Decolorization of Textile Azo Dye via Solid-State Fermented Wheat Bran by Lasiodiplodia sp. YZH1
Abstract
:1. Introduction
2. Materials and Methods
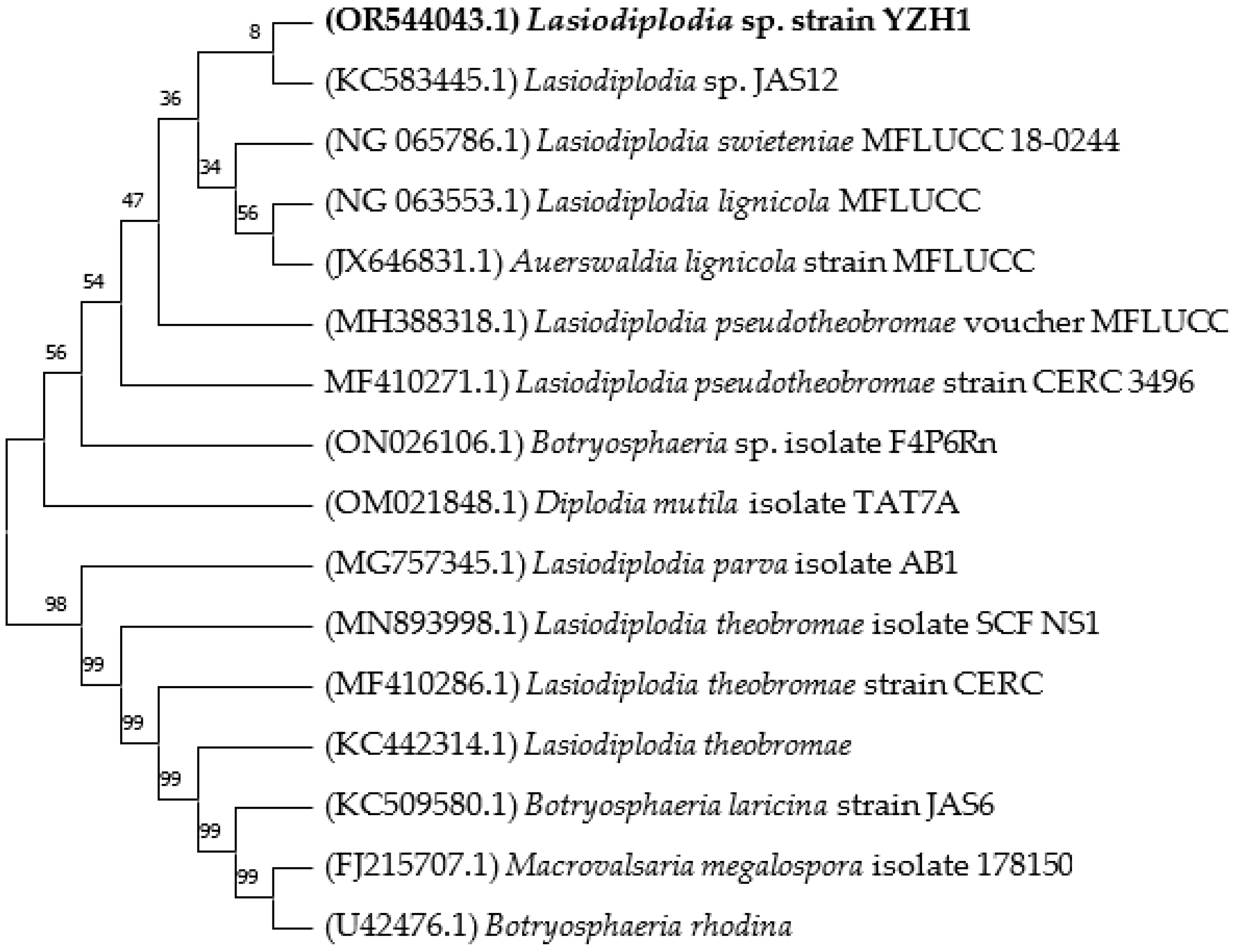
2.1. Isolation and Identification of the Fungal Isolate
2.2. Solid-State Fermentation of Wheat Bran
2.3. Enzyme Extraction and Assay
2.4. Dye Removal Assays
2.5. Optimization of Dye Removal
2.6. Characterization of Wheat Bran
2.7. Dye Removal Assay in Liquid Cultures
3. Results and Discussion
3.1. Isolation and Identification of the Fungal Isolate
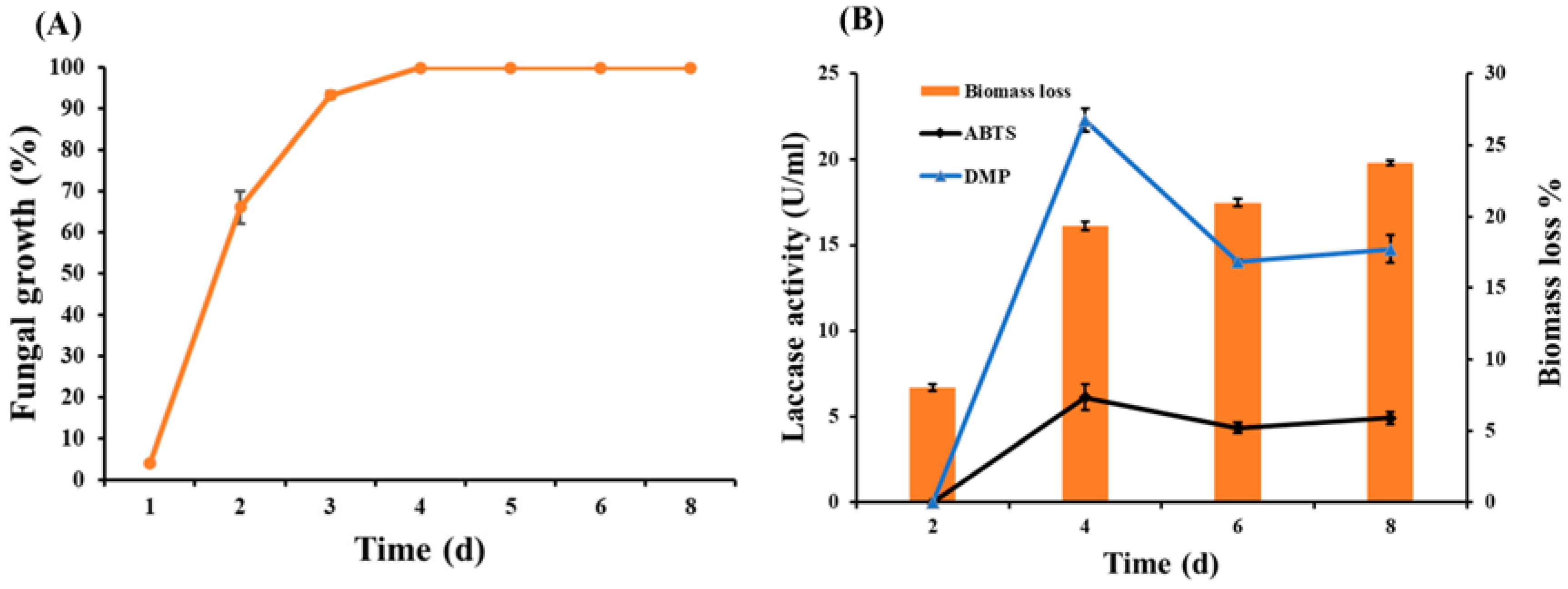
3.2. Fungal Growth, Enzyme Assay, and Biomass Loss during SSF of Wheat Bran
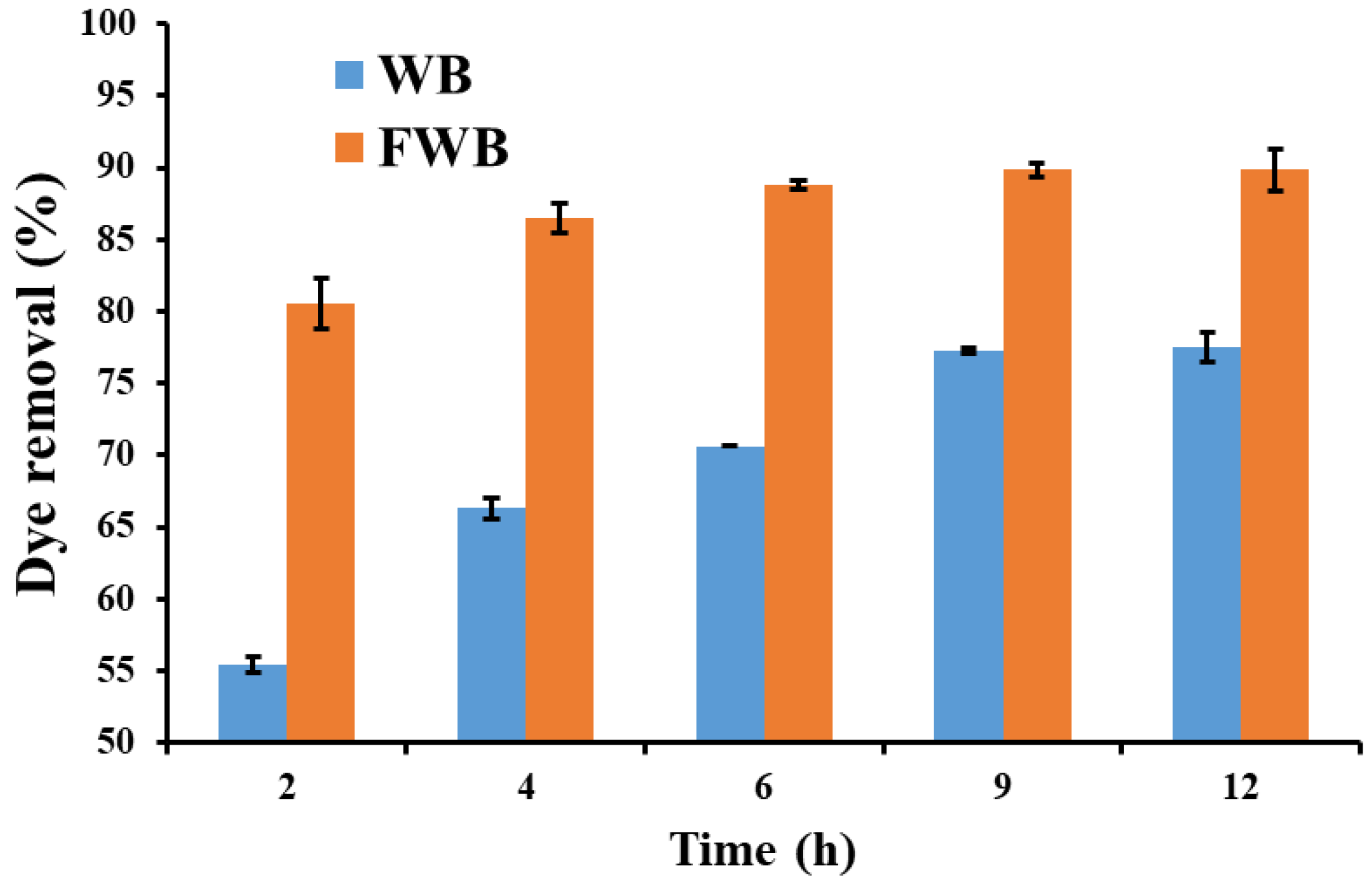
3.3. Dye Removal Efficiency
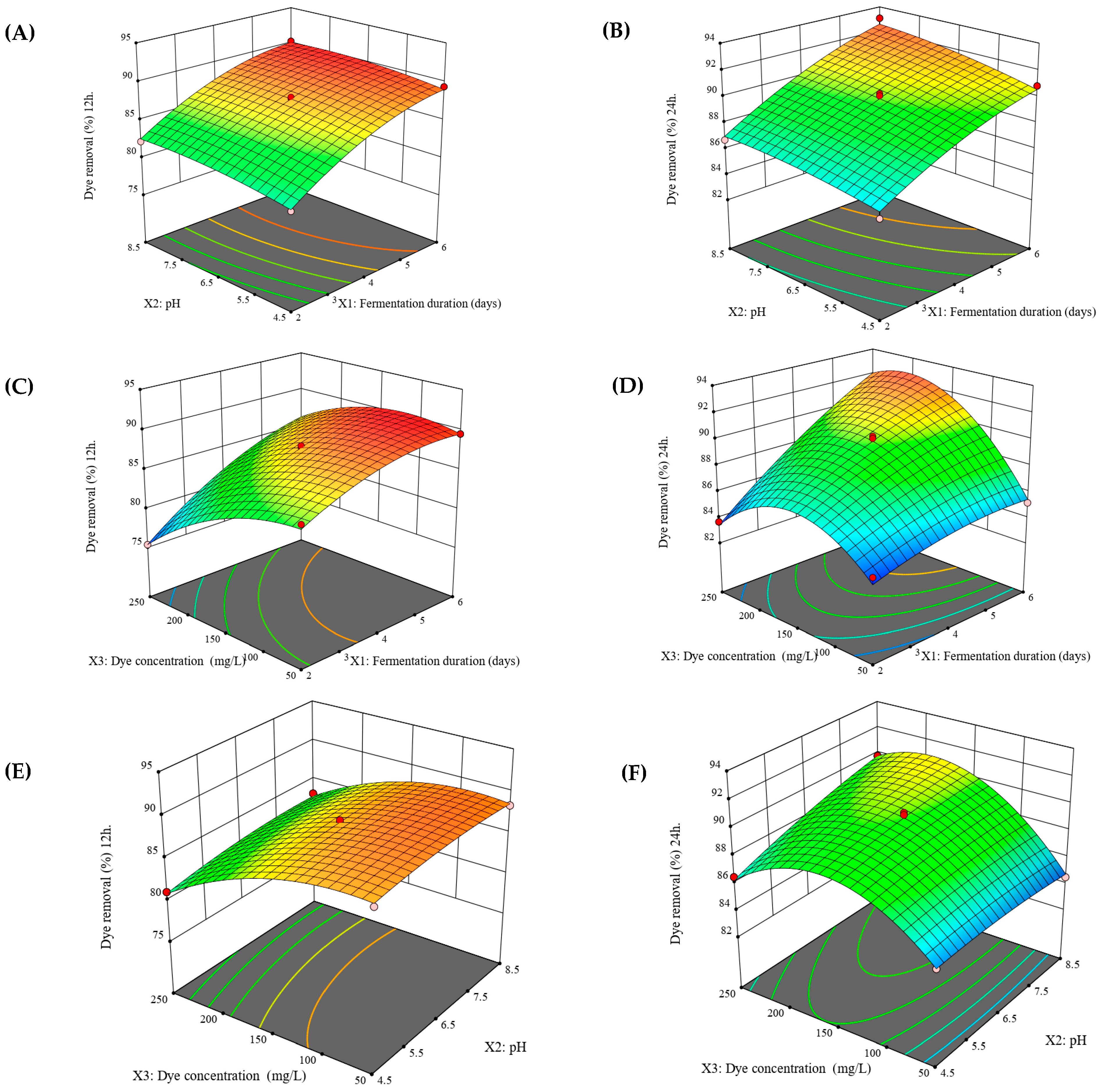
3.4. Optimization of Dye Removal
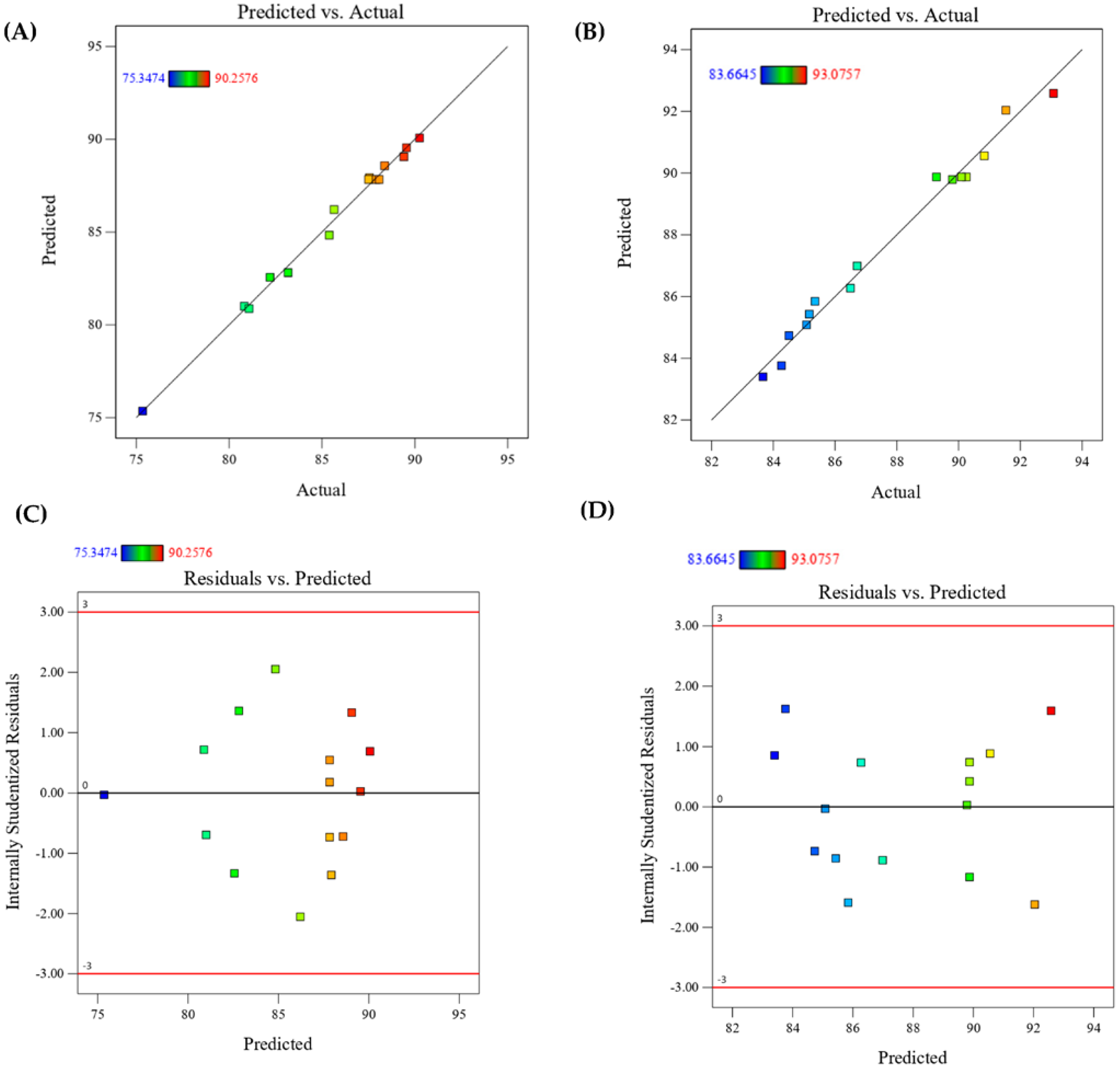
3.5. Validation of the Design Model
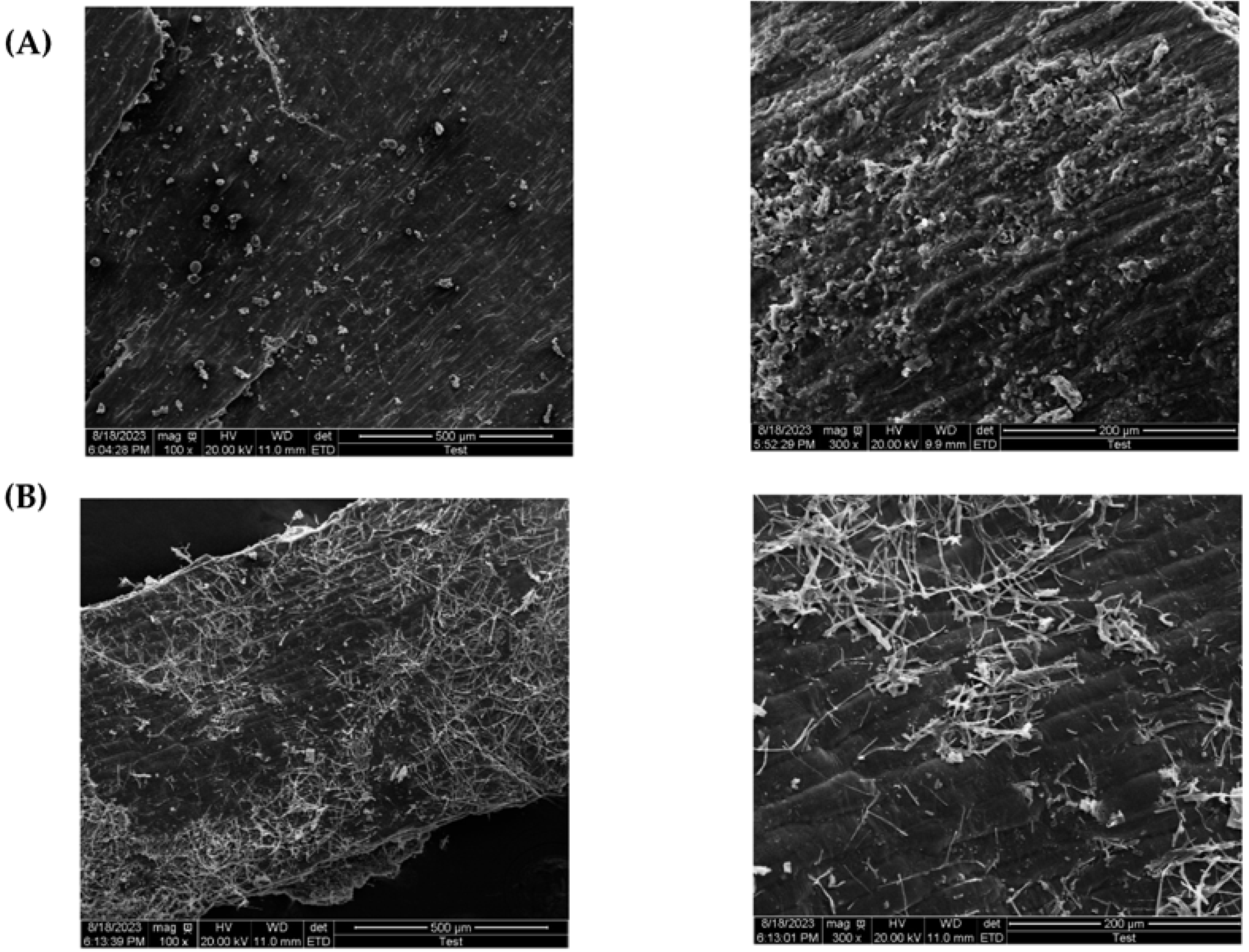
3.6. Characterization of Wheat Bran
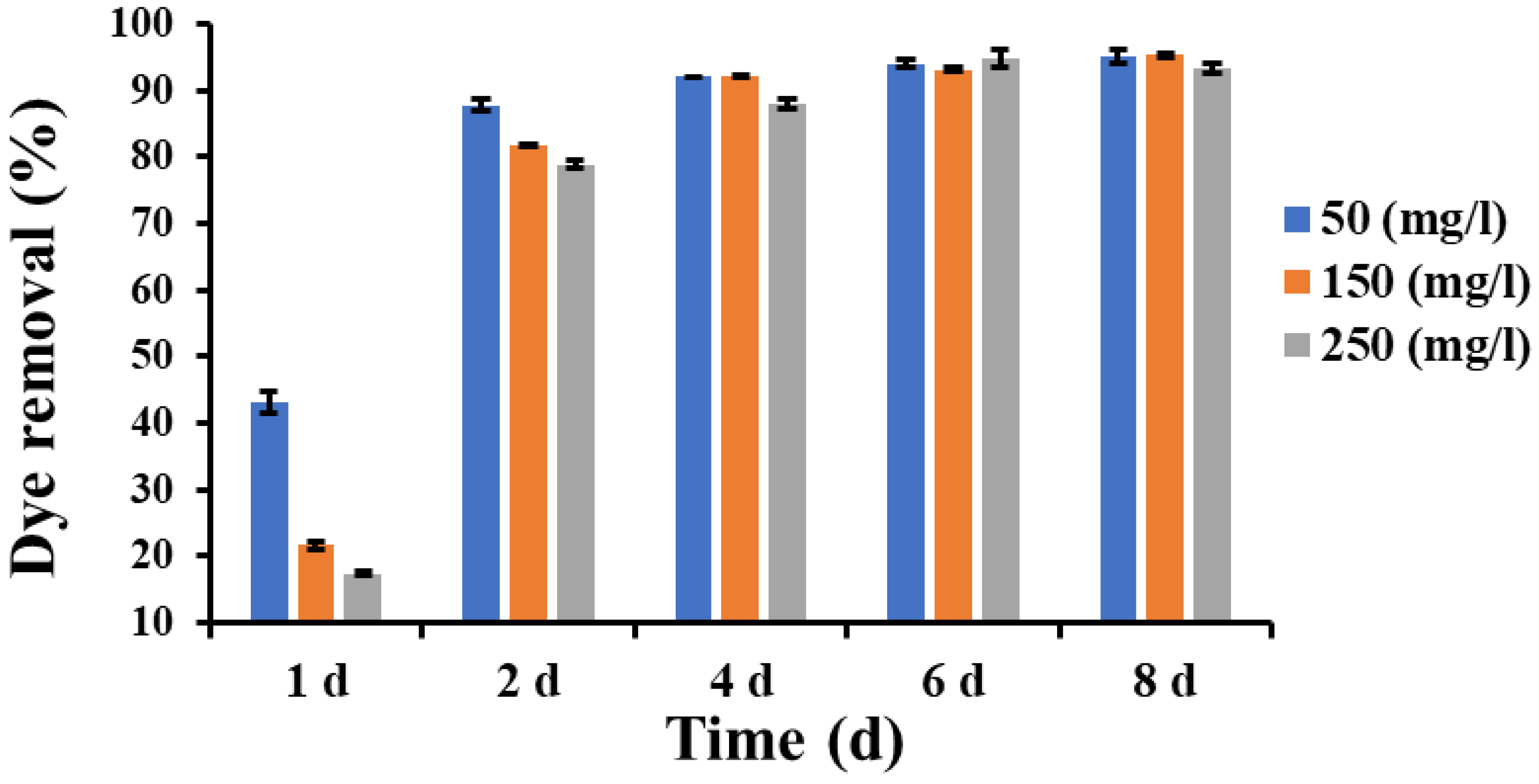
3.7. Dye Removal Assay in Liquid Cultures
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhou, Y.; Lu, J.; Zhou, Y.; Liu, Y. Recent advances for dyes removal using novel adsorbents: A review. Environ. Pollut. 2019, 252, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Ardila-Leal, L.D.; Poutou-Pinales, R.A.; Pedroza-Rodriguez, A.M.; Quevedo-Hidalgo, B.E. A Brief History of Colour, the Environmental Impact of Synthetic Dyes and Removal by Using Laccases. Molecules 2021, 26, 3813. [Google Scholar] [CrossRef] [PubMed]
- Lellis, B.; Fávaro-Polonio, C.Z.; Pamphile, J.A.; Polonio, J.C. Effects of textile dyes on health and the environment and bioremediation potential of living organisms. Biotechnol. Res. Innov. 2019, 3, 275–290. [Google Scholar] [CrossRef]
- Ehrampoush, M.; Ghanizadeh, G.; Ghaneian, M. Equilibrium and kinetics study of reactive red 123 dye removal from aqueous solution by adsorption on eggshell. J. Environ. Health Sci. Eng. 2011, 8, 101–106. [Google Scholar]
- Zhang, J.; Feng, M.; Jiang, Y.; Hu, M.; Li, S.; Zhai, Q. Efficient decolorization/degradation of aqueous azo dyes using buffered H2O2 oxidation catalyzed by a dosage below ppm level of chloroperoxidase. Chem. Eng. J. 2012, 191, 236–242. [Google Scholar] [CrossRef]
- Chung, K.-T. Azo dyes and human health: A review. J. Environ. Sci. Health Part C 2016, 34, 233–261. [Google Scholar] [CrossRef] [PubMed]
- Ajaz, M.; Elahi, A.; Rehman, A. Degradation of azo dye by bacterium, Alishewanella sp. CBL-2 isolated from industrial effluent and its potential use in decontamination of wastewater. J. Water Reuse Desalination 2018, 8, 507–515. [Google Scholar] [CrossRef]
- Baena-Baldiris, D.; Montes-Robledo, A.; Baldiris-Avila, R. Franconibacter sp., 1MS: A new strain in decolorization and degradation of azo dyes ponceau s red and methyl orange. ACS Omega 2020, 5, 28146–28157. [Google Scholar] [CrossRef]
- Devi, P.; Saroha, A.K. Utilization of sludge based adsorbents for the removal of various pollutants: A review. Sci. Total Environ. 2017, 578, 16–33. [Google Scholar] [CrossRef]
- Routoula, E.; Patwardhan, S.V. Degradation of anthraquinone dyes from effluents: A review focusing on enzymatic dye degradation with industrial potential. Environ. Sci. Technol. 2020, 54, 647–664. [Google Scholar] [CrossRef]
- Wanyonyi, W.C.; Onyari, J.M.; Shiundu, P.M.; Mulaa, F.J. Biodegradation and detoxification of malachite green dye using novel enzymes from Bacillus cereus strain KM201428: Kinetic and metabolite analysis. Energy Procedia 2017, 119, 38–51. [Google Scholar] [CrossRef]
- Vishwakarma, S.; Singh, M.; Srivastava, A.; Pandey, V. Azo dye (direct blue 14) decolorization by immobilized extracellular enzymes of Pleurotus species. Cell. Mol. Biol. 2012, 58, 21–25. [Google Scholar] [PubMed]
- Gupta, V. Application of low-cost adsorbents for dye removal–a review. J. Environ. Manag. 2009, 90, 2313–2342. [Google Scholar] [CrossRef]
- Abdolali, A.; Guo, W.; Ngo, H.; Chen, S.; Nguyen, N.; Tung, K. Typical lignocellulosic wastes and by-products for biosorption process in water and wastewater treatment: A critical review. Bioresour. Technol. 2014, 160, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.-L.; Wang, R.-Z.; Liu, Y.-G.; Zeng, G.-M.; Lai, C.; Xu, P.; Lu, B.-A.; Xu, J.-J.; Wang, C.; Huang, C. Application of molecularly imprinted polymers in wastewater treatment: A review. Environ. Sci. Pollut. Res. 2015, 22, 963–977. [Google Scholar] [CrossRef] [PubMed]
- Hokkanen, S.; Bhatnagar, A.; Sillanpää, M. A review on modification methods to cellulose-based adsorbents to improve adsorption capacity. Water Res. 2016, 91, 156–173. [Google Scholar] [CrossRef]
- Choi, J.; Won, W.; Capareda, S.C. The economical production of functionalized Ashe juniper derived-biochar with high hazardous dye removal efficiency. Ind. Crops Prod. 2019, 137, 672–680. [Google Scholar] [CrossRef]
- Kadam, A.A.; Telke, A.A.; Jagtap, S.S.; Govindwar, S.P. Decolorization of adsorbed textile dyes by developed consortium of Pseudomonas sp. SUK1 and Aspergillus ochraceus NCIM-1146 under solid state fermentation. J. Hazard. Mater. 2011, 189, 486–494. [Google Scholar] [CrossRef]
- Kadam, A.A.; Kamatkar, J.D.; Khandare, R.V.; Jadhav, J.P.; Govindwar, S.P. Solid-state fermentation: Tool for bioremediation of adsorbed textile dyestuff on distillery industry waste-yeast biomass using isolated Bacillus cereus strain EBT1. Environ. Sci. Pollut. Res. 2013, 20, 1009–1020. [Google Scholar] [CrossRef]
- Waghmare, P.R.; Kadam, A.A.; Saratale, G.D.; Govindwar, S.P. Enzymatic hydrolysis and characterization of waste lignocellulosic biomass produced after dye bioremediation under solid state fermentation. Bioresour. Technol. 2014, 168, 136–141. [Google Scholar] [CrossRef]
- Singh, G.; Dwivedi, S.K. Decolorization and degradation of Direct Blue-1 (Azo dye) by newly isolated fungus Aspergillus terreus GS28, from sludge of carpet industry. Environ. Technol. Innov. 2020, 18, 100751. [Google Scholar] [CrossRef]
- Asses, N.; Ayed, L.; Hkiri, N.; Hamdi, M. Congo Red Decolorization and Detoxification by Aspergillus niger: Removal Mechanisms and Dye Degradation Pathway. BioMed Res. Int. 2018, 2018, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Reda, F.M.; El-Mekkawy, R.M.; Hassan, N.S. Detoxification and Bioremediation of Sulfa Drugs and Synthetic Dyes by Streptomyces mutabilis A17 Laccase Produced in Solid State Fermentation. J. Pure Appl. Microbiol. 2019, 13, 85–96. [Google Scholar] [CrossRef]
- Karimifard, S.; Moghaddam, M.R.A. Application of response surface methodology in physicochemical removal of dyes from wastewater: A critical review. Sci. Total Environ. 2018, 640, 772–797. [Google Scholar] [CrossRef]
- Archin, S. Optimization of process parameters by response surface methodology for methylene blue removal using cellulose dusts. Civ. Eng. J. 2018, 4, 3. [Google Scholar]
- Abdulhameed, A.S.; Mohammad, A.-T.; Jawad, A.H. Application of response surface methodology for enhanced synthesis of chitosan tripolyphosphate/TiO2 nanocomposite and adsorption of reactive orange 16 dye. J. Clean. Prod. 2019, 232, 43–56. [Google Scholar] [CrossRef]
- Mohammad, A.-T.; Abdulhameed, A.S.; Jawad, A.H. Box-Behnken design to optimize the synthesis of new crosslinked chitosan-glyoxal/TiO2 nanocomposite: Methyl orange adsorption and mechanism studies. Int. J. Biol. Macromol. 2019, 129, 98–109. [Google Scholar] [CrossRef]
- Gadekar, M.R.; Ahammed, M.M. Modelling dye removal by adsorption onto water treatment residuals using combined response surface methodology-artificial neural network approach. J. Environ. Manag. 2019, 231, 241–248. [Google Scholar] [CrossRef]
- Karri, R.R.; Tanzifi, M.; Yaraki, M.T.; Sahu, J. Optimization and modeling of methyl orange adsorption onto polyaniline nano-adsorbent through response surface methodology and differential evolution embedded neural network. J. Environ. Manag. 2018, 223, 517–529. [Google Scholar] [CrossRef]
- Duan, X.; Liu, X.; Xiao, S.; Du, C.; Yan, B. Fe-Trimesic Acid/Melamine Gel-Derived Fe/N-Doped Carbon Nanotubes as Catalyst of Peroxymonosulfate to Remove Sulfamethazine. Water 2023, 15, 381. [Google Scholar] [CrossRef]
- Yang, Y.; Zheng, Z.; Zhang, D.; Zhang, X. Response surface methodology directed adsorption of chlorate and chlorite onto MIEX resin and study of chemical properties. Environ. Sci. Water Res. Technol. 2020, 6, 2454–2464. [Google Scholar] [CrossRef]
- Shahamat, Y.D.; Masihpour, M.; Borghei, P.; Rahmati, S.H. Removal of azo red-60 dye by advanced oxidation process O3/UV from textile wastewaters using Box-Behnken design. Inorg. Chem. Commun. 2022, 143, 109785. [Google Scholar] [CrossRef]
- Prabhakar, Y.; Gupta, A.; Kaushik, A. Microbial degradation of reactive red-35 dye: Upgraded progression through Box–Behnken design modeling and cyclic acclimatization. J. Water Process Eng. 2021, 40, 101782. [Google Scholar] [CrossRef]
- Choińska-Pulit, A.; Sobolczyk-Bednarek, J.; Łaba, W. Optimization of copper, lead and cadmium biosorption onto newly isolated bacterium using a Box-Behnken design. Ecotoxicol. Environ. Saf. 2018, 149, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Parmar, P.; Shukla, A.; Goswami, D.; Patel, B.; Saraf, M. Optimization of cadmium and lead biosorption onto marine Vibrio alginolyticus PBR1 employing a Box-Behnken design. Chem. Eng. J. Adv. 2020, 4, 100043. [Google Scholar] [CrossRef]
- Matrawy, A.A.; Marey, H.S.; Embaby, A.M. The Agro-industrial Byproduct Wheat Bran as an Inducer for Alkaline Protease (ALK-PR23) Production by Pschyrotolerant Lysinibacillus sphaericus Strain AA6 EMCCN3080. Waste Biomass Valorization 2023, 1–16. [Google Scholar] [CrossRef]
- Kler, R.; Gangurde, R.; Elmirzaev, S.; Hossain, M.S.; Vo, N.V.; Nguyen, T.V.; Kumar, P.N. Optimization of Meat and Poultry Farm Inventory Stock Using Data Analytics for Green Supply Chain Network. Discret. Dyn. Nat. Soc. 2022, 2022, 1–8. [Google Scholar] [CrossRef]
- Liu, J.; Sidhu, S.S.; Wang, M.L.; Tonnis, B.; Habteselassie, M.; Mao, J.; Huang, Q. Evaluation of various fungal pretreatment of switchgrass for enhanced saccharification and simultaneous enzyme production. J. Clean. Prod. 2015, 104, 480–488. [Google Scholar] [CrossRef]
- Osma, J.F.; Herrera, J.L.T.; Couto, S.R. Banana skin: A novel waste for laccase production by Trametes pubescens under solid-state conditions. Application to synthetic dye decolouration. Dye. Pigment. 2007, 75, 32–37. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, J.; Ning, Y.; Liao, X.; Jia, Y. Eichhornia crassipes: Agro-waster for a novel thermostable laccase production by Pycnoporus sanguineus SYBC-L1. J. Biosci. Bioeng. 2017, 123, 163–169. [Google Scholar] [CrossRef]
- Torres-Farradá, G.; Manzano Leon, A.M.; Rineau, F.; Ledo Alonso, L.L.; Sánchez-López, M.I.; Thijs, S.; Colpaert, J.; Ramos-Leal, M.; Guerra, G.; Vangronsveld, J. Diversity of ligninolytic enzymes and their genes in strains of the genus Ganoderma: Applicable for biodegradation of xenobiotic compounds? Front. Microbiol. 2017, 8, 898. [Google Scholar] [CrossRef] [PubMed]
- Dayi, B.; Kyzy, A.D.; Abduloglu, Y.; Cikrikci, K.; Ardag Akdogan, H. Investigation of the ability of immobilized cells to different carriers in removal of selected dye and characterization of environmentally friendly laccase of Morchella esculenta. Dye. Pigment. 2018, 151, 15–21. [Google Scholar] [CrossRef]
- Latif, W.; Ciniglia, C.; Iovinella, M.; Shafiq, M.; Papa, S. Role of White Rot Fungi in Industrial Wastewater Treatment: A Review. Appl. Sci. 2023, 13, 8318. [Google Scholar] [CrossRef]
- Jami, F.; Slippers, B.; Wingfield, M.J.; Gryzenhout, M. Greater Botryosphaeriaceae diversity in healthy than associated diseased Acacia karroo tree tissues. Australas. Plant Pathol. 2013, 42, 421–430. [Google Scholar] [CrossRef]
- Arunprasath, T.; Sudalai, S.; Meenatchi, R.; Jeyavishnu, K.; Arumugam, A. Biodegradation of triphenylmethane dye malachite green by a newly isolated fungus strain. Biocatal. Agric. Biotechnol. 2019, 17, 672–679. [Google Scholar] [CrossRef]
- Deng, Z.; Zhang, R.; Shi, Y.; Hu, L.a.; Tan, H.; Cao, L. Characterization of Cd-, Pb-, Zn-resistant endophytic Lasiodiplodia sp. MXSF31 from metal accumulating Portulaca oleracea and its potential in promoting the growth of rape in metal-contaminated soils. Environ. Sci. Pollut. Res. 2014, 21, 2346–2357. [Google Scholar] [CrossRef]
- Aminin, A.L.; Ulvairoh, U.; Mulyani, N.S.; Gunawan, G. Optimization of solid state fermentation of banana peel for removal of crystal violet dye from aqueous solution. In AIP Conference Proceedings; AIP Publishing: New York, NY, USA, 2022. [Google Scholar]
- Liu, J.; Wang, Z.; Li, H.; Hu, C.; Raymer, P.; Huang, Q. Effect of solid state fermentation of peanut shell on its dye adsorption performance. Bioresour. Technol. 2018, 249, 307–314. [Google Scholar] [CrossRef]
- Ortiz-Monsalve, S.; Gutterres, M.; Valente, P.; Plácido, J.; Bustamante-López, S.; Kelly, D.; Kelly, S.L. Degradation of a leather-dye by the combination of depolymerised wood-chip biochar adsorption and solid-state fermentation with Trametes villosa SCS-10. Bioresour. Bioprocess. 2020, 7, 61. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc. A Guide Methods Appl. 1990, 18, 315–322. [Google Scholar]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547. [Google Scholar] [CrossRef]
- Bharti, V.; Vikrant, K.; Goswami, M.; Tiwari, H.; Sonwani, R.K.; Lee, J.; Tsang, D.C.; Kim, K.-H.; Saeed, M.; Kumar, S. Biodegradation of methylene blue dye in a batch and continuous mode using biochar as packing media. Environ. Res. 2019, 171, 356–364. [Google Scholar] [CrossRef]
- Copete-Pertuz, L.S.; Alandete-Novoa, F.; Placido, J.; Correa-Londono, G.A.; Mora-Martinez, A.L. Enhancement of ligninolytic enzymes production and decolourising activity in Leptosphaerulina sp. by co-cultivation with Trichoderma viride and Aspergillus terreus. Sci. Total Environ. 2019, 646, 1536–1545. [Google Scholar] [CrossRef]
- Ferreira, T.; Rasband, W. ImageJ user guide. ImageJ 2012, 1, 155–161. [Google Scholar]
- Smits, J.P.; Rinzema, A.; Tramper, J.; Van Sonsbeek, H.M.; Knol, W. Solid-state fermentation of wheat bran by Trichoderma reesei QM9414—Substrate composition changes, C balance, enzyme production, growth and kinetics. Appl. Microbiol. Biotechnol. 1996, 46, 489–496. [Google Scholar] [CrossRef]
- Dayi, B.; Onac, C.; Kaya, A.; Akdogan, H.A.; Rodriguez-Couto, S. New Type Biomembrane: Transport and Biodegradation of Reactive Textile Dye. ACS Omega 2020, 5, 9813–9819. [Google Scholar] [CrossRef] [PubMed]
- Forootanfar, H.; Rezaei, S.; Zeinvand-Lorestani, H.; Tahmasbi, H.; Mogharabi, M.; Ameri, A.; Faramarzi, M.A. Studies on the laccase-mediated decolorization, kinetic, and microtoxicity of some synthetic azo dyes. J. Environ. Health Sci. Eng. 2016, 14, 7. [Google Scholar] [CrossRef] [PubMed]
- Shedbalkar, U.; Dhanve, R.; Jadhav, J. Biodegradation of triphenylmethane dye cotton blue by Penicillium ochrochloron MTCC 517. J. Hazard. Mater. 2008, 157, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wang, Q.; Jiang, Z.; Yang, X.-x.; Ji, Y. Enzymatic hydrolysis of pretreated soybean straw. Biomass Bioenergy 2007, 31, 162–167. [Google Scholar] [CrossRef]
- Li, Q.; Chen, C.; Penttinen, P.; Xiong, C.; Zheng, L.; Huang, W. Microbial diversity associated with Tricholoma matsutake fruiting bodies. Microbiology 2016, 85, 531–539. [Google Scholar] [CrossRef]
- Cheng, J.-T.; Wang, H.-M.; Yu, J.-H.; Sun, C.-F.; Cao, F.; Jiang, X.-H.; Chen, X.-A.; Zhao, Q.-W.; Gan, L.-S.; Xie, R.-R. Discovery of a Potential Liver Fibrosis Inhibitor from a Mushroom Endophytic Fungus by Genome Mining of a Silent Biosynthetic Gene Cluster. J. Agric. Food Chem. 2021, 69, 11303–11310. [Google Scholar] [CrossRef]
- Khaled, J.M.; Alyahya, S.A.; Govindan, R.; Chelliah, C.K.; Maruthupandy, M.; Alharbi, N.S.; Kadaikunnan, S.; Issac, R.; Murugan, S.; Li, W.J. Laccase producing bacteria influenced the high decolorization of textile azo dyes with advanced study. Environ. Res 2022, 207, 112211. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-X.; Zhang, R.-J.; Tang, L.; Zhang, J.-H.; Mao, Z.-G. In vivo and in vitro decolorization of synthetic dyes by laccase from solid state fermentation with Trametes sp. SYBC-L4. Bioprocess Biosyst. Eng. 2014, 37, 2597–2605. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Couto, S.; Osma, J.F.; Toca-Herrera, J.L. Removal of synthetic dyes by an eco-friendly strategy. Eng. Life Sci. 2009, 9, 116–123. [Google Scholar] [CrossRef]
- Asgher, M.; Noreen, S.; Bilal, M. Enhancing catalytic functionality of Trametes versicolor IBL-04 laccase by immobilization on chitosan microspheres. Chem. Eng. Res. Des. 2017, 119, 1–11. [Google Scholar] [CrossRef]
- Li, X.; Jia, R. Decolorization and biosorption for Congo red by system rice hull- Schizophyllum sp. F17 under solid-state condition in a continuous flow packed-bed bioreactor. Bioresour. Technol. 2008, 99, 6885–6892. [Google Scholar] [CrossRef]
- Ozmen, N.; Yesilada, O. Valorization and biodecolorization of dye adsorbed on lignocellulosics using white rot fungi. BioResources 2012, 7, 1656–1665. [Google Scholar] [CrossRef]
- Nigam, P.; Armour, G.; Banat, I.M.; Singh, D.; Marchant, R. Physical removal of textile dyes from effluents and solid-state fermentation of dye-adsorbed agricultural residues. Bioresour. Technol. 2000, 72, 219–226. [Google Scholar] [CrossRef]
- Dil, E.A.; Ghaedi, M.; Asfaram, A.; Mehrabi, F.; Bazrafshan, A.A.; Tayebi, L. Synthesis and application of Ce-doped TiO2 nanoparticles loaded on activated carbon for ultrasound-assisted adsorption of Basic Red 46 dye. Ultrason. Sonochem. 2019, 58, 104702. [Google Scholar]
- Noshadi, I.; Amin, N.; Parnas, R.S. Continuous production of biodiesel from waste cooking oil in a reactive distillation column catalyzed by solid heteropolyacid: Optimization using response surface methodology (RSM). Fuel 2012, 94, 156–164. [Google Scholar] [CrossRef]
- Tejada-Tovar, C.; Villabona-Ortíz, Á.; Gonzalez-Delgado, Á.D. Adsorption of azo-anionic dyes in a solution using modified coconut (Cocos nucifera) mesocarp: Kinetic and equilibrium study. Water 2021, 13, 1382. [Google Scholar] [CrossRef]
- Rodríguez-Arellano, G.; Barajas-Fernández, J.; García-Alamilla, R.; Lagunes-Gálvez, L.M.; Lara-Rivera, A.H.; García-Alamilla, P. Evaluation of cocoa beans shell powder as a bioadsorbent of Congo red dye aqueous solutions. Materials 2021, 14, 2763. [Google Scholar] [CrossRef] [PubMed]
- Zonoozi, M.H.; Moghaddam, M.A.; Arami, M. Coagulation/flocculation of dye-containing solutions using polyaluminium chloride and alum. Water Sci. Technol. 2009, 59, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- Sadri, M.S.; Alavi, M.M.; Arami, M. Decolorization of an acidic dye from synthetic wastewater by sludge of water treatment. Plant 2010, 7, 437–442. [Google Scholar]
- Mishra, A.; Tripathi, B.D.; Rai, A.K. Biosorption of Cr (VI) and Ni (II) onto Hydrilla verticillata dried biomass. Ecol. Eng. 2014, 73, 713–723. [Google Scholar] [CrossRef]
- Li, D.; Xu, X.; Yu, H.; Han, X. Characterization of Pb2+ biosorption by psychrotrophic strain Pseudomonas sp. I3 isolated from permafrost soil of Mohe wetland in Northeast China. J. Environ. Manag. 2017, 196, 8–15. [Google Scholar] [CrossRef]
- Shen, D.; Liu, G.; Zhao, J.; Xue, J.; Guan, S.; Xiao, R. Thermo-chemical conversion of lignin to aromatic compounds: Effect of lignin source and reaction temperature. J. Anal. Appl. Pyrolysis 2015, 112, 56–65. [Google Scholar] [CrossRef]
- Boakye, P.; Tran, H.N.; Lee, D.S.; Woo, S.H. Effect of water washing pretreatment on property and adsorption capacity of macroalgae-derived biochar. J. Environ. Manag. 2019, 233, 165–174. [Google Scholar] [CrossRef]
- Plaza, C.; Nogales, R.; Senesi, N.; Benitez, E.; Polo, A. Organic matter humification by vermicomposting of cattle manure alone and mixed with two-phase olive pomace. Bioresour. Technol. 2008, 99, 5085–5089. [Google Scholar] [CrossRef]
- Liu, X.; Bai, X.; Dong, L.; Liang, J.; Jin, Y.; Wei, Y.; Li, Y.; Huang, S.; Qu, J. Composting enhances the removal of lead ions in aqueous solution by spent mushroom substrate: Biosorption and precipitation. J. Clean. Prod. 2018, 200, 1–11. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, Y.; Shaaban, M.; Zhu, D.; Hu, C.; Chen, Z.; Wang, Y. Evaluation of microbial inoculants pretreatment in straw and manure co-composting process enhancement. J. Clean. Prod. 2019, 239, 118078. [Google Scholar] [CrossRef]
- Stefke, B.; Windeisen, E.; Schwanninger, M.; Hinterstoisser, B. Determination of the weight percentage gain and of the acetyl group content of acetylated wood by means of different infrared spectroscopic methods. Anal. Chem. 2008, 80, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Teng, C.; Yu, S.; Song, T.; Dong, L.; Liang, J.; Bai, X.; Liu, X.; Hu, X.; Qu, J. Batch and fixed-bed biosorption of Cd(II) from aqueous solution using immobilized Pleurotus ostreatus spent substrate. Chemosphere 2018, 191, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Drumm, F.C.; Franco, D.S.P.; Georgin, J.; Grassi, P.; Jahn, S.L.; Dotto, G.L. Macro-fungal (Agaricus bisporus) wastes as an adsorbent in the removal of the acid red 97 and crystal violet dyes from ideal colored effluents. Environ. Sci. Pollut. Res. Int. 2021, 28, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, D.; Sheng, F.; Qing, H. Adsorption characteristics of Copper (II), Zinc (II) and Mercury (II) by four kinds of immobilized fungi residues. Ecotoxicol. Environ. Saf. 2018, 147, 357–366. [Google Scholar] [CrossRef]
- Georgin, J.; Franco, D.S.; Grassi, P.; Tonato, D.; Piccilli, D.G.; Meili, L.; Dotto, G.L. Potential of Cedrella fissilis bark as an adsorbent for the removal of red 97 dye from aqueous effluents. Environ. Sci. Pollut. Res. 2019, 26, 19207–19219. [Google Scholar] [CrossRef]
- Zazycki, M.A.; Godinho, M.; Perondi, D.; Foletto, E.L.; Collazzo, G.C.; Dotto, G.L. New biochar from pecan nutshells as an alternative adsorbent for removing reactive red 141 from aqueous solutions. J. Clean. Prod. 2018, 171, 57–65. [Google Scholar] [CrossRef]
- Behl, K.; Sinha, S.; Sharma, M.; Singh, R.; Joshi, M.; Bhatnagar, A.; Nigam, S. One-time cultivation of Chlorella pyrenoidosa in aqueous dye solution supplemented with biochar for microalgal growth, dye decolorization and lipid production. Chem. Eng. J. 2019, 364, 552–561. [Google Scholar] [CrossRef]
- Li, N.; Wang, S.; Wang, T.; Liu, R.; Zhi, Z.; Wu, T.; Sui, W.; Zhang, M. Valorization of Wheat Bran by Three Fungi Solid-State Fermentation: Physicochemical Properties, Antioxidant Activity and Flavor Characteristics. Foods 2022, 11, 1722. [Google Scholar] [CrossRef]

| Variable | Range and Level | |||
|---|---|---|---|---|
| Name | Unit | −1 | 0 | 1 |
| Wheat bran fermentation duration (X1) | day | 2 | 4 | 6 |
| pH of the dye solution (X2) | − | 4.5 | 6.5 | 8.5 |
| Dye concentration (X3) | mg L−1 | 50 | 150 | 250 |
| Run | Variables | Response 1 Dye Removal Percentage after 12 h (%) | Response 2 Dye Removal Percentage after 24 h (%) | ||||
|---|---|---|---|---|---|---|---|
| Fermentation Duration (X1) | Solution pH (X2) | Dye Concentration (X3) | Experimental | Predicted | Experimental | Predicted | |
| 1 | 6 | 6.5 | 50 | 89.55 | 89.54 | 85.17 | 85.43 |
| 2 | 4 | 6.5 | 150 | 87.51 | 87.83 | 89.28 | 89.87 |
| 3 | 4 | 4.5 | 50 | 87.56 | 87.93 | 85.07 | 85.08 |
| 4 | 4 | 6.5 | 150 | 87.91 | 87.83 | 90.25 | 89.87 |
| 5 | 2 | 8.5 | 150 | 82.21 | 82.57 | 86.71 | 86.99 |
| 6 | 2 | 6.5 | 250 | 75.35 | 75.35 | 83.66 | 83.40 |
| 7 | 4 | 8.5 | 50 | 88.38 | 88.57 | 84.51 | 84.73 |
| 8 | 6 | 4.5 | 150 | 89.41 | 89.06 | 90.83 | 90.56 |
| 9 | 2 | 4.5 | 150 | 80.81 | 81.00 | 85.35 | 85.84 |
| 10 | 2 | 6.5 | 50 | 85.39 | 84.84 | 84.27 | 83.76 |
| 11 | 4 | 6.5 | 150 | 88.08 | 87.83 | 90.09 | 89.87 |
| 12 | 6 | 8.5 | 150 | 90.26 | 90.07 | 93.08 | 92.58 |
| 13 | 4 | 8.5 | 250 | 83.18 | 82.81 | 89.80 | 89.79 |
| 14 | 4 | 4.5 | 250 | 81.07 | 80.88 | 86.50 | 86.27 |
| 15 | 6 | 6.5 | 250 | 85.66 | 86.21 | 91.53 | 92.04 |
| Source | Sums of Squares | df | Mean Square | F-Value | p-Value | Remarks |
|---|---|---|---|---|---|---|
| Model | 242.83 | 9 | 26.98 | 92.75 | <0.0001 | Significant |
| X1 | 121.05 | 1 | 121.05 | 416.13 | <0.0001 | Significant |
| X2 | 3.32 | 1 | 3.32 | 11.43 | 0.0197 | Significant |
| X3 | 82.07 | 1 | 82.07 | 282.12 | <0.0001 | Significant |
| X1X2 | 0.0753 | 1 | 0.0753 | 0.2588 | 0.6326 | Not significant |
| X1X3 | 9.46 | 1 | 9.46 | 32.52 | 0.0023 | Significant |
| X2X3 | 0.4139 | 1 | 0.4139 | 1.42 | 0.2865 | Not significant |
| X12 | 9.59 | 1 | 9.59 | 32.97 | 0.0022 | Significant |
| X22 | 1.11 | 1 | 1.11 | 3.82 | 0.1082 | Not significant |
| X32 | 18.46 | 1 | 18.46 | 63.47 | 0.0005 | Significant |
| Residual | 1.45 | 5 | 0.2909 | |||
| Lack of Fit | 1.29 | 3 | 0.4286 | 5.08 | 0.1689 | Not significant |
| Pure Error | 0.1688 | 2 | 0.0844 | |||
| Cor Total | 244.29 | 14 | ||||
| Std. Dev. | 0.5394 | R2 = | 0.9940 | |||
| Mean | 85.49 | Adj. R2 = | 0.9833 | |||
| C.V. % | 0.6309 | Pred. R2 = | 0.9142 | |||
| PRESS | 20.95 | Adeq. Precision | 33.4159 | |||
| Source | Sums of Squares | df | Mean Square | F-Value | p-Value | Remarks |
|---|---|---|---|---|---|---|
| Model | 130.11 | 9 | 14.46 | 37.53 | 0.0005 | Significant |
| X1 | 53.10 | 1 | 53.10 | 137.83 | <0.0001 | Significant |
| X2 | 5.02 | 1 | 5.02 | 13.04 | 0.0154 | Significant |
| X3 | 19.48 | 1 | 19.48 | 50.56 | 0.0009 | Significant |
| X1X2 | 0.1932 | 1 | 0.1932 | 0.5016 | 0.5104 | Not significant |
| X1X3 | 12.14 | 1 | 12.14 | 31.53 | 0.0025 | Significant |
| X2X3 | 3.74 | 1 | 3.74 | 9.72 | 0.0263 | Significant |
| X12 | 1.31 | 1 | 1.31 | 3.41 | 0.1240 | Not significant |
| X22 | 0.2984 | 1 | 0.2984 | 0.7746 | 0.4191 | Not significant |
| X32 | 35.96 | 1 | 35.96 | 93.33 | 0.0002 | Significant |
| Residual | 1.93 | 5 | 0.3852 | |||
| Lack of Fit | 1.39 | 3 | 0.4632 | 1.73 | 0.3873 | Not significant |
| Pure Error | 0.5367 | 2 | 0.2683 | |||
| Cor Total | 132.04 | 14 | ||||
| Std. Dev. | 0.5394 | R2 = | 0.9940 | |||
| Mean | 85.49 | Adj. R2 = | 0.9833 | |||
| C.V. % | 0.6309 | Pred. R2 = | 0.9142 | |||
| PRESS | 20.95 | Adeq. Precision | 33.4159 | |||
| Analysis | Predicted Mean | Predicted Median | Std Dev | SE Pred | 95% PI Low | Data Mean | 95% PI High |
|---|---|---|---|---|---|---|---|
| 12 h | 89.877 | 89.877 | 0.539 | 0.689 | 88.106 | 89.485 | 91.648 |
| 24 h | 92.882 | 92.882 | 0.594 | 0.713 | 91.138 | 93.402 | 95.114 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borham, A.; Okla, M.K.; El-Tayeb, M.A.; Gharib, A.; Hafiz, H.; Liu, L.; Zhao, C.; Xie, R.; He, N.; Zhang, S.; et al. Decolorization of Textile Azo Dye via Solid-State Fermented Wheat Bran by Lasiodiplodia sp. YZH1. J. Fungi 2023, 9, 1069. https://doi.org/10.3390/jof9111069
Borham A, Okla MK, El-Tayeb MA, Gharib A, Hafiz H, Liu L, Zhao C, Xie R, He N, Zhang S, et al. Decolorization of Textile Azo Dye via Solid-State Fermented Wheat Bran by Lasiodiplodia sp. YZH1. Journal of Fungi. 2023; 9(11):1069. https://doi.org/10.3390/jof9111069
Chicago/Turabian StyleBorham, Ali, Mohammad K. Okla, Mohamed A. El-Tayeb, Ahmed Gharib, Hanan Hafiz, Lei Liu, Chen Zhao, Ruqing Xie, Nannan He, Siwen Zhang, and et al. 2023. "Decolorization of Textile Azo Dye via Solid-State Fermented Wheat Bran by Lasiodiplodia sp. YZH1" Journal of Fungi 9, no. 11: 1069. https://doi.org/10.3390/jof9111069
APA StyleBorham, A., Okla, M. K., El-Tayeb, M. A., Gharib, A., Hafiz, H., Liu, L., Zhao, C., Xie, R., He, N., Zhang, S., Wang, J., & Qian, X. (2023). Decolorization of Textile Azo Dye via Solid-State Fermented Wheat Bran by Lasiodiplodia sp. YZH1. Journal of Fungi, 9(11), 1069. https://doi.org/10.3390/jof9111069






