Virulence Factors and Antifungal Susceptibility Profile of C. tropicalis Isolated from Various Clinical Specimens in Alexandria, Egypt
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Identification
2.3. Antifungal Susceptibility Testing
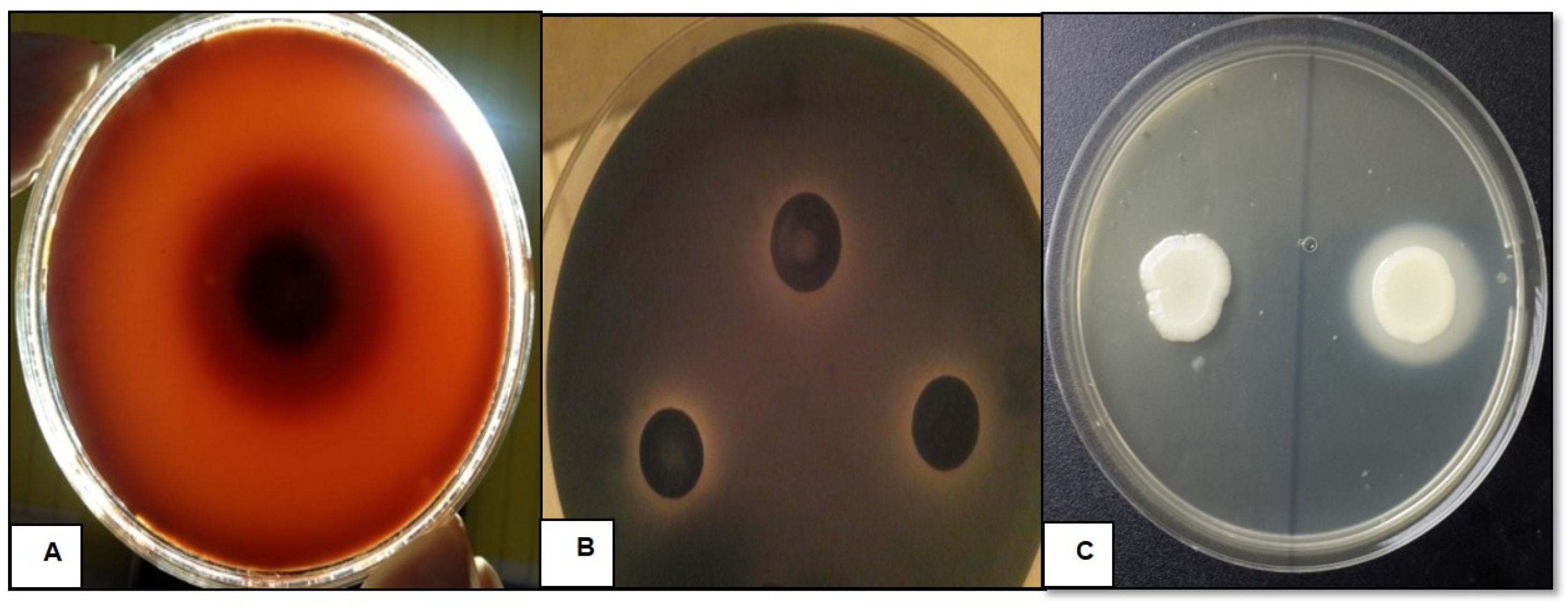
2.4. Assessment of Virulence Factors
2.5. Determination of Biofilm Formation
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statements
Acknowledgments
Conflicts of Interest
References
- Deorukhkar, S.C.; Saini, S.; Mathew, S. Non-albicans Candida Infection: An Emerging Threat. Interdiscip. Perspect. Infect. Dis. 2014, 2014, 615958. [Google Scholar] [CrossRef] [PubMed]
- Jackson, B.E.; Wilhelmus, K.R.; Mitchell, B.M. Genetically regulated filamentation contributes to Candida albicans virulence during corneal infection. Microb. Pathog. 2007, 42, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Achkar, J.M.; Fries, B.C. Candida infections of the genitourinary tract. Clin. Microbiol. Rev. 2010, 23, 253–273. [Google Scholar] [CrossRef] [PubMed]
- Rosenbach, A.; Dignard, D.; Pierce, J.V.; Whiteway, M.; Kumamoto, C.A. Adaptations of Candida albicans for growth in the mammalian intestinal tract. Eukaryot. Cell 2010, 9, 1075–1086. [Google Scholar] [CrossRef] [PubMed]
- Naglik, J.R.; Moyes, D.L.; Wachtler, B.; Hube, B. Candida albicans interactions with epithelial cells and mucosal immunity. Microbes Infect. 2011, 13, 963–976. [Google Scholar] [CrossRef] [PubMed]
- Lamoth, F.; Lockhart, S.R.; Berkow, E.L.; Calandra, T. Changes in the epidemiological landscape of invasive candidiasis. J. Antimicrob. Chemother. 2018, 73, i4–i13. [Google Scholar] [CrossRef]
- Tan, T.Y.; Tan, A.L.; Tee, N.W.; Ng, L.S.; Chee, C.W. The increased role of non-albicans species in candidaemia: Results from a 3-year surveillance study. Mycoses 2010, 53, 515–521. [Google Scholar] [CrossRef]
- Sipsas, N.V.; Lewis, R.E.; Tarrand, J.; Hachem, R.; Rolston, K.V.; Raad, I.I.; Kontoyiannis, D.P. Candidemia in patients with hematologic malignancies in the era of new antifungal agents (2001-2007): Stable incidence but changing epidemiology of a still frequently lethal infection. Cancer 2009, 115, 4745–4752. [Google Scholar] [CrossRef]
- Maccallum, D.M. Hosting infection: Experimental models to assay Candida virulence. Int. J. Microbiol. 2012, 2012, 363764. [Google Scholar] [CrossRef]
- Silva, S.; Negri, M.; Henriques, M.; Oliveira, R.; Williams, D.W.; Azeredo, J. Candida glabrata, Candida parapsilosis and Candida tropicalis: Biology, epidemiology, pathogenicity and antifungal resistance. FEMS Microbiol. Rev. 2012, 36, 288–305. [Google Scholar] [CrossRef]
- Pahwa, N.; Kumar, R.; Nirkhiwale, S.; Bandi, A. Species distribution and drug susceptibility of Candida in clinical isolates from a tertiary care centre at Indore. Indian J. Med. Microbiol. 2014, 32, 44–48. [Google Scholar] [CrossRef]
- Negri, M.; Silva, S.; Henriques, M.; Oliveira, R. Insights into Candida tropicalis nosocomial infections and virulence factors. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1399–1412. [Google Scholar] [CrossRef] [PubMed]
- Kothalawala, M.; Jayaweera, J.; Arunan, S.; Jayathilake, A. The emergence of non-albicans candidemia and evaluation of HiChrome Candida differential agar and VITEK2 YST® platform for differentiation of Candida bloodstream isolates in teaching hospital Kandy, Sri Lanka. BMC Microbiol. 2019, 19, 136. [Google Scholar] [CrossRef]
- Chai, L.Y.; Denning, D.W.; Warn, P. Candida tropicalis in human disease. Crit. Rev. Microbiol. 2010, 36, 282–298. [Google Scholar] [CrossRef] [PubMed]
- Hurley, R.; Winner, H. The pathogenicity of Candida tropicalis. J. Pathol. Bacteriol. 1962, 84, 33–88. [Google Scholar] [CrossRef]
- Vandeputte, P.; Ferrari, S.; Coste, A.T. Antifungal resistance and new strategies to control fungal infections. Int. J. Microbiol. 2012. [Google Scholar] [CrossRef]
- Melhem, M.; Bertoletti, A.; Lucca, H.; Silva, R.; Meneghin, F.; Szeszs, M. Use of the VITEK 2 system to identify and test the antifungal susceptibility of clinically relevant yeast species. Braz. J. Microbiol. 2013, 44, 1257–1266. [Google Scholar] [CrossRef]
- CLSI. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts: Fourth Informational Supplement M27-S4; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Pfaller, M.A.; Espinel-Ingroff, A.; Canton, E.; Castanheira, M.; Cuenca-Estrella, M.; Diekema, D.J.; Fothergill, A.; Fuller, J.; Ghannoum, M.; Jones, R.N.; et al. Wild-type MIC distributions and epidemiological cutoff values for amphotericin B, flucytosine, and itraconazole and Candida spp. as determined by CLSI broth microdilution. J. Clin. Microbiol. 2012, 50, 2040–2046. [Google Scholar] [CrossRef] [PubMed]
- Manns, J.M.; Mosser, D.M.; Buckley, H.R. Production of a hemolytic factor by Candida albicans. Infect. Immun. 1994, 62, 5154–5156. [Google Scholar] [CrossRef]
- Price, M.F.; Wilkinson, I.D.; Gentry, L.O. Plate method for detection of phospholipase activity in Candida albicans. Sabouraudia 1982, 20, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Ito-Kuwa, S.; Nakamura, Y.; Masuhara, T. Comparative pathogenicity of a wild-type strain and respiratory mutants of Candida albicans in mice. Zent. Bakteriol. 1990, 273, 332–343. [Google Scholar] [CrossRef]
- Samaranayake, L.P.; Raeside, J.M.; MacFarlane, T.W. Factors affecting the phospholipase activity of Candida species in vitro. Sabouraudia 1984, 22, 201–207. [Google Scholar] [CrossRef]
- Ramos Lde, S.; Barbedo, L.S.; Braga-Silva, L.A.; dos Santos, A.L.; Pinto, M.R.; Sgarbi, D.B. Protease and phospholipase activities of Candida spp. isolated from cutaneous candidiasis. Rev. Iberoam. Micol. 2015, 32, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Yigit, N.; Aktas, E.; Dagistan, S.; Ayyildiz, A. Investigating biofilm production, coagulase and hemolytic activity in Candida species isolated from denture stomatitis patients. Eurasian J. Med. 2011, 43, 27. [Google Scholar] [CrossRef]
- Silva, S.; Henriques, M.; Martins, A.; Oliveira, R.; Williams, D.; Azeredo, J. Biofilms of non-Candida albicans Candida species: Quantification, structure and matrix composition. Med. Mycol. 2009, 47, 681–689. [Google Scholar] [CrossRef]
- Stepanović, S.; Ćirković, I.; Ranin, L.; Svabić-Vlahović, M. Biofilm formation by Salmonella spp. and Listeria monocytogenes on plastic surface. Lett. Appl. Microbiol. 2004, 38, 428–432. [Google Scholar] [CrossRef]
- Singh, R.; Chakrabarti, A. Invasive Candidiasis in the Southeast-Asian Region. In Candida Albicans: Cellular and Molecular Biology; Prasad, R., Ed.; Springer International Publishing: Cham, Germany, 2017; pp. 25–40. [Google Scholar]
- Wynn, J.L.; Tan, S.; Gantz, M.G.; Das, A.; Goldberg, R.N.; Adams-Chapman, I.; Stoll, B.J.; Shankaran, S.; Walsh, M.C.; Auten, K.J.; et al. Outcomes following candiduria in extremely low birth weight infants. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2012, 54, 331–339. [Google Scholar] [CrossRef]
- Hegazi, M.; Abdelkader, A.; Zaki, M.; El-Deek, B. Characteristics and risk factors of candidemia in pediatric intensive care unit of a tertiary care children’s hospital in Egypt. J. Infect. Dev. Ctries. 2014, 8, 624–634. [Google Scholar] [CrossRef]
- Helmi, H. Bloodstream infections due to Candida Species and antifungal susceptibility profile. Egypt. J. Med. Microbiol. 2009, 18, 13–22. [Google Scholar]
- Esmat, M.M.; Mohamed, T.; Abdelrahman, A.H. Species Identification and Antifungal Susceptibility Profile of Candida Isolates from ICU Patients in Sohag University Hospital, Upper Egypt. Egypt. J. Med. Microbiol. 2015, 38, 1–9. [Google Scholar] [CrossRef]
- Shawky, S.M.; Gaballah, A.H.; Abdallah, A.; Fadel, S.; El Kholy, M.A. Automated Identification and Antifungal Susceptibility Testing of Candida Species using Vitek 2 Compact System in ICUs and Pediatric Oncology Unit, Alexandria, Egypt. Egypt. J. Med. Microbiol. 2017, 26, 101–109. [Google Scholar] [CrossRef]
- Eddouzi, J.; Lohberger, A.; Vogne, C.; Manai, M.; Sanglard, D. Identification and antifungal susceptibility of a large collection of yeast strains isolated in Tunisian hospitals. Med. Mycol. 2013, 51, 737–746. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ibrahim, N.H.; Melake, N.A.; Somily, A.M.; Zakaria, A.S.; Baddour, M.M.; Mahmoud, A.Z. The effect of antifungal combination on transcripts of a subset of drug-resistance genes in clinical isolates of Candida species induced biofilms. Saudi Pharm. J. 2015, 23, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.; Fan, X.; Chen, S.C.; Wang, H.; Sun, Z.Y.; Liao, K.; Chen, S.L.; Yan, Y.; Kang, M.; Hu, Z.D.; et al. Antifungal susceptibilities of Candida glabrata species complex, Candida krusei, Candida parapsilosis species complex and Candida tropicalis causing invasive candidiasis in China: 3 year national surveillance. J. Antimicrob. Chemother. 2015, 70, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.T.; Liu, C.Y.; Liao, C.H.; Chung, K.P.; Sheng, W.H.; Hsueh, P.R. Antifungal susceptibilities of Candida isolates causing bloodstream infections at a medical center in Taiwan, 2009–2010. Antimicrob. Agents Chemother. 2014, 58, 3814–3819. [Google Scholar] [CrossRef]
- Orasch, C.; Marchetti, O.; Garbino, J.; Schrenzel, J.; Zimmerli, S.; Mühlethaler, K.; Pfyffer, G.; Ruef, C.; Fehr, J.; Zbinden, R. Candida species distribution and antifungal susceptibility testing according to European Committee on Antimicrobial Susceptibility Testing and new vs. old Clinical and Laboratory Standards Institute clinical breakpoints: A 6-year prospective candidaemia survey from the fungal infection network of Switzerland. Clin. Microbiol. Infect. 2014, 20, 698–705. [Google Scholar]
- Shurtleff, D.B.; Peterson, W.; Sherris, J.C. Systemic Candida tropicalis infection treated with amphotericin. N. Engl. J. Med. 1963, 269, 1112–1115. [Google Scholar] [CrossRef]
- Yu, S.; Li, W.; Che, J.; Bian, F.; Lu, J.; Wu, Y. Study on virulence factors of Candida tropicalis isolated from clinical samples. Zhonghua Liu Xing Bing Xue Za Zhi 2015, 36, 1162–1166. [Google Scholar]
- de Melo Riceto, É.B.; de Paula Menezes, R.; Penatti, M.P.A.; dos Santos Pedroso, R. Enzymatic and hemolytic activity in different Candida species. Rev. Iberoam. Micol. 2015, 32, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Deorukhkar, S.; Saini, S. Non albicans Candida species: Its isolation pattern, species distribution, virulence factors and antifungal susceptibility profile. Int. J. Med. Sci. Public Health 2013, 2, 533–539. [Google Scholar] [CrossRef]
- Luo, G.; Samaranayake, L.P.; Yau, J.Y. Candida species exhibit differential in vitro hemolytic activities. J. Clin. Microbiol. 2001, 39, 2971–2974. [Google Scholar] [CrossRef] [PubMed]
- Favero, D.; Furlaneto-Maia, L.; Franca, E.J.; Goes, H.P.; Furlaneto, M.C. Hemolytic factor production by clinical isolates of Candida species. Curr. Microbiol. 2014, 68, 161–166. [Google Scholar] [CrossRef]
- Furlaneto-Maia, L.; Specian, A.F.; Bizerra, F.C.; de Oliveira, M.T.; Furlaneto, M.C. In vitro evaluation of putative virulence attributes of oral isolates of Candida spp. obtained from elderly healthy individuals. Mycopathologia 2008, 166, 209–217. [Google Scholar] [CrossRef]
- Kumar, V.; Latha, R.; Vedhagiri, K.; Sathiamoorthi, T.; Jayarani, G.; Sasikala, R.; Selvin, J.; Natarajaseenivasan, K. Phospholipase C, proteinase and hemolytic activities of Candida spp. isolated from pulmonary tuberculosis patients. J. Mycol. Médicale 2009, 19, 3–10. [Google Scholar] [CrossRef]
- Dostál, J.; Hamal, P.; Pavlíčková, L.; Souček, M.; Ruml, T.; Pichová, I.; Hrušková-Heidingsfeldová, O. Simple method for screening Candida species isolates for the presence of secreted proteinases: A tool for the prediction of successful inhibitory treatment. J. Clin. Microbiol. 2003, 41, 712–716. [Google Scholar] [CrossRef]
- Galan-Ladero, M.; Blanco, M.; Sacristan, B.; Fernandez-Calderon, M.; Perez-Giraldo, C.; Gomez-Garcia, A. Enzymatic activities of Candida tropicalis isolated from hospitalized patients. Med. Mycol. 2010, 48, 207–210. [Google Scholar] [CrossRef]
- Deorukhkar, S.C.; Saini, S.; Mathew, S. Virulence Factors Contributing to Pathogenicity of Candida tropicalis and Its Antifungal Susceptibility Profile. Int. J. Microbiol. 2014, 2014, 456878. [Google Scholar] [CrossRef]
- Negri, M.; Martins, M.; Henriques, M.; Svidzinski, T.I.; Azeredo, J.; Oliveira, R. Examination of potential virulence factors of Candida tropicalis clinical isolates from hospitalized patients. Mycopathologia 2010, 169, 175–182. [Google Scholar] [CrossRef]
- da Costa, K.R.C.; Ferreira, J.C.; Lavrador, M.A.S.; Baruffi, M.D.; Candido, R.C. Virulence attributes and genetic variability of oral Candida albicans and Candida tropicalis isolates. Mycoses 2012, 55, e97–e105. [Google Scholar] [CrossRef] [PubMed]
- Udayalaxmi; Jacob, S.; D’Souza, D. Comparison Between Virulence Factors of Candida albicans and Non-Albicans Species of Candida Isolated from Genitourinary Tract. J. Clin. Diagn. Res. JCDR 2014, 8, DC15–DC17. [Google Scholar] [CrossRef] [PubMed]
- Mane, A.; Pawale, C.; Gaikwad, S.; Bembalkar, S.; Risbud, A. Adherence to buccal epithelial cells, enzymatic and hemolytic activities of Candida isolates from HIV-infected individuals. Med. Mycol. 2011, 49, 548–551. [Google Scholar] [PubMed]
- Aher, C.S. Species distribution, virulence factors and antifungal susceptibility profile of Candida isolated from Oropharyngeal lesions of HIV infected patients. Int. J. Curr. Microbiol. Appl. Sci 2014, 3, 453–460. [Google Scholar]
- Tellapragada, C.; Eshwara, V.K.; Johar, R.; Shaw, T.; Malik, N.; Bhat, P.V.; Kamath, A.; Mukhopadhyay, C. Antifungal susceptibility patterns, in vitro production of virulence factors, and evaluation of diagnostic modalities for the speciation of pathogenic Candida from blood stream infections and vulvovaginal candidiasis. J. Pathog. 2014, 2014, 142864. [Google Scholar] [CrossRef]
- Agwan, V.; Butola, R.; Madan, M. Comparison of biofilm formation in clinical isolates of Candida species in a tertiary care center, North India. Indian J. Pathol. Microbiol. 2015, 58, 475–478. [Google Scholar] [CrossRef] [PubMed]
- Tumbarello, M.; Fiori, B.; Trecarichi, E.M.; Posteraro, P.; Losito, A.R.; De Luca, A.; Sanguinetti, M.; Fadda, G.; Cauda, R.; Posteraro, B. Risk factors and outcomes of candidemia caused by biofilm-forming isolates in a tertiary care hospital. PLoS ONE 2012, 7, e33705. [Google Scholar] [CrossRef]
- Marcos-Zambrano, L.J.; Escribano, P.; Bouza, E.; Guinea, J. Production of biofilm by Candida and non-Candida spp. isolates causing fungemia: Comparison of biomass production and metabolic activity and development of cut-off points. Int. J. Med. Microbiol. IJMM 2014, 304, 1192–1198. [Google Scholar] [CrossRef]
- Guembe, M.; Cruces, R.; Pelaez, T.; Munoz, P.; Bouza, E.; GEIDI study group. Assessment of biofilm production in Candida isolates according to species and origin of infection. Enferm. Infecc. Microbiol. Clin. 2017, 35, 37–40. [Google Scholar] [CrossRef]
- Negri, M.; Silva, S.; Capoci, I.R.; Azeredo, J.; Henriques, M. Candida tropicalis Biofilms: Biomass, Metabolic Activity and Secreted Aspartyl Proteinase Production. Mycopathologia 2016, 181, 217–224. [Google Scholar] [CrossRef]
- Bhatt, M.; Sarangi, G.; Paty, B.P.; Mohapatra, D.; Chayani, N.; Mahapatra, A.; Das, P.; Sahoo, D. Biofilm as a virulence marker in Candida species in Nosocomial blood stream infection and its correlation with antifungal resistance. Indian J. Med. Microbiol. 2015, 33, 112–114. [Google Scholar] [CrossRef]
- Negri, M.; Silva, S.; Breda, D.; Henriques, M.; Azeredo, J.; Oliveira, R. Candida tropicalis biofilms: Effect on urinary epithelial cells. Microb. Pathog. 2012, 53, 95–99. [Google Scholar] [CrossRef]

| Antifungal Agent | Sensitivity | Dose-Dependent Susceptibility | Resistance |
|---|---|---|---|
| n (%) | n (%) | n (%) | |
| (I) Azoles | |||
| Fluconazole | 41(57.75) | 4(5.63) | 26(36.62) |
| Voriconazole | 43(60.56) | 23(32.39) | 5(7.04) |
| (II) Echinocandins | |||
| Caspofungin | 71(100) | 0(0) | 0(0) |
| Micafungin | 71(100) | 0(0) | 0(0) |
| (III) Flucytosine | 71(100) | 0(0) | 0(0) |
| (IV) Amphotericin B | 71(100) | 0(0) | 0(0) |
| MIC (µg/mL) | C. tropicalis Isolates (n = 71) | Percentage (%) |
|---|---|---|
| Fluconazole | ||
| ≤1 | 41 | 57.75 |
| 2 | 0 | 0 |
| 4 | 4 | 5.63 |
| 8 | 13 | 18.31 |
| 16 | 1 | 1.41 |
| 32 | 10 | 14.08 |
| ≥64 | 2 | 2.82 |
| Voriconazole | ||
| ≤0.12 | 43 | 60.56 |
| 0.25 | 14 | 19.72 |
| 0.5 | 9 | 12.68 |
| 1 | 3 | 4.23 |
| 2 | 2 | 2.82 |
| Sample Type | Biofilm Non-Forming Isolates. n (%) | Biofilm Forming Isolates. n (%) | ||
|---|---|---|---|---|
| Weak | Moderate | Strong | ||
| Urine (n = 49) | 4(8.16%) | 22(44.9%) | 23(46.94%) | |
| Respiratory (n = 13) | 1(7.69%) | 3(23.08%) | 1(7.69%) | 8 61.54%) |
| Blood (n = 7) | 1(14.29%) | 6(85.71%) | ||
| Skin and soft tissue (n = 2) | 1(50%) | 1(50%) | ||
| Total (n = 71) | 1(1.4%) | 7(9.86%) | 25(35.21%) | 38(53.52%) |
| Biofilm Formation | Azole Susceptible Isolates n (%) | Azole Non-Susceptible Isolates n (%) |
|---|---|---|
| No | 0(0%) | 1(1.4%) |
| Weak | 7(9.86%) | 0(0%) |
| Moderate | 4(5.63%) | 21(29.58%) |
| Strong | 30(42.25%) | 8(11.27%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Kholy, M.A.; Helaly, G.F.; El Ghazzawi, E.F.; El-Sawaf, G.; Shawky, S.M. Virulence Factors and Antifungal Susceptibility Profile of C. tropicalis Isolated from Various Clinical Specimens in Alexandria, Egypt. J. Fungi 2021, 7, 351. https://doi.org/10.3390/jof7050351
El-Kholy MA, Helaly GF, El Ghazzawi EF, El-Sawaf G, Shawky SM. Virulence Factors and Antifungal Susceptibility Profile of C. tropicalis Isolated from Various Clinical Specimens in Alexandria, Egypt. Journal of Fungi. 2021; 7(5):351. https://doi.org/10.3390/jof7050351
Chicago/Turabian StyleEl-Kholy, Mohammed A., Ghada F. Helaly, Ebtisam F. El Ghazzawi, Gamal El-Sawaf, and Sherine M. Shawky. 2021. "Virulence Factors and Antifungal Susceptibility Profile of C. tropicalis Isolated from Various Clinical Specimens in Alexandria, Egypt" Journal of Fungi 7, no. 5: 351. https://doi.org/10.3390/jof7050351
APA StyleEl-Kholy, M. A., Helaly, G. F., El Ghazzawi, E. F., El-Sawaf, G., & Shawky, S. M. (2021). Virulence Factors and Antifungal Susceptibility Profile of C. tropicalis Isolated from Various Clinical Specimens in Alexandria, Egypt. Journal of Fungi, 7(5), 351. https://doi.org/10.3390/jof7050351






