Dual Detection of the Chytrid Fungi Batrachochytrium spp. with an Enhanced Environmental DNA Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Collection of Field Samples
2.2. DNA Extraction and qPCR
2.3. gBlock In Vitro Testing
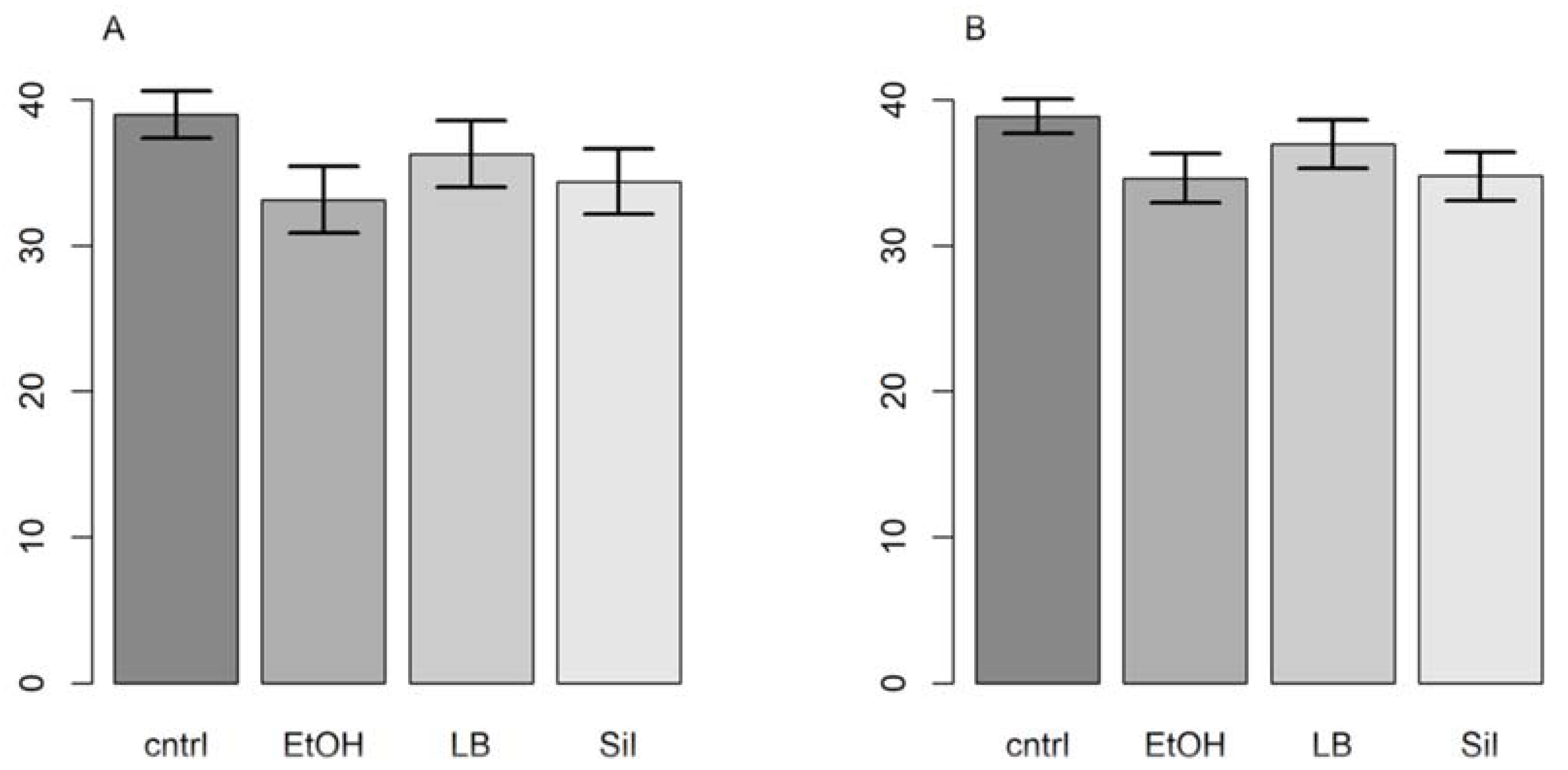
2.4. Storage Experiment for eDNA Samples
2.5. Cost Assessment
2.6. Statistical Analyses
3. Results
3.1. Field Sampling
3.2. gBlock In Vitro Testing
3.3. Storage Experiment for eDNA Samples
3.4. Cost Assessment
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- MacKenzie, D.I.; Kendall, W. How should detection probability be incorporated into estimates of relative abundance? Ecology 2002, 83, 2387–2393. [Google Scholar] [CrossRef]
- Bailey, L.L.; Simons, T.R.; Pollock, K.H. Estimating site occupancy and species detection probability parameters for terrestrial salamanders. Ecol. Appl. 2004, 14, 692–702. [Google Scholar] [CrossRef]
- Thomsen, P.F.; Willerslev, E. Environmental DNA—An emerging tool in conservation for monitoring past and present biodiversity. Biol. Conserv. 2015, 183, 4–18. [Google Scholar] [CrossRef]
- Goldberg, C.S.; Pilliod, D.S.; Arkle, R.S.; Waits, L.P. Molecular Detection of Vertebrates in Stream Water: A Demonstration Using Rocky Mountain Tailed Frogs and Idaho Giant Salamanders. PLoS ONE 2011, 6, e22746. [Google Scholar] [CrossRef] [PubMed]
- Laramie, M.B.; Pilliod, D.S.; Goldberg, C.S. Characterizing the distribution of an endangered salmonid using environmental DNA analysis. Biol. Conserv. 2015, 183, 29–37. [Google Scholar] [CrossRef]
- Ficetola, G.F.; Miaud, C.; Pompanon, F.; Taberlet, P. Species detection using environmental DNA from water samples. Biol. Lett. 2008, 4, 423–425. [Google Scholar] [CrossRef] [PubMed]
- Ahne, W.; Bjorklund, H.V.; Essbauer, S.; Fijan, N.; Kurath, G.; Winton, J.R. Spring viremia of carp (SVC). Dis. Aquat. Org. 2002, 52, 261–272. [Google Scholar] [CrossRef]
- Foster, G.; Malnick, H.; Lawson, P.A.; Kirkwood, J.; MacGregor, S.K.; Collins, M.D. Suttonella ornithocola sp. nov., from birds of the tit families, and emended description of the genus Suttonella. Int. J. Syst. Evol. Microbiol. 2005, 55, 2269–2272. [Google Scholar] [CrossRef]
- Gargas, A.; Trest, M.; Christensen, M.; Volk, T.; Blehert, D. Geomyces destructans sp. nov. associated with bat white-nose syndrome. Mycotaxon 2009, 108, 147–154. [Google Scholar] [CrossRef]
- Allain, S.J.R.; Duffus, A.L.J. Emerging infectious disease threats to European herpetofauna. Herpetol. J. 2019, 29, 189–206. [Google Scholar] [CrossRef]
- Scheele, B.C.; Pasmans, F.; Skerratt, L.F.; Berger, L.; Martel, A.; Beukema, W.; Acevedo, A.A.; Burrowes, P.A.; Carvalho, T.; Catenazzi, A.; et al. Amphibian fungal panzootic causes catastrophic and ongoing loss of biodiversity. Science 2019, 363, 1459–1463. [Google Scholar] [CrossRef] [PubMed]
- Berger, L.; Speare, R.; Daszak, P.; Green, D.E.; Cunningham, A.A.; Goggin, C.L.; Slocombe, R.; Ragan, M.A.; Hyatt, A.D.; McDonald, K.R.; et al. Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc. Natl. Acad. Sci. USA 1998, 95, 9031–9036. [Google Scholar] [CrossRef] [PubMed]
- Longcore, J.E.; Pessier, A.P.; Nichols, D.K. Batrachochytrium dendrobatidis gen. et sp. nov., a Chytrid Pathogenic to Amphibians. Mycologia 1999, 91, 219–227. [Google Scholar] [CrossRef]
- Martel, A.; Der Sluijs, A.S.-V.; Blooi, M.; Bert, W.; Ducatelle, R.; Fisher, M.C.; Woeltjes, A.; Bosman, W.; Chiers, K.; Bossuyt, F.; et al. Batrachochytrium salamandrivorans sp. nov. causes lethal chytridiomycosis in amphibians. Proc. Natl. Acad. Sci. USA 2013, 110, 15325–15329. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.F.; Baldi Salas, M.; Jenkins, D.; Garner, T.W.J.; Cunningham, A.A.; Hyatt, A.D.; Bosch, J.; Fisher, M.C. Environmental detection of Batrachochytrium dendrobatidis in a temperate climate. Dis. Aquat. Organ. 2007, 77, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Kirshtein, J.D.; Anderson, C.W.; Wood, J.S.; E Longcore, J.; A Voytek, M. Quantitative PCR detection of Batrachochytrium dendrobatidis DNA from sediments and water. Dis. Aquat. Org. 2007, 77, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Kamoroff, C.; Goldberg, C.S. Using environmental DNA for early detection of amphibian chytrid fungus Batrachochytrium dendrobatidis prior to a ranid die-off. Dis. Aquat. Organ. 2017, 127, 75–79. [Google Scholar] [CrossRef]
- Mosher, B.A.; Huyvaert, K.P.; Chestnut, T.; Kerby, J.L.; Madison, J.D.; Bailey, L.L. Design-and model-based recommendations for detecting and quantifying an amphibian pathogen in environmental samples. Ecol. Evol. 2017, 7, 10952–10962. [Google Scholar] [CrossRef]
- Spitzen-van der Sluijs, A.; Stark, T.; DeJean, T.; Verbrugghe, E.; Herder, J.; Gilbert, M.; Janse, J.; Martel, A.; Pasmans, F.; Valentini, A. Using environmental DNA for detection of Batrachochytrium salamandrivorans in natural water. Environ. DNA 2020, 2, 1–7. [Google Scholar] [CrossRef]
- Boyle, D.G.; Boyle, D.B.; Olsen, V.; Morgan, J.A.T.; Hyatt, A.D. Rapid quantitative detection of chytridiomycosis (Batrachochytrium dendrobatidis) in amphibian samples using real-time Taqman PCR assay. Dis. Aquat. Org. 2004, 60, 141–148. [Google Scholar] [CrossRef]
- Blooi, M.; Pasmans, F.; Longcore, J.E.; Spitzen-van der Sluijs, A.; Vercammen, F.; Martel, A. Duplex real-time PCR for rapid simultaneous detection of Batrachochytrium dendrobatidis and Batrachochytrium salamandrivorans in amphibian samples. J. Clin. Microbiol. 2013, 51, 4173–4177. [Google Scholar] [CrossRef]
- Goka, K.; Yokoyama, J.; Une, Y.; Kuroki, T.; Suzuki, K.; Nakahara, M.; Kobayashi, A.; Inaba, S.; Mizutani, T.; Hyatt, A.D. Amphibian chytridiomycosis in Japan: Distribution, haplotypes and possible route of entry into Japan. Mol. Ecol. 2009, 18, 4757–4774. [Google Scholar] [CrossRef]
- Goldberg, C.S.; Turner, C.R.; Deiner, K.; Klymus, K.E.; Thomsen, P.F.; Murphy, M.A.; Spear, S.F.; McKee, A.; Oyler-McCance, S.J.; Cornman, R.S.; et al. Critical considerations for the application of environmental DNA methods to detect aquatic species. Methods Ecol. Evol. 2016, 7, 1299–1307. [Google Scholar] [CrossRef]
- Renshaw, M.A.; Olds, B.P.; Jerde, C.L.; McVeigh, M.M.; Lodge, D.M. The room temperature preservation of filtered environmental DNA samples and assimilation into a phenol-chloroform-isamyl alcohol DNA extraction. Mol. Ecol. Resour. 2015, 15, 168–176. [Google Scholar] [CrossRef]
- Spens, J.; Evans, A.R.; Halfmaerten, D.; Knudsen, S.W.; Sengupta, M.E.; Mak, S.S.T.; Sigsgaard, E.E.; Hellström, M. Comparison of capture and storage methods for aqueous macrobial eDNA using an optimized extraction protocol: Advantage of enclosed filter. Methods Ecol. Evol. 2017, 8, 635–645. [Google Scholar] [CrossRef]
- Hyman, O.J.; Collins, J.P. Evaluation of a filtration-based method for detecting Batrachochytrium dendrobatidis in natural bodies of water. Dis. Aquat. Org. 2012, 97, 185–195. [Google Scholar] [CrossRef]
- Lastra González, D.; Baláž, V.; Solský, M.; Thumsová, B.; Kolenda, K.; Najbar, A.; Najbar, B.; Kautman, M.; Chajma, P.; Balogová, M.; et al. Recent Findings of Potentially Lethal Salamander Fungus Batrachochytrium salamandrivorans. Emerg. Infect. Dis. 2019, 25, 1416–1418. [Google Scholar] [CrossRef]
- Hyatt, A.D.; Boyle, D.B.; Olsen, V.; Boyle, A.D.H.D.G.; Berger, L.; Obendorf, D.; Dalton, A.; Kriger, K.; Hero, J.-M.; Hines, H.; et al. Diagnostic assays and sampling protocols for the detection of Batrachochytrium dendrobatidis. Dis. Aquat. Org. 2007, 73, 175–192. [Google Scholar] [CrossRef] [PubMed]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Gelman, A.; Su, Y.S. Arm: Data Analysis Using Regression and Multilevel/Hierarchical Models. R Package Version 1.10-1. 2018. Available online: https://CRAN.R-project.org/package=arm (accessed on 2 September 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 2 September 2020).
- Dalbeck, L.; Düssell-Siebert, H.; Kerres, A.; Kirst, K.; Koch, A.; Lötters, S.; Ohlhoff, D.; Sabino-Pinto, J.; Preißler, K.; Schulte, U.; et al. The salamander plague and its pathogen Batrachochytrium salamandrivorans (Bsal): Current status in Germany. Z. Feldherpetol. 2018, 25, 1–22. [Google Scholar]
- Lötters, S.; Wagner, N.; Albaladejo, G.; Böning, P.; Dalbeck, L.; Düssel, H.; Feldmeier, S.; Guschal, M.; Kirst, K.; Ohlhoff, D.; et al. The amphibian pathogen Batrachochytrium salamandrivorans in the hotspot of its European invasive range: Past-present-future. Salamandra 2020, 56, 173–188. [Google Scholar]
- Spitzen-van der Sluijs, A.; Stegen, G.; Bogaerts, S.; Canessa, S.; Steinfartz, S.; Janssen, N.; Bosman, W.; Pasmans, F.; Martel, A. Post-epizootic salamander persistence in a disease-free refugium suggests poor dispersal ability of Batrachochytrium salamandrivorans. Sci. Rep. 2018, 8, 3800. [Google Scholar] [CrossRef]
- Takahashi, M.K.; Meyer, M.J.; McPhee, C.; Gaston, J.R.; Venesky, M.D.; Case, B.F. Seasonal and diel signature of eastern hellbender environmental DNA. J. Wildl. Manag. 2018, 82, 217–225. [Google Scholar] [CrossRef]
- Goldberg, C.S.; Strickler, K.M.; Pilliod, D.S. Moving environmental DNA methods from concept to practice for monitoring aquatic macroorganisms. Biol. Conserv. 2015, 183, 1–3. [Google Scholar] [CrossRef]
- Valentini, A.; Taberlet, P.; Miaud, C.; Civade, R.; Herder, J.; Thomsen, P.F.; Bellemain, E.; Besnard, A.; Coissac, E.; Boyer, F.; et al. Next-generation monitoring of aquatic biodiversity using environmental DNA metabarcoding. Mol. Ecol. 2016, 25, 929–942. [Google Scholar] [CrossRef] [PubMed]
- Chestnut, T.; Anderson, C.; Popa, R.; Blaustein, A.R.; Voytek, M.; Olson, D.H.; Kirshtein, J. Heterogeneous Occupancy and Density Estimates of the Pathogenic Fungus Batrachochytrium dendrobatidis in Waters of North America. PLoS ONE 2014, 9, e106790. [Google Scholar] [CrossRef]
- Thomas, V.; Blooi, M.; Van Rooij, P.; Van Praet, S.; Verbrugghe, E.; Grasselli, E.; Lukac, M.; Smith, S.; Pasmans, F.; Martel, A. Recommendations on diagnostic tools for Batrachochytrium salamandrivorans. Transbound. Emerg. Dis. 2018, 65, 1–11. [Google Scholar] [CrossRef]
- Bletz, M.C.; Rebollar, E.A.; Harris, R.N. Differential efficiency among DNA extraction methods influences detection of the amphibian pathogen Batrachochytrium dendrobatidis. Dis. Aquat. Organ. 2015, 113, 1–8. [Google Scholar] [CrossRef] [PubMed]
- A Brannelly, L.; Wetzel, D.P.; West, M.; Richards-Zawacki, C.L. Optimized Batrachochytrium dendrobatidis DNA extraction of swab samples results in imperfect detection particularly when infection intensities are low. Dis. Aquat. Org. 2020, 139, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Sabino-Pinto, J.; Krause, E.T.; Bletz, M.C.; Martel, A.; Pasmans, F.; Steinfartz, S.; Vences, M. Detectability vs. time and costs in pooled DNA extraction of cutaneous swabs: A study on the amphibian chytrid fungi. Amphibia-Reptilia 2019, 40, 29–39. [Google Scholar] [CrossRef]
- OIE (World Organisation for Animal Health). Manual of Diagnostic Tests for Aquatic Animals. 2016. Available online: http://www.oie.int/international-standard-setting/aquatic-manual/access-online/ (accessed on 10 September 2020).
- Klymus, K.E.; Merkes, C.M.; Allison, M.J.; Goldberg, C.S.; Helbing, C.C.; Hunter, M.E.; Jackson, C.A.; Lance, R.F.; Mangan, A.M.; Monroe, E.M.; et al. Reporting the limits of detection and quantification for environmental DNA assays. Environ. DNA 2019, 2, 1–12. [Google Scholar] [CrossRef]
- Bedwell, M.E.; Goldberg, C.S. Spatial and temporal patterns of environmental DNA detection to inform sampling protocols in lentic and lotic systems. Ecol. Evol. 2020, 10, 1602–1612. [Google Scholar] [CrossRef]
- Martel, A.; Vila-Escale, M.; Fernández-Giberteau, D.; Martínez-Silvestre, A.; Canessa, S.; Van Praet, S.; Pannon, P.; Chiers, K.; Ferran, A.; Kelly, M.; et al. Integral chain management of wildlife diseases. Conserv. Lett. 2020, 13, e12707. [Google Scholar] [CrossRef]
- Harper, L.R.; Buxton, A.S.; Rees, H.C.; Bruce, K.; Brys, R.; Halfmaerten, D.; Read, D.S.; Watson, H.V.; Sayer, C.D.; Jones, E.P.; et al. Prospects and challenges of environmental DNA (eDNA) monitoring in freshwater ponds. Hydrobiologia 2019, 826, 25–41. [Google Scholar] [CrossRef]
- Hinlo, R.; Gleeson, D.; Lintermans, M.; Furlan, E. Methods to maximise recovery of environmental DNA from water samples. PLoS ONE 2017, 12, e0179251. [Google Scholar] [CrossRef]
- Wegleitner, B.J.; Jerde, C.L.; Tucker, A.; Chadderton, W.L.; Mahon, A.R. Long duration, room temperature preservation of filtered eDNA samples. Conserv. Genet. Resour. 2015, 7, 789–791. [Google Scholar] [CrossRef]
- Brannelly, L.A.; Wetzel, D.P.; Ohmer, M.E.B.; Zimmerman, L.; Saenz, V.; Richards-Zawacki, C.L. Evaluating environmental DNA as a tool for detecting an amphibian pathogen using an optimized extraction method. Oecologia 2020, 194, 267–281. [Google Scholar] [CrossRef] [PubMed]
- Sabino-Pinto, J.; Veith, M.; Vences, M.; Steinfartz, S. Asymptomatic infection of the fungal pathogen Batrachochytrium salamandrivorans in captivity. Sci. Rep. 2018, 8, 11767. [Google Scholar] [CrossRef]
| Country | Locality | First Bsal Positive | N Filters | N Swabs | Bsal DNA Copies | Bd (DNA Copies) |
|---|---|---|---|---|---|---|
| Spain | Ampuero | * 2017 | 1/1 | 5/5 | 10.81 ‡/8.05–23 § | |
| Spain | Teverga 1 | * 2017 | 1/1 | 1/19 | 96.55 ‡/15.05 § | |
| Spain | Teverga 2 | * 2017 | 1/1 | 0 | 15.70 ‡ | |
| Spain | Teverga 3 † | 2019 | 2/2 | 2/2 | 12.2–12.45 ‡/3.27–17 § | |
| Spain | Ruente 1 | * 2017 | 1/2 | 0/20 | 12 ‡ | |
| Spain | Ruente 2 | 2019 | 1/1 | 0/11 | 6.16 ‡ | 28.8 ‡ |
| Spain | Suances | * 2017 | 2/2 | 0/25 | 22.35–96.05 ‡ | 31.2; 764.4 ‡ |
| Spain | Ponga † | 2019 | 2/2 | 0/15 | 1.51–24.2 ‡ | |
| Spain | Cieza | 2019 | 1/1 | 0/11 | 33.3 ‡ | |
| Czech R. | Stare Mesto | N/A | 2/2 | 11/30 | N/A | 0.1–7.3 ‡/1.91–44.55 § |
| Czech R. | Sokolov | N/A | 0/32 | N/A | N/A |
| Storage Type | Primers from Blooi et al., 2013 | Primers from Spitzen-van der Sluijs et al., 2020 |
|---|---|---|
| Control, room temperature | 0.01% * | 0.1% * |
| Longmire’s buffer | 0.04% | 0.2% * |
| Silica gel | 0.19% | 1.1% |
| 96% EtOH | 0.34% | 1.1% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lastra González, D.; Baláž, V.; Vojar, J.; Chajma, P. Dual Detection of the Chytrid Fungi Batrachochytrium spp. with an Enhanced Environmental DNA Approach. J. Fungi 2021, 7, 258. https://doi.org/10.3390/jof7040258
Lastra González D, Baláž V, Vojar J, Chajma P. Dual Detection of the Chytrid Fungi Batrachochytrium spp. with an Enhanced Environmental DNA Approach. Journal of Fungi. 2021; 7(4):258. https://doi.org/10.3390/jof7040258
Chicago/Turabian StyleLastra González, David, Vojtech Baláž, Jiří Vojar, and Petr Chajma. 2021. "Dual Detection of the Chytrid Fungi Batrachochytrium spp. with an Enhanced Environmental DNA Approach" Journal of Fungi 7, no. 4: 258. https://doi.org/10.3390/jof7040258
APA StyleLastra González, D., Baláž, V., Vojar, J., & Chajma, P. (2021). Dual Detection of the Chytrid Fungi Batrachochytrium spp. with an Enhanced Environmental DNA Approach. Journal of Fungi, 7(4), 258. https://doi.org/10.3390/jof7040258






