Occurrence, Diversity and Anti-Fungal Resistance of Fungi in Sand of an Urban Beach in Slovenia—Environmental Monitoring with Possible Health Risk Implications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling of Sand and Seawater
2.2. Cultivation of Fungi and Storage of Viable Strains
2.3. DNA Extraction and Molecular Identification
2.4. Antifungal Susceptibility Testing of Selected Aspergillus Strains
2.5. Using Machine Learning to Relate Environmental Conditions and Fungal Profiles
2.6. Predictive Modelling
3. Results
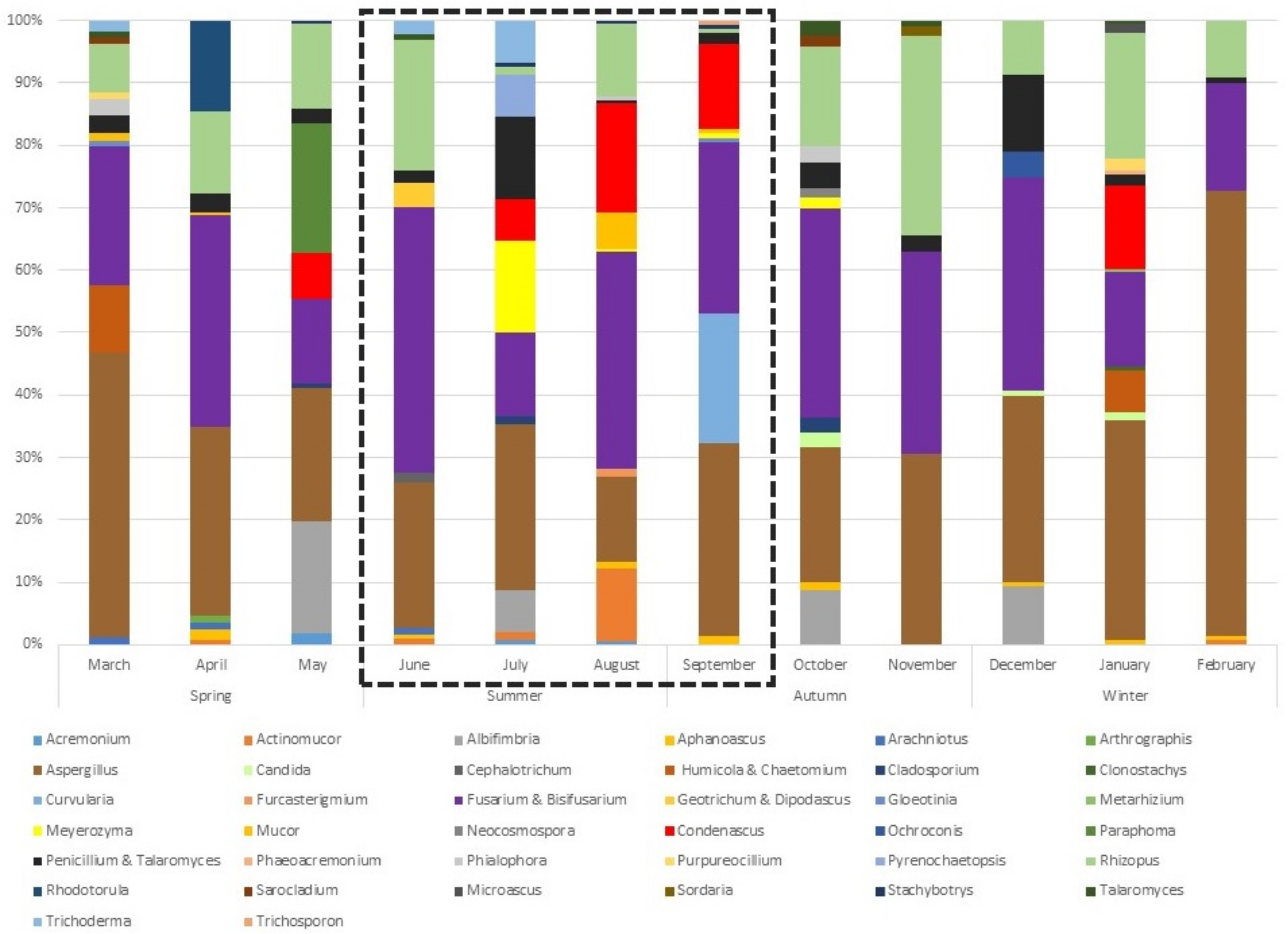
3.1. Artificially Designed Sandy Beach Harbours a Great Diversity of Fungi
3.2. Differences in Diversity of Culturable Fungi in Seawater and Beach Sand
3.3. Machine Learning Models Reveal Connections between Environmental Changes and Fungal Presence
3.4. Factors Influencing the Stability of Fungal Community in Beach Sand
3.5. Potential Human Pollution Indicators for the Bathing Season
3.6. Aspergillus Niger Strains Isolated from Beach Sand Are Resistant to Amphotericin B
4. Discussion
4.1. Fungi Isolated from Seawater Differ from Sand Mycobiota
4.2. Fungal Abundance in Beach Sand Varies According to Seasonal Weather Conditions
4.3. Core Fungal Genera in the Sand Are Affected by the Seawater Temperature
4.4. Fungi in Sand as Possible Indicators of Anthropogenic Pollution during the Bathing Season
4.5. Beach Sand Harbours Amphotericin B-Resistant Strains of Aspergillus Section Nigri
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Naranjo-Ortiz, M.A.; Gabaldón, T. Fungal evolution: Major ecological adaptations and evolutionary transitions. Biol. Rev. Camb. Philos. Soc. 2019, 94, 1443–1476. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.D.; Xu, J.; Rapior, S.; Jeewon, R.; Lumyong, S.; Niego, A.G.T.; Abeywickrama, P.D.; Aluthmuhandiram, J.V.S.; Brahamanage, R.S.; Brooks, S.; et al. The amazing potential of fungi: 50 ways we can exploit fungi industrially. Fungal Divers. 2019, 97, 1–136. [Google Scholar] [CrossRef]
- Murgia, M.; Fiamma, M.; Barac, A.; Deligios, M.; Mazzarello, V.; Paglietti, B.; Cappuccinelli, P.; Al-Qahtani, A.; Squartini, A.; Rubino, S.; et al. Biodiversity of fungi in hot desert sands. MicrobiologyOpen 2019, 8, e00595. [Google Scholar] [CrossRef] [PubMed]
- Coleine, C.; Stajich, E.J.; de los Ríos, A.; Selbmann, L. Beyond the extremes: Rocks as ultimate refuge for fungi in drylands. Mycologia 2021, 113, 108–133. [Google Scholar] [CrossRef]
- Zajc, J.; Zalar, P.; Plemenitaš, A.; Gunde-Cimerman, N. The mycobiota of the salterns. Prog. Mol. Subcell. Biol. 2012, 53, 133–158. [Google Scholar] [CrossRef]
- Azpiazu-Muniozguren, M.; Perez, A.; Rementeria, A.; Martinez-Malaxetxebarria, I.; Alonso, R.; Laorden, L.; Gamboa, J.; Bikandi, J.; Garaizar, J.; Martinez-Ballesteros, I. Fungal diversity and composition of the continental solar saltern in Añana Salt Valley (Spain). J. Fungi 2021, 7, 1074. [Google Scholar] [CrossRef]
- Gunde-Cimerman, N.; Zalar, P. Fungi in solar salterns. Food Technol. Biotechnol. 2014, 52, 170–179. [Google Scholar]
- Magan, N. Fungi in extreme environments. In Environmental and Microbial Relationships, The Mycota, 1st ed.; Kubicek, C., Druzhinina, I., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; Volume 4, pp. 85–103. [Google Scholar] [CrossRef]
- Rampelotto, P. Resistance of microorganisms to extreme environmental conditions and its contribution to astrobiology. Sustainability 2010, 2, 1602–1623. [Google Scholar] [CrossRef]
- Gostinčar, C.; Grube, M.; De Hoog, S.; Zalar, P.; Gunde-Cimerman, N. Extremotolerance in fungi: Evolution on the edge. FEMS Microbiol. Ecol. 2009, 71, 2–11. [Google Scholar] [CrossRef]
- Pessoa, M.F.; Lidon, F.C. Impact of human activities on coastal vegetation—A review. Emir. J. Food Agric. 2013, 25, 926–944. [Google Scholar] [CrossRef]
- Solo-Gabriele, H.M.; Harwood, V.J.; Kay, D.; Fujioka, R.S.; Sadowsky, M.J.; Whitman, R.L.; Wither, A.; Caniça, M.; da Fonseca, R.C.; Duarte, A.; et al. Beach sand and the potential for infectious disease transmission: Observations and recommendations. J. Mar. Biol. Assoc. UK 2016, 96, 101–120. [Google Scholar] [CrossRef]
- Brandão, J.; Gangneux, J.P.; Arikan-Akdagli, S.; Barac, A.; Bostanaru, A.C.; Brito, S.; Bull, M.; Çerikçioğlu, N.; Chapman, B.; Efstratiou, M.A.; et al. Mycosands: Fungal diversity and abundance in beach sand and recreational waters—Relevance to human health. Sci. Total Environ. 2021, 781, 146598. [Google Scholar] [CrossRef] [PubMed]
- Byappanahalli, M.N.; Nevers, M.B.; Korajkic, A.; Staley, Z.R.; Harwood, V.J. Enterococci in the environment. Microbiol. Mol. Biol. Rev. 2012, 76, 685–706. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Hur, H.G.; Sadowsky, M.J.; Byappanahalli, M.N.; Yan, T.; Ishii, S. Environmental Escherichia coli: Ecology and public health implications—A review. J. Appl. Microbiol. 2017, 123, 570–581. [Google Scholar] [CrossRef]
- Hurst, C.J. Dirt and disease: The ecology of soil fungi and plant fungi that are infectious for vertebrates. In Understanding Terrestrial Microbial Communities; Advances in Environmental Microbiology; Hurst, C., Ed.; Springer: Cham, Switzerland, 2019; Volume 6, pp. 289–405. [Google Scholar] [CrossRef]
- Friedman, D.Z.; Schwartz, I.S. Emerging fungal infections: New patients, new patterns, and new pathogens. J. Fungi 2019, 5, 67. [Google Scholar] [CrossRef]
- Romão, D.; Sabino, R.; Veríssimo, C.; Viegas, C.; Barroso, H.; Duarte, A.; Solo-Gabriele, H.; Gunde-Cimerman, N.; Babič, M.N.; Marom, T.; et al. Children and sand play: Screening of potential harmful microorganisms in sandboxes, parks, and beaches. Curr. Fungal Infect. Rep. 2015, 9, 155–163. [Google Scholar] [CrossRef]
- Sabino, R.; Verissimo, C.; Cunha, M.A.; Wergikoski, B.; Ferreira, F.C.; Rodrigues, R.; Parada, H.; Falcão, L.; Rosado, L.; Pinheiro, C.; et al. Pathogenic fungi: An unacknowledged risk at coastal resorts? New insights on microbiological sand quality in Portugal. Mar. Pollut. Bull. 2011, 62, 1506–1511. [Google Scholar] [CrossRef]
- Azevedo, E.; Figueira, D.; Caeiro, M.F.; Barata, M. Biodiversity of filamentous fungi on soils and sands. In Progress in Mycology; Rai, M., Kövics, G., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 233–257. [Google Scholar]
- Libkind, D.; Buzzini, P.; Turchetti, B.; Rosa, C.A. Yeasts in continental and seawater. In Yeasts in Natural Ecosystems: Diversity; Buzzini, P., Lachance, M.A., Yurkov, A., Eds.; Springer: Cham, Switzerland, 2017; pp. 1–61. [Google Scholar]
- Van den Ende, A.H.G.; de Hoog, G.S. Variability and molecular diagnostics of the neurotropic species Cladophialophora bantiana. Stud. Mycol. 1999, 43, 151–162. [Google Scholar]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Glass, N.L.; Donaldson, G.C. Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl. Environ. Microbiol. 1995, 61, 1323–1330. [Google Scholar] [CrossRef]
- O’Donnell, K.; Kistler, H.C.; Cigelnik, E.; Ploetz, R.C. Multiple evolutionary origins of the fungus causing Panama disease of banana: Concordant evidence from nuclear and mitochondrial gene genealogies. Proc. Natl. Acad. Sci. USA 1998, 95, 2044–2049. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Boekhout, T.; Kurtzman, C.P. Principles and methods used in yeast classification, and an overview of currently accepted yeast genera. In Nonconventional Yeasts in Biotechnology; Wolf, K., Ed.; Springer: Berlin, Germany, 1996; pp. 1–81. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- ARSO. Slovenian Environmental Agency, Ministry of the Environment and Spatial Planning. Available online: https://meteo.arso.gov.si/met/sl/app/webmet/#webmet=vUHcs9WYkN3LtVGdl92LhBHcvcXZi1WZ09Cc1p2cvAncvd2LyVWYs12L3VWY0hWZy9SaulGdugXbsx3cs9mdl5WahxHf (accessed on 8 July 2022).
- Blockeel, H.; De Raedt, L. Top-down induction of first-order logical decision trees. Artif. Intell. 1998, 101, 285–297. [Google Scholar] [CrossRef]
- Petković, M.; Kocev, D.; Džeroski, S. Feature ranking for multi-target regression. Mach. Learn. 2020, 109, 1179–1204. [Google Scholar] [CrossRef]
- Kocev, D.; Vens, C.; Struyf, J.; Džeroski, S. Tree ensembles for predicting structured outputs. Pattern Recognit. 2013, 46, 817–833. [Google Scholar] [CrossRef]
- de Hoog, G.S.; Guarro, J.; Gené, J.; Ahmed, S.; Al-Hatmi, A.M.S.; Figueras, M.J.; Vitale, R.G. Atlas of Clinical Fungi, 4th ed.; Foundation Atlas of Clinical Fungi: Utrecht, The Netherlands, 2020. [Google Scholar]
- ATCC. American Type Culture Collection. Available online: https://www.atcc.org/ (accessed on 18 January 2022).
- Siopi, M.; Pournaras, S.; Meletiadis, J. Comparative evaluation of Sensititre YeastOne and CLSI M38-A2 reference method for antifungal susceptibility testing of Aspergillus spp. against echinocandins. J. Clin. Microbiol. 2017, 55, 1714–1719. [Google Scholar] [CrossRef]
- EUCAST. European Committee on Antimicrobial Susceptibility Testing. Breakpoint Tables for Interpretation of MICs for Antifungal Agents, Version 10.0. Available online: https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/AFST/Clinical_breakpoints/AFST_BP_v10.0_200204_updatd_links_200924.pdf (accessed on 8 July 2022).
- Lumibao, C.Y.; Formel, S.; Elango, V.; Pardue, J.H.; Blum, M.; Van Bael, S.A. Persisting responses of salt marsh fungal communities to the Deepwater Horizon oil spill. Sci. Total Environ. 2018, 642, 904–913. [Google Scholar] [CrossRef]
- Neto, J.A.B.; Gaylarde, C.; Beech, I.; Bastos, A.C.; da Silva Quaresma, V.; de Carvalho, D.G. Microplastics and attached microorganisms in sediments of the Vitória bay estuarine system in SE Brazil. Ocean Coast. Manag. 2019, 169, 247–253. [Google Scholar] [CrossRef]
- Pérez-Alvelo, K.M.; Llegus, E.M.; Forestier-Babilonia, J.M.; Elías-Arroyo, C.V.; Pagán-Malavé, K.N.; Bird-Rivera, G.J.; Rodríguez-Sierra, C.J. Microplastic pollution on sandy beaches of Puerto Rico. Mar. Pollut. Bull. 2021, 164, 112010. [Google Scholar] [CrossRef]
- Bridson, J.H.; Patel, M.; Lewis, A.; Gaw, S.; Parker, K. Microplastic contamination in Auckland (New Zealand) beach sediments. Mar. Pollut. Bull. 2020, 151, 110867. [Google Scholar] [CrossRef] [PubMed]
- Pathakumari, B.; Liang, G.; Liu, W. Immune defence to invasive fungal infections: A comprehensive review. Biomed. Pharmacother. 2020, 130, 110550. [Google Scholar] [CrossRef] [PubMed]
- Bongomin, F.; Gago, S.; Oladele, R.O.; Denning, D.W. Global and multi-national prevalence of fungal diseases-estimate precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef]
- Klun, K.; Falnoga, I.; Mazej, D.; Šket, P.; Faganeli, J. Colloidal organic matter and metal (loid) s in coastal waters (Gulf of Trieste, Northern Adriatic Sea). Aquat. Geochem. 2019, 25, 179–194. [Google Scholar] [CrossRef]
- Perkovic, M.; Hribar, U.; Harsch, R. Oil Pollution in Slovenian Waters: The Threat to the Slovene Coast, Possible Negative Influences of Shipping on an Environment and Its Cultural Heritage. In Oil Pollution in the Mediterranean Sea: Part II. The Handbook of Environmental Chemistry; Carpenter, A., Kostianoy, A., Eds.; Springer: Cham, Switzerland, 2016; Volume 84, pp. 133–157. [Google Scholar] [CrossRef]
- FEE. Foundation for Environmental Education. Pure Water, Clean Coasts, Safety and Access for All. Available online: https://www.blueflag.global/ (accessed on 11 July 2022).
- EEU. Directive 2006/7/EC of the European Parliament and of the Council of 15 February 2006 concerning the management of bathing water quality and repealing directive76/160/EEC. Off. J. Eur. Union 2006, 64, 37–51. [Google Scholar]
- WHO. Guidelines on Recreational Water Quality: Volume 1 Coastal and Fresh Waters; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Weiskerger, C.J.; Brandão, J. Fungal contaminants in water and sand: A new frontier for quantitative microbial risk assessment. Curr. Opin. Environ. Sci. Health 2020, 16, 73–81. [Google Scholar] [CrossRef]
- Londoño, C.O.; Fernández, R.R.; Gulloso, E.R.M. Identification of fungi dermatophytes in the coastal area of district of Riohacha, La Guajira. Contemp. Eng. Sci. 2018, 11, 4691–4699. [Google Scholar] [CrossRef]
- Frenkel, M.; Yunik, Y.; Fleker, M.; Blum, S.E.; Sionov, E.; Elad, D.; Serhan, H.; Segal, E. Fungi in sands of Mediterranean Sea beaches of Israel—Potential relevance to human health and well-being. Mycoses 2020, 63, 1255–1261. [Google Scholar] [CrossRef]
- Michalska, M.; Kurpas, M.; Zorena, K.; Wąż, P.; Marks, R. Mold and Yeast-Like Fungi in the Seaside Air of the Gulf of Gdańsk (Southern Baltic) after an Emergency Disposal of Raw Sewage. J. Fungi 2020, 7, 219. [Google Scholar] [CrossRef]
- Gunde-Cimerman, N.; Ramos, J.; Plemenitaš, A. Halotolerant and halophilic fungi. Mycol. Res. 2009, 113, 1231–1241. [Google Scholar] [CrossRef]
- Maciel, N.O.; Johann, S.; Brandão, L.R.; Kucharíková, S.; Morais, C.G.; Oliveira, A.P.; Freitas, G.J.; Borelli, B.M.; Pellizzari, F.M.; Santos, D.A.; et al. Occurrence, antifungal susceptibility, and virulence factors of opportunistic yeasts isolated from Brazilian beaches. Memórias Inst. Oswaldo Cruz 2019, 114. [Google Scholar] [CrossRef] [PubMed]
- Luchetta, A.; Cantoni, C.; Cozzi, S.; Catalano, G.; Civitarese, G. Dissolved carbon dioxide, nutrients and oxygen in the Adriatic Sea. A Regional Observing Effort. Available online: https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.1054.2313&rep=rep1&type=pdf (accessed on 10 July 2022).
- Pombeiro-Sponchiado, S.R.; Sousa, G.S.; Andrade, J.C.; Lisboa, H.F.; Gonçalves, R.C. Production of melanin pigment by fungi and its biotechnological applications. In Melanin, 1st ed.; Blumenberg, M., Ed.; IntechOpen: London, UK, 2017; Volume 1, pp. 47–75. [Google Scholar]
- Kaewkrajay, C.; Chanmethakul, T.; Limtong, S. Assessment of diversity of culturable marine yeasts associated with corals and zoanthids in the Gulf of Thailand, South China Sea. Microorganisms 2020, 8, 474. [Google Scholar] [CrossRef]
- Sousa, L.; Camacho, I.C.; Grinn-Gofroń, A.; Camacho, R. Monitoring of anamorphic fungal spores in Madeira region (Portugal), 2003–2008. Aerobiologia 2016, 32, 303–315. [Google Scholar] [CrossRef]
- Liu, B.; Fu, R.; Wu, B.; Liu, X.; Xiang, M. Rock-inhabiting fungi: Terminology, diversity, evolution and adaptation mechanisms. Mycology 2022, 13, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Abreu, R.; Figueira, C.; Romão, D.; Brandão, J.; Freitas, M.C.; Andrade, C.; Calado, G.; Ferreira, C.; Campos, A.; Prada, S. Sediment characteristics and microbiological contamination of beach sand—A case-study in the archipelago of Madeira. Sci. Total Environ. 2016, 573, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Velegraki, A.; Efstratiou, M.A.; Arabatzis, M.; Prodromou, P. A survey on the occurrence of dermatophytes in recreational beach sand. In Proceedings of the International Society for Microbial Ecology (ISME), Copenhagen, Denmark, 19–24. August 2012. [Google Scholar]
- Vega, F.E. Insect pathology and fungal endophytes. J. Invertebr. Pathol. 2008, 98, 277–279. [Google Scholar] [CrossRef]
- Alpar, B.; Unlu, S. Volatile Aromatic Compounds (BTEX) in Sediments Offshore Zonguldak Industrial Region, Black Sea, Turkey. Asian J. Chem. 2010, 22, 3531. [Google Scholar]
- van Rhijn, N.; Bromley, M. The consequences of our changing environment on life threatening and debilitating fungal diseases in humans. J. Fungi 2021, 7, 367. [Google Scholar] [CrossRef]
- Loureiro, S.T.A.; Cavalcanti, M.A.D.Q.; Neves, R.P.; Passavante, J.Z.D.O. Yeasts isolated from sand and sea water in beaches of Olinda, Pernambuco state, Brazil. Braz. J. Microbiol. 2005, 36, 333–337. [Google Scholar] [CrossRef]
- Grishkan, I.; Lázaro, R.; Kidron, G.J. Cultured microfungal communities in biological soil crusts and bare soils at the Tabernas Desert, Spain. Soil Syst. 2019, 3, 36. [Google Scholar] [CrossRef]
- Zupančič, N.; Skobe, S. Anthropogenic environmental impact in the Mediterranean coastal area of Koper/Capodistria, Slovenia. J. Soils Sediments 2014, 14, 67–77. [Google Scholar] [CrossRef]
- Nosratabadi, M.; Kordbacheh, P.; Kachuei, R.; Safara, M.; Rezaie, S.; Afshari, M.A. Isolation of keratinophilic fungi from the soil of Greater Tunb, Abu-Musa, and Sirri islands in the Persian Gulf, Iran. Curr. Med. Mycol. 2017, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.D.; Rautemaa-Richardson, R. Biotic environments supporting the persistence of clinically relevant Mucormycetes. J. Fungi 2019, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Timmusk, S.; Nevo, E.; Ayele, F.; Noe, S.; Niinemets, Ü. Fighting Fusarium pathogens in the era of climate change: A conceptual approach. Pathogens 2020, 9, 419. [Google Scholar] [CrossRef]
- Alwathnani, H.A.; Perveen, K. Biological control of Fusarium wilt of tomato by antagonist fungi and cyanobacteria. Afr. J. Biotechnol. 2012, 11, 1100–1105. [Google Scholar] [CrossRef]
- Wang, X.W.; Bai, F.Y.; Bensch, K.; Meijer, M.; Sun, B.D.; Han, Y.F.; Crous, P.W.; Samson, R.A.; Yang, F.Y.; Houbraken, J. Phylogenetic re-evaluation of Thielavia with the introduction of a new family Podosporaceae. Stud. Mycol. 2019, 93, 155–252. [Google Scholar] [CrossRef]
- Walther, G.; Wagner, L.; Kurzai, O. Updates on the Taxonomy of Mucorales with an Emphasis on Clinically Important Taxa. J. Fungi 2019, 5, 106. [Google Scholar] [CrossRef]
- CDC. Centers for Disease Control and Prevention, National Center for Emerging and Zoonotic Infectious Diseases (NCEZID), Division of Foodborne, Waterborne, and Environmental Diseases (DFWED). Antifungal-Resistant Aspergillus. Available online: https://www.cdc.gov/fungal/diseases/aspergillosis/antifungal-resistant.html (accessed on 11 July 2022).
- Espinel-Ingroff, A.; Fothergill, A.; Fuller, J.; Johnson, E.; Pelaez, T.; Turnidge, J. Wild-type MIC distributions and epidemiological cutoff values for caspofungin and Aspergillus spp. for the CLSI broth microdilution method (M38-A2 document). Antimicrob. Agents Chemother. 2011, 55, 2855–2859. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Friberg, N.; Mares, M.; Kahlmeter, G.; Meletiadis, J.; Guinea, J.; Andersen, C.T.; Arikan-Akdagli, S.; Barchiesi, F.; Chryssanthou, E.; et al. How to interpret MICs of antifungal compounds according to the revised clinical breakpoints v. 10.0 European committee on antimicrobial susceptibility testing (EUCAST). Clin. Microbiol. Infect. 2020, 26, 1464–1472. [Google Scholar] [CrossRef]
- Wang, H.C.; Hsieh, M.I.; Choi, P.C.; Wu, C.J. Comparison of the Sensititre YeastOne and CLSI M38-A2 microdilution methods in determining the activity of amphotericin B, itraconazole, voriconazole, and posaconazole against Aspergillus species. J. Clin. Microbiol. 2018, 56, e00780-18. [Google Scholar] [CrossRef]





| Identification | Genetic Barcode | EXF-No. 1 | GenBank No. | Month | Habitat | Biosafety Level(BSL) 5 | Other Characteristics or Role in Habitats 6 |
|---|---|---|---|---|---|---|---|
| Acremonium masseei | ITS | EXF-14878 | MT280604 | August 2,3 | BS | No data | colonising plants, soil, freshwater |
| Acremonium sp. | ITS | EXF-14657 | MT280605 | July 2,3 | BS | BSL-1 | colonising plants, soil, freshwater |
| Actinomucor elegans | ITS | EXF-14384 EXF-14449 EXF-14621 EXF-14845 EXF-14849 EXF-14639 | MT280606 MT280607 MT280608 MT280609 MT280610 MT280611 | February, April, June 2,3, July 2,3, August 2,3 | BS | BSL-2 | colonising plants, soil, freshwater, insect-related, potential human pollution indicator |
| Albifimbria verrucaria | ITS | EXF-14006 EXF-14159 EXF-14548 EXF-14648 | MT280612 MT280613 MT280614 MT280615 | May 3, July 2,3, October 4, December | BS | BSL-1 | colonising plants, soil, freshwater |
| Alternaria sp. | ITS | EXF-13999 | MT280685 | October4 | SW | BSL-1 | colonising plants, soil, freshwater |
| Aphanoascus fulvescens | ITS | EXF-14459 EXF-14870 EXF-14871 | MT280616 MT280617 MT280618 | April, September 2,3 | SW | BSL-2 | dermatophyte, “core-genus” in the sand |
| Aphanoascus reticulisporus | ITS | EXF-14632 | MT280619 | June 2,3 | BS | BSL-1 | dermatophyte,“core-genus” in the sand |
| Aphanoascus terreus | ITS | EXF-13894 EXF-14308 EXF-14461 | MT280627 MT280628MT280629 | January, April, October 4 | BS | BSL-1 | dermatophyte,“core-genus” in the sand |
| Arachniotus flavoluteus | ITS | EXF-14420 EXF-14458 EXF-14628 | MT280620 MT280621 MT280622 | March, April, June 2,3 | BS | BSL-1 | dermatophyte |
| Arthrographis curvata | ITS | EXF-14460 | MT280623 | April | BS | BSL-1 | dermatophyte |
| Aspergillus calidoustus | benA | EXF-14640 | MT328464 | July 2,3 | BS | BSL-1 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus destruens | benA | EXF-14429 | MT328468 | February | SW | BSL-1 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus flavus | benA | EXF-13896 EXF-14002 EXF-13898 EXF-13905 EXF-13907 EXF-14163 EXF-14011 EXF-14285 EXF-14296 EXF-14383 EXF-14389 EXF-14404 EXF-14862 | MT328469 MT328470 MT328471 MT328472 MT328473 MT328474 MT328475 MT328476 MT328477 MT328478 MT328479 MT328480 MT328481 | January, February, March, May 3, June 2,3, July 2,3, August 2,3, September 2,3, October 4, November, December | BS | BSL-2 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus floccosus | benA | EXF-14455 EXF-14615 | MT328455 MT328456 | April, June 2,3 | BS | BSL-1 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus jensenii | benA | EXF-14541 EXF-14542 | MT328465 MT328466 | May3 | SW | BSL-1 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus lentulus | ITS | EXF-13910 | MT280624 | October4 | BS | BSL-2 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus nidulans | benA | EXF-14003 EXF-13911 EXF-14014 EXF-14167 EXF-14298 EXF-14388 EXF-14392 EXF-14393 EXF-14405 EXF-14410 EXF-14452 EXF-14643 EXF-14850 EXF-14854 EXF-14465 | MT328430 MT328431 MT328432 MT328433 MT328434 MT328435 MT328436 MT328437 MT328438 MT328439 MT328440 MT328441 MT328442 MT328443 MT328444 | January, February, March, July 2,3, August 2,3, September 2,3, October 4, November, December | BS | BSL-1 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus niger | benA | EXF-14015 EXF-14297 EXF-14558 EXF-14618 EXF-14873 | MT328457 MT328458 MT328459 MT328460 MT328461 | January, May 3, June 2,3, August 2,3, November | BS | BSL-1 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus pseudoglaucus | benA | EXF-14397 | MT328467 | March | SW | BSL-1 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus terreus | benA | EXF-14294 EXF-14387 EXF-14863 | MT328445 MT328446 MT328447 | January, February, September2,3 | BS | BSL-2 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus tubingensis | benA | EXF-14165 EXF-14289 EXF-14381 EXF-14453 EXF-14464 EXF-14555 EXF-14543 | MT328448 MT328449 MT328450 MT328451MT328452 MT328453 MT328454 | January, February, April, May3, December | BS, SW | BSL-1 | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aspergillus welwitchiae | benA | EXF-14400 EXF-14864 | MT328462 MT328463 | March, September 2,3 | BS | No data | dematiaceous, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Aureobasidium melanogenum | ITS | EXF-13886 | MT280686 | October4 | SW | BSL-1 | dematiaceous, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Aureobasidium pullulans | ITS | EXF-14611 EXF-14638 | MT280687 MT280688 | June 2,3, July 2,3 | SW | BSL-1 | dematiaceous, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Bisifusarium delphinoides | tef1 | EXF-14645 EXF-14166 | MT292641 MT292642 | July 2,3, December | BS | BSL-1 | “core-genus” in the sand, colonising plants, soil, freshwater |
| Candida parapsilosis | LSU | EXF-13884 EXF-13882 EXF-13881 EXF-14282 EXF-14300 | MT273264 MT273265 MT273266 MT273267 MT273268 | October 4, December, January | BS | BSL-1 | colonising humans, colonising plants, soil, freshwater |
| Candida tropicalis | LSU | EXF-14299 | MT273269 | January | BS | BSL-2 | colonising humans, colonising plants, soil, freshwater |
| Cephalotrichum gorgonifer | ITS | EXF-14630 | MT280625 | June 2,3 | BS | BSL-1 | rock-inhabiting |
| Chaetomidium fimeti | ITS | EXF-14301 | MT280626 | January | BS | BSL-1 | rock-inhabiting, dematiaceous, colonising plants, soil, freshwater |
| Cladosporium cladosporioides | act | EXF-13913 EXF-13912 EXF-14378 | MT292643 MT292644 MT292645 | February, October 4 | SW | BSL-1 | dematiaceous, psychrotolerant, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Cladosporium halotolerans | act | EXF-14442 EXF-14443 EXF-14004 EXF-14562 | MT292646 MT292647 MT292648 MT292649 | April, May 3 | BS, SW | BSL-1 | dematiaceous, psychrotolerant, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Cladosporium pseudocladosporioides | act | EXF-13897 EXF-14379 | MT292650 MT292651 | February, October4 | SW | BSL-1 | dematiaceous, psychrotolerant, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Cladosporium ramotenellum | act | EXF-13916 EXF-14380 | MT292652 MT292653 | February, October4 | SW | BSL-1 | dematiaceous, psychrotolerant, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Cladosporium sphaerospermum | act | EXF-14540 | MT292656 | May3 | SW | BSL-1 | dematiaceous, psychrotolerant, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Cladosporium tenellum | act | EXF-14001 | MT292654 | October4 | SW | BSL-1 | dematiaceous, psychrotolerant, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Cladosporium velox | act | EXF-13914 | MT292655 | October 4 | SW | BSL-1 | dematiaceous, psychrotolerant, halotolerant, BTEX degrader, colonising plants, soil, freshwater |
| Clonostachys rosea | ITS | EXF-14306 | MT280630 | January | BS | BSL-1 | insect-related, colonising plants, soil, freshwater |
| Condenascus tortuosus | ITS | EXF-14287 EXF-14553 EXF-14554 EXF-14653 EXF-14844 EXF-14858 | MT280631 MT280632 MT280633 MT280634 MT280635 MT280636 | January, May 3, July 2,3, August 2,3, September 2,3 | BS | No data | indicator in bathing season, colonising plants, soil, freshwater |
| Curvularia sp. | ITS | EXF-14855 EXF-14856 | MT280637 MT280638 | September 2,3 | BS | BSL-1 | dematiaceous, colonising plants, soil, freshwater |
| Cystofilobasidium macerans | LSU | EXF-14427 | MT273278 | February | SW | BSL-1 | psychrotolerant, halotolerant |
| Didymella glomerata | ITS | EXF-14613 | MT280689 | June 2,3 | SW | BSL-1 | dematiaceous, colonising plants, soil, freshwater |
| Dipodascus geotrichum | ITS | EXF-14631 EXF-14624 | MT280640 MT280641 | June 2,3 | BS | BSL-1 | colonising humans, plants |
| Exophiala xenobiotica | ITS | EXF-13891 | MT280690 | October 4 | SW | BSL-1 | rock-inhabiting, dematiaceous, BTEX degrader, colonising humans, freshwater |
| Furcasterigmium furcatum | ITS | EXF-14876 | MT280639 | August 2,3 | BS | BSL-1 | rock-inhabiting, colonising plants, soil, freshwater |
| Fusarium brachygibbosum | tef1 | EXF-14451 | MT292640 | April | BS | BSL-1 | “core-genus” in the sand, colonising plants, soil, freshwater |
| Fusarium ipomoeae | tef1 | EXF-14157 | MT292639 | December | BS | BSL-1 | “core-genus” in the sand, colonising plants, soil, freshwater |
| Fusarium nirenbergiae | tef1 | EXF-13888 EXF-14164 EXF-14385 EXF-14399 EXF-14411 EXF-14447 EXF-14448 EXF-14547 EXF-14549 EXF-14626 EXF-14641 EXF-14860 EXF-14843 | MT292623 MT292624 MT292625 MT292626 MT292627 MT292628 MT292629 MT292630 MT292631 MT292632 MT292633 MT292634 MT292635 | February, March, April, May 3, June 2,3, July 2,3, August 2,3, September 2,3, October 4, December | BS | BSL-1 | “core-genus” in the sand, colonising plants, soil, freshwater |
| Fusarium solani | tef1 | EXF-14848 EXF-14005 EXF-14288 | MT292636 MT292637 MT292638 | January, August 2,3, October 4 | BS | BSL-2 | “core-genus” in the sand, colonising plants, soil, freshwater |
| Geotrichum silvicola | ITS | EXF-14629 | MT280642 | June 2,3 | BS | BSL-1 | colonising humans, colonising plants, soil, freshwater |
| Gliomastix roseogrisea | ITS | EXF-14564 | MT280643 | May 3 | BS | BSL-1 | BTEX degrader, dematiaceous, insect-related, colonising plants, soil, freshwater |
| Gloeotinia sp. | ITS | EXF-14407 EXF-14868 | MT280644 MT280645 | March, September 2,3 | BS | No data | psychrotolerant, colonising plants, soil, freshwater |
| Hortaea werneckii | ITS | EXF-14000 | MT280691 | October 4 | SW | BSL-1 | dematiaceous, halophilic |
| Humicola homopilata | ITS | EXF-14398 | MT280646 | March | BS | No data | rock-inhabiting, dematiaceous, colonising plants, soil, freshwater |
| Lenzites betulinus | ITS | EXF-14377 | MT280692 | February | SW | No data | colonising plants, soil, freshwater |
| Metarhizium marquandii | ITS | EXF-14305 | MT280647 | January | BS | BSL-1 | insect-related, colonising plants, soil, freshwater |
| Meyerozyma caribbica | LSU | EXF-14647 EXF-14650 EXF-14655 EXF-14881 EXF-14867 EXF-13879 | MT273270 MT273271 MT273272 MT273273 MT273274 MT273275 | July 2,3, August 2,3, September 2,3, October 4 | BS | BSL-1 | colonising humans, colonising plants, soil, freshwater, potential human pollution indicator |
| Meyerozyma guilliermondii | LSU | EXF-13885 | MT273279 | October 4 | SW | BSL-1 | colonising humans, colonising plants, soil, freshwater, potential human pollution indicator |
| Microascus brevicaulis | ITS | EXF-14307 | MT280648 | January | BS | BSL-2 | colonising plants, soil, freshwater |
| Mucor circinelloides | ITS | EXF-14403 EXF-14853 EXF-14450 EXF-14847 | MT280649 MT280650 MT280651 MT280652 | March, April, August 2,3, September 2,3 | BS | BSL-1 | colonising plants, soil, freshwater |
| Neocosmospora rubicola | ITS | EXF-13893 | MT280653 | October 4 | BS | No data | colonising plants, soil, freshwater |
| Neomicrosphaeropsis italica | ITS | EXF-14074 | MT280693 | November | SW | No data | colonising plants, soil, freshwater |
| Ochroconis sp. | ITS | EXF-14160 | MT280654 | December | BS | BSL-1 | dematiaceous, BTEX degrader, colonising plants, soil, freshwater |
| Papiliotrema fonsecae | LSU | EXF-14426 | MT273280 | February | SW | BSL-1 | halotolerant |
| Paraconiothyrium cyclothyrioides | ITS | EXF-13917 EXF-14007 EXF-14614 | MT280694 MT280695 MT280696 | June 2,3, October 4, November | SW | BSL-1 | halotolerant |
| Paraphoma radicina | ITS | EXF-14550 | MT280655 | May 3 | BS | BSL-1 | dematiceaous, colonising plants, soil, freshwater |
| Penicillium aurantiogriseum | benA | EXF-13906 EXF-14386 EXF-14402 EXF-14644 | MT328501 MT328502 MT328503 MT328504 | February, March, July 2,3, October 4 | BS | BSL-1 | psychrotolerant, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Penicillium crustosum | benA | EXF-14646 EXF-14444 | MT328495 MT328496 | April, July 2,3 | BS, SW | BSL-1 | psychrotolerant, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Penicillium glabrum | benA | EXF-14169 | MT328513 | December | BS | BSL-1 | psychrotolerant, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Penicillium griseofulvum | benA | EXF-13899 EXF-14170 EXF-14172 EXF-14156 EXF-14158 EXF-14423 EXF-14552 EXF-14563 | MT328505 MT328506 MT328507 MT328508 MT328509 MT328510 MT328511 MT328512 | March, May 3, October 4, December | BS | BSL-1 | psychrotolerant, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Penicillium oxalicum | benA | EXF-14292 EXF-14445 | MT328493 MT328494 | January, April | BS | BSL-1 | psychrotolerant, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Penicillium sizovae | benA | EXF-14880 | MT328498 | August 2,3 | BS | BSL-1 | psychrotolerant, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Penicillium sp. | benA | EXF-14610 | MT328497 | June 2,3 | SW | BSL-1 | psychrotolerant, halotolerant, “core-genus” in the sand, colonising plants, soil, freshwater |
| Phaeoacremonium iranianum | ITS | EXF-14302 | MT280656 | January | BS | No data | colonising plants, soil, freshwater |
| Phialophora americana | ITS | EXF-14409 EXF-14879 | MT280659 MT280660 | March, August 2,3 | BS | BSL-2 | dematiaceous, BTEX degrader, colonising plants, soil, freshwater |
| Phialophora sp. | ITS | EXF-13890 EXF-14073 | MT280657 MT280658 | October 4 | BS | BSL-1 | dematiaceous, colonising plants, soil, freshwater |
| Purpureocillium sp. | benA | EXF-14303 EXF-14421 | MT328499 MT328500 | January, March | BS | BSL-1/-2 | rock-inhabiting, halotolerant, colonising plants, soil, freshwater |
| Pyrenochaetopsis tabarestanensis | ITS | EXF-14642 | MT280661 | July 2,3 | BS | No data | colonising plants, soil, freshwater |
| Rhizopus arrhizus | ITS | EXF-13903 EXF-13909 EXF-14009 EXF-14012 EXF-14168 EXF-14290 EXF-14291 EXF-14293 EXF-14390 EXF-14454 EXF-14556 EXF-14627 EXF-14649 EXF-14846 EXF-14852 | MT280662 MT280663 MT280664 MT280665 MT280666 MT280667 MT280668 MT280669 MT280670 MT280671 MT280672 MT280673 MT280674 MT280675 MT280676 | January, February, April, May 3, June 2,3, July 2,3, August 2,3, September 2,3, October 4, November, December | BS | BSL-2 | “core-genus” in the sand, colonising plants, soil, freshwater |
| Rhodotorula graminis | LSU | EXF-14539 | MT273282 | January | SW | BSL-1 | psychrotolerant, halotolerant, BTEX-degrader, colonising plants, soil, freshwater |
| Rhodotorula mucilaginosa | LSU | EXF-14446 | MT273276 | April | BS | BSL-1 | halotolerant, BTEX-degrader, colonising human, plants, soil, freshwater |
| Sarocladium kiliense | ITS | EXF-13880 EXF-14408 | MT280677 MT280678 | March, October4 | BS | BSL-2 | insect-related, colonising plants, soil, freshwater |
| Sordaria nodulifera | ITS | EXF-14010 | MT280679 | November | BS | No data | colonising plants, soil, freshwater |
| Stachybotrys sp. | ITS | EXF-14565 EXF-14654 EXF-14875 | MT280680 MT280681 MT280682 | May 3, July 2,3, August 2,3, September 2,3 | BS | BSL-1 | colonising plants, soil, freshwater, potential human pollution indicator |
| Stereum hirsutum | ITS | EXF-13915 | MT280697 | October4 | SW | BSL-1 | colonising plants, soil, freshwater |
| Talaromyces atroroseus | benA | EXF-14286 | MT328482 | January | BS | BSL-1 | insect-related, colonising plants, soil, freshwater, “core-genus” in the sand |
| Talaromyces pinophilus | benA | EXF-14406 EXF-13900 EXF-14017 EXF-13904 EXF-13902 EXF-14415 EXF-13901 EXF-14620 EXF-14633 | MT328483 MT328484 MT328485 MT328486 MT328487 MT328488 MT328489 MT328490 MT328491 | March, May 3, October 4, November | BS | BSL-1 | insect-related, colonising plants, soil, freshwater, “core-genus” in the sand |
| Talaromyces purpureogenus | benA | EXF-13895 | MT328492 | October 4 | BS | BSL-1 | insect-related, colonising plants, soil, freshwater, “core-genus” in the sand |
| Trichoderma citrinoviride | ITS | EXF-14412 EXF-14619 | MT280683 MT280684 | March, June 2,3 | BS | BSL-1 | colonising plants, soil, freshwater |
| Trichosporon asahii | LSU | EXF-14865 | MT273277 | September 2,3 | BS | BSL-2 | dermatophyte |
| Wickerhamomyces anomalus | LSU | EXF-14842 | MT273283 | August 2,3 | SW | BSL-2 | colonising humans, plants, soil, freshwater |
| Identification | EXF-No. | Range of Minimal Inhibitory Concentration (MIC) [µg/mL] | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Anidulafungin (AND), 24 h | Micafungin (MF), 24 h | Caspofungin (CAS), 24 h | Amphotericin B (AB), 48 h | 5-Flucytosine (FC), 48 h | Posaconazole (PZ), 48 h | Voriconazole (VOR), 48 h | Itraconazole (IZ), 48 h | Fluconazole (FZ), 48 h | ||
| Aspergillus flavus | EXF-14617 | 0.03 4 | 0.06 4 | 1 4 | 2–4 | >64 4 | 0.06 4 | 0.5–1 | 0.06 4 | ≥256 4 |
| EXF-14651 | ||||||||||
| EXF-14874 | ||||||||||
| EXF-14862 | ||||||||||
| ECOFF 1 | IE 2 | IE 2 | IE 2 | 4 | no data | 0.5 | 2 | 1 | / 3 | |
| Aspergillus nidulans | EXF-14643 | 0.03 4 | 0.12 4 | 0.25 4 | 2–4 | >64 4 | 0.06–0.12 | 0.12–0.25 | 0.03–0.06 | ≥256 4 |
| EXF-14850 | ||||||||||
| EXF-14854 | ||||||||||
| ECOFF 1 | IE 2 | IE 2 | IE 2 | 4 | no data | 0.5 | 1 | 1 | / 3 | |
| Aspergillus niger | EXF-14618 | 0.03 4 | 0.12 4 | 1 4 | 2 4 | 32 4 | 0.12 4 | 1 4 | 0.25 4 | ≥256 4 |
| EXF-14873 | ||||||||||
| ECOFF 1 | IE 2 | IE 2 | IE 2 | 0.5–1 | no data | 0.25–0.5 | 2 | 4 | / 3 | |
| Aspergillus welwitschiae | EXF-14864 | 0.015 | 0.06 | 0.5 | 2 | 16 | 0.12 | 0.5 | 0.12 | ≥256 |
| ECOFF 1 | no data | no data | no data | no data | no data | no data | no data | no data | no data | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novak Babič, M.; Gunde-Cimerman, N.; Breskvar, M.; Džeroski, S.; Brandão, J. Occurrence, Diversity and Anti-Fungal Resistance of Fungi in Sand of an Urban Beach in Slovenia—Environmental Monitoring with Possible Health Risk Implications. J. Fungi 2022, 8, 860. https://doi.org/10.3390/jof8080860
Novak Babič M, Gunde-Cimerman N, Breskvar M, Džeroski S, Brandão J. Occurrence, Diversity and Anti-Fungal Resistance of Fungi in Sand of an Urban Beach in Slovenia—Environmental Monitoring with Possible Health Risk Implications. Journal of Fungi. 2022; 8(8):860. https://doi.org/10.3390/jof8080860
Chicago/Turabian StyleNovak Babič, Monika, Nina Gunde-Cimerman, Martin Breskvar, Sašo Džeroski, and João Brandão. 2022. "Occurrence, Diversity and Anti-Fungal Resistance of Fungi in Sand of an Urban Beach in Slovenia—Environmental Monitoring with Possible Health Risk Implications" Journal of Fungi 8, no. 8: 860. https://doi.org/10.3390/jof8080860
APA StyleNovak Babič, M., Gunde-Cimerman, N., Breskvar, M., Džeroski, S., & Brandão, J. (2022). Occurrence, Diversity and Anti-Fungal Resistance of Fungi in Sand of an Urban Beach in Slovenia—Environmental Monitoring with Possible Health Risk Implications. Journal of Fungi, 8(8), 860. https://doi.org/10.3390/jof8080860








