Keratinases Produced by Aspergillus stelliformis, Aspergillus sydowii, and Fusarium brachygibbosum Isolated from Human Hair: Yield and Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Keratin Powder Preparation
2.2. Preliminary Experiment/Submerged Fermentation
2.3. Keratinase Activity Assay
2.4. Molecular Identification of the Potent Strains
2.5. Experiment to Produce Keratinases Using the Three Most Active Strains
2.6. Effect of PH and Temperature on the Activity of Partially Purified Keratinases
3. Results
3.1. Preliminary Experiment
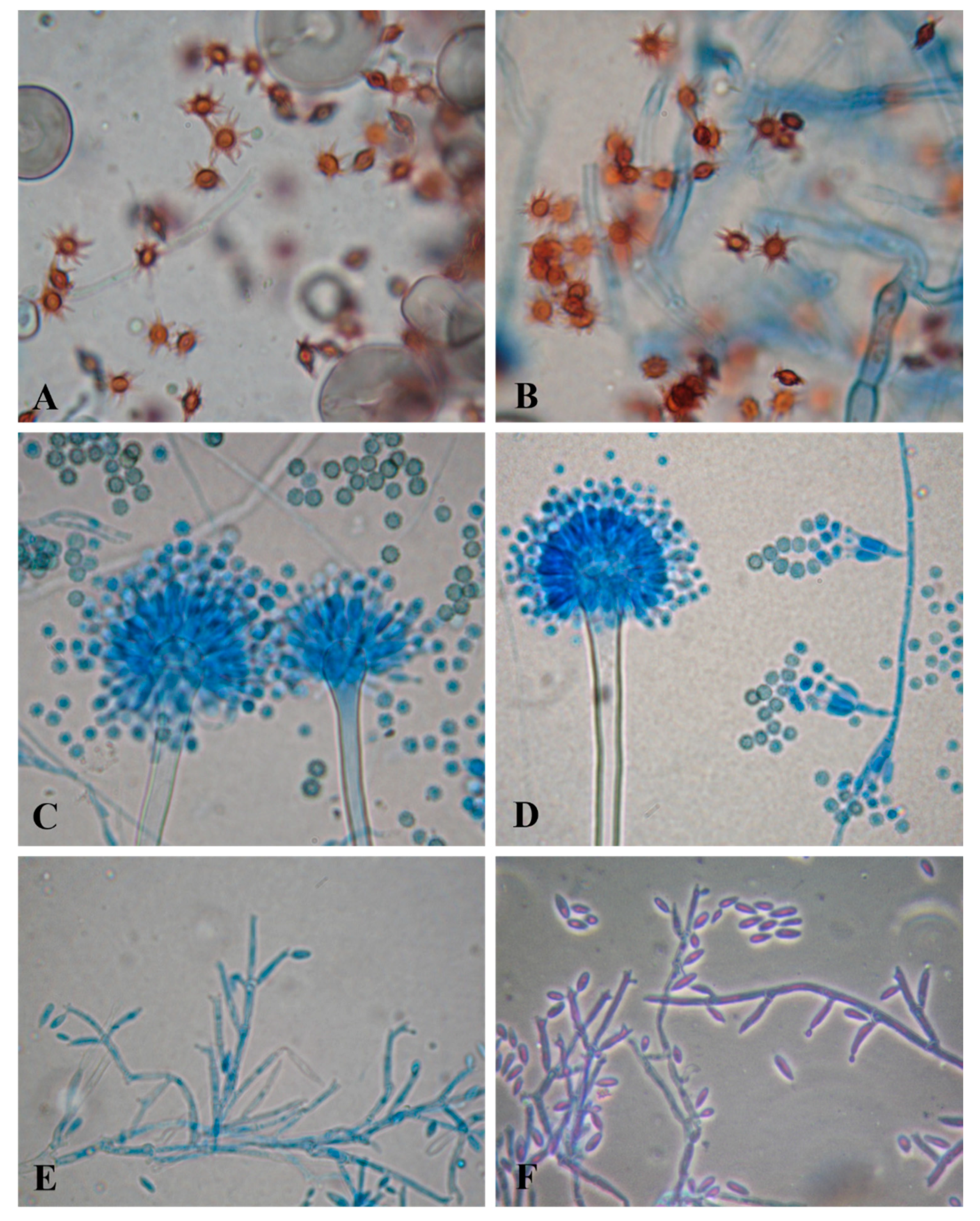
3.2. Fungi Producing Active Microbial Keratinases
3.3. Yield and Activity of Keratinases
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharma, S.; Gupta, A. Sustainable management of keratin waste biomass: Applications and future perspectives. Brazilian Arch. Biol. Technol. 2016, 59. [Google Scholar] [CrossRef] [Green Version]
- Verma, A.; Singh, H.; Anwar, S.; Chattopadhyay, A.; Tiwari, K.K.; Kaur, S.; Dhilon, G.S. Microbial keratinases: Industrial enzymes with waste management potential. Crit. Rev. Biotechnol. 2017, 37, 476–491. [Google Scholar] [CrossRef]
- Reddy, C.C.; Khilji, I.A.; Gupta, A.; Bhuyar, P.; Mahmood, S.; AL-Japairai, K.A.S.; Chua, G.K. Valorization of keratin waste biomass and its potential applications. J. Water Process Eng. 2021, 40, 101707. [Google Scholar] [CrossRef]
- Hassan, M.A.; Abol-Fotouh, D.; Omer, A.M.; Tamer, T.M.; Abbas, E. Comprehensive insights into microbial keratinases and their implication in various biotechnological and industrial sectors: A review. Int. J. Biol. Macromol. 2020, 154, 567–583. [Google Scholar] [CrossRef]
- Gafar, A.; Khayat, M.E.; Ahmad, S.A.; Yasid, N.A.; Shukor, M.Y. Response Surface Methodology for the Optimization of Keratinase Production in Culture Medium Containing Feathers by Bacillus sp. UPM-AAG1. Catalysts 2020, 10, 848. [Google Scholar] [CrossRef]
- Bohacz, J.; Korniłłowicz-Kowalska, T.; Kitowski, I.; Ciesielska, A. Degradation of chicken feathers by Aphanoascus keratinophilus and Chrysosporium tropicum strains from pellets of predatory birds and its practical aspect. Int. Biodeterior. Biodegrad. 2020, 151, 104968. [Google Scholar] [CrossRef]
- Nnolim, N.E.; Udenigwe, C.C.; Okoh, A.I.; Nwodo, U.U. Microbial Keratinase: Next Generation Green Catalyst and Prospective Applications. Front. Microbiol. 2020, 11, 3280. [Google Scholar] [CrossRef] [PubMed]
- Günyar, O.A.; Kiraç, S.; Aldi, B.; Ergin, C. Isolation and identification of keratinophilic fungi in soil samples from excavation area of ancient city of Stratonikeia, Turkey and determination of its enzyme potentials. J. Environ. Biol. 2020, 41, 1521–1525. [Google Scholar] [CrossRef]
- Fang, Z.; Zhang, J.; Liu, B.; Du, G.; Chen, J. Biodegradation of wool waste and keratinase production in scale-up fermenter with different strategies by Stenotrophomonas maltophilia BBE11-1. Bioresour. Technol. 2013, 140, 286–291. [Google Scholar] [CrossRef] [Green Version]
- Akhter, M.; Wal Marzan, L.; Akter, Y.; Shimizu, K. Microbial bioremediation of feather waste for keratinase production: An outstanding solution for leather dehairing in tanneries. Microbiol. Insights 2020, 13, 1178636120913280. [Google Scholar] [CrossRef] [PubMed]
- Akram, F.; ul Haq, I.; Jabbar, Z. Production and characterization of a novel thermo-and detergent stable keratinase from Bacillus sp. NKSP-7 with perceptible applications in leather processing and laundry industries. Int. J. Biol. Macromol. 2020, 164, 371–383. [Google Scholar] [CrossRef]
- Avdiyuk, K.V.; Varbanets, L.D. Keratinolytic enzymes: Producers, physical and chemical properties. Application for biotechnology. Biotechnol. Acta 2019, 12, 27–45. [Google Scholar]
- Abdel-Fattah, A.M.; El-Gamal, M.S.; Ismail, S.A.; Emran, M.A.; Hashem, A.M. Biodegradation of feather waste by keratinase produced from newly isolated Bacillus licheniformis ALW1. J. Genet. Eng. Biotechnol. 2018, 16, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Brandelli, A.; Daroit, D.J.; Riffel, A. Biochemical features of microbial keratinases and their production and applications. Appl. Microbiol. Biotechnol. 2010, 85, 1735–1750. [Google Scholar] [CrossRef]
- Moridshahi, R.; Bahreini, M.; Sharifmoghaddam, M.; Asoodeh, A. Biochemical characterization of an alkaline surfactant-stable keratinase from a new keratinase producer, Bacillus zhangzhouensis. Extremophiles 2020, 24, 693–704. [Google Scholar] [CrossRef]
- Vidmar, B.; Vodovnik, M. Microbial keratinases: Enzymes with promising biotechnological applications. Food Technol. Biotechnol. 2018, 56, 312–328. [Google Scholar] [CrossRef] [PubMed]
- Köhler, J.R.; Casadevall, A.; Perfect, J. The spectrum of fungi that infects humans. Cold Spring Harb. Perspect. Med. 2015, 5, a019273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abirami, S.; Ragavi, R.; Antony, V.S. Utilization of keratinolytic Lichtheimia corymbifera AS1 for degradation of cattle hoove—A slaughter house waste to use in plant growth. Biointerface Res. Appl. Chem 2020, 10, 6417–6426. [Google Scholar]
- Sousa, M.; Souza, O.; Maciel, M.; Cruz, R.; Rêgo, M.G.; Magalhães, O.; Pessoa-Júnior, A.; Porto, A.; Souza-Motta, C. Keratinolytic potential of fungi isolated from soil preserved at the Micoteca URM. Eur. J. Biotechnol. Biosci. 2015, 3, 10–15. [Google Scholar]
- Paul, T.; Das, A.; Mandal, A.; Halder, S.K.; DasMohapatra, P.K.; Pati, B.R.; Mondal, K.C. Production and purification of keratinase using chicken feather bioconversion by a newly isolated Aspergillus fumigatus TKF1: Detection of valuable metabolites. Biomass Convers. Biorefinery 2014, 4, 137–148. [Google Scholar] [CrossRef]
- Bagewadi, Z.K.; Mulla, S.I.; Ninnekar, H.Z. Response surface methodology based optimization of keratinase production from Trichoderma harzianum isolate HZN12 using chicken feather waste and its application in dehairing of hide. J. Environ. Chem. Eng. 2018, 6, 4828–4839. [Google Scholar] [CrossRef]
- Gradišar, H.; Friedrich, J.; Križaj, I.; Jerala, R. Similarities and specificities of fungal keratinolytic proteases: Comparison of keratinases of Paecilomyces marquandii and Doratomyces microsporus to some known proteases. Appl. Environ. Microbiol. 2005, 71, 3420–3426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alwakeel, S.S. Characterization of keratinophilic fungal species and other non-dermatophytes in hair and nail samples in riyadh, Saudi Arabia. Ass. Univ. Bull. Environ. Res. 2018, 21, 3–22. [Google Scholar] [CrossRef]
- Łaba, W.; Szczekala, K.B. Keratinolytic proteases in biodegradation of pretreated feathers. Polish J. Environ. Stud. 2013, 22, 1101–1109. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Moubasher, A.H.; Ismail, M.; Mohamed, R.; Al-Bedak, O. Production and purification of extreme xylanase from Aspergillus flavus AUMC 10331 in sub-merged fermentation. Eur. J. Biol. Res. 2019, 9, 20–28. [Google Scholar]
- Al-Bedak, O.A.; Moubasher, A.H. Aspergillus gaarensis, a new addition to section Circumdati from soil of Lake El-Gaar in Wadi-El-Natron, Egypt. Stud. Fungi 2020, 5, 59–65. [Google Scholar] [CrossRef]
- Ameen, F.; Moslem, M.; Hadi, S.; Al-Sabri, A.E. Biodegradation of diesel fuel hydrocarbons by mangrove fungi from Red Sea Coast of Saudi Arabia. Saudi J. Biol. Sci. 2016, 23. [Google Scholar] [CrossRef] [Green Version]
- El-Fakharany, E.M.; Hassan, M.A.; Taha, T.H. Production and application of extracellular laccase produced by Fusarium oxysporum EMT. Int. J. Agric. Biol. 2016, 18, 939–947. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [Green Version]
- Criscuolo, A.; Gribaldo, S. BMGE (Block Mapping and Gathering with Entropy): A new software for selection of phylogenetic informative regions from multiple sequence alignments. BMC Evol. Biol. 2010, 10, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [Green Version]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar]
- Lefort, V.; Longueville, J.-E.; Gascuel, O. SMS: Smart model selection in PhyML. Mol. Biol. Evol. 2017, 34, 2422–2424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rambaut, A.; Drummond, A.J. FigTree, version 1.4.0.; University of Edinburgh: Edinburgh, UK, 2012.
- Kothari, D.; Rani, A.; Goyal, A. Keratinases. In Current Developments in Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2017; pp. 447–469. [Google Scholar]
- Kang, D.; Herschend, J.; Al-Soud, W.A.; Mortensen, M.S.; Gonzalo, M.; Jacquiod, S.; Sørensen, S.J. Enrichment and characterization of an environmental microbial consortium displaying efficient keratinolytic activity. Bioresour. Technol. 2018, 270, 303–310. [Google Scholar] [CrossRef]
- Kalaikumari, S.S.; Vennila, T.; Monika, V.; Chandraraj, K.; Gunasekaran, P.; Rajendhran, J. Bioutilization of poultry feather for keratinase production and its application in leather industry. J. Clean. Prod. 2019, 208, 44–53. [Google Scholar] [CrossRef]
- Menon, S.; Savur, B.; Kasat, D.; Mavani, U.; Singh, S.; Rane, M. Management and utilization of Keratin Waste--A review. Int. J. Adv. Res. Ideas Innov. Technol. 2020, 6, 511–515. [Google Scholar]
- Reddy, M.R.; Reddy, K.S.; Chouhan, Y.R.; Bee, H.; Reddy, G. Effective feather degradation and keratinase production by Bacillus pumilus GRK for its application as bio-detergent additive. Bioresour. Technol. 2017, 243, 254–263. [Google Scholar] [CrossRef]
- Peng, Z.; Mao, X.; Zhang, J.; Du, G.; Chen, J. Biotransformation of keratin waste to amino acids and active peptides based on cell-free catalysis. Biotechnol. Biofuels 2020, 13, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.; Huang, Y.; Nesme, J.; Herschend, J.; Jacquiod, S.; Kot, W.; Hansen, L.H.; Lange, L.; Sørensen, S.J. Metagenomic analysis of a keratin-degrading bacterial consortium provides insight into the keratinolytic mechanisms. Sci. Total Environ. 2021, 761, 143281. [Google Scholar] [CrossRef]
- Peng, Z.; Zhang, J.; Du, G.; Chen, J. Keratin waste recycling based on microbial degradation: Mechanisms and prospects. ACS Sustain. Chem. Eng. 2019, 7, 9727–9736. [Google Scholar] [CrossRef]
- Hassan, M.A.; Taha, T.H.; Hamad, G.M.; Hashem, M.; Alamri, S.; Mostafa, Y.S. Biochemical characterisation and application of keratinase from Bacillus thuringiensis MT1 to enable valorisation of hair wastes through biosynthesis of vitamin B-complex. Int. J. Biol. Macromol. 2020, 153, 561–572. [Google Scholar] [CrossRef]
- Koutb, M.; Morsy, F.M.; Bagy, M.M.K.; Hassan, E.A. Optimization of extracellular keratinase production by Aspergillus terreus isolated from chicken’s litter. J. Adv. Lab. Res. Biol. 2012, 3, 210–216. [Google Scholar]
- Mazotto, A.M.; Couri, S.; Damaso, M.C.T.; Vermelho, A.B. Degradation of feather waste by Aspergillus niger keratinases: Comparison of submerged and solid-state fermentation. Int. Biodeterior. Biodegrad. 2013, 85, 189–195. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.-D. Purification and characterization of a keratinase from a feather-degrading fungus, Aspergillus flavus Strain K-03. Mycobiology 2007, 35, 219–225. [Google Scholar] [CrossRef] [Green Version]
- El-Ghonemy, D.H.; Ali, T.H. Optimization of physico-chemical parameters for hyper keratinase production from a newly isolated Aspergillus sp. DHE7 using chicken feather as substrate-management of biowaste. J. Appl. Pharm. Sci 2017, 7, 171–178. [Google Scholar]
- Sharaf, E.F.; Khalil, N.M. Keratinolytic activity of purified alkaline keratinase produced by Scopulariopsis brevicaulis (Sacc.) and its amino acids profile. Saudi J. Biol. Sci. 2011, 18, 117–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Isaac, G.S.; Abu-Tahon, M.A. Dehairing capability of alkaline keratinase produced by new isolated Cochliobolus hawaiiensis AUMC 8606 grown on chicken feather. Rom. Biotechnol. Lett. 2016, 22, 12147–12154. [Google Scholar]
- Kačinová, V.; Kolčáková, V.; Petranová, D. Axiocentric media education as a strategy for the cultivation of media recipients. Eur. J. Sci. Theol. 2014, 10, 103–116. [Google Scholar]
- Tapia, D.M.T.; Simões, M.L.G. Production and partial characterization of keratinase produced by a microorganism isolated from poultry processing plant wastewater. African J. Biotechnol. 2008, 7, 296–300. [Google Scholar]
- Riffel, A.; Brandelli, A.; Bellato, C.d.M.; Souza, G.H.M.F.; Eberlin, M.N.; Tavares, F.C.A. Purification and characterization of a keratinolytic metalloprotease from Chryseobacterium sp. kr6. J. Biotechnol. 2007, 128, 693–703. [Google Scholar] [CrossRef]
- Farag, A.M.; Hassan, M.A. Purification, characterization and immobilization of a keratinase from Aspergillus oryzae. Enzyme Microb. Technol. 2004, 34, 85–93. [Google Scholar] [CrossRef]
- El-Ayouty, Y.M.; El-Said, A.; Salama, A.M. Purification and characterization of a keratinase from the feather-degrading cultures of Aspergillus flavipes. Afr. J. Biotechnol. 2012, 11, 2313–2319. [Google Scholar] [CrossRef]
- Ploetz, R.C. Fusarium wilt of banana. Phytopathology 2015, 105, 1512–1521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al Nadabi, W.; Faisal, M.; Mohammed, M.A. Patient safety culture in Oman: A national study. J. Eval. Clin. Pract. 2020, 26, 1406–1415. [Google Scholar] [CrossRef] [PubMed]
- Ezrari, S.; Lahlali, R.; Radouane, N.; Tahiri, A.; Asfers, A.; Boughalleb-M’Hamdi, N.; Amiri, S.; Lazraq, A. Characterization of Fusarium species causing dry root rot disease of citrus trees in Morocco. J. Plant Dis. Prot. 2020, 128, 1–17. [Google Scholar]
- Nishad, R.; Ahmed, T.A. Survey and identification of date palm pathogens and indigenous biocontrol agents. Plant Dis. 2020, 104, 2498–2508. [Google Scholar] [CrossRef] [PubMed]
- Escrivá, L.; Font, G.; Manyes, L. In vivo toxicity studies of Fusarium mycotoxins in the last decade: A review. Food Chem. Toxicol. 2015, 78, 185–206. [Google Scholar] [CrossRef]
- Gutleb, A.C.; Morrison, E.; Murk, A.J. Cytotoxicity assays for mycotoxins produced by Fusarium strains: A review. Environ. Toxicol. Pharmacol. 2002, 11, 309–320. [Google Scholar] [CrossRef]
- Ein-Gil, N.; Ilan, M.; Carmeli, S.; Smith, G.W.; Pawlik, J.R.; Yarden, O. Presence of Aspergillus sydowii, a pathogen of gorgonian sea fans in the marine sponge Spongia obscura. ISME J. 2009, 3, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Soler-Hurtado, M.M.; Sandoval-Sierra, J.V.; Machordom, A.; Diéguez-Uribeondo, J. Aspergillus sydowii and other potential fungal pathogens in gorgonian octocorals of the Ecuadorian Pacific. PLoS One 2016, 11, e0165992. [Google Scholar] [CrossRef] [Green Version]
- Mousavi, B.; Hedayati, M.T.; Hedayati, N.; Ilkit, M.; Syedmousavi, S. Aspergillus species in indoor environments and their possible occupational and public health hazards. Curr. Med. Mycol. 2016, 2, 36. [Google Scholar] [CrossRef] [Green Version]
- Sabino, R.; Verissimo, C.; Viegas, C.; Viegas, S.; Brandão, J.; Alves-Correia, M.; Borrego, L.-M.; Clemons, K.V.; Stevens, D.A.; Richardson, M. The role of occupational Aspergillus exposure in the development of diseases. Med. Mycol. 2019, 57, S196–S205. [Google Scholar] [CrossRef]
- Peidro-Guzmán, H.; Pérez-Llano, Y.; González-Abradelo, D.; Fernández-López, M.G.; Dávila-Ramos, S.; Aranda, E.; Hernández, D.R.O.; Garcia, A.O.; Lira-Ruan, V.; Pliego, O.R.; et al. Transcriptomic analysis of polyaromatic hydrocarbon degradation by the halophilic fungus Aspergillus sydowii at hypersaline conditions. Environ. Microbiol. 2020. [Google Scholar] [CrossRef]
- González-Abradelo, D.; Pérez-Llano, Y.; Peidro-Guzmán, H.; del Rayo Sánchez-Carbente, M.; Folch-Mallol, J.L.; Aranda, E.; Vaidyanathan, V.K.; Cabana, H.; Gunde-Cimerman, N.; Batista-García, R.A. First demonstration that ascomycetous halophilic fungi (Aspergillus sydowii and Aspergillus destruens) are useful in xenobiotic mycoremediation under high salinity conditions. Bioresour. Technol. 2019, 279, 287–296. [Google Scholar] [CrossRef]
- Zhang, C.; Tao, Y.; Li, S.; Tian, J.; Ke, T.; Wei, S.; Wang, P.; Chen, L. Simultaneous degradation of trichlorfon and removal of Cd (II) by Aspergillus sydowii strain PA F-2. Environ. Sci. Pollut. Res. 2019, 26, 26844–26854. [Google Scholar] [CrossRef] [PubMed]
- Bu, C.; Zhang, Q.; Zeng, J.; Cao, X.; Hao, Z.; Qiao, D.; Cao, Y.; Xu, H. Identification of a novel anthocyanin synthesis pathway in the fungus Aspergillus sydowii H-1. BMC Genomics 2020, 21, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Matkar, K.; Chapla, D.; Divecha, J.; Nighojkar, A.; Madamwar, D. Production of cellulase by a newly isolated strain of Aspergillus sydowii and its optimization under submerged fermentation. Int. Biodeterior. Biodegrad. 2013, 78, 24–33. [Google Scholar] [CrossRef]
- Nair, S.G.; Sindhu, R.; Shashidhar, S. Purification and biochemical characterization of two xylanases from Aspergillus sydowii SBS 45. Appl. Biochem. Biotechnol. 2008, 149, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, K.K.; Albuquerque, W.W.; Costa, R.M.; Batista, J.M.S.; Marques, D.A.; Bezerra, R.P.; Herculano, P.N.; Porto, A.L. Biotechnological potential of a novel tannase-acyl hydrolase from Aspergillus sydowii using waste coir residue: Aqueous two-phase system and chromatographic techniques. Biocatal. Agric. Biotechnol. 2020, 23, 101453. [Google Scholar] [CrossRef]
- Cong, B.; Wang, N.; Liu, S.; Liu, F.; Yin, X.; Shen, J. Isolation, characterization and transcriptome analysis of a novel Antarctic Aspergillus sydowii strain MS-19 as a potential lignocellulosic enzyme source. BMC Microbiol. 2017, 17, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sklenávr, F.; Jurjević, Ž.; Peterson, S.W.; Kolavr’ik, M.; Nováková, A.; Flieger, M.; Stoduulková, E.; Kubátová, A.; Hubka, V. Increasing the species diversity in the Aspergillus section Nidulantes: Six novel species mainly from the indoor environment. Mycologia 2020, 112, 342–370. [Google Scholar] [CrossRef] [PubMed]




| Fungal Species | AUMC No. | Relative Activity IU mL−1 min−1 |
|---|---|---|
| Alternaria alternata (Fries) Keissler | 10926 | 689 ± 42 |
| Alternaria alternata (Fries) Keissler | 10932 | 1680 ± 120 |
| Alternaria botrytis Woudenberg and Crous | 10931 | 1211 ± 98 |
| Alternaria botrytis Woudenberg and Crous | 10936 | 1018 ± 87 |
| Alternaria chlamydosporigena Woudenberg and Crous | 10915 | 1398 ± 154 |
| Aspergillus nidulans Winter | 10933 | 935 ± 70 |
| Aspergillus niger van Tieghem | 10912 | 52 ± 23 |
| Aspergillus stelliformis F. Sklenar, Jurjević and Hubka | 10920 | 3336 ± 169 |
| Aspergillus sydowii (Bainier and Sartory) Thom and Church | 10935 | 3523 ± 188 |
| Aspergillus ustus (Bainier) Thom and Church | 10934 | 2091 ± 197 |
| Aureobasidium pullulans (de Bary) Arnaud | 10914 | 129 ± 11 |
| Chaetomium globosum Kunze | 10941 | 1600 ± 156 |
| Curvularia tsudae (Tsuda & Ueyama) Deng, Tan and Shivas | 10940 | 570 ± 85 |
| Fusarium brachygibbosum Padwick | 10937 | 3554 ± 189 |
| Nodulisporium sp. | 10916 | 270 ± 20 |
| Penicillium chrysogenum Thom | 10913 | 589 ± 11 |
| Penicillium glabrum (Wehmer) Westling | 10929 | 218 ± 49 |
| Phoma herbarum Westend | 10919 | 258 ± 12 |
| Pyrenophora dematioidea (Bubák & Wróbl.) Rossman and K.D. Hyde | 10930 | 580 ± 33 |
| Pyrenophora dematioidea (Bubák & Wróbl.) Rossman and K.D. Hyde | 10938 | 512 ± 15 |
| Control Activity KUg−1 Enzyme | 113 | 121 | 130 |
|---|---|---|---|
| Inhibitor | A. stelliformis | A. sydowii | F. brachygibbosum |
| FeSO4 | 28 ± 4 | 27 ± 2 | 24 ± 3 |
| ZnSO4 | 29 ± 3 | 18 ± 2 | 23 ± 4 |
| CaCl2 | 34 ± 6 | 22 ± 6 | 22 ± 4 |
| CuSO4 | 28 ± 3 | 22 ± 5 | 23 ± 2 |
| NiCl2 | 30 ± 4 | 58 ± 9 | 45 ± 4 |
| CoCl2 | 76 ± 8 | 33 ± 8 | 27 ± 4 |
| MgSO4 | 27 ± 5 | 17 ± 5 | 53 ± 8 |
| EDTA | 7 ± 1 | 17 ± 4 | 52 ± 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alwakeel, S.S.; Ameen, F.; Al Gwaiz, H.; Sonbol, H.; Alghamdi, S.; Moharram, A.M.; Al-Bedak, O.A. Keratinases Produced by Aspergillus stelliformis, Aspergillus sydowii, and Fusarium brachygibbosum Isolated from Human Hair: Yield and Activity. J. Fungi 2021, 7, 471. https://doi.org/10.3390/jof7060471
Alwakeel SS, Ameen F, Al Gwaiz H, Sonbol H, Alghamdi S, Moharram AM, Al-Bedak OA. Keratinases Produced by Aspergillus stelliformis, Aspergillus sydowii, and Fusarium brachygibbosum Isolated from Human Hair: Yield and Activity. Journal of Fungi. 2021; 7(6):471. https://doi.org/10.3390/jof7060471
Chicago/Turabian StyleAlwakeel, Suaad S., Fuad Ameen, Hussah Al Gwaiz, Hana Sonbol, Salma Alghamdi, Ahmad M. Moharram, and Osama A. Al-Bedak. 2021. "Keratinases Produced by Aspergillus stelliformis, Aspergillus sydowii, and Fusarium brachygibbosum Isolated from Human Hair: Yield and Activity" Journal of Fungi 7, no. 6: 471. https://doi.org/10.3390/jof7060471






