cellophane with the fungal colonies was transferred to a new sterile Petri dish using sterile forceps, ensuring the mycelium side faced upwards. A 3 mL solution of 1.5% (w/v) lywallzyme (Guangdong Microbiology Institute) was then applied to the mycelia. The solution was prepared using a 0.6 mol/L mannitol solution. Enzymatic digestion was conducted at 33 °C for 70 min to obtain protoplasts, which were subsequently counted using a hemocytometer. Then, 50 μL of the enzyme solution containing 105 protoplasts/mL was spread onto regeneration medium plates, which were prepared with 200.00 g of potato, 20.00 g of glucose, 20.00 g of agar, 205.38 g of sucrose, and 1.00 g of maltose, made up to a final volume of 1000 mL. The plates were incubated in the dark at 25 °C. After the mycelia had developed, single colonies were picked and transferred to PDA plates. The monokaryotic strains without any clamp connection were selected under 1000× magnification using an optical microscope (Eclipse 80i, Nikon, Tokyo, Japan). Strains CchA and CchB were two monokaryotic strains with compatible mating types. They were capable of forming a clamp connection when co-cultured. Both the dikaryotic and monokaryotic strains were cultivated in the dark on PDA medium at 25 °C.
For the genome sequencing, the monokaryotic strains CchA and CchB were cultured in Potato Dextrose Broth (PDB) at 25 °C in the dark for 14 d. For the transcriptome sequencing, the mycelia of A. chaxingu AS-5 were cultured on a PDA medium for 7 d. Under conditions of 25 °C and scattered light, A. chaxingu AS-5 formed primordia on the substrate (63% sawdust, 20% cottonseed hulls, 15% bran, 1% lime, and 1% gypsum) and then grew for an additional 5 d to form mature fruiting bodies. Then, the mycelia and fruiting bodies were collected and washed with sterile water, and promptly frozen in liquid nitrogen for subsequent nucleic acid extraction. Genomic DNA was extracted using the QIAGEN® Genomic Kit (Qiagen, Dusseldorf, Germany) according to the manufacturer’s instructions. Total RNA extraction was performed using TRIzol reagent (Invitrogen, Carlsbad, CA, USA).
2.2. Genome Sequencing and Heterozygosity Estimation
Both the DNA samples from strains CchA and CchB were sequenced using the MGISEQ-T7 (with the construction of a 350 bp library) and the PacBio Sequel II (with the PacBio HiFi construction of a 15 Kb SMRTbell library) platforms at Wuhan Grandomics Biosciences Co., Ltd. (Wuhan, Hubei, China). The DNA of strain CchA was subjected to Hi-C sequencing on the Illumina NovaSeq 6000 platform at Shanghai Biozeron Biotech. Co., Ltd. (Shanghai, China), with paired-end reads of 150 bp. The RNA from the mycelia of the strains CchA and A. chaxingu AS-5 and the fruiting bodies of the strain A. chaxingu AS-5 were used for transcriptome sequencing on the Illumina NovaSeq 6000 platform at Shanghai Biozeron Biotech. Co., Ltd. (Shanghai, China).
For the estimation of genome heterozygosity, Jellyfish v 2.1.3 [15] was utilized for K-mer distribution analysis based on next-generation sequencing reads (NGS). The results were then imported into GenomeScope v1.0 [16] with a K-mer length of 21 and a ploidy of 2.
2.3. De Novo Genome Assembly and Quality Assessment
The de novo genome assembly of the strains CchA and CchB primarily involves error correction to identify haplotypes, the construction of assembly graphs, and the generation of assembled sequences. Firstly, we used Smrt link v7.0 (https://github.com/WenchaoLin/SMRT-Link, accessed on 6 July 2023) to pre-process the PacBio raw reads, removing adapter sequences, low-quality regions, and sequences containing sequencing errors. We also employed the default parameters of the CCS software v3.0.0 (https://github.com/PacificBiosciences/ccs, accessed on 10 July 2023) to generate HiFi reads. Using the Hifiasm v0.13 software [17] with default parameters, we performed de novo assembly of PacBio HiFi long-reads. Subsequently, short reads were employed and further error correction of the assembled genome was conducted using the Pilon v1.23 [18]. ALLHIC software (https://github.com/tanghaibao/allhic, accessed on 21 August 2023) was used to link the genome contigs or scaffolds.
To address the missing telomeres, a manual identification process was used to extract the telomeric repeat sequences ‘CCCTAA’ and ‘TTAGGG’ from the PacBio HiFi reads. The extracted sequences of the missing telomeres were assembled using the Hifiasm software v0.12 [17] with default parameters, and they were subsequently matched with each chromosome. Further error correction was performed using Racon [19] and Pilon [18] to ensure assembly quality.
The quality assessment of each genome primarily included the long terminal repeat (LTR) assembly index (LAI) [20], mapping rate of reads, overall base accuracy estimates, and Benchmarking Universal Single-Copy Orthologs (BUSCO) completeness analysis [21].
2.4. Genome Annotation
De novo prediction, homology-based prediction, and transcriptome prediction were employed for gene prediction. De novo gene prediction was performed using Augustus v3.3.1 [22]. For homology prediction, the protein sequences of Psilocybe cubensis (the National Center for Biotechnology Information GenBank: GCA_017499595.2), Agrocybe pediades (GCA_015484485.1), Pholiota molesta (GCA_014925825.1), and Hypholoma sublateritium (GCA_000827495.1) were mapped to the A. chaxingu genome using GeMoMa v1.6.2 [23] with default parameters. To facilitate gene prediction using transcriptome sequencing (RNA-seq), we used PASA v2.3.3 [24] to map RNA-seq data onto the A. chaxingu genome. Finally, EVM [25] was used to integrate these data to eliminate redundancy in the final gene set.
We used the rRNAmmer v1.2 [26] to detect rRNA in the A. chaxingu genome by ab initio prediction and homologous sequences downloaded from the Ensembl database [27]. The tRNAscan-SE with the default parameters was employed to identify the genes related to tRNA [28]. For non-coding RNA such as snRNA and miRNA, we utilized the Infernal [29] with default parameters for annotation, based on the Rfam database [30].
RepeatModeler v2.0.2a [31] and RepeatMasker v4.1.2 [32] softwares were utilized to predict transposon sequences by aligning the assembled genome of A. chaxingu against the transposon Repbase database [33]. Additionally, LTR_ finder [34] and LTR_ harvest [35] were employed for de novo searching of LTR retrotransposons. The unknown repetitive sequences were classified using DeepTE [36].
For functional annotation of the predicted protein-coding genes, BLASTP v2.7.1 [37] was used to align protein sequences against databases including Protein FAMilies Database (PFAM) [38], Integrated Resource of Protein Domains and Functional Sites (InterPro) [39], Clusters of Orthologous Groups (COGs) [40], Gene Ontology Terms (GO Terms) [41], BUSCO [21], and Evolutionary Genealogy of Genes: Non-supervised Orthologous Groups (EggNOG) [42]. InterProScan 5.32-71.0 [43] was used to identify protein domains and obtain the annotations of GO using default parameters. Kyoto Encyclopedia of Genes and Genomes (KEGG) annotation was performed using KAAS [44].
The carbohydrate-active enzymes (CAZymes) were analyzed using the CAZyme database (http://www.cazy.org/, accessed on 25 August 2023). Fungal AntiSMASH 7.1.0 (https://fungismash.secondarymetabolites.org/, accessed on 2 September 2023) was utilized to predict secondary metabolite gene clusters.
2.5. Identification of Mating-Type Locus
The identification of mating-type A (MatA) locus involved utilizing genome annotation results, conducting homology searches in closely related species, and performing homology searches in model species for mitochondrial intermediate peptidase (Mip)-associated genes related to the MatA. The selected closely related species included C. cylindracea [12] and C. aegerita [11], while the model species chosen were Schizophyllum commune [45] and Coprinopsis cinerea [46]. The pheromone receptor (Pr) genes of the mating-type B locus (MatB) were searched for in the annotation results of GO Terms. Simultaneously, the two highly conserved Pr genes reported in the closely related species C. salicacola [47] were used for a homology search to determine the location of MatB, while other Pr proteins were validated through a BLASTP homology comparison in the C. aegerita genome [11]. The sequence length of pheromone precursor (Pp) is typically less than 60 amino acids, and their high variability makes them difficult to identify by BLAST homology searches. Furthermore, gene prediction methods can only predict a small fraction of these genes. Based on the localization of Pp genes in proximity to the Pr [48,49,50], the presence of the CAAX-box motif at the 3′ end, and upstream conserved AF and EA motifs [51,52,53,54], we annotated the Pp genes by utilizing Expasy (https://web.expasy.org/translate/, accessed on 15 September 2023) to annotate the DNA sequences within a 50 kb range of the Pr.
To verify the authenticity of the gene CchA_000467 at MatA locus in the strain CchA, primers (CchA-F: GCTTGGGTGACGTCATTAC/CchA-R3: CARATTGAWGTTTGGGTAC) were designed. PCR reaction was performed in 20 µL of total volume: 0.8 µL of each primer (10 µM), 2 µL of DNA, 10 µL of PrimeSTAR® Max DNA Polymerase (TAKARA), and 6.4 µL of ddH2O. The PCR was performed as follows: 95 °C, 3 min; 35 cycles of 15 s at 95 °C, 15 s at 48 °C, and 55 s at 72 °C; 72 °C, 10 min. The PCR products were checked through electrophoresis on 1.0% agarose gel.
2.6. Phylogenomic and Evolutionary Analyses
Phylogenomic analysis was performed with 15 fungal genomes (Table S1). We identified single-copy orthologous genes using OrthoFinder v2.5.4 [55] and performed sequence alignment using Muscle v3.8.31 [56] with default parameters. Conservative sequences were selected using Gblocks v0.91b [57] and subsequently concatenated into a supermatrix by seqkit v2.2.0 [58]. A maximum-likelihood phylogenetic tree was constructed using RAxML-NG v0.9.0 [59] with the amino acid substitution matrix LG+F+I+G4 selected by ProtTest v3.4.2 [60]. The optimal tree was visualized in FigTree v1.4.4 (https://github.com/rambaut/figtree/, accessed on 6 January 2024). The software r8s v1.81 [61] was utilized to estimate the divergence times of species with an approximate likelihood method by calibrating with two fossil record fungal species of C. cinerea and Laccaria bicolor [62]. CAFE5 v1.1 [63] was employed to calculate expansions or contractions in gene families.
2.7. Comparative Genomic Analysis
Whole-genome synteny analysis of the nucleotide sequences was executed on the monoploid genomes of strains CchA and CchB using MUMmer [64]. The gene collinearity relationships between each chromosome of CchA and CchB were analyzed using MCScanX (E value < 1 × 10−5) [65]. Furthermore, we conducted an analysis using SyRI v1.5 software [66] to identify structural variations larger than 10 kb between homologous chromosomes of two genomes, including translocations, inversions, and duplications. The identification of unique genes between the genome of strains CchA and CchB was carried out using OrthoFinder v2.5.4 [55]. Subsequently, the KEGG enrichment of these specific genes was analyzed using TBtools [67].
To verify the authenticity of the unique genes CchA_005693 and CchA_005699 in the genome of CchA, primers (005693-F: ATGCTGACTTTGAGATATAAGAAGCAG/005693-R: TTATTTCGCATTGGGTGGCG; 005699-F: ATGAAAGGTGGGATGTCGAG/005693-R: TTATTTCGCATTGGGTGGCG, respectively) were designed. PCR reaction was performed in 20 µL of total volume: 0.8 µL of each primer (10 µM), 2 µL of DNA, 10 µL of PrimeSTAR® Max DNA Polymerase (TAKARA), and 6.4 µL of ddH2O. The PCR was performed as follows: 95 °C, 3 min; 35 cycles of 15 s at 95 °C, 15 s at 55 °C, and 15 s at 72 °C; 72 °C, 10 min. The PCR products were checked through electrophoresis on 1.0% agarose gel.
2.8. Transcriptome Analysis
The relative expression levels were recorded as transcripts per kilobase of exon model per million mapped reads (TPM), which were calculated using featureCounts 2.0.6 [68]. The clean RNA-seq reads were mapped to the assembly genomes of strains CchA and CchB using HISAT v2.2.1 [69] for TPM calculation.
3. Results
3.1. Genome Sequencing, T2T Assembly, and Quality Assessment
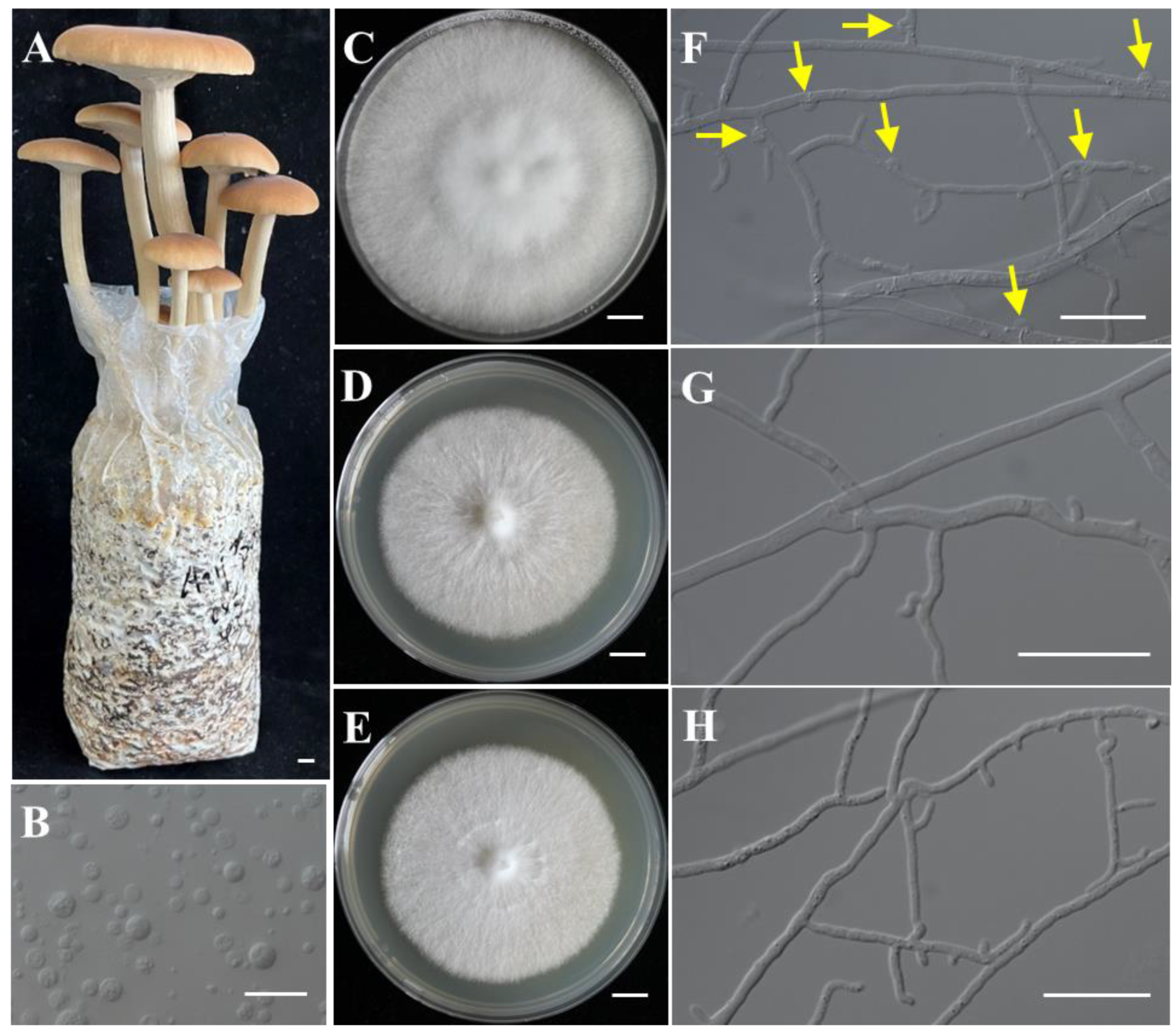
Strains CchA and CchB were two monokaryotic strains with compatible mating types isolated through protoplast monokaryotization from the commercially dominant cultivar “AS-5” (Figure 1A–H). They were capable of forming a clamp connection when co-cultured. Genome sequencing of the monokaryotic strains CchA and CchB (Table S2) was conducted using the MGISEQ-T7 and PacBio Sequel II platforms. A total of 9.55 Gb (sequence coverage ~193.26×) and 6.99 Gb (~138.56×) of NGS reads were generated for strains CchA and CchB, along with 7.30 Gb (~147.73×) and 7.04 Gb (~139.55×) of PacBio HiFi reads, respectively. In addition, Hi-C sequencing of the strain CchA was conducted on the Illumina NovaSeq 6000 platform, generating 5.04 Gb (~101.89×) of clean data. Based on 21 K-mer analysis (Figure S1), it was found that heterozygosity levels of strains CchA and CchB were only 0.03% and 0.04%, respectively. The estimated genome sizes were 45.60 Mb for strain CchA and 47.31 Mb for strain CchB.
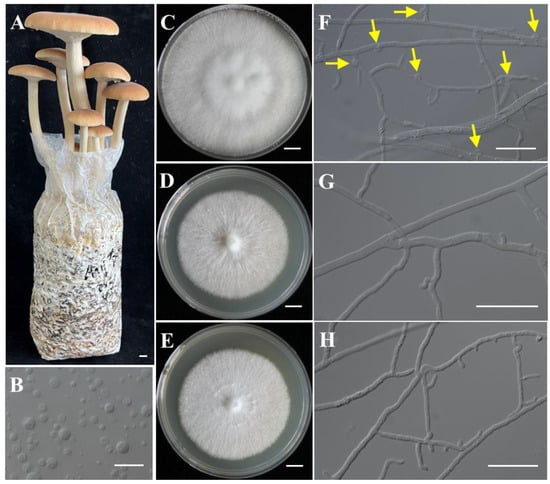
Figure 1.
The fruiting bodies of Agrocybe chaxingu and the monokaryotic strains used for genome sequencing. (A) The fruiting bodies of A. chaxingu. (B) The protoplasts of A. chaxingu. (C–E) Colony of heterokaryotic and two monokaryotic strains. (F) Hyphae of heterokaryotic strain with clamp connections. Yellow arrows indicate the clamp connections. (G,H) Hyphae of monokaryotic strain CchA and CchB. Bars: (A,C–E) = 1 cm; (B,F–H) = 20 μm.
We successfully assembled two T2T chromosome-level haploid genomes of A. chaxingu (Figure 2). The CchA genome had a total length of 50.60 Mb (50,604,502 bp) with 13 chromosomes, N50 of 3.95 Mb, and chromosome lengths ranging from 2.65 Mb to 5.19 Mb (Table S3). The CchB genome measured 51.66 Mb (51,660,018 bp), with 13 chromosomes, N50 of 3.97 Mb, and chromosome lengths ranging from 2.98 Mb to 5.46 Mb (Table S3). The GC content of the CchA genome was 50.99%, while that of CchB was 50.98%. A total of 26 telomeres were detected at both ends of their 13 chromosomes, characterized by (CCCTAA)n and (TTAGGG)n repeats for the genomes of both strains.
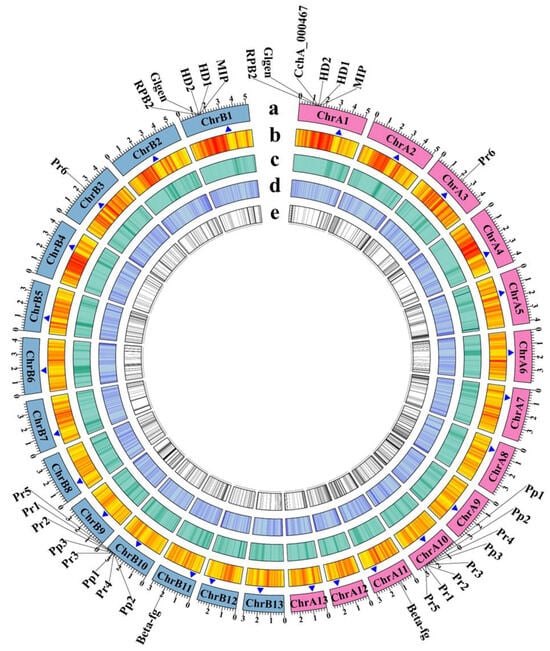
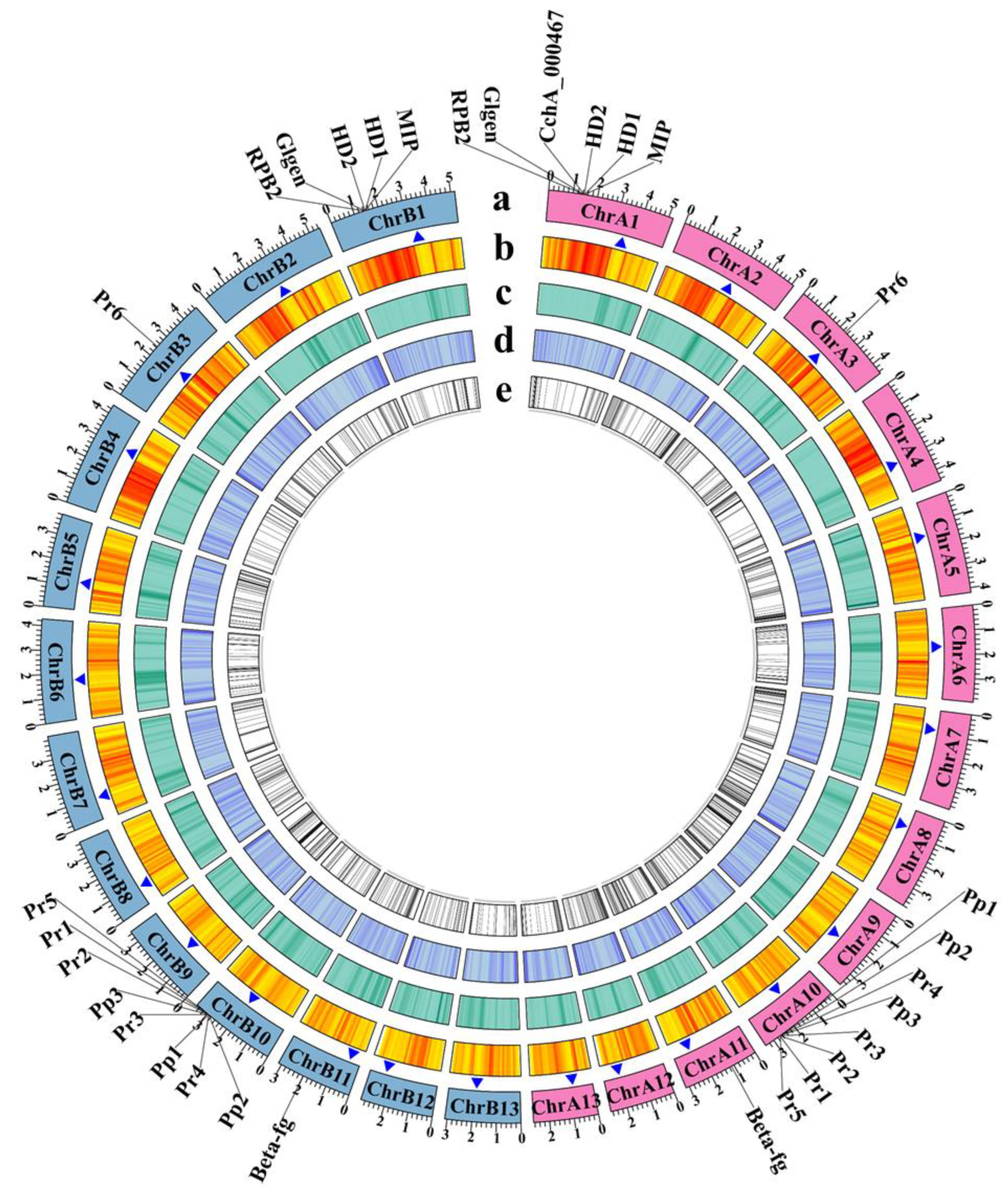
Figure 2.
Genome features of Agrocybe chaxingu strains CchA and CchB. The genes labeled outside the circle were related to mating types and their adjacent genes. HD: Homeodomain, MIP: mitochondrial intermediate peptidase, Glgen: glycosyltransferase family 8 protein, RPB2: RNA polymerase II the second large subunit, Beta-fg: β-flanking, Pr: pheromone receptor, Pp: pheromone precursor. (a) The chromosomes of strains CchA and CchB (Mb). Pink represents the chromosomes of CchA. Light blue represents the chromosomes of CchB. (b) Gene density. (c) Repetitive sequence density. (d) GC content. (e) Unique genes. The blue triangles represent the possible centromere regions.
Ribosomal DNA (rDNA) was predicted at the end of the chromosome 12 (chr12) of both the CchA and CchB genomes (Figure S2A,B). In CchA, the closest Internal Transcribed Spacer (ITS) to the telomere was located 10 kb away, whereas in CchB, the nearest ITS was only 1 kb from the telomere sequences.
The assembly quality and completeness of the CchA and CchB genomes were assessed through various methods. Initially, consistent with the k-mer evaluation, the CchB genome surpassed that of CchA by over 1 Mb. The visualized Hi-C data for the CchA genome exhibited a high degree of consistency across all chromosomes (Figure S2C), indicating the accuracy of their ordering and orientation. Secondly, aligning the HiFi reads from CchA and CchB to the two assembled genomes revealed an average HiFi read coverage of approximately 142.9× and 90× for each chromosome, respectively (Figure S2A,B). The particularly high coverage at the end of chr12 was due to the rDNA sequences. The respective LAI scores for the genomes of CchA and CchB were 23.35 and 25.76, reaching gold quality [20]. Moreover, 99.56% and 99.96% of Illumina reads, as well as 100% and 99.92% of HiFi reads, were successfully mapped to the genomes (including the mitochondrial genome) of CchA and CchB, respectively, indicating sufficiently high coverage (Figure S2D). Using Merqury v1.3 [70], the accuracy of the genome was assessed. The results showed that in the genome of CchA, out of 34,533,832 k-mers (k-mer = 18), only 2611 k-mers were found in the genome but not in the HiFi reads, with a quality value (QV) of 53.5187. The overall base accuracy estimated by T2T assembly was 99.9996%. In the genome of CchB, out of 38,006,749 k-mers (k-mer = 18), only 6606 k-mers were found in the genome but not in the HiFi reads, with a QV of 49.9034. The overall base accuracy estimated by T2T assembly was 99.9990%. Finally, BUSCO analyses revealed that both genomes exhibited a completeness of 98.5%, indicating a high level of genome integrity (Table 1). Overall, these results demonstrated the high quality, accuracy, and reliability of the reference genome for both strains CchA and CchB.

Table 1.
BUSCO assessment results in the genomes of CchA and CchB.
As shown in Table 2, in comparison with the other five genomes within the C. cylindracea species complex, the assembly quality of CchA and CchB has been significantly improved, achieving a chromosome-level genome.

Table 2.
Global statistics for the genomes of CchA, CchB and the previously published strains of C. cylindracea species complex.
3.2. Repeat Annotation, Noncoding RNA Annotation, and Gene Prediction
We identified 8.59 Mb (16.97%) and 9.28 Mb (17.97%) of repetitive sequences in the genome assemblies of the strains CchA and CchB, respectively (Table 3). Among the annotated and classified repetitive sequence elements, the dominant included LTR sequences and Long Interspersed Nuclear Elements (LINEs). In the genome of CchA, LTRs accounted for 3.45% (1.75 Mb), while in CchB, the proportion was 4.36% (2.25 Mb). LTRs comprised two subtypes, Copia and Gypsy. Additionally, LINEs represented 1.73% (0.87 Mb) of the CchA sequence and 1.66% (0.86 Mb) of the CchB sequence. A total of 9.28% (4.97 Mb) and 10.18% (5.26 Mb) of repetitive elements were unclassified in the genome of CchA and CchB, respectively. Furthermore, the CchA and CchB genomes exhibited distinct repetitive elements. Specifically, the L1/CIN4 and Tc1-IS630-Pogo elements were exclusive to CchA, whereas the RTE/Bov-B and Hobo-Activator elements were found solely in CchB. Notably, the Hobo-Activator repetitive element was more prevalent, comprising 0.29% (0.15 Mb) of the CchB genome. Transposons are mobile genetic elements and an important source of gene variation and evolution in many organisms [71], such as Hobo-Activator participating in chromosome rearrangement or breakage [72].

Table 3.
Classification of the repeat sequences in the genome of CchA and CchB.
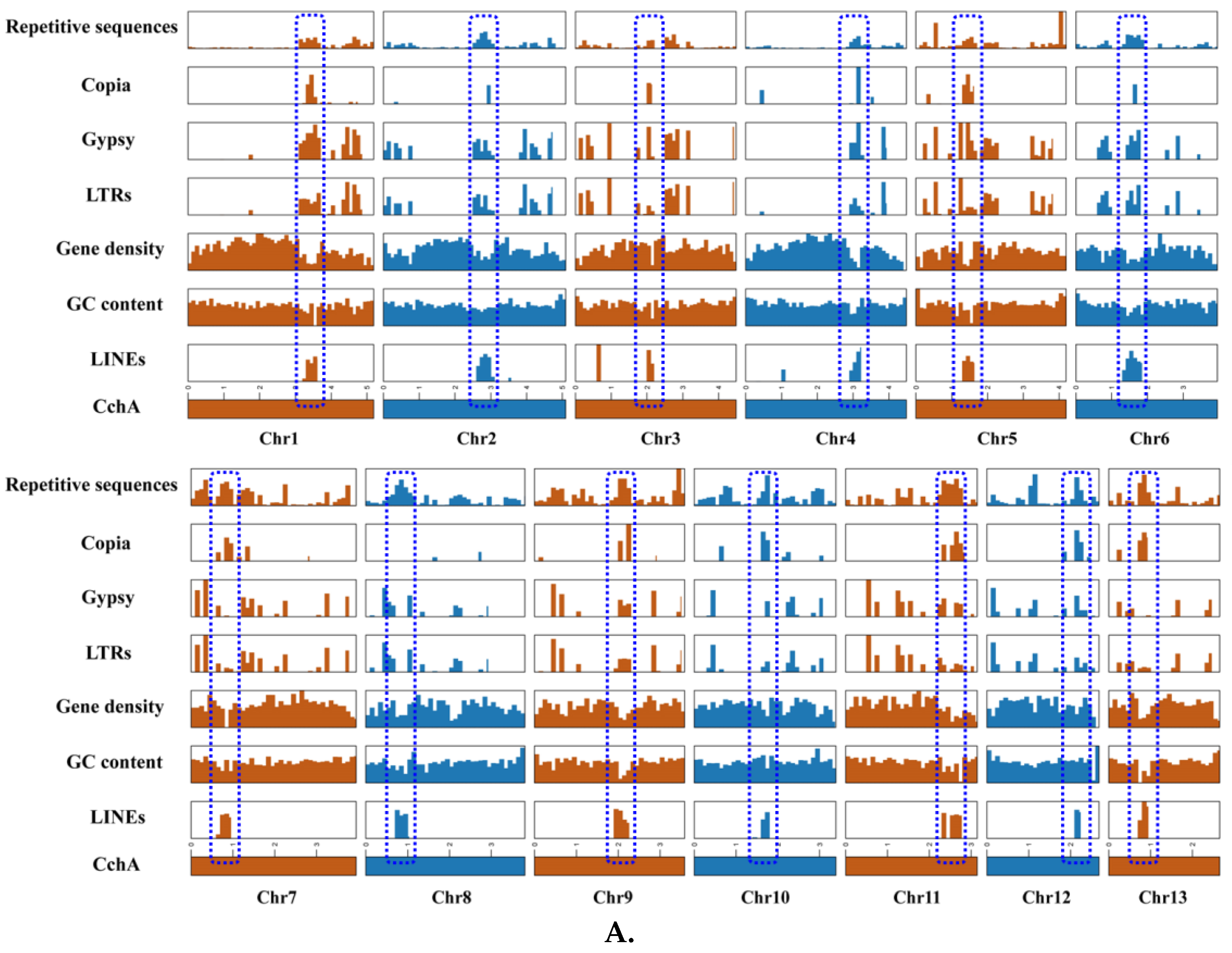
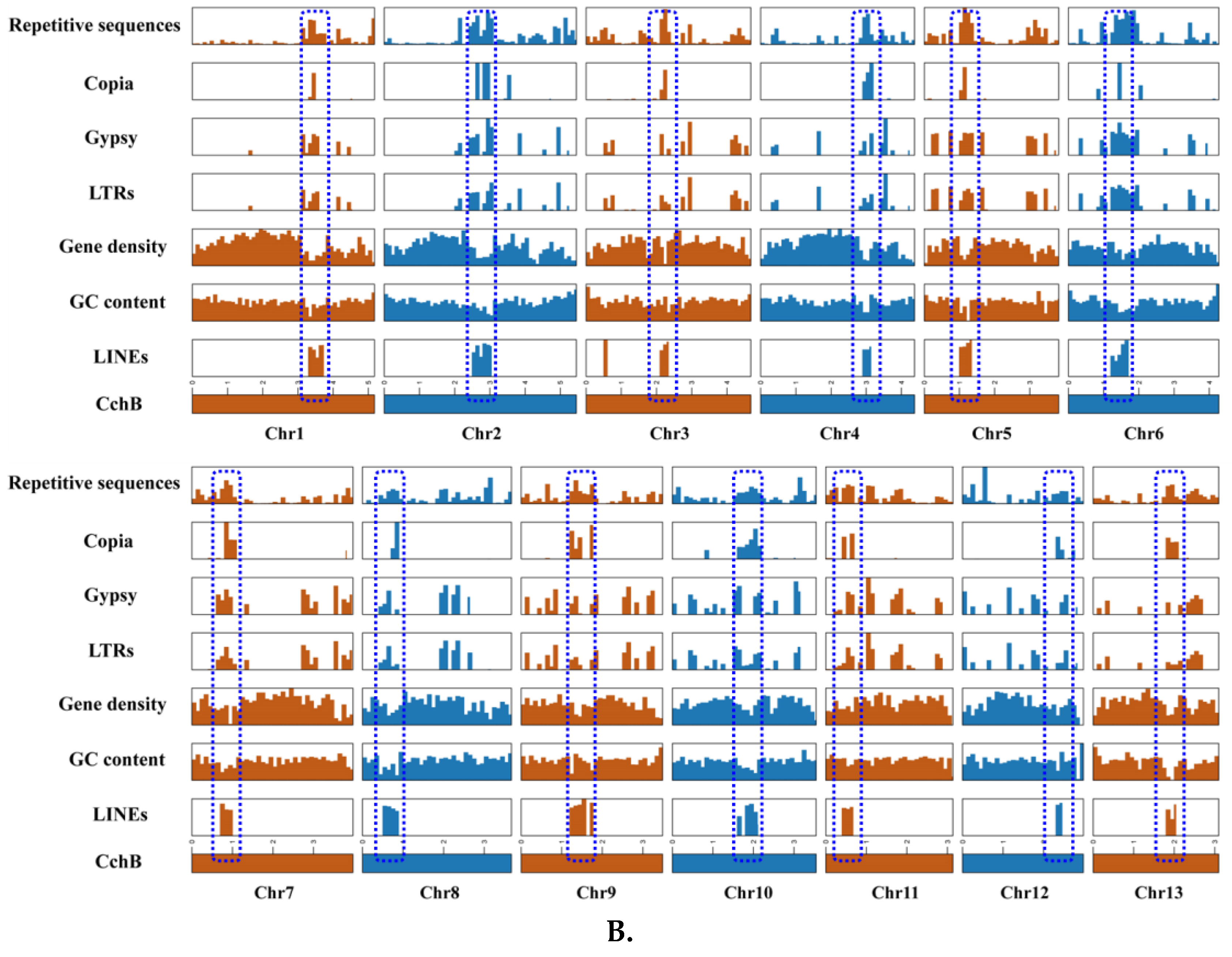
LINEs were highly concentrated in a certain position across all chromosomes of CchA and CchB, primarily spanning a range of 100 to 600 Kb (Figure 3A,B), with minimal presence in other regions. In these regions, both GC content and gene density were relatively low. The Copia LTRs were mainly concentrated in the same regions where LINEs were prevalent. The regions with low GC content, low gene density, and high repetitive sequence content (especially concentrated LINEs) might have represented centromeres (Figure 2 and Figure 3). The predicted centromere regions in the genome of strain CchA were confirmed in its Hi-C heatmap (Figure S2C). These characteristics exhibited some similarities with the putative centromeric regions of Tricholoma matsutake (where LINE-rich regions were GC-rich) [73].
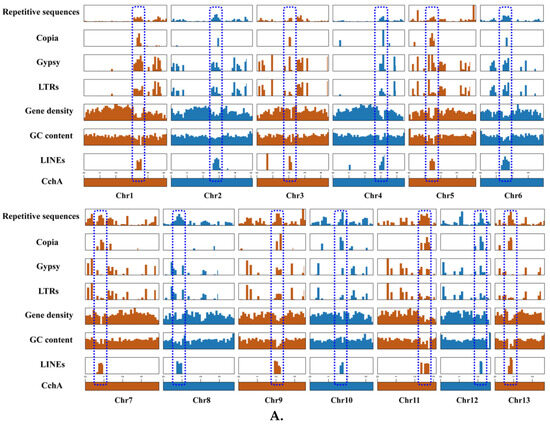
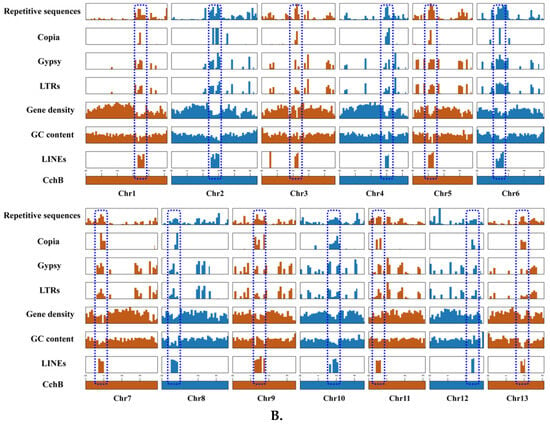
Figure 3.
Features of the major repetitive sequences, GC content (within a 10 kb window), and gene density (within a 100 kb window) in the Agrocybe chaxingu CchA (A) and CchB (B) genomes. The blue dashed box represents the predicted centromere region.
For non-coding RNA (Table S4), 0.0686% (0.03 Mb) and 0.0719% (0.04 Mb) were identified in the genomes of strains CchA and CchB, respectively.
The assembled T2T genomes of strains CchA and CchB were predicted to contain 14,376 and 14,207 protein-coding genes, respectively, and 3.05% (438) and 3.00% (427) CAZyme genes were identified in each strain (Tables S5 and S6).
3.3. Analysis of Mating-Type Genes
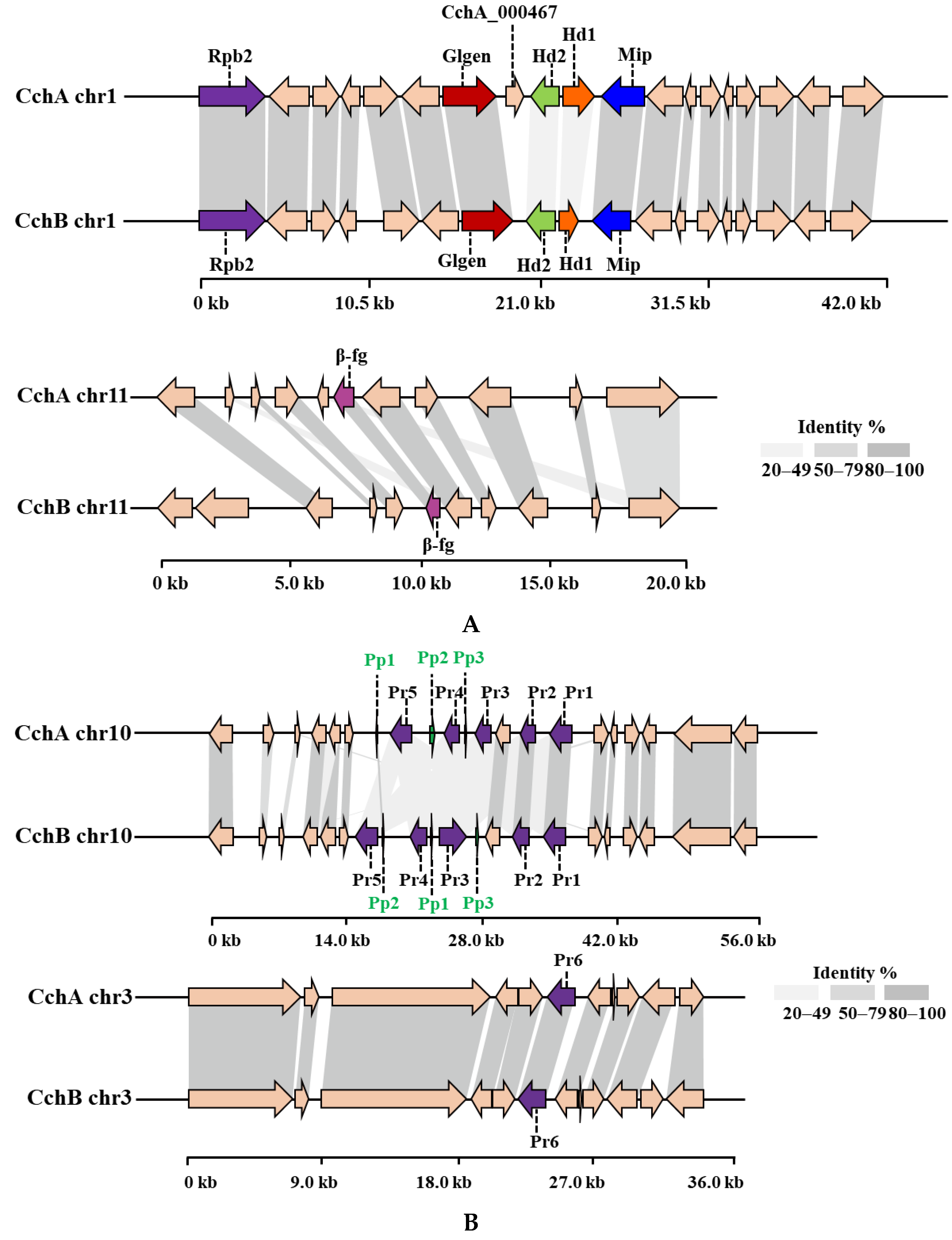
By combining homology-based search and gene function annotation results, the Mip gene was identified. Subsequently, two reverse-oriented genes encoding homeodomain (Hd) proteins adjacent to the Mip gene in the CchA and CchB genomes were detected both on chr1 (Figure 4A). In S. commune [45] and C. cinerea [74,75], multiple Hd genes constituted the two sub-loci of MatA, designated MatAα and MatAβ. In species such as Agaricus bisporus [76], Lentinula edodes [77], Pleurotus ostreatus [78], and Flammulina filiformis [79,80], additional genes containing Hd domain were found located outside the MatA locus. There were no other Hd1 and Hd2 domains identified apart from the MatA locus in the two A. chaxingu strains. On the other side of the Hd gene, a highly conserved glycosyltransferase family 8 gene (Glgen) was identified. No β-flanking (β-fg) genes were found in the regions upstream or downstream of the MatA locus in the CchA and CchB genomes. However, they were identified on the chr11 of CchA and CchB genomes, indicating that β-fg genes are no longer directly linked with the MatA locus in A. chaxingu. A similar phenomenon has been observed in F. filiformis [80] and Sparassis latifolia [81].
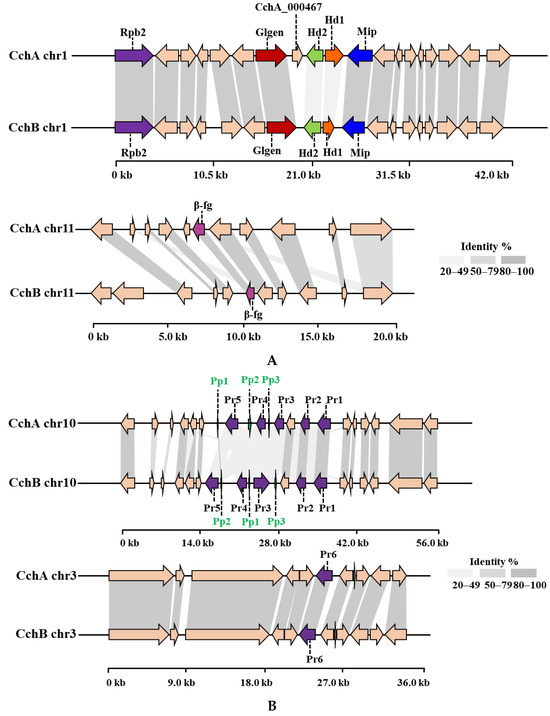
Figure 4.
The protein synteny around the MatA (A) and MatB (B) loci of CchA and CchB. (A) Hd: Homeodomain, Mip: mitochondrial intermediate peptidase, Glgen: glycosyltransferase family 8 protein, Rpb2: RNA polymerase II, the second large subunit, β-fg: β-flanking. (B) Pr: pheromone receptor, Pp: pheromone precursor.
A comparative synteny analysis of the MatA region among several major edible fungi with annotated genomes revealed that over 70% of the genes in the MatA region were conserved, although they exhibited some degree of variation in their relative positions (Figure S3). Furthermore, it was demonstrated that in A. bisporus H97, P. ostreatus PC9, and P. eryngii ATCC90797, the MatA region was surrounded by Mip genes and β-fg genes. In L. edodes L808-1, the Mip gene and β-fg gene in the MatA region were found to be located on the same side of the chromosome. However, in the C. cylindracea species complex, including A. chaxingu and C. aegerita, neither the MatA region nor the entire chromosome exhibited the presence of β-fg genes.
Through a comparative analysis of the collinearity of MatA in the CchA and CchB genomes, it was observed that the similarity of Hd1 proteins with the two genomes was 32.77%, and that of the Hd2 proteins was 35.87%. Additionally, there was no similarity in the intergenic regions between Hd1 and Hd2 genes. The similarity of most other proteins near the MatA locus exceeded 95% between the two genomes. On the chr11 of the CchA and CchB genomes, the similarity of β-fg proteins was 99.04%.
Compared to CchB, the MatA locus in the CchA genome contains an additional gene, CchA_000467, located between the Hd2 and Glgen genes. The existence of the CchA_000467 gene was supported by 122 reads in the original HiFi sequences and was verified through PCR amplification (Figure S4). After conducting a homology search for the CchA_000467 gene, it was found to have similarity to a nuclear receptor coregulator, suggesting that CchA_000467 might have been involved in the process of co-activating or repressing effective transcriptional regulation by nuclear receptors [82].
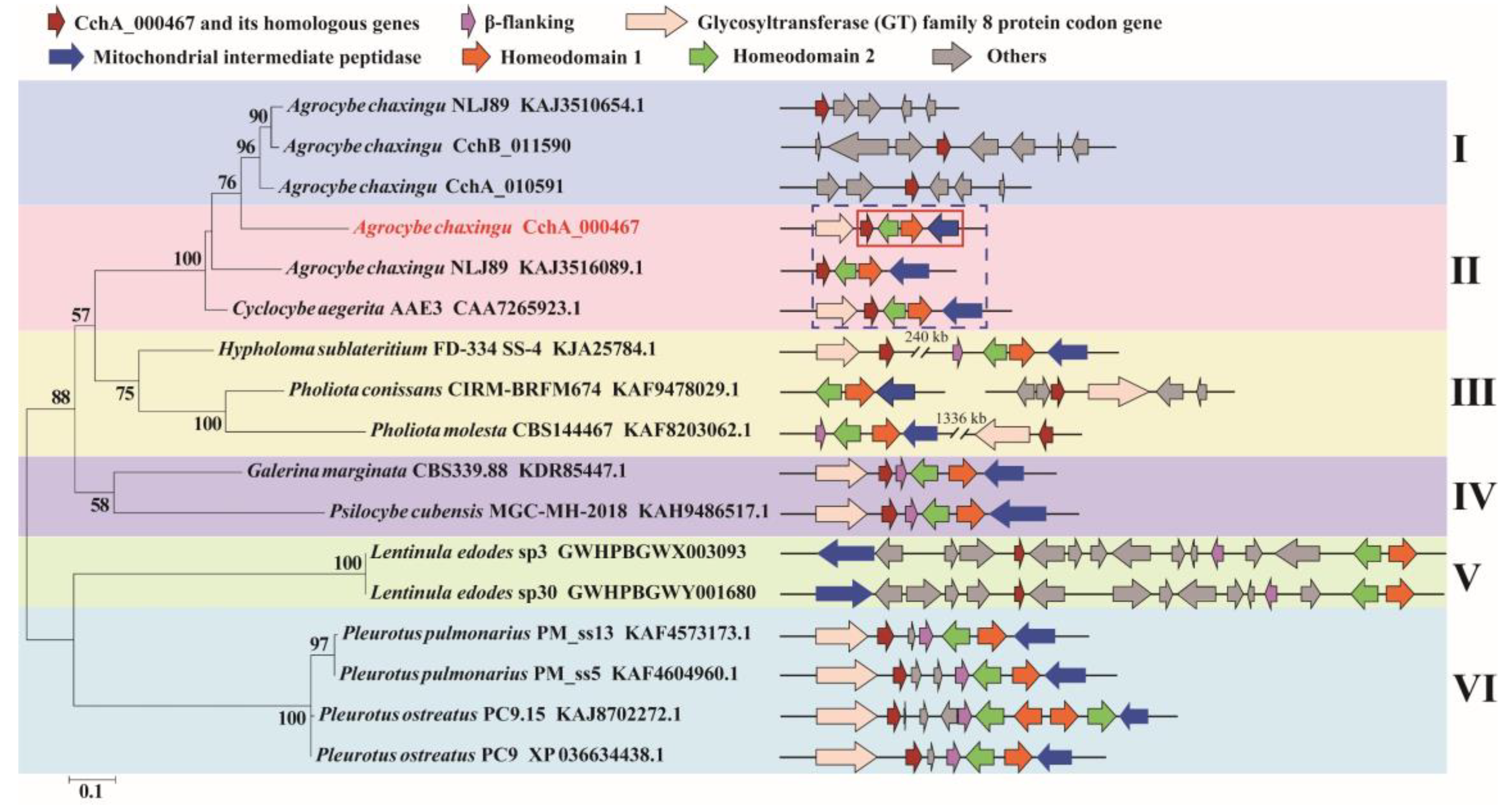
Further analysis revealed that there was a CchA_000467 homologous gene on chr9 in the genomes of both CchA (CchA_010591) and CchB (CchB_011590). Two proteins, CchA_000467 and CchA_010591, exhibited a 68.74% similarity in strain CchA. Subsequently, we investigated the presence of CchA_000467 in other A. chaxingu strains. It was discovered that similar to strain CchA, haploid strain A. chaxingu NLJ89 harbored two proteins in its genome: one located adjacent to Hd2 and the other situated on a different chromosome. Conversely, only one homologous gene of CchA_000467 was found in close proximity to Hd2 in C. aegerita AAE3.
The homologous proteins of CchA_000467 were also found in the genomes of other basidiomycetes (Table S7). The position of these genes was observed to include a considerable distance from the MatA locus, adjacent from the MatA locus, a separation from the MatA locus by multiple additional genes, and a separation from the MatA locus by one or two genes. The occurrence of CchA_000467 or its homologs close to Hd2 has thus far been observed exclusively within the C. cylindracea species complex.
Based on the amino acid sequences of CchA_000467 and its homologs from other species (Table S7), a phylogenetic tree was constructed (Figure 5). The five groups were clustered with strong support as clade I and II, clade III, clade IV, clade V, and clade VI. The division of groups aligns with the classification of this gene and its positional relationship with MatA locus.
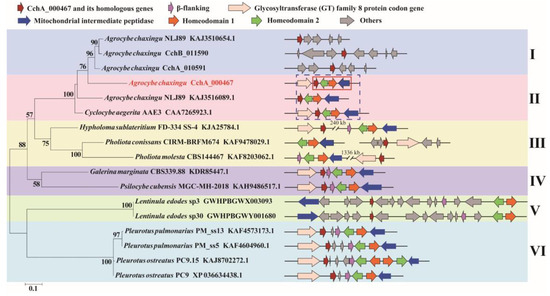
Figure 5.
Phylogenetic analysis based on the amino acid sequences of the CchA_000467 and its homologs from 15 strains of basidiomycetes. The phylogenetic tree was constructed using RAxML (version 8) with 1000 bootstrap replicates. The numbers at the nodes represent the bootstrap percentages. The red fonts indicate the CchA_000467 proteins. The red solid box indicates that the CchA_000467 gene was adjacent to the mating-type A locus. The blue dashed box indicates that in the Cyclocybe cylindracea species complex, the CchA_000467 gene and its homologs were adjacent to the mating-type A locus. The letters Ι, ΙΙ, ΙΙΙ, ΙV, V, and VΙ represent several different types of positional relationships between the CchA_000467 gene and its homologs with the mating-type A locus. In the phylogenetic tree, the text represents species names, strain identifiers, and GenBank numbers from the National Center for Biotechnology Information, excluding strains of CchA and CchB.
In previous studies, the majority of published genome sketches were of a single haploid or diploid, without any accompanying description of the other compatible haploid. In this study, the mating type structure of another haploid, CchB, which is compatible with CchA, was clearly identified, and it was demonstrated that CchB lacks the CchA_000467 gene or its homologs in MatA locus.
Two Pr genes, Pr1 and Pr2, were identified on chr10 in the CchA genome, and homologous genes were also found on chr10 in the CchB genome (Figure 4B). These two Pr proteins in the two genomes showed strong conservation with a similarity of 96.62% and 99.37%, respectively. Three other Pr genes, Pr3, Pr4, and Pr5, on chr10 of the CchA and CchB genome were identified. However, there was low similarity for these proteins (57.90% for Pr3, while other similarities ranged between 20% and 50%). In addition, a Pr6 gene was identified on chr3 in both the CchA and CchB genomes with a protein similarity of 99.78%. Each of the six Pr genes in the CchA and CchB genomes has been validated in gene functional annotation. This study identified six Pr genes, with only Pr6 located independently. A Pr gene situated outside the mating locus has also been reported in the genome of S. commune, where four Pr-like genes were found: brl1, brl2, and brl3 are located at the B mating-type locus, while brl4 is positioned separately [83].
Three Pp genes were successfully identified on chr10 of the CchA and CchB genome. Only Pp1 proteins showed a similarity of 88.89% between the two genomes. No similarity was observed among the other Pp proteins, indicating the differences in the MatA and MatB loci between the strains CchA and CchB.
The collinearity changes in genes within the MatB locus were also notably distinct, with significant variations in gene order and quantity observed among different species. In this study, the MatB of A. chaxingu was similar to, but not identical to that of L. edodes [77,84,85] and F. filiformis [79,80]. In L. edodes, the MatB locus is subdivided into two sub-loci, Bα and Bβ, each containing one Pr gene and one or two Pp genes. Additionally, there were two non-mating-type specific Pr genes located 12 kb away from the MatB, and three Pr genes without accompanying Pp genes were also found on the other scaffold [85,86]. In F. filiformis, there are two typical MatB sub-loci spaced 181 kb apart. At positions 442 kb and 138 kb from the sub-locus, two putative Pr genes were found.
Prior to the acquisition of the complete genome sequence, several scholars researched the genetic patterns of the C. cylindracea species complex through monokaryotic crosses [13,87,88]. This study revealed that the mating type genes of the A. chaxingu strain closely resembled those of its related species, C. cylindracea [12] and C. aegerita [89]. However, it was observed that Hd2 in the MatA of A. chaxingu displayed an opposite orientation compared to C. cylindracea. Additionally, there were slight differences in the arrangement and number of MatB genes in comparison to both species. A profound understanding of the molecular genetic structure of mating-type systems, which govern hybridization and sexual reproduction processes, will aid in revealing the regulatory function of mating-type genes in fruiting body development. This comprehension will also help address breeding-related scientific challenges in the edible mushroom industry [90].
3.4. Prediction of Secondary Metabolite Biosynthetic Gene Clusters
The secondary metabolite biosynthetic gene clusters (SM-BGCs) of the CchA and CchB genomes were predicted using antiSMASH version 7.1.0. In the genome of CchA, 33 SM-BGCs were identified, while CchB contained 34 (Table S8). For CchA, there were 17 terpenes, five fungal ribosomally synthesized and post-translationally modified peptides (Fungal-RiPPs), five non-ribosomal peptide synthases (NRPSs), two indoles, one nonribosomal iron-siderophore (NI-siderophore), one type I polyketide synthase (T1PKS), along with two others. On the other hand, in CchB, predictions included 20 terpenes, five NRPSs, four fungal RiPPs, one indole, one NI-siderophore, one T1PKS, and two others. Notably, within CchA, an extra fungal-RiPP-like biosynthetic gene cluster was observed on chr8, and an additional indole biosynthetic gene cluster was found on chr5. Meanwhile, CchB exhibited one supplementary terpene synthase biosynthetic gene cluster on each of chr8, chr9, and chr10.
Obviously, A. chaxingu has significant potential for terpene biosynthesis. Terpenes are a large class of natural products with a highly diverse range of structures, widely used in pharmaceuticals, food, chemicals, and industrial raw materials [91]. In this study, the cluster of CchA_001922, CchA_009732 in strain CchA, and CchB_000247, CchB_009556 in strain CchB were predicted to be responsible for δ-cadinene biosynsis. δ-cadinene is an important sesquiterpene with the ability to fight against the malaria vector Anopheles stephensi [92], inhibit the growth of ovarian cancer cells [93], and treat respiratory infections [94]. Two BGCs in strains CchA and CchB each (cluster of CchA_009358, CchA_013562, CchB_009236, CchB_014124) were predicted for being responsible for the production of armillyl orsellinate 8α-hydroxy-6-protoilludene. This compound represents a crucial precursor for melleolides, a class of compounds with potential applications as fungicides, antimicrobials, and cancer therapeutics [95,96]. Most of the core genes of these above clusters were expressed according to the transcript data. It was indicated that A. chaxingu was able to produce plenty of terpenes. Many terpenoid compounds have been identified in the C. cylindracea species complex. A total of 22 volatile sesquiterpenes [91] were identified in C. aegerita at different growth stages, including δ-cadinene, cadinane-type sesquiterpenoids, and Δ6-protoilludene. These volatile sesquiterpenes were the precursors of numerous important active substances [97,98]. In C. salicacola, over 20 illudin-type sesquiterpenes [99,100,101,102], three aromadendrane-type sesquiterpenes [103], two Fomannosane-type sesquiterpenoids [101], and one bis-sesquiterpene [104] were isolated and identified. These compounds exhibited various antibacterial, antiviral, or antitumor activities [105]. Three protoilludene sesquiterpenes were isolated from C. cylindracea and exhibited significant inhibitory activity against pathogenic bacteria [106,107].
At present, polysaccharides [4] and steroid compounds [108] have been reported to be derived from A. chaxingu. Notably, six out of the eight steroid compounds identified from the genus Agrocybe were derived from the fruiting bodies of A. chaxingu [91]. The discovery of rich biosynthetic gene clusters for terpenes in A. chaxingu highlighted the necessity for further investigation into the structure and bioactivities of these compounds. Additionally, the complete genome of A. chaxingu will provide a crucial molecular foundation for the precise development of natural products and the analysis of metabolic pathways.
3.5. Whole Genome Comparison and Syntenic Analysis
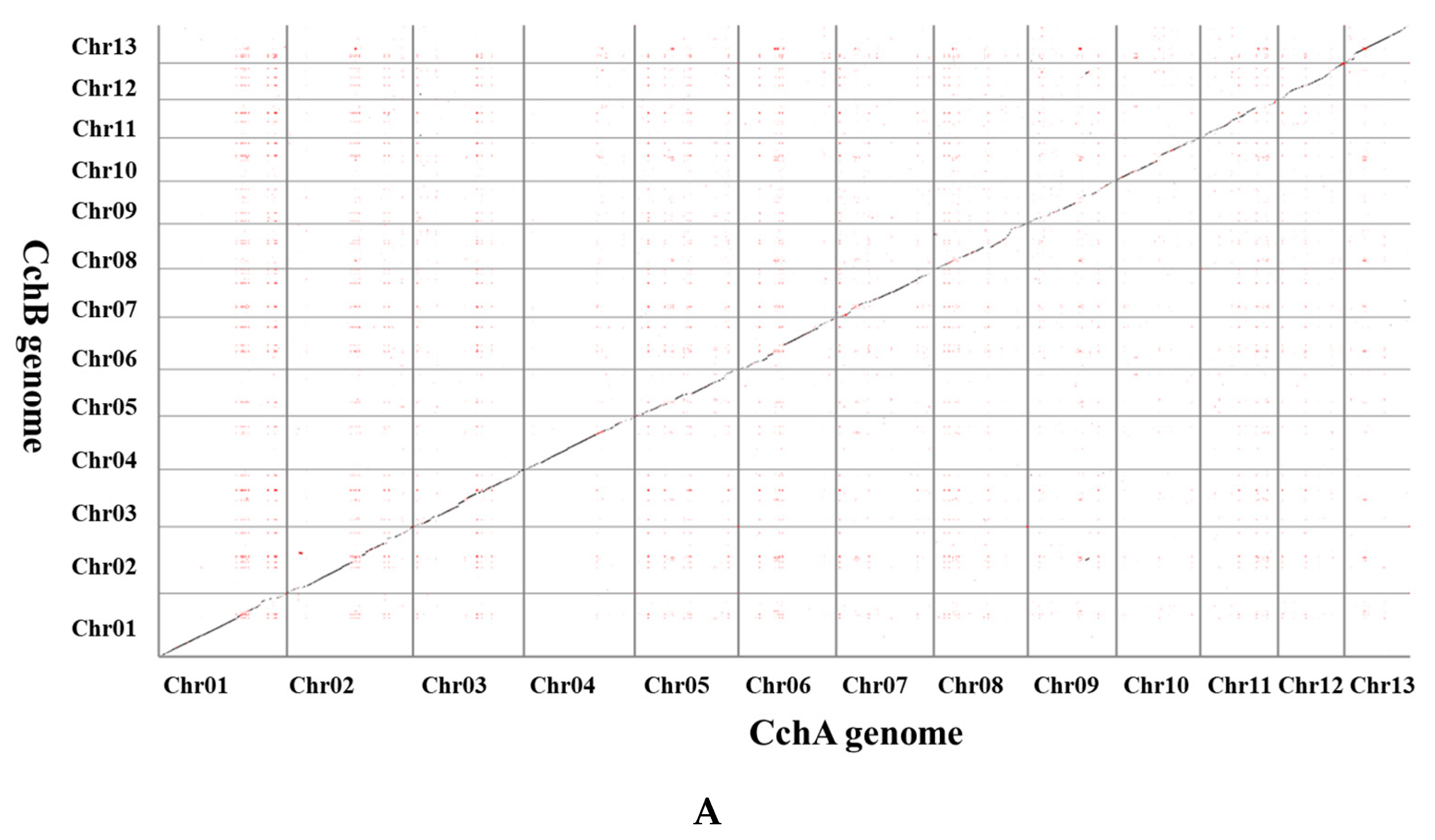
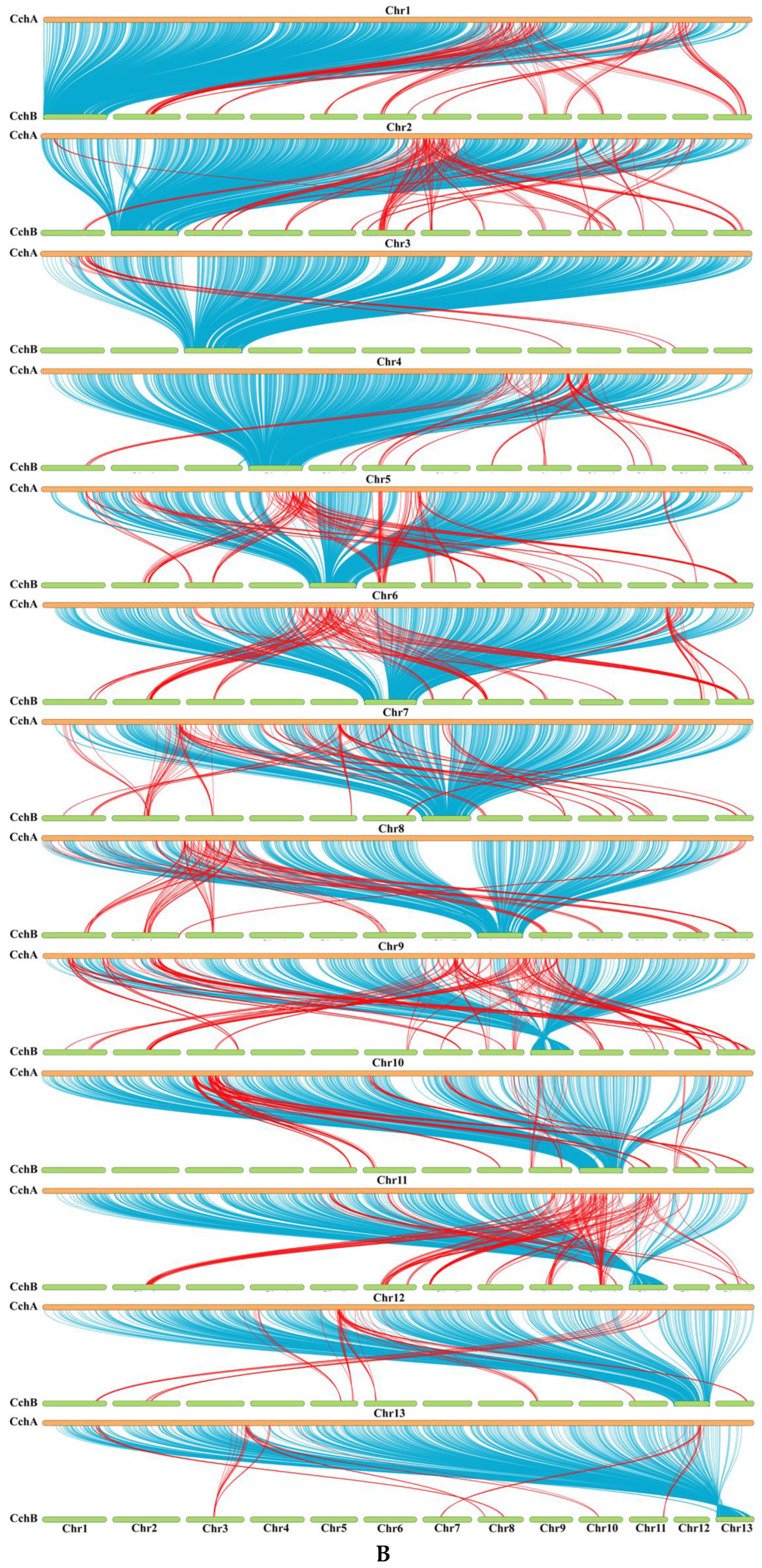
A synteny analysis of the CchA and CchB genomes using MUMmer revealed that these two genomes exhibited a high overall level of synteny, although some minor regions displayed non-synteny (Figure 6A). Further analysis demonstrated that approximately 70.53% of the genes (E value < 1 × 10−5) on CchA exhibited synteny on the homologous chromosomes of CchB (Figure 6B). The highest gene collinearity of 83.09% was observed between chr 4 of the CchA genome and the homologous chromosomes of the CchB genome, while the lowest collinearity of 55.7% occurred on chr 9. Additionally, 858 genes in CchA exhibited collinearity with genes on non-homologous chromosomes in CchB (indicated by red in Figure 6B). It was indicated that the majority of these genes remained relatively stable during evolution, with only a few genes potentially having undergone gene rearrangements. The rearranged genes were found to be significantly enriched in a number of biological processes, including DNA synthesis, cell cycle regulation, antioxidant defense, and cellular protection (Table S9). The rearrangement of genes has been a significant factor in the evolutionary process of individual organisms. This can be observed in the generation of new genes [109], the activation of genes [110], and the provision of valuable resources for phylogenetic studies [111].
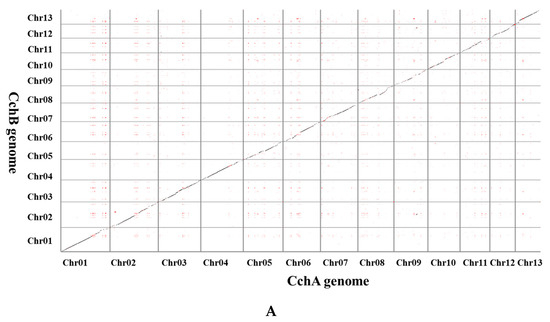
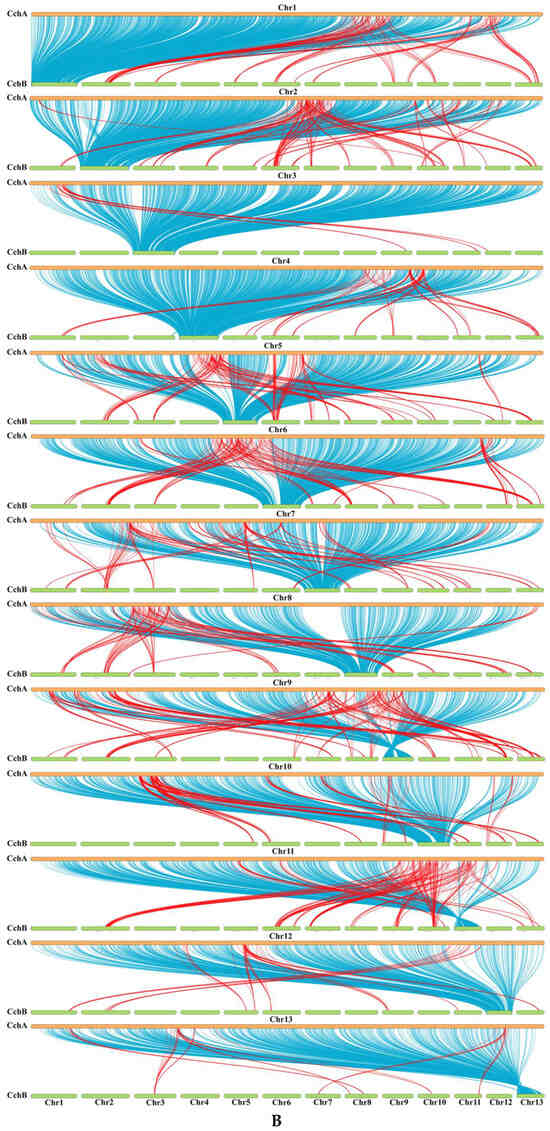
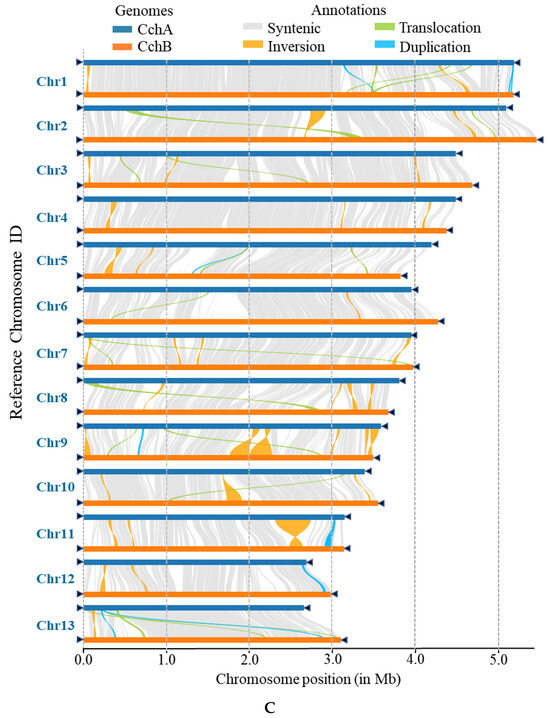
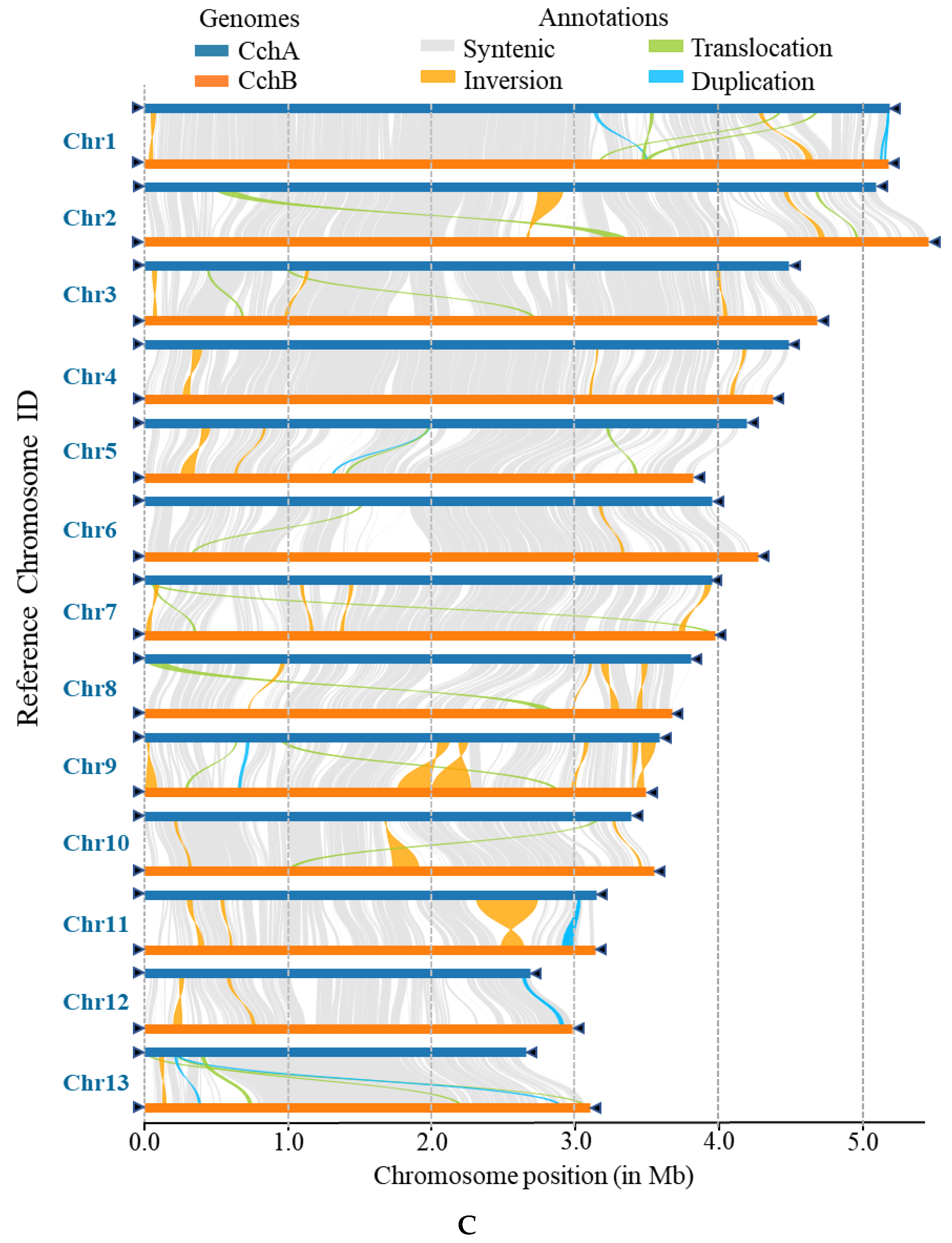
Figure 6.
Whole-genome comparison between the assemblies of CchA and CchB. (A) Dot plot for the syntenic blocks. The black dots represent syntenic regions. The red dots represent the repetitive sequences in the genomes. (B) The gene collinearity between each chromosome of CchA and the 13 chromosomes of CchB. The blue lines represent the gene collinearity between homologous chromosomes. The red lines represent the gene collinearity between non-homologous chromosomes. (C) Chromosome-level local sequence differences. The triangle represents the telomere.
Furthermore, the structural variations between homologous chromosomes of the two genomes were analyzed by SyRI. Structural variations of tens of kilobases in size were identified, including inversions, translocations, and duplications (Figure 6C). The collinear nucleotide sequences between the homologous chromosomes of the CchA and CchB genomes were found to be approximately 33.95 Mb in length, which was consistent with the collinearity ratio of genes. Compared to the CchB genome, the CchA genome exhibited 19 translocations, amounting to 519,232 bp, which accounted for 1.03% of the entire CchA genome. Additionally, there were 36 inversions spanning 1,655,412 bp, constituting 3.27%, as well as 15 duplications totaling 208,799 bp, which accounted for 0.41%. In the genomes of CchA and CchB, the observed rearranged genes and structural variation regions were rich in retrotransposons (Figure 2), so we speculated that these may have been caused by retrotransposons [46,112].
Through comparing the CchA and CchB genes, and considering completely missing genes as unique, the results revealed that there were 613 unique genes for CchA (4.26% of all the genes) and 483 for CchB (3.40% of all the genes). All these unique genes were distributed on each chromosome of both CchA and CchB (Figure 2). The majority of unique genes were classified as hypothetical proteins, representing 88.74% and 82.19% of the unique genes in CchA and CchB, respectively. In contrast, the proportion of hypothetical proteins within the overall genome was relatively low, comprising 17.42% for CchA and 16.61% for CchB. The KEGG enrichment analysis revealed that the unique genes of CchA were significantly enriched in DNA replication and RNA degradation (Table S10), with a total of 32 genes involved in this process. The absence of substantial enrichment in other enrichment analyses may have been attributable to the high proportion of hypothetical proteins present in the unique genes of both CchA and CchB.
Among the unique genes in CchA with annotated functions, seven were annotated as leucine-rich repeat domain superfamily, 11 related to intracellular transport and metabolism, primarily encompassing carbohydrates, lipids, peptides, amino acids, nucleotides, and secondary metabolites. Two unique genes (CchA_005693 and CchA_005699) encoding clitocypin, a fungal cysteine protease inhibitor, were identified in the genome of CchA. The PCR amplification (Figure S5) and sequencing have confirmed that these two genes were genome CchA specific. The function of these two genes is our next project. Among the annotated unique genes in CchB, a total of 36 genes were identified as being primarily involved in maintaining genome stability and the stress response. These included 15 genes involved in DNA replication, recombination and repair, nine genes associated with post-translational modification, five distinct types of zinc finger domains, and seven catalytic proteinase families. Moreover, among the unique genes associated with environmental adaptation and defense, there were 13 genes related to substance transport, nine genes involved in forming defensive cellular scaffolds, and eight genes associated with signal transduction.
Comparative analysis of haploid genomes within the same individual was performed in some species of edible fungi, including L. edodes, Taiwanofungus camphoratus, and Tremella fuciformis. In the compatible monokaryons SP3 and SP30 of L. Edodes [77], approximately 30% of their genomes were non-syntenic, with large-scale segmental rearrangements; their genomes contained 333 (2.91%) and 366 (3.25%) unique genes, respectively, which was lower than in this study. A substantial number of inversions, translocations, and duplications were identified between the genomes of monokaryons Wpm-1 and Wpm-4 of L. Edodes [113]. In T. camphoratus [114], the 2nd and 10th chromosomes of the monokaryon W1 underwent unequal and reciprocal rearrangements in the monokaryon V5, resulting in a longer 2nd chromosome and a shorter 10th chromosome in V5. In the compatible monokaryons A and B of T. fuciformis [115], rearrangements and sequence variations were observed in chromosomes 1, 2, and 5, resulting in the formation of distinct genome structures. The A and B genomes exhibited the presence of 100 (1.18%) and 134 (1.56%) unique genes, respectively. Obviously, high levels of rearrangements were typically observed between chromosomes of monokaryons in edible fungi, and these rearranged gene regions were often rich in retrotransposons [46]. Furthermore, each monokaryon contained its own unique genes, though the proportion of unique genes varied.
3.6. Phylogenomic Analysis and Gene Family Evolution
A phylogenetic analysis was conducted using the genomes of CchA and CchB, along with those of 13 other species from the Agaricales (Table S1). Protein clustering in 15 fungi yielded 1735 single-copy orthologous ones. Maximum likelihood (ML) phylogenetic analysis based on these shared single-copy orthologous proteins produced a super matrix comprising 556,265 amino acid sites. Phylogenomic analysis supported that Agrocybe was not a monophyletic group, which was consistent with a previous study [116]. The two monokaryotic strains of A. chaxingu AS-5 clustered together with A. chaxingu strain GCA_027627235.1 (Figure 7). These three strains formed a group with C. aegerita GCA_902728275.1, and they all belong to the C. cylindracea species complex. Phylogenomic analysis supported A. chaxingu and C. aegerita as distinct species, although they were frequently confused in the market. Additionally, A. pediades, which is also classified within the genus Agrocybe along with A. chaxingu, clustered into a different clade. This result was consistent with the phylogenetic studies using different molecular markers [10,117,118,119].
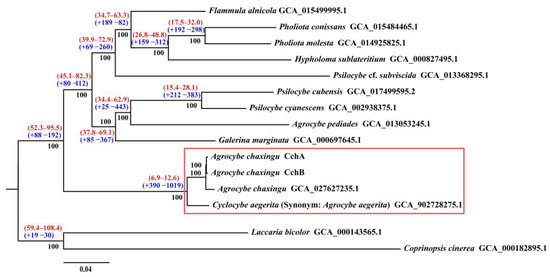
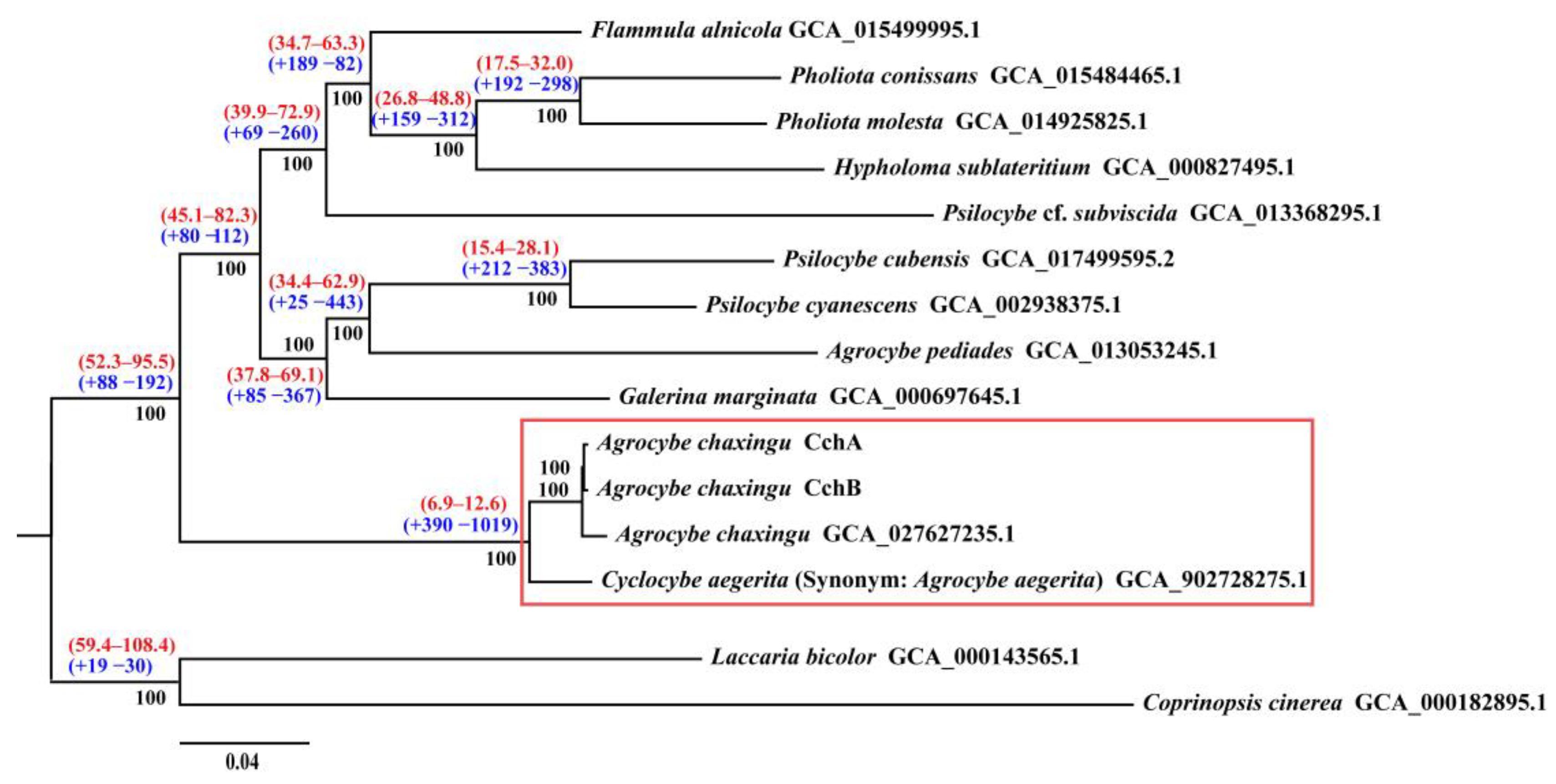
Figure 7.
Phylogenomic analysis among 15 fungal genomes. Bootstrap values are indicated in black, divergence time with 95% CI (confidence interval) in red, and gene family expansion/contraction in blue. The red boxes represent the Cyclocybe cylindracea species complex.
The molecular clock method was used to estimate divergence times, with fossil records of C. cinerea and L. bicolor serving as calibration points [62]. The results revealed that the divergence time between C. cylindracea species complex and other species were estimated to be approximately 6.9–12.6 million years ago (MYA).
Since 2014 when the genus Agrocybe was changed to Cyclocybe [119], the strains in the C. cylindracea species complex have demonstrated diversity in their genus names, resulting in confusion between the use of Agrocybe and Cyclocybe. A. aegerita [11], C. aegerita [10], C. cylindracea [120], and A. cylindracea [12] were all in use. Moreover, the outcomes were suboptimal when straightforward molecular markers were employed to categorize strains within the C. cylindracea species complex. For example, the use of rDNA-ITS to classify species within the genus Agrocybe [117,118] revealed that species such as C. salicacola, C. cylindracea, A. chaxingu, and C. aegerita could not be clearly distinguished. Nevertheless, it was evident that there were discernible genetic variations between the C. cylindracea and A. chaxingu strains, as evidenced by the utilization of three molecular markers: the nuclear ribosomal unit, the mitochondrial SSU-rDNA, and the mitochondrial cob gene [121]. In addition, the use of ITS, the ribosomal RNA large subunit (LSU), the translation elongation factor 1-α gene (TEF1α), and the RNA polymerase II subunit gene (RPB2) as four molecular markers assisted in delineating the European species C. aegerita from a new complex of Asian taxa [10]. The development of multiple molecular markers based on whole-genome sequencing and comparative genomics analysis will facilitate the identification of genetic differences within this species complex [10].
3.7. Gene Expression Analysis
To investigate the gene expression profiles of different nuclei at the stages of mycelia (MY) and fruiting bodies (FB) of the strain A. chaxingu AS-5, RNA-seq was conducted, and a total of 38.5 Gb sequences were generated (Table S11). Using the assembled CchA and CchB genomes as reference genomes, the RNA-seq data was analyzed (Figure 8). At both MY and FB stages, there was no significant difference between the average transcription levels of all genes in CchA and CchB (p > 0.05).
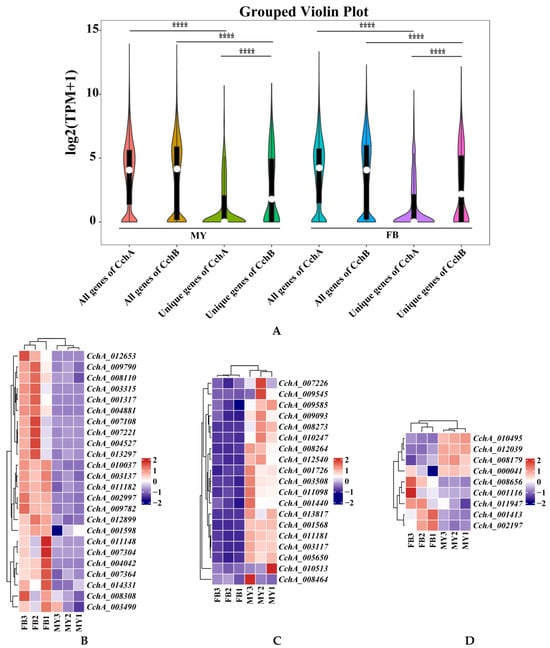
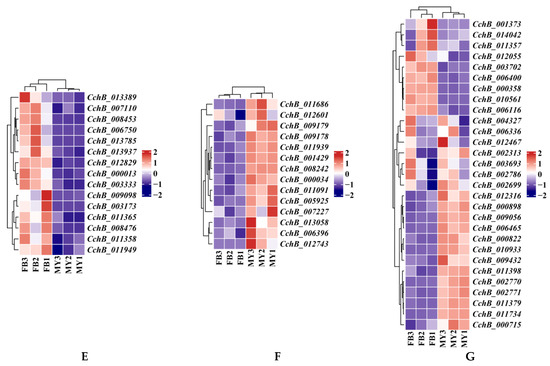
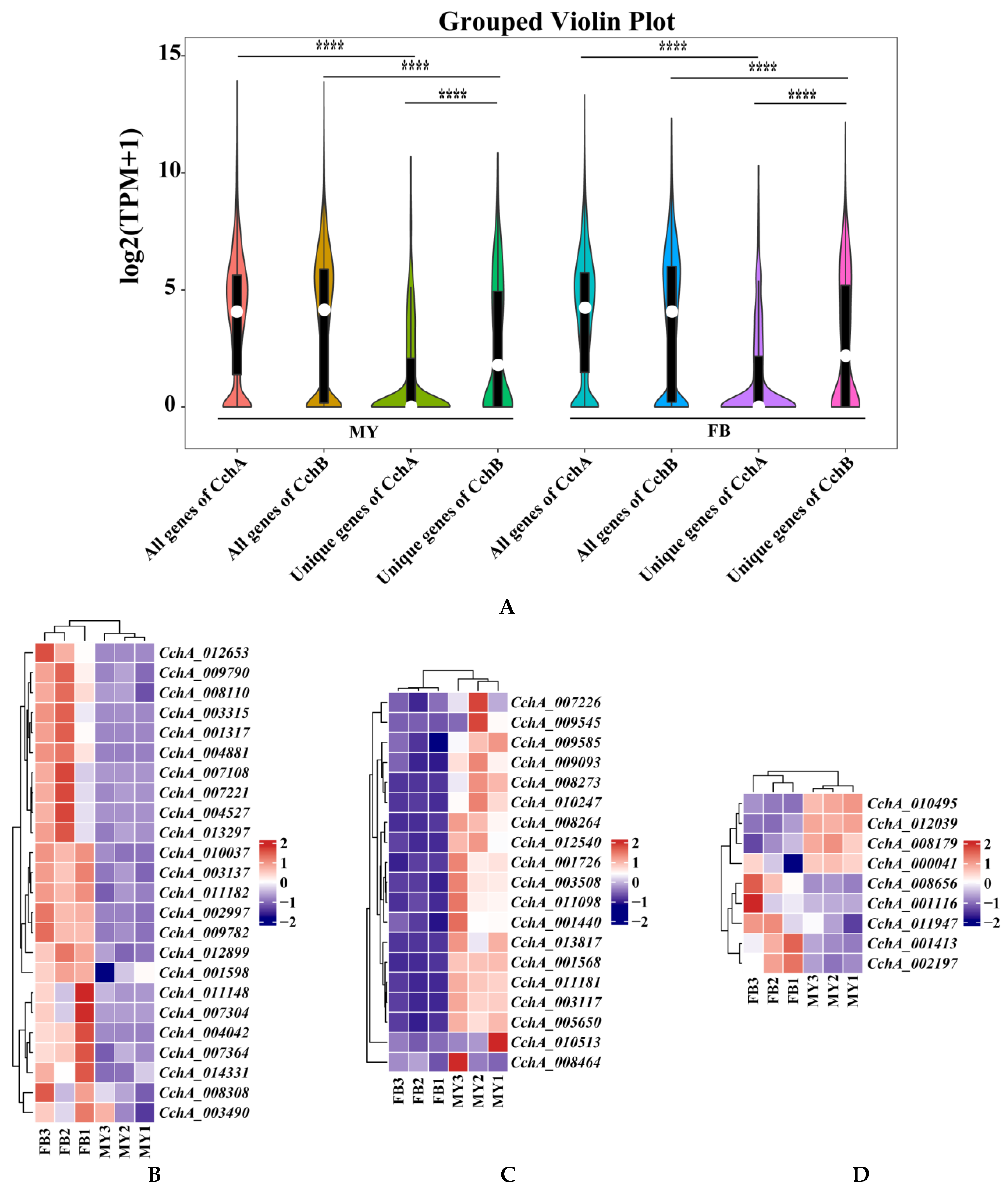
Figure 8.
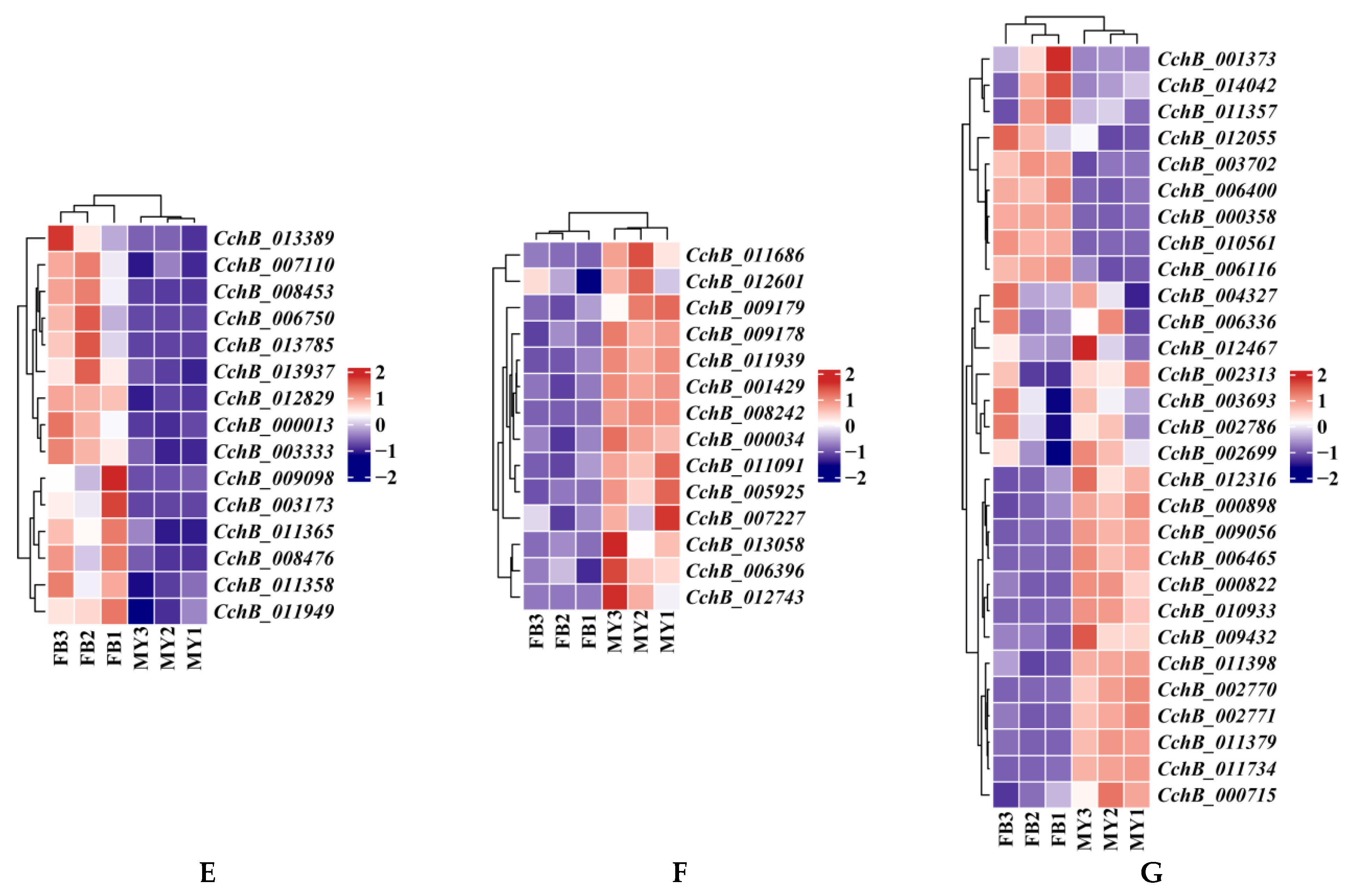
Transcriptome analyses of the strain Agrocybe chaxingu AS-5 between the mycelia and fruiting bodies. (A) Violin plot showing TPM values of the CchA and CchB in MY and FB. MY: mycelia; FB: fruiting bodies. The significance of any differences was assessed by T test. **** represented a significant difference, p < 0.05. (B) Expression of 24 CchA unique genes exclusively at the FB stage. (C) Expression of 19 CchA unique genes exclusively at the MY stage. (D) Nine CchA unique genes expressed at high levels at both stages of MY and FB. (E) Expression of 15 CchB unique genes exclusively at the FB stage. (F) Expression of 14 CchB unique genes exclusively at the MY stage. (G) Twenty-nine CchB unique genes expressed at high levels at both stages of MY and FB.
Furthermore, the expression levels of unique genes in the genomes of CchA and CchB were focused. It was found that the expression levels of all genes at both MY and FB stages were significantly higher than those of unique genes associated with either CchA or CchB (p < 0.05). At both of these stages, the overall expression level of CchB unique genes was significantly higher than that of CchA unique genes (p < 0.05, Figure 8A).
At the MY and FB stages, 66.23% and 65.42%, respectively, of unique genes of CchA were not expressed (TPM < 1 for two of three repetitions), and only 42.65% and 42.44%, respectively, for CchB. Among the 613 unique genes in CchA, 382 of them (62.32%) were not expressed at both stages. These genes may be environmental response genes or genes that are regulated during specific developmental stages, such as spore germination, dormancy, or other developmental stages. A total of 24 unique genes of CchA were expressed exclusively at the FB stage (Figure 8B), 23 of these genes were identified as hypothetical proteins, and one was involved with ribonuclease H-like protein. A total of 19 unique genes were expressed only at the MY stage and not at the FB stage, which may have been related to nutrient absorption, growth, and environmental adaptation of the mycelia (Figure 8C). A total of nine were expressed at high levels (TPM > 100 for two of three repetitions, Figure 8D) at both stages, including CchA_008179, CchA_001116, CchA_001413, CchA_002197, CchA_011947, CchA_008656, CchA_000041, CchA_010495, and CchA_012039. These genes are likely to be associated with fundamental metabolic processes, the synthesis of structural proteins, and other related functions.
Similarly, among the 483 unique genes in CchB, 191 genes (39.54%) were not expressed and 29 genes were expressed at high levels (Figure 8G) at both stages. Additionally, 14 unique genes were expressed only at the MY stage (Figure 8F), and 15 unique genes were expressed only at the FB stage (Figure 8E). The 15 genes associated with fruiting body formation were all hypothetical proteins. Among them, five were unique hypothetical proteins found only in the C. cylindracea species complex, such as CchB_007110, CchB_003173, CchB_011365, CchB_000013, and CchB_003333.
A total of 39 unique genes in CchA and CchB were exclusively expressed at the FB stage (Table S12, Figure 8B,E). The identification of these genes could provide a valuable genetic resource for the study of the fruiting body development mechanism of A. chaxingu.
Consequently, compared to CchA, we found that at both the MY and FB stages, CchB unique genes exhibited higher expression levels and a lower proportion of genes that were not expressed at both stages. This indicated that CchB was likely to have played a more significant role than CchA during the mycelium growth and fruiting body development. Finally, it was demonstrated that CchA and CchB expressed unique genes exclusively during the FB stage, indicating that CchA and CchB required coordinated regulation in the fruiting body development of A. chaxingu.
4. Discussion
This study assembled and compared the T2T genomes of a pair of sexually compatible monokaryons, CchA and CchB, to understand the genetic characteristics of A. chaxingu. Gene transcription at the stages of mycelium and fruiting body were also compared to investigate the genes involved in fruiting body development. The CchA and CchB T2T genomes represented the only high-quality genomes of A. chaxingu available until now. The T2T genome facilitated more accurate identification of structural variations and single nucleotide variations, particularly complex structural variations and variations in repetitive sequences. This was of significant importance for studying genetic diversity, evolutionary mechanisms, and adaptability.
It was found that 70.53% of the genes were collinear between homologous chromosomes in the two genomes, and 858 genes from CchA were rearranged to non-homologous chromosomes in CchB. A comparison of the two monokaryons showed that CchA contained 4.26% unique genes, while CchB had 3.4%. The genetic characteristics of these two sexually compatible monokaryon genomes revealed both similarities and differences when compared to other edible and medicinal fungi. For instance, the collinear gene ratio was similar to that observed in L. edodes [77], but the proportion of unique genes was higher than that identified in L. edodes (2.91% and 3.25%) [77] and T. fuciformis (1.18% and 1.56%) [115]. Furthermore, chromosomal rearrangement phenomena were observed in A. chaxingu, L. edodes [77,113], T. fuciformis [115], and T. camphoratus [114]. This suggested that the two sexually compatible nuclei in dikaryotic edible fungi exhibit distinctive genetic characteristics, primarily reflected in disparate genetic information and chromosomal rearrangements.
The formation of dikaryons is subject to the regulation of mating-type genes, and A. chaxingu displayed distinctive characteristics in its mating-type genes. A difference in the CchA_000467 gene at the MatA locus was identified between the two monokaryons, and the occurrence of CchA_000467 being adjacent to the Hd2 gene was exclusive to the C. cylindracea species complex. Furthermore, the β-fg gene in the A. chaxingu genome was not linked to the MatA locus, contrasting with many other edible fungi.
A comparative analysis of gene expression during the mycelium and fruiting body stages of A. chaxingu revealed that overall gene expression levels were not significantly different between the two monokaryons. However, the overall expression level and proportion of unique genes in CchB were significantly higher than those in CchA. Moreover, CchA and CchB exhibited distinct gene expression patterns during the fruiting body stage. This indicated that A. chaxingu required the coordinated control of both nuclei during fruiting body development, with CchB likely playing a more significant role. A similar phenomenon was observed during the fruiting body development of L. edodes [113], where the two nuclear types coordinated fruiting body development through the dominant expression of different functional genes. Currently, transcriptomics enables the study of dikaryotic edible fungi at various growth and development stages. However, research treating the two nuclei in dikaryons as distinct subjects remains limited [122] In A. bisporus, single-nucleotide polymorphism markers were employed to investigate the expression levels of allelic pairs between the two nuclei of dikaryons, elucidating the dominance-recessiveness relationships and complementary effects between the dikaryotic nuclei [123].
Several scientific issues in the edible mushroom industry require further consideration, such as strain degeneration and maintenance of strain characteristics, as well as the theoretical basis for hybrid breeding. These issues were closely related to the regulation and interaction of the two different types of nuclei in dikaryotic mushroom cells. Although dikaryotic gene expression often mirrors the dominance observed in one of the monokaryons [123], the division of labor between nuclei in dikaryons is crucial. Furthermore, the other nucleus plays a vital role in maintaining the stability of the dikaryon [124] Our findings revealed the unique genetic characteristics and expression profiles between the two sexually compatible monokaryon genomes of A. chaxingu, which is of significant importance for the genetic research.
Therefore, the two monokaryons of A. chaxingu each possess their own independent regulatory systems during dikaryon development, while also allowing for potential mutual regulatory information exchange and additive effects in expression levels. This study has highlighted the distinctive genetic characteristics of the two monokaryons of A. chaxingu and provided insights into fruiting body development.
5. Conclusions
In this study, we employed multiple techniques to sequence the complete genomes of two sexually compatible monokaryons, CchA and CchB, derived from a widely cultivated strain of A. chaxingu AS-5. For the first time, we successfully assembled two gap-free T2T genomes, and analyzed the genetic differences between them, along with their expression levels in growth and development. These high-quality genome assemblies serve as valuable resources for investigating the mechanisms underlying fruiting body development in A. chaxingu. They also open up new avenues for molecular breeding, genetic regulation of important agronomic traits, localization of quantitative trait loci for key genes, and the development of linked molecular markers for specific traits in A. chaxingu.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/jof10090602/s1: Figure S1. The GenomeScope profiles of CchA (A) and CchB (B) based on 21 K-mer; Figure S2. Overview of the A. chaxingu AS-5 assembly genome; Figure S3. Alignment analysis of the mating-type loci A in Agrocybe chaxingu strains CchA and CchB with those of other edible mushrooms; Figure S4. PCR verification of CchA_000467 gene in CchA; Figure S5. PCR verification of CchA_005693 and CchA_005699 unique genes in CchA; Table S1. Information on the 15 species used for constructing the phylogenomic analysis; Table S2. Sequencing data for the genome assembly of CchA and CchB; Table S3. The lengths of the chromosomes in the genomes of CchA and CchB; Table S4. Statistics for non-coding RNA annotation in CchA and CchB; Table S5. Statistics for protein prediction in the genomes of CchA and CchB; Table S6. CAZyme annotation counts for CchA and CchB genomes; Table S7. Information on the 15 strains of basidiomycetes used for constructing the phylogenetic analysis based on the amino acid sequences of CchA_000467 and its homologs; Table S8. The gene cluster comparison of CchA and CchB predicted by antiSMASH; Table S9. Significant GO enrichment results for the rearranged genes of CchA; Table S10. Significant KEGG enrichment results for the unique genes in CchA; Table S11. Summary of the RNA-seq data utilized in this study; Table S12. Unique genes expressed only in the fruiting body stage in CchA and CchB [11,18,46,75,125,126,127,128,129,130,131].
Author Contributions
X.C., G.M., Y.W., C.D. and G.H. conceived and designed the research; X.C. and G.M. performed genome assembly; X.P. and J.D. performed the experiments; X.C., M.W. and G.M. performed the genome analysis and data visualization; X.C. and C.D. wrote the manuscript, which was reviewed by all authors. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Collaborative Innovation Project of Modern Agricultural Research of Jiangxi Province (JXXTCX202409); Project of Science and Technology Department of Jiangxi Province (20212BBF61002, 20212BBF63013); Collaborative Innovation Project of Modern Agricultural Research of Jiangxi Province (JXXTCXBSJJ202213); Crop Seeds Joint Research of Jiangxi Province; and China Agriculture Research System (CARS20).
Institutional Review Board Statement
Not Applicable.
Informed Consent Statement
Not Applicable.
Data Availability Statement
The raw sequences of PacBio Hifi were submitted to NCBI SRA (http://www.ncbi.nlm.nih.gov/sra, accessed on 31 July 2024) under BioProject accession numbers PRJNA1142205.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Liu, Q.M.; Song, H.Y.; Chen, R.X.; Chen, M.H.; Zhai, Z.J.; Zhou, J.P.; Gao, Y.; Hu, D.M. Species concept of Cyclocybe chaxingu, an edible mushroom cultivated in China. Mycosystema 2021, 40, 981–991. [Google Scholar]
- Luo, X.C.; Chen, S.Y. Collection of Mushroom Industry in China, 2nd ed.; Tsinghua University Press: Beijing, China, 2016; pp. 1339–1348. [Google Scholar]
- Lee, B.R.; Kim, S.Y.; Kim, D.W.; An, J.J.; Song, H.Y.; Yoo, K.Y.; Kang, T.C.; Won, M.H.; Lee, K.J.; Kim, K.H.; et al. Agrocybe chaxingu polysaccharide prevent inflammation through the inhibition of COX-2 and NO production. BMB Rep. 2009, 42, 794–799. [Google Scholar] [CrossRef]
- Lee, B.R.; Lee, Y.P.; Kim, D.W.; Song, H.Y.; Yoo, K.Y.; Won, M.H.; Kang, T.C.; Lee, K.J.; Kim, K.H.; Joo, J.H.; et al. Amelioration of streptozotocin-induced diabetes by Agrocybe chaxingu polysaccharide. Mol. Cells 2010, 29, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Zhou, L.W.; Yang, Z.L.; Bau, T.; Li, T.H.; Dai, Y.C. Resource diversity of Chinese macrofungi: Edible, medicinal and poisonous species. Fungal Divers. 2019, 98, 1–76. [Google Scholar]
- Landi, N.; Pacifico, S.; Ragucci, S.; Iglesias, R.; Piccolella, S.; Amici, A.; Di Giuseppe, A.M.A.; Di Maro, A. Purification, characterization and cytotoxicity assessment of Ageritin: The first ribotoxin from the basidiomycete mushroom Agrocybe aegerita. Biochim. Biophys. Acta-Gen. Subj. 2017, 1861, 1113–1121. [Google Scholar] [CrossRef] [PubMed]
- Ragucci, S.; Landi, N.; Russo, R.; Valletta, M.; Pedone, P.V.; Chambery, A.; Di Maro, A. Ageritin from pioppino mushroom: The prototype of ribotoxin-like proteins, a novel family of specific ribonucleases in edible mushrooms. Toxins 2021, 13, 263. [Google Scholar] [CrossRef]
- Ullrich, R.; Nüske, J.; Scheibner, K.; Spantzel, J.; Hofrichter, M. Novel haloperoxidase from the agaric basidiomycete Agrocybe aegerita oxidizes aryl alcohols and aldehydes. Appl. Environ. Microbiol. 2004, 70, 4575–4581. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, L.M.; Rosenthal, K.; Lütz, S. Recent advances in heme biocatalysis engineering. Biotechnol. Bioeng. 2019, 116, 3469–3475. [Google Scholar] [CrossRef]
- Frings, R.A.; Maciá-Vicente, J.G.; Buße, S.; Čmoková, A.; Kellner, H.; Hofrichter, M.; Hennicke, F. Multilocus phylogeny-and fruiting feature-assisted delimitation of European Cyclocybe aegerita from a new Asian species complex and related species. Mycol. Prog. 2020, 19, 1001–1016. [Google Scholar] [CrossRef]
- Gupta, D.K.; Rühl, M.; Mishra, B.; Kleofas, V.; Hofrichter, M.; Herzog, R.; Pecyna, M.J.; Sharma, R.; Kellner, H.; Hennicke, F.; et al. The genome sequence of the commercially cultivated mushroom Agrocybe aegerita reveals a conserved repertoire of fruiting-related genes and a versatile suite of biopolymer-degrading enzymes. BMC Genom. 2018, 19, 48. [Google Scholar] [CrossRef]
- Liang, Y.; Lu, D.X.; Wang, S.; Zhao, Y.H.; Gao, S.H.; Han, R.B.; Yu, J.; Zheng, W.L.; Geng, J.N.; Hu, S.N. Genome assembly and pathway analysis of edible mushroom Agrocybe cylindracea. Genom. Proteom. Bioinform. 2020, 18, 341–351. [Google Scholar] [CrossRef]
- Cai, Y.S.; Fu, J.S.; Luo, F.L.; Ke, L.N.; Huang, X.Z. Studies on the inheritance and breeding technique of Agrocybe chaxingu Huang. Edible Fungi China 2007, 26, 17–20. [Google Scholar]
- Wang, H.X.; Zhang, L.J.; Sun, P.; Hu, J.; Chen, X.T.; Wei, Y.H. Protoplast regeneration and monokaryotation characteristics of Cyclocybe aegerita. Microbiol. China 2022, 49, 2076–2087. [Google Scholar]
- Marcais, G.; Kingsford, C. A fast, lock-free approach for efficient parallel counting of occurrences of k-mers. Bioinformatics 2011, 27, 764–770. [Google Scholar] [CrossRef]
- Vurture, G.W.; Sedlazeck, F.J.; Nattestad, M.; Underwood, C.J.; Fang, H.; Gurtowski, J.; Schatz, M.C. GenomeScope: Fast reference-free genome profiling from short reads. Bioinformatics 2017, 33, 2202–2204. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Concepcion, G.T.; Feng, X.W.; Zhang, H.W.; Li, H. Haplotype-resolved de novo assembly using phased assembly graphs with hifiasm. Nat. Methods 2021, 18, 170–175. [Google Scholar] [CrossRef]
- Walker, B.J.; Abeel, T.; Shea, T.; Priest, M.; Abouelliel, A.; Sakthikumar, S.; Cuomo, C.A.; Zeng, Q.D.; Wortman, J.; Young, S.K.; et al. Pilon: An integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS ONE 2014, 9, e112963. [Google Scholar] [CrossRef]
- Vaser, R.; Sović, I.; Nagarajan, N.; Šikić, M. Fast and accurate de novo genome assembly from long uncorrected reads. Genome Res. 2017, 27, 737–746. [Google Scholar] [CrossRef]
- Ou, S.J.; Chen, J.F.; Jiang, N. Assessing genome assembly quality using the LTR Assembly Index (LAI). Nucleic Acids Res. 2018, 46, e126. [Google Scholar] [CrossRef]
- Simão, F.A.; Waterhouse, R.M.; Ioannidis, P.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO: Assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 2015, 31, 3210–3212. [Google Scholar] [CrossRef] [PubMed]
- Stanke, M.; Schöffmann, O.; Morgenstern, B.; Waack, S. Gene prediction in eukaryotes with a generalized hidden Markov model that uses hints from external sources. BMC Bioinform. 2006, 7, 62. [Google Scholar] [CrossRef] [PubMed]
- Keilwagen, J.; Hartung, F.; Grau, J. GeMoMa: Homology-based gene prediction utilizing intron position conservation and RNA-seq data. Methods Mol. Biol. 2019, 1962, 161–177. [Google Scholar] [PubMed]
- Avram, O.; Kigel, A.; Vaisman-Mentesh, A.; Kligsberg, S.; Rosenstein, S.; Dror, Y.; Pupko, T.; Wine, Y. PASA: Proteomic analysis of serum antibodies web server. PLoS Comput. Biol. 2021, 17, e1008607. [Google Scholar] [CrossRef]
- Haas, B.J.; Salzberg, S.L.; Zhu, W.; Pertea, M.; Allen, J.E.; Orvis, J.; White, O.; Buell, C.R.; Wortman, J.R. Automated eukaryotic gene structure annotation using EVidenceModeler and the program to assemble spliced alignments. Genome Biol. 2008, 9, R7. [Google Scholar] [CrossRef]
- Lagesen, K.; Hallin, P.; Rødland, E.A.; Stærfeldt, H.-H.; Rognes, T.; Ussery, D.W. RNAmmer: Consistent and rapid annotation of ribosomal RNA genes. Nucleic Acids Res. 2007, 35, 3100–3108. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, F.; Allen, J.E.; Allen, J.; Alvarez-Jarreta, J.; Amode, M.R.; Armean, I.M.; Austine-Orimoloye, O.; Azov, A.G.; Barnes, I.; Bennett, R.; et al. Ensembl 2022. Nucleic Acids Res. 2022, 50, D988–D995. [Google Scholar] [CrossRef] [PubMed]
- Lowe, T.M.; Eddy, S.R. tRNAscan-SE: A program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res. 1997, 25, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Nawrocki, E.P.; Kolbe, D.L.; Eddy, S.R. Infernal 1.0: Inference of RNA alignments. Bioinformatics 2009, 25, 1335–1337. [Google Scholar] [CrossRef] [PubMed]
- Kalvari, I.; Nawrocki, E.P.; Argasinska, J.; Quinones-Olvera, N.; Finn, R.D.; Bateman, A.; Petrov, A.I. Non-coding RNA analysis using the Rfam database. Curr. Protoc. Bioinform. 2018, 62, e51. [Google Scholar] [CrossRef]
- Flynn, J.M.; Hubley, R.; Goubert, C.; Rosen, J.; Clark, A.G.; Feschotte, C.; Smit, A.F. RepeatModeler2 for automated genomic discovery of transposable element families. Proc. Natl. Acad. Sci. USA 2020, 117, 9451–9457. [Google Scholar] [CrossRef]
- Tarailo-Graovac, M.; Chen, N.S. Using RepeatMasker to identify repetitive elements in genomic sequences. Curr. Protoc. Bioinform. 2009, 25, 4.10.1–4.10.14. [Google Scholar] [CrossRef]
- Jurka, J.; Kapitonov, V.V.; Pavlicek, A.; Klonowski, P.; Kohany, O.; Walichiewicz, J. Repbase Update, a database of eukaryotic repetitive elements. Cytogenet. Genome Res. 2005, 110, 462–467. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, H. LTR_FINDER: An efficient tool for the prediction of full-length LTR retrotransposons. Nucleic Acids Res. 2007, 35, 265–268. [Google Scholar] [CrossRef]
- Ellinghaus, D.; Kurtz, S.; Willhoeft, U. LTRharvest, an efficient and flexible software for de novo detection of LTR retrotransposons. BMC Bioinform. 2008, 9, 18. [Google Scholar] [CrossRef]
- Yan, H.D.; Bombarely, A.; Li, S. DeepTE: A computational method for de novo classification of transposons with convolutional neural network. Bioinformatics 2020, 36, 4269–4275. [Google Scholar] [CrossRef]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 421. [Google Scholar] [CrossRef]
- Finn, R.D.; Tate, J.; Mistry, J.; Coggill, P.C.; Sammut, S.J.; Hotz, H.R.; Ceric, G.; Forslund, K.; Eddy, S.R.; Sonnhammer, E.L.L.; et al. The Pfam protein families database. Nucleic Acids Res. 2007, 36, D281–D288. [Google Scholar] [CrossRef]
- Paysan-Lafosse, T.; Blum, M.; Chuguransky, S.; Grego, T.; Pinto, B.L.; Salazar, G.A.; Bileschi, M.L.; Bork, P.; Bridge, A.; Colwell, L.; et al. InterPro in 2022. Nucleic Acids Res. 2023, 51, D418–D427. [Google Scholar] [CrossRef]
- Koonin, E.V. The Clusters of Orthologous Groups (COGs) Database: Phylogenetic classification of proteins from complete genomes. NCBI Handb. 2002. Available online: http://www.biodados.icb.ufmg.br/cromatina/NCBI_selected/COGs_ch21d1.pdf (accessed on 2 September 2023).
- Harris, M.A.; Clark, J.; Ireland, A.; Lomax, J.; Ashburner, M.; Foulger, R.; Eilbeck, K.; Lewis, S.; Marshall, B.; Mungall, C.; et al. The Gene Ontology (GO) database and informatics resource. Nucleic Acids Res. 2004, 32, D258–D261. [Google Scholar]
- Jensen, L.J.; Julien, P.; Kuhn, M.; von Mering, C.; Muller, J.; Doerks, T.; Bork, P. eggNOG: Automated construction and annotation of orthologous groups of genes. Nucleic Acids Res. 2007, 36, D250–D254. [Google Scholar] [CrossRef]
- Jones, P.; Binns, D.; Chang, H.Y.; Fraser, M.; Li, W.Z.; McAnulla, C.; McWilliam, H.; Maslen, J.; Mitchell, A.; Nuka, G.; et al. InterProScan 5: Genome-scale protein function classification. Bioinformatics 2014, 30, 1236–1240. [Google Scholar] [CrossRef]
- Moriya, Y.; Itoh, M.; Okuda, S.; Yoshizawa, A.C.; Kanehisa, M. KAAS: An automatic genome annotation and pathway reconstruction server. Nucleic Acids Res. 2007, 35, W182–W185. [Google Scholar] [CrossRef] [PubMed]
- Ohm, R.A.; De Jong, J.F.; Lugones, L.G.; Aerts, A.; Kothe, E.; Stajich, J.E.; De Vries, R.P.; Record, E.; Levasseur, A.; Baker, S.E.; et al. Genome sequence of the model mushroom Schizophyllum commune. Nat. Biotechnol. 2010, 28, 957–963. [Google Scholar] [CrossRef]
- Stajich, J.E.; Wilke, S.K.; Ahrén, D.; Au, C.H.; Birren, B.W.; Borodovsky, M.; Burns, C.; Canbäck, B.; Casselton, L.A.; Cheng, C.K.; et al. Insights into evolution of multicellular fungi from the assembled chromosomes of the mushroom Coprinopsis cinerea (Coprinus cinereus). Proc. Natl. Acad. Sci. USA 2010, 107, 11889–11894. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.M.; Chai, H.M.; Yang, W.X.; Zhang, X.L.; Chen, Y.H.; Zhao, Y.C. Characterization of non-coding regions in b mating loci of Agrocybe Salicacola groups: Target sites for B mating type identification. Curr. Microbiol. 2017, 74, 772–778. [Google Scholar] [CrossRef]
- Brown, A.J.; Casselton, L.A. Mating in mushrooms: Increasing the chances but prolonging the affair. Trends Genet. 2001, 17, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Halsall, J.R.; Milner, M.J.; Casselton, L.A. Three subfamilies of pheromone and receptor genes generate multiple B mating specificities in the mushroom Coprinus cinereus. Genetics 2000, 154, 1115–1123. [Google Scholar] [CrossRef]
- James, T.Y.; Liou, S.R.; Vilgalys, R. The genetic structure and diversity of the A and B mating-type genes from the tropical oyster mushroom, Pleurotus djamor. Fungal Genet. Biol. 2004, 41, 813–825. [Google Scholar] [CrossRef]
- Bölker, M.; Kahmann, R. Sexual pheromones and mating responses in fungi. Plant Cell 1993, 5, 1461. [Google Scholar]
- Caldwell, G.A.; Naider, F.; Becker, J.M. Fungal lipopeptide mating pheromones: A model system for the study of protein prenylation. Microbiol. Rev. 1995, 59, 406–422. [Google Scholar] [CrossRef]
- Casselton, L.A.; Olesnicky, N.S. Molecular genetics of mating recognition in basidiomycete fungi. Microbiol. Mol. Biol. Rev. 1998, 62, 55–70. [Google Scholar] [CrossRef]
- Kües, U.; James, T.Y.; Heitman, J. 6 Mating type in basidiomycetes: Unipolar, bipolar, and tetrapolar patterns of sexuality. In Evolution of Fungi and Fungal-Like Organisms; Springer: Berlin/Heidelberg, Germany, 2011; pp. 97–160. [Google Scholar]
- Emms, D.M.; Kelly, S. OrthoFinder: Solving fundamental biases in whole genome comparisons dramatically improves orthogroup inference accuracy. Genome Biol. 2015, 16, 157. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Castresana, J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol. Biol. Evol. 2000, 17, 540–552. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Le, S.; Li, Y.; Hu, F.Q. SeqKit: A cross-platform and ultrafast toolkit for FASTA/Q file manipulation. PLoS ONE 2016, 11, e0163962. [Google Scholar] [CrossRef]
- Kozlov, A.M.; Darriba, D.; Flouri, T.; Morel, B.; Stamatakis, A. RAxML-NG: A fast, scalable and user-friendly tool for maximum likelihood phylogenetic inference. Bioinformatics 2019, 35, 4453–4455. [Google Scholar] [CrossRef]
- Darriba, D.; Taboada, G.L.; Doallo, R.; Posada, D. ProtTest 3: Fast selection of best-fit models of protein evolution. Bioinformatics 2011, 27, 1164–1165. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, M.J. r8s: Inferring absolute rates of molecular evolution and divergence times in the absence of a molecular clock. Bioinformatics 2003, 19, 301–302. [Google Scholar] [CrossRef]
- Floudas, D.; Binder, M.; Riley, R.; Barry, K.; Blanchette, R.A.; Henrissat, B.; Martínez, A.T.; Otillar, R.; Spatafora, J.W.; Yadav, J.S.; et al. The Paleozoic origin of enzymatic lignin decomposition reconstructed from 31 fungal genomes. Science 2012, 336, 1715–1719. [Google Scholar] [CrossRef]
- De Bie, T.; Cristianini, N.; Demuth, J.P.; Hahn, M.W. CAFE: A computational tool for the study of gene family evolution. Bioinformatics 2006, 22, 1269–1271. [Google Scholar] [CrossRef]
- Marçais, G.; Delcher, A.L.; Phillippy, A.M.; Coston, R.; Salzberg, S.L.; Zimin, A. MUMmer4: A fast and versatile genome alignment system. PLoS Comput. Biol. 2018, 14, e1005944. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, H.; DeBarry, J.D.; Tan, X.; Li, J.; Wang, X.; Lee, T.-H.; Jin, H.; Marler, B.; Guo, H.; et al. MCScanX: A toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res. 2012, 40, e49. [Google Scholar] [CrossRef] [PubMed]
- Goel, M.; Sun, H.Q.; Jiao, W.B.; Schneeberger, K. SyRI: Finding genomic rearrangements and local sequence differences from whole-genome assemblies. Genome Biol. 2019, 20, 277. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An integrative toolkit developed for interactive analyses of big biological data. Mol. Plant. 2020, 13, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Smyth, G.K.; Shi, W. featureCounts: An efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 2014, 30, 923–930. [Google Scholar] [CrossRef]
- Kim, D.; Paggi, J.M.; Park, C.; Bennett, C.; Salzberg, S.L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 2019, 37, 907–915. [Google Scholar] [CrossRef]
- Rhie, A.; Walenz, B.P.; Koren, S.; Phillippy, A.M. Merqury: Reference-free quality, completeness, and phasing assessment for genome assemblies. Genome Biol. 2020, 21, 245. [Google Scholar] [CrossRef]
- Baduel, P.; Leduque, B.; Ignace, A.; Gy, I.; Gil, J., Jr.; Loudet, O.; Colot, V.; Quadrana, L. Genetic and environmental modulation of transposition shapes the evolutionary potential of Arabidopsis thaliana. Genome Biol. 2021, 22, 138. [Google Scholar] [CrossRef]
- Ladeveze, V.; Aulard, S.; Aulard, N.; Périquet, G.; Lemeunier, F. Hobo transposons causing chromosomal breakpoints. Proc. R. Soc. Lond. B 1998, 265, 1157–1159. [Google Scholar] [CrossRef]
- Kurokochi, H.; Tajima, N.; Sato, M.P.; Yoshitake, K.; Asakawa, S.; Isobe, S.; Shirasawa, K. Telomere-to-telomere genome assembly of matsutake (Tricholoma matsutake). DNA Res. 2023, 30, dsad006. [Google Scholar] [CrossRef]
- Kües, U.; Casselton, L.A. The origin of multiple mating types in mushrooms. J. Cell Sci. 1993, 104, 227–230. [Google Scholar] [CrossRef]
- Pardo, E.H.; O’Shea, S.F.; Casselton, L.A. Multiple versions of the A mating type locus of Coprinus cinereus are generated by three paralogous pairs of multiallelic homeobox genes. Genetics 1996, 144, 87–94. [Google Scholar] [CrossRef]
- Morin, E.; Kohler, A.; Baker, A.R.; Foulongne-Oriol, M.; Lombard, V.; Nagy, L.G.; Ohm, R.A.; Patyshakuliyeva, A.; Brun, A.; Aerts, A.L.; et al. Genome sequence of the button mushroom Agaricus bisporus reveals mechanisms governing adaptation to a humic-rich ecological niche. Proc. Natl. Acad. Sci. USA 2012, 109, 17501–17506. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Yan, D.; Song, S.; Fan, Y.Y.; Wang, S.X.; Liu, Y.; Huang, Y.; Rong, C.B.; Guo, Y.; Zhao, S.; et al. Haplotype-resolved genome analyses reveal genetically distinct nuclei within a commercial cultivar of Lentinula edodes. J. Fungi 2022, 8, 167. [Google Scholar] [CrossRef] [PubMed]
- Kües, U. From two to many: Multiple mating types in basidiomycetes. Fungal Biol. Rev. 2015, 29, 126–166. [Google Scholar] [CrossRef]
- Van Peer, A.F.; Park, S.Y.; Shin, P.G.; Jang, K.Y.; Yoo, Y.B.; Park, Y.J.; Lee, B.M.; Sung, G.H.; James, T.Y.; Kong, W.S. Comparative genomics of the mating-type loci of the mushroom Flammulina velutipes reveals widespread synteny and recent inversions. PLoS ONE 2011, 6, e22249. [Google Scholar] [CrossRef]
- Wang, W.; Lian, L.D.; Xu, P.; Chou, T.S.; Mukhtar, I.; Osakina, A.; Waqas, M.; Chen, B.Z.; Liu, X.R.; Liu, F.; et al. Advances in understanding mating type gene organization in the mushroom forming fungus Flammulina velutipes. G3-Genes Genomes Genet. 2016, 6, 3635–3645. [Google Scholar] [CrossRef]
- Li, Y.Q.; Yang, Y.; Huang, X.H.; Huang, J.; Dong, C.H. Molecular and genetic evidence for a tetrapolar mating system in Sparassis latifolia. Fungal Biol. 2020, 124, 1004–1012. [Google Scholar] [CrossRef]
- McKenna, N.J.; Lanz, R.B.; O’Malley, B.W. Nuclear receptor coregulators: Cellular and molecular biology. Endocr. Rev. 1999, 20, 321–344. [Google Scholar]
- Wirth, S.; Freihorst, D.; Krause, K.; Kothe, E. What role might non-mating receptors play in Schizophyllum commune? J. Fungi 2021, 7, 399. [Google Scholar] [CrossRef]
- Au, C.H.; Wong, M.C.; Bao, D.P.; Zhang, M.Y.; Song, C.Y.; Song, W.H.; Law, P.T.W.; Kües, U.; Kwan, H.S. The genetic structure of the A mating-type locus of Lentinula edodes. Gene 2014, 535, 184–190. [Google Scholar] [CrossRef]
- Chen, L.F.; Gong, Y.H.; Cai, Y.L.; Liu, W.; Zhou, Y.; Xiao, Y.; Xu, Z.Y.; Liu, Y.; Lei, X.Y.; Wang, G.Z.; et al. Genome sequence of the edible cultivated mushroom Lentinula edodes (shiitake) reveals insights into lignocellulose degradation. PLoS ONE 2016, 11, e0160336. [Google Scholar] [CrossRef]
- Wu, L.; Peer, A.V.; Song, W.H.; Wang, H.; Chen, M.J.; Tan, Q.; Song, C.Y.; Zhang, M.Y.; Bao, D.P. Cloning of the Lentinula edodes B mating-type locus and identification of the genetic structure controlling B mating. Gene 2013, 531, 270–278. [Google Scholar] [CrossRef]
- Labarère, J.; Noël, T. Mating type switching in the tetrapolar basidiomycete Agrocybe aegerita. Genetics 1992, 131, 307–319. [Google Scholar] [CrossRef]
- Uhart, M.; Albertó, E. Mating tests in Agrocybe cylindracea sensu lato. Recognition of Agrocybe wrightii as a novel species. Mycol. Prog. 2009, 8, 337–349. [Google Scholar] [CrossRef]
- Qian, R.; Chen, W.M.; Chai, H.M.; Tao, N.; Zhao, Y.C.; Chen, Y.H. B Mating Locus from Agrocybe aegerita: Structural Analysis. Chin. Agric. Sci. Bull. 2021, 37, 14–19. [Google Scholar]
- Bao, D.P. Research progress on the mating-typing locus structures of basidiomycete mushrooms. Mycosystema 2019, 38, 2061–2077. [Google Scholar]
- Zhang, L.Q.; Yan, M.X.; Liu, C.W. A comprehensive review of secondary metabolites from the genus Agrocybe: Biological activities and pharmacological implications. Mycology 2024, 15, 162–179. [Google Scholar] [CrossRef]
- Govindarajan, M.; Rajeswary, M.; Benelli, G. δ-Cadinene, calarene and δ-4-carene from Kadsura heteroclita essential oil as novel larvicides against malaria, dengue and filariasis mosquitoes. Comb. Chem. High Throughput Screen 2016, 19, 565–571. [Google Scholar] [CrossRef]
- Hui, L.M.; Zhao, G.D.; Zhao, J.J. δ-Cadinene inhibits the growth of ovarian cancer cells via caspase-dependent apoptosis and cell cycle arrest. Int. J. Clin. Exp. Pathol. 2015, 8, 6046. [Google Scholar]
- Pérez-López, A.; Cirio, A.T.; Rivas-Galindo, V.M.; Aranda, R.S.; de Torres, N.W. Activity against Streptococcus pneumoniae of the essential oil and δ-cadinene isolated from Schinus molle fruit. J. Essent. Oil Res. 2011, 23, 25–28. [Google Scholar] [CrossRef]
- Engels, B.; Heinig, U.; McElroy, C.; Meusinger, R.; Grothe, T.; Stadler, M.; Jennewein, S. Isolation of a gene cluster from Armillaria gallica for the synthesis of armillyl orsellinate-type sesquiterpenoids. Appl. Microbiol. Biotechnol. 2021, 105, 211–224. [Google Scholar] [CrossRef] [PubMed]
- McElroy, C.; Engels, B.; Meusinger, R.; Jennewein, S. CYPArm2 is a CYP450 monooxygenase with protoilludene 13-hydroxylase activity involved in the biosynthesis of armillyl orsellinate-type sesquiterpenoids. ChemBioChem 2023, 24, e202300179. [Google Scholar] [CrossRef]
- Orban, A.; Hennicke, F.; Rühl, M. Volatilomes of Cyclocybe aegerita during different stages of monokaryotic and dikaryotic fruiting. Biol. Chem. 2020, 401, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Q.; Chen, X.X.; Orban, A.; Shukal, S.; Birk, F.; Too, H.P.; Rühl, M. Agrocybe aegerita serves as a gateway for identifying sesquiterpene biosynthetic enzymes in higher fungi. ACS Chem. Biol. 2020, 15, 1268–1277. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.Y.; Zhang, L.; Feng, T.; Li, Z.H.; Dong, Z.J.; Li, X.Y.; Su, J.; Li, Y.; Liu, J.K. Unusual illudin-type sesquiterpenoids from cultures of Agrocybe salicacola. Nat. Product. Bioprospecting 2011, 1, 87–92. [Google Scholar] [CrossRef]
- Wang, G.; Liu, L.Y.; Zhu, Y.C.; Liu, J.K. Illudin T, a new sesquiterpenoid from basidiomycete Agrocybe salicacola. J. Asian Nat. Prod. Res. 2011, 13, 430–433. [Google Scholar] [CrossRef]
- Liu, L.Y.; Li, Z.H.; Dong, Z.J.; Li, X.Y.; Su, J.; Li, Y.; Liu, J.K. Two novel fomannosane-type sesquiterpenoids from the culture of the basidiomycete Agrocybe salicacola. Nat. Product. Bioprospecting 2012, 2, 130–132. [Google Scholar] [CrossRef]
- Dai, Q.; Zhang, F.L.; Du, J.X.; Li, Z.H.; Feng, T.; Liu, J.K. Illudane sesquiterpenoids from edible mushroom Agrocybe salicacola and their bioactivities. Acs Omega 2020, 5, 21961–21967. [Google Scholar] [CrossRef]
- Zhu, Y.C.; Wang, G.; Liu, J.K. Two new sesquiterpenoids from basidiomycete Agrocybe salicacola. J. Asian Nat. Prod. Res. 2010, 12, 464–469. [Google Scholar] [CrossRef]
- Zhu, Y.C.; Wang, G.; Yang, X.L.; Luo, D.Q.; Zhu, Q.C.; Peng, T.; Liu, J.K. Agrocybone, a novel bis-sesquiterpene with a spirodienone structure from basidiomycete Agrocybe salicacola. Tetrahedron Lett. 2010, 51, 3443–3445. [Google Scholar] [CrossRef]
- Reina, M.; Orihuela, J.C.; González-Coloma, A.; de Inés, C.; de la Cruz, M.; del Val, A.G.; Torno, J.R.; Fraga, B.M. Four illudane sesquiterpenes from Coprinopsis episcopalis. Phytochemistry 2004, 65, 381–385. [Google Scholar] [CrossRef]
- Assante, G.; Dallavalle, S.; Anna Martino, P. Protoilludane sesquiterpenoids as scaffold structures for new antimicrobials against Mannheimia haemolytica. J Antibiot 2013, 66, 43–45. [Google Scholar] [CrossRef]
- Surup, F.; Hennicke, F.; Sella, N.; Stroot, M.; Bernecker, S.; Pfütze, S.; Stadler, M.; Rühl, M. New terpenoids from the fermentation broth of the edible mushroom Cyclocybe aegerita. Beilstein J. Org. Chem. 2019, 15, 1000–1007. [Google Scholar] [CrossRef]
- Kawagishi, H.; Akachi, T.; Ogawa, T.; Masuda, K.; Yamaguchi, K.; Yazawa, K.; Takahashi, M. Chaxine A, an osteoclast-forming suppressing substance, from the mushroom Agrocybe chaxingu. Heterocycles 2007, 69, 253–258. [Google Scholar] [CrossRef]
- Burssed, B.; Zamariolli, M.; Bellucco, F.T.; Melaragno, M.I. Mechanisms of structural chromosomal rearrangement formation. Mol. Cytogenet. 2022, 15, 23. [Google Scholar] [CrossRef]
- Sun, L.H.; Jing, Y.Q.; Liu, X.Y.; Li, Q.; Xue, Z.H.; Cheng, Z.K.; Wang, D.W.; He, H.; Qian, W.Q. Heat stress-induced transposon activation correlates with 3D chromatin organization rearrangement in Arabidopsis. Nat. Commun. 2020, 11, 1886. [Google Scholar] [CrossRef] [PubMed]
- Boore, J.L. The use of genome-level characters for phylogenetic reconstruction. Trends Ecol. Evol. 2006, 21, 439–446. [Google Scholar] [CrossRef]
- Bennetzen, J.L.; Wang, H. The contributions of transposable elements to the structure, function, and evolution of plant genomes. Annu. Rev. Plant Biol. 2014, 65, 505–530. [Google Scholar] [CrossRef]
- Shen, N.; Xie, H.Y.; Liu, K.F.; Li, X.R.; Wang, L.; Deng, Y.J.; Chen, L.F.; Bian, Y.B.; Xiao, Y. Near-gapless genome and transcriptome analyses provide insights into fruiting body development in Lentinula edodes. Int. J. Biol. Macromol. 2024, 130610. [Google Scholar] [CrossRef]
- Chen, C.L.; Li, W.C.; Chuang, Y.C.; Liu, H.C.; Huang, C.H.; Lo, K.Y.; Chen, C.Y.; Chang, F.M.; Chang, G.A.; Lin, Y.L.; et al. Sexual crossing, chromosome-level genome sequences, and comparative genomic analyses for the medicinal mushroom Taiwanofungus camphoratus (Syn. Antrodia cinnamomea, Antrodia camphorata). Microbiol. Spectr. 2022, 10, e02032-21. [Google Scholar]
- Deng, Y.J.; Guo, L.; Lin, L.J.; Li, Y.F.; Zhang, J.X.; Zhang, Y.; Yuan, B.; Ke, L.; Xie, B.G.; Ming, R. Meiosis in an asymmetric dikaryotic genome of Tremella fuciformis Tr01 facilitates new chromosome formation. Genome Biol. 2023, 24, 280. [Google Scholar] [CrossRef]
- Kiyashko, A.; Malysheva, E.; Justo, A.; Malysheva, V. Two new species of Agrocybe (Agaricales, Basidiomycota) from South Siberia, Russia. Nova Hedwig. 2022, 115, 181–203. [Google Scholar] [CrossRef]
- Cheong, J.C.; Lee, M.C.; Jang, K.Y.; Jhune, C.S.; Lee, C.J. Phylogenetic relationships of mushroom Agrocybe spp. based on rDNA-ITS analysis. Korean J. Mycol. 2009, 37, 11–18. [Google Scholar] [CrossRef]
- Zhou, P.Q.; Li, L.L.; Shi, H.G.; Wang, L.; Li, Y. Analysis of Phylogeny of Agrocybe Genus Based on Nucleotide Database in NCBI. bioRxiv 2022. [Google Scholar] [CrossRef]
- Vizzini, A.; Angelini, C.; Ercole, E. Le sezioni velatae e aporus di Agrocybe sottogenere aporus: Rivalutazione del genere Cyclocybe velen. ed un nuova specie. Boll. Della Assoc. Micol. Ecol. Romana 2014, 92, 21–38. [Google Scholar]
- Niveiro, N.; Uhart, M.; Albertó, E. Revision of the genera Agrocybe and Cyclocybe (Strophariaceae, Agaricales, Basidiomycota) in Argentina. Rodriguésia 2020, 71, e02272018. [Google Scholar] [CrossRef]
- Callac, P.; Guinberteau, J.; Férandon, C.; Barroso, G. An Asian commercial strain of Agrocybe chaxingu and a European wild strain of Agrocybe cylindracea exhibiting morphological difference and high genetic divergence are interfertile. In Proceedings of the Seventh International Conference on Mushroom Biology and Mushroom Products ICMBMP7, Arcachon, France, 4–7 October 2011; pp. 113–122. [Google Scholar]
- Liu, T.X.; Li, H.R.; Ding, Y.T.; Qi, Y.C.; Gao, Y.Q.; Song, A.D.; Shen, J.W.; Qiu, L.Y. Genome-wide gene expression patterns in dikaryon of the basidiomycete fungus Pleurotus ostreatus. Braz. J. Microbiol. 2017, 48, 380–390. [Google Scholar] [CrossRef]
- Gehrmann, T.; Pelkmans, J.F.; Ohm, R.A.; Vos, A.M.; Sonnenberg, A.S.; Baars, J.J.; Wösten, H.A.; Reinders, M.J.; Abeel, T. Nucleus-specific expression in the multinuclear mushroom-forming fungus Agaricus bisporus reveals different nuclear regulatory programs. Proc. Natl. Acad. Sci. USA 2018, 115, 4429–4434. [Google Scholar] [CrossRef] [PubMed]
- Auxier, B.; Czárán, T.L.; Aanen, D.K. Modeling the consequences of the dikaryotic life cycle of mushroom-forming fungi on genomic conflict. eLife 2022, 11, e75917. [Google Scholar] [CrossRef]
- Riley, R.; Salamov, A.A.; Brown, D.W.; Nagy, L.G.; Floudas, D.; Held, B.W.; Levasseur, A.; Lombard, V.; Morin, E.; Otillar, R.; et al. Extensive sampling of basidiomycete genomes demonstrates inadequacy of the white-rot/brown-rot paradigm for wood decay fungi. Proc. Natl. Acad. Sci. USA 2014, 111, 9923–9928. [Google Scholar] [CrossRef]
- Kohler, A.; Kuo, A.; Nagy, L.G.; Morin, E.; Barry, K.W.; Buscot, F.; Canbäck, B.; Choi, C.; Cichocki, N.; Clum, A.; et al. Convergent losses of decay mechanisms and rapid turnover of symbiosis genes in mycorrhizal mutualists. Nature Genet. 2015, 47, 410–415. [Google Scholar] [CrossRef]
- McKernan, K.; Kane, L.T.; Crawford, S.; Chin, C.S.; Trippe, A.; McLaughlin, S. A draft reference assembly of the Psilocybe cubensis genome. F1000Research 2021, 10, 281. [Google Scholar] [CrossRef] [PubMed]
- Steindorff, A.S.; Carver, A.; Calhoun, S.; Stillman, K.; Liu, H.; Lipzen, A.; He, G.; Yan, M.; Pangilinan, J.; LaButti, K.; et al. Comparative genomics of pyrophilous fungi reveals a link between fire events and developmental genes. Environ. Microbiol. 2021, 23, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Floudas, D.; Bentzer, J.; Ahrén, D.; Johansson, T.; Persson, P.; Tunlid, A. Uncovering the hidden diversity of litter-decomposition mechanisms in mushroom-forming fungi. ISME J. 2020, 14, 2046–2059. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.Y.; Wang, Z.H.; Song, W.D.; Fan, P.M.; Kang, Y.P.; Lei, Y.; Wan, L.Y.; Huai, D.X.; Chen, Y.N.; Wang, X.; et al. Genome sequencing and comparative genomic analysis of highly and weakly aggressive strains of Sclerotium rolfsii, the causal agent of peanut stem rot. BMC Genomics 2021, 22, 276. [Google Scholar] [CrossRef]
- Martin, F.; Aerts, A.; Ahrén, D.; Brun, A.; Danchin, E.G.J.; Duchaussoy, F.; Gibon, J.; Kohler, A.; Lindquist, E.; Pereda, V.; et al. The genome of Laccaria bicolor provides insights into mycorrhizal symbiosis. Nature 2008, 452, 88–92. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).