High-Throughput Microbial Community Analyses to Establish a Natural Fungal and Bacterial Consortium from Sewage Sludge Enriched with Three Pharmaceutical Compounds
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Selective Pressure Experiments
2.2.1. Setting the Flask Content for Selective Pressure Experiments
2.2.2. Setting the Pharmaceutical Pressure of the Flask Experiment
2.3. Analysis of Fungal and Bacterial Communities by Illumina MiSeq Sequencing
2.3.1. Sample Preparation and DNA Isolation
2.3.2. Sequencing Analysis
2.3.3. Data and Bioinformatic Analysis
2.4. Fungal and Bacterial Strains Isolation after Selective Pressure Experiment
2.4.1. Isolation and Molecular Identification of Fungal and Bacterial Strains
2.4.2. Biodegradation Experiments of the Isolated Microorganisms
2.5. Chromatographic Analyses
2.5.1. Characterization of Emerging Contaminants by LC/MS-QTOF in Sewage Sludge Samples
2.5.2. HPLC Analyses of the Removal of PhACs during Pressure Experiment and Biodegradation Experiments
3. Results and Discussion
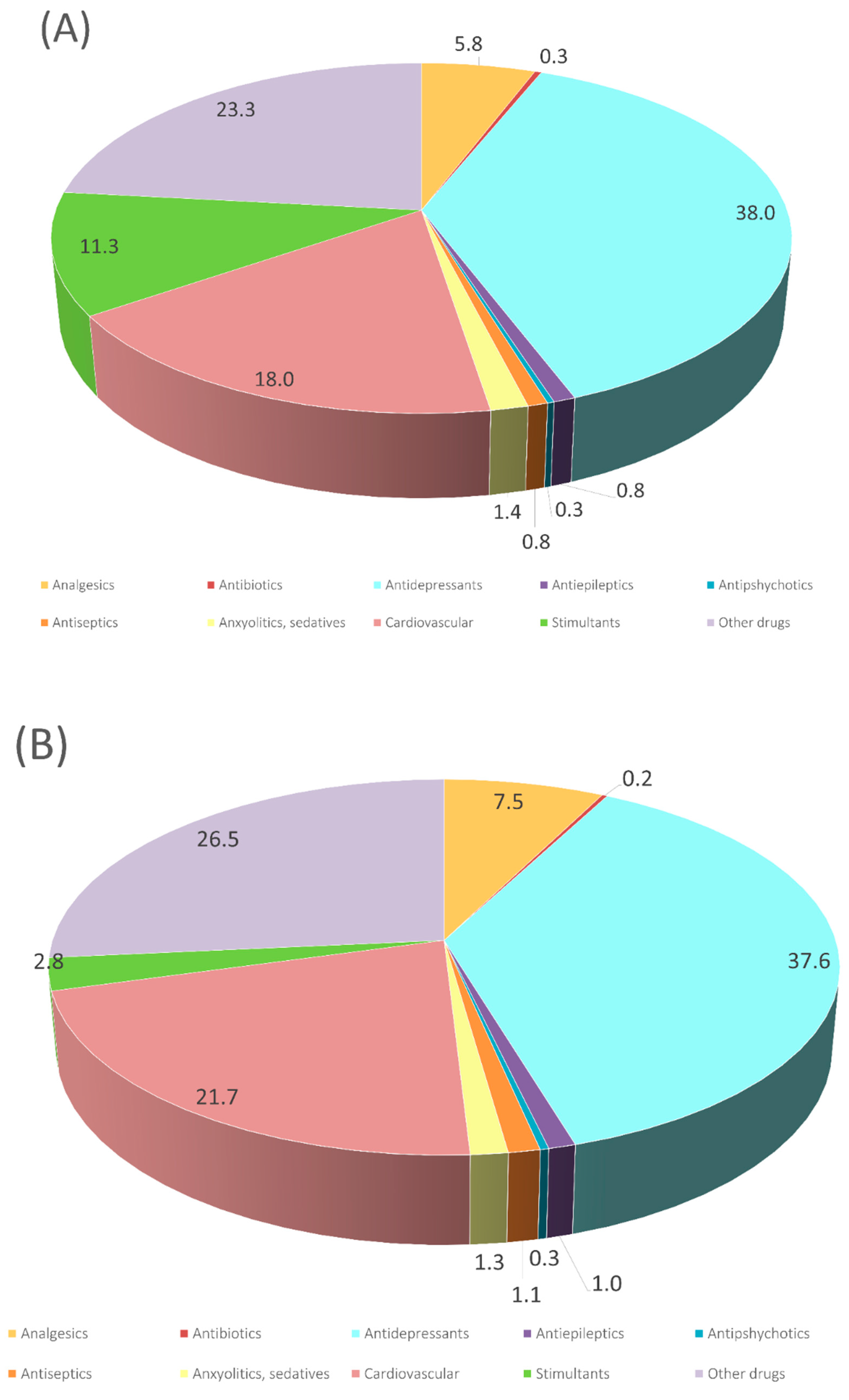
3.1. Occurrence of Emerging Pollutants in Sewage Sludge Samples
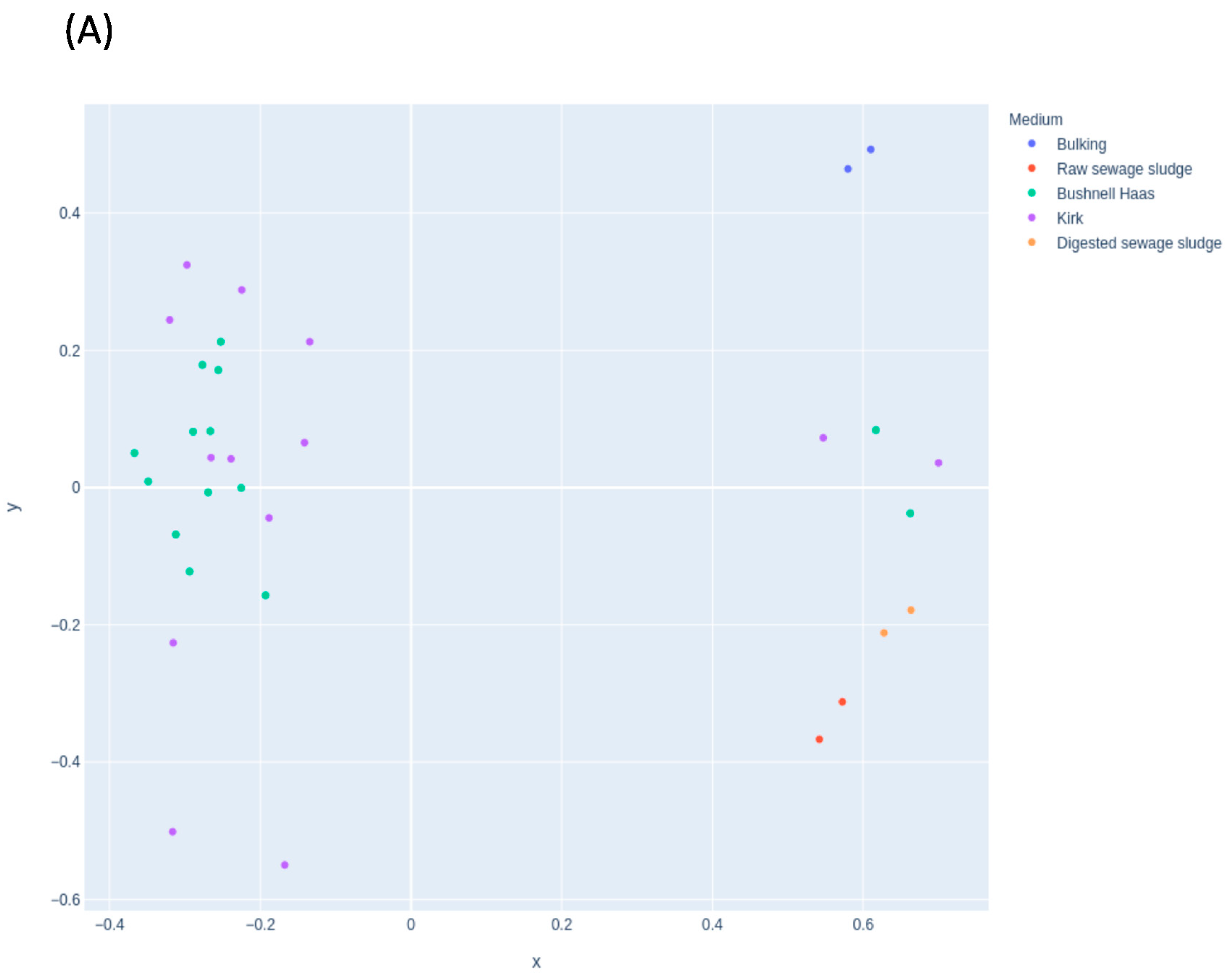
3.2. Shift of Microbial Population during Selective Pressure Experiments
3.3. Effect of the Sewage Sludge and the Medium for the Selection of Microorganisms
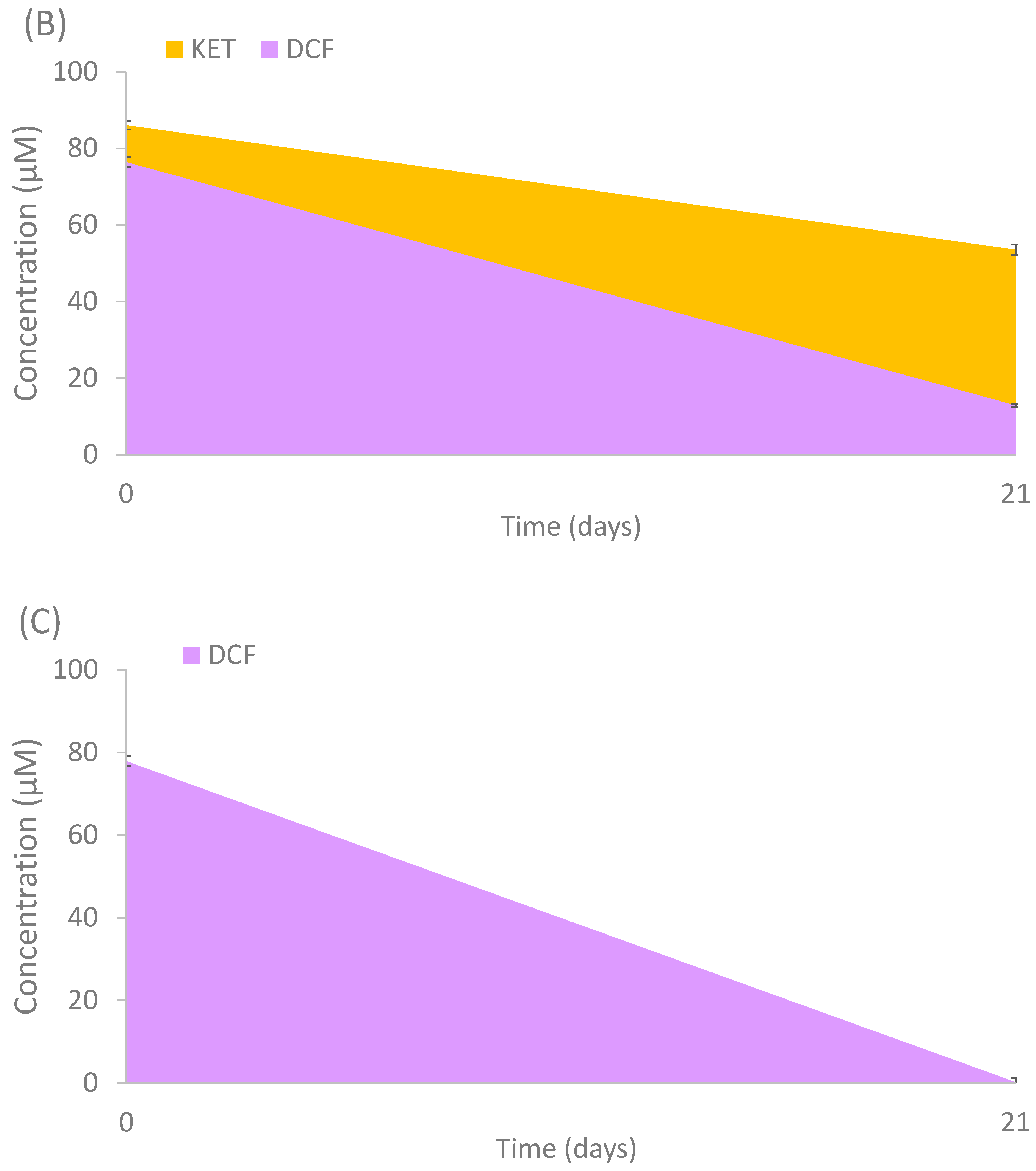
3.4. Pharmaceutical Active Compounds (PhACs) Removal during Selective Pressure Experiment
3.5. Isolation of Microorganisms after Selective Pressure
3.6. Biodegradation Experiments Using the Isolated Strains
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bianchi, E.; Biancalani, A.; Berardi, C.; Antal, A.; Fibbi, D.; Coppi, A.; Lastrucci, L.; Bussotti, N.; Colzi, I.; Renai, L.; et al. Improving the Efficiency of Wastewater Treatment Plants: Bio-Removal of Heavy-Metals and Pharmaceuticals by Azolla filiculoides and Lemna minuta. Sci. Total Environ. 2020, 746, 141219. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Qiang, Z.; Ben, W.; Zhu, B.; Liu, J. Rapid Detection of Multiple Class Pharmaceuticals in Both Municipal Wastewater and Sludge with Ultra High Performance Liquid Chromatography Tandem Mass Spectrometry. J. Environ. Sci. 2014, 26, 1949–1959. [Google Scholar] [CrossRef] [PubMed]
- Castro, G.; Ramil, M.; Cela, R.; Rodríguez, I. Identification and Determination of Emerging Pollutants in Sewage Sludge Driven by UPLC-QTOF-MS Data Mining. Sci. Total Environ. 2021, 778, 146256. [Google Scholar] [CrossRef] [PubMed]
- Comtois-Marotte, S.; Chappuis, T.; Vo Duy, S.; Gilbert, N.; Lajeunesse, A.; Taktek, S.; Desrosiers, M.; Veilleux, É.; Sauvé, S. Analysis of Emerging Contaminants in Water and Solid Samples Using High Resolution Mass Spectrometry with a Q Exactive Orbital Ion Trap and Estrogenic Activity with YES-Assay. Chemosphere 2017, 166, 400–411. [Google Scholar] [CrossRef]
- Kümmerer, K. The Presence of Pharmaceuticals in the Environment Due to Human Use—Present Knowledge and Future Challenges. J. Environ. Manag. 2009, 90, 2354–2366. [Google Scholar] [CrossRef]
- Michalska, M.; Zorena, K.; Marks, R.; Wąż, P. The Emergency Discharge of Sewage to the Bay of Gdańsk as a Source of Bacterial Enrichment in Coastal Air. Sci. Rep. 2021, 11, 20959. [Google Scholar] [CrossRef]
- Ericson, H.; Thorsén, G.; Kumblad, L. Physiological Effects of Diclofenac, Ibuprofen and Propranolol on Baltic Sea Blue Mussels. Aquat. Toxicol. Amst. Neth. 2010, 99, 223–231. [Google Scholar] [CrossRef]
- Radjenović, J.; Petrović, M.; Barceló, D. Fate and Distribution of Pharmaceuticals in Wastewater and Sewage Sludge of the Conventional Activated Sludge (CAS) and Advanced Membrane Bioreactor (MBR) Treatment. Water Res. 2009, 43, 831–841. [Google Scholar] [CrossRef]
- Ramos, R.L.; Lebron, Y.A.R.; Moreira, V.R.; de Souza Santos, L.V.; Amaral, M.C.S. Phenolic Compounds in Surface Water: Methodology and Occurrence in Doce River, Brazil. Environ. Monit. Assess. 2021, 193, 687. [Google Scholar] [CrossRef]
- Tomasi Morgano, M.; Leibold, H.; Richter, F.; Stapf, D.; Seifert, H. Screw Pyrolysis Technology for Sewage Sludge Treatment. Waste Manag. 2018, 73, 487–495. [Google Scholar] [CrossRef]
- Rocha, A.C.; Camacho, C.; Eljarrat, E.; Peris, A.; Aminot, Y.; Readman, J.W.; Boti, V.; Nannou, C.; Marques, A.; Nunes, M.L.; et al. Bioaccumulation of Persistent and Emerging Pollutants in Wild Sea Urchin Paracentrotus lividus. Environ. Res. 2018, 161, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.A.; Khan, S.U.; Ahmed, S.; Farooqi, I.H.; Yousefi, M.; Mohammadi, A.A.; Changani, F. Recent Trends in Disposal and Treatment Technologies of Emerging-Pollutants—A Critical Review. TrAC Trends Anal. Chem. 2020, 122, 115744. [Google Scholar] [CrossRef]
- Garcia-Rodríguez, A.; Sagristà, E.; Matamoros, V.; Fontàs, C.; Hidalgo, M.; Salvadó, V. Determination of Pharmaceutical Compounds in Sewage Sludge Using a Standard Addition Method Approach. Int. J. Environ. Anal. Chem. 2014, 94, 1199–1209. [Google Scholar] [CrossRef]
- Tran, N.H.; Urase, T.; Ngo, H.H.; Hu, J.; Ong, S.L. Insight into Metabolic and Cometabolic Activities of Autotrophic and Heterotrophic Microorganisms in the Biodegradation of Emerging Trace Organic Contaminants. Bioresour. Technol. 2013, 146, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Feng, J.; Liu, J.; Fu, W.; Li, X.; Li, B. Deciphering of Microbial Community and Antibiotic Resistance Genes in Activated Sludge Reactors under High Selective Pressure of Different Antibiotics. Water Res. 2019, 151, 388–402. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, S.; Yang, X.-L.; Yang, Y.-L.; Xu, H.; Li, X.-N.; Song, H.-L. Enhanced Degradation of Bisphenol A and Ibuprofen by an Up-Flow Microbial Fuel Cell-Coupled Constructed Wetland and Analysis of Bacterial Community Structure. Chemosphere 2019, 217, 599–608. [Google Scholar] [CrossRef]
- Zhao, X.; Chen, Z.; Wang, X.; Li, J.; Shen, J.; Xu, H. Remediation of Pharmaceuticals and Personal Care Products Using an Aerobic Granular Sludge Sequencing Bioreactor and Microbial Community Profiling Using Solexa Sequencing Technology Analysis. Bioresour. Technol. 2015, 179, 104–112. [Google Scholar] [CrossRef]
- Iranzo, M.; Gamón, M.; Boluda, R.; Mormeneo, S. Analysis of Pharmaceutical Biodegradation of WWTP Sludge Using Composting and Identification of Certain Microorganisms Involved in the Process. Sci. Total Environ. 2018, 640–641, 840–848. [Google Scholar] [CrossRef]
- Zheng, G.; Yu, B.; Wang, Y.; Ma, C.; Chen, T. Removal of Triclosan during Wastewater Treatment Process and Sewage Sludge Composting—A Case Study in the Middle Reaches of the Yellow River. Environ. Int. 2020, 134, 105300. [Google Scholar] [CrossRef]
- Jia, X.; Dini-Andreote, F.; Falcão Salles, J. Community Assembly Processes of the Microbial Rare Biosphere. Trends Microbiol. 2018, 26, 738–747. [Google Scholar] [CrossRef]
- Pascoal, F.; Magalhães, C.; Costa, R. The Link Between the Ecology of the Prokaryotic Rare Biosphere and Its Biotechnological Potential. Front. Microbiol. 2020, 11, 231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sidhu, C.; Vikram, S.; Pinnaka, A.K. Unraveling the Microbial Interactions and Metabolic Potentials in Pre- and Post-Treated Sludge from a Wastewater Treatment Plant Using Metagenomic Studies. Front. Microbiol. 2017, 8, 1382. [Google Scholar] [CrossRef] [PubMed]
- Robledo-Mahón, T.; Aranda, E.; Pesciaroli, C.; Rodríguez-Calvo, A.; Silva-Castro, G.A.; González-López, J.; Calvo, C. Effect of Semi-Permeable Cover System on the Bacterial Diversity during Sewage Sludge Composting. J. Environ. Manag. 2018, 215, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Robledo-Mahón; Gómez-Silván, C.; Andersen, G.L.; Calvo, C.; Aranda, E. Assessment of Bacterial and Fungal Communities in a Full-Scale Thermophilic Sewage Sludge Composting Pile under a Semipermeable Cover. Bioresour. Technol. 2020, 298, 122550. [Google Scholar] [CrossRef]
- Kirk, T.K.; Schultz, E.; Connors, W.J.; Lorenz, L.F.; Zeikus, J.G. Influence of Culture Parameters on Lignin Metabolism ByPhanerochaete chrysosporium. Arch. Microbiol. 1978, 117, 277–285. [Google Scholar] [CrossRef]
- Bushnell, L.; Haas, H. The Utilization of Certain Hydrocarbons by Microorganisms. J. Bacteriol. 1941, 41, 653–673. [Google Scholar] [CrossRef] [Green Version]
- Abarian, M.; Hassanshahian, M.; Esbah, A. Degradation of Phenol at High Concentrations Using Immobilization of Pseudomonas putida P53 into Sawdust Entrapped in Sodium-Alginate Beads. Water Sci. Technol. J. Int. Assoc. Water Pollut. Res. 2019, 79, 1387–1396. [Google Scholar] [CrossRef]
- Qin, X.; Luo, H.; Zhang, X.; Yao, B.; Ma, F.; Su, X. Dye-Decolorizing Peroxidases in Irpex lacteus Combining the Catalytic Properties of Heme Peroxidases and Laccase Play Important Roles in Ligninolytic System. Biotechnol. Biofuels 2018, 11, 302. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global Patterns of 16S RRNA Diversity at a Depth of Millions of Sequences per Sample. Proc. Natl. Acad. Sci. USA 2011, 108, 4516–4522. [Google Scholar] [CrossRef] [Green Version]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. PCR Protoc. Guide Methods Appl. 1990, 18, 315–322. [Google Scholar]
- Olicón-Hernández, D.R.; Gómez-Silván, C.; Pozo, C.; Andersen, G.L.; González-Lopez, J.; Aranda, E. Penicillium oxalicum XD-3.1 Removes Pharmaceutical Compounds from Hospital Wastewater and Outcompetes Native Bacterial and Fungal Communities in Fluidised Batch Bioreactors. Int. Biodeterior. Biodegrad. 2021, 158, 105179. [Google Scholar] [CrossRef]
- Waksman, S.A. A Method for Counting the Number of Fungi in the Soil. J. Bacteriol. 1922, 7, 339–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, K.S.; Kim, J.-M.; Kim, J.-W.; Jung, B.Y.; Kim, W.; Kim, I.J.; Kook, Y.-H. Identification of Bacillus anthracis by RpoB Sequence Analysis and Multiplex PCR. J. Clin. Microbiol. 2003, 41, 2908–2914. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conejo-Saucedo, U.; Ledezma-Villanueva, A.; Ángeles de Paz, G.; Herrero-Cervera, M.; Calvo, C.; Aranda, E. Evaluation of the Potential of Sewage Sludge Mycobiome to Degrade High Diclofenac and Bisphenol-A Concentrations. Toxics 2021, 9, 115. [Google Scholar] [CrossRef]
- Anastassiades, M.; Lehotay, S.J.; Stajnbaher, D.; Schenck, F.J. Fast and Easy Multiresidue Method Employing Acetonitrile Extraction/Partitioning and “Dispersive Solid-Phase Extraction” for the Determination of Pesticide Residues in Produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [CrossRef] [Green Version]
- Official Journal of the European Union. Directive 2013/39/EU Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013 Amending Directives 2000/60/EC and 2008/105/EC as Regards Priority Substances in the Field of Water Policy; Official Journal of the European Union: Brussels, Belgium, 2013. [Google Scholar]
- Ort, C.; Lawrence, M.G.; Rieckermann, J.; Joss, A. Sampling for Pharmaceuticals and Personal Care Products (PPCPs) and Illicit Drugs in Wastewater Systems: Are Your Conclusions Valid? A Critical Review. Environ. Sci. Technol. 2010, 44, 6024–6035. [Google Scholar] [CrossRef]
- Ort, C.; Lawrence, M.G.; Reungoat, J.; Mueller, J.F. Sampling for PPCPs in Wastewater Systems: Comparison of Different Sampling Modes and Optimization Strategies. Environ. Sci. Technol. 2010, 44, 6289–6296. [Google Scholar] [CrossRef]
- Dennhardt, A.A.; Murphy, J.G. Prevention and Treatment of College Student Drug Use: A Review of the Literature. Addict. Behav. 2013, 38, 2607–2618. [Google Scholar] [CrossRef]
- Gerrity, D.; Trenholm, R.A.; Snyder, S.A. Temporal Variability of Pharmaceuticals and Illicit Drugs in Wastewater and the Effects of a Major Sporting Event. Water Res. 2011, 45, 5399–5411. [Google Scholar] [CrossRef]
- Lai, F.Y.; Bruno, R.; Hall, W.; Gartner, C.; Ort, C.; Kirkbride, P.; Prichard, J.; Thai, P.K.; Carter, S.; Mueller, J.F. Profiles of Illicit Drug Use during Annual Key Holiday and Control Periods in Australia: Wastewater Analysis in an Urban, a Semi-Rural and a Vacation Area. Addict. Abingdon Engl. 2013, 108, 556–565. [Google Scholar] [CrossRef] [Green Version]
- Lai, F.Y.; Thai, P.K.; O’Brien, J.; Gartner, C.; Bruno, R.; Kele, B.; Ort, C.; Prichard, J.; Kirkbride, P.; Hall, W.; et al. Using Quantitative Wastewater Analysis to Measure Daily Usage of Conventional and Emerging Illicit Drugs at an Annual Music Festival. Drug Alcohol Rev. 2013, 32, 594–602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasprzyk-Hordern, B.; Baker, D.R. Enantiomeric Profiling of Chiral Drugs in Wastewater and Receiving Waters. Environ. Sci. Technol. 2012, 46, 1681–1691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasprzyk-Hordern, B.; Baker, D.R. Estimation of Community-Wide Drugs Use via Stereoselective Profiling of Sewage. Sci. Total Environ. 2012, 423, 142–150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrie, B.; Barden, R.; Kasprzyk-Hordern, B. A Review on Emerging Contaminants in Wastewaters and the Environment: Current Knowledge, Understudied Areas and Recommendations for Future Monitoring. Water Res. 2015, 72, 3–27. [Google Scholar] [CrossRef]
- Cortés, J.M.; Larsson, E.; Jönsson, J.Å. Study of the Uptake of Non-Steroid Anti-Inflammatory Drugs in Wheat and Soybean after Application of Sewage Sludge as a Fertilizer. Sci. Total Environ. 2013, 449, 385–389. [Google Scholar] [CrossRef]
- Gallardo-Altamirano, M.J.; Maza-Márquez, P.; Montemurro, N.; Pérez, S.; Rodelas, B.; Osorio, F.; Pozo, C. Insights into the Removal of Pharmaceutically Active Compounds from Sewage Sludge by Two-Stage Mesophilic Anaerobic Digestion. Sci. Total Environ. 2021, 789, 147869. [Google Scholar] [CrossRef]
- Baratpour, P.; Moussavi, G. The Accelerated Biodegradation and Mineralization of Acetaminophen in the H2O2-Stimulated Upflow Fixed-Bed Bioreactor (UFBR). Chemosphere 2018, 210, 1115–1123. [Google Scholar] [CrossRef]
- Thelusmond, J.-R.; Strathmann, T.J.; Cupples, A.M. Carbamazepine, Triclocarban and Triclosan Biodegradation and the Phylotypes and Functional Genes Associated with Xenobiotic Degradation in Four Agricultural Soils. Sci. Total Environ. 2019, 657, 1138–1149. [Google Scholar] [CrossRef]
- Karn, S.K.; Kumar, A. Sludge: Next Paradigm for Enzyme Extraction and Energy Generation. Prep. Biochem. Biotechnol. 2019, 49, 105–116. [Google Scholar] [CrossRef]
- Robledo-Mahón, T.; Calvo, C.; Aranda, E. Enzymatic Potential of Bacteria and Fungi Isolates from the Sewage Sludge Composting Process. Appl. Sci. 2020, 10, 7763. [Google Scholar] [CrossRef]
- Naghdi, M.; Taheran, M.; Brar, S.K.; Kermanshahi-pour, A.; Verma, M.; Surampalli, R.Y. Removal of Pharmaceutical Compounds in Water and Wastewater Using Fungal Oxidoreductase Enzymes. Environ. Pollut. 2018, 234, 190–213. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Lam, S.S.; Iqbal, H.M.N. Biocatalytic Remediation of Pharmaceutically Active Micropollutants for Environmental Sustainability. Environ. Pollut. 2022, 293, 118582. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.V.; Cabana, H. Towards High Potential Magnetic Biocatalysts for On-Demand Elimination of Pharmaceuticals. Bioresour. Technol. 2016, 200, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, M.; El-sheekh, M.; Ma, Y.; Pugazhendhi, A.; Natarajan, D.; Kandasamy, G.; Raja, R.; Saravana Kumar, R.M.; Kumarasamy, S.; Sathiyan, G.; et al. Current Status of Microbes Involved in the Degradation of Pharmaceutical and Personal Care Products (PPCPs) Pollutants in the Aquatic Ecosystem. Environ. Pollut. 2022, 300, 118922. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; de Toledo, R.A.; Wang, S.; Shim, H. Removal of Carbamazepine and Naproxen by Immobilized Phanerochaete chrysosporium under Non-Sterile Condition. New Biotechnol. 2015, 32, 282–289. [Google Scholar] [CrossRef]
- Rodríguez-Rodríguez, C.E.; Marco-Urrea, E.; Caminal, G. Degradation of Naproxen and Carbamazepine in Spiked Sludge by Slurry and Solid-Phase Trametes versicolor Systems. Bioresour. Technol. 2010, 101, 2259–2266. [Google Scholar] [CrossRef]





| Compound | Formula | CAS Number | Application | rSS (ng/g, d.w.) | dSS (ng/g, d.w.) | RSD (%) | Percentage of Removal |
|---|---|---|---|---|---|---|---|
| Acetaminophen | C8H9NO2 | 103-90-2 | Analgesic | 45.3 | 31.8 | 20.3 | 29.9 |
| Acridone | C13H9NO | 578-95-0 | Antiviral agent | 3.9 | 0.3 | 13.0 | 91.3 |
| Amlodipine | C20H25ClN2O5 | 88150-42-9 | Cardiovascular | BLOQ | 148.3 | 6.4 | |
| Atenolol | C14H22N2O3 | 29122-68-7 | 48.7 | 0 | 100.0 | ||
| 1.2.3-Benzotriazole | C6H5N3 | 95-14-7 | Drug precursor | 448.9 | 773.9 | 13.0 | −72.4 |
| Caffeine | C8H10N4O2 | 58-08-2 | Stimulant | BLOQ | 37.6 | 43.2 | |
| Carbamazepine | C15H12N2O | 298-46-4 | Antiepileptic | 8.7 | 6.7 | 4.8 | 23.6 |
| Carbamazepine-10.11-epoxide | C15H12N2O2 | 36507-30-9 | Carbamazepine metabolite | 13.5 | 16.8 | 5.3 | −24.4 |
| Chlorpromazine | C17H19ClN2S | 50-53-3 | Antipsychotics | 8.4 | 9.3 | 14.3 | −11.0 |
| Dexamethasone | C22H29FO5 | 50-02-2 | Analgesic (corticosteroids) | 117.8 | 148.7 | 29.3 | −26.2 |
| Diclofenac | C14H11Cl2NO2 | 15307-86-5 | Analgesic | 17.3 | 38.9 | 16.4 | −124.7 |
| Diltiazem | C22H26N2O4S | 42399-41-7 | Cardiovascular | 17.5 | 3.1 | 9.6 | 82.0 |
| Estrone | C18H22O2 | 53-16-7 | Hormone | 302.7 | 18.1 | 25.7 | 94.0 |
| Fenofibrate | C20H21ClO4 | 49562-28-9 | Cardiovascular | 142.4 | 318.5 | 35.2 | −123.7 |
| Fluoxetine | C17H18F3NO | 54910-89-3 | Antidepressant | 57.5 | 135.9 | 1.8 | −136.4 |
| Ketamine | C13H16ClNO | 6740-88-1 | Anesthetic | 1.0 | 0.6 | 41.9 | 37.6 |
| Ketoprofen | C16H14O3 | 22071-15-4 | Analgesic | 10.0 | 5.7 | 11.1 | 42.6 |
| Lamotrigine | C9H7Cl2N5 | 84057-84-1 | Antiepileptic. | 4.9 | 5.7 | 9.7 | −17.0 |
| Loratadine | C22H23ClN2O2 | 79794-75-5 | Antihistamine | 4.2 | 0 | 100.0 | |
| Lormetazepam | C16H12Cl2N2O2 | 848-75-9 | Anxiolytic, sedative | 9.8 | 12.0 | 8.0 | −22.7 |
| Mephedrone | C11H15NO | 1189805-46-6 | Stimulant drug | 313.0 | 18.1 | 6.4 | 94.2 |
| Methadone | C21H27NO | 76-99-3 | Stimulant drug | 54.6 | 24.4 | 24.0 | 55.3 |
| Metoprolol | C15H25NO3 | 37350-58-6 | Cardiovascular | 11.5 | 21.7 | 15.6 | −88.9 |
| Oxazepam | C15H11N2O2Cl | 604-75-1 | Anxiolytic | 20.7 | 17.7 | 6.3 | 14.8 |
| Paroxetine | C19H20FNO3 | 61869-08-7 | Antidepressant | 54.1 | 121.9 | 17.5 | −125.2 |
| Propranolol | C16H21NO2 | 525-66-6 | Cardiovascular | 33.5 | 44.1 | 17.2 | −31.7 |
| Sertraline | C17H17Cl2N | 79617-96-2 | Antidepressant | 1100.0 | 864.4 | 3.5 | 21.4 |
| Sulfapyridine | C11H11N3O2S | 144-83-2 | Antibiotic | 10.9 | 7.5 | 18.9 | 31.7 |
| Temazepam | C16H13ClN2O2 | 846-50-4 | Anxiolytic, sedative | 10.4 | 4.9 | 5.9 | 52.4 |
| Triclocarban | C13H9Cl3N2O | 101-20-2 | Antiseptic | 24.6 | 34.1 | 21.7 | −38.5 |
| Valsartan | C24H29N5O3 | 137862-53-4 | Cardiovascular | 58.2 | 112.3 | 8.8 | −93.0 |
| Valsartan acid | C14H10N4O2 | Valsartan metabolite | 275.5 | 0 | 100.0 | ||
| Venlafaxine | C17H27NO2 | 93413-69-5 | Antidepressant | 30.8 | 2.8 | 7.0 | 91.0 |
| Zolpidem | C19H21N3O | 82626-48-0 | Insomnia treatment | 5.3 | 5.4 | 19.8 | −2.0 |
| Isolate ID | >99% Database Similarity | Gene Bank Accession Number | Phylum | |
|---|---|---|---|---|
| Fungal strains | H1 | Cladosporium cladosporioides | MT773569 | Ascomycota |
| H2 | Cladosporium limoniforme | MT773579 | Ascomycota | |
| H3 | Cladosporium halotolerans | MT773604 | Ascomycota | |
| H4 | Alternaria alternata | MT776719 | Ascomycota | |
| H5 | Aspergillus montevidensis | Ascomycota | ||
| H6 | Penicillium raistrickii | Ascomycota | ||
| H7 | Purpureocillium lilacinum | MT773618 | Ascomycota | |
| Bacterial strains | C1 | Bacillus simplex | MT773382 | Firmicutes |
| C2 | Corynebacterium efficiens | MT773417 | Actinobacteria | |
| C3 | Corynebacterium humireducens | MT773419 | Actinobacteria | |
| C4 | Gordonia hirsuta | MT773437 | Actinobacteria | |
| M1 | Alcaligenes faecalis | MT773443 | Proteobacteria | |
| K1 | Micrococcus yunnanensis | MT773451 | Actinobacteria | |
| K4 | Enterococcus faecium | MT773438 | Firmicutes | |
| T1 | Paenalcaligenes hominis | MT773452 | Proteobacteria | |
| T4 | Oligella ureolytica | MT773453 | Proteobacteria | |
| T15 | Sphingobacterium jejuense | MT773454 | Bacteroidetes | |
| T19 | Staphylococcus hominis | MT773567 | Firmicutes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ledezma-Villanueva, A.; Robledo-Mahón, T.; Gómez-Silván, C.; Angeles-De Paz, G.; Pozo, C.; Manzanera, M.; Calvo, C.; Aranda, E. High-Throughput Microbial Community Analyses to Establish a Natural Fungal and Bacterial Consortium from Sewage Sludge Enriched with Three Pharmaceutical Compounds. J. Fungi 2022, 8, 668. https://doi.org/10.3390/jof8070668
Ledezma-Villanueva A, Robledo-Mahón T, Gómez-Silván C, Angeles-De Paz G, Pozo C, Manzanera M, Calvo C, Aranda E. High-Throughput Microbial Community Analyses to Establish a Natural Fungal and Bacterial Consortium from Sewage Sludge Enriched with Three Pharmaceutical Compounds. Journal of Fungi. 2022; 8(7):668. https://doi.org/10.3390/jof8070668
Chicago/Turabian StyleLedezma-Villanueva, Alejandro, Tatiana Robledo-Mahón, Cinta Gómez-Silván, Gabriela Angeles-De Paz, Clementina Pozo, Maximino Manzanera, Concepción Calvo, and Elisabet Aranda. 2022. "High-Throughput Microbial Community Analyses to Establish a Natural Fungal and Bacterial Consortium from Sewage Sludge Enriched with Three Pharmaceutical Compounds" Journal of Fungi 8, no. 7: 668. https://doi.org/10.3390/jof8070668








