Abstract
Strobilomyces, one of the most noticeable genera of Boletaceae (Boletales), is both ecologically and economically important. Although many studies have focused on Strobilomyces in China, the diversity still remains incompletely understood. In the present study, several collections of Strobilomyces from Hainan Island, tropical China were studied based on morphology and molecular phylogenetic analyses. Four species are described as new, viz. S. baozhengii, S. conicus, S. hainanensis, and S. pachycystidiatus. Detailed descriptions, color photos of fresh basidiomata, and line drawings of microstructures of the four species are presented.
1. Introduction
Strobilomyces Berk., typified by S. strobilaceus (Scop.) Berk., is one of the most noticeable genera of Boletaceae (Boletales), which is characterized by its blackish, blackish-brown, reddish-brown or yellowish-brown pileus covered with scales, subglobose to elliptic basidiospores with reticulate, semi-reticulate, flat-roofed conical or echinate ornamentation, and an obvious reddening or blackening discoloration of tissues when bruised [1,2,3,4,5,6,7]. The genus is divided into two sections: Echinati L.H. Han, Zhu L. Yang & Ndolo Ebika and Strobilomyces Berk. Species of Strobilomyces are diverse; 80 names under the genus were recorded in the Index Fungorum database [http://www.Indexfungorum.org (accessed on 7 November 2023)] [7,8,9]. Interestingly, most species of the genus are reported from tropical and subtropical areas of Asia [1,6,7,8,10,11,12,13,14,15].
Strobilomyces plays an important role in maintaining the biodiversity of forest ecosystems, for species of the genus are usually symbiotic with many plants including Dipterocarpaceae, Fabaceae, Fagaceae, Myrtaceae, and Pinaceae [3,5,6,7]. Besides ecological value, the edible and medicinal values of the genus were also noted, as for example, S. confusus Singer, S. glabriceps W.F. Chiu, and S. strobilaceus (Scop.) Berk. were put on the Chinese edible mushroom list; S. strobilaceus was believed to have anticancer activity [16,17].
In China, species of Strobilomyces are also abundant. Currently, approximately 32 species of the genus have been uncovered [3,5,6,7,13,18,19,20,21,22]. Among them, most are from temperate and subtropical China, while a few species are from tropical regions of China. It is well known, tropical China especially Hainan Island is a biodiversity hotspot [23,24]. With more field investigations in the region, more species of the genus are expected to be revealed. During field investigations of Hainan Island, several collections of Strobilomyces were compiled; further morphology and molecular phylogenetic analyses confirm that these collections represent four novel species. They are described in an effort to further demonstrate the species diversity of Boletaceae in tropical China.
2. Materials and Methods
2.1. Morphological Studies
The fresh basidiomata were described and photographed in the field in daylight, then dried at 60 °C for 12 h. Dried specimens were deposited in the Fungal Herbarium of Hainan Medical University, Haikou City, Hainan Province of China (FHMU). Color codes comply with Kornerup and Wanscher [25]. The pileipellis sections were cut between the center and the margin of the pileus and the stipitipellis sections were taken from the middle part along the longitudinal axis of the stipe [26]. Samples were rehydrated using 5% KOH. All microscopic structures were drawn by freehand from rehydrated material. For basidiospores, the notation ‘n/m/p’ indicates ‘n’ basidiospores from ‘m’ basidiomata of ‘p’ collections. Dimensions of basidiospores are expressed as (a–)b–c(–d), where the range b–c represents a minimum of 90% of the measured values (5th to 95th percentile), and extreme values (a and d), whenever present (a < 5th percentile, d > 95th percentile), are expressed in parentheses. Q indicates the length/width ratio of basidiospores; Qm indicates the average Q of basidiospores and is given with standard deviation. The basidiospores were examined by Zeiss Sigma 300 scanning electron microscopy (SEM) using a small piece of hymenophore (2–5 mm diameter) from one dried specimen, which was sprayed with gold by ion sputtering.
2.2. Molecular Procedures
Plant Genomic DNA Kit (CWBIO, Beijing, China) was used to extract total genomic DNA from materials dried with silica gel according to the manufacturer’s instructions. Primer pairs used for amplification were as follows: LR0R/LR5 [27,28] for nuc 28S rDNA D1–D2 domains (28S), ITS5/ITS4 [29] for nuc rDNA region encompassing the internal transcribed spacers 1 and 2 along with the 5.8S rDNA, the translation elongation factor 1-α gene (tef1) with EF1-F/EF1-R [30], and polymerase II second largest subunit gene (rpb2) with RPB2-B-F1/RPB2-B-R [19]. The PCR thermal cycling programs for 28S, ITS, tef1, and rpb2 amplification were conducted as follows: 95 °C for 4 min, 35 cycles of 94 °C for 60 s, annealing at an appropriate temperature (50 °C for 28S and ITS, 53 °C for tef1, and 52 °C for rpb2) for 30 s, 72 °C for 4 min, and 72 °C for 7 min. The PCR products were purified (TIANGEN, Beijing, China) and then sequenced using a BigDye Terminator v3.1 Kit and an ABI 3730xl DNA Analyzer (Guangzhou Branch of BGI, China). Forward and reverse sequences were compiled with BioEdit v7.0.9.1 [31].
2.3. Dataset Assembly
A total of forty-five sequences (thirteen of 28S, thirteen of ITS, thirteen of tef1, and six of rpb2) from fifteen specimens were newly generated and edited sequences were deposited in GenBank, and accession numbers of 28S, ITS, tef1, and rpb2 are provided in Table 1. The sequences were aligned with selected sequences from previous studies and GenBank (Table 1). Anthracoporus holophaeus (Corner) Yan C. Li & Zhu L. Yang (HKAS50508 and HKAS59407) was chosen as outgroup following Wu et al. [20]. Sequences of 28S, ITS, tef1, and rpb2 were aligned, using MUSCLE (Edgar 2004) separately to test for phylogenetic conflict. There were no conflicts in the topologies of these trees, indicating that phylogenetic signals from different gene fragments were congruent. The sequences of the different genes were concatenated using Phyutility v. 2.2 for further analyses [32].

Table 1.
Taxa, vouchers, locations, and GenBank accession numbers of DNA sequences used in phylogenetic analyses. The new sequences are in bold.
2.4. Phylogenetic Analyses
Maximum likelihood (ML) and Bayesian inference (BI) were used to reconstruct phylogenetic trees based on the combined nuclear dataset (28S + ITS + tef1 + rpb2). ML analysis was conducted with the program RAxML 7.2.6 running 1000 replicates combined with an ML search. Bayesian analysis was conducted with MrBayes 3.1 using the Markov Chain Monte Carlo technique and parameters predetermined with MrModeltest 2.3 [41]. The best-fit likelihood models for the combined dataset were GTR + I + G, GTR + G, SYM + I + G, and K80 + I + G for 28S, ITS, tef1, and rpb2, respectively. Bayesian analysis of the combined nuclear dataset (28S + ITS + tef1 + rpb2) was repeated for 30 million generations. Trees sampled from the first 25% of the generations were discarded as burn-in. The average SD of split frequencies was constrained to be below 0.01, and Bayesian posterior probabilities (PP) were then calculated for a majority consensus tree of the retained Bayesian trees.
3. Results
3.1. Molecular Data
The combined dataset (28S + ITS + tef1 + rpb2) included 140 taxa with 2679 nucleotide sites, and the alignment was deposited in TreeBASE (S30585) [https://treebase.org/treebase-web/home.html (accessed on 22 July 2023)]. Bayesian analyses resulted in identical topologies to the ML analysis, while statistical supports showed slight differences (Figure 1). The present molecular data indicated that the Chinese species of Strobilomyces were grouped into thirty-six independent lineages (Figure 1). A total of four new lineages were identified in this study (Lineages 3, 6, 23, and 25 of Figure 1). Lineage 3 comprised one collection (FHMU2171) from southern China; lineage 6, with strong statistical support (BS = 100%, PP = 1.0), included two new collections (FHMU1937 and FHMU3129) from southern China; lineage 23, with strong statistical support (BS = 100%, PP = 1.0), comprised nine collections (FHMU1493, FHMU1900, FHMU1907, FHMU1909, FHMU1948, FHMU4666, FHMU4692, FHMU4706 and FHMU4751) from southern China; lineage 25 comprised one collection (FHMU2077) also from southern China (Figure 1).
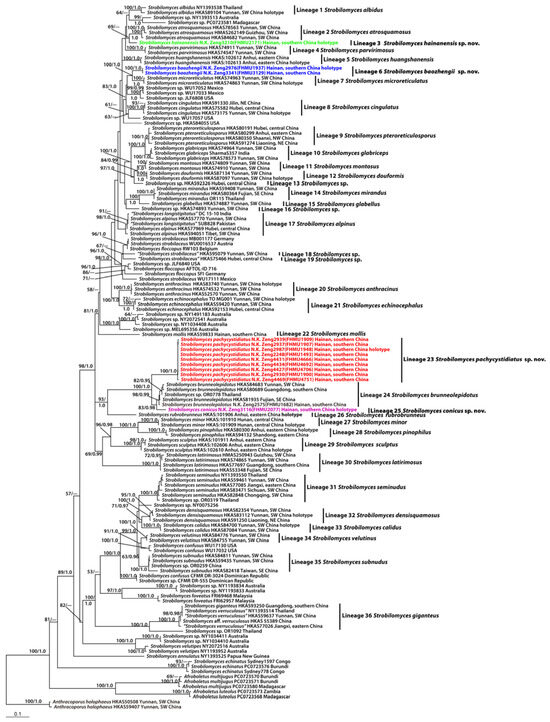
Figure 1.
Phylogram inferred from a combined dataset (28S, ITS, tef1, and rpb2) using RAxML. RAxML likelihood bootstrap (BS ≥ 50%) and Bayesian posterior probabilities (PP ≥ 0.95) are indicated above or below the branches as RAxML BS/PP.
3.2. Taxonomy
- Strobilomyces baozhengii N.K. Zeng, Hui Deng, Yi Wang & Zhi Q. Liang, sp. nov. Figure 2a,b, Figure 3a,b and Figure 4.
 Figure 2. Basidiomata of Strobilomyces species. (a,b) S. baozhengii (FHMU1937, holotype); (c,d) S. conicus (FHMU2077, holotype); (e,f) S. hainanensis (FHMU2171, holotype); (g,h) S. pachycystidiatus (FHMU1948, holotype). Bars = 1 μm. Photos: N.K. Zeng.
Figure 2. Basidiomata of Strobilomyces species. (a,b) S. baozhengii (FHMU1937, holotype); (c,d) S. conicus (FHMU2077, holotype); (e,f) S. hainanensis (FHMU2171, holotype); (g,h) S. pachycystidiatus (FHMU1948, holotype). Bars = 1 μm. Photos: N.K. Zeng. Figure 3. Scanning electron microscopy (SEM) photographs of basidiospores from Strobilomyces species. (a,b) S. baozhengii (FHMU1937, holotype); (c,d) S. conicus (FHMU2077, holotype); (e,f) S. hainanensis (FHMU2171, holotype); (g,h) S. pachycystidiatus (FHMU1948, holotype). Bars = 2 μm. Photos: H. Deng.
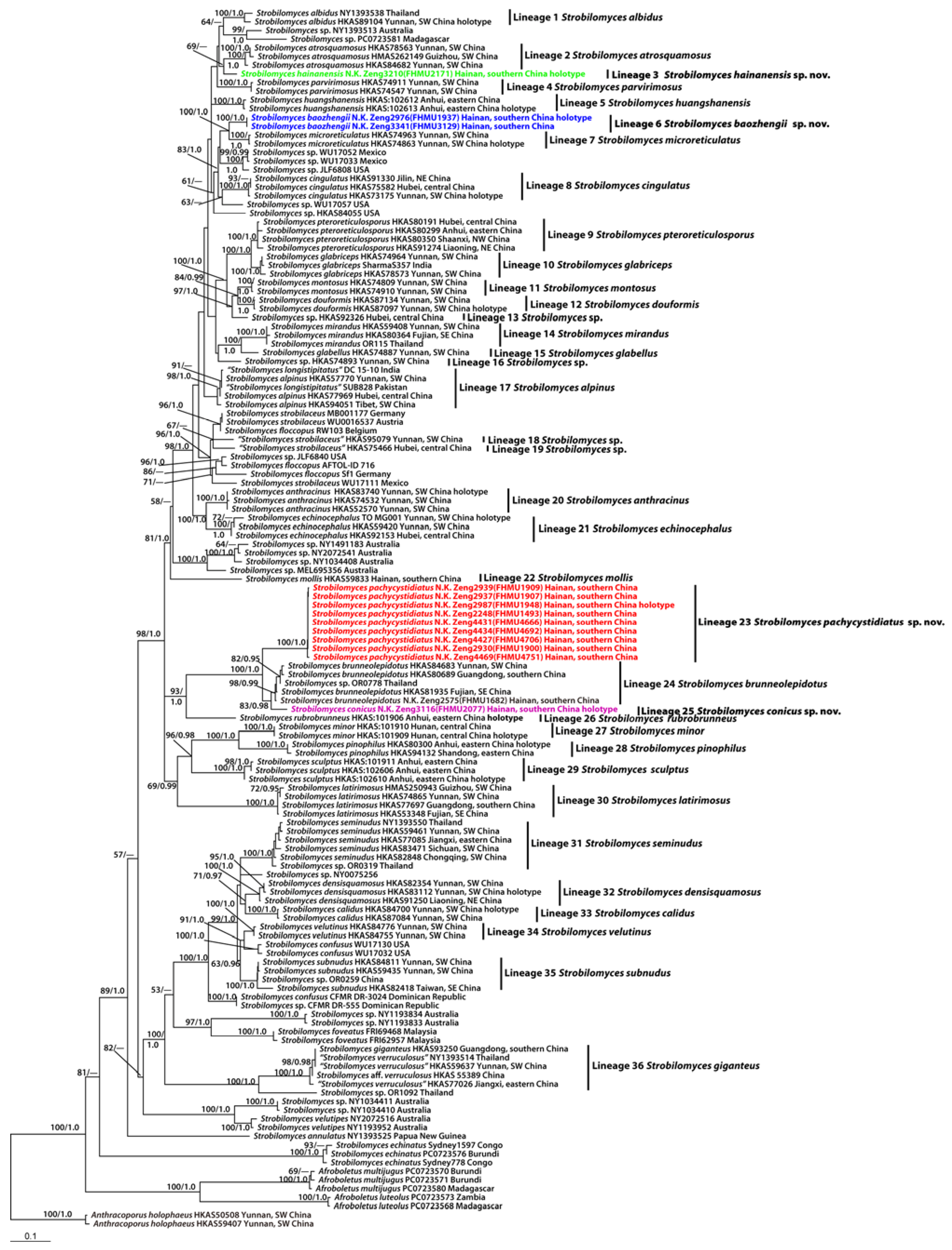
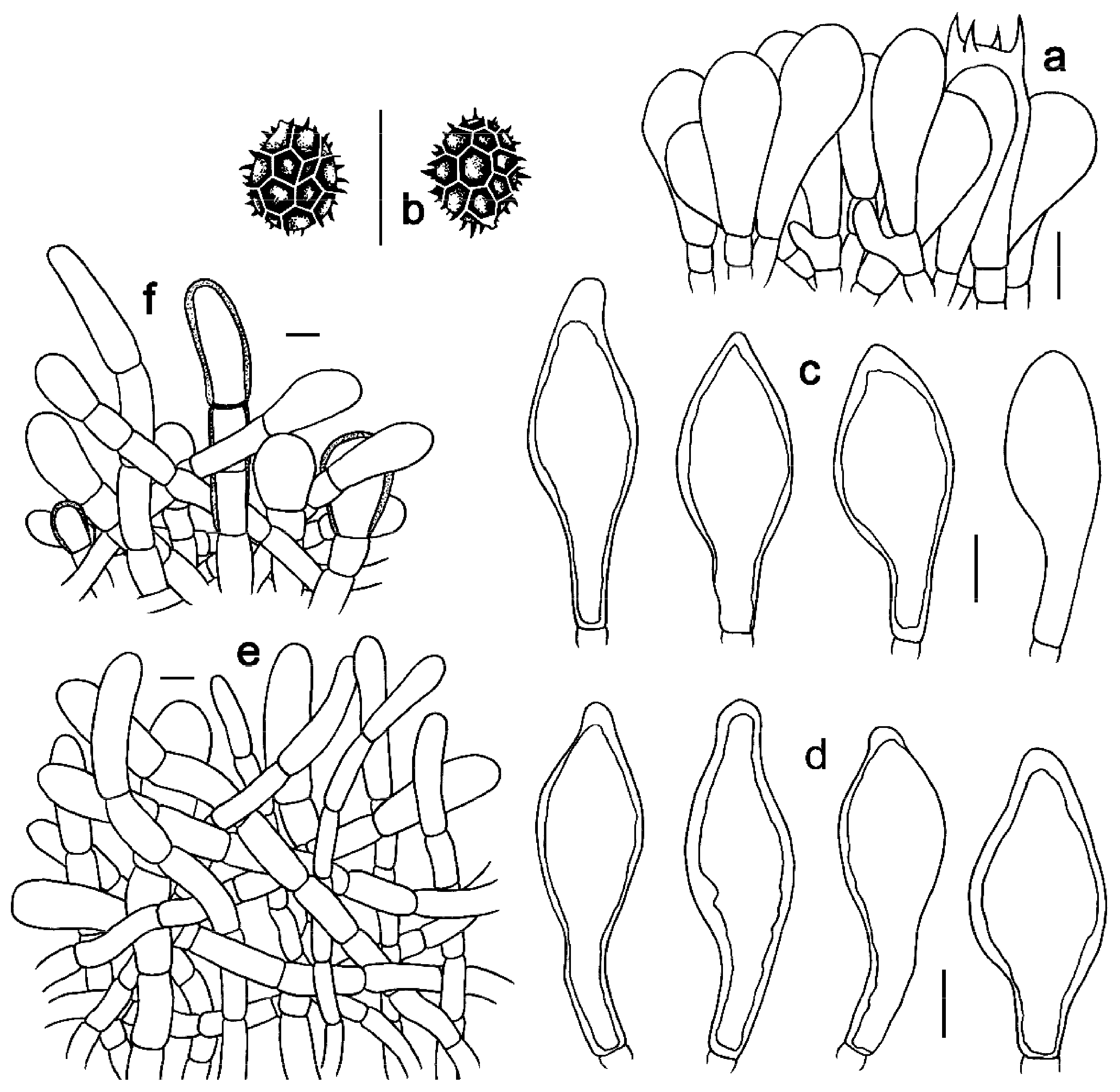
Figure 3. Scanning electron microscopy (SEM) photographs of basidiospores from Strobilomyces species. (a,b) S. baozhengii (FHMU1937, holotype); (c,d) S. conicus (FHMU2077, holotype); (e,f) S. hainanensis (FHMU2171, holotype); (g,h) S. pachycystidiatus (FHMU1948, holotype). Bars = 2 μm. Photos: H. Deng.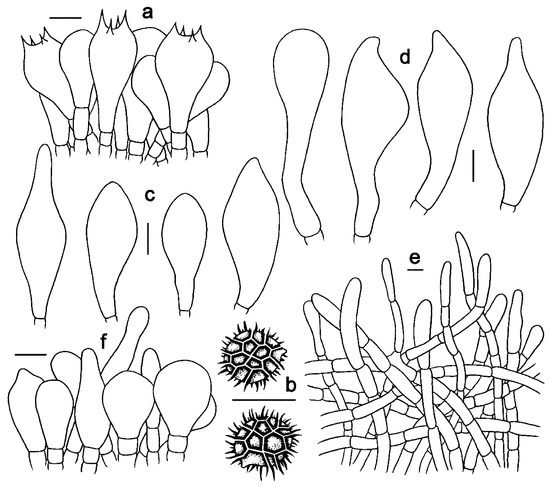 Figure 4. Microscopic features of Strobilomyces baozhengii (FHMU1937, holotype). (a) Basidia. (b) Basidiospores. (c) Cheilocystidia. (d) Pleurocystidia. (e) Pileipellis. (f) Stipitipellis. Bars = 10 μm. Drawings by Y. Wang.
Figure 4. Microscopic features of Strobilomyces baozhengii (FHMU1937, holotype). (a) Basidia. (b) Basidiospores. (c) Cheilocystidia. (d) Pleurocystidia. (e) Pileipellis. (f) Stipitipellis. Bars = 10 μm. Drawings by Y. Wang.
MycoBank no: MB 850251
Etymology: “baozhengii” is named for Bao Zheng, a historic Chinese righteous officer, said to have a dark face.
Diagnosis: Differs from closest species of Strobilomyces by small basidioma (4.3–6 cm diameter), pileus densely covered with patch-like to appressed, light black to black scales, grey to black stipe covered with patch-like to appressed, blackish-brown scales or reticula, small hymenophoral pores (approximately 0.1 cm diameter), small- to medium-sized reticulate basidiospores (8–10.5 × 7–8.5μm), and its association with fagaceous trees.
Holotype: CHINA. Hainan Province: Yinggeling of Hainan Tropical Rainforest National Park, elev. 550 m, 26 May 2017, N.K. Zeng2976 (FHMU1937).
Description: Pileus 4.3–6 cm diameter, subhemispherical when young, then convex to applanate; surface dirty white, densely covered with light black (11C1) to black (11F1), patch-like to appressed scales; margin usually appendiculate with thick and triangular or irregular lacy veil remnants concolorous with pileal surface; context 0.7–1.5 cm in thickness in the center of the pileus, white (2A1) to greyish-white (7B1), turning red (9E5) slowly then black (9F1) quickly when injured. Hymenophore tubulate, slightly depressed around the apex of the stipe; pores angular, approximately 0.1 cm diameter, tubes 0.6–0.7 cm long, greyish-white (2A1) to greyish-black (7D1), turning reddish (9E5) then black (9F1) when injured. Stipe 4.5–6 × 1–1.5 cm, central, subcylindrical, solid, grey (7B1) to black (7F1); surface covered with patch-like to appressed blackish-brown (7E1) scales or reticula; context greyish-white (11F1), turning reddish (9E5) or reddish-brown (9E7) then black (9F1) when injured; annulus at the apex but not very obvious; basal mycelium greyish-white (7A1). Odor indistinct.
Basidia 24–38 × 12–19.5 μm, subclavate or clavate, thin-walled, sometimes slightly thick-walled (up to 0.5 μm), colorless, pale yellowish-brown to yellowish-brown in KOH; sterigmata 2–7 μm in length. Basidiospores [60/3/2] 11–13.5 × (10–)10.5–12(–14) μm, Q = (0.96–)1.00–1.18(–1.20), Qm = 1.07 ± 0.06 (including ornamentation); [40/2/2] (7–)8–10.5(–11) × 7–8.5 μm, Q = 1.00–1.31 (–1.47), Qm = 1.16 ± 0.10 (excluding ornamentation), spherical to broad ellipsoid, yellowish-brown to darkish-brown in KOH, reticulate, with meshes 1–5 μm high. Hymenophoral trama boletoid, composed of colorless to pale yellow in KOH, 4–15 μm wide, thin- to slightly thick-walled (up to 0.5 μm) hyphae. Cheilocystidia 37–55 × 13–20 μm, fusiform or subfusiform, thin- to slightly thick-walled (up to 1 μm), colorless, pale yellowish-brown to yellowish-brown in KOH. Pleurocystidia 50–65 × 16–21 μm, fusiform, subfusiform or subclavate, thin- to slightly thick-walled (up to 0.5 μm), yellowish-brown in KOH. Pileipellis an intricate trichodermium 150–280 μm thick, composed of 5–14 μm wide, colorless to yellowish-brown in KOH, thin- to slightly thick-walled (up to 1 μm) hyphae; terminal cells 24–57 × 5–10 μm, clavate or subcylindrical, with obtuse apex. Pileal trama composed of hyphae 4–16 μm in diameter, thin- to slightly thick-walled (up to 0.5 μm), yellow in KOH. Stipitipellis a trichoderm-like structure 60–230 μm thick, composed of thin-walled, colorless, pale yellowish-brown to yellowish-brown in KOH, emergent hyphae with clavate or subglobose terminal cells (19–44 × 6–22 μm). Stipe trama composed of parallel hyphae 4–12 μm wide, cylindrical, thin- to slightly thick-walled (up to 0.5 μm), colorless or pale yellowish-brown in KOH. Clamp connections absent in all tissues.
Additional specimen examined: CHINA. Hainan Province: Yinggeling of Hainan Tropical Rainforest National Park, elev. 550 m, 6 May 2018, N.K. Zeng3341 (FHMU3129).
Habitat: Scattered on the ground in forests dominated by fagaceous trees.
Known distribution: Southern China (Hainan Province).
Notes: Strobilomyces baozhengii is phylogenetically related to S. microreticulatus Li H. Han & Zhu L. Yang (Figure 1). However, S. microreticulatus, originally described from Yunnan Province, southwestern China, differs by a stipe with grey-white and grey-black flosses above and below the annulus, and larger basidiospores (9–11 × 7–8 μm) with lower meshes (1 μm high) [7]. Morphologically, S. baozhengii is similar to Chinese S. echinocephalus Gelardi & Vizzini and S. parvirimosus J.Z. Ying, Malaysian S. mollis Corner, and Indian S. nigricans Berk. However, S. echinocephalus has a pileus covered with erect scales and smaller basidiospores measuring 8–11.5 × 6.8–9.9 μm (including ornamentation) [8]; S. parvirimosus has a pileus covered with erect pyramidal scales and basidiospores with lower meshes (up to 1 μm) [7,42]; S. mollis has a blackish-brown to vinaceous pileus with erect conical scales and smaller basidiospores (7.5–9.5 × 6.5–8 μm) [1,7]; S. nigricans has larger basidiospores (9.5–12 × 7.5–9.5 μm) and grows in association with Abies spp. [7,43].
- Strobilomyces conicus N.K. Zeng, Hui Deng, Yi Wang & Zhi Q. Liang, sp. nov. Figure 2c,d, Figure 3c,d and Figure 5.
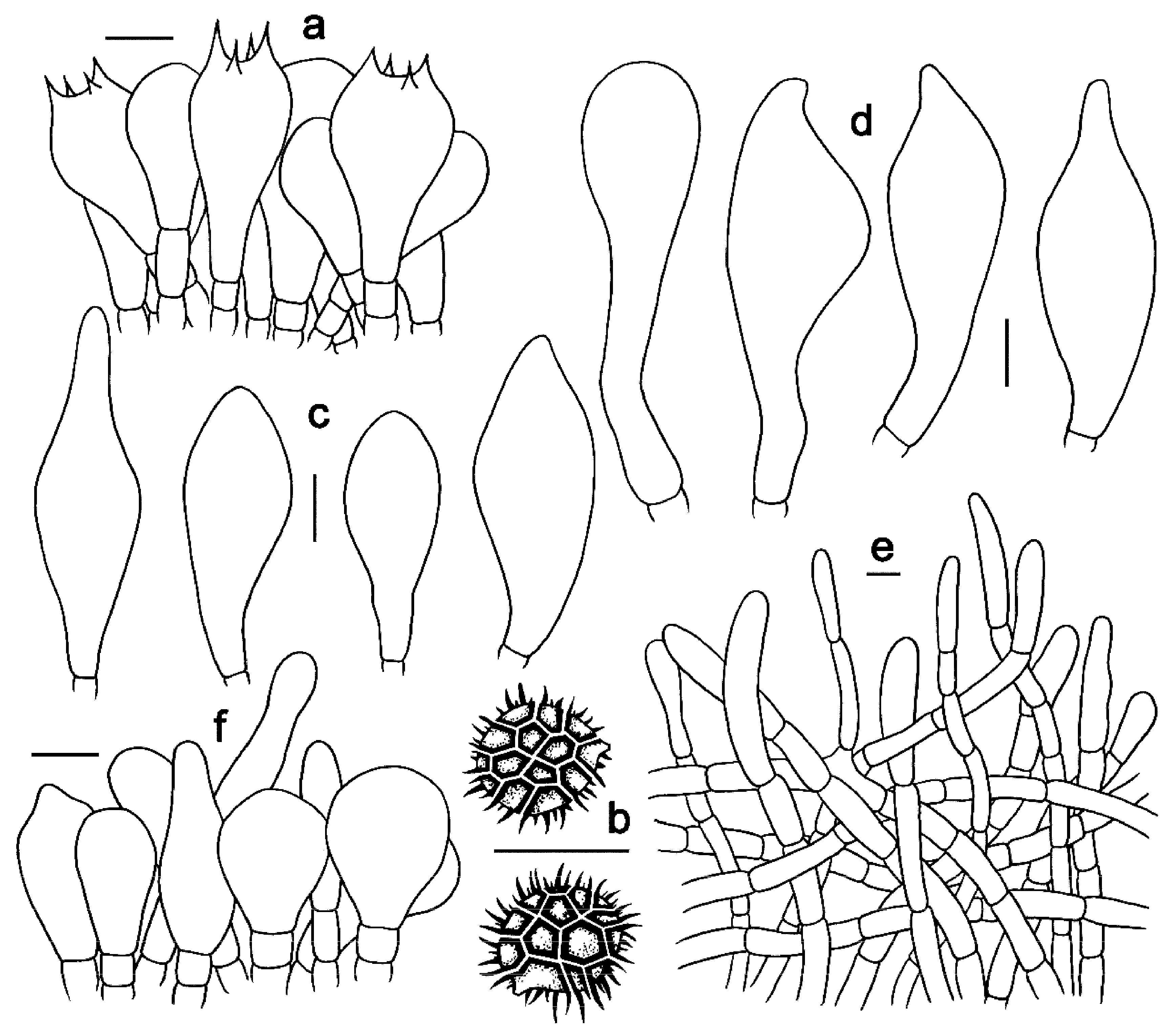
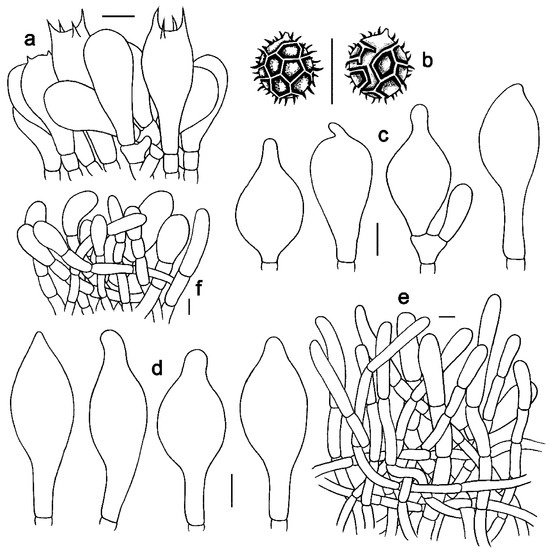 Figure 5. Microscopic features of Strobilomyces conicus (FHMU2077, holotype). (a) Basidia. (b) Basidiospores. (c) Cheilocystidia. (d) Pleurocystidia. (e) Pileipellis. (f) Stipitipellis. Bars = 10 μm. Drawings by Y. Wang.
Figure 5. Microscopic features of Strobilomyces conicus (FHMU2077, holotype). (a) Basidia. (b) Basidiospores. (c) Cheilocystidia. (d) Pleurocystidia. (e) Pileipellis. (f) Stipitipellis. Bars = 10 μm. Drawings by Y. Wang.
MycoBank no: MB 850252
Etymology: “conicus” refers to the conical scales on pileus.
Diagnosis: Differs from closest species of Strobilomyces by medium-sized basidioma (approximately 6.5 cm diameter), pileus densely covered with brownish-black to black, erect conical scales or villi, small hymenophoral pores (0.05–0.1 cm diameter), small incomplete reticulate to reticulate basidiospores (7.5–9 × 7–7.5 μm), and its association with fagaceous trees.
Holotype: CHINA. Hainan Province: Limushan of Hainan Tropical Rainforest National Park, elev. 650 m, 26 July 2017, N.K. Zeng3116 (FHMU2077).
Description: Pileus approximately 6.5 cm diameter, subhemispherical when young, then convex to applanate; surface dirty white, densely covered with brownish-black (7D3) to black (7F1), erect conical scales or villi; margin usually appendiculate with thick and triangular remnants concolorous with pileal surface; context approximately 1 cm in thickness in the center of the pileus, greyish-white (2B2), immediately turning reddish-brown (7D5) then black (7F1) when injured. Hymenophore tubulate, adnate; pores angular, 0.05–0.1 cm diameter, tubes 0.2–0.7 cm long, white (2A1) or greyish-white (2B2), turning reddish-brown (7E4) slowly then black (7F1) quickly when injured. Stipe 6.5 × 0.8–1 cm, central, subcylindrical, solid; surface covered with black (7F1) scales or villi; context greyish-black (5E1), unchanging in color when injured; basal mycelium white (2A1). Odor indistinct.
Basidia 27–43 × 11–20 μm, subclavate or clavate, thin-walled, colorless to pale yellowish-brown in KOH; sterigmata 2–6 μm in length. Basidiospores [20/1/1] (9–)10–11(–12) × (9–)10–11(–11.5) μm, Q = 1.00–1.10, Qm = 1.01 ± 0.03 (including ornamentation); [20/1/1] 7.5–9 × 7–7.5(–8.5) μm, Q = (1.00–)1.06–1.20(–1.21), Qm = 1.11 ± 0.06 (excluding ornamentation), spherical to broad ellipsoid, yellowish-brown to darkish-brown in KOH, incomplete reticulate to reticulate, with meshes 1–3 μm high. Hymenophoral trama boletoid; composed of colorless to yellowish-brown in KOH, 5–16 μm wide, thin-walled hyphae. Cheilocystidia 36–54 × 15–23 μm, ventricose, fusiform or subfusiform, thin- to slightly thick-walled (up to 1 μm), colorless to pale yellowish-brown in KOH. Pleurocystidia 41–61 × 16–25 μm, ventricose, fusiform or subfusiform, thin- to slightly thick-walled (up to 1 μm), colorless to yellowish-brown in KOH. Pileipellis an intricate trichodermium 160–350 μm thick, composed of 5–17 μm wide, yellow in KOH, thin- to slightly thick-walled (up to 1 μm) hyphae; terminal cells 27–70 × 6–15 μm, clavate or subclavate, with obtuse apex. Pileal trama composed of hyphae 3–16 μm in diameter, thin- to slightly thick-walled (up to 0.5 μm), colorless in KOH. Stipitipellis a trichoderm-like structure 80–240 μm thick, composed of thin- to thick-walled (up to 2 μm), yellowish-brown in KOH, emergent hyphae with spherality, pyriform, clavate or subclavate terminal cells (21–47 × 6–20 μm). Stipe trama composed of parallel hyphae 6–28 μm wide, cylindrical, thin- to slightly thick-walled (up to 0.5 μm), and colorless to pale yellowish-brown in KOH. Clamp connections absent in all tissues.
Habitat: Scattered on the ground in forests dominated by fagaceous trees.
Known distribution: Southern China (Hainan Province).
Notes: Phylogenetically, S. conicus is related to S. brunneolepidotus Har. Takah. & Taneyama and S. pachycystidiatus N.K. Zeng, Hui Deng, Yi Wang & Zhi Q. Liang (Figure 1). However, S. brunneolepidotus, originally described from Japan, differs by its reddish-brown scales on pileus [7,44]; S. pachycystidiatus, also described from tropical China, is distinguished by its larger hymenophoral pores (0.1–0.5 cm diameter), thicker cystidia (thick to 2–3 μm), and basidiospores with lower meshes (0.5–2.5 μm high). Morphologically, S. conicus is similar to S. anthracinus Li H. Han, J. Xu & Zhu L. Yang and S. calidus Li H. Han, J. Xu & Zhu L. Yang, S. echinocephalus, S. microreticulatus, S. mollis, S. nigricans, and S. parvirimosus. However, S. anthracinus has a stipe with thin fluffy flosses and completely reticulate basidiospores with lower meshes (1–1.5 μm high) [7]; S. calidus has a larger basidioma, larger hymenophoral pores (0.1–0.2 cm diameter), larger basidiospores with lower meshes (0.5–1 μm high) [7]; S. echinocephalus has a smaller basidioma and smaller basidiospores measuring 8–11.5 × 6.8–9.9 μm (including ornamentation) [8]; S. microreticulatus has a smaller basidioma, a stipe with grey-white and grey-black flosses above and below annulus, and larger basidiospores (9–11 × 7–8 μm) with lower meshes (1 μm high) [7]; S. mollis has larger hymenophoral pores (1–1.5 mm diameter) and completely reticulate basidiospores [1,7]; S. nigricans has larger basidiospores (9.5–12 × 7.5–9.5 μm) and associates with Abies spp. [7,43]; S. parvirimosus has larger basidiospores (8–10 × 6.5–8 μm) with lower meshes (1 μm high) [7,42].
- Strobilomyces hainanensis N.K. Zeng, Hui Deng, Yi Wang & Zhi Q. Liang, sp. nov. Figure 2e,f, Figure 3e,f and Figure 6.
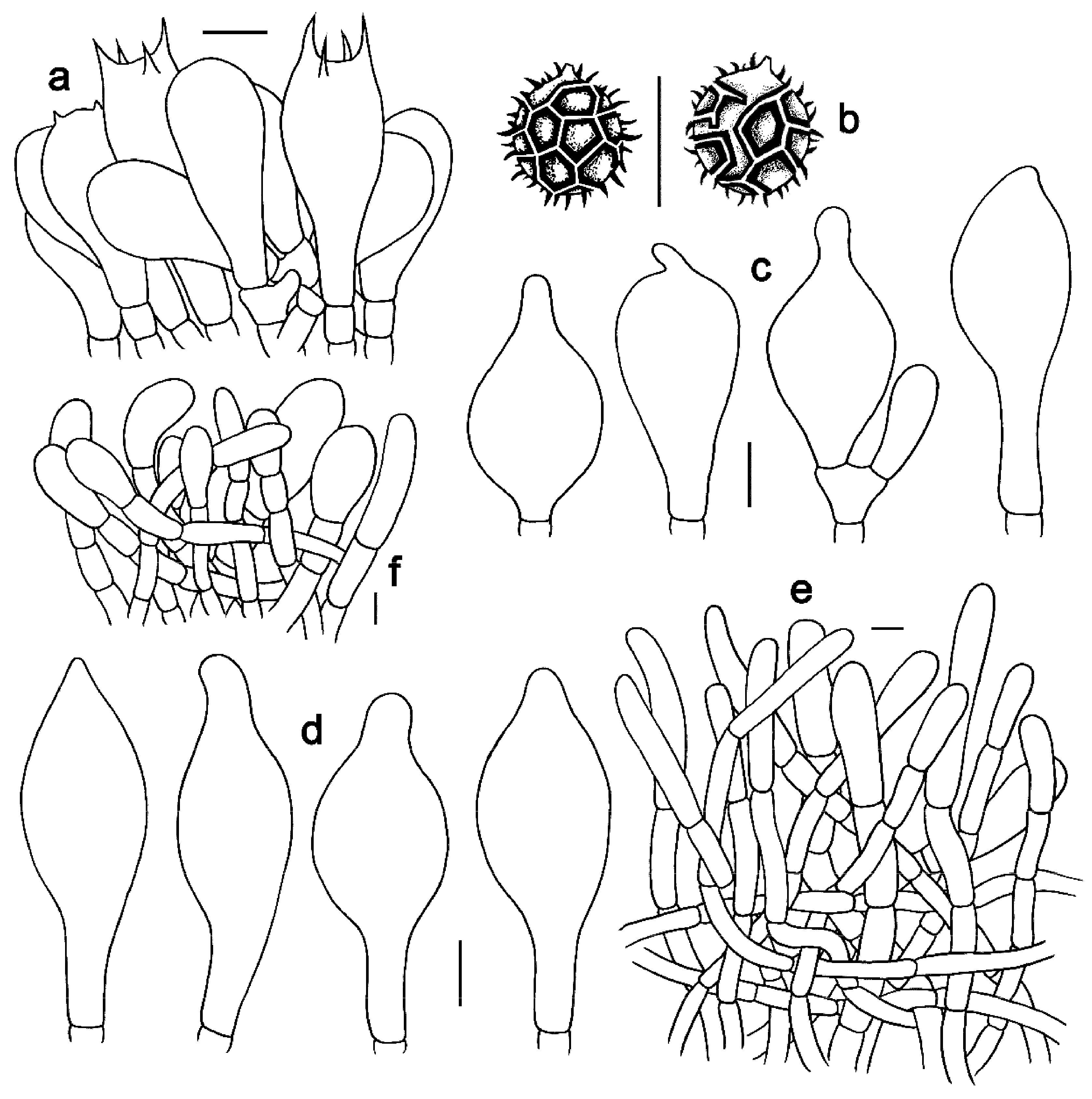
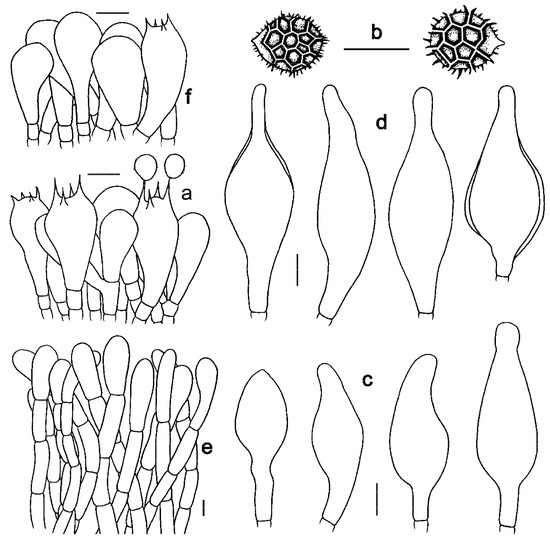 Figure 6. Microscopic features of Strobilomyces hainanensis (FHMU2171, holotype). (a) Basidia. (b) Basidiospores. (c) Cheilocystidia. (d) Pleurocystidia. (e) Pileipellis. (f) Stipitipellis. Bars = 10 μm. Drawings by Y. Wang.
Figure 6. Microscopic features of Strobilomyces hainanensis (FHMU2171, holotype). (a) Basidia. (b) Basidiospores. (c) Cheilocystidia. (d) Pleurocystidia. (e) Pileipellis. (f) Stipitipellis. Bars = 10 μm. Drawings by Y. Wang.
MycoBank no: MB 850253.
Etymology: “hainanensis” refers to Hainan Province, China, holotype locality.
Diagnosis: Differs from closest species of Strobilomyces by smaller basidioma (approximately 4 cm diameter), pileus densely covered with black, erect conical to pyramidal scales, stipe covered with black fluffy flosses, small hymenophoral pores (approximately 0.1 cm diameter), medium-sized reticulate basidiospores (9–11.5 × 8–10 μm), and its association with fagaceous trees.
Holotype: CHINA. Hainan Province: Jianfengling of Hainan Tropical Rainforest National Park, elev. 950 m, 31 July 2017, N.K. Zeng3210 (FHMU2171).
Description: Pileus approximately 4 cm diameter, subhemispherical when young, then convex to applanate; surface dirty white, densely covered with black (11F1), conical to pyramidal scales; appendiculate thick and triangular veil remnants along the margin, dark reddish-brown (11C3); context approximately 0.3 cm in thickness in the center of the pileus, turning reddish (9E5) then black (9F1) when injured. Hymenophore tubulate, slightly depressed around the apex of the stipe; pores angular, approximately 0.1 cm diameter, tubes approximately 0.6 cm long, greyish-white (2A1) to greyish-black (7D1), turning reddish (9E5) then black (9F1) when injured. Stipe 4–5 × 0.5–0.7 cm, central, subcylindrical or slightly enlarged at base, solid; surface covered with black (12F1) fluffy flosses, context greyish-white (11F1), turning reddish (9E5) or reddish-brown (9E7) then black (9F1) when injured; basal mycelium white (1A1). Odor indistinct.
Basidia 23–40 × 11–20 μm, clavate, thin-walled, colorless to yellowish-brown in KOH; sterigmata 4–6 μm in length. Basidiospores [20/1/1] (10.5)11–13(–14) × 10–13 μm, Q = 1.00–1.14(–1.30), Qm = 1.08 ± 0.07 (including ornamentation); [20/1/1] (8.5–)9–11.5(–12) × 8–10 μm, Q = 1.06–1.24 (–1.25), Qm = 1.15 ± 0.06 (excluding ornamentation), spherical to broad ellipsoid, yellowish-brown to darkish-brown in KOH, reticulate, with meshes 1–4 μm high. Hymenophoral trama boletoid; composed of colorless to yellowish-brown in KOH, 3–14 μm wide, thin- to slightly thick-walled (up to 1 μm) hyphae. Cheilocystidia 21–63 × 12–20 μm, abundant, lageniform, ventricose or subfusiform, thin- to slightly thick-walled (up to 1 μm), yellowish-brown in KOH. Pleurocystidia 36.5–68 × 12.5–28 μm, abundant, fusiform or subfusiform, thin- to slightly thick-walled (up to 1 μm), yellowish-brown to darkish-brown in KOH. Pileipellis a trichoderm approximately 300 μm thick, composed of more or less vertically arranged, 7–19 μm wide, pale yellowish-brown to yellowish-brown in KOH, thin- to slightly thick-walled (up to 1 μm) hyphae; terminal cells 25–68 × 10–17 μm, clavate or subcylindrical, with obtuse apex. Pileal trama composed of hyphae 4–31 μm in diameter, thin- to slightly thick-walled (up to 0.5 μm), colorless or pale yellow in KOH. Stipitipellis a trichoderm-like structure 50–100 μm thick, composed of thin- to slightly thick-walled (up to 0.5 μm), yellowish-brown in KOH, emergent hyphae with subclavate or clavate terminal cells (25–35 × 12–18 μm). Stipe trama composed of parallel hyphae 4–12 μm wide, cylindrical, thin- to slightly thick-walled (up to 0.5 μm), and colorless to yellowish-brown in KOH. Clamp connections absent in all tissues.
Habitat: Solitary on the ground in forests dominated by Lithocarpus spp., Leucobryum javense (Brid.) Mitt., and Calypogeia sp.
Known distribution: Southern China (Hainan Province).
Notes: Our new species S. hainanensis formed a separate species-level branch at the phylogenetic tree (Figure 1), indicating that it is in an independent phylogenetic position. Morphologically, S. hainanensis is similar to S. echinocephalus, S. microreticulatus, S. mollis, S. nigricans, and S. parvirimosus. However, S. echinocephalus has a larger basidioma and smaller basidiospores measuring 8–11.5 × 6.8–9.9 μm (including ornamentation) [8]; S. microreticulatus has a pileus with dirty white scales on periphery, a stipe with grey-white flosses above annulus, and narrower basidiospores (9–11 × 7–8 μm) [7]; S. mollis has smaller basidiospores (7.5–9.5 × 6.5–8 μm) [1,7]; S. nigricans is associated with Abies spp. in subalpine areas in northern India [7,43]; S. parvirimosus has smaller basidiospores (8–10 × 6.5–8 μm) with lower meshes (up to 1 μm) [7,42].
- Strobilomyces pachycystidiatus N.K. Zeng, Hui Deng, Yi Wang & Zhi Q. Liang, sp. nov. Figure 2g,h, Figure 3g,h and Figure 7.
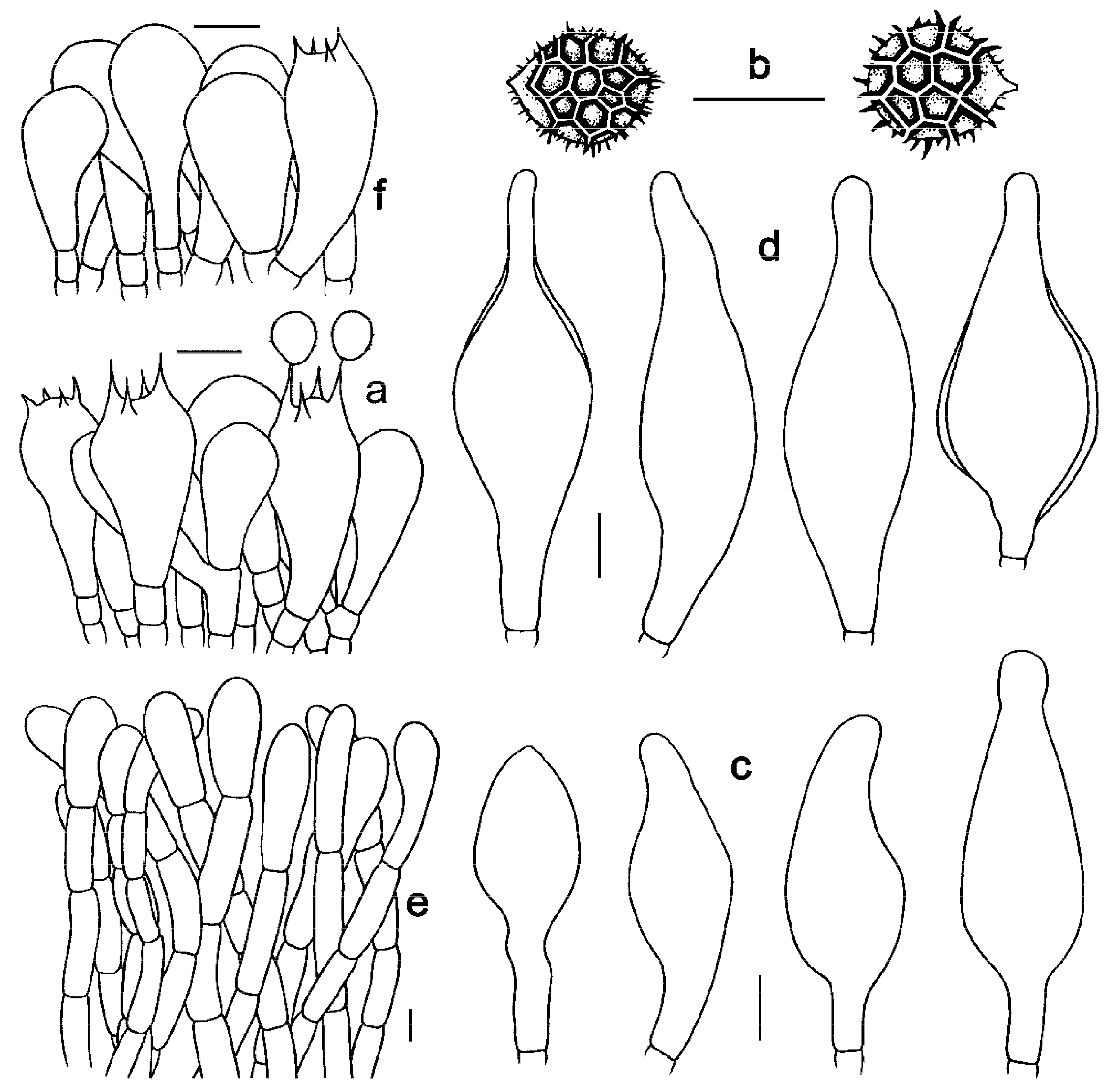
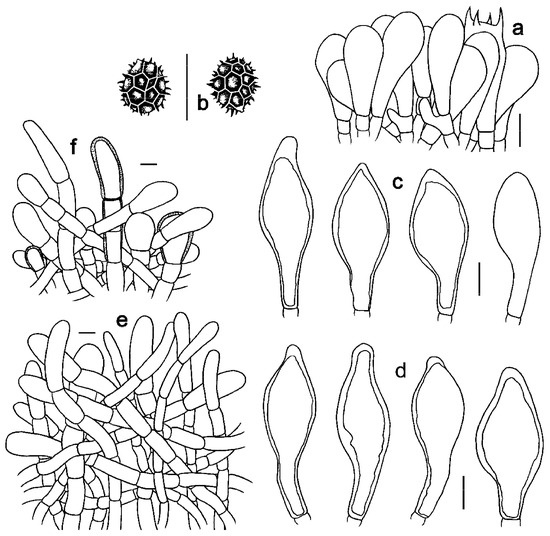 Figure 7. Microscopic features of Strobilomyces pachycystidiatus (FHMU1948, holotype). (a) Basidia. (b) Basidiospores. (c) Cheilocystidia. (d) Pleurocystidia. (e) Pileipellis. (f) Stipitipellis. Bars = 10 μm. Drawings by Y. Wang.
Figure 7. Microscopic features of Strobilomyces pachycystidiatus (FHMU1948, holotype). (a) Basidia. (b) Basidiospores. (c) Cheilocystidia. (d) Pleurocystidia. (e) Pileipellis. (f) Stipitipellis. Bars = 10 μm. Drawings by Y. Wang.
MycoBank no: MB 850254
Etymology: Latin “pachy” meaning thick, “cystidiatus” meaning cystidium, “pachycystidiatus” refers to the thick-walled cystidia.
Diagnosis: Differs from closest species of Strobilomyces by tiny to medium basidioma (2.7–7.5 cm diam), pileus densely covered with blackish-brown to black conical scales, stipe covered with darkish-brown to black scales or fluffy flosses, large hymenophoral pores (0.1–0.5 cm diameter), small- to medium-sized reticulate basidiospores (6.5–11 × 4.5–9 μm), thick-walled (2–3 μm) cystidia, and its association with fagaceous trees.
Holotype: CHINA. Hainan Province: Yinggeling of Hainan Tropical Rainforest National Park, elev. 550 m, 26 July 2017, N.K. Zeng2987 (FHMU1948).
Description: Pileus 2.7–7.5 cm diameter, subhemispherical when young, then convex to applanate; surface dirty white, densely covered with blackish-brown (7F1) to black (11F1), conical scales; margin usually appendiculate with thick and triangular or irregular lacy veil remnants concolorous with pileal surface; context 0.6–1.4 cm in thickness in the center of the pileus, white (2A1) to greyish-white (5A1), turning reddish-brown (5B3) then black (7F1) when injured. Hymenophore tubulate, slightly depressed around the apex of the stipe; pores angular, 0.1–0.5 cm diameter, tubes 0.4–1.1 cm long, white (3A1) or greyish-white (5A1), turning reddish-brown (5B3) then black (7F1) when injured. Stipe 4.6–9 × 0.6–1.4 cm, central, subcylindrical, solid; surface densely covered with darkish-brown (7E1) to black (7F1) scales or fluffy flosses; context white (2A1) to greyish-white (5A1), turning reddish-brown (5B3) then black (7F1) when injured; basal mycelium white (2A1) or greyish-white (7A1). Odor indistinct.
Basidia 19–33 × 10–15 μm, subclavate or clavate, thin- to slightly thick-walled (up to 0.5 μm), colorless to pale yellowish-brown in KOH; sterigmata 2–6 μm in length. Basidiospores [300/15/9] 8.5–11(–12) × (7–)8–11 μm, Q = 1.00–1.13(–1.21), Qm = 1.06 ± 0.05 (including ornamentation); [300/15/9] 7–9 × 6–8(–8.5) μm, Q = 1.00–1.33(–1.42), Qm = 1.18 ± 0.11 (excluding ornamentation), spherical to broad ellipsoid, yellowish-brown to darkish-brown in KOH, reticulate, with meshes 0.5–2.5 μm high. Hymenophoral trama boletoid; composed of colorless to yellowish-brown in KOH, 4–20 μm wide, thin- to slightly thick-walled (up to 0.5 μm) hyphae. Cheilocystidia 30–54 × 13–20 μm, abundant, ventricose, fusiform or subfusiform, thin- to thick-walled 5 (up to 2 μm), colorless to yellowish-brown in KOH. Pleurocystidia 35–57 × 13–20 μm, abundant, ventricose, fusiform or subfusiform, thin- to thick-walled (up to 3 μm), yellowish-brown in KOH. Pileipellis an intricate trichodermium 130–700 μm thick, composed of 5–21 μm wide, pale yellowish-brown to yellowish-brown in KOH, thin-walled hyphae; terminal cells 31–71 × 7–19 μm, pyriform, clavate or subclavate, with obtuse apex. Pileal trama composed of hyphae 5–17 μm in diameter, thin- to slightly thick-walled (up to 0.5 μm), colorless to yellowish-brown in KOH. Stipitipellis a trichoderm-like structure 50–400 μm thick, composed of thin- to thick-walled (up to 2 μm), yellowish-brown in KOH, emergent hyphae with spherality, clavate or subclavate terminal cells (15–52 × 10–30 μm). Stipe trama composed of longitudinally arranged, parallel hyphae 3–15 μm wide, cylindrical, thin- to slightly thick-walled (up to 0.5 μm), colorless, pale yellowish-brown to darkish-brown in KOH. Clamp connections absent in all tissues.
Additional specimens examined: CHINA. Hainan Province: Yinggeling of Hainan Tropical Rainforest National Park, elev. 550 m, 30 July 2015, N.K. Zeng2248 (FHMU1493); same location, 9 September 2016, N.K. Zeng2930 (FHMU1900); same location, 10 September 2016, N.K. Zeng2937, 2939 (FHMU1907, 1909); same location, 3 July 2020, N.K. Zeng4427, 4431, 4434, 4469 (FHMU4706, 4666, 4692, 4683, 4751).
Habitat: Solitary or scattered on the ground in forests dominated by fagaceous trees.
Known distribution: Southern China (Hainan Province).
Notes: Strobilomyces pachycystidiatus is phylogenetically closely related to S. brunneolepidotus. However, S. brunneolepidotus has a basidioma with reddish-brown scales and thin-walled cystidia [7,44]. Morphologically, S. pachycystidiatus is similar to S. echinocephalus, S. microreticulatus, S. mollis, S. nigricans, and S. parvirimosus. However, S. echinocephalus has a stipe densely covered by cottony to woolly floccules, larger basidiospores measuring 8–11.5 × 6.8–9.9 μm (including ornamentation) and smaller hymenophoral pores (approximately 0.1 cm diameter) [8]; S. microreticulatus has a smaller basidioma, a stipe with grey-white flosses above annulus, larger basidiospores (9–11 × 7–8 μm), and smaller hymenophoral pores (0.05–0.1cm diameter) [7]; S. mollis has a stipe with blackish-brown to vinaceous, thin fluffy flosses, basidiospores with higher meshes (2–3.5 μm high), and smaller hymenophoral pores (0.1–0.15 cm diameter) [1,7]; S. nigricans has larger basidiospores (9.5–12 × 7.5–9.5 μm), its association with Abies spp., and its distribution in subalpine areas in northern India [7,43]; S. parvirimosus has smaller basidiospores (8–10 × 6.5–8 μm) with lower meshes (1 μm high), and smaller hymenophoral pores (0.05–0.1 diameter) [7,42].
4. Discussion
Although the diagnosis of Strobilomyces is relatively easy, species within the genus are difficult to identify because of an absence of molecular data as well as morphological convergence documented in this group in previous studies [11,12]. With the development of molecular phylogenetic analyses, many previously described taxa have been re-evaluated, which enhanced our understanding of Strobilomyces diversity worldwide [7,8,14]. For example, S. confusus and S. strobilaceus, two taxa originally described from North America and Europe, respectively [45], were previously thought to be widely distributed species. However, recent studies have indicated that S. confusus and S. strobilaceus actually represent species complexes [7,8,14]. Our molecular data also showed that specimens identified as S. confusus or S. strobilaceus were present in different parts of the tree (Figure 1).
High species diversity of Strobilomyces in China was revealed in the present study, and thirty-six lineages of Strobilomyces were identified (Figure 1). Four (lineages 3, 6, 23, and 25) were described as new species: S. hainanensis, S. baozhengii, S. pachycystidiatus, and S. conicus; twenty-eight (lineages 1, 2, 4, 5, 7, 8, 9, 10, 11, 12, 14, 15, 17, 20, 21, 22, 24, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, and 36) represent previously described taxa (Table 2); four (lineage 13, 16, 18, and 19) are awaiting further identification (Figure 1). Together with previously described species of Strobilomyces in China (Table 2), our new species S. baozhengii, S. conicus, S. hainanensis, and S. pachycystidiatus are all members of sect. Strobilomyces (Figure 1). Interestingly, species of sect. Echinati have not been uncovered in China hitherto.

Table 2.
Accepted species, and locality of Strobilomyces (sect. Strobilomyces) in China.
Recent phylogenetic studies have provided new perspectives into the phylogeny and geography of Strobilomyces [5,7]. Besides revealing four new species of Strobilomyces, our molecular data have also contributed to other knowledge of this group (Figure 1). The molecular data indicated the affinities of Strobilomyces species between China and Europe, with two Chinese collections both labeled as S. strobilaceus (lineages 18 and 19) being related to the European taxa (Figure 1). Moreover, we also noted that some Chinese species are closely related to the North America taxa; for example, S. velutinus (lineage 34) from southwestern China is affiliated with one taxon labeled as S. confusus from USA. Interestingly, the Asian S. albidus is related to two unidentified collections from Australia and Africa, respectively (Figure 1). In addition, we noted there are several common taxa from China and Thailand, i.e., S. albidus, S. brunneolepidotus, S. giganteus, S. mirandus, and S. seminudus (Figure 1). Strobilomyces alpinus is widely distributed, occurring in the south of China, Pakistan, and India (Figure 1). The affinities of S. glabriceps between the south of China and India are evident (Figure 1).
5. Conclusions
Although Strobilomyces is widely distributed in the world, the diversity of this genus has not been completely resolved. In this work, four new species of Strobilomyces, viz. S. baozhengii, S. conicus, S. hainanensis, and S. pachycystidiatus are described based on morphological and molecular phylogenetic analysis, which provides us with further understanding of this genus diversity in tropical China. In the future, we hope that more and more species of the genus will be uncovered.
- Key to the species of Strobilomyces in China.
| 1. Pileus with blackish-brown, reddish-brown, yellowish-brown, brown or golden-tawny scales | 2 |
| 1. Pileus with black, greyish-black, grey or dirty white scales | 17 |
| 2. Pileus with golden-tawny to golden-orange scales; basidiospores smaller measuring 6–8 × 5.5–7 μm | S. mirandus |
| 2. Pileus with blackish-brown, reddish-brown, brown or yellowish-brown scales; basidiospores larger, more than 7 μm in length | 3 |
| 3. Pileus smaller (up to 4 cm), with yellowish-brown scales | S. minor |
| 3. Pileus larger (up to 12 cm), with blackish-brown, reddish-brown or brown scales | 4 |
| 4. Pileus with reddish-brown or brown scales | 5 |
| 4. Pileus with blackish-brown scales | 9 |
| 5. Stipe with dirty white to light reddish-brown thin flosses; hymenophoral pores smaller, 0.05–0.1 cm diameter | S. glabellus |
| 5. Stipe with reddish-brown, brown, dark brown to blackish-brown fluffy flosses; hymenophoral pores larger, 0.1–0.3 cm diameter | 6 |
| 6. Pileus with reddish-brown to vinaceous brown on the lower part to dark brown to blackish-brown on the upper part, crowded, more or less erect pyramidal scales; stipe with dark brown to blackish-brown fluffy flosses | S. atrosquamosus |
| 6. Pileus with reddish-brown or brown, scattered, erect conical scales; stipe with concolorous fluffy flosses and conical scales | 7 |
| 7. Hymenophoral pores smaller, 0.05–0.1 cm diameter; stipe with light grayish-brown to chocolate fluff on the upper part and reddish-brown to chocolate fluff on the lower, basidiospores larger measuring 8–10 × 7–8 μm | S. rubrobrunneus |
| 7. Hymenophoral pores larger, 0.1–0.3 cm diameter; stipe with brown or reddish fluffy flosses or scales, basidiospores smaller, less than 9 μm in length | 8 |
| 8. Scales on pileus brown, without reddish tinge; stipe surface reticulate with shallow and elongate meshes on the upper part, covered with clustered tomentose scales on the lower part; basidiospores smaller measuring 7–8 × 6.5–7 μm | S. sculptus |
| 8. Scales on pileus with reddish tinge; stipe with concolorous thick fluffy flosses and erect conical scales; basidiospores larger measuring 7.5–9 × 6.5–8 μm | S. brunneolepidotus |
| 9. Pileus with more or less erect conical to pyramidal scales | 10 |
| 9. Pileus with patch-like to appressed scales or flosses | 15 |
| 10. Basidiospores with complete reticula; hymenophoral pores larger, 0.05–0.5 cm diameter | 11 |
| 10. Basidiospores with incomplete reticula; hymenophoral pores smaller, 0.05–0.1 cm diameter | S. conicus |
| 11. Hymenophoral pores larger, 0.1–0.5 cm diameter; cystidia thick-walled (up to 3 μm); tropical distribution | S. pachycystidiatus |
| 11. Hymenophoral pores smaller, 0.05–0.15 cm diameter; cystidia thin-walled, less than 1 μm; temperate, tropical or subtropical distribution | 12 |
| 12. Stipe with blackish-brown thin fluffy flosses | 13 |
| 12. Stipe with sooty-brown to blackish, grey to dirty white or greyish-black flosses | 14 |
| 13. Pileus with blackish-brown to vinaceous, soft erect conical scales; basidiospores with larger meshes (2–3.5 μm diameter) | S. mollis |
| 13. Pileus with black-brown, more or less erect pyramidal scales; basidiospores with smaller meshes (1–2 μm diameter) | S. parvirimosus |
| 14. Pileus with sooty-brown to blackish scales; stipe without an annulus, with cottony or woolly, sooty-brown to blackish floccules | S. echinocephalus |
| 14. Pileus with dirty white to light blackish-brown scales; stipe with an annulus, with grey to dirty white on the upper part and greyish-black flosses on the lower part | S. microreticulatus |
| 15. Hymenophoral pores larger, 0.1–0.2 mm diameter; basidiospores larger measuring 11.5–14 × 9.5–11 μm, with larger meshes (2–4 μm diameter) | S. alpinus |
| 15. Hymenophoral pores smaller, 0.05–0.1 mm diameter; basidiospores smaller, less than 11 μm in length, with smaller meshes (1–3 μm diameter) | 16 |
| 16. Pileus with blackish-brown at apex and light brown to dirty white at base, scales or flosses; stipe with an annulus, with greyish-white fluffy flosses on the upper part and dark blackish-brown on the lower arranged in spiral; basidiospores larger measuring 9–11 × 7–8.5 μm; subtropical to temperate distribution | S. cingulatus |
| 16. Pileus with blackish-brown to dark chocolate scales or flosses; stipe without an annulus, with dirty white thin flosses evenly distributed; basidiospores smaller measuring 7–9 × 6–7 μm; tropical distribution | S. albidus |
| 17. Pileus with black scales | 18 |
| 17. Pileus with greyish-black, grey to dirty white scales | 24 |
| 18. Pileus with more or less erect conical to pyramidal scales | 19 |
| 18. Pileus with patch-like to appressed scales or flosses | 23 |
| 19. Basidiospores with complete reticula | 20 |
| 19. Basidiospores with incomplete reticula | 22 |
| 20. Pileus larger (up to 9.7 cm); stipe with thick flosses arranged in spiral | S. douformis |
| 20. Pileus smaller (up to 5 cm); stipe with thin flosses evenly distributed | 21 |
| 21. Pileus with charcoal black, smaller, more or less erect pyramidal scales; basidiospores smaller measuring 8.5–10 × 6.5–8(–9) μm; subtropical distribution | S. anthracinus |
| 21. Pileus with black erect conical to pyramidal scales; basidiospores larger measuring 9–11.5 × 8–10 μm; tropical distribution | S. hainanensis |
| 22. Pileus with soft, more or less erect conical to pyramidal scales; stipe without an annular zone, entirely with black granular scales; echinate basidiospores with confluent tubercles and irregular incomplete reticulations | S. calidus |
| 22. Pileus with hard, more or less erect conical scales; stipe with an annular zone at apex, with black minutely conical scales and fluffy flosses; basidiospores with semireticulate ornamentation | S. giganteus |
| 23. Pileus larger (up to 6 cm); stipe with patch-like to appressed, blackish-brown scales or reticula; tropical distribution | S. baozhengii |
| 23. Pileus smaller (up to 4.5 cm); stipe with black tomentose scales; subtropical distribution | S. huangshanensis |
| 24. Pileus with more or less erect conical to pyramidal scales | 25 |
| 24. Pileus with patch-like to appressed scales or flosses | 29 |
| 25. Basidiospores with complete reticula | 26 |
| 25. Basidiospores with incomplete reticula | 28 |
| 26. Stipe without an annular zone; tropical to subtropical distribution | S. montosus |
| 26. Stipe with an annular zone; subtropical to temperate distribution | 27 |
| 27. Pileus larger (up to 11 cm), with dirty white, small, erect conical scales and greyish-black apex; hymenophoral pores larger, 0.1–0.2 cm diameter | S. pteroreticulosporus |
| 27. Pileus smaller (up to 7 cm), with greyish-black, more or less erect pyramidal scales; hymenophoral pores smaller, 0.05–0.1 cm diameter | S. pinophilus |
| 28. Pileus larger (up to 12 cm); stipe with greyish-white flosses on the upper part and black on the lower; echinate basidiospores with irregular short ribs; subtropical to temperate distribution | S. densisquamosus |
| 28. Pileus smaller (up to 6 cm); stipe with black scattered granular scales; semireticulate basidiospores | S. velutinus |
| 29. Basidiospores with complete reticula | 30 |
| 29. Basidiospores with incomplete reticula | 31 |
| 30. Pileus larger (up to 15 cm); stipe with an annulus, with grey to dirty white flosses on the upper part and greyish-black on the lower; basidiospores with larger meshes (2–4 μm diameter); subtropical distribution | S. glabriceps |
| 30. Pileus smaller (up to 9 cm); stipe without an annular zone, entirely with whitish thick fluffy flosses; basidiospores with smaller meshes (1–2 μm diameter); tropical and subtropical distribution | S. latirimosus |
| 31. Stipe with an annular zone; basidiospores smaller measuring 7–9 × 6.5–8.5 μm; tropical to subtropical distribution | S. seminudus |
| 31. Stipe without an annular zone; basidiospores larger measuring 8–10 × 7.5–8.5 μm; subtropical distribution | S. subnudus |
Author Contributions
Conceptualization, Z.-Q.L. and N.-K.Z.; methodology, performing the experiment and formal analysis, H.D. and Y.W.; resources and data curation, N.-K.Z.; writing—original draft preparation, H.D., Y.W., J.-R.L. and Z.-Z.C.; writing—review and editing, Z.-Q.L. and N.-K.Z.; supervision, N.-K.Z.; project administration, N.-K.Z.; funding acquisition, N.-K.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the National Natural Science Foundation of China (No. 32160001), the Hainan Provincial Natural Science Foundation of China (Nos. 322MS024, 820RC633, 822QN316), and the Integrated Survey and Monitoring of Resources in Hainan Tropical Rainforest National Park (No. 1074688).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The names of the new species were formally registered in the MycoBank [https://www.mycobank.org/ (accessed on 1 October 2023)]. Specimens were deposited in the Fungal Herbarium of Hainan Medical University, Haikou City, Hainan Province of China (FHMU). The sequence data generated in this study are deposited in GenBank [https://www.ncbi.nlm.nih.gov/genbank/ (accessed on 30 June 2023)].
Acknowledgments
The authors are very grateful to the forest rangers, Yinggeling of Hainan Tropical Rainforest National Park, for their kind help during the field investigations.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Corner, E.J.H. Boletus in Malaysia; Government Printing Office: Singapore, 1972; pp. 1–450.
- Singer, R. The Agaricales in Modern Taxonomy, 4th ed.; Koeltz Scientific Books: Königstein, Germany, 1986; pp. 1–981. [Google Scholar]
- Sato, H.; Hattori, T.; Lee, S.S.; Murakami, N. Two species of Strobilomyces (Boletaceae, Boletales), S. seminudus and S. hongoi sp. nov. from Japan. Mycologia 2011, 103, 598–609. [Google Scholar] [CrossRef] [PubMed]
- Han, L.H.; Buyck, B.; Yorou, N.S.; Halling, R.E.; Yang, Z.L. Afroboletus sequestratus (Boletales), the first species with sequestrate basidioma in the genus. Phytotaxa 2017, 305, 11–20. [Google Scholar] [CrossRef]
- Han, L.H.; Feng, B.; Wu, G.; Halling, R.E.; Buyck, B.; Yorou, N.S.; Ebika, S.T.N.; Yang, Z.L. African origin and global distribution patterns: Evidence inferred from phylogenetic and biogeographical analyses of ectomycorrhizal fungal genus Strobilomyces. J. Biogeogr. 2018, 45, 201–212. [Google Scholar] [CrossRef]
- Han, L.H.; Hao, Y.J.; Liu, C.; Dai, D.Q.; Zhao, K.; Tang, L.Z. Strobilomyces rubrobrunneus (Boletaceae), a new species with reddish brown scales from eastern China. Phytotaxa 2018, 376, 167–176. [Google Scholar] [CrossRef]
- Han, L.H.; Wu, G.; Horak, E.; Halling, R.E.; Xu, J.; Ndolo, E.S.T.; Sato, H.; Fechner, N.; Sharma, Y.P.; Yang, Z.L. Phylogeny and species delimitation of Strobilomyces (Boletaceae), with an emphasis on the Asian species. Persoonia 2020, 44, 113–139. [Google Scholar] [CrossRef]
- Gelardi, M.; Vizzini, A.; Ercole, E.; Voyron, S.; Wu, G.; Liu, X.Z. Strobilomyces echinocephalus sp. nov. (Boletales) from south-western China, and a key to the genus Strobilomyces worldwide. Mycol. Prog. 2013, 12, 575–588. [Google Scholar] [CrossRef]
- Tibpromma, S.; Hyde, K.D.; Jeewon, R.; Maharachchikumbura, S.S.N.; Liu, J.K.; Bhat, D.J.; Jones, E.B.G.; McKenzie, E.H.C.; Camporesi, E.; Bulgakov, T.S.; et al. Fungal diversity notes 491–602: Taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers. 2017, 83, 1–261. [Google Scholar] [CrossRef]
- Pegler, D.N. A preliminary Agaric Flora of East Africa. Kew Bull. Addit. Ser. 1977, 6, 1–615. [Google Scholar]
- Ying, J.Z.; Ma, Q.M. New taxa and records of the genus Strobilomyces in China. Acta Mycol. Sin. 1985, 4, 95–102. [Google Scholar]
- Zang, M. Notes on the Boletales from eastern Himalayas and adjacent of China. Acta Bot. Yunnanica 1985, 7, 383–401. [Google Scholar]
- Sato, H.; Yumoto, T.; Murakami, N. Cryptic species and host specificity in the ectomycorrhizal genus Strobilomyces (Strobilomycetaceae). Am. J. Bot. 2007, 94, 1630–1641. [Google Scholar] [CrossRef]
- Sato, H.; Tanabe, A.S.; Toju, H. Host shifts enhance diversification of ectomycorrhizal fungi: Diversification rate analysis of the ectomycorrhizal fungal genera Strobilomyces and Afroboletus with an 80-gene phylogeny. New Phytol. 2017, 214, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Antonin, V.; Vizzini, A.; Ercole, E.; Leonardi, M. Strobilomyces pteroreticulosporus (Boletales), a new species of the S. strobilaceus complex from the Republic of Korea and remarks on the variability of S. confusus. Phytotaxa 2015, 219, 78–86. [Google Scholar] [CrossRef]
- Dai, Y.C.; Zhou, L.W.; Yang, Z.L.; Wen, H.A.; Tolgor, B.; Li, T.H. A revised checklist of edible fungi in China. Mycosystema 2010, 29, 1–21. [Google Scholar]
- Dai, Y.C.; Yang, Z.L. A revised checklist of medicinal fungi in China. Mycosystema 2008, 27, 801–824. [Google Scholar]
- Gao, C.; Shi, N.N.; Liu, Y.X.; Peay, K.G.; Zheng, Y.; Ding, Q.; Mi, X.C.; Ma, K.P.; Wubet, T.; Buscot, F.; et al. Host plant genus-level diversity is the best predictor of ectomycorrhizal fungal diversity in a Chinese subtropical forest. Mol. Ecol. 2013, 22, 3403–3414. [Google Scholar] [CrossRef]
- Wu, G.; Feng, B.; Xu, J.P.; Zhu, X.T.; Li, Y.C.; Zeng, N.K.; Hosen, M.I.; Yang, Z.L. Molecular phylogenetic analyses redefine seven major clades and reveal 22 new generic clades in the fungal family Boletaceae. Fungal Divers. 2014, 69, 93–115. [Google Scholar] [CrossRef]
- Wu, G.; Li, Y.C.; Zhu, X.T.; Zhao, K.; Han, L.H.; Cui, Y.Y.; Li, F.; Xu, J.P.; Yang, Z.L. One hundred noteworthy boletes from China. Fungal Divers. 2016, 81, 25–188. [Google Scholar] [CrossRef]
- Han, L.H.; Zhao, K.; Liu, C.; Chu, H.L.; Tang, L.Z. Strobilomyces minor (Boletaceae), a new species associated with fagaceous plants in Central China. Phytotaxa 2019, 397, 55–64. [Google Scholar] [CrossRef]
- Han, L.H.; Guo, T.; Yang, R.H.; Liu, C.; Tang, L.Z. Strobilomyces sculptus sp. nov. (Boletaceae) from eastern China with morphological and molecular evidence. Nova Hedwig. 2019, 109, 111–120. [Google Scholar] [CrossRef]
- Liu, H.; Chen, Q.; Liu, X.; Xu, Z.; Dai, Y.; Liu, Y.; Chen, Y. Variation patterns of plant composition/diversity in Dacrydium pectinatum communities and their driving factors in a biodiversity hotspot on Hainan Island, China. Glob. Ecol. Conserv. 2020, 22, e01034. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Da Fonseca, G.A.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Kornerup, A.; Wanscher, J.H. Taschenlexikon der Farben, 3rd ed.; Muster-Schmidt Verlag: Göttingen, Germany, 1981; pp. 1–242. [Google Scholar]
- Ladurner, H.; Simonini, G. Xerocomus s.l.; Edizioni Candusso: Alassio, Italy, 2003. [Google Scholar]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef]
- James, T.Y.; Kauff, F.; Schoch, C.L.; Matheny, P.B.; Hofstetter, V.; Cox, C.J.; Celio, G.; Gueidan, C.; Fraker, E.; Miadlikowska, J.; et al. Reconstructing the early evolution of Fungi using a six-gene phylogeny. Nature 2006, 443, 818–822. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.D.; Lee, S.; Taylor, J. Amplifification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelflfland, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Mikheyev, A.S.; Mueller, U.G.; Abbot, P. Cryptic sex and many-to-one coevolution in the fungus-growing ant symbiosis. Proc. Natl. Acad. Sci. USA 2006, 103, 10702–10706. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analyses program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Smith, S.A.; Dunn, C.W. Phyutility: A phyloinformatics tool for trees, alignments and molecular data. Bioinformatics 2008, 24, 715–716. [Google Scholar] [CrossRef]
- Kuo, M.; Ortiz-Santana, B. Revision of leccinoid fungi, with emphasis on North American taxa, based on molecular and morphological data. Mycologia 2020, 112, 197–211. [Google Scholar] [CrossRef]
- Binder, M.; Hibbett, D.S. Molecular systematics and biological diversification of Boletales. Mycologia 2006, 98, 971–981. [Google Scholar] [CrossRef]
- Halling, R.E.; Nuhn, M.; Fechner, N.A.; Osmundson, T.W.; Soytong, K.; Arora, D.; Hibbett, D.S.; Binder, M. Sutorius: A new genus for Boletus eximius. Mycologia 2012, 104, 951–961. [Google Scholar] [CrossRef]
- Raspé, O.; Vadthanarat, S.; Kesel, D.A.; Degreef, J.; Hyde, K.D.; Lumyong, S. Pulveroboletus fragrans, a new Boletaceae species from Northern Thailand, with a remarkable aromatic odor. Mycol. Prog. 2016, 15, 38. [Google Scholar] [CrossRef]
- Song, J.; Liang, J.F.; Mehrabi-Koushki, M.; Krisai-Greilhuber, I.; Ali, B.; Bhatt, V.K.; Cerna-Mendoza, A.; Chen, B.; Chen, X.Z.; Chu, H.L.; et al. Fungal systematics and evolution: FUSE 5. Sydowia 2019, 71, 141–245. [Google Scholar] [PubMed]
- Ullah, S.; Vizzini, A.; Fiaz, M.; Rehman, H.U.; Sher, H.; Khalid, A.N. Strobilomyces longistipitatus (Boletaceae) newly recorded from Hindukush and Himalayan moist temperate forests of Pakistan. Nova Hedwig. 2019, 108, 243–254. [Google Scholar] [CrossRef]
- Vadthanarat, S.; Lumyong, S.; Raspé, O. Cacaoporus, a new Boletaceae genus, with two new species from Thailand. MycoKeys 2019, 54, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Vadthanarat, S.; Raspé, O.; Lumyong, S. Phylogenetic affinities of the sequestrate genus Rhodactina (Boletaceae), with a new species, R. rostratispora from Thailand. MycoKeys 2018, 29, 63–80. [Google Scholar] [CrossRef]
- Zeng, N.K.; Chai, H.; Jiang, S.; Xue, R.; Wang, Y.; Hong, D.; Liang, Z.Q. Retiboletus nigrogriseus and Tengioboletus fujianensis, two new boletes from the south of China. Phytotaxa 2018, 367, 45–54. [Google Scholar] [CrossRef]
- Ying, J.Z. Supplement notes on genus Strobilomyces from China. Acta Mycol. Sin. 1986, (Suppl. I), 305–308. [Google Scholar]
- Berkeley, M.J. Decades of fungi. Decades XXXIV. Sikkim Himalayan Fungi collected by Dr. Hooker. Hook J. Bot. 1851, 3, 77–84. [Google Scholar]
- Takahashi, H.; Taneyama, Y.; Kobayashi, T.; Oba, Y.; Hadano, E.; Hadano, A.; Kurogi, S.; Wada, S.; Terashima, Y. The Agaric Flora in Southwestern Japan; Tokai Daigaku: Kanagawa, Japan, 2016. [Google Scholar]
- Petersen, R.H.; Hughes, K.W.; Adamčík, S.; Tkalčec, Z.; Mešić, A. Typification of three European species epithets attributable to Strobilomyces (Boletales). Czech Mycol. 2012, 64, 141–163. [Google Scholar] [CrossRef]
- Wen, H.A.; Ying, J.Z. Supplementary notes on the genus Strobilomyces from China II. Mycosystema 2001, 20, 297–300. [Google Scholar]
- Chiu, W.F. The boletes of Yunnan. Mycologia 1948, 40, 199–231. [Google Scholar] [CrossRef]
- Hongo, T. Materials for the fungus flora of Japan (32). Trans. Mycol. Soc. Jpn. 1982, 23, 195–200. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).