Talaromyces marneffei Infection: Virulence, Intracellular Lifestyle and Host Defense Mechanisms
Abstract
1. Introduction
2. Establishment of T. marneffei Infection
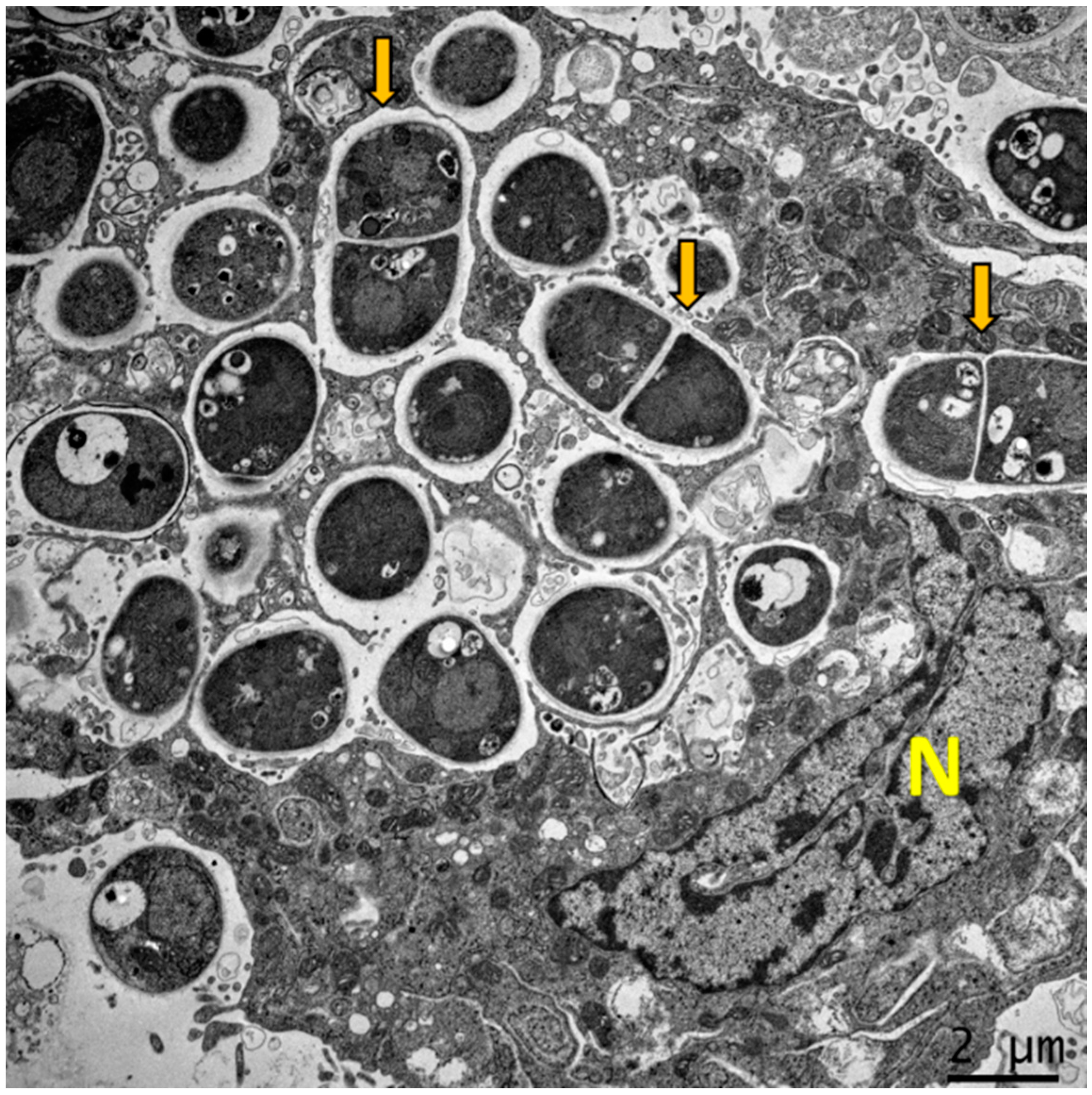
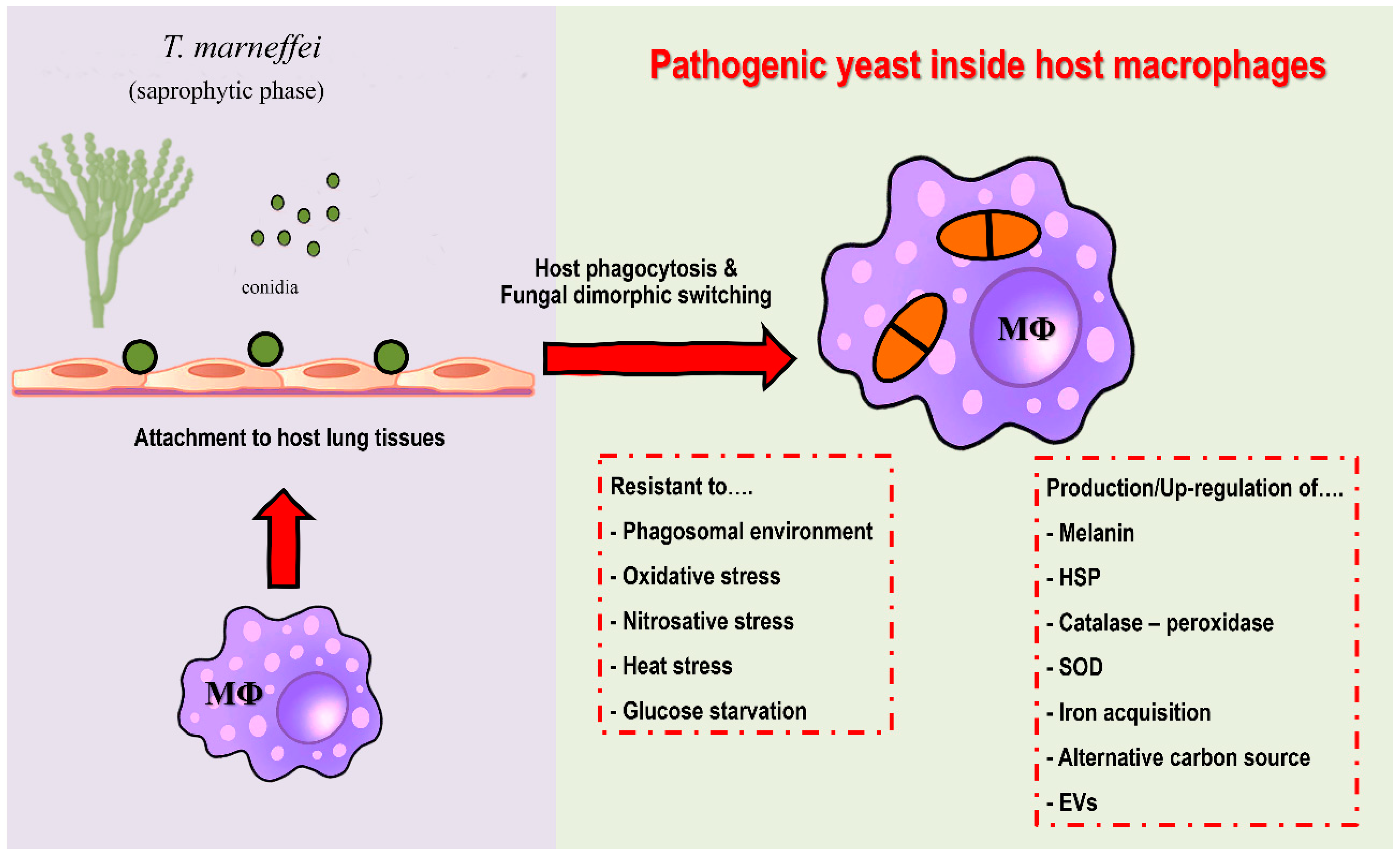
3. Dimorphism and Intracellular Lifestyle of T. marneffei
4. Host Defense Mechanisms
4.1. Innate Immunity
4.2. Acquired Immunity
5. Conclusions and Future Perspective
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Narayanasamy, S.; Dougherty, J.; van Doorn, H.R.; Le, T. Pulmonary talaromycosis: A window into the immunopathogenesis of an endemic mycosis. Mycopathologia 2021, 186, 707–715. [Google Scholar] [CrossRef]
- Segretain, G. Penicillium marneffei n.sp., agent of a mycosis of the reticuloendothelial system. Mycopathol. Mycol. Appl. 1959, 11, 327–353. [Google Scholar] [CrossRef] [PubMed]
- Drouhet, E. Central nervous system mycoses in pediatrics. Minerva Pediatr. 1992, 44, 69–77. [Google Scholar]
- DiSalvo, A.F.; Fickling, A.M.; Ajello, L. Infection caused by Penicillium marneffei: Description of first natural infection in man. Am. J. Clin. Pathol. 1973, 60, 259–263. [Google Scholar] [CrossRef][Green Version]
- Jayanetra, P.; Nitiyanant, P.; Ajello, L.; Padhye, A.A.; Lolekha, S.; Atichartakarn, V.; Vathesatogit, P.; Sathaphatayavongs, B.; Prajaktam, R. Penicilliosis marneffei in Thailand: Report of five human cases. Am. J. Trop. Med. Hyg. 1984, 33, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Supparatpinyo, K.; Khamwan, C.; Baosoung, V.; Nelson, K.E.; Sirisanthana, T. Disseminated Penicillium marneffei infection in southeast Asia. Lancet 1994, 344, 110–113. [Google Scholar] [CrossRef]
- Headley, S.A.; Pretto-Giordano, L.G.; Lima, S.C.; Suhett, W.G.; Pereira, A.H.T.; Freitas, L.A.; Suphoronski, S.A.; Oliveira, T.E.S.; Alfieri, A.F.; Pereira, E.C.; et al. Pneumonia due to Talaromyces marneffei in a dog from Southern Brazil with concomitant canine distemper virus infection. J. Comp. Pathol. 2017, 157, 61–66. [Google Scholar] [CrossRef]
- Capponi, M.; Segretain, G.; Sureau, P. Penicillosis from Rhizomys sinensis. Bull. Soc. Pathol. Exot. Fil. 1956, 49, 418–421. [Google Scholar]
- Deng, Z.L.; Yun, M.; Ajello, L. Human penicilliosis marneffei and its relation to the bamboo rat (Rhizomys pruinosus). J. Med. Vet. Mycol. 1986, 24, 383–389. [Google Scholar] [CrossRef]
- Chariyalertsak, S.; Vanittanakom, P.; Nelson, K.E.; Sirisanthana, T.; Vanittanakom, N. Rhizomys sumatrensis and Cannomys badius, new natural animal hosts of Penicillium marneffei. J. Med. Vet. Mycol. 1996, 34, 105–110. [Google Scholar] [CrossRef]
- Ajello, L.; Padhye, A.A.; Sukroongreung, S.; Nilakul, C.H.; Tantimavanic, S. Occurrence of Penicillium marneffei infections among wild bamboo rats in Thailand. Mycopathologia 1995, 131, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; He, G.; Lu, S.; Liang, Y.; Xi, L. Role of Rhizomys pruinosus as a natural animal host of Penicillium marneffei in Guangdong, China. Microb. Biotechnol. 2015, 8, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Rooney, P.J.; Klein, B.S. Linking fungal morphogenesis with virulence. Cell. Microbiol. 2002, 4, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Vanittanakom, N.; Cooper, C.R., Jr.; Fisher, M.C.; Sirisanthana, T. Penicillium marneffei infection and recent advances in the epidemiology and molecular biology aspects. Clin. Microbiol. Rev. 2006, 19, 95–110. [Google Scholar] [CrossRef]
- Chariyalertsak, S.; Sirisanthana, T.; Supparatpinyo, K.; Nelson, K.E. Seasonal variation of disseminated Penicillium marneffei infections in northern Thailand: A clue to the reservoir? J. Infect. Dis. 1996, 173, 1490–1493. [Google Scholar] [CrossRef]
- Le, T.; Wolbers, M.; Chi, N.H.; Quang, V.M.; Chinh, N.T.; Lan, N.P.; Lam, P.S.; Kozal, M.J.; Shikuma, C.M.; Day, J.N.; et al. Epidemiology, seasonality, and predictors of outcome of AIDS-associated Penicillium marneffei infection in Ho Chi Minh City, Viet Nam. Clin. Infect. Dis. 2011, 52, 945–952. [Google Scholar] [CrossRef]
- Narayanasamy, S.; Dat, V.Q.; Thanh, N.T.; Ly, V.T.; Chan, J.F.; Yuen, K.Y.; Ning, C.; Liang, H.; Li, L.; Chowdhary, A.; et al. A global call for talaromycosis to be recognised as a neglected tropical disease. Lancet Glob. Health 2021, 9, e1618–e1622. [Google Scholar] [CrossRef]
- Chan, Y.F.; Chow, T.C. Ultrastructural observations on Penicillium marneffei in natural human infection. Ultrastruct. Pathol. 1990, 14, 439–452. [Google Scholar] [CrossRef]
- Gilbert, A.S.; Wheeler, R.T.; May, R.C. Fungal pathogens: Survival and replication within macrophages. Cold. Spring Harb. Perspect Med. 2014, 5, a019661. [Google Scholar] [CrossRef]
- Pruksaphon, K.; Ching, M.M.N.; Nosanchuk, J.D.; Kaltsas, A.; Ratanabanangkoon, K.; Roytrakul, S.; Martinez, L.R.; Youngchim, S. Characterization of a novel yeast phase-specific antigen expressed during in vitro thermal phase transition of Talaromyces marneffei. Sci. Rep. 2020, 10, 21169. [Google Scholar] [CrossRef]
- Weerasinghe, H.; Bugeja, H.E.; Andrianopoulos, A. The novel Dbl homology/BAR domain protein, MsgA, of Talaromyces marneffei regulates yeast morphogenesis during growth inside host cells. Sci. Rep. 2021, 11, 2334. [Google Scholar] [CrossRef] [PubMed]
- Pongpom, M.; Vanittanakom, P.; Nimmanee, P.; Cooper, C.R., Jr.; Vanittanakom, N. Adaptation to macrophage killing by Talaromyces marneffei. Future Sci. OA 2017, 3, Fso215. [Google Scholar] [CrossRef] [PubMed]
- Klein, B.S.; Tebbets, B. Dimorphism and virulence in fungi. Curr. Opin. Microbiol. 2007, 10, 314–319. [Google Scholar] [CrossRef]
- Romani, L. Overview of the Fungal Pathogens. In Immunology of Infectious Diseases; ASM Press: Washington, DC, USA, 2001; pp. 25–37. [Google Scholar]
- Walsh, T.J.; Dixon, D.M. Spectrum of Mycoses. In Medical Microbiology, 4th ed.; Baron, S., Ed.; University of Texas Medical Branch at Galveston: Galveston, TX, USA, 1996; Chapter 75. [Google Scholar]
- Lau, S.K.; Tse, H.; Chan, J.S.; Zhou, A.C.; Curreem, S.O.; Lau, C.C.; Yuen, K.Y.; Woo, P.C. Proteome profiling of the dimorphic fungus Penicillium marneffei extracellular proteins and identification of glyceraldehyde-3-phosphate dehydrogenase as an important adhesion factor for conidial attachment. FEBS J. 2013, 280, 6613–6626. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, A.J.; Jeavons, L.; Youngchim, S.; Vanittanakom, N.; Hay, R.J. Sialic acid-dependent recognition of laminin by Penicillium marneffei conidia. Infect. Immun. 1998, 66, 6024–6026. [Google Scholar] [CrossRef]
- Hamilton, A.J.; Jeavons, L.; Youngchim, S.; Vanittanakom, N. Recognition of fibronectin by Penicillium marneffei conidia via a sialic acid-dependent process and its relationship to the interaction between conidia and laminin. Infect. Immun. 1999, 67, 5200–5205. [Google Scholar] [CrossRef]
- Srinoulprasert, Y.; Kongtawelert, P.; Chaiyaroj, S.C. Chondroitin sulfate B and heparin mediate adhesion of Penicillium marneffei conidia to host extracellular matrices. Microb. Pathog. 2006, 40, 126–132. [Google Scholar] [CrossRef]
- Supparatpinyo, K.; Sirisanthana, T. Disseminated Penicillium marneffei infection diagnosed on examination of a peripheral blood smear of a patient with human immunodeficiency virus infection. Clin. Infect. Dis. 1994, 18, 246–247. [Google Scholar] [CrossRef]
- Kudeken, N.; Kawakami, K.; Saito, A. Mechanisms of the in vitro fungicidal effects of human neutrophils against Penicillium marneffei induced by granulocyte-macrophage colony-stimulating factor (GM-CSF). Clin. Exp. Immunol. 2000, 119, 472–478. [Google Scholar] [CrossRef]
- Cogliati, M.; Roverselli, A.; Boelaert, J.R.; Taramelli, D.; Lombardi, L.; Viviani, M.A. Development of an in vitro macrophage system to assess Penicillium marneffei growth and susceptibility to nitric oxide. Infect. Immun. 1997, 65, 279–284. [Google Scholar] [CrossRef]
- Lu, S.; Hu, Y.; Lu, C.; Zhang, J.; Li, X.; Xi, L. Development of in vitro macrophage system to evaluate phagocytosis and intracellular fate of Penicillium marneffei conidia. Mycopathologia 2013, 176, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Ellett, F.; Pazhakh, V.; Pase, L.; Benard, E.L.; Weerasinghe, H.; Azabdaftari, D.; Alasmari, S.; Andrianopoulos, A.; Lieschke, G.J. Macrophages protect Talaromyces marneffei conidia from myeloperoxidase-dependent neutrophil fungicidal activity during infection establishment in vivo. PLoS Pathog. 2018, 14, e1007063. [Google Scholar] [CrossRef] [PubMed]
- Stathakis, A.; Lim, K.P.; Boan, P.; Lavender, M.; Wrobel, J.; Musk, M.; Heath, C.H. Penicillium marneffei infection in a lung transplant recipient. Transpl. Infect. Dis. 2015, 17, 429–434. [Google Scholar] [CrossRef] [PubMed]
- De Monte, A.; Risso, K.; Normand, A.C.; Boyer, G.; L’Ollivier, C.; Marty, P.; Gari-Toussaint, M. Chronic pulmonary penicilliosis due to Penicillium marneffei: Late presentation in a french traveler. J. Travel. Med. 2014, 21, 292–294. [Google Scholar] [CrossRef][Green Version]
- Hart, J.; Dyer, J.R.; Clark, B.M.; McLellan, D.G.; Perera, S.; Ferrari, P. Travel-related disseminated Penicillium marneffei infection in a renal transplant patient. Transpl. Infect. Dis. 2012, 14, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.; Beliavsky, A.; Gough, K. Talaromyces marneffei fungemia after travel to China in a Canadian patient with AIDS. CMAJ 2020, 192, E92–E95. [Google Scholar] [CrossRef]
- Limper, A.H.; Adenis, A.; Le, T.; Harrison, T.S. Fungal infections in HIV/AIDS. Lancet Infect. Dis. 2017, 17, e334–e343. [Google Scholar] [CrossRef]
- Chan, J.F.; Lau, S.K.; Yuen, K.Y.; Woo, P.C. Talaromyces (Penicillium) marneffei infection in non-HIV-infected patients. Emerg. Microbes Infect. 2016, 5, e19. [Google Scholar] [CrossRef]
- Guo, J.; Ning, X.Q.; Ding, J.Y.; Zheng, Y.Q.; Shi, N.N.; Wu, F.Y.; Lin, Y.K.; Shih, H.P.; Ting, H.T.; Liang, G.; et al. Anti-IFN-γ autoantibodies underlie disseminated Talaromyces marneffei infections. J. Exp. Med. 2020, 217, e20190502. [Google Scholar] [CrossRef]
- Wongkulab, P.; Wipasa, J.; Chaiwarith, R.; Supparatpinyo, K. Autoantibody to interferon-gamma associated with adult-onset immunodeficiency in non-HIV individuals in Northern Thailand. PLoS ONE 2013, 8, e76371. [Google Scholar] [CrossRef]
- Le, T.; Huu Chi, N.; Kim Cuc, N.T.; Manh Sieu, T.P.; Shikuma, C.M.; Farrar, J.; Day, J.N. AIDS-associated Penicillium marneffei infection of the central nervous system. Clin. Infect. Dis. 2010, 51, 1458–1462. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Y.; Dong, R.J.; Shrestha, S.; Upadhyay, P.; Li, H.Q.; Kuang, Y.Q.; Yang, X.P.; Zhang, Y.G. AIDS-associated Talaromyces marneffei central nervous system infection in patients of southwestern China. AIDS Res. 2020, 17, 26. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, C.A. Central nervous system Infection with other endemic mycoses: Rare manifestation of blastomycosis, paracoccidioidomycosis, talaromycosis, and sporotrichosis. J. Fungi 2019, 5, 64. [Google Scholar] [CrossRef] [PubMed]
- Karkowska-Kuleta, J.; Rapala-Kozik, M.; Kozik, A. Fungi pathogenic to humans: Molecular bases of virulence of Candida albicans, Cryptococcus neoformans and Aspergillus fumigatus. Acta Biochim. Pol. 2009, 56, 211–224. [Google Scholar] [CrossRef]
- Boral, H.; Metin, B.; Döğen, A.; Seyedmousavi, S.; Ilkit, M. Overview of selected virulence attributes in Aspergillus fumigatus, Candida albicans, Cryptococcus neoformans, Trichophyton rubrum, and Exophiala dermatitidis. Fungal. Genet. Biol. 2018, 111, 92–107. [Google Scholar] [CrossRef]
- Rappleye, C.A.; Goldman, W.E. Defining virulence genes in the dimorphic fungi. Annu. Rev. Microbiol. 2006, 60, 281–303. [Google Scholar] [CrossRef]
- Falkow, S. Molecular Koch’s postulates applied to microbial pathogenicity. Rev. Infect. Dis. 1988, 10 (Suppl. S2), S274–S276. [Google Scholar] [CrossRef]
- Andrianopoulos, A. Control of morphogenesis in the human fungal pathogen Penicillium marneffei. Int. J. Med. Microbiol. 2002, 292, 331–347. [Google Scholar] [CrossRef]
- Cooper, C.R.; Vanittanakom, N. Insights into the pathogenicity of Penicillium marneffei. Future Microbiol. 2008, 3, 43–55. [Google Scholar] [CrossRef]
- Boyce, K.J.; Andrianopoulos, A. Fungal dimorphism: The switch from hyphae to yeast is a specialized morphogenetic adaptation allowing colonization of a host. FEMS Microbiol. Rev. 2015, 39, 797–811. [Google Scholar] [CrossRef]
- Bugeja, H.E.; Boyce, K.J.; Weerasinghe, H.; Beard, S.; Jeziorowski, A.; Pasricha, S.; Payne, M.; Schreider, L.; Andrianopoulos, A. Tools for high efficiency genetic manipulation of the human pathogen Penicillium marneffei. Fungal. Genet. Biol. 2012, 49, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Hu, X.; Jan, S.; Rasheed, S.M.; Zhang, Y.; Du, M.; Yang, E. Development of CRISPR-Cas9 genome editing system in Talaromyces marneffei. Microb. Pathog. 2021, 154, 104822. [Google Scholar] [CrossRef] [PubMed]
- Van Dyke, M.C.C.; Teixeira, M.M.; Barker, B.M. Fantastic yeasts and where to find them: The hidden diversity of dimorphic fungal pathogens. Curr. Opin. Microbiol. 2019, 52, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, G.M. Dimorphism in fungal pathogens of mammals, plants, and insects. PLoS Pathog. 2015, 11, e1004608. [Google Scholar] [CrossRef]
- Heung, L.J. Monocytes and the host response to fungal pathogens. Front. Cell. Infect. Microbiol. 2020, 10, 34. [Google Scholar] [CrossRef]
- Cao, C.; Li, R.; Wan, Z.; Liu, W.; Wang, X.; Qiao, J.; Wang, D.; Bulmer, G.; Calderone, R. The effects of temperature, pH, and salinity on the growth and dimorphism of Penicillium marneffei. Med. Mycol. 2007, 45, 401–407. [Google Scholar] [CrossRef]
- Boyce, K.J.; Andrianopoulos, A. Morphogenetic circuitry regulating growth and development in the dimorphic pathogen Penicillium marneffei. Eukaryot. Cell 2013, 12, 154–160. [Google Scholar] [CrossRef][Green Version]
- Mead, M.E.; Borowsky, A.T.; Joehnk, B.; Steenwyk, J.L.; Shen, X.X.; Sil, A.; Rokas, A. Recurrent loss of abaA, a master regulator of asexual development in filamentous fungi, correlates with changes in genomic and morphological traits. Genome Biol. Evol. 2020, 12, 1119–1130. [Google Scholar] [CrossRef]
- Borneman, A.R.; Hynes, M.J.; Andrianopoulos, A. The abaA homologue of Penicillium marneffei participates in two developmental programmes: Conidiation and dimorphic growth. Mol. Microbiol. 2000, 38, 1034–1047. [Google Scholar] [CrossRef]
- Boyce, K.J.; Schreider, L.; Kirszenblat, L.; Andrianopoulos, A. The two-component histidine kinases DrkA and SlnA are required for in vivo growth in the human pathogen Penicillium marneffei. Mol. Microbiol. 2011, 82, 1164–1184. [Google Scholar] [CrossRef]
- Boyce, K.J.; Hynes, M.J.; Andrianopoulos, A. The CDC42 homolog of the dimorphic fungus Penicillium marneffei is required for correct cell polarization during growth but not development. J. Bacteriol. 2001, 183, 3447–3457. [Google Scholar] [CrossRef] [PubMed]
- Boyce, K.J.; Hynes, M.J.; Andrianopoulos, A. The Ras and Rho GTPases genetically interact to co-ordinately regulate cell polarity during development in Penicillium marneffei. Mol. Microbiol. 2005, 55, 1487–1501. [Google Scholar] [CrossRef] [PubMed]
- Boyce, K.J.; Andrianopoulos, A. A p21-activated kinase is required for conidial germination in Penicillium marneffei. PLoS Pathog. 2007, 3, e162. [Google Scholar] [CrossRef] [PubMed]
- Boyce, K.J.; Schreider, L.; Andrianopoulos, A. In vivo yeast cell morphogenesis is regulated by a p21-activated kinase in the human pathogen Penicillium marneffei. PLoS Pathog. 2009, 5, e1000678. [Google Scholar] [CrossRef]
- Fridovich, I. Oxygen toxicity: A radical explanation. J. Exp. Biol. 1998, 201, 1203–1209. [Google Scholar] [CrossRef]
- Missall, T.A.; Lodge, J.K.; McEwen, J.E. Mechanisms of resistance to oxidative and nitrosative stress: Implications for fungal survival in mammalian hosts. Eukaryot. Cell 2004, 3, 835–846. [Google Scholar] [CrossRef]
- Youngchim, S.; Vanittanakom, N.; Hamilton, A.J. Analysis of the enzymatic activity of mycelial and yeast phases of Penicillium marneffei. Med. Mycol. 1999, 37, 445–450. [Google Scholar] [CrossRef]
- Taramelli, D.; Tognazioli, C.; Ravagnani, F.; Leopardi, O.; Giannulis, G.; Boelaert, J.R. Inhibition of intramacrophage growth of Penicillium marneffei by 4-aminoquinolines. Antimicrob. Agents Chemother. 2001, 45, 1450–1455. [Google Scholar] [CrossRef]
- Unissa, A.N.; Doss, C.G.; Kumar, T.; Sukumar, S.; Lakshmi, A.R.; Hanna, L.E. Significance of catalase-peroxidase (KatG) mutations in mediating isoniazid resistance in clinical strains of Mycobacterium tuberculosis. J. Glob. Antimicrob. Resist. 2018, 15, 111–120. [Google Scholar] [CrossRef]
- Paris, S.; Wysong, D.; Debeaupuis, J.P.; Shibuya, K.; Philippe, B.; Diamond, R.D.; Latgé, J.P. Catalases of Aspergillus fumigatus. Infect. Immun. 2003, 71, 3551–3562. [Google Scholar] [CrossRef]
- Pongpom, P.; Cooper, C.R., Jr.; Vanittanakom, N. Isolation and characterization of a catalase-peroxidase gene from the pathogenic fungus, Penicillium marneffei. Med. Mycol. 2005, 43, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Pongpom, M.; Sawatdeechaikul, P.; Kummasook, A.; Khanthawong, S.; Vanittanakom, N. Antioxidative and immunogenic properties of catalase-peroxidase protein in Penicillium marneffei. Med. Mycol. 2013, 51, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Thirach, S.; Cooper, C.R., Jr.; Vanittanakom, P.; Vanittanakom, N. The copper, zinc superoxide dismutase gene of Penicillium marneffei: Cloning, characterization, and differential expression during phase transition and macrophage infection. Med. Mycol. 2007, 45, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Haas, A. The phagosome: Compartment with a license to kill. Traffic 2007, 8, 311–330. [Google Scholar] [CrossRef] [PubMed]
- Thirach, S.; Cooper, C.R.; Vanittanakom, N. Molecular analysis of the Penicillium marneffei glyceraldehyde-3-phosphate dehydrogenase-encoding gene (gpdA) and differential expression of gpdA and the isocitrate lyase-encoding gene (acuD) upon internalization by murine macrophages. J. Med. Microbiol. 2008, 57, 1322–1328. [Google Scholar] [CrossRef] [PubMed]
- Cánovas, D.; Andrianopoulos, A. Developmental regulation of the glyoxylate cycle in the human pathogen Penicillium marneffei. Mol. Microbiol. 2006, 62, 1725–1738. [Google Scholar] [CrossRef] [PubMed]
- Pasricha, S.; Schafferer, L.; Lindner, H.; Joanne Boyce, K.; Haas, H.; Andrianopoulos, A. Differentially regulated high-affinity iron assimilation systems support growth of the various cell types in the dimorphic pathogen Talaromyces marneffei. Mol. Microbiol. 2016, 102, 715–737. [Google Scholar] [CrossRef]
- Taramelli, D.; Brambilla, S.; Sala, G.; Bruccoleri, A.; Tognazioli, C.; Riviera-Uzielli, L.; Boelaert, J.R. Effects of iron on extracellular and intracellular growth of Penicillium marneffei. Infect. Immun. 2000, 68, 1724–1726. [Google Scholar] [CrossRef]
- Pongpom, M.; Amsri, A.; Sukantamala, P.; Suwannaphong, P.; Jeenkeawpieam, J. Expression of Talaromyces marneffei acuM and acuK Genes in gluconeogenic substrates and various iron concentrations. J. Fungi 2020, 6, 102. [Google Scholar] [CrossRef]
- Amsri, A.; Jeenkeawpieam, J.; Sukantamala, P.; Pongpom, M. Role of acuK in control of iron acquisition and gluconeogenesis in Talaromyces marneffei. J. Fungi 2021, 7, 798. [Google Scholar] [CrossRef]
- Tiwari, S.; Thakur, R.; Shankar, J. Role of heat-shock proteins in cellular function and in the biology of fungi. Biotechnol. Res. Int. 2015, 2015, 132635. [Google Scholar] [CrossRef] [PubMed]
- Kummasook, A.; Pongpom, P.; Vanittanakom, N. Cloning, characterization and differential expression of an hsp70 gene from the pathogenic dimorphic fungus, Penicillium marneffei. DNA Seq. 2007, 18, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Vanittanakom, N.; Pongpom, M.; Praparattanapan, J.; Cooper, C.R.; Sirisanthana, T. Isolation and expression of heat shock protein 30 gene from Penicillium marneffei. Med. Mycol. 2009, 47, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, M.H.; Bell, A.A. Melanins and their importance in pathogenic fungi. Curr. Top. Med. Mycol. 1988, 2, 338–387. [Google Scholar] [CrossRef] [PubMed]
- Taborda, C.P.; da Silva, M.B.; Nosanchuk, J.D.; Travassos, L.R. Melanin as a virulence factor of Paracoccidioides brasiliensis and other dimorphic pathogenic fungi: A minireview. Mycopathologia 2008, 165, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Youngchim, S.; Zamith-Miranda, D.; Nosanchuk, J.D. Fungal melanin and the mammalian immune system. J. Fungi 2021, 7, 264. [Google Scholar] [CrossRef] [PubMed]
- Nosanchuk, J.D.; Casadevall, A. Impact of melanin on microbial virulence and clinical resistance to antimicrobial compounds. Antimicrob. Agents Chemother. 2006, 50, 3519–3528. [Google Scholar] [CrossRef]
- Youngchim, S.; Hay, R.J.; Hamilton, A.J. Melanization of Penicillium marneffei in vitro and in vivo. Microbiology 2005, 151, 291–299. [Google Scholar] [CrossRef]
- Tam, E.W.; Tsang, C.C.; Lau, S.K.; Woo, P.C. Polyketides, toxins and pigments in Penicillium marneffei. Toxins 2015, 7, 4421–4436. [Google Scholar] [CrossRef]
- Woo, P.C.; Tam, E.W.; Chong, K.T.; Cai, J.J.; Tung, E.T.; Ngan, A.H.; Lau, S.K.; Yuen, K.Y. High diversity of polyketide synthase genes and the melanin biosynthesis gene cluster in Penicillium marneffei. FEBS J. 2010, 277, 3750–3758. [Google Scholar] [CrossRef]
- Thurston, C.F. The structure and function of fungal laccases. Microbiology 1994, 1, 19–26. [Google Scholar] [CrossRef]
- Sapmak, A.; Boyce, K.J.; Andrianopoulos, A.; Vanittanakom, N. The pbrB gene encodes a laccase required for DHN-melanin synthesis in conidia of Talaromyces (Penicillium) marneffei. PLoS ONE 2015, 10, e0122728. [Google Scholar] [CrossRef] [PubMed]
- Sapmak, A.; Kaewmalakul, J.; Nosanchuk, J.D.; Vanittanakom, N.; Andrianopoulos, A.; Pruksaphon, K.; Youngchim, S. Talaromyces marneffei laccase modifies THP-1 macrophage responses. Virulence 2016, 7, 702–717. [Google Scholar] [CrossRef] [PubMed]
- Bugeja, H.E.; Hynes, M.J.; Andrianopoulos, A. The RFX protein RfxA is an essential regulator of growth and morphogenesis in Penicillium marneffei. Eukaryot. Cell 2010, 9, 578–591. [Google Scholar] [CrossRef] [PubMed]
- Suwunnakorn, S.; Cooper, C.R.; Kummasook, A.; Vanittanakom, N. Role of the yakA Gene in Morphogenesis and Stress Response in Penicillium marneffei. Microbiology 2014, 160, 1929–1939. [Google Scholar] [CrossRef]
- Suwunnakorn, S.; Cooper, C.R., Jr.; Kummasook, A.; Pongpom, M.; Vanittanakom, P.; Vanittanakom, N. Role of the rttA gene in morphogenesis, stress response, and virulence in the human pathogenic fungus Penicillium marneffei. Med. Mycol. 2015, 53, 119–131. [Google Scholar] [CrossRef][Green Version]
- Nimmanee, P.; Woo, P.C.; Vanittanakom, P.; Youngchim, S.; Vanittanakom, N. Functional analysis of atfA gene to stress response in pathogenic thermal dimorphic fungus Penicillium marneffei. PLoS ONE 2014, 9, e111200. [Google Scholar] [CrossRef]
- Nimmanee, P.; Woo, P.C.; Kummasook, A.; Vanittanakom, N. Characterization of sakA gene from pathogenic dimorphic fungus Penicillium marneffei. Int. J. Med. Microbiol. 2015, 305, 65–74. [Google Scholar] [CrossRef]
- Feng, J.; He, L.; Xiao, X.; Chen, Z.; Chen, C.; Chu, J.; Lu, S.; Li, X.; Mylonakis, E.; Xi, L. Methylcitrate cycle gene MCD is essential for the virulence of Talaromyces marneffei. Med. Mycol. 2020, 58, 351–361. [Google Scholar] [CrossRef]
- Rizzo, J.; Rodrigues, M.L.; Janbon, G. Extracellular vesicles in fungi: Past, present, and future perspectives. Front. Cell. Infect. Microbiol. 2020, 10, 346. [Google Scholar] [CrossRef]
- Wolf, J.M.; Casadevall, A. Challenges posed by extracellular vesicles from eukaryotic microbes. Curr. Opin. Microbiol. 2014, 22, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Wang, J.; Jiang, H.; Lin, H.; Ou, Z.; Ullah, A.; Hua, Y.; Chen, J.; Lin, X.; Hu, X.; et al. Extracellular vesicles derived from Talaromyces marneffei yeasts mediate inflammatory response in macrophage cells by bioactive protein components. Front. Microbiol. 2020, 11, 603183. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, L.; Xiao, S.; Chen, W.; Hu, F.; Li, F.; Guo, P.; Chen, X.; Cai, W.; Tang, X. The association of TLR2, TLR3, and TLR9 gene polymorphisms with susceptibility to talaromycosis among han Chinese AIDS patients in Guangdong. Front. Cell. Infect. Microbiol. 2021, 11, 625461. [Google Scholar] [CrossRef] [PubMed]
- Srinoulprasert, Y.; Pongtanalert, P.; Chawengkirttikul, R.; Chaiyaroj, S.C. Engagement of Penicillium marneffei conidia with multiple pattern recognition receptors on human monocytes. Microbiol. Immunol. 2009, 53, 162–172. [Google Scholar] [CrossRef]
- Kudeken, N.; Kawakami, K.; Saito, A. Cytokine-induced fungicidal activity of human polymorphonuclear leukocytes against Penicillium marneffei. FEMS Immunol. Med. Microbiol. 1999, 26, 115–124. [Google Scholar] [CrossRef]
- Tang, Y.; Zhang, H.; Xu, H.; Zeng, W.; Qiu, Y.; Tan, C.; Tang, S.; Zhang, J. Dendritic cells promote Treg expansion but not Th17 generation in response to Talaromyces marneffei yeast cells. Infect. Drug Resist. 2020, 13, 805–813. [Google Scholar] [CrossRef]
- Roilides, E.; Lyman, C.A.; Sein, T.; Petraitiene, R.; Walsh, T.J. Macrophage colony-stimulating factor enhances phagocytosis and oxidative burst of mononuclear phagocytes against Penicillium marneffei conidia. FEMS Immunol. Med. Microbiol. 2003, 36, 19–26. [Google Scholar] [CrossRef]
- He, J.; Li, J.S.; Xu, H.Y.; Kuang, Y.Q.; Li, J.; Li, H.B.; Li, Z.; Zhou, H.L.; Wang, R.R.; Li, Y.Y. A Reliable murine model of disseminated infection induced by Talaromyces marneffei. Mycopathologia 2022, 187, 53–64. [Google Scholar] [CrossRef]
- Kudeken, N.; Kawakami, K.; Saito, A. Different susceptibilities of yeasts and conidia of Penicillium marneffei to nitric oxide (NO)-mediated fungicidal activity of murine macrophages. Clin. Exp. Immunol. 1998, 112, 287–293. [Google Scholar] [CrossRef]
- Rongrungruang, Y.; Levitz, S.M. Interactions of Penicillium marneffei with human leukocytes in vitro. Infect. Immun. 1999, 67, 4732–4736. [Google Scholar] [CrossRef]
- Othman, J.; Brown, C.M. Talaromyces marneffei and dysplastic neutrophils on blood smear in newly diagnosed HIV. Blood 2018, 131, 269. [Google Scholar] [CrossRef] [PubMed]
- Klebanoff, S.J. Antimicrobial mechanisms in neutrophilic polymorphonuclear leukocytes. Semin. Hematol. 1975, 12, 117–142. [Google Scholar] [PubMed]
- Linnerz, T.; Hall, C.J. The diverse roles of phagocytes during bacterial and fungal infections and sterile inflammation: Lessons from zebrafish. Front. Immunol. 2020, 11, 1094. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Khan, F.A.; Pandupuspitasari, N.S.; Zhang, S. Immune evasion strategies of pathogens in macrophages: The potential for limiting pathogen transmission. Curr. Issues Mol. Biol. 2017, 21, 21–40. [Google Scholar] [PubMed]
- Lawrence, T.; Natoli, G. Transcriptional regulation of macrophage polarization: Enabling diversity with identity. Nat. Rev. Immunol. 2011, 11, 750–761. [Google Scholar] [CrossRef]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000Prime Rep. 2014, 6, 13. [Google Scholar] [CrossRef]
- Wagener, J.; MacCallum, D.M.; Brown, G.D.; Gow, N.A. Candida albicans chitin increases arginase-1 activity in human macrophages, with an impact on macrophage antimicrobial functions. mBio 2017, 8, e01820-16. [Google Scholar] [CrossRef]
- Heung, L.J.; Hohl, T.M. Inflammatory monocytes are detrimental to the host immune response during acute infection with Cryptococcus neoformans. PLoS Pathog. 2019, 15, e1007627. [Google Scholar] [CrossRef]
- Dai, X.; Mao, C.; Lan, X.; Chen, H.; Li, M.; Bai, J.; Deng, J.; Liang, Q.; Zhang, J.; Zhong, X.; et al. Acute Penicillium marneffei infection stimulates host M1/M2a macrophages polarization in BALB/c mice. BMC Microbiol. 2017, 17, 177. [Google Scholar] [CrossRef]
- Chen, R.; Xi, L.; Huang, X.; Ma, T.; Ren, H.; Ji, G. Effect of Jun N-terminal kinase 1 and 2 on the replication of Penicillium marneffei in human macrophages. Microb. Pathog. 2015, 82, 1–6. [Google Scholar] [CrossRef]
- Yang, D.; Shen, L.X.; Chen, R.F.; Fu, Y.; Xu, H.Y.; Zhang, L.N.; Liu, D.H. The Effect of Talaromyces marneffei Infection on CD86 Expression in THP-1 Cells. Infect. Drug Resist. 2021, 14, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Ning, C.; Huang, J.; Wang, G.; Lai, J.; Han, J.; He, J.; Zhang, H.; Liang, B.; Liao, Y.; et al. Talaromyces marneffei promotes M2-like polarization of human macrophages by downregulating SOCS3 expression and activating the TLR9 pathway. Virulence 2021, 12, 1997–2012. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Li, X.; Lu, S.; Ma, T.; Huang, X.; Mylonakis, E.; Liang, Y.; Xi, L. Role of extracellular signal-regulated kinases 1 and 2 and p38 mitogen-activated protein kinase pathways in regulating replication of Penicillium marneffei in human macrophages. Microbes Infect. 2014, 16, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, H.; Li, S.; Li, Y.; Liu, G.; Bai, J.; Luo, H.; Lan, X.; He, Z. LncSSBP1 functions as a negative regulator of IL-6 through interaction with hnRNPK in bronchial epithelial cells infected with Talaromyces marneffei. Front. Immunol. 2019, 10, 2977. [Google Scholar] [CrossRef]
- Sze, K.H.; Lam, W.H.; Zhang, H.; Ke, Y.H.; Tse, M.K.; Woo, P.C.Y.; Lau, S.K.P.; Lau, C.C.Y.; Cai, J.P.; Tung, E.T.K.; et al. Talaromyces marneffei Mp1p is a virulence factor that binds and sequesters a key proinflammatory lipid to dampen host innate immune response. Cell. Chem. Biol. 2017, 24, 182–194. [Google Scholar] [CrossRef]
- Lam, W.H.; Sze, K.H.; Ke, Y.; Tse, M.K.; Zhang, H.; Woo, P.C.Y.; Lau, S.K.P.; Lau, C.C.Y.; Xu, S.; Lai, P.M.; et al. Talaromyces marneffei Mp1 protein, a novel virulence factor, carries two arachidonic acid-binding domains to suppress inflammatory responses in hosts. Infect. Immun. 2019, 87, e00679-18. [Google Scholar] [CrossRef]
- Lin, J.S.; Yang, C.W.; Wang, D.W.; Wu-Hsieh, B.A. Dendritic cells cross-present exogenous fungal antigens to stimulate a protective CD8 T cell response in infection by Histoplasma capsulatum. J. Immunol. 2005, 174, 6282–6291. [Google Scholar] [CrossRef]
- Nakamura, K.; Miyazato, A.; Koguchi, Y.; Adachi, Y.; Ohno, N.; Saijo, S.; Iwakura, Y.; Takeda, K.; Akira, S.; Fujita, J.; et al. Toll-like receptor 2 (TLR2) and dectin-1 contribute to the production of IL-12p40 by bone marrow-derived dendritic cells infected with Penicillium marneffei. Microbes Infect. 2008, 10, 1223–1227. [Google Scholar] [CrossRef]
- Qin, Y.; Li, Y.; Liu, W.; Tian, R.; Guo, Q.; Li, S.; Li, H.; Zhang, D.; Zheng, Y.; Wu, L.; et al. Penicillium marneffei-stimulated dendritic cells enhance HIV-1 trans-infection and promote viral infection by activating primary CD4+ T cells. PLoS ONE 2011, 6, e27609. [Google Scholar] [CrossRef]
- Dong, R.J.; Zhang, Y.G.; Zhu, L.; Liu, H.L.; Liu, J.; Kuang, Y.Q.; Wang, R.R.; Li, Y.Y. Innate immunity acts as the major regulator in Talaromyces marneffei coinfected AIDS patients: Cytokine profile surveillance during initial 6-month antifungal therapy. Open Forum. Infect. Dis. 2019, 6, ofz205. [Google Scholar] [CrossRef]
- Ma, H.; Chan, J.F.W.; Tan, Y.P.; Kui, L.; Tsang, C.C.; Pei, S.L.C.; Lau, Y.L.; Woo, P.C.Y.; Lee, P.P. NLRP3 inflammasome contributes to host defense against Talaromyces marneffei infection. Front. Immunol. 2021, 12, 760095. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Tanaka, R.; Taguchi, H.; Sano, A.; Ito, E.; Fukushima, K.; Takeo, K.; Yoshida, S.; Nishimura, K.; Miyaji, M. Histopathological and electron microscopical studies on experimental Penicillium marneffei infection in mice. J. Med. Vet. Mycol. 1997, 35, 347–353. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kudeken, N.; Kawakami, K.; Kusano, N.; Saito, A. Cell-mediated immunity in host resistance against infection caused by Penicillium marneffei. J. Med. Vet. Mycol. 1996, 34, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Yousukh, A.; Jutavijittum, P.; Pisetpongsa, P.; Chitapanarux, T.; Thongsawat, S.; Senba, M.; Toriyama, K. Clinicopathologic study of hepatic Penicillium marneffei in Northern Thailand. Arch. Pathol. Lab. Med. 2004, 128, 191–194. [Google Scholar] [CrossRef]
- Tsui, W.M.; Ma, K.F.; Tsang, D.N. Disseminated Penicillium marneffei infection in HIV-infected subject. Histopathology 1992, 20, 287–293. [Google Scholar] [CrossRef]
- Tang, B.S.; Chan, J.F.; Chen, M.; Tsang, O.T.; Mok, M.Y.; Lai, R.W.; Lee, R.; Que, T.L.; Tse, H.; Li, I.W.; et al. Disseminated penicilliosis, recurrent bacteremic nontyphoidal salmonellosis, and burkholderiosis associated with acquired immunodeficiency due to autoantibody against gamma interferon. Clin. Vaccine Immunol. 2010, 17, 1132–1138. [Google Scholar] [CrossRef]
- Browne, S.K.; Burbelo, P.D.; Chetchotisakd, P.; Suputtamongkol, Y.; Kiertiburanakul, S.; Shaw, P.A.; Kirk, J.L.; Jutivorakool, K.; Zaman, R.; Ding, L.; et al. Adult-onset immunodeficiency in Thailand and Taiwan. N. Engl. J. Med. 2012, 367, 725–734. [Google Scholar] [CrossRef]
- Lee, P.P.; Lao-Araya, M.; Yang, J.; Chan, K.W.; Ma, H.; Pei, L.C.; Kui, L.; Mao, H.; Yang, W.; Zhao, X.; et al. Application of flow cytometry in the diagnostics pipeline of primary immunodeficiencies underlying disseminated Talaromyces marneffei infection in HIV-negative children. Front. Immunol. 2019, 10, 2189. [Google Scholar] [CrossRef]
- Sisto, F.; Miluzio, A.; Leopardi, O.; Mirra, M.; Boelaert, J.R.; Taramelli, D. Differential cytokine pattern in the spleens and livers of BALB/c mice infected with Penicillium marneffei: Protective role of gamma interferon. Infect. Immun. 2003, 71, 465–473. [Google Scholar] [CrossRef]
- Lyadova, I.V.; Panteleev, A.V. Th1 and Th17 cells in tuberculosis: Protection, pathology, and biomarkers. Mediat. Inflamm. 2015, 2015, 854507. [Google Scholar] [CrossRef]
- McDermott, A.J.; Klein, B.S. Helper T-cell responses and pulmonary fungal infections. Immunology 2018, 155, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Pan, F.; Fan, H.; Lu, L.; Liu, Z.; Jiang, S. The yin and yang of signaling in Tregs and TH17 cells. Sci. Signal. 2011, 4, mr4. [Google Scholar] [CrossRef] [PubMed]
- Romani, L. Immunity to fungal infections. Nat. Rev. Immunol. 2011, 11, 275–288. [Google Scholar] [CrossRef] [PubMed]
- Kroetz, D.N.; Deepe, G.S., Jr. CCR5 dictates the equilibrium of proinflammatory IL-17+ and regulatory Foxp3+ T cells in fungal infection. J. Immunol. 2010, 184, 5224–5231. [Google Scholar] [CrossRef] [PubMed]
- Galdino, N.A.L.; Loures, F.V.; de Araújo, E.F.; da Costa, T.A.; Preite, N.W.; Calich, V.L.G. Depletion of regulatory T cells in ongoing paracoccidioidomycosis rescues protective Th1/Th17 immunity and prevents fatal disease outcome. Sci. Rep. 2018, 8, 16544. [Google Scholar] [CrossRef]
- Wong, S.S.; Wong, K.H.; Hui, W.T.; Lee, S.S.; Lo, J.Y.; Cao, L.; Yuen, K.Y. Differences in clinical and laboratory diagnostic characteristics of penicilliosis marneffei in human immunodeficiency virus (HIV)- and non-HIV-infected patients. J. Clin. Microbiol. 2001, 39, 4535–4540. [Google Scholar] [CrossRef][Green Version]
- Mihu, M.R.; Nosanchuk, J.D. Histoplasma virulence and host responses. Int. J. Microbiol. 2012, 2012, 268123. [Google Scholar] [CrossRef]
- Wong, L.P.; Woo, P.C.; Wu, A.Y.; Yuen, K.Y. DNA immunization using a secreted cell wall antigen Mp1p is protective against Penicillium marneffei infection. Vaccine 2002, 20, 2878–2886. [Google Scholar] [CrossRef]
- Pruksaphon, K.; Intaramat, A.; Ratanabanangkoon, K.; Nosanchuk, J.D.; Vanittanakom, N.; Youngchim, S. Diagnostic laboratory immunology for talaromycosis (penicilliosis): Review from the bench-top techniques to the point-of-care testing. Diagn. Microbiol. Infect. Dis. 2020, 96, 114959. [Google Scholar] [CrossRef]
- Jeavons, L.; Hamilton, A.J.; Vanittanakom, N.; Ungpakorn, R.; Evans, E.G.; Sirisanthana, T.; Hay, R.J. Identification and purification of specific Penicillium marneffei antigens and their recognition by human immune sera. J. Clin. Microbiol. 1998, 36, 949–954. [Google Scholar] [CrossRef]
- Cao, L.; Chen, D.L.; Lee, C.; Chan, C.M.; Chan, K.M.; Vanittanakom, N.; Tsang, D.N.; Yuen, K.Y. Detection of specific antibodies to an antigenic mannoprotein for diagnosis of Penicillium marneffei penicilliosis. J. Clin. Microbiol. 1998, 36, 3028–3031. [Google Scholar] [CrossRef] [PubMed]
- Pongpom, M.; Vanittanakom, N. Characterization of an MPLP6, a gene coding for a yeast phase specific, antigenic mannoprotein in Penicillium marneffei. Med. Mycol. 2011, 49, 32–39. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Y.F.; Cai, J.P.; Wang, Y.D.; Dong, H.; Hao, W.; Jiang, L.X.; Long, J.; Chan, C.; Woo, P.C.; Lau, S.K.; et al. Immunoassays based on Penicillium marneffei Mp1p derived from Pichia pastoris expression system for diagnosis of penicilliosis. PLoS ONE 2011, 6, e28796. [Google Scholar] [CrossRef] [PubMed]
- Chongtrakool, P.; Chaiyaroj, S.C.; Vithayasai, V.; Trawatcharegon, S.; Teanpaisan, R.; Kalnawakul, S.; Sirisinha, S. Immunoreactivity of a 38-kilodalton Penicillium marneffei antigen with human immunodeficiency virus-positive sera. J. Clin. Microbiol. 1997, 35, 2220–2223. [Google Scholar] [CrossRef]
- Parker, D.C. T cell-dependent B cell activation. Annu. Rev. Immunol. 1993, 11, 331–360. [Google Scholar] [CrossRef]
- Kaw, S.; Ananth, S.; Tsopoulidis, N.; Morath, K.; Coban, B.M.; Hohenberger, R.; Bulut, O.C.; Klein, F.; Stolp, B.; Fackler, O.T. HIV-1 infection of CD4 T cells impairs antigen-specific B cell function. EMBO J. 2020, 39, e105594. [Google Scholar] [CrossRef]
- Arvanitis, M.; Anagnostou, T.; Fuchs, B.B.; Caliendo, A.M.; Mylonakis, E. Molecular and nonmolecular diagnostic methods for invasive fungal infections. Clin. Microbiol. Rev. 2014, 27, 490–526. [Google Scholar] [CrossRef]
- Tsang, C.C.; Lau, S.K.P.; Woo, P.C.Y. Sixty years from Segretain’s description: What have we learned and should learn about the basic mycology of Talaromyces marneffei? Mycopathologia 2019, 184, 721–729. [Google Scholar] [CrossRef]
- Giusiano, G. The Trojan horse model in Paracoccidioides: A fantastic pathway to survive infecting human cells. Front. Cell. Infect. Microbiol. 2021, 11, 605679. [Google Scholar] [CrossRef]
- Nosanchuk, J.D. Protective antibodies and endemic dimorphic fungi. Curr. Mol. Med. 2005, 5, 435–442. [Google Scholar] [CrossRef]
- Garner, W.; Samanta, P.; Haidar, G. Invasive fungal infections after anti-CD19 chimeric antigen receptor-modified T-cell therapy: State of the evidence and future directions. J. Fungi 2021, 23, 156. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pruksaphon, K.; Nosanchuk, J.D.; Ratanabanangkoon, K.; Youngchim, S. Talaromyces marneffei Infection: Virulence, Intracellular Lifestyle and Host Defense Mechanisms. J. Fungi 2022, 8, 200. https://doi.org/10.3390/jof8020200
Pruksaphon K, Nosanchuk JD, Ratanabanangkoon K, Youngchim S. Talaromyces marneffei Infection: Virulence, Intracellular Lifestyle and Host Defense Mechanisms. Journal of Fungi. 2022; 8(2):200. https://doi.org/10.3390/jof8020200
Chicago/Turabian StylePruksaphon, Kritsada, Joshua D. Nosanchuk, Kavi Ratanabanangkoon, and Sirida Youngchim. 2022. "Talaromyces marneffei Infection: Virulence, Intracellular Lifestyle and Host Defense Mechanisms" Journal of Fungi 8, no. 2: 200. https://doi.org/10.3390/jof8020200
APA StylePruksaphon, K., Nosanchuk, J. D., Ratanabanangkoon, K., & Youngchim, S. (2022). Talaromyces marneffei Infection: Virulence, Intracellular Lifestyle and Host Defense Mechanisms. Journal of Fungi, 8(2), 200. https://doi.org/10.3390/jof8020200







